Sensitization of MCF7 Cells with High Notch1 Activity by Cisplatin and Histone Deacetylase Inhibitors Applied Together
Abstract
1. Introduction
2. Results
2.1. Expression and Activity of Notch1
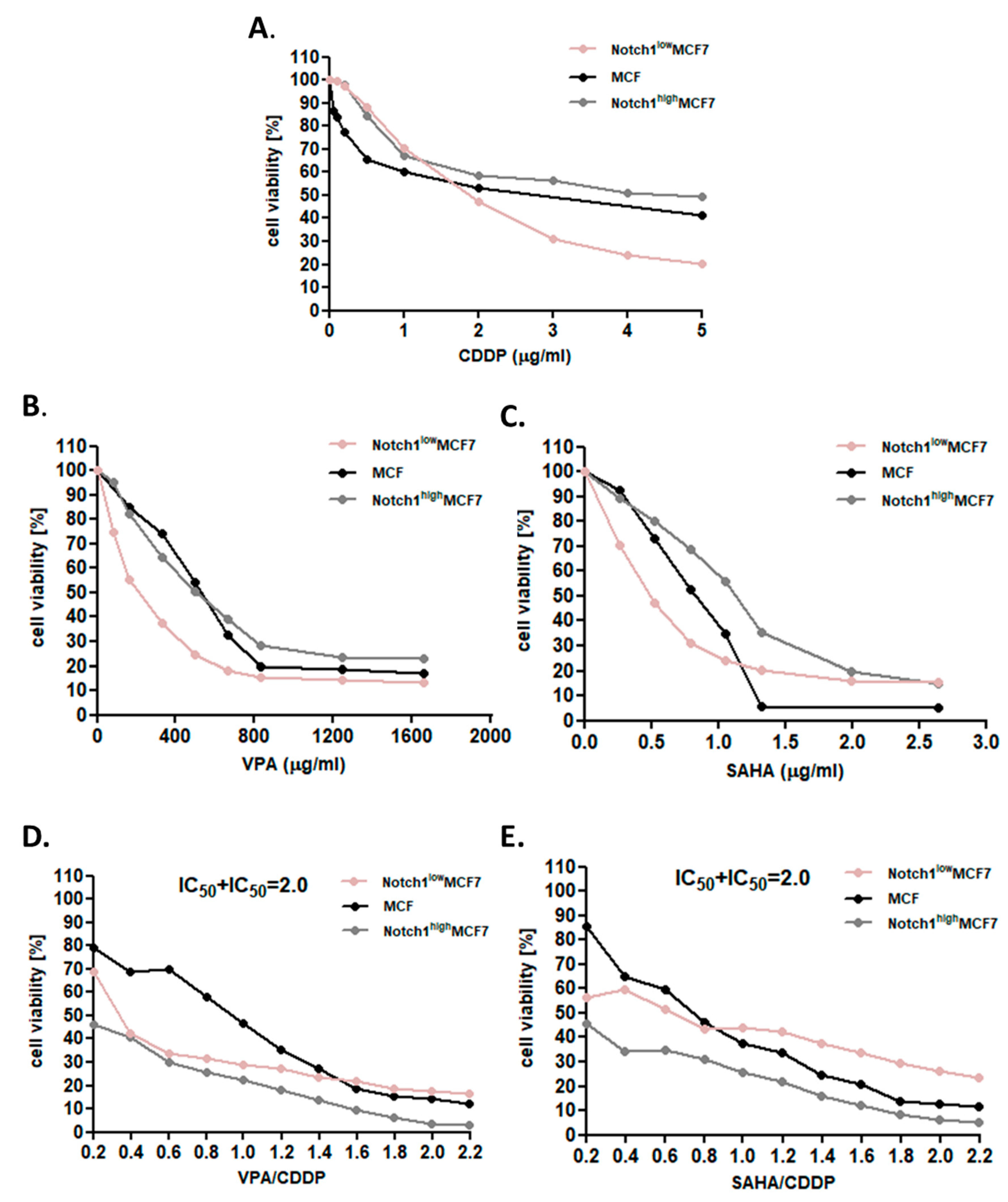
2.2. Dose-Dependent Growth Inhibition of Native and Transfected MCF7 Breast Cancer Cells after CDDP and HDIs Treatment
2.3. Effect of SAHA or VPA on the Anti-Proliferative Effects of CDDP in MCF7 cells with Increased Activity of Notch1 (Notch1highMCF7)
2.4. Effect of SAHA or VPA on the Anti-Proliferative Effects of CDDP on MCF7 cells with Decreased Activity of Notch1 (Notch1lowMCF7)
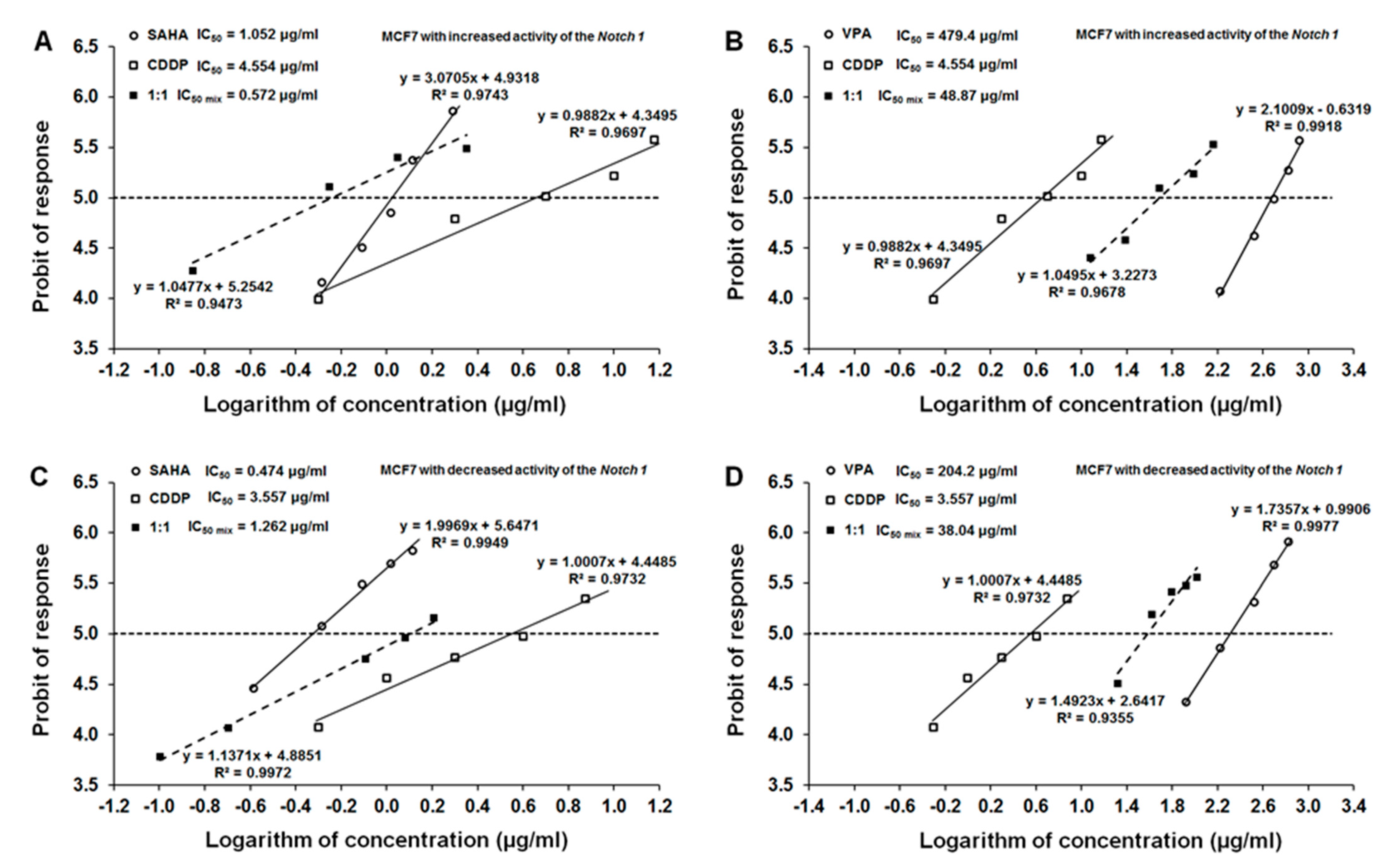
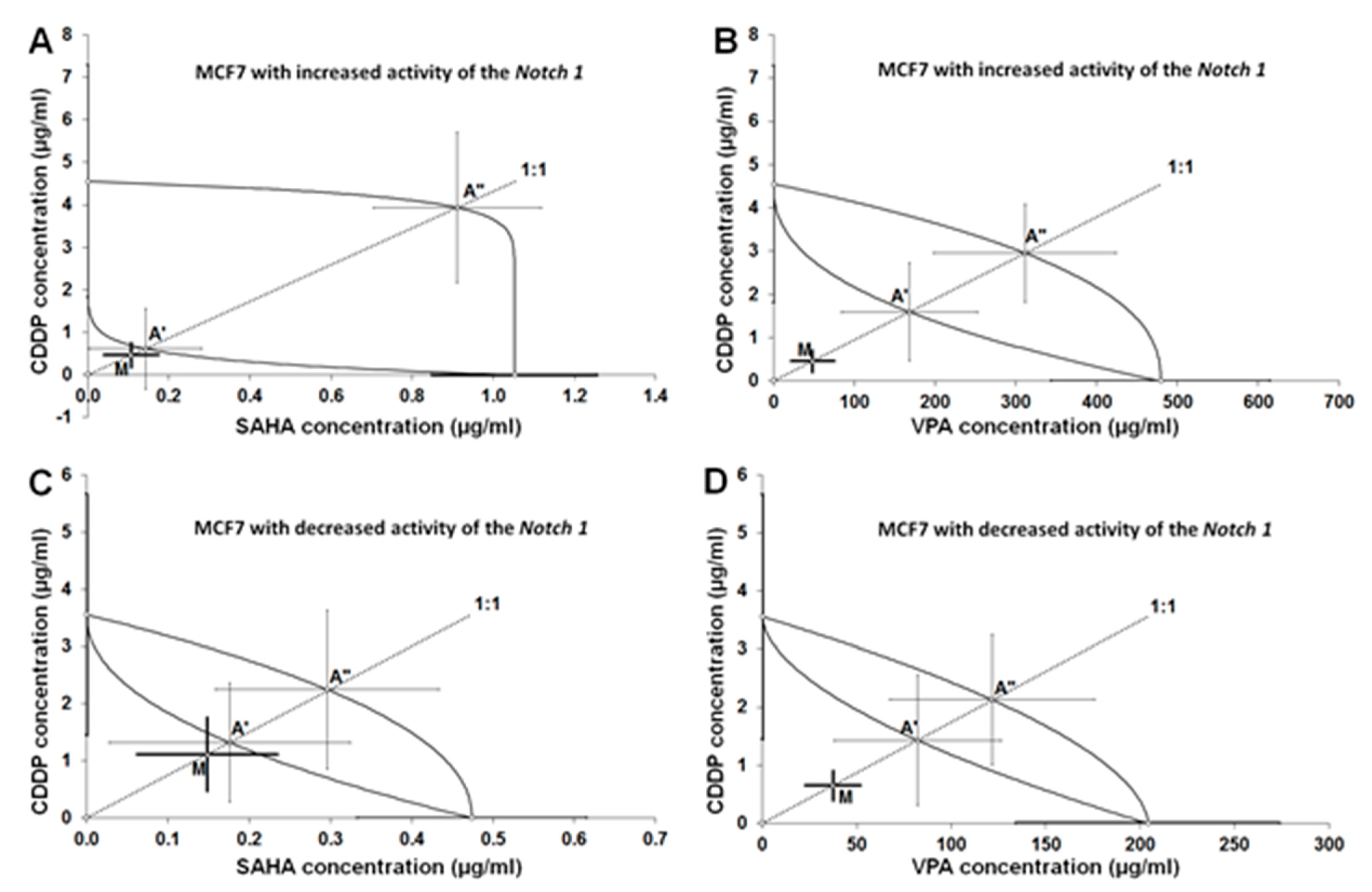
2.5. Type I Isobolographic Analysis of Interaction for the Combinations of CDDP with SAHA or VPA on Notch1highMCF7 Cells
