Pseudoephedrine—Benefits and Risks
Abstract
1. Introduction
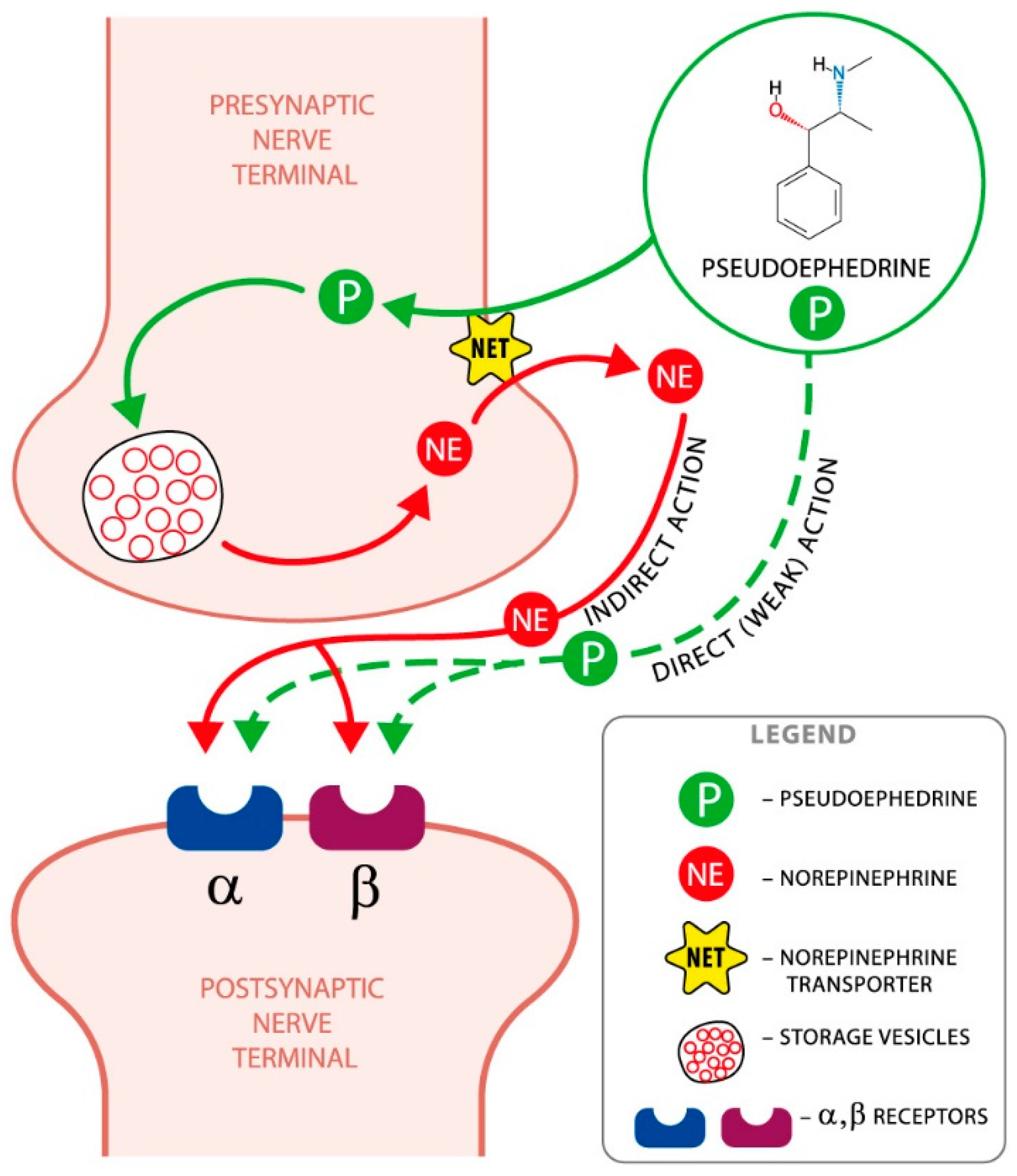
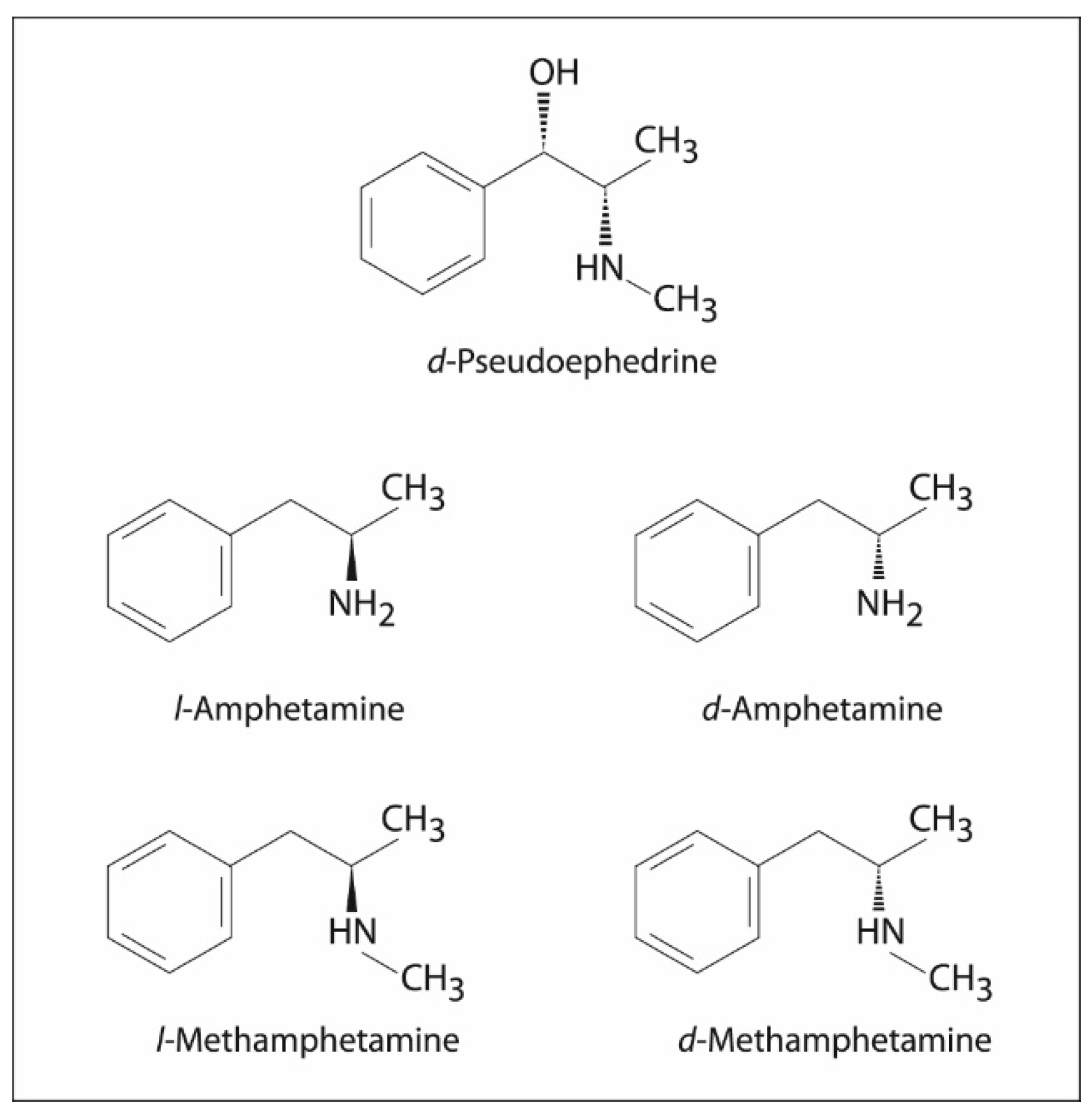
2. Mechanism of Action
3. Pharmacokinetics
4. Special Risk Populations
5. Consequences of Pseudoephedrine Use
6. Clinical Use and Contraindications
7. Dosage
8. Overdose
9. Adverse Reactions
10. Interactions
11. Self-Medication
12. Non-Medical Use
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Krizevski, R.; Bar, E.; Shalit, O.; Sitrit, Y.; Ben-Schabat, S.; Lewinsohn, E. Composition and stereochemistry of ephedrine alkaloids accumulation in Ephedra sinica Stapf. Phytochemistry 2010, 71, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Szumny, D.; Szypuła, E.; Szydłowski, M.; Chlebda, E.; Skrzypiec-Spring, M.; Szumny, A. Herbal Drugs Used in Respiratory System Diseases. Dent. Med. Probl. 2007, 44, 507–515. [Google Scholar]
- Lee, M.R. The history of Ephedra (ma-huang). J. R. Coll. Physicians Edinb. 2011, 41, 78–84. [Google Scholar] [CrossRef]
- Bucci, L.E. Selected herbals and human exercise performance. Am. J. Clin. Nuir. 2000, 72, 624S–636S. [Google Scholar] [CrossRef] [PubMed]
