New Potential Biomarkers for Chronic Kidney Disease Management—A Review of the Literature
Abstract
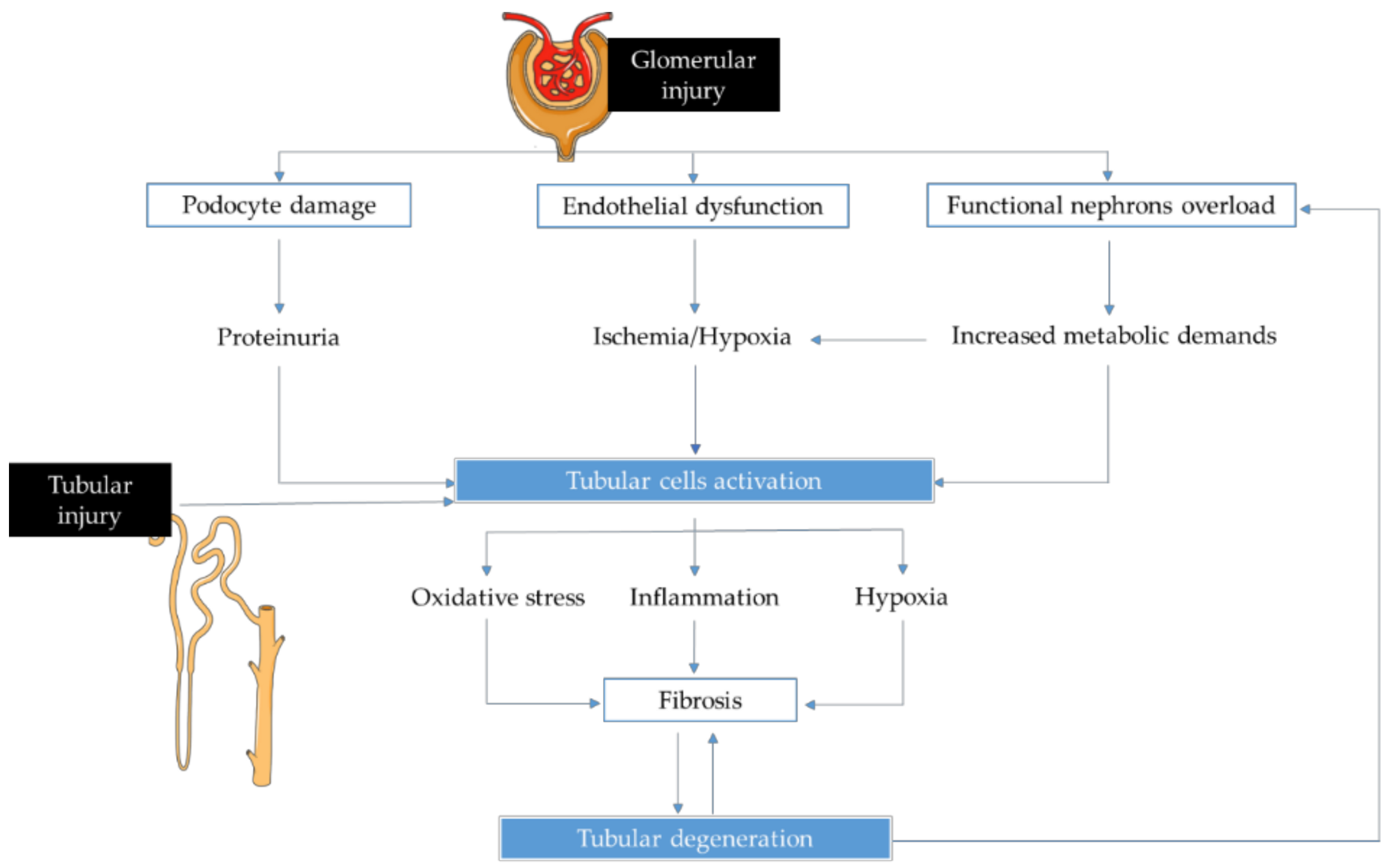
1. Introduction
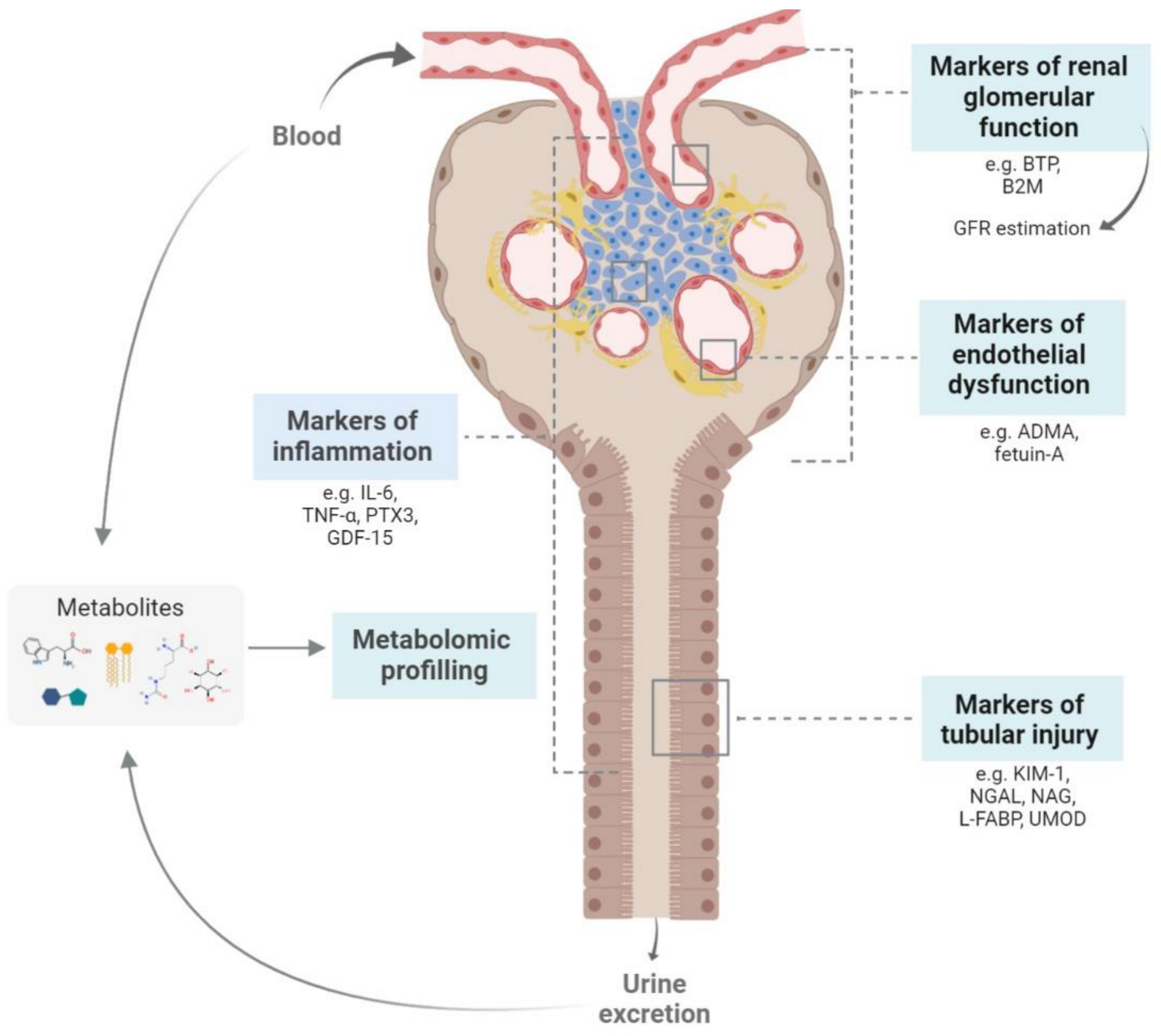
2. New Biomarkers for Chronic Kidney Disease Management
2.1. Biomarkers of Renal Function
2.1.1. Beta Trace Protein (BTP) and β2-Microglobulin (B2M)
2.1.2. Klotho
2.2. Biomarkers of Tubular Lesions
2.2.1. Neutrophil Gelatinase-Associated Lipocalin (NGAL), Kidney Injury Molecule-1 (KIM-1) and N-acetyl-β-D-glucosaminidase (NAG)
2.2.2. Liver-Type Fatty Acid Binding Protein (L-FABP)
2.2.3. Uromodulin (UMOD)
2.3. Biomarkers of Endothelial Dysfunction
2.3.1. Asymmetric Dimethylarginine (ADMA)
2.3.2. Fetuin-A
2.4. Biomarkers of Inflammation
2.5. Metabolomic Studies on CKD Biomarkers
3. Future Perspectives and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CKD | Chronic kidney disease |
| ESRD | End-stage renal disease |
| CVD | Cardiovascular disease |
| GFR | Glomerular filtration rate |
| BTP | Beta trace protein |
| B2M | β2-microglobulin |
| T2D | Type 2 diabetes |
| eGFR | Estimated glomerular filtration rate |
| NGAL | Neutrophil gelatinase-associated lipocalin |
| KIM-1 | Kidney injury molecule-1 |
| NAG | N-acetyl-β-D-glucosaminidase |
| L-FABP | Liver-type fatty acid binding protein |
| UMOD | Uromodulin |
| T1D | Type 1 diabetes |
| AKI | Acute kidney injury |
| NO | Nitric oxide |
| ADMA | Asymmetric dimethylarginine |
| IL-6 | Interleukin-6 |
| TNF-α | Tumor necrosis factor-α |
| PTX3 | Pentraxin 3 |
| CRP | C-reactive protein |
| GDF-15 | Growth differentiation factor-15 |
| cf-DNA | Cell-free DNA |
References
- Foundation, N.K. K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am. J. Kidney Dis. 2002, 39, S1–S266. [Google Scholar]
- Eckardt, K.U.; Coresh, J.; Devuyst, O.; Johnson, R.J.; Kottgen, A.; Levey, A.S.; Levin, A. Evolving importance of kidney disease: From subspecialty to global health burden. Lancet 2013, 382, 158–169. [Google Scholar] [CrossRef]
- Schlondorff, D.O. Overview of factors contributing to the pathophysiology of progressive renal disease. Kidney Int. 2008, 74, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, J.; Tanaka, T.; Nangaku, M. Recent advances in understanding of chronic kidney disease. F1000Res 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.R.; Parikh, C.R. Biomarkers of Acute and Chronic Kidney Disease. Annu. Rev. Physiol. 2019, 81, 309–333. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Stevens, P.E.; Bilous, R.W.; Coresh, J.; De Francisco, A.L.M.; De Jong, P.E.; Griffith, K.E.; Hemmelgarn, B.R.; Iseki, K.; Lamb, E.J.; et al. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2012, 3, 1–150. [Google Scholar] [CrossRef]
- Levey, A.S.; Coresh, J. Chronic kidney disease. Lancet 2012, 379, 165–180. [Google Scholar] [CrossRef]
- Santos-Silva, A.; Costa, E.; Alves, R. Chronic Kidney Disease. In Biomarkers of Cardiometabolic Risk, Inflammation and Disease; Palavra, F., Reis, F., Marado, D., Sena, A., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 95–111. [Google Scholar]
- George, J.A.; Gounden, V. Novel glomerular filtration markers. Adv. Clin. Chem. 2019, 88, 91–119. [Google Scholar] [CrossRef] [PubMed]
- Inker, L.A.; Tighiouart, H.; Coresh, J.; Foster, M.C.; Anderson, A.H.; Beck, G.J.; Contreras, G.; Greene, T.; Karger, A.B.; Kusek, J.W.; et al. GFR Estimation Using beta-Trace Protein and beta2-Microglobulin in CKD. Am. J. Kidney Dis. 2016, 67, 40–48. [Google Scholar] [CrossRef]
- Foster, M.C.; Levey, A.S.; Inker, L.A.; Shafi, T.; Fan, L.; Gudnason, V.; Katz, R.; Mitchell, G.F.; Okparavero, A.; Palsson, R.; et al. Non-GFR Determinants of Low-Molecular-Weight Serum Protein Filtration Markers in the Elderly: AGES-Kidney and MESA-Kidney. Am. J. Kidney Dis. 2017, 70, 406–414. [Google Scholar] [CrossRef]
- Filler, G.; Kusserow, C.; Lopes, L.; Kobrzyński, M. Beta-trace protein as a marker of GFR--history, indications, and future research. Clin. Biochem. 2014, 47, 1188–1194. [Google Scholar] [CrossRef] [PubMed]
- Spanaus, K.S.; Kollerits, B.; Ritz, E.; Hersberger, M.; Kronenberg, F.; von Eckardstein, A. Serum creatinine, cystatin C, and beta-trace protein in diagnostic staging and predicting progression of primary nondiabetic chronic kidney disease. Clin. Chem. 2010, 56, 740–749. [Google Scholar] [CrossRef] [PubMed]
- Donadio, C. Serum and urinary markers of early impairment of gfr in chronic kidney disease patients: Diagnostic accuracy of urinary β-trace protein. Am. J. Physiol. Renal. Physiol. 2010, 299, f1407–f1423. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, n.; Shi, h.; Zhang, L.; Zuo, L.; Xie, J.; Xie, D.; Karger, A.B.; Miao, S.; Ren, H.; Zhang, W.; et al. GFR Estimation Using a Panel of Filtration Markers in Shanghai and Beijing. Kidney Med. 2020, 2, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Donadio, C.; Bozzoli, L. Urinary beta-trace protein: A unique biomarker to screen early glomerular filtration rate impairment. Medicine 2016, 95, e5553. [Google Scholar] [CrossRef] [PubMed]
- Ewadh, M. Beta trace protein level as a better diagnostic marker of renal impairment in patients with chronic kidney disease, diabetes mellitus, and renal transplants. J. Pharm. Sci. Res. 2018, 10, 1615–1618. [Google Scholar]
- Motawi, T.K.; Shehata, N.I.; ElNokeety, M.M.; El-Emady, Y.F. Potential serum biomarkers for early detection of diabetic nephropathy. Diabetes Res. Clin. Pract. 2018, 136, 150–158. [Google Scholar] [CrossRef]
- Dajak, M.; Ignjatovic, S.; Stojimirovic, B.; Gajic, S.; Majkic-Singh, N. Evaluation of renal damage by urinary beta-trace protein in patients with chronic kidney disease. Clin. Lab. 2011, 57, 29–36. [Google Scholar]
- Drüeke, T.B.; Massy, Z.A. Beta2-microglobulin. Semin. Dial. 2009, 22, 378–380. [Google Scholar] [CrossRef]
- Stefanović, V.; Djukanović, L.; Cukuranović, R.; Bukvić, D.; Ležaić, V.; Marić, I.; Ogrizovic, S.S.; Jovanović, I.; Vlahovic, P.; Pešić, I.; et al. Beta2-microglobulin and alpha1-microglobulin as markers of Balkan endemic nephropathy, a worldwide disease. Ren. Fail. 2011, 33, 176–183. [Google Scholar] [CrossRef]
- Foster, M.C.; Inker, L.A.; Hsu, C.Y.; Eckfeldt, J.H.; Levey, A.S.; Pavkov, M.E.; Myers, B.D.; Bennett, P.H.; Kimmel, P.L.; Vasan, R.S.; et al. Filtration markers as predictors of ESRD and mortality in Southwestern American Indians with type 2 diabetes. Am. J. Kidney Dis. 2015, 66, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Rebholz, C.M.; Inker, L.A.; Chen, Y.; Liang, M.; Foster, M.C.; Eckfeldt, J.H.; Kimmel, P.L.; Vasan, R.S.; Feldman, H.I.; Sarnak, M.J.; et al. Risk of ESRD and Mortality Associated with Change in Filtration Markers. Am. J. Kidney Dis. 2017, 70, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Rebholz, C.M.; Grams, M.E.; Matsushita, K.; Selvin, E.; Coresh, J. Change in novel filtration markers and risk of ESRD. Am. J. Kidney Dis. 2015, 66, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Pottel, H.; Schaeffner, E.; Ebert, N. Evaluating the diagnostic value of rescaled beta-trace protein in combination with serum creatinine and serum cystatin C in older adults. Clin. Chim. Acta 2018, 480, 206–213. [Google Scholar] [CrossRef] [PubMed]
- White, C.A.; Allen, C.M.; Akbari, A.; Collier, C.P.; Holland, D.C.; Day, A.G.; Knoll, G.A. Comparison of the new and traditional CKD-EPI GFR estimation equations with urinary inulin clearance: A study of equation performance. Clin. Chim. Acta 2019, 488, 189–195. [Google Scholar] [CrossRef]
- Inker, L.A.; Coresh, J.; Sang, Y.; Hsu, C.Y.; Foster, M.C.; Eckfeldt, J.H.; Karger, A.B.; Nelson, R.G.; Liu, X.; Sarnak, M.; et al. Filtration Markers as Predictors of ESRD and Mortality: Individual Participant Data Meta-Analysis. Clin. J. Am. Soc. Nephrol. 2017, 12, 69–78. [Google Scholar] [CrossRef]
- Buchanan, S.; Combet, E.; Stenvinkel, P.; Shiels, P.G. Klotho, Aging, and the Failing Kidney. Front. Endocrinol. 2020, 11, 560. [Google Scholar] [CrossRef]
- Hu, M.-C.; Kuro-o, M.; Moe, O.W. Klotho and kidney disease. J. Nephrol. 2010, 23 (Suppl. 16), S136–S144. [Google Scholar]
- Zou, D.; Wu, W.; He, Y.; Ma, S.; Gao, J. The role of klotho in chronic kidney disease. BMC Nephrol. 2018, 19, 285. [Google Scholar] [CrossRef]
- Qian, J.; Zhong, J.; Yan, M.; Cheng, P.; Shi, H.; Hao, C.; Gu, Y.; Lai, L. Circulating α-Klotho is Related to Plasma Aldosterone and Its Follow-Up Change Predicts CKD Progression. Kidney Blood Press. Res. 2018, 43, 836–846. [Google Scholar] [CrossRef]
- Wang, Q.; Su, W.; Shen, Z.; Wang, R. Correlation between Soluble α-Klotho and Renal Function in Patients with Chronic Kidney Disease: A Review and Meta-Analysis. Biomed. Res. Int. 2018, 2018, 9481475. [Google Scholar] [CrossRef] [PubMed]
- Inci, A.; Sari, F.; Coban, M.; Olmaz, R.; Dolu, S.; Sarıkaya, M.; Yılmaz, N. Soluble Klotho and fibroblast growth factor 23 levels in diabetic nephropathy with different stages of albuminuria. J. Investig. Med. 2016, 64, 1128–1133. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.P.; Mendes, F.; Carias, E.; Gonçalves, R.B.; Fragoso, A.; Dias, C.; Tavares, N.; Café, H.M.; Santos, N.; Rato, F.; et al. Plasmatic Klotho and FGF23 Levels as Biomarkers of CKD-Associated Cardiac Disease in Type 2 Diabetic Patients. Int. J. Mol. Sci. 2019, 20, 1536. [Google Scholar] [CrossRef] [PubMed]
- Memmos, E.; Sarafidis, P.; Pateinakis, P.; Tsiantoulas, A.; Faitatzidou, D.; Giamalis, P.; Vasilikos, V.; Papagianni, A. Soluble Klotho is associated with mortality and cardiovascular events in hemodialysis. BMC Nephrol. 2019, 20, 217. [Google Scholar] [CrossRef]
