Involvement of Huntingtin in Development and Ciliary Beating Regulation of Larvae of the Sea Urchin, Hemicentrotus pulcherrimus
Abstract
1. Introduction
2. Results
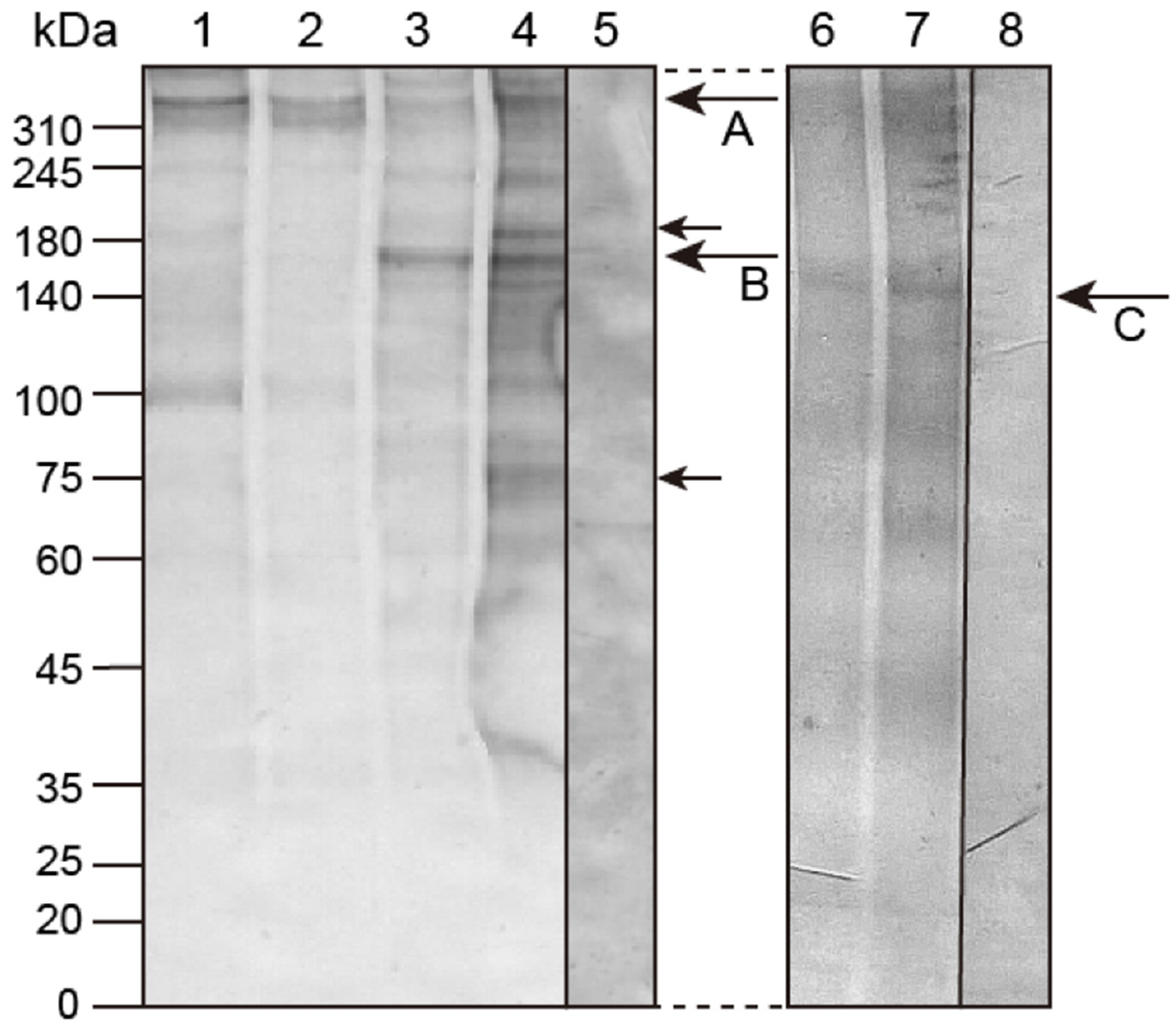
2.1. Molecular Modification of Hp-Htt during Early Development
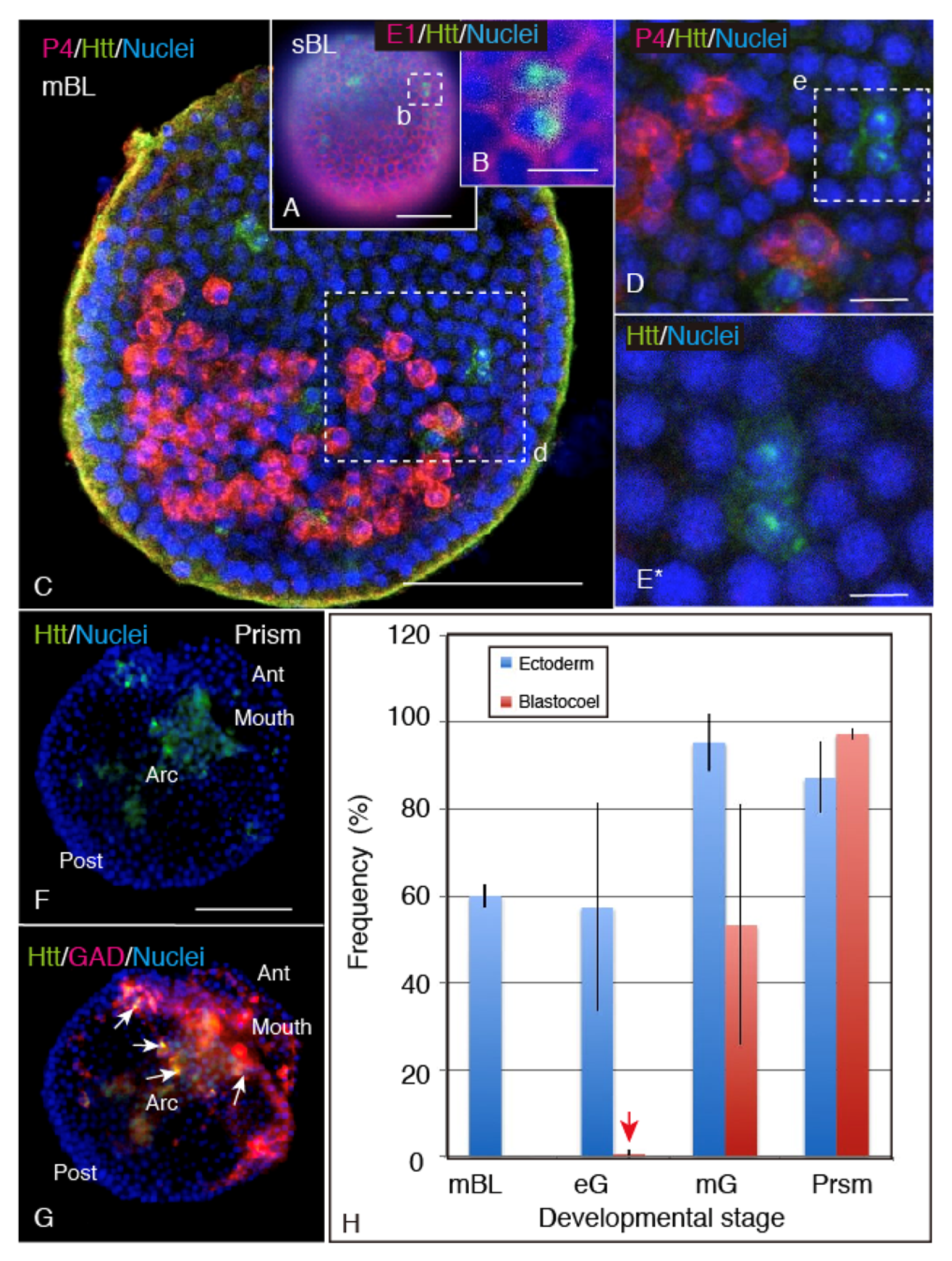
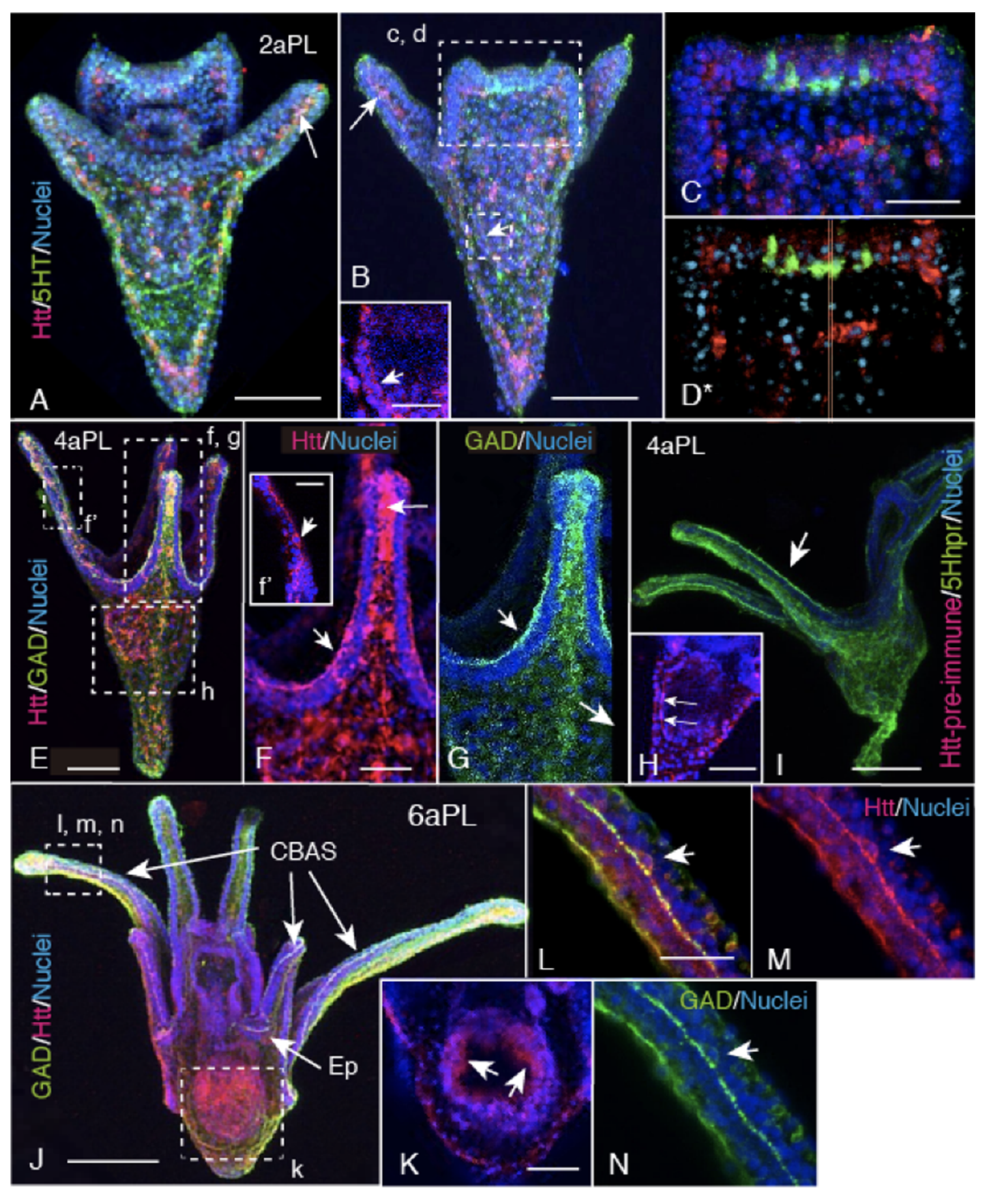
2.2. Spatiotemporal HP-Htt Expression Pattern
2.3. Hp-Htt Gene Knockdown by Its Morpholino Antisense Oligomers
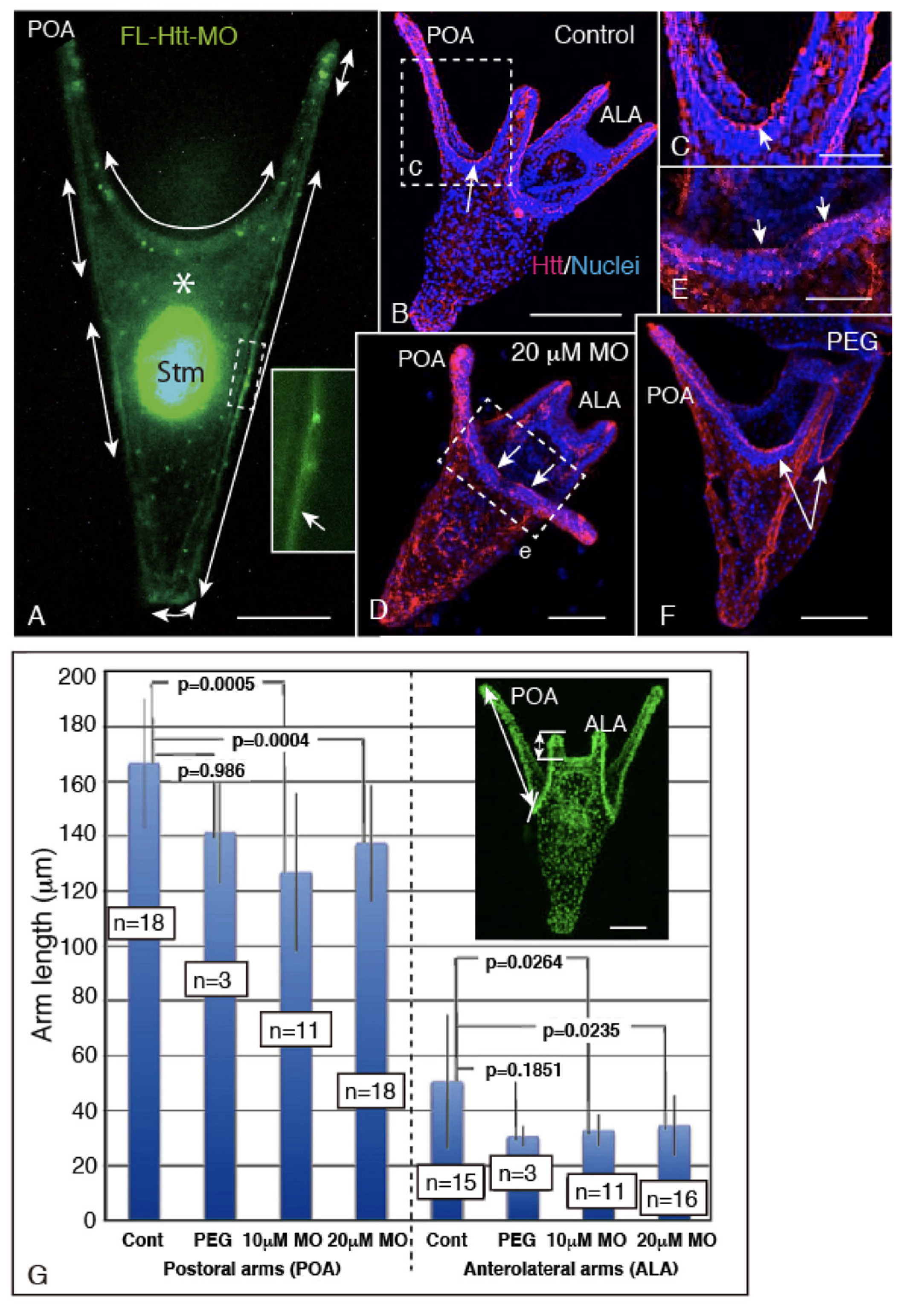
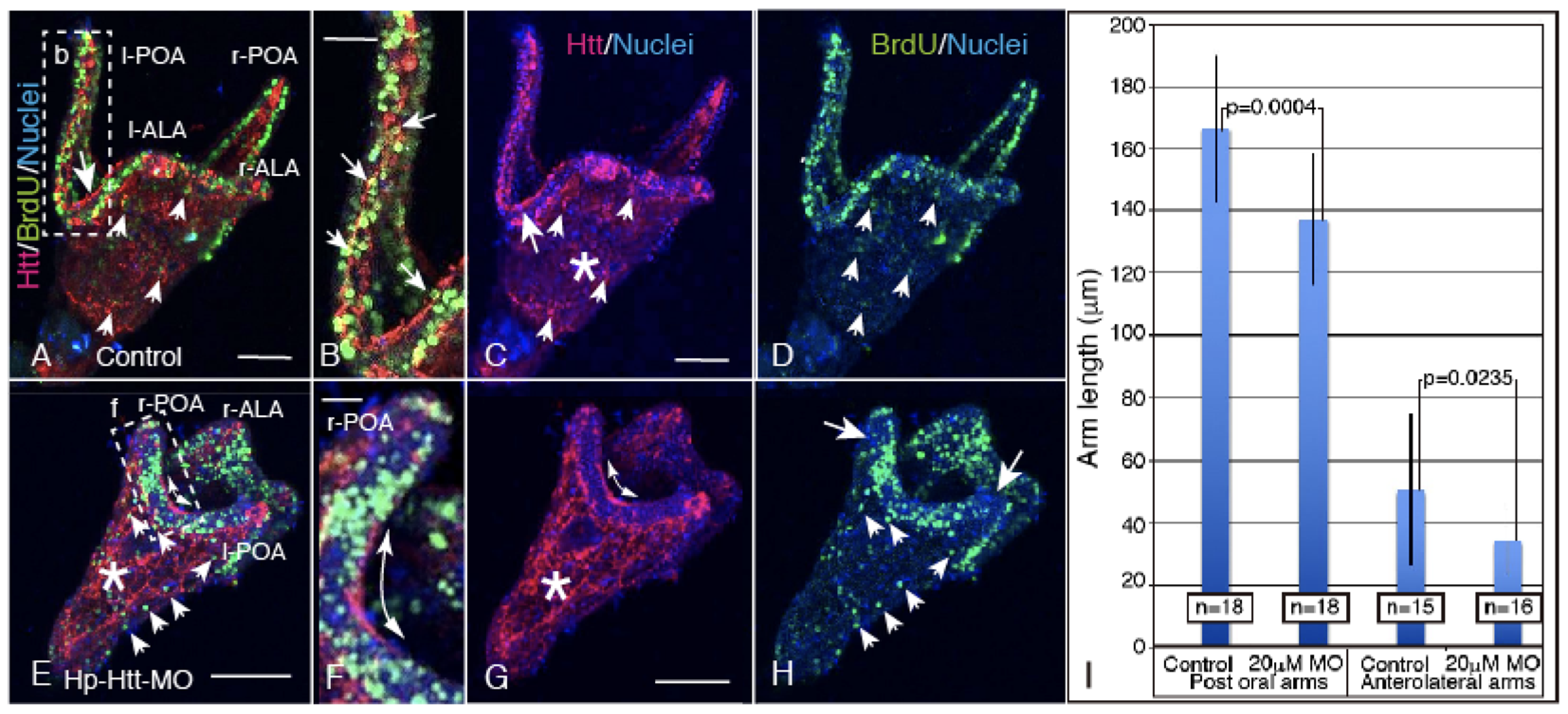
2.4. FL-Htt-MO Formed Shortened Larval Arms and Showed Decreased Cell Proliferation
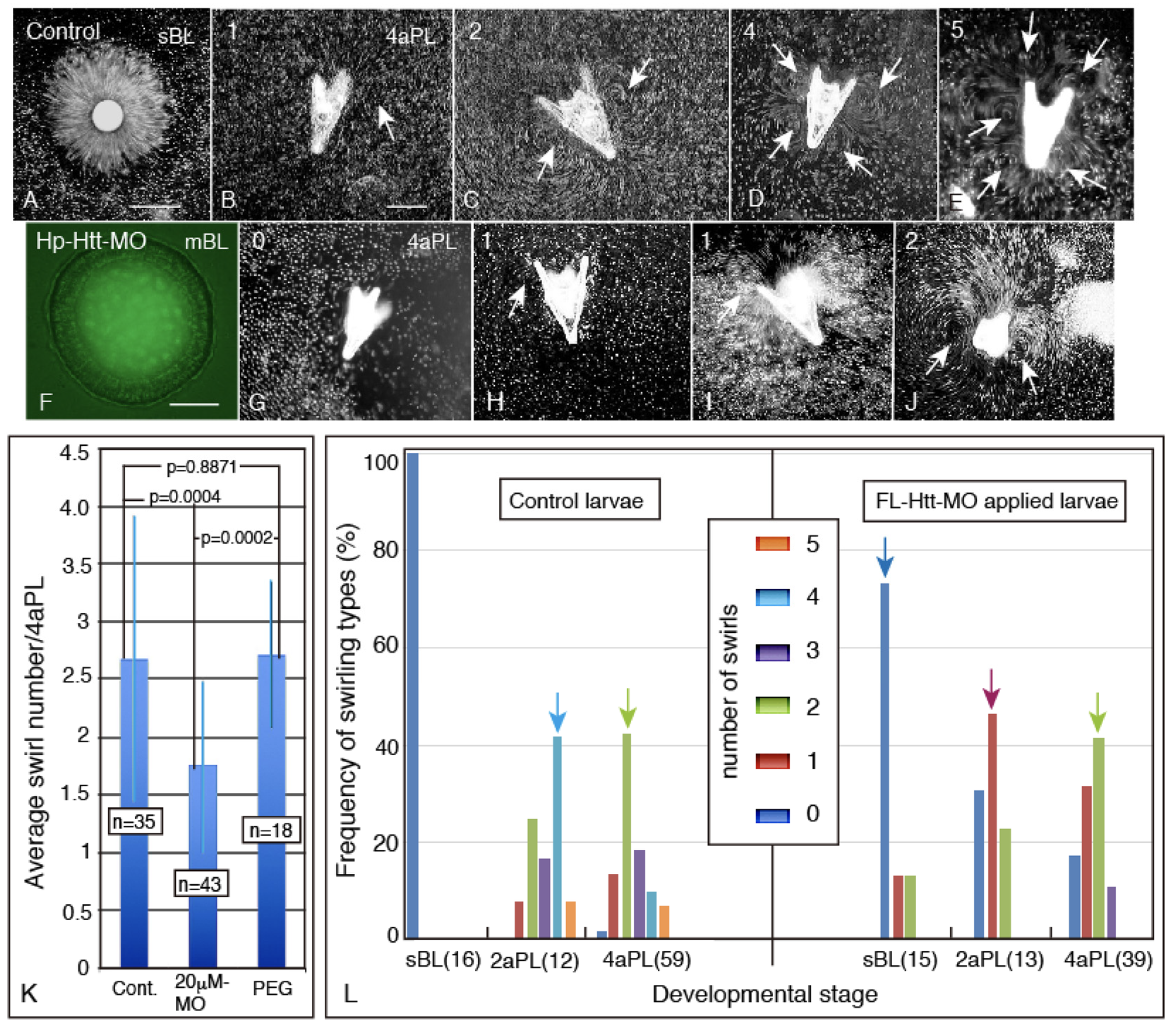
2.5. Disruption of the Swirling Track Pattern Created by the Ciliary Beating Activity in Hp-Htt-MO Embryo and Plutei
3. Discussion
3.1. Hp-Htt Expression Pattern during Development
3.2. Swirling Patterns
3.3. Htt in Morphogenesis
4. Materials and Methods
4.1. Animal Preparation
4.2. Raising Antibody
4.3. Immunoblotting
4.4. Whole-Mount Immunohistochemistry
4.5. FL-Htt-MO Application
4.6. Cell Proliferation Analysis Using BrdU Incorporation
4.7. Swirling Track Pattern Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CBAS | Ciliary band-associated strand |
| FL-Htt-MO | Fluorescein-tagged Hp-Htt morpholino |
| GAD | Glutamic acid decarboxylase |
| HTT | Huntingtin (Huntington’s disease protein) |
| Hp-Htt | Huntington’s disease protein of the sea urchin Hemicentrotus pulcherrimus |
| l-, r-ALA | Left- and right-anterolateral arm |
| l-, r-POA | Left- and right-postoral arm |
| PEG | Endo-Porter-Polyethylene glycol |
| Tjp1 | Tight junction protein 1 |
| 2aPL | Two-arm pluteus larva |
| 4aPL | Four-arm pluteus larva |
| 5HT | Serotonin |
| 5HThpr | Serotonin receptor |
References
- The Huntington’s Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 1993, 72, 971–983. [Google Scholar] [CrossRef]
- Zhang, S.; Feany, M.B.; Saraswati, S.; Littleton, J.T.; Perrimon, N. Inactivation of Drosophila Huntingtin affects long-term adult functioning and the pathogenesis of a Huntington’s disease model. Dis. Model. Mech. 2009, 2, 247–266. [Google Scholar] [CrossRef] [PubMed]