- Wee, S.; Ordway, G.A.; Woolverton, W.L. Reinforcing effect of pseudoephedrine isomers and the mechanism of action. Eur. J. Pharmacol. 2004, 493, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Laccourreye, O.; Werner, A.; Giroud, J.P.; Couloigner, V.; Bonfils, P.; Bondon-Guitton, E. Benefits, limits and danger of ephedrine and pseudoephedrine as nasal decongestants. Europ. Annals Otorhinolaatyngol. Head Neck Dis. 2015, 132, 31–34. [Google Scholar]
- Eccles, R. Substitution of phenylephrine for pseudoephedrine as a nasal decongestant. An illogical way to control methamphetamine abuse. Br. J. Clin. Pharmacol. 2006, 63, 10–14. [Google Scholar] [CrossRef]
- Wellington, K.; Jarvis, B. Cetirizine/Pseudoephedrine. Drugs 2001, 61, 2231–2240. [Google Scholar] [CrossRef]
- Piątek, A.; Koziarska-Rościszewska, M.; Zawilska, J.B. Recreational use of over-the counter drugs: The doping of the brain. Alcohol. Drug Addict. 2015, 28, 65–77. [Google Scholar] [CrossRef]
- Rice, J.; Proctor, K.; Lopardo, L.; Evans, S.; Kasprzyk-Hordern, B. Stereochemistry of ephedrine and its environmental significance: Exposure and effects directed approach. J. Hazard. Mater. 2018, 348, 39–46. [Google Scholar] [CrossRef]
- Chiarugi, A.; Camaioni, A. Update on the pathophysiology and treatment of rhinogenic headache: Focus on the ibuprofen/pseudoephedrine combination. Acta Otorinol. Ital. 2019, 39, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Trinh, K.V.; Kim, J.; Ritsma, A. Effect of pseudoephedrine in sport: A systemic review. BMJ Open Sport Exerc. Med. 2015, 1, e000066. [Google Scholar] [CrossRef]