- He, T.; Xiong, J.; Huang, Y.; Zheng, C.; Liu, Y.; Bi, X.; Liu, C.; Han, W.; Yang, K.; Xiao, T.; et al. Klotho restrain RIG-1/NF-κB signaling activation and monocyte inflammatory factor release under uremic condition. Life Sci. 2019, 231, 116570. [Google Scholar] [CrossRef]
- Zheng, S.; Chen, Y.; Zheng, Y.; Zhou, Z.; Li, Z. Correlation of serum levels of fibroblast growth factor 23 and Klotho protein levels with bone mineral density in maintenance hemodialysis patients. Eur. J. Med. Res. 2018, 23, 18. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, H.; Chen, X.; Chen, H.; Wang, Y.; Wang, T.; Cai, L.; Hong, Y.; Ke, H.; Zheng, J. Relationship between cFGF23/Klotho ratio and phosphate levels in patients with chronic kidney disease. Int. Urol. Nephrol. 2019, 51, 503–507. [Google Scholar] [CrossRef]
- Hong, Y.A.; Lim, J.H.; Kim, M.Y.; Kim, Y.; Yang, K.S.; Chung, B.H.; Chung, S.; Choi, B.S.; Yang, C.W.; Kim, Y.S.; et al. Assessment of tubular reabsorption of phosphate as a surrogate marker for phosphate regulation in chronic kidney disease. Clin. Exp. Nephrol. 2015, 19, 208–215. [Google Scholar] [CrossRef]
- Foster, M.C.; Coresh, J.; Hsu, C.Y.; Xie, D.; Levey, A.S.; Nelson, R.G.; Eckfeldt, J.H.; Vasan, R.S.; Kimmel, P.L.; Schelling, J.; et al. Serum beta-Trace Protein and beta2-Microglobulin as Predictors of ESRD, Mortality, and Cardiovascular Disease in Adults With CKD in the Chronic Renal Insufficiency Cohort (CRIC) Study. Am. J. Kidney Dis. 2016, 68, 68–76. [Google Scholar] [CrossRef]
- Drew, D.A.; Katz, R.; Kritchevsky, S.; Ix, J.; Shlipak, M.; Gutiérrez, O.M.; Newman, A.; Hoofnagle, A.; Fried, L.; Semba, R.D.; et al. Association between Soluble Klotho and Change in Kidney Function: The Health Aging and Body Composition Study. J. Am. Soc. Nephrol. 2017, 28, 1859–1866. [Google Scholar] [CrossRef]
- Ebert, N.; Koep, C.; Schwarz, K.; Martus, P.; Mielke, N.; Bartel, J.; Kuhlmann, M.; Gaedeke, J.; Toelle, M.; van der Giet, M.; et al. Beta Trace Protein does not outperform Creatinine and Cystatin C in estimating Glomerular Filtration Rate in Older Adults. Sci. Rep. 2017, 7, 12656. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, N.; Asgari, M.; Khalessi, N.; Hoseini, R.; Khosravi, N. Serum Beta-Trace Protein for Assessment of Kidney Function in Neonates. Iran. J. Kidney Dis. 2018, 12, 11–13. [Google Scholar] [PubMed]
- Liu, Q.F.; Yu, L.X.; Feng, J.H.; Sun, Q.; Li, S.S.; Ye, J.M. The Prognostic Role of Klotho in Patients with Chronic Kidney Disease: A Systematic Review and Meta-analysis. Dis. Markers 2019, 2019, 6468729. [Google Scholar] [CrossRef] [PubMed]
- Yue, L.; Pan, B.; Shi, X.; Du, X. Comparison between the Beta-2 Microglobulin-Based Equation and the CKD-EPI Equation for Estimating GFR in CKD Patients in China: ES-CKD Study. Kidney Dis. 2020, 6, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Ro, H.; Chang, J.H.; Kim, A.J.; Lee, H.H.; Han, S.H.; Yoo, T.H.; Lee, K.B.; Kim, Y.H.; Kim, S.W.; et al. Mediation of the relationship between proteinuria and serum phosphate: Insight from the KNOW-CKD study. PLoS ONE 2020, 15, e0235077. [Google Scholar] [CrossRef]
- Devarajan, P. Neutrophil gelatinase-associated lipocalin (NGAL): A new marker of kidney disease. Scand J. Clin. Lab. Invest. Suppl. 2008, 241, 89–94. [Google Scholar] [CrossRef]
- Huo, W.; Zhang, K.; Nie, Z.; Li, Q.; Jin, F. Kidney injury molecule-1 (KIM-1): A novel kidney-specific injury molecule playing potential double-edged functions in kidney injury. Transplant. Rev. 2010, 24, 143–146. [Google Scholar] [CrossRef]
- Ning, M.; Mao, X.; Niu, Y.; Tang, B.; Shen, H. Usefulness and limitations of neutrophil gelatinase-associated lipocalin in the assessment of kidney diseases. J. Lab. Precis. Med. 2018, 3, 1. [Google Scholar] [CrossRef]
- Bolignano, D.; Donato, V.; Coppolino, G.; Campo, S.; Buemi, A.; Lacquaniti, A.; Buemi, M. Neutrophil gelatinase-associated lipocalin (NGAL) as a marker of kidney damage. Am. J. Kidney Dis. 2008, 52, 595–605. [Google Scholar] [CrossRef]
- Abbasi, F.; Moosaie, F.; Khaloo, P.; Dehghani Firouzabadi, F.; Fatemi Abhari, S.M.; Atainia, B.; Ardeshir, M.; Nakhjavani, M.; Esteghamati, A. Neutrophil Gelatinase-Associated Lipocalin and Retinol-Binding Protein-4 as Biomarkers for Diabetic Kidney Disease. Kidney Blood Press. Res. 2020, 45, 222–232. [Google Scholar] [CrossRef]
- Kapoula, G.V.; Kontou, P.I.; Bagos, P.G. Diagnostic Accuracy of Neutrophil Gelatinase-Associated Lipocalin for Predicting Early Diabetic Nephropathy in Patients with Type 1 and Type 2 Diabetes Mellitus: A Systematic Review and Meta-analysis. J. Appl. Lab. Med. 2019, 4, 78–94. [Google Scholar] [CrossRef] [PubMed]
- Sabbisetti, V.S.; Waikar, S.S.; Antoine, D.J.; Smiles, A.; Wang, C.; Ravisankar, A.; Ito, K.; Sharma, S.; Ramadesikan, S.; Lee, M.; et al. Blood kidney injury molecule-1 is a biomarker of acute and chronic kidney injury and predicts progression to ESRD in type I diabetes. J. Am. Soc. Nephrol. 2014, 25, 2177–2186. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.R.; Craven, T.E.; Malhotra, R.; Cheung, A.K.; Chonchol, M.; Drawz, P.; Sarnak, M.J.; Parikh, C.R.; Shlipak, M.G.; Ix, J.H. Kidney Damage Biomarkers and Incident Chronic Kidney Disease During Blood Pressure Reduction: A Case-Control Study. Ann. Intern. Med. 2018, 169, 610–618. [Google Scholar] [CrossRef] [PubMed]
- Żyłka, A.; Dumnicka, P.; Kuśnierz-Cabala, B.; Gala-Błądzińska, A.; Ceranowicz, P.; Kucharz, J.; Ząbek-Adamska, A.; Maziarz, B.; Drożdż, R.; Kuźniewski, M. Markers of Glomerular and Tubular Damage in the Early Stage of Kidney Disease in Type 2 Diabetic Patients. Mediat. Inflamm. 2018, 2018, 7659243. [Google Scholar] [CrossRef]
- Bjornstad, P.; Pyle, L.; Cherney, D.Z.I.; Johnson, R.J.; Sippl, R.; Wong, R.; Rewers, M.; Snell-Bergeon, J.K. Plasma biomarkers improve prediction of diabetic kidney disease in adults with type 1 diabetes over a 12-year follow-up: CACTI study. Nephrol. Dial. Transplant. 2018, 33, 1189–1196. [Google Scholar] [CrossRef]
- Skálová, S. The diagnostic role of urinary N-acetyl-beta-D-glucosaminidase (NAG) activity in the detection of renal tubular impairment. Acta Med. 2005, 48, 75–80. [Google Scholar]
- Jungbauer, C.G.; Uecer, E.; Stadler, S.; Birner, C.; Buchner, S.; Maier, L.S.; Luchner, A. N-acteyl-ss-D-glucosaminidase and kidney injury molecule-1: New predictors for long-term progression of chronic kidney disease in patients with heart failure. Nephrology 2016, 21, 490–498. [Google Scholar] [CrossRef]
- Lobato, G.R.; Lobato, M.R.; Thome, F.S.; Veronese, F.V. Performance of urinary kidney injury molecule-1, neutrophil gelatinase-associated lipocalin, and N-acetyl-beta-D-glucosaminidase to predict chronic kidney disease progression and adverse outcomes. Braz. J. Med. Biol. Res. 2017, 50, e6106. [Google Scholar] [CrossRef]
- Xu, Y.; Xie, Y.; Shao, X.; Ni, Z.; Mou, S. L-FABP: A novel biomarker of kidney disease. Clin. Chim. Acta 2015, 445, 85–90. [Google Scholar] [CrossRef]
- Negishi, K.; Noiri, E.; Doi, K.; Maeda-Mamiya, R.; Sugaya, T.; Portilla, D.; Fujita, T. Monitoring of Urinary L-Type Fatty Acid-Binding Protein Predicts Histological Severity of Acute Kidney Injury. Am. J. Pathol. 2009, 174, 1154–1159. [Google Scholar] [CrossRef]
- Suzuki, G.; Ichibayashi, R.; Yamamoto, S.; Nakamichi, Y.; Watanabe, M.; Honda, M. Clinical significance of urinary L-FABP in the emergency department. Int. J. Emerg. Med. 2019, 12, 24. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, D.; Kamijo-Ikemori, A.; Sugaya, T.; Ohata, K.; Hisamichi, M.; Hoshino, S.; Kimura, K.; Shibagaki, Y. Utility of urinary tubular markers for monitoring chronic tubulointerstitial injury after ischemia-reperfusion. Nephrology 2018, 23, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Khatir, D.S.; Bendtsen, M.D.; Birn, H.; Norregaard, R.; Ivarsen, P.; Jespersen, B.; Buus, N.H. Urine liver fatty acid binding protein and chronic kidney disease progression. Scand. J. Clin. Lab. Invest. 2017, 77, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Matsui, K.; Kamijo-Ikemori, A.; Imai, N.; Sugaya, T.; Yasuda, T.; Tatsunami, S.; Toyama, T.; Shimizu, M.; Furuichi, K.; Wada, T.; et al. Clinical significance of urinary liver-type fatty acid-binding protein as a predictor of ESRD and CVD in patients with CKD. Clin. Exp. Nephrol. 2016, 20, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Suzuki, A.; Ishii, J.; Sekiguchi-Ueda, S.; Shibata, M.; Yoshino, Y.; Asano, S.; Hayakawa, N.; Nakamura, K.; Akiyama, Y.; et al. Level of urinary liver-type fatty acid-binding protein is associated with cardiac markers and electrocardiographic abnormalities in type-2 diabetes with chronic kidney disease stage G1 and G2. Heart Vessel. 2015, 30, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Devuyst, O.; Olinger, E.; Rampoldi, L. Uromodulin: From physiology to rare and complex kidney disorders. Nat. Rev. Nephrol. 2017, 13, 525–544. [Google Scholar] [CrossRef]
- Fedak, D.; Kuźniewski, M.; Fugiel, A.; Wieczorek-Surdacka, E.; Przepiórkowska-Hoyer, B.; Jasik, P.; Miarka, P.; Dumnicka, P.; Kapusta, M.; Solnica, B.; et al. Serum uromodulin concentrations correlate with glomerular filtration rate in patients with chronic kidney disease. Pol. Arch. Med. Wewn. 2016, 126, 995–1004. [Google Scholar] [CrossRef]
- Steubl, D.; Block, M.; Herbst, V.; Nockher, W.A.; Schlumberger, W.; Satanovskij, R.; Angermann, S.; Hasenau, A.L.; Stecher, L.; Heemann, U.; et al. Plasma Uromodulin Correlates With Kidney Function and Identifies Early Stages in Chronic Kidney Disease Patients. Medicine 2016, 95, e3011. [Google Scholar] [CrossRef]
- Scherberich, J.E.; Gruber, R.; Nockher, W.A.; Christensen, E.I.; Schmitt, H.; Herbst, V.; Block, M.; Kaden, J.; Schlumberger, W. Serum uromodulin-a marker of kidney function and renal parenchymal integrity. Nephrol. Dial. Transplant. 2018, 33, 284–295. [Google Scholar] [CrossRef]