- Haremaki, T.; Deglincerti, A.; Brivanlou, A.H. Huntingtin is required for ciliogenesis and neurogenesis during early Xenopus development. Dev. Biol. 2015, 408, 305–315. [Google Scholar] [CrossRef]
- Strehlow, A.N.; Li, J.Z.; Myers, R.M. Wild-type huntingtin participates in protein trafficking between the Golgi and the extracellular space. Hum. Mol. Genet. 2006, 16, 391–409. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-B.; Kadakkuzha, B.M.; Liu, X.-A.; Akhmedov, K.; Kandel, E.R.; Puthanveettil, S.V. Huntingtin Is Critical Both Pre- and Postsynaptically for Long-Term Learning-Related Synaptic Plasticity in Aplysia. PLoS ONE 2014, 9, e103004. [Google Scholar] [CrossRef] [PubMed]
- Keryer, G.; Pineda, J.R.; Liot, G.; Kim, J.; Dietrich, P.; Benstaali, C.; Smith, K.; Cordelières, F.P.; Spassky, N.; Ferrante, R.J.; et al. Ciliogenesis is regulated by a huntingtin-HAP1-PCM1 pathway and is altered in Huntington disease. J. Clin. Investig. 2011, 121, 4372–4382. [Google Scholar] [CrossRef] [PubMed]
- Hagenlocher, C.; Walentek, P.; Müller, C.; Thumberger, T.; Feistel, K. Ciliogenesis and cerebrospinal fluid flow in the developing Xenopus brain are regulated by foxj1. Cilia 2013, 2, 12. [Google Scholar] [CrossRef] [PubMed]
- Thouvenin, O.L.; Keiser, L.; CantautBelarif, Y.; Carbo-Tano, M.; Verweij, F.; JurischYaksi, N.; Bardet, P.-L.; Van Niel, G.; Gallaire, F.; Wyart, C. Origin of the bidirectionality of cerebrospinal fluid flow and impact on long-range transport between brain and spinal cord. BioRxiv Prepr. 2019. [Google Scholar] [CrossRef]
- Eichele, G.; Bodenschatz, E.; Ditte, Z.; Günther, A.-K.; Kapoor, S.; Wang, Y.; Westendorf, C. Cilia-driven flows in the brain third ventricle. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190154. [Google Scholar] [CrossRef]
- Liu, G.; Zhou, Y.; Liu, D.; Wang, Q.; Ruan, Z.; He, Q.; Zhang, L. Global Transcriptome Analysis of the Tentacle of the Jellyfish Cyanea capillata Using Deep Sequencing and Expressed Sequence Tags: Insight into the Toxin- and Degenerative Disease-Related Transcripts. PLoS ONE 2015, 10, e0142680. [Google Scholar] [CrossRef]