- Gheorghiev, M.D.; Hosseini, F.; Moran, J.; Cooper, C.E. Effects of pseudoephedrine on parameters affecting exercise performance: A meta-analysis. Sports Med. Open 2018, 4, 44. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Feng, F.; Wang, X.Q.; Zhu, L. Influences of urinary pH on the pharmacokinetics of three amphetamine-type stimulants using a new high-performance liquid chromatographic method. J. Pharm. Sci. 2009, 98, 728–738. [Google Scholar] [CrossRef] [PubMed]
- Pawlaczyk, M.; Korzeniowska, K.; Jabłecka, A. Safety and efficacy of pseudoephedrine. Farm. WSP 2017, 10, 67–71. [Google Scholar]
- Kale, P. Pharmacokinetics and bioavailability of single dose ibuprofen and pseudoephedrine alone or in combination: A randomized three-period, cross-over trial in healthy Indian volunteers. Front. Pharmacol. 2014, 5. [Google Scholar] [CrossRef]
- Flanagan, S.; Minassian, S.L.; Prokocimer, P. Pharmacokinetics of Tedizolid and Pseudoephedrine Administered Alone or in Combination in Healthy Volunteers. J. Clin. Med. 2018, 7, 150. [Google Scholar] [CrossRef] [PubMed]
- Pseudoephedrine. Available online: https://www.drugs.com/monograph/pseudoephedrine.html (accessed on 21 March 2021).
