Shiftwork and Light at Night Negatively Impact Molecular and Endocrine Timekeeping in the Female Reproductive Axis in Humans and Rodents
Abstract
1. Introduction
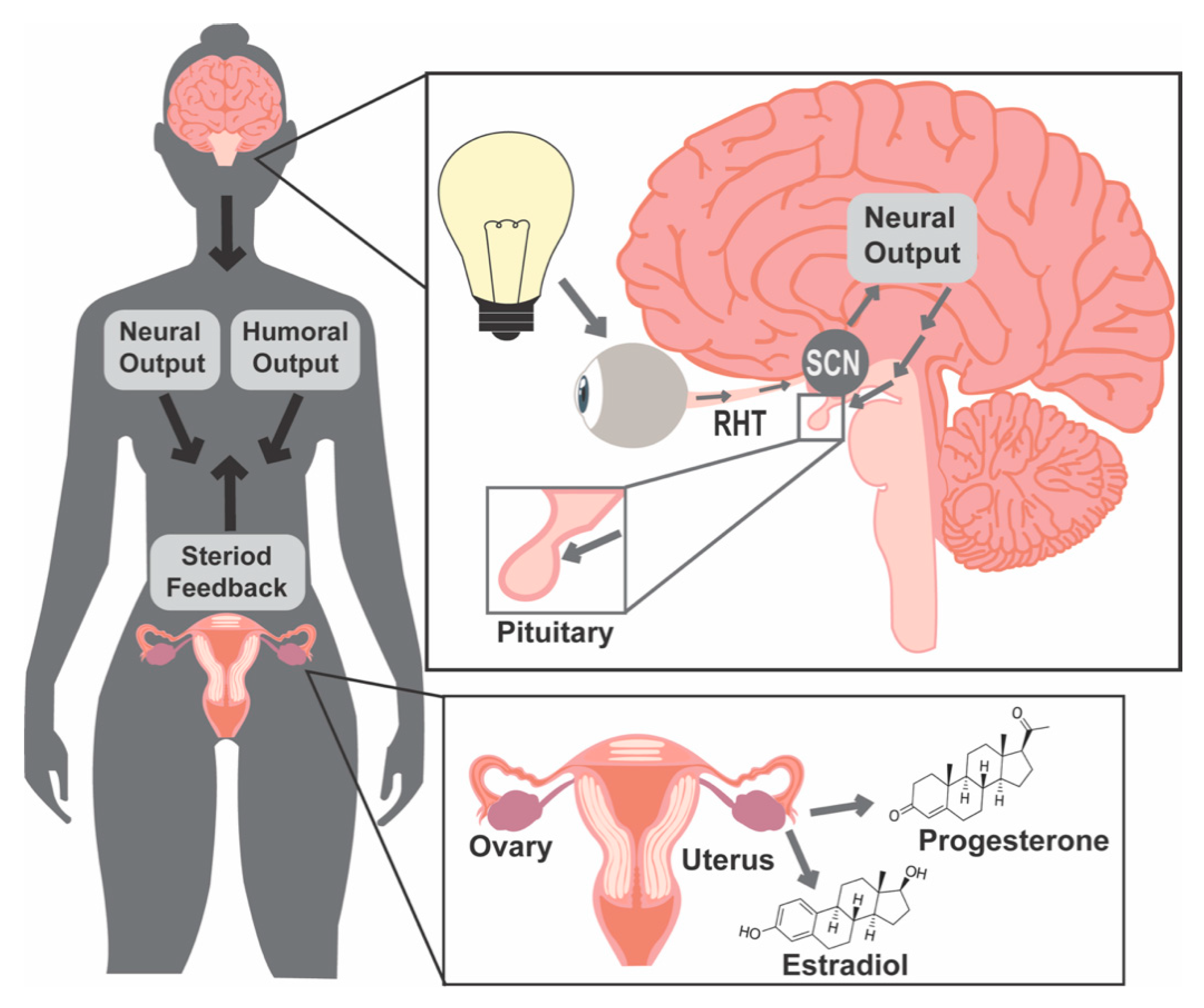
1.1. Light and Molecular Clocks
1.2. Female Reproductive Success Requires Precise Coordination of Hormone Release Patterns
1.3. Light and Food Timing Can Both Contribute to Circadian Disruption
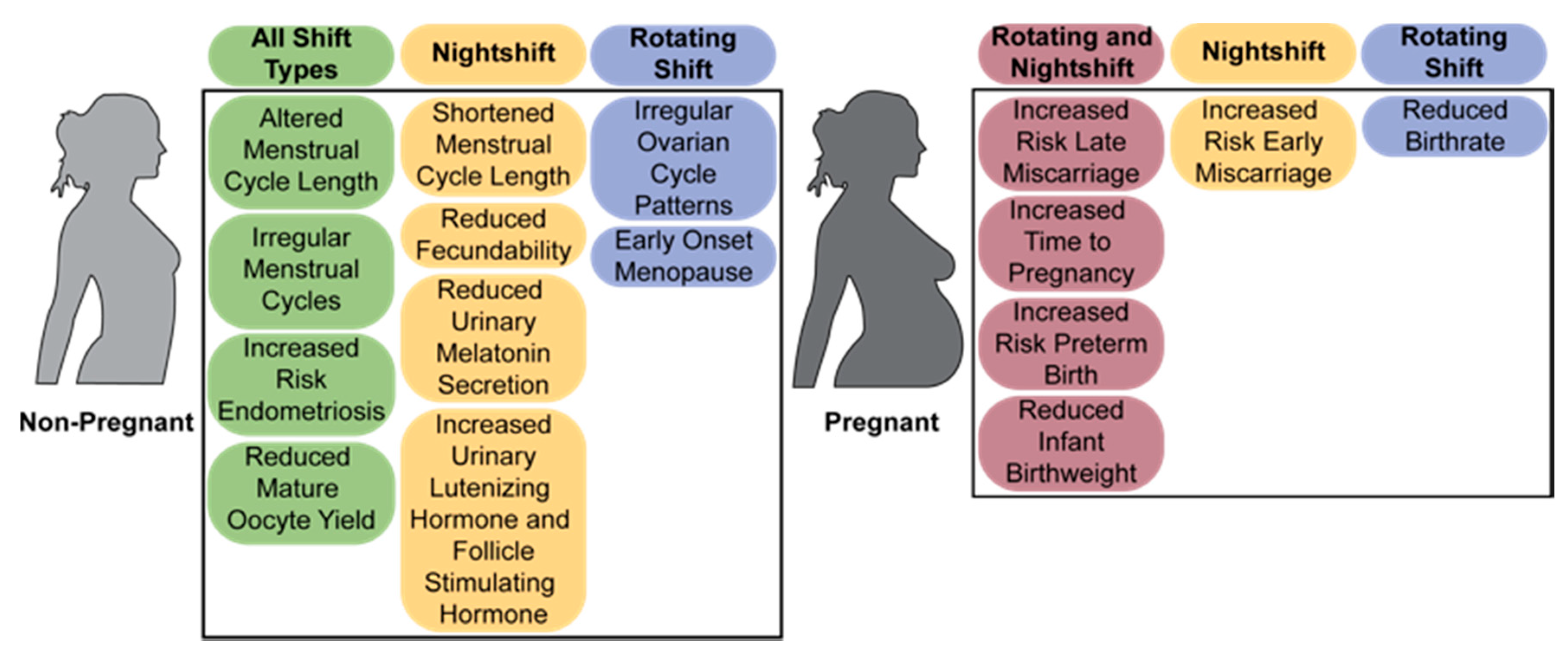
2. Chronic Shiftwork Negatively Impacts Fertility in Women
2.1. Human Literature Search Criteria
2.2. The Menstrual Cycle
2.3. Fertility and Miscarriage
2.4. Labor and Birth
2.5. Menopause and Reproductive Disorders
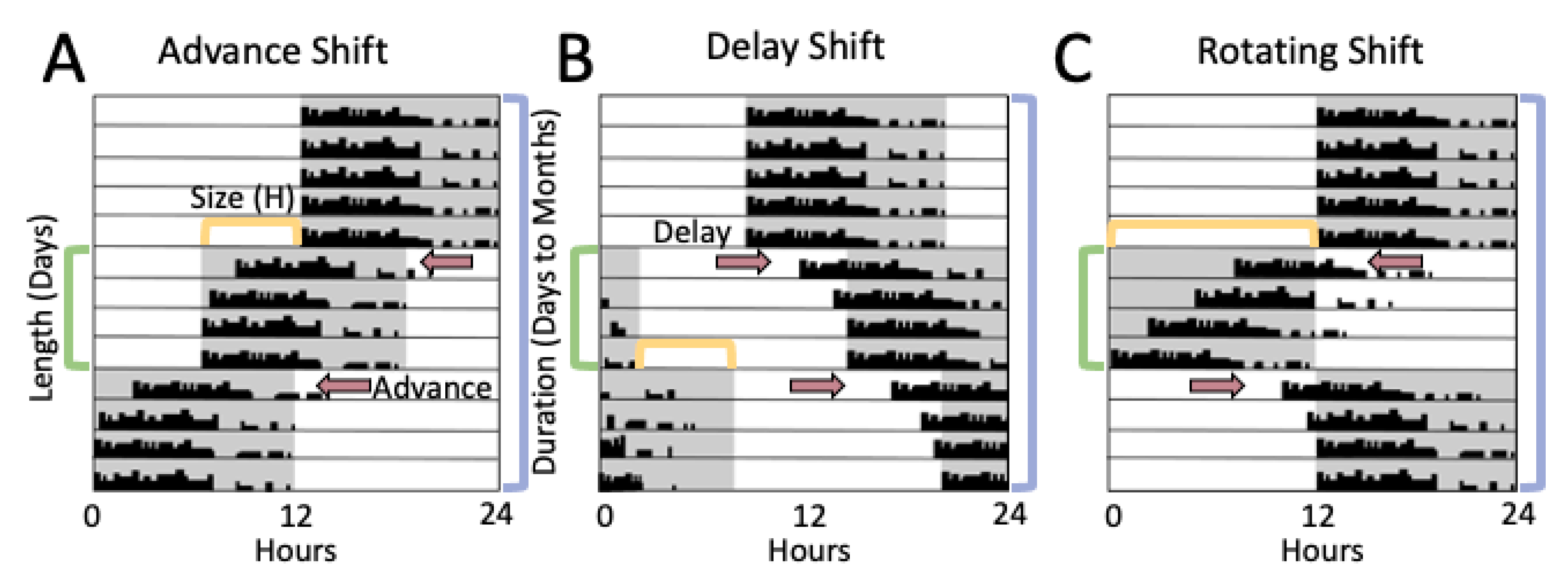
3. Rodent Models of Shiftwork
3.1. Methods of Evaluating Rodent Light-Shift Studies
3.2. Light Shifts Rodent Models and Estrous Cycling
3.3. Light-Shift Rodent Models, Pregnancy, and Labor
4. Discussion
4.1. Connections and Limitations between Human Studies and Rodent Light-Shift Models
4.2. Menstrual Cycle/Estrous Cycling
4.3. Pregnancy and Labor
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Labour Office. Work for a Brighter Future–Global Commission on the Future of Work 2019; International Labour Office: Geneva, Switzerland, 2019. [Google Scholar]
- Liu, K.; Hou, G.; Wang, X.; Chen, H.; Shi, F.; Liu, C.; Zhang, X.; Han, F.; Yang, H.; Zhou, N. Adverse effects of circadian desynchrony on the male reproductive system: An epidemiological and experimental study. Hum. Reprod. 2020, 35, 1515–1528. [Google Scholar] [CrossRef]
- Liu, K.; Hou, G.; Wang, X.; Chen, H.; Shi, F.; Liu, C.; Zhang, X.; Han, F.; Yang, H.; Zhou, N. Social Jetlag and Damage to Male Reproductive System: Epidemiological Observation in European and Chinese Populations and Biochemical Analyses in Mice. CURRENT-BIOLOGY-D-19-01835 2019. [Google Scholar] [CrossRef]
- Lawson, C.C.; Whelan, E.A.; Hibert, E.N.L.; Spiegelman, D.; Schernhammer, E.S.; Rich-Edwards, J.W. Rotating shift work and menstrual cycle characteristics. Epidemiology 2011, 22, 305. [Google Scholar] [CrossRef]
- Chung, F.-F.; Yao, C.-C.C.; Wan, G.-H. The associations between menstrual function and life style/working conditions among nurses in Taiwan. J. Occup. Health 2005, 47, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Labyak, S.; Lava, S.; Turek, F.; Zee, P. Effects of shiftwork on sleep and menstrual function in nurses. Health Care Women Int. 2002, 23, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Wan, G.; Chung, F. Working conditions associated with ovarian cycle in a medical center nurses: A Taiwan study. Japan J. Nurs. Sci. 2012, 9, 112–118. [Google Scholar] [CrossRef]
- Harlow, S.D.; Matanoski, G.M. The association between weight, physical activity, and stress and variation in the length of the menstrual cycle. Am. J. Epidemiol. 1991, 133, 38–49. [Google Scholar] [CrossRef]
- Hatch, M.C.; Figa-Talamanca, I.; Salerno, S. Work stress and menstrual patterns among American and Italian nurses. Scand. J. Work. Environ. Health 1999, 144–150. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Chen, M.-H.; Hsieh, C.-J.; Chen, P.-C. Effect of rotating shift work on childbearing and birth weight: A study of women working in a semiconductor manufacturing factory. World J. Pediatr. 2011, 7, 129–135. [Google Scholar] [CrossRef]
- Grajewski, B.; Whelan, E.A.; Lawson, C.C.; Hein, M.J.; Waters, M.A.; Anderson, J.L.; MacDonald, L.A.; Mertens, C.J.; Tseng, C.-Y.; Cassinelli, R.T. Miscarriage among flight attendants. Epidemiology 2015, 26, 192. [Google Scholar] [CrossRef]
- Whelan, E.A.; Lawson, C.C.; Grajewski, B.; Hibert, E.N.; Spiegelman, D.; Rich-Edwards, J.W. Work schedule during pregnancy and spontaneous abortion. Epidemiology 2007, 18, 350–355. [Google Scholar] [CrossRef]
- Begtrup, L.M.; Specht, I.O.; Hammer, P.E.C.; Flachs, E.M.; Garde, A.H.; Hansen, J.; Hansen, Å.M.; Kolstad, H.A.; Larsen, A.D.; Bonde, J.P. Night work and miscarriage: A Danish nationwide register-based cohort study. Occup Env. Med. 2019, 76, 302–308. [Google Scholar] [CrossRef]
- Lee, K.A.; Gay, C.L. Sleep in late pregnancy predicts length of labor and type of delivery. Am. J. Obstet. Gynecol. 2004, 191, 2041–2046. [Google Scholar] [CrossRef]
- Wilcox, A.J. On the importance—and the unimportance—of birthweight. Int. J. Epidemiol. 2001, 30, 1233–1241. [Google Scholar] [CrossRef]
- Xu, X.; Ding, M.; Li, B.; Christiani, D.C. Association of rotating shiftwork with preterm births and low birth weight among never smoking women textile workers in China. Occup. Environ. Med. 1994, 51, 470–474. [Google Scholar] [CrossRef]
- Zhu, J.L.; Hjollund, N.H.; Olsen, J. Shift work, duration of pregnancy, and birth weight: The National Birth Cohort in Denmark. Am. J. Obstet. Gynecol. 2004, 191, 285–291. [Google Scholar] [CrossRef]
- Bodin, L.; Axelsson, G.; Ahlborg Jr, G. The association of shift work and nitrous oxide exposure in pregnancy with birth weight and gestational age. Epidemiology 1999, 10, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Yang, C.; Tong, X.; Sun, H.; Cong, Y.; Yin, X.; Li, L.; Cao, S.; Dong, X.; Gong, Y.; et al. Shift work and diabetes mellitus: A meta-analysis of observational studies. Occup. Environ. Med. 2015, 72, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Knutsson, A.; Kempe, A. Shift work and diabetes–a systematic review. Chronobiol. Int. 2014, 31, 1146–1151. [Google Scholar] [CrossRef] [PubMed]
- Esquirol, Y.; Perret, B.; Ruidavets, J.B.; Marquie, J.C.; Dienne, E.; Niezborala, M.; Ferrieres, J. Shift work and cardiovascular risk factors: New knowledge from the past decade. Arch. Cardiovasc. Dis. 2011, 104, 636–668. [Google Scholar] [CrossRef]
- Puttonen, S.; Härmä, M.; Hublin, C. Shift work and cardiovascular disease—pathways from circadian stress to morbidity. Scand. J. Work. Environ. Health 2010, 36, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Bøggild, H.; Knutsson, A. Shift work, risk factors and cardiovascular disease. Scand. J. Work. Environ. Health 1999, 25, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Kamdar, B.B.; Tergas, A.I.; Mateen, F.J.; Bhayani, N.H.; Oh, J. Night-shift work and risk of breast cancer: A systematic review and meta-analysis. Breast Cancer Res. Treat. 2013, 138, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.; Mirick, D.K.; Stevens, R.G. Night shift work, light at night, and risk of breast cancer. J. Natl. Cancer Inst. 2001, 93. [Google Scholar] [CrossRef] [PubMed]
- Ijaz, S.; Verbeek, J.; Seidler, A.; Lindbohm, M.-L.; Ojajärvi, A.; Orsini, N.; Costa, G.; Neuvonen, K. Night-shift work and breast cancer—a systematic review and meta-analysis. Scand. J. Work. Environ. Health 2013, 39, 431–447. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.; Stevens, R.G. Case–control study of shift-work and breast cancer risk in Danish nurses: Impact of shift systems. Eur. J. Cancer 2012, 48, 1722–1729. [Google Scholar] [CrossRef] [PubMed]
- Partch, C.L.; Green, C.B.; Takahashi, J.S. Molecular architecture of the mammalian circadian clock. Trends Cell Biol. 2014, 24, 90–99. [Google Scholar] [CrossRef]
- Lowrey, P.L.; Takahashi, J.S. Genetics of the mammalian circadian system: Photic entrainment, circadian pacemaker mechanisms, and posttranslational regulation. Annu. Rev. Genet. 2000, 34, 533–562. [Google Scholar] [CrossRef]