- Candiani, S.; Pestarino, M.; Cattaneo, E.; Tartari, M. Characterization, developmental expression and evolutionary features of the huntingtin gene in the amphioxus Branchiostoma floridae. BMC Dev. Biol. 2007, 7, 127. [Google Scholar] [CrossRef]
- Gissi, C.; Pesole, G.; Cattaneo, E.; Tartari, M. Huntingtin gene evolution in Chordata and its peculiar features in the ascidian Ciona genus. BMC Genom. 2006, 7, 288. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, J.S.; Zinovyeva, A.; Yagi, K.; Makabe, K.W.; Raff, R.A. Neural expression of the Huntington’s disease gene as a chordate evolutionary novelty. J. Exp. Zool. Part B Mol. Dev. Evol. 2003, 297, 57–64. [Google Scholar] [CrossRef]
- Tartari, M.; Gissi, C.; Sardo, V.L.; Zuccato, C.; Picardi, E.; Pesole, G.; Cattaneo, E. Phylogenetic Comparison of Huntingtin Homologues Reveals the Appearance of a Primitive polyQ in Sea Urchin. Mol. Biol. Evol. 2008, 25, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Zuccato, C.; Valenza, M.; Cattaneo, E. Molecular Mechanisms and Potential Therapeutical Targets in Huntington’s Disease. Physiol. Rev. 2010, 90, 905–981. [Google Scholar] [CrossRef]
- Belardi, B.; Hamkins-Indik, T.; Harris, A.R.; Kim, J.; Xu, K.; Fletcher, D.A. A Weak Link with Actin Organizes Tight Junctions to Control Epithelial Permeability. Dev. Cell 2020, 54, 792–804.e7. [Google Scholar] [CrossRef]
- Katow, H.; Uemura, I.; Yoshida, H.; Katow, T. Tjp1 expression at the ciliary band-associated strand (CBAS) of sea urchin larva. In Proceedings of the Annual Meeting of Zoological Society of Japan, Tohoku Branch Conference (Aomori), Yamagata, Japan, 29 July 2017. [Google Scholar]
- Katow, H.; Abe, K.; Katow, T.; Zamani, A.; Abe, H. Development of the GABA-ergic signaling system and its role in larval swimming in sea urchin. J. Exp. Biol. 2013, 216, 1704–1716. [Google Scholar] [CrossRef]