- Summary of Product Characteristics Sudafed. Available online: https://rejestrymedyczne.ezdrowie.gov.pl/rpl/search/public (accessed on 21 March 2021).
- Werler, M.M. Teratogen update: Pseudoephedrine. Birth Defects Res. (Part 1) 2006, 76, 445–452. [Google Scholar] [CrossRef]
- Hernandez, R.K.; Mitchell, A.A.; Werler, M.M. Decongestant use during pregnancy and its association with preterm delivery. Birth Defects Res. (Part A) 2010, 88, 715–721. [Google Scholar] [CrossRef]
- Gelotte, C.K.; Albrecht, H.H.; Hynson, J.; Gallagher, V. A Multicenter, Randomized, Placebo-Controlled Study of Pseudoephedrine for the Temporary Relief of Nasal Congestion in Children with the Common Cold. J. Clin. Pharmacol. 2019, 59, 1573–1583. [Google Scholar] [CrossRef]
- What is the Role of Alpha Agonists in Urinary Incontinence Treatment? Available online: https://www.medscape.com/answers/452289-172460/what-is-the-role-of-alpha-agonists-in-urinary-incontinence-treatment (accessed on 20 March 2021).
- Barroso, O.; Goudreault, D.; Banus, M.C.; Ayotte, C.; Mazzoni, I.; Boghosian, T.; Rabin, O. Determination of urinary concentrations of pseudoephedrine and cathine after therapeutic administration of pseudoephedrine-containing medications to healthy subjects: Implications for doping control analysis of these stimulants banned in sport. Drug Test. Analysis 2011, 4, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Sherkat, A.A.; Sardana, N.; Safaee, S.B.; Lehman, E.B.; Craig, T.J. The role of pseudoephedrine on a daytime somnolence in patients suffering from perennial allergic rhinitis (PAR). Ann. Allergy Asthma Immunol. 2011, 106, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Salerno, S.M.; Jackson, J.L.; Berbano, E.P. Effect of oral pseudoephedrine on blood pressure and heart rate. Arch. Intern. Med. 2009, 6, 1686–1694. [Google Scholar] [CrossRef]
- Serhat, A.; Metehan, O. Acute coronary syndrome presenting after pseudoephedrine use and regression with beta-blocker therapy. Can. J. Cardiol. 2008, 24, e86–e88. [Google Scholar]
- Celic, A. ST elevation myocardial infarction presenting after use of pseudoephedrine. Cardiovasc. Toxicol. 2009, 9, 103–104. [Google Scholar] [CrossRef]
- Bharatula, A.; New, P.W. Cough mixture dependence and stroke: Implications for pseudoephedrine regulation. Med. J. Aust. 2011, 194, 427. [Google Scholar] [CrossRef]
- Ismailogullari, S.; Yetkin, M.F.; Erdogan, F.; Delibas, E.; Aksu, M.; Ersoy, A.O. Letter to the Editor, Pseudoephedrine-induced nonconvulsive status epilepticus. Epilepsy Behav. 2011, 20, 739–740. [Google Scholar] [CrossRef]
- Aziz, M.; Pervez, A.; Fatima, R.; Bansal, A. Case Report Pseudoephedrine Induced Ischemic Colitis: A Case Report and Review of Literature. Case Rep. Gastrointest. Med. 2018, 2018. [Google Scholar] [CrossRef]
- Özkaya, E.; Elinç-Aslan, M.S. Pseudoephedrine May Cause “Pigmenting” Fixed Drug Eruption. Dermatitis 2011, 22, 7–9. [Google Scholar] [CrossRef]
- Ozdemir, H.; Celik, N.G.; Tapisis, A.; Akay, B.N.; Ciftci, E.; Ince, E.; Dogru, U. Baboon syndrom induced by oral antitussive-decongestant agent in a child. Turk. J. Pediatr. 2010, 52, 659–661. [Google Scholar]
- Pseudoephedrine Side Effects. Available online: http://www.drugs.com/sfx/pseudoephedrine-side-effects.html (accessed on 19 April 2021).