2.6. Type I Isobolographic Analysis of Interaction for the Combinations of CDDP with SAHA or VPA on Notch1lowMCF7 Cells
2.7. Analysis of the Types of Pharmacological Interaction between CDDP and HDIs on Cells with Altered Notch1 Activity with Reference to Native MCF7 Cells
3. Discussion
4. Materials and Methods
4.1. Drugs
4.2. Cells
4.3. Transfection Procedure and Development of MCF7 Breast Cancer Cell Lines with Increased (Notch1high) and Decreased (Notch1low) Notch1 Activity
4.4. Western Blotting Analysis and Luciferase Reporter Assay: Evaluation of the Effectiveness of the Transfection Procedure and Development of MCF7 Breast Cancer Cell Lines with Increased (Notch1high) and Decreased (Notch1low) Notch1 Activity
4.5. Cell Viability Assay
4.6. Isobolographic Analysis of Pharmacological Interactions between HDIs and CDDP
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, caac.21660. [Google Scholar] [CrossRef]
- Guido, L.P.; Gomez-Fernandez, C. Advances in the Molecular Taxonomy of Breast Cancer. Arch. Med Res. 2020, 51, 777–783. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.J.; Swain, S.M. Luminal A Breast Cancer and Molecular Assays: A Review. Oncologist 2018, 23, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-H.; Hu, P.-H.; Tu, J.-H.; Yu, N.-S. Luminal B breast cancer: Patterns of recurrence and clinical outcome. Oncotarget 2016, 7, 65024–65033. [Google Scholar] [CrossRef]
- Lim, E.; Winer, E.P. Adjuvant Chemotherapy in Luminal Breast Cancers. 2011. Available online: http://www.adjuvantonline.com (accessed on 28 March 2021).