- Yaguchi, S.; Katow, H. Expression of Tryptophan 5-Hydroxylase Gene during Sea Urchin Neurogenesis and Role of Serotonergic Nervous System in Larval Behavior. J. Comp. Neurol. 2003, 466, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Shook, D.; Keller, R. Mechanisms, mechanics and function of epithelial–mesenchymal transitions in early development. Mech. Dev. 2003, 120, 1351–1383. [Google Scholar] [CrossRef] [PubMed]
- Goffredo, D.; Rigamonti, D.; Tartari, M.; De Micheli, A.; Verderio, C.; Matteol, M.; Zuccato, C.; Cattaneo, E. Calcium-dependent cleavage of endogenous wild-type Huntingtin in primary cortical neurons. J. Biol. Chem. 2002, 277, 39594–39598. [Google Scholar] [CrossRef]
- Katow, H.; Yoshida, H.; Katow, T.; Kiyomoto, M. Ontogeny of a synaptophysin-mediated GABA transmission mechanism from the ciliary band-associated strand to the ciliary band during the development of the sea urchin Hemicentrotus pulcher-rimus. PeerJ Prepr. 2018. [Google Scholar] [CrossRef]
- Katow, H.; Yoshida, H.; Kiyomoto, M. Initial report of γ-aminobutyric acidergic locomotion regulatory system and its 3-mercaptopropionic acid-sensitivity in metamorphic juvenile of sea urchin, Hemicentrotus pulcherrimus. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Katow, H.; Abe, K.; Katow, T.; Yoshida, H.; Kiyomoto, M. Involvement of Netrin/Unc-5 Interaction in Ciliary Beating and in Pattern Formation of the Ciliary Band-Associated Strand (CBAS) in the Sea Urchin, Hemicentrotus pulcherrimus. Int. J. Mol. Sci. 2020, 21, 6587. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Lee, D.H.; Taylor, J.; Vandelft, M.; Truant, R. Huntingtin contains a highly conserved nuclear export signal. Hum. Mol. Genet. 2003, 12, 1393–1403. [Google Scholar] [CrossRef]
- Schulte, J.; Littleton, J.T. The biological function of the Huntingtin protein and its relevance to Huntington’s Disease pathology. Curr. Trends Neurol. 2011, 5, 65–78. [Google Scholar]