- Fidan, S.; Izci, S.; Tellice, M.; Alizade, E.; Acar, G. ST elevation myocardial infarction after use of pseudoephedrine. Herz 2015, 40, 144–146. [Google Scholar] [CrossRef] [PubMed]
- Azzaro, A.A.; Van Den Berg, C.M.; Ziemniak, J.; Kemper, E.M.; Blob, L.F.; Campbell, B.J. Evaluation of the potential for pharmacodynamic and pharmacokinetic drug interactions between selegiline transdermal system and two sympathomimetic agents (pseudoephedrine and phenylpropanolamine) in healthy volunteers. J. Clin. Pharmacol. 2007, 47, 978–990. [Google Scholar] [CrossRef]
- Medications & Substances Causing False Positives. Available online: http://www.passyourdrugtest.com/false-positives.htm. (accessed on 1 February 2021).
- Moeller, K.E.; Lee, K.C.; Kissack, J.C. Urine drug screening: Practical guide for clinicans. Mayo Clin Proc. 2008, 83, 66–76. [Google Scholar] [CrossRef]
- Gołda, A.; Dymek, J.; Polak, W.; Uram, A.; Uman-Ntuk, E.; Skowron, A. An assessment of counsalting quality provided by community pharmacies to patients during expedition of pseudoephedrine. Farm. Pol. 2019, 75, 111–118. [Google Scholar] [CrossRef]
- Sobczak, Ł.; Goryński, K. Drugs in pharmacies and the issue of doping in sports. Farm. Pol. 2018, 74, 199–205. [Google Scholar] [CrossRef]
- Gradidge, P.J.I.; Constantinou, D.; Heard, S.M.; King, C.; Morris-Eyton, H. Effect of a therapeutic dose of pseudoephedrine on swimmers’s performance. S. Afr. J. SM. 2013, 25, 43–46. [Google Scholar] [CrossRef]
- Suchecka, D.; Kucharska-Mazur, J.; Groszewska, K.; Mak, M.; Samochowiec, J.; Samochowiec, A. Analysis of the phenomenon of over-the counter drug abuse and not controlled herbs trade by polish adolescents: Part, I. Med. Prac. 2017, 68, 413–422. [Google Scholar] [CrossRef]
- Brzeczko, A.W.; Leech, R.; Stark, J.G. The advent of a new pseudoephedrine product to combat methamphetamine abuse. Am. J. Drug Alcohol Abuse 2013, 39, 284–290. [Google Scholar] [CrossRef]
- Zuba, D. Medicines Containing Ephedrine and Pseudoephedrine as a Source of Methcathinone. Probl. Forensic Sci. 2007, 71, 323–333. [Google Scholar]
- Kałwa, A.; Habrat, B. Cognitive dysfunctions caused by excessive exposure to manganese compounds. Cognitive disturbances in intravenous users of ephedrone (methcathinone) with manganese compounds. Psychiatr. Pol. 2015, 49, 305–314. [Google Scholar] [CrossRef]
- Stepens, A.; Logina, I.; Liguts, V.; Aldins, P.; Eksteina, I.; Platkajis, A.; Martinsone, I.; Terauds, E.; Rozentale, B.; Donaghy, M. A parkinsonian syndrome in methcathinone users and the role of manganese. N. Engl. J. Med. 2008, 358, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Sikk, K.; Haldre, S.; Aquilonius, S.T.; Taba, P. Manganese–Induced Parkinsonism due to Ephedrone Abuse. Parkinsons. Dis. 2011, 865319. [Google Scholar] [CrossRef]
- Presley, B.; Bianchi, B.; Coleman, J.; Diamond, F.; McNally, G. Efficiency of extraction and conversion of pseudoephedrine to methamphetamine from tamper-resistant and non-tamper-resistant formulations. J. Pharmaceut. Biomed. Anal. 2018, 156, 16–22. [Google Scholar] [CrossRef] [PubMed]
| Pharmacokinetic Parameters of Pseudoephedrine | |
|---|---|
| Onset of action | 30 min |
| Time to reach Cmax | 1–4 h |