- Lim, W.H.; Lewis, J.R.; Wong, G.; Teo, R.; Lim, E.M.; Byrnes, E.; Prince, R.L. Plasma neutrophil gelatinase-associated lipocalin and kidney function decline and kidney disease-related clinical events in older women. Am. J. Nephrol. 2015, 41, 156–164. [Google Scholar] [CrossRef]
- Lv, L.; Wang, J.; Gao, B.; Wu, L.; Wang, F.; Cui, Z.; He, K.; Zhang, L.; Chen, M.; Zhao, M.H. Serum uromodulin and progression of kidney disease in patients with chronic kidney disease. J. Transl. Med. 2018, 16, 316. [Google Scholar] [CrossRef] [PubMed]
- Seibert, F.S.; Sitz, M.; Passfall, J.; Haesner, M.; Laschinski, P.; Buhl, M.; Bauer, F.; Babel, N.; Pagonas, N.; Westhoff, T.H. Prognostic Value of Urinary Calprotectin, NGAL and KIM-1 in Chronic Kidney Disease. Kidney Blood Press. Res. 2018, 43, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Nie, L.M.; Pang, Y.; Wu, W.J.; Tan, Y.; Yu, F.; Zhao, M.H. Composite urinary biomarkers to predict pathological tubulointerstitial lesions in lupus nephritis. Lupus 2018, 27, 1778–1789. [Google Scholar] [CrossRef] [PubMed]
- Steubl, D.; Block, M.; Herbst, V.; Nockher, W.A.; Schlumberger, W.; Kemmner, S.; Bachmann, Q.; Angermann, S.; Wen, M.; Heemann, U.; et al. Urinary uromodulin independently predicts end-stage renal disease and rapid kidney function decline in a cohort of chronic kidney disease patients. Medicine 2019, 98, e15808. [Google Scholar] [CrossRef] [PubMed]
- Steubl, D.; Buzkova, P.; Garimella, P.S.; Ix, J.H.; Devarajan, P.; Bennett, M.R.; Chaves, P.H.M.; Shlipak, M.G.; Bansal, N.; Sarnak, M.J. Association of Serum Uromodulin With ESKD and Kidney Function Decline in the Elderly: The Cardiovascular Health Study. Am. J. Kidney Dis. 2019, 74, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Plesiński, K.; Adamczyk, P.; Świętochowska, E.; Morawiec-Knysak, A.; Gliwińska, A.; Korlacki, W.; Szczepańska, M. Evaluation of liver-type fatty acid binding protein (L-FABP) and interleukin 6 in children with renal cysts. Adv. Clin. Exp. Med. 2019, 28, 1675–1682. [Google Scholar] [CrossRef] [PubMed]
- An, C.; Akankwasa, G.; Liu, J.; Wang, D.; Cheng, G.; Zhang, J.; Qin, X. Urine markers of renal tubular injury in idiopathic membranous nephropathy: A cross sectional study. Clin. Chim. Acta 2019, 492, 7–11. [Google Scholar] [CrossRef]
- Li, A.; Yi, B.; Liu, Y.; Wang, J.; Dai, Q.; Huang, Y.; Li, Y.C.; Zhang, H. Urinary NGAL and RBP Are Biomarkers of Normoalbuminuric Renal Insufficiency in Type 2 Diabetes Mellitus. J. Immunol. Res. 2019, 2019, 5063089. [Google Scholar] [CrossRef]
- Garimella, P.S.; Lee, A.K.; Ambrosius, W.T.; Bhatt, U.; Cheung, A.K.; Chonchol, M.; Craven, T.; Hawfield, A.T.; Jotwani, V.; Killeen, A.; et al. Markers of kidney tubule function and risk of cardiovascular disease events and mortality in the SPRINT trial. Eur. Heart J. 2019, 40, 3486–3493. [Google Scholar] [CrossRef]
- Zhang, D.; Ye, S.; Pan, T. The role of serum and urinary biomarkers in the diagnosis of early diabetic nephropathy in patients with type 2 diabetes. PeerJ 2019, 7, e7079. [Google Scholar] [CrossRef]
- Schulz, C.A.; Engström, G.; Nilsson, J.; Almgren, P.; Petkovic, M.; Christensson, A.; Nilsson, P.M.; Melander, O.; Orho-Melander, M. Plasma kidney injury molecule-1 (p-KIM-1) levels and deterioration of kidney function over 16 years. Nephrol. Dial. Transplant. 2020, 35, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Malyszko, J. Mechanism of endothelial dysfunction in chronic kidney disease. Clin. Chim. Acta 2010, 411, 1412–1420. [Google Scholar] [CrossRef] [PubMed]
- Quoc Hoang, T.A.; Tam, V.; Thang, H.V. Plasma asymmetric dimethylarginine and its association with some of cardiovascular disease risk factors in chronic kidney disease. Med. J. Malays. 2019, 74, 209–214. [Google Scholar]
- Triches, C.B.; Quinto, M.; Mayer, S.; Batista, M.; Zanella, M.T. Relation of asymmetrical dimethylarginine levels with renal outcomes in hypertensive patients with and without type 2 diabetes mellitus. J. Diabetes Complicat. 2018, 32, 316–320. [Google Scholar] [CrossRef]
- Ashokachakkaravarthy, K.; Rajappa, M.; Parameswaran, S.; Satheesh, S.; Priyadarshini, G.; Mohan Raj, P.S.; Revathy, G.; Priyadarssini, M. Asymmetric dimethylarginine and angiopoietin-like protein-2 are independent predictors of cardiovascular risk in pre-dialysis non-diabetic chronic kidney disease patients. Int. Urol. Nephrol. 2020. [Google Scholar] [CrossRef]
- Martens, C.R.; Kirkman, D.L.; Edwards, D.G. The Vascular Endothelium in Chronic Kidney Disease: A Novel Target for Aerobic Exercise. Exerc. Sport Sci. Rev. 2016, 44, 12–19. [Google Scholar] [CrossRef]
- Moe, S.M.; Chen, N.X. Mechanisms of vascular calcification in chronic kidney disease. J. Am. Soc. Nephrol. 2008, 19, 213–216. [Google Scholar] [CrossRef]
- Oliva-Damaso, E.; Oliva-Damaso, N.; Rodriguez-Esparragon, F.; Payan, J.; Baamonde-Laborda, E.; Gonzalez-Cabrera, F.; Santana-Estupiñan, R.; Rodriguez-Perez, J.C. Asymmetric (ADMA) and Symmetric (SDMA) Dimethylarginines in Chronic Kidney Disease: A Clinical Approach. Int. J. Mol. Sci. 2019, 20, 3668. [Google Scholar] [CrossRef]
- Schlesinger, S.; Sonntag, S.R.; Lieb, W.; Maas, R. Asymmetric and Symmetric Dimethylarginine as Risk Markers for Total Mortality and Cardiovascular Outcomes: A Systematic Review and Meta-Analysis of Prospective Studies. PLoS ONE 2016, 11, e0165811. [Google Scholar] [CrossRef]
- Choi, H.R.; Lee, S.W.; Jeon, D.H.; Hur, N.W.; Youm, Y.; Kim, H.C. Association between estimated glomerular filtration rate (eGFR) and asymmetric dimethylarginine (ADMA) concentrations among the elderly in a rural community: A cross-sectional study. BMC Geriatr. 2019, 19, 370. [Google Scholar] [CrossRef]
- Wang, F.; Xiong, R.; Feng, S.; Lu, X.; Li, H.; Wang, S. Association of Circulating Levels of ADMA with Carotid Intima-Media Thickness in Patients with CKD: A Systematic Review and Meta-Analysis. Kidney Blood Press. Res. 2018, 43, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Bartnicki, P.; Kowalczyk, M.; Franczyk-Skóra, B.; Baj, Z.; Rysz, J. Evaluation of Endothelial (dys)Function, Left Ventricular Structure and Function in Patients with Chronic Kidney Disease. Curr. Vasc. Pharmacol. 2016, 14, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Seliger, S.L.; Salimi, S.; Pierre, V.; Giffuni, J.; Katzel, L.; Parsa, A. Microvascular endothelial dysfunction is associated with albuminuria and CKD in older adults. BMC Nephrol. 2016, 17, 82. [Google Scholar] [CrossRef] [PubMed]
- Caglar, K.; Yilmaz, M.I.; Saglam, M.; Cakir, E.; Kilic, S.; Sonmez, A.; Eyileten, T.; Yenicesu, M.; Oguz, Y.; Tasar, M.; et al. Serum fetuin-a concentration and endothelial dysfunction in chronic kidney disease. Nephron. Clin. Pract. 2008, 108, c233–c240. [Google Scholar] [CrossRef]
- Cozzolino, M.; Galassi, A.; Biondi, M.L.; Turri, O.; Papagni, S.; Mongelli, N.; Civita, L.; Gallieni, M.; Brancaccio, D. Serum fetuin-A levels link inflammation and cardiovascular calcification in hemodialysis patients. Am. J. Nephrol. 2006, 26, 423–429. [Google Scholar] [CrossRef]
- Mutluay, R.; Konca Değertekin, C.; Işıktaş Sayılar, E.; Derici, Ü.; Gültekin, S.; Gönen, S.; Arınsoy, S.T.; Sindel, M. Serum fetuin-A is associated with the components of MIAC(malnutrition, inflammation, atherosclerosis, calcification) syndrome in different stages of chronic kidney disease. Turk. J. Med. Sci. 2019, 49, 327–335. [Google Scholar] [CrossRef]
- Zhou, Z.; Ji, Y.; Ju, H.; Chen, H.; Sun, M. Circulating Fetuin-A and Risk of All-Cause Mortality in Patients with Chronic Kidney Disease: A Systematic Review and Meta-Analysis. Front. Physiol. 2019, 10, 966. [Google Scholar] [CrossRef]
- Ciaccio, M.; Bivona, G.; Di Sciacca, R.; Iatrino, R.; Di Natale, E.; Li Vecchi, M.; Bellia, C. Changes in serum fetuin-A and inflammatory markers levels in end-stage renal disease (ESRD): Effect of a single session haemodialysis. Clin. Chem. Lab. Med. 2008, 46, 212–214. [Google Scholar] [CrossRef]
- Wang, A.Y.; Woo, J.; Lam, C.W.; Wang, M.; Chan, I.H.; Gao, P.; Lui, S.F.; Li, P.K.; Sanderson, J.E. Associations of serum fetuin-A with malnutrition, inflammation, atherosclerosis and valvular calcification syndrome and outcome in peritoneal dialysis patients. Nephrol. Dial. Transplant. 2005, 20, 1676–1685. [Google Scholar] [CrossRef]
- Pérez-Sotelo, D.; Roca-Rivada, A.; Larrosa-García, M.; Castelao, C.; Baamonde, I.; Baltar, J.; Crujeiras, A.B.; Seoane, L.M.; Casanueva, F.F.; Pardo, M. Visceral and subcutaneous adipose tissue express and secrete functional alpha2hsglycoprotein (fetuin a) especially in obesity. Endocrine 2017, 55, 435–446. [Google Scholar] [CrossRef]
- Zha, Y.; Qian, Q. Protein Nutrition and Malnutrition in CKD and ESRD. Nutrients 2017, 9, 208. [Google Scholar] [CrossRef]
- Iorember, F.M. Malnutrition in Chronic Kidney Disease. Front. Pediatr. 2018, 6, 161. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hamm, L.L.; Mohler, E.R.; Hudaihed, A.; Arora, R.; Chen, C.S.; Liu, Y.; Browne, G.; Mills, K.T.; Kleinpeter, M.A.; et al. Interrelationship of Multiple Endothelial Dysfunction Biomarkers with Chronic Kidney Disease. PLoS ONE 2015, 10, e0132047. [Google Scholar] [CrossRef]
- Kanbay, M.; Afsar, B.; Siriopol, D.; Unal, H.U.; Karaman, M.; Saglam, M.; Eyileten, T.; Gezer, M.; Verim, S.; Oguz, Y.; et al. Relevance of uric acid and asymmetric dimethylarginine for modeling cardiovascular risk prediction in chronic kidney disease patients. Int. Urol. Nephrol. 2016, 48, 1129–1136. [Google Scholar] [CrossRef] [PubMed]