- Cox, K.H.; Takahashi, J.S. Circadian clock genes and the transcriptional architecture of the clock mechanism. J. Mol. Endocrinol. 2019, 63. [Google Scholar] [CrossRef]
- Koike, N.; Yoo, S.H.; Huang, H.C.; Kumar, V.; Lee, C.; Kim, T.K.; Takahashi, J.S. Transcriptional architecture and chromatin landscape of the core circadian clock in mammals. Science 2012, 338. [Google Scholar] [CrossRef]
- Mereness, A.L.; Murphy, Z.C.; Forrestel, A.C.; Butler, S.; Ko, C.M.; Richards, J.A.S.; Sellix, M.T. Conditional deletion of Bmal1 in ovarian theca cells disrupts ovulation in female mice. Endocrinology 2016, 157, 913–927. [Google Scholar] [CrossRef] [PubMed]
- Hoyle, N.P.; Seinkmane, E.; Putker, M.; Feeney, K.A.; Krogager, T.P.; Chesham, J.E.; Bray, L.K.; Thomas, J.M.; Dunn, K.; Blaikley, J.; et al. Circadian actin dynamics drive rhythmic fibroblast mobilization during wound healing. Sci. Transl. Med. 2017, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bedont, J.L.; LeGates, T.A.; Slat, E.A.; Byerly, M.S.; Wang, H.; Hu, J.; Rupp, A.C.; Qian, J.; Wong, G.W.; Herzog, E.D.; et al. Lhx1 Controls Terminal Differentiation and Circadian Function of the Suprachiasmatic Nucleus. Cell Rep. 2014, 7, 609–622. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, C.K.; Boehle, K.L.; Muglia, L.J. Impaired steroidogenesis and implantation failure in Bmal1−/− mice. Endocrinology 2009, 150, 1879–1885. [Google Scholar] [CrossRef]
- Kriegsfeld, L.J.; Williams III, W.P. Circadian control of neuroendocrine circuits regulating female reproductive function. Front. Endocrinol. (Lausanne) 2012, 3, 60. [Google Scholar]
- Mahoney, M.M.; Sisk, C.; Ross, H.E.; Smale, L. Circadian regulation of gonadotropin-releasing hormone neurons and the preovulatory surge in luteinizing hormone in the diurnal rodent, Arvicanthis niloticus, and in a nocturnal rodent, Rattus norvegicus. Biol. Reprod. 2004, 70, 1049–1054. [Google Scholar] [CrossRef]
- Chappell, P.E. Clocks and the black box: Circadian influences on gonadotropin-releasing hormone secretion. J. Neuroendocrinol. 2005, 17, 119–130. [Google Scholar] [CrossRef]
- Gibson, E.M.; Humber, S.A.; Jain, S.; Williams, W.P.; Zhao, S.; Bentley, G.E.; Tsutsui, K.; Kriegsfeld, L.J. Alterations in RFamide-related peptide expression are coordinated with the preovulatory luteinizing hormone surge. Endocrinology 2008, 149. [Google Scholar] [CrossRef]
- De La Iglesia, H.O.; Schwartz, W.J. Minireview: Timely ovulation: Circadian regulation of the female hypothalamo-pituitary-gonadal axis. Endocrinology 2006, 147, 1148–1153. [Google Scholar] [CrossRef]
- Kriegsfeld, L.J.; Silver, R. The regulation of neuroendocrine function: Timing is everything. Horm. Behav. 2006, 49, 557–574. [Google Scholar] [CrossRef]
- Christian, C.A.; Moenter, S.M. The neurobiology of preovulatory and estradiol-induced gonadotropin-releasing hormone surges. Endocr. Rev. 2010, 31, 544–577. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.H.; Takahashi, J.S. Central circadian control of female reproductive function. Front. Endocrinol. (Lausanne) 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Burger, L.L.; Haisenleder, D.J.; Aylor, K.W.; Marshall, J.C. Regulation of Intracellular Signaling Cascades by GNRH Pulse Frequency in the Rat Pituitary: Roles for CaMK II, ERK, and JNK Activation1. Biol. Reprod. 2008, 79. [Google Scholar] [CrossRef] [PubMed]
- Haisenleder, D.J.; Burger, L.L.; Walsh, H.E.; Stevens, J.; Aylor, K.W.; Shupnik, M.A.; Marshall, J.C. Pulsatile gonadotropin-releasing hormone stimulation of gonadotropin subunit transcription in rat pituitaries: Evidence for the involvement of Jun N-terminal kinase but not p38. Endocrinology 2008, 149. [Google Scholar] [CrossRef] [PubMed]
- Dalkin, A.C.; Haisenleder, D.J.; Ortolano, G.A.; Ellis, T.R.; Marshall, J.C. The frequency of gonadotropin-releasing-hormone stimulation differentially regulates gonadotropin subunit messenger ribonucleic acid expression. Endocrinology 1989, 125. [Google Scholar] [CrossRef]
- Haisenleder, D.J.; Katt, J.A.; Ortolano, G.A.; El-Gewely, M.R.; Duncan, J.A.; Dee, C.; Marshall, J.C. Influence of gonadotropin-releasing hormone pulse amplitude, frequency, and treatment duration on the regulation of luteinizing hormone (LH) subunit messenger ribonucleic acids and LH secretion. Mol. Endocrinol. 1988, 2. [Google Scholar] [CrossRef]
- Papavasiliou, S.S.; Zmeili, S.; Khoury, S.; Landefeld, T.D.; Chin, W.W.; Marshall, J.C. Gonadotropin-releasing hormone differentially regulates expression of the genes for luteinizing hormone α and β subunits in male rats. Proc. Natl. Acad. Sci. USA 1986, 83. [Google Scholar] [CrossRef]
- Sen, A.; Hoffmann, H.M. Role of core circadian clock genes in hormone release and target tissue sensitivity in the reproductive axis. Mol. Cell. Endocrinol. 2020, 501. [Google Scholar] [CrossRef]
- Han, S.Y.; Cheong, I.; McLennan, T.; Herbison, A.E. Neural determinants of pulsatile luteinizing hormone secretion in male mice. Endocrinology 2020, 161. [Google Scholar] [CrossRef]
- Han, S.Y.; Kane, G.; Cheong, I.; Herbison, A.E. Characterization of GnRH pulse generator activity in male mice using GCaMP fiber photometry. Endocrinology 2019, 160. [Google Scholar] [CrossRef]
- Chen, H.; Zhao, L.; Kumazawa, M.; Yamauchi, N.; Shigeyoshi, Y.; Hashimoto, S.; Hattori, M.-a. Downregulation of core clock gene Bmal1 attenuates expression of progesterone and prostaglandin biosynthesis-related genes in rat luteinizing granulosa cells. AJP Cell Physiol. 2013, 304. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhao, L.; Chu, G.; Kito, G.; Yamauchi, N.; Shigeyoshi, Y.; Hashimoto, S.; Hattori, M. aki FSH induces the development of circadian clockwork in rat granulosa cells via a gap junction protein Cx43-dependent pathway. Am. J. Physiol. Endocrinol. Metab. 2013, 304. [Google Scholar] [CrossRef] [PubMed]
- Sellix, M.T.; Yoshikawa, T.; Menaker, M. A circadian egg timer gates ovulation. Curr. Biol. 2010, 20, 266–267. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.H.; Lim, A.; Fernando, D.; Day, M.L. Circadian clockwork genes are expressed in the reproductive tract and conceptus of the early pregnant mouse. Reprod. Biomed. Online 2002, 4. [Google Scholar] [CrossRef]
- Liu, Y.; Johnson, B.P.; Shen, A.L.; Wallisser, J.A.; Krentz, K.J.; Moran, S.M.; Sullivan, R.; Glover, E.; Parlow, A.F.; Drinkwater, N.R.; et al. Loss of BMAL1 in ovarian steroidogenic cells results in implantation failure in female mice. Proc. Natl. Acad. Sci. USA 2014, 111. [Google Scholar] [CrossRef] [PubMed]
- Gamble, K.L.; Resuehr, D.; Johnson, C.H. Shift work and circadian dysregulation of reproduction. Front. Endocrinol. (Lausanne) 2013, 4. [Google Scholar] [CrossRef]
- Gorman, M.R. Temporal organization of pineal melatonin signaling in mammals. Mol. Cell. Endocrinol. 2020, 503. [Google Scholar] [CrossRef]
- Tamura, H.; Nakamura, Y.; Terron, M.P.; Flores, L.J.; Manchester, L.C.; Tan, D.X.; Sugino, N.; Reiter, R.J. Melatonin and pregnancy in the human. Reprod. Toxicol. 2008, 25, 291–303. [Google Scholar] [CrossRef]
- Nakamura, Y.; Tamura, H.; Kashida, S.; Takayama, H.; Yamagata, Y.; Karube, A.; Sugino, N.; Kato, H. Changes of serum melatonin level and its relationship to feto-placental unit during pregnancy. J. Pineal Res. 2001, 30, 29–33. [Google Scholar] [CrossRef]