- McKinstry, S.U.; Karadeniz, Y.B.; Worthington, A.K.; Hayrapetyan, V.Y.; Ozlu, M.I.; Serafin-Molina, K.; Risher, W.C.; Ustunkaya, T.; Dragatsis, I.; Zeitlin, S.; et al. Huntingtin Is Required for Normal Excitatory Synapse Development in Cortical and Striatal Circuits. J. Neurosci. 2014, 34, 9455–9472. [Google Scholar] [CrossRef] [PubMed]
- Elias, S.; Thion, M.S.; Yu, H.; Sousa, C.M.; Lasgi, C.; Morin, X.; Humbert, S. Huntingtin Regulates Mammary Stem Cell Division and Differentiation. Stem Cell Rep. 2014, 2, 491–506. [Google Scholar] [CrossRef]
- Martin, D.D.O.; Schmidt, M.E.; Nguyen, Y.T.; Lazic, N.; Hayden, M.R. Identification of a novel caspase cleavage site in huntingtin that regulates mutant huntingtin clearance. FASEB J. 2018, 33, 3190–3197. [Google Scholar] [CrossRef]
- Rigamonti, D.; Bauer, J.H.; De-Fraja, C.; Conti, L.; Sipione, S.; Sciorati, C.; Clementi, E.; Hackam, A.; Hayden, M.R.; Li, Y.; et al. Wild-Type Huntingtin Protects from Apoptosis Upstream of Caspase-3. J. Neurosci. 2000, 20, 3705–3713. [Google Scholar] [CrossRef]
- Graham, R.K.; Deng, Y.; Slow, E.J.; Haigh, B.; Bissada, N.; Lu, G.; Pearson, J.; Shehadeh, J.; Bertram, L.; Murphy, Z.; et al. Cleavage at the Caspase-6 Site Is Required for Neuronal Dysfunction and Degeneration Due to Mutant Huntingtin. Cell 2006, 125, 1179–1191. [Google Scholar] [CrossRef] [PubMed]
- Wellington, C.L.; Ellerby, L.M.; Gutekunst, C.-A.; Rogers, D.; Warby, S.; Graham, R.K.; Loubser, O.; Van Raamsdonk, J.; Singaraja, R.; Yang, Y.-Z.; et al. Caspase Cleavage of Mutant Huntingtin Precedes Neurodegeneration in Huntington’s Disease. J. Neurosci. 2002, 22, 7862–7872. [Google Scholar] [CrossRef]
- Katow, H.; Yaguchi, S.; Kiyomoto, M.; Washio, M. The 5-HT receptor cell is a new member of secondary mesenchyme cell descendants and forms a major blastocoelar network in sea urchin larvae. Mech. Dev. 2004, 121, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Katow, H.; Suyemitsu, T.; Ooka, S.; Yaguchi, J.; Jin-Nai, T.; Kuwahara, I.; Katow, T.; Yaguchi, S.; Abe, H. Development of a dopaminergic system in sea urchin embryos and larvae. J. Exp. Biol. 2010, 213, 2808–2819. [Google Scholar] [CrossRef]