- Alderson, H.V.; Ritchie, J.P.; Middleton, R.; Larsson, A.; Larsson, T.E.; Kalra, P.A. FGF-23 and Osteoprotegerin but not Fetuin-A are associated with death and enhance risk prediction in non-dialysis chronic kidney disease stages 3-5. Nephrology 2016, 21, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Emrich, I.E.; Zawada, A.M.; Martens-Lobenhoffer, J.; Fliser, D.; Wagenpfeil, S.; Heine, G.H.; Bode-Böger, S.M. Symmetric dimethylarginine (SDMA) outperforms asymmetric dimethylarginine (ADMA) and other methylarginines as predictor of renal and cardiovascular outcome in non-dialysis chronic kidney disease. Clin. Res. Cardiol. 2018, 107, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Amdur, R.L.; Feldman, H.I.; Gupta, J.; Yang, W.; Kanetsky, P.; Shlipak, M.; Rahman, M.; Lash, J.P.; Townsend, R.R.; Ojo, A.; et al. Inflammation and Progression of CKD: The CRIC Study. Clin. J. Am. Soc. Nephrol. 2016, 11, 1546–1556. [Google Scholar] [CrossRef] [PubMed]
- Gupta, J.; Mitra, N.; Kanetsky, P.A.; Devaney, J.; Wing, M.R.; Reilly, M.; Shah, V.O.; Balakrishnan, V.S.; Guzman, N.J.; Girndt, M.; et al. Association between albuminuria, kidney function, and inflammatory biomarker profile in CKD in CRIC. Clin. J. Am. Soc. Nephrol. 2012, 7, 1938–1946. [Google Scholar] [CrossRef] [PubMed]
- Mihai, S.; Codrici, E.; Popescu, I.D.; Enciu, A.-M.; Albulescu, L.; Necula, L.G.; Mambet, C.; Anton, G.; Tanase, C. Inflammation-Related Mechanisms in Chronic Kidney Disease Prediction, Progression, and Outcome. J. Immunol. Res. 2018, 2018, 2180373. [Google Scholar] [CrossRef] [PubMed]
- Bash, L.D.; Erlinger, T.P.; Coresh, J.; Marsh-Manzi, J.; Folsom, A.R.; Astor, B.C. Inflammation, hemostasis, and the risk of kidney function decline in the Atherosclerosis Risk in Communities (ARIC) Study. Am. J. Kidney Dis. 2009, 53, 596–605. [Google Scholar] [CrossRef]
- Sun, J.; Axelsson, J.; Machowska, A.; Heimbürger, O.; Bárány, P.; Lindholm, B.; Lindström, K.; Stenvinkel, P.; Qureshi, A.R. Biomarkers of Cardiovascular Disease and Mortality Risk in Patients with Advanced CKD. Clin. J. Am. Soc. Nephrol. 2016, 11, 1163–1172. [Google Scholar] [CrossRef] [PubMed]
- Romanova, Y.; Laikov, A.; Markelova, M.; Khadiullina, R.; Makseev, A.; Hasanova, M.; Rizvanov, A.; Khaiboullina, S.; Salafutdinov, I. Proteomic Analysis of Human Serum from Patients with Chronic Kidney Disease. Biomolecules 2020, 10, 257. [Google Scholar] [CrossRef] [PubMed]
- Kamińska, J.; Stopiński, M.; Mucha, K.; Jędrzejczak, A.; Gołębiowski, M.; Niewczas, M.A.; Pączek, L.; Foroncewicz, B. IL 6 but not TNF is linked to coronary artery calcification in patients with chronic kidney disease. Cytokine 2019, 120, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Milas, O.; Gadalean, F.; Vlad, A.; Dumitrascu, V.; Velciov, S.; Gluhovschi, C.; Bob, F.; Popescu, R.; Ursoniu, S.; Jianu, D.C.; et al. Pro-inflammatory cytokines are associated with podocyte damage and proximal tubular dysfunction in the early stage of diabetic kidney disease in type 2 diabetes mellitus patients. J. Diabetes Complicat. 2020, 34, 107479. [Google Scholar] [CrossRef]
- Malhotra, R.; Katz, R.; Jotwani, V.; Ambrosius, W.T.; Raphael, K.L.; Haley, W.; Rastogi, A.; Cheung, A.K.; Freedman, B.I.; Punzi, H.; et al. Urine Markers of Kidney Tubule Cell Injury and Kidney Function Decline in SPRINT Trial Participants with CKD. Clin. J. Am. Soc. Nephrol. 2020, 15, 349–358. [Google Scholar] [CrossRef]
- Bae, E.; Cha, R.H.; Kim, Y.C.; An, J.N.; Kim, D.K.; Yoo, K.D.; Lee, S.M.; Kim, M.H.; Park, J.T.; Kang, S.W.; et al. Circulating TNF receptors predict cardiovascular disease in patients with chronic kidney disease. Medicine 2017, 96, e6666. [Google Scholar] [CrossRef]
- Murakoshi, M.; Gohda, T.; Suzuki, Y. Circulating Tumor Necrosis Factor Receptors: A Potential Biomarker for the Progression of Diabetic Kidney Disease. Int. J. Mol. Sci. 2020, 21, 1957. [Google Scholar] [CrossRef]
- Tong, M.; Carrero, J.J.; Qureshi, A.R.; Anderstam, B.; Heimbürger, O.; Bárány, P.; Axelsson, J.; Alvestrand, A.; Stenvinkel, P.; Lindholm, B.; et al. Plasma pentraxin 3 in patients with chronic kidney disease: Associations with renal function, protein-energy wasting, cardiovascular disease, and mortality. Clin. J. Am. Soc. Nephrol. 2007, 2, 889–897. [Google Scholar] [CrossRef]
- Speeckaert, M.M.; Speeckaert, R.; Carrero, J.J.; Vanholder, R.; Delanghe, J.R. Biology of human pentraxin 3 (PTX3) in acute and chronic kidney disease. J. Clin. Immunol. 2013, 33, 881–890. [Google Scholar] [CrossRef]
- Sjöberg, B.; Qureshi, A.R.; Heimbürger, O.; Stenvinkel, P.; Lind, L.; Larsson, A.; Bárány, P.; Ärnlöv, J. Association between levels of pentraxin 3 and incidence of chronic kidney disease in the elderly. J. Intern. Med. 2016, 279, 173–179. [Google Scholar] [CrossRef]
- Valente, M.J.; Rocha, S.; Coimbra, S.; Catarino, C.; Rocha-Pereira, P.; Bronze-da-Rocha, E.; Oliveira, J.G.; Madureira, J.; Fernandes, J.C.; do Sameiro-Faria, M.; et al. Long Pentraxin 3 as a Broader Biomarker for Multiple Risk Factors in End-Stage Renal Disease: Association with All-Cause Mortality. Mediat. Inflamm. 2019, 2019, 3295725. [Google Scholar] [CrossRef] [PubMed]
- Krzanowski, M.; Krzanowska, K.; Gajda, M.; Dumnicka, P.; Dziewierz, A.; Woziwodzka, K.; Litwin, J.A.; Sułowicz, W. Pentraxin 3 as a new indicator of cardiovascular-related death in patients with advanced chronic kidney disease. Pol. Arch. Intern. Med. 2017, 127, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Nair, V.; Robinson-Cohen, C.; Smith, M.R.; Bellovich, K.A.; Bhat, Z.Y.; Bobadilla, M.; Brosius, F.; de Boer, I.H.; Essioux, L.; Formentini, I.; et al. Growth Differentiation Factor-15 and Risk of CKD Progression. J. Am. Soc. Nephrol. 2017, 28, 2233–2240. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N.; Zelnick, L.; Shlipak, M.G.; Anderson, A.; Christenson, R.; Deo, R.; deFilippi, C.; Feldman, H.; Lash, J.; He, J.; et al. Cardiac and Stress Biomarkers and Chronic Kidney Disease Progression: The CRIC Study. Clin. Chem. 2019, 65, 1448–1457. [Google Scholar] [CrossRef]
- Tuegel, C.; Katz, R.; Alam, M.; Bhat, Z.; Bellovich, K.; de Boer, I.; Brosius, F.; Gadegbeku, C.; Gipson, D.; Hawkins, J.; et al. GDF-15, Galectin 3, Soluble ST2, and Risk of Mortality and Cardiovascular Events in CKD. Am. J. Kidney Dis. 2018, 72, 519–528. [Google Scholar] [CrossRef]
- Mirna, M.; Topf, A.; Wernly, B.; Rezar, R.; Paar, V.; Jung, C.; Salmhofer, H.; Kopp, K.; Hoppe, U.C.; Schulze, P.C.; et al. Novel Biomarkers in Patients with Chronic Kidney Disease: An Analysis of Patients Enrolled in the GCKD-Study. J. Clin. Med. 2020, 9, 886. [Google Scholar] [CrossRef]
- Ho, J.E.; Hwang, S.J.; Wollert, K.C.; Larson, M.G.; Cheng, S.; Kempf, T.; Vasan, R.S.; Januzzi, J.L.; Wang, T.J.; Fox, C.S. Biomarkers of cardiovascular stress and incident chronic kidney disease. Clin. Chem. 2013, 59, 1613–1620. [Google Scholar] [CrossRef]
- Celec, P.; Vlková, B.; Lauková, L.; Bábíčková, J.; Boor, P. Cell-free DNA: The role in pathophysiology and as a biomarker in kidney diseases. Expert Rev. Mol. Med. 2018, 20, e1. [Google Scholar] [CrossRef]
- Nuzzo, P.V.; Berchuck, J.E.; Korthauer, K.; Spisak, S.; Nassar, A.H.; Abou Alaiwi, S.; Chakravarthy, A.; Shen, S.Y.; Bakouny, Z.; Boccardo, F.; et al. Detection of renal cell carcinoma using plasma and urine cell-free DNA methylomes. Nat. Med. 2020. [Google Scholar] [CrossRef]
- Merkle, J.; Daka, A.; Deppe, A.C.; Wahlers, T.; Paunel-Görgülü, A. High levels of cell-free DNA accurately predict late acute kidney injury in patients after cardiac surgery. PLoS ONE 2019, 14, e0218548. [Google Scholar] [CrossRef]
- Coimbra, S.; Santos-Silva, A.; Costa, E.; Bronze-da-Rocha, E. DNA Damage in End-Stage Renal Disease Patients. Assessment by In Vitro Comet Assay and by Cell-Free DNA Quantification. In Genotoxicity—A Predictable Risk to Our Actual World; Larramendy, L.M., Soloneski, S., Eds.; Intech, Open Access Publisher: London, UK, 2017. [Google Scholar] [CrossRef]
- Chang, C.C.; Chiu, P.F.; Wu, C.L.; Kuo, C.L.; Huang, C.S.; Liu, C.S.; Huang, C.H. Urinary cell-free mitochondrial and nuclear deoxyribonucleic acid correlates with the prognosis of chronic kidney diseases. BMC Nephrol. 2019, 20, 391. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hu, R.; Luo, T.; Peng, C.; Gong, L.; Hu, J.; Yang, S.; Li, Q. Serum cell-free DNA and progression of diabetic kidney disease: A prospective study. BMJ Open Diabetes Res. Care 2020, 8. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.; Yang, J.Y.C.; Sarwal, R.D.; Sigdel, T.K.; Liberto, J.M.; Damm, I.; Louie, V.; Sigdel, S.; Livingstone, D.; Soh, K.; et al. A Novel Multi-Biomarker Assay for Non-Invasive Quantitative Monitoring of Kidney Injury. J. Clin. Med. 2019, 8, 499. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Shin, M.H.; Kim, H.N.; Lee, Y.H.; Choi, S.W.; Ahn, H.R.; Kweon, S.S. Relationship between plasma pentraxin 3 level and risk of chronic kidney disease in the Korean elderly: The Dong-gu study. Int. Urol. Nephrol. 2017, 49, 2027–2033. [Google Scholar] [CrossRef]
- Frimodt-Møller, M.; von Scholten, B.J.; Reinhard, H.; Jacobsen, P.K.; Hansen, T.W.; Persson, F.I.; Parving, H.H.; Rossing, P. Growth differentiation factor-15 and fibroblast growth factor-23 are associated with mortality in type 2 diabetes—An observational follow-up study. PLoS ONE 2018, 13, e0196634. [Google Scholar] [CrossRef]
- Anderson, C.E.; Hamm, L.L.; Batuman, G.; Kumbala, D.R.; Chen, C.S.; Kallu, S.G.; Siriki, R.; Gadde, S.; Kleinpeter, M.A.; Krane, N.K.; et al. The association of angiogenic factors and chronic kidney disease. BMC Nephrol. 2018, 19, 117. [Google Scholar] [CrossRef]
- Lee, J.; Choi, J.Y.; Kwon, Y.K.; Lee, D.; Jung, H.Y.; Ryu, H.M.; Cho, J.H.; Ryu, D.H.; Kim, Y.L.; Hwang, G.S. Changes in serum metabolites with the stage of chronic kidney disease: Comparison of diabetes and non-diabetes. Clin. Chim. Acta 2016, 459, 123–131. [Google Scholar] [CrossRef]