- Lok, R.; van Koningsveld, M.J.; Gordijn, M.C.M.; Beersma, D.G.M.; Hut, R.A. Daytime melatonin and light independently affect human alertness and body temperature. J. Pineal Res. 2019, 67. [Google Scholar] [CrossRef]
- Dollins, A.B.; Zhdanova, I.V.; Wurtman, R.J.; Lynch, H.J.; Deng, M.H. Effect of inducing nocturnal serum melatonin concentrations in daytime on sleep, mood, body temperature, and performance. Proc. Natl. Acad. Sci. USA 1994, 91. [Google Scholar] [CrossRef] [PubMed]
- Auld, F.; Maschauer, E.L.; Morrison, I.; Skene, D.J.; Riha, R.L. Evidence for the efficacy of melatonin in the treatment of primary adult sleep disorders. Sleep Med. Rev. 2017, 34, 10–22. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, R.T.; Jungheim, E.S.; Fay, J.C.; Bates, K.; Herzog, E.D.; England, S.K. Riding the rhythm of melatonin through pregnancy to deliver on time. Front. Endocrinol. (Lausanne) 2019, 10, 616. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.J.K.; Vidafar, P.; Burns, A.C.; McGlashan, E.M.; Anderson, C.; Rajaratnam, S.M.W.; Lockley, S.W.; Cain, S.W. High sensitivity and interindividual variability in the response of the human circadian system to evening light. Proc. Natl. Acad. Sci. USA 2019, 116, 12019–12024. [Google Scholar] [CrossRef]
- Rahman, S.A.; Marcu, S.; Shapiro, C.M.; Brown, T.J.; Casper, R.F. Spectral modulation attenuates molecular, endocrine, and neurobehavioral disruption induced by nocturnal light exposure. Am. J. Physiol. Metab. 2011, 300, E518–E527. [Google Scholar] [CrossRef]
- Kayumov, L.; Casper, R.F.; Hawa, R.J.; Perelman, B.; Chung, S.A.; Sokalsky, S.; Shapiro, C.M. Blocking low-wavelength light prevents nocturnal melatonin suppression with no adverse effect on performance during simulated shift work. J. Clin. Endocrinol. Metab. 2005, 90, 2755–2761. [Google Scholar] [CrossRef]
- Rahman, S.A.; Kollara, A.; Brown, T.J.; Casper, R.F. Selectively filtering short wavelengths attenuates the disruptive effects of nocturnal light on endocrine and molecular circadian phase markers in rats. Endocrinology 2008, 149, 6125–6135. [Google Scholar] [CrossRef]
- Schug, T.T.; Janesick, A.; Blumberg, B.; Heindel, J.J. Endocrine disrupting chemicals and disease susceptibility. J. Steroid Biochem. Mol. Biol. 2011, 127, 204–215. [Google Scholar] [CrossRef]
- Longo, V.D.; Panda, S. Fasting, Circadian Rhythms, and Time-Restricted Feeding in Healthy Lifespan. Cell Metab. 2016, 23. [Google Scholar] [CrossRef]
- Vollmers, C.; Gill, S.; DiTacchio, L.; Pulivarthy, S.R.; Le, H.D.; Panda, S. Time of feeding and the intrinsic circadian clock drive rhythms in hepatic gene expression. Proc. Natl. Acad. Sci. USA 2009, 106, 21453–21458. [Google Scholar] [CrossRef]
- Panda, S. Circadian physiology of metabolism. Science 2016, 354, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Chaix, A.; Lin, T.; Le, H.D.; Chang, M.W.; Panda, S. Time-Restricted Feeding Prevents Obesity and Metabolic Syndrome in Mice Lacking a Circadian Clock. Cell Metab. 2019, 29. [Google Scholar] [CrossRef] [PubMed]
- Stephan, F.K. Phase shifts of circadian rhythms in activity entrained to food access. Physiol. Behav. 1984, 32, 663–671. [Google Scholar] [CrossRef]
- Mistlberger, R.E. Circadian food-anticipatory activity: Formal models and physiological mechanisms. Neurosci. Biobehav. Rev. 1994, 18, 171–195. [Google Scholar] [CrossRef]
- Swamy, S.; Xie, X.; Kukino, A.; Calcagno, H.E.; Lasarev, M.R.; Park, J.H.; Butler, M.P. Circadian disruption of food availability significantly reduces reproductive success in mice. Horm. Behav. 2018, 105. [Google Scholar] [CrossRef]
- Lowden, A.; Moreno, C.; Holmbäck, U.; Lennernäs, M.; Tucker, P. Eating and shift work—effects on habits, metabolism, and performance. Scand. J. Work. Environ. Health 2010, 36, 150–162. [Google Scholar] [CrossRef]
- Opperhuizen, A.L.; van Kerkhof, L.W.M.; Proper, K.I.; Rodenburg, W.; Kalsbeek, A. Rodent models to study the metabolic effects of shiftwork in humans. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef]
- Tsang, A.H.; Astiz, M.; Leinweber, B.; Oster, H. Rodent models for the analysis of tissue clock function in metabolic rhythms research. Front. Endocrinol. (Lausanne) 2017, 8, 27. [Google Scholar] [CrossRef]
- Fonken, L.K.; Nelson, R.J. The effects of light at night on circadian clocks and metabolism. Endocr. Rev. 2014, 35, 648–670. [Google Scholar] [CrossRef]
- Sati, L. Chronodisruption: Effects on reproduction, transgenerational health of offspring and epigenome. Reproduction 2020, 160, R79–R94. [Google Scholar] [CrossRef]
- Su, S.; Lu, C.; Kao, Y.; Guo, H. Effects of 12-hour rotating shifts on menstrual cycles of photoelectronic workers in Taiwan. Chronobiol. Int. 2008, 25, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Gu, F.; Deng, M.; Guo, L.; Lu, C.; Zhou, C.; Chen, S.; Xu, Y. Rotating shift work and menstrual characteristics in a cohort of Chinese nurses. BMC Womens Health 2016, 16, 24. [Google Scholar] [CrossRef] [PubMed]
- Albert-Sabater, J.A.; Martínez, J.M.; Baste, V.; Moen, B.E.; Ronda-Perez, E. Comparison of menstrual disorders in hospital nursing staff according to shift work pattern. J. Clin. Nurs. 2016, 25, 3291–3299. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, R.C.; Moore, V.M.; Marino, J.L.; Whitrow, M.J.; Davies, M.J. Night Shift Among Women: Is It Associated With Difficulty Conceiving a First Birth? Front. Public Heal. 2020, 8, 676. [Google Scholar]
- Mínguez-Alarcón, L.; Souter, I.; Williams, P.L.; Ford, J.B.; Hauser, R.; Chavarro, J.E.; Gaskins, A.J. Occupational factors and markers of ovarian reserve and response among women at a fertility centre. Occup. Environ. Med. 2017, 74, 426–431. [Google Scholar] [CrossRef]
- Ahlborg Jr, G.; AXELSSON, G.; Bodin, L. Shift work, nitrous oxide exposure and subfertility among Swedish midwives. Int. J. Epidemiol. 1996, 25, 783–790. [Google Scholar] [CrossRef]
- Sponholtz, T.R.; Bethea, T.N.; Ruiz-Narváez, E.A.; Boynton-Jarrett, R.; Palmer, J.R.; Rosenberg, L.; Wise, L.A. Night Shift Work and Fecundability in Late Reproductive-Aged African American Women. J. Women’s Heal. 2020. [Google Scholar] [CrossRef]
- Gaskins, A.J.; Rich-Edwards, J.W.; Lawson, C.C.; Schernhammer, E.S.; Missmer, S.A.; Chavarro, J.E. Work schedule and physical factors in relation to fecundity in nurses. Occup. Environ. Med. 2015, 72, 777–783. [Google Scholar] [CrossRef]
- Zhu, J.L.; Hjollund, N.H.; Boggild, H.; Olsen, J. Shift work and subfecundity: A causal link or an artefact? Occup. Environ. Med. 2003, 60, e12. [Google Scholar] [CrossRef]
- Xu, G.; Wu, Y.; Yang, L.; Yuan, L.; Guo, H.; Zhang, F.; Guan, Y.; Yao, W. Risk factors for early miscarriage among Chinese: A hospital-based case-control study. Fertil. Steril. 2014, 101, 1663–1670. [Google Scholar] [CrossRef]
- Infante-Rivard, C.; David, M.; Gauthier, R.; Rivard, G.-E. Pregnancy loss and work schedule during pregnancy. Epidemiology 1993, 4, 73–75. [Google Scholar] [CrossRef]
- Lawson, C.C.; Rocheleau, C.M.; Whelan, E.A.; Hibert, E.N.L.; Grajewski, B.; Spiegelman, D.; Rich-Edwards, J.W. Occupational exposures among nurses and risk of spontaneous abortion. Am. J. Obstet. Gynecol. 2012, 206. [Google Scholar] [CrossRef]
- Zhu, J.L.; Hjollund, N.H.; Andersen, A.-M.N.; Olsen, J. Shift work, job stress, and late fetal loss: The National Birth Cohort in Denmark. J. Occup. Environ. Med. 2004, 46, 1144–1149. [Google Scholar] [CrossRef]