- Hoyer, D.; Clarke, D.E.; Fozard, J.R.; Hartig, P.R.; Martin, G.R.; Mylecharane, E.J.; Saxena, P.R.; Humphrey, P.P. In-ternational Union of Pharmacology classification of receptors for 5-hydroxytryptamine (Serotonin). Pharmacol. Rev. 1994, 46, 157–203. [Google Scholar] [PubMed]
- Olstad, E.W.; Ringers, C.; Hansen, J.N.; Wens, A.; Brandt, C.; Wachten, D.; Yaksi, E.; Jurisch-Yaksi, N. Ciliary Beating Compartmentalizes Cerebrospinal Fluid Flow in the Brain and Regulates Ventricular Development. Curr. Biol. 2019, 29, 229–241.e6. [Google Scholar] [CrossRef]
- Maiuri, T.; Woloshansky, T.; Xia, J.; Truant, R. The huntingtin N17 domain is a multifunctional CRM1 and Ran-dependent nuclear and cilial export signal. Hum. Mol. Genet. 2013, 22, 1383–1394. [Google Scholar] [CrossRef] [PubMed]
- Mahuzier, A.; Shihavuddin, A.; Fournier, C.; Lansade, P.; Faucourt, M.; Menezes, N.; Meunier, A.; Garfa-Traoré, M.; Carlier, M.-F.; Voituriez, R.; et al. Ependymal cilia beating induces an actin network to protect centrioles against shear stress. Nat. Commun. 2018, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Godin, J.D.; Colombo, K.; Molina-Calavita, M.; Keryer, G.; Zala, D.; Charrin, B.C.; Dietrich, P.; Volvert, M.-L.; Guillemot, F.; Dragatsis, I.; et al. Huntingtin Is Required for Mitotic Spindle Orientation and Mammalian Neurogenesis. Neuron 2010, 67, 392–406. [Google Scholar] [CrossRef]
- Rodrigues, F.B.; Byrne, L.M.; De Vita, E.; Johnson, E.B.; Hobbs, N.Z.; Thornton, J.S.; Scahill, R.I.; Wild, E.J. Cerebrospinal fluid flow dynamics in Huntington’s disease evaluated by phase contrast MRI. Eur. J. Neurosci. 2019, 49, 1632–1639. [Google Scholar] [CrossRef]
- Saudou, F.; Humbert, S. The Biology of Huntingtin. Neuron 2016, 89, 910–926. [Google Scholar] [CrossRef] [PubMed]
- Marinković, M.; Berger, J.; Jékely, G. Neuronal coordination of motile cilia in locomotion and feeding. Philos. Trans. R. Soc. B Biol. Sci. 2020, 375, 20190165. [Google Scholar] [CrossRef]