- McMahon, G.M.; Hwang, S.J.; Clish, C.B.; Tin, A.; Yang, Q.; Larson, M.G.; Rhee, E.P.; Li, M.; Levy, D.; O’Donnell, C.J.; et al. Urinary metabolites along with common and rare genetic variations are associated with incident chronic kidney disease. Kidney Int. 2017, 91, 1426–1435. [Google Scholar] [CrossRef]
- Guo, Y.; Yu, H.; Chen, D.; Zhao, Y.Y. Machine learning distilled metabolite biomarkers for early stage renal injury. Metabolomics 2019, 16, 4. [Google Scholar] [CrossRef]
- Lee, H.; Jang, H.B.; Yoo, M.G.; Park, S.I.; Lee, H.J. Amino Acid Metabolites Associated with Chronic Kidney Disease: An Eight-Year Follow-Up Korean Epidemiology Study. Biomedicines 2020, 8, 222. [Google Scholar] [CrossRef]
- Konje, V.C.; Rajendiran, T.M.; Bellovich, K.; Gadegbeku, C.A.; Gipson, D.S.; Afshinnia, F.; Mathew, A.V.; Michigan Kidney Translational Core CPROBE Investigator Group. Tryptophan levels associate with incident cardiovascular disease in chronic kidney disease. Clin. Kidney J. 2020. [Google Scholar] [CrossRef]
- Debnath, S.; Velagapudi, C.; Redus, L.; Thameem, F.; Kasinath, B.; Hura, C.E.; Lorenzo, C.; Abboud, H.E.; O’Connor, J.C. Tryptophan Metabolism in Patients With Chronic Kidney Disease Secondary to Type 2 Diabetes: Relationship to Inflammatory Markers. Int. J. Tryptophan Res. 2017, 10, 1178646917694600. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Karnovsky, A.; Afshinnia, F.; Wigginton, J.; Rader, D.J.; Natarajan, L.; Sharma, K.; Porter, A.C.; Rahman, M.; He, J.; et al. Differential network enrichment analysis reveals novel lipid pathways in chronic kidney disease. Bioinformatics 2019, 35, 3441–3452. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.-R.; Coresh, J.; Inker, L.A.; Levey, A.S.; Zheng, Z.; Rebholz, C.M.; Tin, A.; Appel, L.J.; Chen, J.; Sarnak, M.J.; et al. Serum metabolites are associated with all-cause mortality in chronic kidney disease. Kidney Int. 2018, 94, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Grams, M.E.; Tin, A.; Rebholz, C.M.; Shafi, T.; Köttgen, A.; Perrone, R.D.; Sarnak, M.J.; Inker, L.A.; Levey, A.S.; Coresh, J. Metabolomic Alterations Associated with Cause of CKD. Clin. J. Am. Soc. Nephrol. 2017, 12, 1787–1794. [Google Scholar] [CrossRef] [PubMed]
- Tavares, G.; Venturini, G.; Padilha, K.; Zatz, R.; Pereira, A.C.; Thadhani, R.I.; Rhee, E.P.; Titan, S.M.O. 1,5-Anhydroglucitol predicts CKD progression in macroalbuminuric diabetic kidney disease: Results from non-targeted metabolomics. Metabolomics 2018, 14, 39. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Coresh, J.; Tin, A.; Rebholz, C.M.; Appel, L.J.; Chen, J.; Vasan, R.S.; Anderson, A.H.; Feldman, H.I.; Kimmel, P.L.; et al. Serum Metabolomic Alterations Associated with Proteinuria in CKD. Clin. J. Am. Soc. Nephrol. 2019, 14, 342–353. [Google Scholar] [CrossRef]
- Hallan, S.; Afkarian, M.; Zelnick, L.R.; Kestenbaum, B.; Sharma, S.; Saito, R.; Darshi, M.; Barding, G.; Raftery, D.; Ju, W.; et al. Metabolomics and Gene Expression Analysis Reveal Down-regulation of the Citric Acid (TCA) Cycle in Non-diabetic CKD Patients. EBioMedicine 2017, 26, 68–77. [Google Scholar] [CrossRef]
- Hocher, B.; Adamski, J. Metabolomics for clinical use and research in chronic kidney disease. Nat. Rev. Nephrol. 2017, 13, 269–284. [Google Scholar] [CrossRef]
- Cañadas-Garre, M.; Anderson, K.; McGoldrick, J.; Maxwell, A.P.; McKnight, A.J. Proteomic and metabolomic approaches in the search for biomarkers in chronic kidney disease. J. Proteom. 2019, 193, 93–122. [Google Scholar] [CrossRef]
- Good, D.M.; Zürbig, P.; Argilés, A.; Bauer, H.W.; Behrens, G.; Coon, J.J.; Dakna, M.; Decramer, S.; Delles, C.; Dominiczak, A.F.; et al. Naturally occurring human urinary peptides for use in diagnosis of chronic kidney disease. Mol. Cell Proteom. 2010, 9, 2424–2437. [Google Scholar] [CrossRef] [PubMed]
- Pontillo, C.; Mischak, H. Urinary peptide-based classifier CKD273: Towards clinical application in chronic kidney disease. Clin. Kidney J. 2017, 10, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Tofte, N.; Lindhardt, M.; Adamova, K.; Bakker, S.J.L.; Beige, J.; Beulens, J.W.J.; Birkenfeld, A.L.; Currie, G.; Delles, C.; Dimos, I.; et al. Early detection of diabetic kidney disease by urinary proteomics and subsequent intervention with spironolactone to delay progression (PRIORITY): A prospective observational study and embedded randomised placebo-controlled trial. Lancet Diabetes Endocrinol. 2020, 8, 301–312. [Google Scholar] [CrossRef]
- Karu, N.; McKercher, C.; Nichols, D.S.; Davies, N.; Shellie, R.A.; Hilder, E.F.; Jose, M.D. Tryptophan metabolism, its relation to inflammation and stress markers and association with psychological and cognitive functioning: Tasmanian Chronic Kidney Disease pilot study. BMC Nephrol. 2016, 17, 171. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Yasuda, K.; Yamamoto, R.; Soga, T.; Rakugi, H.; Hayashi, T.; Isaka, Y. Identification of biomarkers for development of end-stage kidney disease in chronic kidney disease by metabolomic profiling. Sci. Rep. 2016, 6, 26138. [Google Scholar] [CrossRef]
- Rhee, E.P.; Clish, C.B.; Wenger, J.; Roy, J.; Elmariah, S.; Pierce, K.A.; Bullock, K.; Anderson, A.H.; Gerszten, R.E.; Feldman, H.I. Metabolomics of Chronic Kidney Disease Progression: A Case-Control Analysis in the Chronic Renal Insufficiency Cohort Study. Am. J. Nephrol. 2016, 43, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Sekula, P.; Goek, O.N.; Quaye, L.; Barrios, C.; Levey, A.S.; Römisch-Margl, W.; Menni, C.; Yet, I.; Gieger, C.; Inker, L.A.; et al. A Metabolome-Wide Association Study of Kidney Function and Disease in the General Population. J. Am. Soc. Nephrol. 2016, 27, 1175–1188. [Google Scholar] [CrossRef]
- Boelaert, J.; Lynen, F.; Glorieux, G.; Schepers, E.; Neirynck, N.; Vanholder, R. Metabolic profiling of human plasma and urine in chronic kidney disease by hydrophilic interaction liquid chromatography coupled with time-of-flight mass spectrometry: A pilot study. Anal. Bioanal. Chem. 2017, 409, 2201–2211. [Google Scholar] [CrossRef]
- Gil, R.B.; Ortiz, A.; Sanchez-Niño, M.D.; Markoska, K.; Schepers, E.; Vanholder, R.; Glorieux, G.; Schmitt-Kopplin, P.; Heinzmann, S.S. Increased urinary osmolyte excretion indicates chronic kidney disease severity and progression rate. Nephrol. Dial. Transplant. 2018, 33, 2156–2164. [Google Scholar] [CrossRef]
- Wang, F.; Sun, L.; Sun, Q.; Liang, L.; Gao, X.; Li, R.; Pan, A.; Li, H.; Deng, Y.; Hu, F.B.; et al. Associations of Plasma Amino Acid and Acylcarnitine Profiles with Incident Reduced Glomerular Filtration Rate. Clin. J. Am. Soc. Nephrol. 2018, 13, 560–568. [Google Scholar] [CrossRef]
- Kordalewska, M.; Macioszek, S.; Wawrzyniak, R.; Sikorska-Wiśniewska, M.; Śledziński, T.; Chmielewski, M.; Mika, A.; Markuszewski, M.J. Multiplatform metabolomics provides insight into the molecular basis of chronic kidney disease. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2019, 1117, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Nierenberg, J.L.; He, J.; Li, C.; Gu, X.; Shi, M.; Razavi, A.C.; Mi, X.; Li, S.; Bazzano, L.A.; Anderson, A.H.; et al. Novel associations between blood metabolites and kidney function among Bogalusa Heart Study and Multi-Ethnic Study of Atherosclerosis participants. Metabolomics 2019, 15, 149. [Google Scholar] [CrossRef] [PubMed]
- Titan, S.M.; Venturini, G.; Padilha, K.; Goulart, A.C.; Lotufo, P.A.; Bensenor, I.J.; Krieger, J.E.; Thadhani, R.I.; Rhee, E.P.; Pereira, A.C. Metabolomics biomarkers and the risk of overall mortality and ESRD in CKD: Results from the Progredir Cohort. PLoS ONE 2019, 14, e0213764. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.Y.; Lin, C.J.; Pan, H.C.; Lee, C.C.; Lu, S.C.; Hsieh, Y.T.; Huang, S.Y.; Huang, H.Y. Clinical association between the metabolite of healthy gut microbiota, 3-indolepropionic acid and chronic kidney disease. Clin. Nutr. 2019, 38, 2945–2948. [Google Scholar] [CrossRef]
- Dekker, S.E.I.; Verhoeven, A.; Soonawala, D.; Peters, D.J.M.; de Fijter, J.W.; Mayboroda, O.A. Urinary metabolites associate with the rate of kidney function decline in patients with autosomal dominant polycystic kidney disease. PLoS ONE 2020, 15, e0233213. [Google Scholar] [CrossRef]
- Levey, A.S.; Perrone, R.D.; Madias, N.E. Serum Creatinine and Renal Function. Annu. Rev. Med. 1988, 39, 465–490. [Google Scholar] [CrossRef]
- Ichii, O.; Horino, T. MicroRNAs associated with the development of kidney diseases in humans and animals. J. Toxicol. Pathol. 2018, 31, 23–34. [Google Scholar] [CrossRef]
- Metzinger-Le Meuth, V.; Burtey, S.; Maitrias, P.; Massy, Z.A.; Metzinger, L. microRNAs in the pathophysiology of CKD-MBD: Biomarkers and innovative drugs. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 337–345. [Google Scholar] [CrossRef]
- Shang, F.; Wang, S.C.; Hsu, C.Y.; Miao, Y.; Martin, M.; Yin, Y.; Wu, C.C.; Wang, Y.T.; Wu, G.; Chien, S.; et al. MicroRNA-92a Mediates Endothelial Dysfunction in CKD. J. Am. Soc. Nephrol. 2017, 28, 3251–3261. [Google Scholar] [CrossRef]
- Lv, W.; Fan, F.; Wang, Y.; Gonzalez-Fernandez, E.; Wang, C.; Yang, L.; Booz, G.W.; Roman, R.J. Therapeutic potential of microRNAs for the treatment of renal fibrosis and CKD. Physiol. Genom. 2018, 50, 20–34. [Google Scholar] [CrossRef]
- Peters, L.J.F.; Floege, J.; Biessen, E.A.L.; Jankowski, J.; van der Vorst, E.P.C. MicroRNAs in Chronic Kidney Disease: Four Candidates for Clinical Application. Int. J. Mol. Sci. 2020, 21, 6547. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.Y.; Ballard, S.; Batlle, D.; Bonventre, J.V.; Bottinger, E.P.; Feldman, H.I.; Klein, J.B.; Coresh, J.; Eckfeldt, J.H.; Inker, L.A.; et al. Cross-Disciplinary Biomarkers Research: Lessons Learned by the CKD Biomarkers Consortium. Clin. J. Am. Soc. Nephrol. 2015, 10, 894–902. [Google Scholar] [CrossRef] [PubMed]