- Axelsson, G.; Ahlborg, G.; Bodin, L. Shift work, nitrous oxide exposure, and spontaneous abortion among Swedish midwives. Occup. Environ. Med. 1996, 53, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, G.; Rylander, R.; Molin, I. Outcome of pregnancy in relation to irregular and inconvenient work schedules. Occup. Environ. Med. 1989, 46, 393–398. [Google Scholar] [CrossRef] [PubMed]
- McDonald, A.D.; McDonald, J.C.; Armstrong, B.; Cherry, N.M.; Nolin, A.D.; Robert, D. Prematurity and work in pregnancy. Occup. Environ. Med. 1988, 45, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Abeysena, C.; Jayawardana, P.; DE A SENEVIRATNE, R. Maternal sleep deprivation is a risk factor for small for gestational age: A cohort study. Aust. New Zeal. J. Obstet. Gynaecol. 2009, 49, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Niedhammer, I.; O’Mahony, D.; Daly, S.; Morrison, J.J.; Kelleher, C.C.; Group, L.C.C.S.S. Occupational predictors of pregnancy outcomes in Irish working women in the Lifeways cohort. BJOG Int. J. Obstet. Gynaecol. 2009, 116, 943–952. [Google Scholar] [CrossRef]
- Croteau, A.; Marcoux, S.; Brisson, C. Work activity in pregnancy, preventive measures, and the risk of delivering a small-for-gestational-age infant. Am. J. Public Health 2006, 96, 846–855. [Google Scholar] [CrossRef]
- Nurminen, T. Shift work, fetal development and course of pregnancy. Scand. J. Work. Environ. Health 1989, 15, 395–403. [Google Scholar] [CrossRef]
- Pompeii, L.A.; Savitz, D.A.; Evenson, K.R.; Rogers, B.; McMahon, M. Physical exertion at work and the risk of preterm delivery and small-for-gestational-age birth. Obstet. Gynecol. 2005, 106, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Buen, M.; Amaral, E.; Souza, R.T.; Passini, R.; Lajos, G.J.; Tedesco, R.P.; Nomura, M.L.; Dias, T.Z.; Rehder, P.M.; Sousa, M.H. Maternal Work and Spontaneous Preterm Birth: A Multicenter Observational Study in Brazil. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bonzini, M.; Coggon, D.; Godfrey, K.; Inskip, H.; Crozier, S.; Palmer, K.T. Occupational physical activities, working hours and outcome of pregnancy: Findings from the Southampton Women’s Survey. Occup. Environ. Med. 2009, 66, 685–690. [Google Scholar] [CrossRef] [PubMed]
- van Melick, M.J.G.J.; van Beukering, M.D.M.; Mol, B.W.; Frings-Dresen, M.H.W.; Hulshof, C.T.J. Shift work, long working hours and preterm birth: A systematic review and meta-analysis. Int. Arch. Occup. Environ. Health 2014, 87, 835–849. [Google Scholar] [CrossRef]
- Specht, I.O.; Hammer, P.E.C.; Flachs, E.M.; Begtrup, L.M.; Larsen, A.D.; Hougaard, K.S.; Hansen, J.; Hansen, Å.M.; Kolstad, H.A.; Rugulies, R. Night work during pregnancy and preterm birth—A large register-based cohort study. PLoS ONE 2019, 14. [Google Scholar] [CrossRef]
- Mozurkewich, E.L.; Luke, B.; Avni, M.; Wolf, F.M. Working conditions and adverse pregnancy outcome: A meta-analysis. Obstet. Gynecol. 2000, 95, 623–635. [Google Scholar] [CrossRef]
- Stock, D.; Knight, J.A.; Raboud, J.; Cotterchio, M.; Strohmaier, S.; Willett, W.; Eliassen, A.H.; Rosner, B.; Hankinson, S.E.; Schernhammer, E. Rotating night shift work and menopausal age. Hum. Reprod. 2019, 34, 539–548. [Google Scholar] [CrossRef]
- Schernhammer, E.S.; Laden, F.; Speizer, F.E.; Willett, W.C.; Hunter, D.J.; Kawachi, I.; Colditz, G.A. Rotating night shifts and risk of breast cancer in women participating in the nurses’ health study. J. Natl. cancer Inst. 2001, 93, 1563–1568. [Google Scholar] [CrossRef]
- Viswanathan, A.N.; Hankinson, S.E.; Schernhammer, E.S. Night shift work and the risk of endometrial cancer. Cancer Res. 2007, 67, 10618–10622. [Google Scholar] [CrossRef]
- Bhatti, P.; Cushing-Haugen, K.L.; Wicklund, K.G.; Doherty, J.A.; Rossing, M.A. Nightshift work and risk of ovarian cancer. Occup. Environ. Med. 2013, 70, 231–237. [Google Scholar] [CrossRef]
- Carter, B.D.; Diver, W.R.; Hildebrand, J.S.; Patel, A.V.; Gapstur, S.M. Circadian disruption and fatal ovarian cancer. Am. J. Prev. Med. 2014, 46, S34–S41. [Google Scholar] [CrossRef] [PubMed]
- Poole, E.M.; Schernhammer, E.S.; Tworoger, S.S. Rotating night shift work and risk of ovarian cancer. Cancer Epidemiol. Prev. Biomarkers 2011, 20, 934–938. [Google Scholar] [CrossRef] [PubMed]
- Marino, J.L.; Holt, V.L.; Chen, C.; Davis, S. Shift work, hCLOCK T3111C polymorphism, and endometriosis risk. Epidemiology 2008, 19, 477. [Google Scholar] [CrossRef] [PubMed]
- Schernhammer, E.S.; Rosner, B.; Willett, W.C.; Laden, F.; Colditz, G.A.; Hankinson, S.E. Epidemiology of urinary melatonin in women and its relation to other hormones and night work. Cancer Epidemiol. Prev. Biomarkers 2004, 13, 936–943. [Google Scholar]
- Gómez-Acebo, I.; Dierssen-Sotos, T.; Papantoniou, K.; García-Unzueta, M.T.; Santos-Benito, M.F.; Llorca, J. Association between exposure to rotating night shift versus day shift using levels of 6-sulfatoxymelatonin and cortisol and other sex hormones in women. Chronobiol. Int. 2015, 32, 128–135. [Google Scholar] [CrossRef]
- Baumgartner, A.; Dietzel, M.; Saletu, B.; Wolf, R.; Campos-Barros, A.; Gräf, K.-J.; Kürten, I.; Mannsmann, U. Influence of partial sleep deprivation on the secretion of thyrotropin, thyroid hormones, growth hormone, prolactin, luteinizing hormone, follicle stimulating hormone, and estradiol in healthy young women. Psychiatry Res. 1993, 48, 153–178. [Google Scholar] [CrossRef]
- Davis, S.; Mirick, D.K.; Chen, C.; Stanczyk, F.Z. Night shift work and hormone levels in women. Cancer Epidemiol. Prev. Biomarkers 2012, 21, 609–618. [Google Scholar] [CrossRef]
- Yamauchi, H. Effects of night work on urinary excretion rates of 6-sulfatoxymelatonin, norepinephrine and estriol in pregnant women. Ind. Health 2004, 42, 268–276. [Google Scholar] [CrossRef]
- Michels, K.A.; Mendola, P.; Schliep, K.C.; Yeung, E.H.; Ye, A.; Dunietz, G.L.; Wactawski-Wende, J.; Kim, K.; Freeman, J.R.; Schisterman, E.F. The influences of sleep duration, chronotype, and nightwork on the ovarian cycle. Chronobiol. Int. 2020, 37, 260–271. [Google Scholar] [CrossRef]
- Peplonska, B.; Bukowska, A.; Lie, J.A.; Gromadzinska, J.; Zienolddiny, S. Night shift work and other determinants of estradiol, testosterone, and dehydroepiandrosterone sulfate among middle-aged nurses and midwives. Scand. J. Work. Environ. Health 2016, 42, 435–446. [Google Scholar] [CrossRef]
- Baker, F.C.; Driver, H.S. Circadian rhythms, sleep, and the menstrual cycle. Sleep Med. 2007, 8, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.A.; Wright, K.P.; Lockley, S.W.; Czeisler, C.A.; Gronfier, C. Characterizing the temporal Dynamics of Melatonin and Cortisol Changes in Response to Nocturnal Light Exposure. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Christiani, D.C.; Niu, T.; Xu, X. Occupational stress and dysmenorrhea in women working in cotton textile mills. Int. J. Occup. Environ. Health 1995, 1, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Shortridge, L.A.; Lemasters, G.K.; Valanis, B.; Hertzberg, V. Menstrual cycles in nurses handling antineoplastic drugs. Cancer Nurs. 1995, 18, 439–444. [Google Scholar] [CrossRef]