- Wada, Y.; Mogami, Y.; Baba, S. Modification of ciliary beating in sea urchin larvae induced by neurotransmitters: Beat-plane rotation and control of frequency fluctuation. J. Exp. Biol. 1997, 200, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.-H.J.; Frey, A.S.; Alsdorf, S.A.; Kerner, J.A.; Kosinski, C.M.; Mangiarini, L.; Penney, J.B., Jr.; Davies, S.W.; Bates Gp Young, A.B. Altered neurotransmitter receptor expression in transgenic mouse models of Huntington’s disease. Phil. Trans. R. Soc. Lond. B 1999, 354, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Garret, M.; Du, Z.; Chazalon, M.; Cho, Y.H.; Baufreton, J. Alteration of GABAergic neurotransmission in Huntington’s dis ease. CNS Neurosci. Ther. 2018, 24, 292–300. [Google Scholar] [CrossRef]
- Molina-Calavita, M.; Barnat, M.; Elias, S.; Aparicio, E.; Piel, M.; Humbert, S. Mutant Huntingtin Affects Cortical Progenitor Cell Division and Development of the Mouse Neocortex. J. Neurosci. 2014, 34, 10034–10040. [Google Scholar] [CrossRef]
- Sidik, H.; Ang, C.J.; Pouladi, M.A. Huntingtin confers fitness but is not embryonically essential in zebrafish development. Dev. Biol. 2020, 458, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Kinjo, S.; Kiyomoto, M.; Yamamoto, T.; Ikeo, K.; Yaguchi, S. HpBase: A genome database of a sea urchin, Hemicentrotus pulcherrimus. Dev. Growth Differ. 2018, 60, 174–182. [Google Scholar] [CrossRef]
- Shimizu, K.; Noro, N.; Matsuda, R. Micromere Differentiation in the Sea Urchin Embryo: Expression of Primary Mesenchyme Cell Specific Antigen during Development. (sea urchin/micromere/primary mesenchyme cell/monoclonal antibody). Dev. Growth Differ. 1988, 30, 35–47. [Google Scholar] [CrossRef]
- Wakayama, N.; Katow, T.; Katow, H. Characterization and Endocytic Internalization of Epith-2 Cell Surface Glycoprotein during the Epithelial-to-Mesenchymal Transition in Sea Urchin Embryos. Front. Endocrinol. 2013, 4, 112. [Google Scholar] [CrossRef]
- Showman, R.M.; Foerder, C.A. Removal of the fertilization membrane of sea urchin embryos employing aminotriazole. Exp. Cell Res. 1979, 120, 253–255. [Google Scholar] [CrossRef]

| Antigen | Host | Antibody Type | Applied Dilution | References |