| Year | Study Type | Study Population | Biomarker (s) | Study Outcomes | Reference |
|---|---|---|---|---|---|
| 2015 | Prospective cohort | 9703 participants from the ARIC | serum B2M | Greater than 30% decline in B2M may be less common, but appears to be more specific for ESRD than equivalent changes in eGFR based on serum creatinine | [24] |
| 2015 | Prospective cohort | 250 Pima Indians with T2D | serum BTP, B2M | BTP and to a lesser extent B2M were associated with ESRD; only higher serum concentrations of B2M were associated with increased mortality risk in this population | [22] |
| 2015 | Cross-sectional | 93 CKD patients at stages 1–5 | urinary klotho | Decreased tubular phosphate reabsorption was associated with decreased eGFR, but it was not associated with urinary klotho levels | [39] |
| 2016 | Cross-sectional | 355 CKD patients, classified in the different stages of CKD | urinary BTP | BTP is present in the urine of patients with normal GFR, and its urinary excretion progressively increases along with the reduction of GFR; clearance of BTP progressively increases with the reduction of GFR | [16] |
| 2016 | Cross-sectional | 109 CKD patients with T2D and 32 healthy controls | serum klotho | Serum klotho levels were significantly elevated in diabetic patients; klotho levels decreased with increasing albumin excretion | [33] |
| 2016 | Retrospective cohort | 3551 participants with CKD from MDRD, AASK and CRIC studies | serum BTP, B2M | BTP and B2M are less influenced by age, sex and race than creatinine and less influenced by race than cystatin C, but provide less accurate GFR estimates | [10] |
| 2016 | Prospective cohort | 3613 adults from the CRIC study | serum BTP, B2M | BTP and B2M were independent predictors of ESRD and all-cause mortality, but only B2M was an independent predictor cardiovascular events | [40] |
| 2017 | Prospective cohort | 2496 participants from the Health Aging and Body Composition study | serum klotho | Higher klotho levels were associated with lower odds of kidney function decline, but not with incident CKD | [41] |
| 2017 | Cross-sectional | 50 individuals with type 2 diabetes and 25 healthy controls | serum BTP | BTP level was significantly higher in T2Dwith the microalbuminuria group than T2DM with normoalbuminuria and control groups | [18] |
| 2017 | Cross-sectional | 566 individuals aged 70+ from the Berlin Initiative Study | serum BTP | Combination of creatinine, cystatin C and BTP showed the best prediction of GFR; single usage of BTP showed the worst prediction within models with only one biomarker | [42] |
| 2017 | Prospective cohort | 317 participants from MDRD and 373 from AASK | serum BTP, B2M | Declines in eGFR based on the average of four filtration markers (creatinine, cystatin C, BTP, and B2M) were consistently associated with progression to ESRD; only the decline in eGFR-BTP was significantly more strongly associated with ESRD risk | [23] |
| 2017 | Meta-analysis | 23,318 individuals from six different studies | serum BTP, B2M | eGFR-BTP, eGFR-B2M, and their average showed stronger risk associations with ESRD and all-cause mortality when compared with eGFRcr | [27] |
| 2017 | Cross-sectional | Elderly participants from the AGES-Kidney study (683) and the MESA-Kidney (273) | serum BTP, B2M | eGFR-cys, eGFR-B2M and eGFR-BTP had significantly less strong residual associations with age and sex than eGFRcr | [11] |
| 2018 | Cross-sectional | 125 maintenance hemodialysis patients | serum klotho | Klotho levels were associated with the degree of bone mineral density; osteoporosis groups presented lower levels than the normal bone mass group | [37] |
| 2018 | Prospective cohort | 112 adults with stages 1–5 CKD | serum klotho | Klotho levels were positively associated with baseline eGFR; reduction in klotho levels was associated with renal function decline | [31] |
| 2018 | Cross-sectional | 150 patients with CKD at stages 1–4 and 50 healthy controls | serum BTP | Increased BTP concentrations in CKD patients are highly significantly correlated with the concentrations of Cr and Cys; BTP had a higher value of correlation with mGFR | [17] |
| 2018 | Systematic review and meta-analysis | 9 publications, comprising 1457 CKD patients | serum klotho | There was a positive correlation between serum klotho levels and eGFR; no significant correlations were found between serum klotho levels and calcium and phosphorus circulating levels | [32] |
| 2018 | Cross-sectional | 566 individuals aged 70+ from the Berlin Initiative Study | serum BTP | The addition of BTP to serum creatinine-based eGFR equations does not result in the same improvement as the addition of Cys | [25] |
| 2018 | Cross-sectional | 50 healthy term neonates | serum BTP | BTP concentrations were positively associated with the concentrations of serum Cr level; inverse serum BTP is associated with estimated GFR level among neonates | [43] |
| 2019 | Prospective cohort | 86 adults with stable CKD | serum BTP, B2M | The addition of BTP/B2M eGFR to Cr/cysC eGFR equations did not improve GFR estimation | [26] |
| 2019 | Systematic Review and Meta-analysis | 8 cohort studies with 3586 participants | serum klotho | Klotho levels were positively correlated with the eGFR; lower klotho levels were significantly associated with an increased risk of poor kidney outcomes | [44] |
| 2019 | Prospective cohort | 107 diabetic patients with CKD at stages 2 and 3 | serum klotho | Lower levels of klotho were associated with cardiac pathological changes and higher CVD risk | [34] |
| 2019 | Prospective cohort | 79 CKD patients on hemodialysis | serum klotho | Lower klotho levels were associated with the risk of CVD, independently from factors associated with mineral bone disease | [35] |
| 2019 | Cross-sectional | 286 CKD patients at stages 2–5 | serum klotho | The serum levels of inflammatory markers were negatively associated with klotho levels | [36] |
| 2019 | Cross-sectional | 152 patients with CKD at stages 3–5 and 30 healthy controls | serum klotho | eGFR reduction was associated with decreased klotho levels; serum phosphate levels were negatively associated with klotho levels | [38] |
| 2020 | Cross-sectional | 1066 participants with Cr and Cys and 666 with all 4 markers | serum BTP, B2M | eGFR-B2M and eGFR-BTP were not more accurate than eGFR-cr and eGFR-cys; accuracy was significantly better for the eGFR equation considering the four markers when compared to eGFRcr-cys equation | [15] |
| 2020 | Prospective cohort | 830 Chinese CKD patients | serum B2M | The B2M equation had smaller bias in the subgroup of GFR 60–89 mL/min/1.73 m2, but a larger bias and worse precision and accuracy in the subgroup of GFR > 90 mL/min/1.73 m2 when compared to the CKD-EPI equation | [45] |
| 2020 | Cross-sectional | 1793 patients from the KNOW-CKD study | serum klotho | Decreased klotho levels correlated negatively with phosphate levels and with the degree of proteinuria | [46] |
| Year | Study Type | Study Population | Biomarker (s) | Study Outcomes | Reference |
|---|---|---|---|---|---|
| 2015 | Prospective cohort | 1245 women aged ≥ 70 from the general population | plasma NGAL | NGAL is of modest clinical utility in predicting renal function decline and acute or chronic renal disease-related events in individuals with mild-to-moderate CKD | [71] |
| 2015 | Cross-sectional | 276 type 2 diabetic patients with CKD at stage 1 and 2 | urinary L-FABP | Urinary L-FABP was significantly correlated with UACR in the early stages of CKD | [66] |
| 2015 | Prospective cohort | 138 patients with CHF | urinary KIM-1, NGAL, NAG | In patients with CKD progression, KIM-1 and NAG were elevated in contrast to NGAL; KIM-1 and NAG were negatively correlated with ejection fraction and eGFR | [58] |
| 2016 | Cross-sectional | 355 patients with CKD at stages 1–5 and 71 patients without CKD | plasma and urinary UMOD | UMOD allowed the identification of patients without CKD and patients at any stage of CKD; Plasma UMOD appears to outperform urinary uromodulin as a CKD marker | [69] |
| 2016 | Prospective cohort | 244 adult patients with CKD | urinary NAG, L-FABP | Elevated urinary L-FABP and low eGFR were associated with the development of ESRD and CVD, irrespective of diabetes | [65] |
| 2016 | Cross-sectional | 170 patients with CKD at stages 1 to 5 and 30 healthy individuals | serum UMOD | Serum UMOD concentrations in CKD patients were lower than in healthy subjects, and the lower concentrations were associated with more advanced stages | [68] |
| 2017 | Case–control | 74 adult CKD patients (stages 3–5) and 25 healthy subjects | urinary L-FABP | L-FABP shows a negative correlation with GFR and a positive correlation with UAC; in patients without albuminuria, L-FABP was associated with renal function decline | [64] |
| 2017 | Prospective cohort | 250 patients with CKD at stages 1–5, including 111 on hemodialysis | urinary KIM-1, NGAL, NAG | NGAL was moderately correlated with the 5 stages of CKD, while KIM-1 and NAG were also correlated, but weakly | [59] |
| 2018 | Cross-sectional | 80 patients with T2D without significant decrease in eGFR and albuminuria | urinary NGAL, KIM-1, UMOD | Urinary NGAL and KIM-1 correlated positively with albuminuria; all markers differed significantly between patients with moderately increased albuminuria compared to those with normal to mildly increased albuminuria | [55] |
| 2018 | Prospective cohort | 2813 patients from C-STRIDE study | serum UMOD | Higher incidence rates of ESRD, CVD and death were associated with decreased UMOD levels | [72] |