- Kolstad, H.A.; Bonde, J.P.; Hjøllund, N.H.; Jensen, T.K.; Henriksen, T.B.; Ernst, E.; Giwercman, A.; Skakkebæk, N.E.; Olsen, J. Menstrual cycle pattern and fertility: A prospective follow-up study of pregnancy and early embryonal loss in 295 couples who were planning their first pregnancy. Fertil. Steril. 1999, 71, 490–496. [Google Scholar] [CrossRef]
- Kauffman, A.S.; Hoffmann, H.M. Editorial: Hormone release patterns in mammals. Mol. Cell. Endocrinol. 2020, 507. [Google Scholar] [CrossRef]
- Kumar, P.; Magon, N. Hormones in pregnancy. Niger. Med. J. J. Niger. Med. Assoc. 2012, 53. [Google Scholar] [CrossRef]
- Okada, H.; Tsuzuki, T.; Murata, H. Decidualization of the human endometrium. Reprod. Med. Biol. 2018, 17, 220–227. [Google Scholar] [CrossRef]
- Callaghan, W.M.; MacDorman, M.F.; Rasmussen, S.A.; Qin, C.; Lackritz, E.M. The contribution of preterm birth to infant mortality rates in the United States. Pediatrics 2006, 118, 1566–1573. [Google Scholar] [CrossRef]
- Behrman, R.E.; Butler, A.S. Preterm Birth: Causes, Consequences, and Prevention; National Academies Press: Washington, DC, USA, 2007. [Google Scholar] [CrossRef]
- Nehme, P.A.; Amaral, F.G.; Middleton, B.; Lowden, A.; Marqueze, E.; França-Junior, I.; Antunes, J.L.F.; Cipolla-Neto, J.; Skene, D.J.; Moreno, C.R.C. Melatonin profiles during the third trimester of pregnancy and health status in the offspring among day and night workers: A case series. Neurobiol. Sleep Circadian Rhythm. 2019, 6. [Google Scholar] [CrossRef]
- Hilder, L.; Costeloe, K.; Thilaganathan, B. Prolonged pregnancy: Evaluating gestation-specific risks of fetal and infant mortality. BJOG Int. J. Obstet. Gynaecol. 1998, 105, 169–173. [Google Scholar] [CrossRef] [PubMed]
- El Marroun, H.; Zeegers, M.; Steegers, E.A.P.; van der Ende, J.; Schenk, J.J.; Hofman, A.; Jaddoe, V.W.V.; Verhulst, F.C.; Tiemeier, H. Post-term birth and the risk of behavioural and emotional problems in early childhood. Int. J. Epidemiol. 2012, 41, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Cagnacci, A.; Soldani, R.; Melis, G.B.; Volpe, A. Diurnal rhythms of labor and delivery in women: Modulation by parity and seasons. Am. J. Obstet. Gynecol. 1998, 178, 140–145. [Google Scholar] [CrossRef]
- Olcese, J. Circadian aspects of mammalian parturition: A review. Mol. Cell. Endocrinol. 2012, 349, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Olcese, J.; Lozier, S.; Paradise, C. Melatonin and the Circadian Timing of Human Parturition. Reprod. Sci. 2013, 20, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Bosc, M.J. Photoperiodic regulation of the time of birth in rats: Involvement of circadian endogenous mechanisms. Physiol. Behav. 1990, 48, 441–446. [Google Scholar] [CrossRef]
- Hirst, J.J.; Haluska, G.J.; Cook, M.J.; Novy, M.J. Plasma oxytocin and nocturnal uterine activity: Maternal but not fetal concentrations increase progressively during late pregnancy and delivery in rhesus monkeys. Am. J. Obstet. Gynecol. 1993, 169, 415–422. [Google Scholar] [CrossRef]
- Yellon, S.M.; Longo, L.D. Melatonin rhythms in fetal and maternal circulation during pregnancy in sheep. Am. J. Physiol. 1987, 252, E799–E802. [Google Scholar] [CrossRef]
- Kennaway, D.J.; Voultsios, A.; Varcoe, T.J.; Moyer, R.W. Melatonin in mice: Rhythms, response to light, adrenergic stimulation, and metabolism. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 282, R358–R365. [Google Scholar] [CrossRef][Green Version]
- Wierrani, F.; Grin, W.; Hlawka, B.; Kroiss, A.; Grünberger, W. Elevated serum melatonin levels during human late pregnancy and labour. J. Obstet. Gynaecol. (Lahore) 1997, 17, 449–451. [Google Scholar] [CrossRef]
- Sharkey, J.T.; Puttaramu, R.; Word, R.A.; Olcese, J. Melatonin synergizes with oxytocin to enhance contractility of human myometrial smooth muscle cells. J. Clin. Endocrinol. Metab. 2009, 94, 421–427. [Google Scholar] [CrossRef]
- Olcese, J.; Beesley, S. Clinical significance of melatonin receptors in the human myometrium. Fertil. Steril. 2014, 102. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, K.; Klerman, E.B.; Phillips, A.J.K. Are Individual Differences in Sleep and Circadian Timing Amplified by Use of Artificial Light Sources? J. Biol. Rhythms 2017, 32, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.A.; Bibbo, C.; Olcese, J.; Czeisler, C.A.; Robinson, J.N.; Klerman, E.B. Relationship between endogenous melatonin concentrations and uterine contractions in late third trimester of human pregnancy. J. Pineal Res. 2019, 66, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Beesley, S.; Lee, J.; Olcese, J. Circadian clock regulation of melatonin MTNR1B receptor expression in human myometrial smooth muscle cells. Mol. Hum. Reprod. 2015, 21, 662–671. [Google Scholar] [CrossRef]
- Backe, B. A circadian variation in the observed duration of labor. Acta Obstet. Gynecol. Scand. 1991, 70, 465–468. [Google Scholar] [CrossRef]
- Murphy, V.E.; Smith, R.; Giles, W.B.; Clifton, V.L. Endocrine regulation of human fetal growth: The role of the mother, placenta, and fetus. Endocr. Rev. 2006, 27, 141–169. [Google Scholar] [CrossRef]
- Mucci, L.A.; Lagiou, P.; Tamimi, R.M.; Hsieh, C.-C.; Adami, H.-O.; Trichopoulos, D. Pregnancy estriol, estradiol, progesterone and prolactin in relation to birth weight and other birth size variables (United States). Cancer Causes Control 2003, 14, 311–318. [Google Scholar] [CrossRef]
- McKinlay, S.M.; Brambilla, D.J.; Posner, J.G. The normal menopause transition. Maturitas 1992, 14, 103–115. [Google Scholar] [CrossRef]
- Gold, E.B. The timing of the age at which natural menopause occurs. Obstet. Gynecol. Clin. 2011, 38, 425–440. [Google Scholar] [CrossRef]
- Haus, E.L.; Smolensky, M.H. Shift work and cancer risk: Potential mechanistic roles of circadian disruption, light at night, and sleep deprivation. Sleep Med. Rev. 2013, 17, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Brinton, L.A.; Gridley, G.; Persson, I.; Baron, J.; Bergqvist, A. Cancer risk after a hospital discharge diagnosis of endometriosis. Am. J. Obstet. Gynecol. 1997, 176, 572–579. [Google Scholar] [CrossRef]
- Bahougne, T.; Kretz, M.; Angelopoulou, E.; Jeandidier, N.; Simonneaux, V. Impact of Circadian Disruption on Female Mice Reproductive Function. Endocrinol. (United States) 2020, 161. [Google Scholar] [CrossRef] [PubMed]
- Takasu, N.N.; Nakamura, T.J.; Tokuda, I.T.; Todo, T.; Block, G.D.; Nakamura, W. Recovery from Age-Related Infertility under Environmental Light-Dark Cycles Adjusted to the Intrinsic Circadian Period. Cell Rep. 2015, 12, 1407–1413. [Google Scholar] [CrossRef] [PubMed]
- Yoshinaka, K.; Yamaguchi, A.; Matsumura, R.; Node, K.; Tokuda, I.; Akashi, M. Effect of different light–dark schedules on estrous cycle in mice, and implications for mitigating the adverse impact of night work. Genes Cells 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Endo, A.; Watanabe, T. Effects of non-24-hour days on reproductive efficacy and embryonic development in mice. Gamete Res. 1989, 22, 435–441. [Google Scholar] [CrossRef] [PubMed]
- McCORMACK, C.E. Acute effects of altered photoperiods on the onset of ovulation in gonadotropin-treated immature rats. Endocrinology 1973, 93, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Varcoe, T.J.; Wight, N.; Voultsios, A.; Salkeld, M.D.; Kennaway, D.J. Chronic Phase Shifts of the Photoperiod throughout Pregnancy Programs Glucose Intolerance and Insulin Resistance in the Rat. PLoS ONE 2011, 6, e18504. [Google Scholar] [CrossRef]