- Huang, L.; Liu, Q.; Chen, S.; Shao, Z. Cisplatin versus carboplatin in combination with paclitaxel as neoadjuvant regimen for triple negative breast cancer. OncoTargets Ther. 2017, ume10, 5739–5744. [Google Scholar] [CrossRef] [PubMed]
- Aldossary, S.A. Review on Pharmacology of Cisplatin: Clinical Use, Toxicity and Mechanism of Resistance of Cisplatin. Biomed. Pharmacol. J. 2019, 12, 7–15. [Google Scholar] [CrossRef]
- Wawruszak, A.; Kalafut, J.; Okon, E.; Czapinski, J.; Halasa, M.; Przybyszewska, A.; Miziak, P.; Okla, K.; Rivero-Muller, A.; Stepulak, A. Histone Deacetylase Inhibitors and Phenotypical Transformation of Cancer Cells. Cancers 2019, 11, 148. [Google Scholar] [CrossRef] [PubMed]
- Romoli, M.; Mazzocchetti, P.; D’Alonzo, R.; Siliquini, S.; Rinaldi, V.E.; Verrotti, A.; Calabresi, P.; Costa, C. Valproic Acid and Epilepsy: From Molecular Mechanisms to Clinical Evidences. Curr. Neuropharmacol. 2019, 17, 926–946. [Google Scholar] [CrossRef]
- Fedele, P.; Orlando, L.; Cinieri, S. Targeting triple negative breast cancer with histone deacetylase inhibitors. Expert Opin. Investig. Drugs 2017, 26, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Munster, P.; Marchion, D.; Bicaku, E.; Schmitt, M.; Lee, J.H.; DeConti, R.; Simon, G.; Fishman, M.; Minton, S.; Garrett, C.; et al. Phase I Trial of Histone Deacetylase Inhibition by Valproic Acid Followed by the Topoisomerase II Inhibitor Epirubicin in Advanced Solid Tumors: A Clinical and Translational Study. J. Clin. Oncol. 2007, 25, 1979–1985. [Google Scholar] [CrossRef] [PubMed]
- Luu, T.; Kim, K.-P.; Blanchard, S.; Anyang, B.; Hurria, A.; Yang, L.; Beumer, J.H.; Somlo, G.; Yen, Y. Phase IB trial of ixabepilone and vorinostat in metastatic breast cancer. Breast Cancer Res. Treat. 2017, 167, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, L.J.; Zhao, F.; Wang, M.; Swaby, R.F.; Sparano, J.A.; Meropol, N.J.; Bhalla, K.N.; Pellegrino, C.M.; Alpaugh, R.K.; Falkson, C.I.; et al. A Phase I/II study of suberoylanilide hydroxamic acid (SAHA) in combination with trastuzumab (Herceptin) in patients with advanced metastatic and/or local chest wall recurrent HER2-amplified breast cancer: A trial of the ECOG-ACRIN Cancer Research Group (E1104). Breast Cancer Res. Treat. 2017, 165, 375–382. [Google Scholar] [CrossRef]
- Tu, Y.; Hershman, D.L.; Bhalla, K.; Fiskus, W.; Pellegrino, C.M.; Andreopoulou, E.; Makower, D.; Kalinsky, K.; Fehn, K.; Fineberg, S.; et al. A phase I-II study of the histone deacetylase inhibitor vorinostat plus sequential weekly paclitaxel and doxorubicin-cyclophosphamide in locally advanced breast cancer. Breast Cancer Res. Treat. 2014, 146, 145–152. [Google Scholar] [CrossRef]
- Chiu, H.-W.; Yeh, Y.-L.; Wang, Y.-C.; Huang, W.-J.; Chen, Y.-A.; Chiou, Y.-S.; Ho, S.-Y.; Lin, P.; Wang, Y.-J. Suberoylanilide Hydroxamic Acid, an Inhibitor of Histone Deacetylase, Enhances Radiosensitivity and Suppresses Lung Metastasis in Breast Cancer In Vitro and In Vivo. PLoS ONE 2013, 8, e76340. [Google Scholar] [CrossRef] [PubMed]