| 2018 | Cross-sectional | 132 patients with CKD at stages 1–5 and 33 patients without CKD | serum UMOD | UMOD levels inversely correlated with creatinine and creatinine/cystatin C-based eGFR | [70] |
| 2018 | Case–control | 324 participants from the SPRINT trial (162 who developed CKD during the follow-up and 162 matched controls) | urinary KIM-1, NGAL, UMOD | Only baseline concentrations of KIM-1 were associated with the development of incident CKD during the follow-up | [54] |
| 2018 | Prospective cohort | 527 adults with type 1 diabetes from the CACTI study | plasma KIM-1, NGAL, UMOD | NGAL and UMOD were associated with UACR and incident impaired GFR over the 12-year follow-up period | [56] |
| 2018 | Prospective cohort | 143 patients with stable CKD with diverse etiologies | urinary NGAL, KIM-1 | Neither NGAL nor KIM-1 provided robust prognostic information on the loss or renal function in a heterogeneous CKD population | [73] |
| 2018 | Cross-sectional | 109 biopsy-proven lupus nephritis patients and 50 healthy individuals | urinary NGAL, KIM-1 | Patients with active lupus nephritis exhibited elevated urinary levels of KIM-1 and NGAL compared with patients in remission and controls; KIM-1 levels correlated with tubular atrophy and interstitial inflammatory lesions | [74] |
| 2019 | Prospective cohort | 230 CKD patients stages 1 to 5 | urinary UMOD | UMOD concentrations were positively associated with eGFR and inversely associated with proteinuria; UMOD levels were independently associated with ESRD or rapid loss of eGFR | [75] |
| 2019 | Prospective cohort | 933 individuals aged ≥65 years from the CHS study | serum UMOD | Lower UMOD was associated with the development of ESRD independently of eGFR, UACR, and cardiovascular and CKD risk factors | [76] |
| 2019 | Cross-sectional | 39 children with kidney cysts, including 20 subjects with ADPKD, and 20 controls | urinary and serum L-FABP | Higher concentration of L-FABP in serum and urine indicated early damage to the renal parenchyma, detectable before the onset of hypertension and other organ damage | [77] |
| 2019 | Cross-sectional | 165 biopsy-proven CKD patients and 64 healthy controls | urinary NAG, KIM-1, NGAL | All biomarkers were significantly increased in patients, but their values were similar for patients with moderate and severe tubular injury | [78] |
| 2019 | Systematic review and meta-analysis | 22 studies involving 683 healthy individuals and 3249 diabetic patients | serum and urinary NGAL | Both urinary and serum NGAL showed an increasing trend, in parallel with albuminuria and progression of the disease, estimated by eGFR; the highest concentrations were achieved in patients with the highest severity of diabetic nephropathy | [52] |
| 2019 | Cross-sectional | 209 T2D normoalbuminuric patients with or without CKD | urinary NGAL | Levels of urinary NGAL were elevated in patients with renal insufficiency and negatively related to eGFR in T2D patients with normoalbuminuria | [79] |
| 2019 | Prospective cohort | 2377 participants from SPRINT trial with non-diabetic CKD | urinary UMOD | Lower uromodulin levels were associated with higher risk of CVD events and mortality, independently of eGFR, UACR, and other risk factors | [80] |
| 2019 | Cross-sectional | 287 T2D patients and 42 healthy controls | urinary NGAL | Urinary NGAL was significantly correlated with the UACR in patients with T2D | [81] |
| 2020 | Cross-sectional | 133 patients with diabetes and 39 healthy controls | urinary NGAL | Patients with severely increased albuminuria had higher levels of NGAL compared to patients with normal albuminuria and controls | [51] |
| 2020 | Prospective cohort | 4739 participants of the population-based Malmö Diet and Cancer Study | plasma KIM-1 | Plasma KIM-1 was able to predict the future decline of eGFR and the risk of CKD in healthy participants | [82] |
| Year | Study Type | Study Population | Biomarker (s) | Study Outcomes | Reference |
|---|---|---|---|---|---|
| 2015 | Cross-sectional | 201 patients with CKD and 201 controls | plasma ADMA | Plasma ADMA levels were associated with CKD severity measured by eGFR and/or albuminuria | [104] |
| 2016 | Prospective cohort | 259 patients with CKD at stages 1–5 | serum ADMA | Patients with ADMA levels above the median value had an increased risk of all-cause mortality and CVE | [105] |
| 2016 | Cross-sectional | 35 pre-dialysis CKD patients, 40 on hemodialysis, and 15 healthy subjects | plasma ADMA | Plasma ADMA concentration was associated with disadvantageous changes in left ventricular structure and function | [93] |
| 2016 | Prospective cohort | 463 individuals with CKD at stages 3–5 from the CRISIS Study | plasma fetuin-A | There was no clear association between fetuin-A and risk for RRT, CVE, and death | [106] |
| 2018 | Prospective cohort | 528 adult CKD patients at stages 2–4 | plasma ADMA | eGFR was inversely correlated with plasma levels of ADMA | [107] |
| 2018 | Systematic review and meta-analysis | 6 articles, involving 616 CKD patients | plasma ADMA | Levels of circulating ADMA were positively related to CIMT in CKD patients | [92] |
| 2018 | Prospective cohort | 162 hypertensive CKD patients, free from albuminuria | plasma ADMA | High ADMA levels were associated with the progression of albuminuria in hypertensive patients, with and without type 2 diabetes | [85] |
| 2019 | Cross-sectional | 651 elderly subjects from KSHAP cohort study | plasma ADMA | eGFR levels were inversely associated with ADMA concentrations | [91] |
| 2019 | Cross-sectional | 176 CKD patients and 64 control subjects | plasma ADMA | Plasma ADMA levels were similar in the control group and stage 1 CKD patients; in other stages, ADMA levels were significantly higher in comparison to the control subjects | [84] |
| 2019 | Systematic review and meta-analysis | 13 studies comprising 5169 CKD patients | serum fetuin-A | CKD patients with the lowest fetuin-A levels had a 92% greater risk of all-cause mortality compared with those with the highest levels | [98] |
| 2019 | Cross-sectional | 238 CKD patients (stages 3–5) | serum fetuin-A | Fetuin-A levels in ESRD patients were significantly lower than those from patients at stages 3 and 4 CKD; fetuin-A was negatively correlated with vascular calcification score and CIMT | [97] |
| 2020 | Cross-sectional | 43 adult patients with CKD and 43 healthy controls | plasma ADMA | Levels of ADMA positively correlate with CKD severity; FMD was significantly decreased in CKD patients, and negatively correlated with ADMA levels | [86] |
| Year | Study Type | Study Population | Biomarker (s) | Study Outcomes | Reference |
|---|---|---|---|---|---|
| 2016 | Prospective cohort | 746 individuals with GFR > 60mL/min/1.73 m2 | serum PTX3 | Higher PTX3 levels are associated with lower GFR and independently predict incident CKD in the elderly | [121] |
| 2016 | Prospective cohort | 3430 patients with reduced eGFR from the CRIC study | plasma IL-6, TNF-α | Elevated plasma levels of TNF-α were associated with rapid loss of kidney function in CKD patients | [108] |
| 2016 | Prospective cohort | 543 patients with stage 5 CKD | plasma IL-6, TNF- α | IL-6 and TNF-a could predict all-cause mortality risk; only IL-6 could classify clinical CVD | [112] |
| 2017 | Prospective cohort | 521 adults with CKD from the C-PROBE and the SKS studies | plasma or serum GDF-15 | Circulating GDF-15 levels were strongly correlated with intrarenal expression of GDF15 and significantly associated with increased risk of CKD progression | [124] |
| 2017 | Prospective cohort | 984 CKD patients stages 1–5 | serum TNFR1, TNFR2 | TNFR1 and 2 were associated with CVD and other risk factors in CKD, independently of eGFR | [117] |
| 2017 | Cross-sectional | 1816 community residents randomly selected from the Dong-gu study | plasma PTX3 | A significantly higher risk of CKD was found in the group with the highest plasma levels of PTX3 when compared to the group with the lowest levels | [136] |
| 2017 | Prospective cohort | 78 stage 5 CKD patients (51 on hemodialysis and 27 on pre-dialysis) | serum PTX3, IL-6, CRP | In contrast to CRP levels, baseline PTX3 levels predicted CV mortality independently of classic CV risk factors; PTX3 levels also significantly predicted mortality | [123] |
| 2017 | Prospective cohort | 39 ESRD patients under HD and 15 healthy controls | serum cfDNA | ESRD patients had a significantly higher value when compared to controls; cfDNA correlated positively with CRP levels in ESRD patients | [132] |
| 2018 | Prospective cohort | 883 adults at any stage CKD from the SKS or the C-PROBE studies | serum GDF-15 | Adults with CKD and higher circulating levels of GDF-15 presented greater mortality; elevated GDF-15 was also associated with an increased rate of HF | [126] |
| 2018 | Prospective cohort | 200 patients with T2D | plasma GDF-15 | Higher GDF-15 improved risk prediction of decline in kidney function; in patients with T2D and microalbuminuria, higher GDF-15 was independently associated with all-cause mortality | [137] |
| 2018 | Cross-sectional | 201 patients with CKD and 201 controls | plasma PTX3 | Plasma PTX3 levels were increased in patients with CKD when compared to controls | [138] |
| 2019 | Prospective cohort | 3664 participants with CKD from the CRIC study | plasma GDF-15 | GDF-15 was significantly associated with an increased risk of CKD progression | [125] |
| 2019 | Prospective cohort | 57 CKD patients at stages 3–5 and 19 healthy controls | serum IL-6, TNF- α | TNF and IL-6 were significantly higher in more advanced CKD stages; IL-6, but not TNF- α, was associated with 5-year risk of all-cause mortality in CKD patients | [114] |