|---|---|---|---|---|
| Sp-Htt (Hp-Htt) | Rabbit | Polyclonal | 1:500 | This study |
| Hp-GAD | Rabbit | Polyclonal | 1:500 | [23] |
| Hp-Tjp1 | Rabbit | Polyclonal | 1:1000 | [18] |
| Hp-5HThpr | Mouse | Polyclonal | 1:200 | [19] |
| P4 | Mouse | Monoclonal | No dilution | [49] |
| Serotonin | Rabbit | Polyclonal | 1:1000 | Sigma-Aldrich, St. Louse, MO, USA |
| BrdU | Mouse | Polyclonal | 1:250 | Sigma-Aldrich, St. Louse, MO, USA |
| Epith-2 | Mouse | Monoclonal | 1:100 | [50] |
| Antigen | Labeled Fluorescent Dye | Applied Dilution |
|---|---|---|
| Rabbit | Alexa Fluor 488 | 1:500 |
| Rabbit | Alexa Fluor 568 | 1:500 |
| Rabbit | Alexa Fluor 594 | 1:500 |
| Rabbit | Zenon Alexa Fluor 488 | 1:300 |
| Rabbit | Zenon Alexa Fluor 568 | 1:300 |
| Rabbit | Zenon Alexa Fluor 594 | 1:300 |
| Mouse | Zenon Alexa Fluor 488 | 1:300 |
| Mouse | Zenon Alexa Fluor 568 | 1:300 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katow, H.; Katow, T.; Yoshida, H.; Kiyomoto, M. Involvement of Huntingtin in Development and Ciliary Beating Regulation of Larvae of the Sea Urchin, Hemicentrotus pulcherrimus. Int. J. Mol. Sci. 2021, 22, 5116. https://doi.org/10.3390/ijms22105116
Katow H, Katow T, Yoshida H, Kiyomoto M. Involvement of Huntingtin in Development and Ciliary Beating Regulation of Larvae of the Sea Urchin, Hemicentrotus pulcherrimus. International Journal of Molecular Sciences. 2021; 22(10):5116. https://doi.org/10.3390/ijms22105116
Chicago/Turabian StyleKatow, Hideki, Tomoko Katow, Hiromi Yoshida, and Masato Kiyomoto. 2021. "Involvement of Huntingtin in Development and Ciliary Beating Regulation of Larvae of the Sea Urchin, Hemicentrotus pulcherrimus" International Journal of Molecular Sciences 22, no. 10: 5116. https://doi.org/10.3390/ijms22105116
APA StyleKatow, H., Katow, T., Yoshida, H., & Kiyomoto, M. (2021). Involvement of Huntingtin in Development and Ciliary Beating Regulation of Larvae of the Sea Urchin, Hemicentrotus pulcherrimus. International Journal of Molecular Sciences, 22(10), 5116. https://doi.org/10.3390/ijms22105116