- Varcoe, T.J.; Boden, M.J.; Voultsios, A.; Salkeld, M.D.; Rattanatray, L.; Kennaway, D.J. Characterisation of the maternal response to chronic phase shifts during gestation in the rat: Implications for fetal metabolic programming. PLoS ONE 2013, 8, e53800. [Google Scholar] [CrossRef]
- Summa, K.C.; Vitaterna, M.H.; Turek, F.W. Environmental perturbation of the Circadian clock disrupts pregnancy in the mouse. PLoS ONE 2012, 7, e37668. [Google Scholar] [CrossRef]
- Moline, M.L.; Albers, H.E. Response of circadian locomotor activity and the proestrous luteinizing hormone surge to phase shifts of the light-dark cycle in the hamster. Physiol. Behav. 1988, 43, 435–440. [Google Scholar] [CrossRef]
- Roseboom, P.H.; Namboodiri, M.A.A.; Zimonjic, D.B.; Popescu, N.C.; Rodriguez, I.R.; Gastel, J.A.; Klein, D.C. Natural melatoninknockdown’in C57BL/6J mice: Rare mechanism truncates serotonin N-acetyltransferase. Mol. Brain Res. 1998, 63, 189–197. [Google Scholar] [CrossRef]
- Kondratova, A.A.; Kondratov, R. V The circadian clock and pathology of the ageing brain. Nat. Rev. Neurosci. 2012, 13, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.J.; Nakamura, W.; Yamazaki, S.; Kudo, T.; Cutler, T.; Colwell, C.S.; Block, G.D. Age-related decline in circadian output. J. Neurosci. 2011, 31, 10201–10205. [Google Scholar] [CrossRef] [PubMed]
- Weinert, D. Age-dependent changes of the circadian system. Chronobiol. Int. 2000, 17, 261–283. [Google Scholar] [CrossRef]
- Pilorz, V.; Steinlechner, S. Low reproductive success in Per1 and Per2 mutant mouse females due to accelerated ageing? Reproduction 2008, 135. [Google Scholar] [CrossRef]
- Vitaterna, M.H.; Ko, C.H.; Chang, A.-M.; Buhr, E.D.; Fruechte, E.M.; Schook, A.; Antoch, M.P.; Turek, F.W.; Takahashi, J.S. The mouse Clock mutation reduces circadian pacemaker amplitude and enhances efficacy of resetting stimuli and phase–response curve amplitude. Proc. Natl. Acad. Sci. USA 2006, 103, 9327–9332. [Google Scholar] [CrossRef]
- Schwartz, W.J.; Zimmerman, P. Circadian timekeeping in BALB/c and C57BL/6 inbred mouse strains. J. Neurosci. 1990, 10, 3685–3694. [Google Scholar] [CrossRef]
- Pittendrigh, C.S.; Daan, S. A functional analysis of circadian pacemakers in nocturnal rodents. II. The variability of phase response curves. J Comp Physiol 1976, 106, 253–266. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Z.; Wang, F.; Tian, X.; Ji, P.; Liu, G. Effects of melatonin administration on embryo implantation and offspring growth in mice under different schedules of photoperiodic exposure. Reprod. Biol. Endocrinol. 2017, 15, 1–9. [Google Scholar] [CrossRef]
- Motta-Teixeira, L.C.; Machado-Nils, A.V.; Battagello, D.S.; Diniz, G.B.; Andrade-Silva, J.; Silva, S.; Matos, R.A.; do Amaral, F.G.; Xavier, G.F.; Bittencourt, J.C.; et al. The absence of maternal pineal melatonin rhythm during pregnancy and lactation impairs offspring physical growth, neurodevelopment, and behavior. Horm. Behav. 2018, 105. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.J.; Sellix, M.T.; Kudo, T.; Nakao, N.; Yoshimura, T.; Ebihara, S.; Colwell, C.S.; Block, G.D. Influence of the estrous cycle on clock gene expression in reproductive tissues: Effects of fluctuating ovarian steroid hormone levels. Steroids 2010, 75, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Loh, D.H.; Kuljis, D.A.; Azuma, L.; Wu, Y.; Truong, D.; Wang, H.B.; Colwell, C.S. Disrupted reproduction, estrous cycle, and circadian rhythms in female mice deficient in vasoactive intestinal peptide. J. Biol. Rhythms 2014, 29, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Yaw, A.M.; Duong, T.V.; Nguyen, D.; Hoffmann, H.M. Circadian rhythms in the mouse reproductive axis during the estrous cycle and pregnancy. J. Neurosci. Res. 2020, 99. [Google Scholar] [CrossRef]
- Alvarez, J.D.; Hansen, A.; Ord, T.; Bebas, P.; Chappell, P.E.; Giebultowicz, J.M.; Williams, C.; Moss, S.; Sehgal, A. The circadian clock protein BMAL1 is necessary for fertility and proper testosterone production in mice. J. Biol. Rhythms 2008, 23. [Google Scholar] [CrossRef]
- Dolatshad, H.; Campbell, E.A.; O’Hara, L.; Maywood, E.S.; Hastings, M.H.; Johnson, M.H.; O’hara, L.; Maywood, E.S.; Hastings, M.H.; Johnson, M.H. Developmental and reproductive performance in circadian mutant mice. Hum. Reprod. 2006, 21, 68–79. [Google Scholar] [CrossRef]
- Kennaway, D.J.; Boden, M.J.; Voultsios, A. Reproductive performance in female ClockΔ19 mutant mice. Reprod. Fertil. Dev. 2004, 16. [Google Scholar] [CrossRef]
- Lv, S.; Wang, N.; Ma, J.; Li, W.; Chen, Z.; Zhang, C. Impaired decidualization caused by downregulation of circadian clock gene BMAL1 contributes to human recurrent miscarriage †. Biol. Reprod. 2019, 4519. [Google Scholar] [CrossRef]
- Sadek, K.; Bruce, K.; Macklon, N.; Cagampang, F.; Cheong, Y. O-239 Clock gene expression in the human endometrium: Impact of phase of menstrual cycle, bmi and sleep. Hum. Reprod. 2012, 27, ii93–ii95. [Google Scholar]
- Panda, S.; Hogenesch, J.B.; Kay, S.A. Circadian rhythms from flies to human. Nature 2002, 417. [Google Scholar] [CrossRef]
- Connolly, M.S.; Lynch, C.B. Circadian variation of strain differences in body temperature and activity in mice. Physiol. Behav. 1981, 27, 1045–1049. [Google Scholar] [CrossRef]
- Possidente, B.; Stephan, F.K. Circadian period in mice: Analysis of genetic and maternal contributions to inbred strain differences. Behav. Genet. 1988, 18, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ebihara, S.; Tsuji, K.; Kondo, K. Strain differences of the mouse’s free-running circadian rhythm in continuous darkness. Physiol. Behav. 1978, 20, 795–799. [Google Scholar] [CrossRef]
| Publication | Demo-Graphic Location | Mean Age (years) | Study Size (Number of) | Variables | Results | ||
|---|---|---|---|---|---|---|---|
| Rotating Shifts | Night Work | Control | |||||
| [4] | United States | 28–45 | 13,349 | 57,728 | Menstrual Cycle Length | Shiftwork associated with cycles <21 d or >40 d | |
| Menstrual Cycle Regularity | Increased risk of irregular menstrual cycles for women of ages 28–30 and 41–45 and women with continuous rotating schedule | ||||||
| [6] | Midwest United States | 29.97 ± 0.6 | 68 | N/A | N/A | Menstrual Cycle Length | 22% of shiftwork nurses had a change in menstrual cycle length, including shortened, lengthened, and varied |
| Dysmenorrhea | 18% of shiftwork nurses had an increase in pain | ||||||
| Changes in Menstrual Cycle | 53% of shiftwork nurses reported some type of change to their menstrual cycle | ||||||
| Sleep | Decrease in sleep, increase in number of awakenings from sleep, 31% of reported menstrual changes also had increase in sleep disturbances | ||||||
| [5] | Taiwan | 27.7 ± 5.3 | 50 | 12 | 72 | Menstrual Cycle Regularity | 35% of shiftwork nurses had irregular menstrual cycle |
| Menstrual Cycle Length | 60% of fixed nights shifts and 21.7% of rotating shifts had cycles <25 d, no long cycles | ||||||
| Dysmenorrhea | 30% regularly experienced pain with menstrual cycle | ||||||
| [82] | Taiwan | >17 | 280 | 49 | Menstrual Cycle Length | Increased risk of short (9.7% vs. 6.0%; p = 0.004) and prolonged cycles (26.0% vs. 17.9%; p = 0.04) for shiftwork | |
| Menstrual Cycle Regularity | Increased risk of irregularity (MCL <25 days or >35 days) for shiftwork | ||||||