| 2019 | Prospective cohort | 318 ESRD patients, undergoing HD and 22 healthy controls | plasma PTX3, IL-6, TNF- α, CRP | When comparing inflammatory mediators, the increase in PTX3 levels was the only predictor of all-cause mortality in dialysis patients | [122] |
| 2019 | Prospective cohort | 124 patients with CKD (stages 1–5) | plasma and urinary cfDNA | No correlations were found between cfDNA levels and CKD staging; higher urinary levels of cfDNA were associated with worse renal outcomes at 6 months | [133] |
| 2020 | Cross-sectional | 219 adult CKD patients (stages 2–5) from the GCKD study | plasma GDF-15 | GDF-15 was significantly elevated in CKD patients and showed a significant inverse correlation with eGFR | [127] |
| 2020 | Cross-sectional | 117 T2D patients and 11 healthy controls | serum and urinary IL-8, IL-18 | Serum and urinary levels of IL-8 and IL-18 were positively correlated with podocyte damage, peritubular dysfunction, and albuminuria, and negatively correlated with eGFR | [115] |
| 2020 | Prospective cohort | 2428 SPRINT participants with CKD | urinary IL-18 | Urinary IL-18 was associated with eGFR decline and may help to detect subtle changes in eGFR | [116] |
| 2020 | Prospective cohort | 160 patients with DN | serum cfDNA | Serum cfDNA levels were significantly negatively associated with the eGFR changes during the follow-up | [134] |
| Year | Study Type | Study Population | Study Outcomes | Reference |
|---|---|---|---|---|
| 2016 | Cross-sectional | 27 patients with CKD at stages 3–5 | Kidney function decline was associated with an increase in the inflammation marker neopterin and the metabolism of tryptophan via the kynurenine pathway | [156] |
| 2016 | Prospective cohort | 118 patients with CKD at stages 3–5 | Sixteen metabolites, from variable metabolic pathways, were related to higher risk of kidney function deterioration in advanced CKD patients | [157] |
| 2016 | Case–control | 200 patients with rapid renal disease progression and 200 stable controls | Ten metabolites were associated with CKD progression; six (uric acid, glucuronate, 4-hydroxymandelate, 3-methyladipate/pimelate, cytosine, and homogentisate) were higher in cases than controls, whereas four (threonine, methionine, phenylalanine, and arginine) were lower | [158] |
| 2016 | Prospective cohort | 1735 participants in the KORA F4 study | Six metabolites (N-acetylalanine, N-acetylcarnosine, C-mannosyltryptophan, erythronate, pseudouridine, and O-sulfo-L-tyrosine) were associated with eGFR and CKD in both studies and showed high correlation with established kidney function markers | [159] |
| 1164 individuals in the TwinsUK registry | ||||
| 2016 | Cross-sectional | 291 pre-dialysis CKD patients and 56 healthy controls | The presence of diabetes affects the metabolic phenotypes of CKD patients at an early stage, and those differences are attenuated with CKD progression | [139] |
| 2017 | Case–control | 193 patients with incident CKD from the Framingham Study and 193 matched controls | Lower urinary levels of glycine and histidine were associated with a higher risk of incident CKD; moreover, the authors identified several novel associations with urinary metabolites and genetic variations | [140] |
| 2017 | Cross-sectional | 60 T2D patients with all stages of CKD from the FIND study | Tryptophan levels were inversely correlated with CKD staging, while its metabolites were positively associated with the severity of kidney disease; kynurenine was positively correlated with TNF-a levels | [144] |
| 2017 | Cross-sectional | 589 CKD patients from the MDRD study | Five metabolite associations (kynurenate, homovanillate sulfate, hippurate, N2,N2-dimethylguanosine, and 16-hydroxypalmitate) showed consistently higher levels in ADPKD compared with glomerular disease and CKD of other causes | [147] |
| 2017 | Cross-sectional | 22 non-diabetic CKD stage 3–4 patients and 10 healthy controls | Urinary levels of 27 metabolites and plasma concentration of 33 metabolites differed significantly in CKD patients compared to controls; the citric acid cycle pathway was the most affected, with reduced urinary excretion of citrate, cis-aconitate, isocitrate, 2-oxoglutarate and succinate | [150] |
| 2017 | Cross-sectional | 20 CKD patients at stage 3 and 20 at stage 5, and 20 healthy controls | Glycoursodeoxycholic acid and 2-hydroxyethane sulfonate were downregulated in the urine of patients, and pregnenolone sulfate was also found to be decreased in plasma when compared to controls | [160] |
| 2018 | Prospective cohort | 56 Brazilian macroalbuminuric CKD patients | Lower levels of 1,5-AG, norvaline and l-aspartic acid were significantly associated with the risk of a combined outcome of mortality, dialysis need or creatinine doubling | [148] |
| 2018 | Retrospective cohort | 227 patients with CKD and a nested subgroup of 57 for follow up | Eleven metabolites from various metabolic pathways were associated with reduced eGFR; increased urinary concentrations of betaine and myo-inositol were found to be prognostic markers of CKD progression | [161] |
| 2018 | Prospective cohort | 299 CKD patients from the MDRD study and 963 from the AASK cohort | Serum metabolites fumarate, allantoin, and ribonate were associated with a higher risk of mortality in two cohorts of patients with CKD | [146] |
| 2018 | Prospective cohort | 1765 Chinese adults with eGFR ≥ 60 mL/min per 1.73 m2 | Elevated plasma levels of cysteine and several acylcarnitines were associated with eGFR reduction, independent of baseline eGFR and other conventional risk factors | [162] |
| 2019 | Cross-sectional | 587 adults with all stages of CKD and 116 healthy controls | Five serum metabolites (5-MTP, canavaninosuccinate, acetylcarnitine, tiglylcarnitine and taurine) were identified to estimate kidney filtration and enhance earlier CKD prediction | [141] |
| 2019 | Prospective cohort | 1582 participants from the AASK and MDRD studies | The serum metabolites 4-hydroxychlorthalonil and 1,5-AG and the phosphatidylethanolamine metabolic pathway were strongly associated with proteinuria in CKD | [149] |
| 2019 | Cross-sectional | 30 patients with CKD at stages 3 and 4 and 30 healthy volunteers | More significant changes in acylcarnitines, carbohydrates (such as glucose and myo-inositol), and glycerophospholipid metabolism pathways were found in CKD patients than in controls | [163] |
| 2019 | Retrospective cohort | 214 CKD patients from the CPROBE and 200 from the CRIC studies | In CKD patients, changes in the triacylglycerols and cardiolipins-phosphatidylethanolamines preceded the clinical outcomes of ESRD by several years | [145] |
| 2019 | Cross-sectional | 1243 participants from the BHS and 260 from the MESA studies | This study identified 39 novel metabolites in sub-pathways previously associated with kidney function, and 12 novel metabolites in sub-pathways with novel associations | [164] |
| 2019 | Retrospective cohort | 454 patients with CKD at stages 3 and 4 from the Progredir Cohort Study | D-malic acid, acetohydroxamic acid, butanoic acid, ribose, glutamine, trans-aconitic acid, lactose and an unidentified molecule (m/z 273) were positively related to the risk of overall mortality, while docosahexaenoic acid was inversely related to this risk; lactose, 2-O-glycerol-α-d-galactopyranoside, and tyrosine were associated with ESRD progression | [165] |
| 2019 | Cross-sectional | 140 CKD patients and 144 healthy subjects | CKD patients presented significantly lower serum levels of 3-indolepropionic acid and higher serum levels of indoxyl sulfate and p-cresol sulfate when compared to controls | [166] |
| 2020 | Prospective cohort | 1741 subjects from the Ansan-Ansung population study | Researchers found 22 metabolites associated with eGFR and CKD prevalence; citrulline, kynurenine, and the kynurenine/tryptophan ratio were associated with incident CKD | [142] |
| 2020 | Prospective cohort | 184 patients with CKD at stages 1–5 from the CPROBE study | Kynurenic acid, 3-hydroxykynurenine and kynurenine were increased with CKD stage progression; higher tryptophan levels at baseline were associated with lower odds of incident CVD | [143] |
| 2020 | Prospective cohort | 501 patients with ADPKD, with different stages of CKD | Four urinary metabolites (myo-inositol, 3-hydroxyisovalerate, ADMA and creatinine) were strongly associated with baseline eGFR; the urinary alanine/citrate ratio showed the best association with eGFR decline | [167] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lousa, I.; Reis, F.; Beirão, I.; Alves, R.; Belo, L.; Santos-Silva, A. New Potential Biomarkers for Chronic Kidney Disease Management—A Review of the Literature. Int. J. Mol. Sci. 2021, 22, 43. https://doi.org/10.3390/ijms22010043
Lousa I, Reis F, Beirão I, Alves R, Belo L, Santos-Silva A. New Potential Biomarkers for Chronic Kidney Disease Management—A Review of the Literature. International Journal of Molecular Sciences. 2021; 22(1):43. https://doi.org/10.3390/ijms22010043
Chicago/Turabian StyleLousa, Irina, Flávio Reis, Idalina Beirão, Rui Alves, Luís Belo, and Alice Santos-Silva. 2021. "New Potential Biomarkers for Chronic Kidney Disease Management—A Review of the Literature" International Journal of Molecular Sciences 22, no. 1: 43. https://doi.org/10.3390/ijms22010043
APA StyleLousa, I., Reis, F., Beirão, I., Alves, R., Belo, L., & Santos-Silva, A. (2021). New Potential Biomarkers for Chronic Kidney Disease Management—A Review of the Literature. International Journal of Molecular Sciences, 22(1), 43. https://doi.org/10.3390/ijms22010043