- Fortunati, N.; Bertino, S.; Costantino, L.; Bosco, O.; Vercellinatto, I.; Catalano, M.G.; Boccuzzi, G. Valproic acid is a selective antiproliferative agent in estrogen-sensitive breast cancer cells. Cancer Lett. 2008, 259, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Jin, H.; Roy, M.; Ma, A.L.; Gong, S.; Jaskula-Sztul, R.; Chen, H. Antineoplastic effects of histone deacetylase inhibitors in neuroendocrine cancer cells are mediated through transcriptional regulation of Notch1 by activator protein 1. Cancer Med. 2017, 6, 2142–2152. [Google Scholar] [CrossRef] [PubMed]
- Wawruszak, A.; Luszczki, J.J.; Kalafut, J.; Okla, K.; Halasa, M.; Rivero-Muller, A.; Stepulak, A. Additive Pharmacological Interaction between Cisplatin (CDDP) and Histone Deacetylase Inhibitors (HDIs) in MDA-MB-231 Triple Negative Breast Cancer (TNBC) Cells with Altered Notch1 Activity—An Isobolographic Analysis. Int. J. Mol. Sci. 2019, 20, 3663. [Google Scholar] [CrossRef] [PubMed]
- Andersson, E.R.; Sandberg, R.; Lendahl, U. Notch signaling: Simplicity in design, versatility in function. Development 2011, 138, 3593–3612. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.-L.; Tian, Y.; Lu, C.; Guo, H.; Zhao, X.-W.; Guo, Y.-W.; Wang, L.-Q.; Du, Q.-L.; Liu, C.-P. Effects of notch-1 down-regulation on malignant behaviors of breast cancer stem cells. Acta Acad. Med. Wuhan 2014, 34, 195–200. [Google Scholar] [CrossRef]
- Yuan, X.; Zhang, M.; Wu, H.; Xu, H.; Han, N.; Chu, Q.; Yu, S.; Chen, Y.; Wu, K. Expression of Notch1 Correlates with Breast Cancer Progression and Prognosis. PLoS ONE 2015, 10, e0131689. [Google Scholar] [CrossRef] [PubMed]
- Wawruszak, A.; Luszczki, J.J.; Grabarska, A.; Gumbarewicz, E.; Dmoszynska-Graniczka, M.; Polberg, K.; Stepulak, A. Assessment of Interactions between Cisplatin and Two Histone Deacetylase Inhibitors in MCF7, T47D and MDA-MB-231 Human Breast Cancer Cell Lines—An Isobolographic Analysis. PLoS ONE 2015, 10, e0143013. [Google Scholar] [CrossRef]
- Wawruszak, A.; Gumbarewicz, E.; Okon, E.; Jeleniewicz, W.; Czapinski, J.; Halasa, M.; Okla, K.; Smok-Kalwat, J.; Bocian, A.; Rivero-Muller, A.; et al. Histone deacetylase inhibitors reinforce the phenotypical markers of breast epithelial or mesenchymal cancer cells but inhibit their migratory properties. Cancer Manag. Res. 2019, ume11, 8345–8358. [Google Scholar] [CrossRef] [PubMed]
- Landor, S.K.-J.; Mutvei, A.P.; Mamaeva, V.; Jin, S.; Busk, M.; Borra, R.; Grönroos, T.J.; Kronqvist, P.; Lendahl, U.; Sahlgren, C.M. Hypo- and hyperactivated Notch signaling induce a glycolytic switch through distinct mechanisms. Proc. Natl. Acad. Sci. USA 2011, 108, 18814–18819. [Google Scholar] [CrossRef]
- Tallarida, R.J. Revisiting the Isobole and Related Quantitative Methods for Assessing Drug Synergism. J. Pharmacol. Exp. Ther. 2012, 342, 2–8. [Google Scholar] [CrossRef]
- Tallarida, R.J. An Overview of Drug Combination Analysis with Isobolograms. J. Pharmacol. Exp. Ther. 2006, 319, 1–7. [Google Scholar] [CrossRef]