| [83] | China | 21–46 | 334 | 139 | Menstrual Cycle Length | Significantly higher proportion of nurses with irregularity for shift workers | |
| Menstrual Cycle Regularity | Having a cycle of 25–31 days decreased from 81.7% to 67.8% after changing to shiftwork | ||||||
| [84] | Spain | <40 | 113 | N/A | 75 | Menstrual Disorders | No increased risk of having menstrual disorders (duration, dysmenorrhea, excessive bleeding) |
| [7] | Taiwan | 27.7 | 50 | 29 | 72 | Ovarian Cycle Regularity | 52.6% of rotating shiftwork nurses had irregular ovarian cycle patterns |
| Dysmenorrhea | 30% had regular dysmenorrhea | ||||||
| [10] | Taiwan | Daytime Work: 26.7 ± 6.3, Intermittent Rotating Work: 30.2 ± 8.7, Persistent Rotating Work: 27.5 ± 8.5 | 303 | N/A | 137 | Childbearing Rate | Decrease in childbearing rates for rotating shift workers |
| Birthweight | Persistent rotating work decreased birth weight by 250 g. Thirty five percent of babies born were in the lightest quintile birth weight (1950–2834 g) | ||||||
| [85] | Australia | N/A | N/A | 11,000 | 84,991 | Fertility | Night shift workers ≤35 years more likely to require fertility treatment, no associations among women >35 years; among women who did fertility treatment, night shift workers were more likely than day workers to have menstrual irregularity or endometriosis |
| [86] | United States | 35 | 36 | 426 | Fertility | Evening/night/rotating shifts had close to 25% less mature oocytes | |
| [87] | Sweden | <29–35+ | 457 | 100 | 189 | Fertility | Night shift, two-shift, or three-shift rotation workers had decreased fertility |
| [88] | United States | 34.9 ± 2.7 | N/A | 196 | 364 | Fecundity | Women who reported ever working night shifts had 20% lower fecundability compared with those who never reported night shift work |
| [89] | United States and Canada | 33 | 344 | 298 | 1006 | Fecundity | No association between types of shift and duration of pregnancy attempt |
| [90] | Denmark | <25–35+ | 4763 | 809 (evening) 290 (night) | 24,605 | Fecundity | Fixed night and evenings had longer time to pregnancy |
| [11] | United States | 38 | 673 (flight attendants) | 91 (teachers) | Miscarriage | Increased risk for flight attendants who flew 15+ h during normal sleeping period from 10 PM to 8 AM | |
| [13] | Denmark | 30.5 ± 3.9 | N/A | 10,047 | 12,697 | Miscarriage | Dose-dependent relationship between increased risk and successive night shifts, miscarriage rate for weeks 3–21 of pregnancy was lower for night shift group |
| [12] | United States | 25–42 | 1766 | 680 | 5242 | Miscarriage | Early miscarriage (<12 weeks): no increased risk for rotating shiftwork, 60% increase for nightshifts. Late miscarriage (12–20 weeks): 80% increase for nights only and 50% increase for rotating shiftwork, no nights |
| [91] | China | 29.5 | N/A | 1860 | N/A | Miscarriage | Working night shifts was independently associated with increased risk of miscarriage |
| [92] | Canada | N/A | 90 | 75 (evening) 11 (night) | 700 | Miscarriage | Four times increased risk of pregnancy loss with evening work and more than twice as high for fixed nights |
| [93] | United States | 25–42 | 1709 | 664 | 5109 | Miscarriage | Failed pregnancies more likely from working night shift and long hours during first trimester |
| [94] | Denmark | <25–35+ | 6577 | 420 | 33,694 | Miscarriage | Fixed night shifts associated with fetal loss |
| [95] | Sweden | <29–35+ | 1571 | 367 | 567 | Miscarriage | Increased odds ratio for night shift and three-shift workers, increased odds ratio of late miscarriage for night shift workers |
| [96] | Sweden | N/A | 44 (rotating) 285 (irregular) | 11 (evening) 202 (night) | 215 | Miscarriage | Rotating shifts had slight increased risk of miscarriage |
| Birthweight | Irregular work hours had significantly lower birth weights | ||||||
| [14] | United States | 32.1 ± 4.5 | N/A | N/A | 131 | Labor | Mean labor duration with: severe sleep disturbance, 26 h; normal sleep disturbance, 18.3 h; less than 6 h of sleep per night, 29 h; more than 7 h of sleep per night (normal), 17.7 h |
| Delivery Type | Cesarean rate with: severe sleep disturbance, 39%; normal sleep disturbance, 10.3%; less than 6 h of sleep per night, 36.8%; more than 7 h of sleep per night (normal), 10.8% | ||||||
| [17] | Denmark | N/A | 3197 | 400 | 32,465 | Birthweight | Fixed nights: decrease 30 g in birthweight. Rotating shiftwork: no significant change |
| Term Birth | No preterm association for rotating shiftwork, high risk of post-term for night workers | ||||||
| [97] | Canada | <20–29+ | 660 (changing shifts) | N/A | 22,101 | Birthweight | Increased risk for fatal ovarian cancer among rotating shift workers, but nightshift had no association |
| Term Birth | Changing shift work less strongly associated with preterm birth | ||||||
| [16] | China | 23.8 ± 2.5 | 127 | N/A | 38 | Birthweight | All live birth: −79 g for rotating shiftwork. Production workers only: −109 g for rotating shiftwork |
| Term Birth | All live birth incidence: 20% rotating shiftwork, 15.1% regular schedule. Production workers only incidence: 20.2% rotating shiftwork, 15.6% regular schedule | ||||||
| [98] | South Asia | 26.4 ± 5.5 | 74 | 690 | Birthweight | Shiftwork was a risk factor for small for gestational age less than 5th centile | |
| [99] | Ireland | 16–44 | 133 | 529 | Birthweight | Working 40+ hours per week and shiftwork was associated with a birthweight of 3000 g or less | |
| [100] | Canada | <25–>35 | N/A | 177 | 900 | Birthweight | Irregular shiftwork schedule increased risk for small for gestational age |
| [101] | Finland | N/A | 368 | N/A | 1979 | Birthweight | Increased number of small for gestational age for rotating shiftwork |
| [102] | United States | >16 | N/A | 166 | 1630 | Term Birth | Night shifts have a 50% increased risk of preterm birth |
| [103] | Brazil | N/A | N/A | 134 | 2164 | Term Birth | No correlation with spontaneous preterm birth and nightshift |
| [104] | England | 30.3 ± 3.8 | N/A | 131 | 1196 | Term Birth | Preterm birth had little association with long working hours or shift work |
| [105] | Systematic review and meta-analysis | Term Birth | Night shifts or shifts not significantly associated with increased risk of preterm birth | ||||
| [106] | Denmark | 30.8 | N/A | 10,203 | 6298 | Term Birth | No increased risk of preterm birth for night shift, risk not related to number, duration, or consecutive night shifts; women changing from night shifts in the first trimester to day work only in the second trimester had weak increased risk of preterm birth |
| [107] | Meta-analysis | Term Birth | Shiftwork and night shifts significantly associated with preterm birth | ||||
| [108] | United States | 35.5 ± 4.7 | 18,635 | 62,205 | Menopause | Increased risk of early menopause. Women <45 years with exposure of 11–20+ years of rotating night shiftwork have 22% increased risk for early menopause | |
| [109] | United States | 30–55 | 46,801 | 31,761 | Breast Cancer | Premenopausal: (years of shiftwork) 1–14 years had 23% increased risk 15+ years had 30% increased risk Postmenopausal: 30+ years had 45% increased risk | |
| [110] | United States | 30–55 | 31,442 | 22,045 | Endometrial Cancer | 20+ years of shiftwork had 47% increased risk, 2-fold increased risk for shiftwork women with a BMI of 30+ | |