| Time to reach Cmax after administration of the extended-release formulation | 2–6 h |
| Duration of action | 4–12 h |
| Distribution coefficient | 2.64–3.51 l/kg |
| Biological half-life | 3–16 h |
| Renal clearance | 0.44–0.46 l/h/kg, 7.3–7.7 mL/min/kg |
| Pseudoephedrine Adverse Effects |
|---|
| CNS stimulation—sleep disturbances (>30%), anxiety, headache, muscle tremor, confusion |
| Dryness of mucous membranes of the mouth, nose and throat (>15%) |
| Digestive tract dysfunction—indigestion, nausea, vomiting, decreased appetite, irritation of the gastric mucosa (5%) |
| Cardiac arrhythmias, tachycardia, increased blood pressure |
| Excessive sweating, hyperglycemia, urination disorders |
| Allergic reactions—redness, rashes |
| Psychological dependence |
| Pseudoephedrine Interactions | |
|---|---|
| Other Concomitantly Used Medicines and Substances | Type and Consequence of Interaction |
| Antacids (e.g., aluminium hydroxide), proton pump inhibitors | Increase in PSE absorption rate |
| Kaolin clay | Decreased rate of absorption of PSE due to its adsorption on the surface of kaolin clay |
| Digitalis glycosides | Increased ectopic activity of the heart’s conducting system, arrhythmia |
| MAO inhibitors (phenelzine, selegiline, tranylcypromine, procarbazine) | Synergistic sympathomimetic effect, significant increase in blood pressure, hypertensive crisis, bradycardia— 14-day interval between drugs is required |
| Tricyclic antidepressants | Increased effect of PSE, increased risk of hypertension and cardiac arrhythmias—concomitant use is not recommended |
| Methyldopa, guanethidine, reserpine | PSE reduces the antihypertensive effect in addition to the drugs—concomitant use is not recommended |
| Appetite suppressants | Risk of increased blood pressure, increased heart rate—concomitant use is not recommended |
| Ergotamine, dihydroergotamine, linezolid, oxytocin, ephedrine, phenylephrine, bromocriptine | Risk of vasoconstriction and increase in blood pressure—concomitant use is not recommended |
| Urine alkalinisation (e.g., sodium bicarbonate) | Urine alkalinisation increases the reabsorption of PSE, the risk of seizures, anxiety, restlessness, insomnia, tachycardia |
| Inhalation agents for general anaesthesia | Acute hypertensive reaction in the perioperative period—discontinuation of PSE is recommended 24 h before the planned general anaesthesia |
| Caffeine | Elevated body temperature, hyperglycaemia, insulinaemia, increased C-peptide levels |
| Ethyl alcohol | Acute psychosis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Głowacka, K.; Wiela-Hojeńska, A. Pseudoephedrine—Benefits and Risks. Int. J. Mol. Sci. 2021, 22, 5146. https://doi.org/10.3390/ijms22105146
Głowacka K, Wiela-Hojeńska A. Pseudoephedrine—Benefits and Risks. International Journal of Molecular Sciences. 2021; 22(10):5146. https://doi.org/10.3390/ijms22105146
Chicago/Turabian StyleGłowacka, Krystyna, and Anna Wiela-Hojeńska. 2021. "Pseudoephedrine—Benefits and Risks" International Journal of Molecular Sciences 22, no. 10: 5146. https://doi.org/10.3390/ijms22105146
APA StyleGłowacka, K., & Wiela-Hojeńska, A. (2021). Pseudoephedrine—Benefits and Risks. International Journal of Molecular Sciences, 22(10), 5146. https://doi.org/10.3390/ijms22105146