- Tallarida, R.J. Interactions between drugs and occupied receptors. Pharmacol. Ther. 2007, 113, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Weigelt, B.; Geyer, F.C.; Reis-Filho, J.S. Histological types of breast cancer: How special are they? Mol. Oncol. 2010, 4, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Daniyal, A.; Santoso, I.; Gunawan, N.H.P.; Barliana, M.I.; Abdulah, R. Genetic Influences in Breast Cancer Drug Resistance. Breast Cancer Targets Ther. 2021, ume13, 59–85. [Google Scholar] [CrossRef] [PubMed]
- Gumbarewicz, E.; Luszczki, J.J.; Wawruszak, A.; Dmoszynska-Graniczka, M.; Grabarska, A.J.; Jarząb, A.M.; Polberg, K.; Stepulak, A. Isobolographic analysis demonstrates additive effect of cisplatin and HDIs combined treatment augmenting their anti-cancer activity in lung cancer cell lines. Am. J. Cancer Res. 2016, 6, 2831–2845. [Google Scholar] [PubMed]
- Yadav, B.S.; Sharma, S.C.; Chanana, P.; Jhamb, S. Systemic treatment strategies for triple-negative breast cancer. World J. Clin. Oncol. 2014, 5, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Grabarska, A.; Nowosadzka, E.; Jeleniewicz, W.; Kupisz, K.; Łuszczki, J.J.; Gumbarewicz, E.; Dmoszynska-Graniczka, M.; Kowalczuk, K.; Polberg, K.; Stepulak, A. Histone Deacetylase Inhibitor SAHA as Potential Targeted Therapy Agent for Larynx Cancer Cells. J. Cancer 2017, 8, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Kunnimalaiyaan, S.; Sokolowski, K.; Gamblin, T.C.; Kunnimalaiyaan, M. Suberoylanilide hydroxamic Acid, a histone deacetylase inhibitor, alters multiple signaling pathways in hepatocellular carcinoma cell lines. Am. J. Surg. 2017, 213, 645–651. [Google Scholar] [CrossRef]
- Sun, G.; Mackey, L.V.; Coy, D.H.; Yu, C.-Y.; Sun, L. The Histone Deacetylase Inhibitor Vaproic Acid Induces Cell Growth Arrest in Hepatocellular Carcinoma Cells via Suppressing Notch Signaling. J. Cancer 2015, 6, 996–1004. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Yang, Y.; Zhao, R.-J.; Wang, L.-H.; Zhao, L.; Yan, L.-N.; Zhang, Z.-H.; Hao, C.-L. [Effects of VPA on the Expression of Notch Signaling Pathway in Multiple Myeloma RPMI 8226 Cell Line]. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2016, 24, 1449–1453. [Google Scholar] [PubMed]
- Adler, J.T.; Hottinger, D.G.; Kunnimalaiyaan, M.; Chen, H. Histone deacetylase inhibitors upregulate Notch-1 and inhibit growth in pheochromocytoma cells. Surgery 2008, 144, 956–962. [Google Scholar] [CrossRef]
- Greenblatt, D.Y.; Vaccaro, A.M.; Jaskula-Sztul, R.; Ning, L.; Haymart, M.; Kunnimalaiyaan, M.; Chen, H. Valproic Acid Activates Notch-1 Signaling and Regulates the Neuroendocrine Phenotype in Carcinoid Cancer Cells. Oncologist 2007, 12, 942–951. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; He, Q.; Tsai, C.; Lei, J.; Chen, J.; Makcey, L.V.; Coy, D.H. HDAC inhibitors suppressed small cell lung cancer cell growth and enhanced the suppressive effects of receptor-targeting cytotoxins via upregulating somatostatin receptor II. Am. J. Transl. Res. 2018, 10, 545–553. Available online: www.ajtr.org/ISSN:1943-8141/AJTR0068980 (accessed on 30 March 2021).
- Stockhausen, M.; Sjolund, J.; Manetopoulos, C.; Axelson, H. Effects of the histone deacetylase inhibitor valproic acid on Notch signalling in human neuroblastoma cells. Br. J. Cancer 2005, 92, 751–759. [Google Scholar] [CrossRef]
- Marra, A.; Trapani, D.; Viale, G.; Criscitiello, C.; Curigliano, G. Practical classification of triple-negative breast cancer: Intratumoral heterogeneity, mechanisms of drug resistance, and novel therapies. NPJ. Breast Cancer 2020, 6, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Deng, H.-Y.; Jiang, R. Effect of trastuzumab on Notch-1 signaling pathway in breast cancer SK-BR3 cells. Chin. J. Cancer Res. 2012, 24, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Wróblewska-Łuczka, P.; Grabarska, A.; Florek-Łuszczki, M.; Plewa, Z.; Łuszczki, J.J. Synergy, Additivity, and Antagonism between Cisplatin and Selected Coumarins in Human Melanoma Cells. Int. J. Mol. Sci. 2021, 22, 537. [Google Scholar] [CrossRef] [PubMed]
| Cell Line | CDDP | SAHA | VPA |
|---|---|---|---|
| Notch1highMCF7 | 4.554 | 1.052 | 479.4 |
| MCF7 (native) | 2.495 | 0.746 | 465.68 |
| Notch1lowMCF7 | 3.557 | 0.474 | 204.2 |
| Cell Line | Combination | IC50 mix (μg/mL) | nmix | L IC50 add (μg/mL) | nadd | U IC50 add (μg/mL) | nadd |
|---|---|---|---|---|---|---|---|
| Notch1highMCF7 | CDDP+SAHA | 0.572 ± 0.362 | 24 | 0.759 ± 1.084 | 56 | 4.846 ± 1.970 | 56 |
| Notch1highMCF7 | CDDP+VPA | 48.87 ± 27.65 | 30 | 169.9 ± 85.13 | 56 | 314.4 ± 114.0 | 56 |
| Notch1lowMCF7 | CDDP+SAHA | 1.262 ± 0.737 | 24 | 1.497 ± 1.180 | 56 | 2.545 ± 1.522 | 56 |
| Notch1lowMCF7 | CDDP+VPA | 38.04 ± 15.14 | 30 | 83.75 ± 45.30 | 56 | 123.8 ± 55.30 | 56 |
| Combination | Notch1highMCF7 | MCF7 | Notch1lowMCF7 |
|---|---|---|---|
| CDDP+SAHA | additivity | additivity | additivity |
| CDDP+VPA | additivity with tendencytowards synergy | additivity | additivity with tendencytowards synergy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wawruszak, A.; Luszczki, J.; Halasa, M.; Okon, E.; Landor, S.; Sahlgren, C.; Rivero-Muller, A.; Stepulak, A. Sensitization of MCF7 Cells with High Notch1 Activity by Cisplatin and Histone Deacetylase Inhibitors Applied Together. Int. J. Mol. Sci. 2021, 22, 5184. https://doi.org/10.3390/ijms22105184
Wawruszak A, Luszczki J, Halasa M, Okon E, Landor S, Sahlgren C, Rivero-Muller A, Stepulak A. Sensitization of MCF7 Cells with High Notch1 Activity by Cisplatin and Histone Deacetylase Inhibitors Applied Together. International Journal of Molecular Sciences. 2021; 22(10):5184. https://doi.org/10.3390/ijms22105184
Chicago/Turabian StyleWawruszak, Anna, Jarogniew Luszczki, Marta Halasa, Estera Okon, Sebastian Landor, Cecilia Sahlgren, Adolfo Rivero-Muller, and Andrzej Stepulak. 2021. "Sensitization of MCF7 Cells with High Notch1 Activity by Cisplatin and Histone Deacetylase Inhibitors Applied Together" International Journal of Molecular Sciences 22, no. 10: 5184. https://doi.org/10.3390/ijms22105184
APA StyleWawruszak, A., Luszczki, J., Halasa, M., Okon, E., Landor, S., Sahlgren, C., Rivero-Muller, A., & Stepulak, A. (2021). Sensitization of MCF7 Cells with High Notch1 Activity by Cisplatin and Histone Deacetylase Inhibitors Applied Together. International Journal of Molecular Sciences, 22(10), 5184. https://doi.org/10.3390/ijms22105184