| [111] | United States | 35–74 | N/A | 831 | 2491 | Invasive Ovarian Cancer | Any nightshift: 1.24-fold increased risk |
| Borderline Ovarian Tumors | Any nightshift: 1.48-fold increased risk | ||||||
| [112] | United States | 50.3 | 10,552 | 1754 | 141,637 | Ovarian Cancer | Increased risk for fatal ovarian cancer among rotating shift workers, but nightshift had no association |
| [113] | United States | 30–55 (NHS) and 25–42 (NHS11) | 15,928 | 55,369 | Ovarian Cancer | Rotating night shiftwork duration saw no association | |
| [114] | United States | 18–49 | N/A | 198 | 229 | Endometriosis | Night shift work had 50% increased risk of endometriosis; risk doubled when women worked more than half of the shifts at night |
| [115] | United States | 42.4 ± 3.7 | 419 | 244 | Hormones | Recent night work (within last 2 weeks) had 56% decrease in urinary melatonin, long-term rotating shiftwork is associated with increased estradiol in postmenopausal women, no effect on progesterone levels | |
| [116] | Spain | 43.4 ± 12.15 | 63 | 73 | Hormones | Rotating night shifts increased estradiol and progesterone levels, decreased testosterone levels, no significant difference in cortisol levels | |
| [117] | Austria and Germany | 24.2 | 10 subjects put under partial sleep deprivation | Hormones | LH, estriol, and thyroid-stimulating hormone levels increased, thyroid-stimulating hormone increasing significantly and remaining at high levels following sleep deprivation | ||
| [118] | United States | 34.5 | N/A | 172 | 151 | Hormones | Night shift workers: 6-sulfatoxymelatonin levels were lower and reproductive hormone levels were higher during daytime sleep and nighttime work, relative to nighttime sleep |
| [119] | Japan | 22–39 | 9 (3 pregnant, 6 non-pregnant) | N/A | Hormones | Both: excretion profiles of urinary 6-sulfatoxymelatonin alter on night shift; urinary estriol level was not significantly affected by shift. Pregnant: urinary norepinephrine level during the night work was considerably higher; effect of night work on urinary estriol level of the pregnant women remained uncertain | |
| [120] | United States | 24 | 77 | 103 | Hormones | Women having night/shift work had lower testosterone and increased LH relative to non-night/shift work women | |
| [121] | Poland | 40–60 | N/A | 263 | 269 | Hormones | Frequency of night shift work did not determine hormone concentrations, total duration of night work tended to be positively associated with estradiol concentration, postmenopausal women with night work >15 years had increased estradiol levels |
| Publication | Rodent Species | Strain | Control Light Cycle | Experimental Light Cycle | Variables Measured | Results |
|---|---|---|---|---|---|---|
| [155] | Mouse | C57BL/6J | Stable LD12:12 | Single 10 h phase shifts (advance/delay) and Chronic shifting (10 h advance for 3 days then 10 h delay for 4 days, for up to 9 months) | Estrous cycles, LH secretion profiles, natural fertility with 1 wk mating | Acute light shifts altered the LH surge and estrous cycling. Chronic shifting increased estrous acyclicity and pattern/amplitude of LH surge. Chronic shifting mice had reduced birth rate, number of pups/litter, and total number of pups |
| [76] | Mouse | Per2:Luciferase on C57 background | Food during dark phase | Normal; restricted food to light phase | Litter success rates, time to first litter, number of pups born and weaned, estrous cycling, mating, pregnancy maintenance | Light-restricted feeding reduced litters, reduced number of copulatory plugs, delayed time to first litter and did not change the number of pups born or weaned, estrous cycles, or pregnancy maintenance |
| [156] | Mouse | C57BL/6J Jms Slc and backcrossed with Cry1 or Cry2 KO | 3 h delay of darkness onset for 2 days and then returned to the former phase of the LD cycles for 5 days | Estrous cycles, birth success | In Cry1 and Cry2 KO, light shifts altered estrous cycles and lowered pregnancy rates in older, but not young mice | |
| [157] | Mouse | C57BL6 | LD unchanged | 12 h shift reversals for 36 days at the interval of 3, 6 or 12 days | Estrous cycles | Degree of circadian disruption was dependent on shift interval and estrous cycling was disrupted in all shift intervals |
| [158] | Mouse | Jcl:ICR | 24 h day 12:12 | 22 (L11:D11) or 26 h day (L13: D13) light/ dark cycles for at least 2 weeks before mating and/or during pregnancy | Mating rate, pregnancy rate, resorption rates, number of pups | Mice on 22 h days or 26 h days had decreased mating rates, increased resorption and reduced pup rate with no change in pregnancy rate as compared to mice on 24 h days |
| [159] | Rats | Unknown | LD12:12 | Long days (15 h light) reduced by 5 h either in AM or PM | Ovulation counts | Ovulation is adaptive to light. In the transition from long to shorter photoperiods, subtracting light from the AM (phase delay) results in delayed ovulation. Subtracting light from PM portion advanced ovulation |
| [160] | Rats | Albino Wistar | Stable LD12:12 | Complete reversal of lights every 3–4 days | Pregnancy outcomes, growth and development; Offspring: metabolic, circadian, anxiety-like and behavioral despair | No differences in gestation length, litter size, survival to weaning, or birth weight. Offspring exhibited sex-specific metabolic effects |
| [161] | Rats | Albino Wistar | Stable LD12:12 | Complete reversal of lights every 3–4 days | Pregnancy outcomes, metabolic function, circadian and metabolic gene expression | Light shifts disrupt weight gain across gestation (with similar food consumption), normal circadian melatonin, altered corticosterone, glucose, insulin, leptin, cholesterol, and free fatty acids. No change to glucose and insulin tolerance, Maternal liver clock gene expression (Bmal1 not rhythmic in shift, Per1 rhythmic but altered expression, Per2 not rhythmic, Rev-erba not rhythmic). Altered expression of metabolic genes in liver. Fetal gene expression also altered |
| [162] | Mouse | C57BL/6J | Stable LD 12:12 | 6 h advance or delay (4 times) every 5–6 days | Pregnancy outcomes, locomotor activity | Light advance group experienced the greatest reduction in the number of pregnancies caried to term. Light delays reduced the number of pregnancies caried to term as compared to no shifts |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaw, A.M.; McLane-Svoboda, A.K.; Hoffmann, H.M. Shiftwork and Light at Night Negatively Impact Molecular and Endocrine Timekeeping in the Female Reproductive Axis in Humans and Rodents. Int. J. Mol. Sci. 2021, 22, 324. https://doi.org/10.3390/ijms22010324
Yaw AM, McLane-Svoboda AK, Hoffmann HM. Shiftwork and Light at Night Negatively Impact Molecular and Endocrine Timekeeping in the Female Reproductive Axis in Humans and Rodents. International Journal of Molecular Sciences. 2021; 22(1):324. https://doi.org/10.3390/ijms22010324
Chicago/Turabian StyleYaw, Alexandra M., Autumn K. McLane-Svoboda, and Hanne M. Hoffmann. 2021. "Shiftwork and Light at Night Negatively Impact Molecular and Endocrine Timekeeping in the Female Reproductive Axis in Humans and Rodents" International Journal of Molecular Sciences 22, no. 1: 324. https://doi.org/10.3390/ijms22010324
APA StyleYaw, A. M., McLane-Svoboda, A. K., & Hoffmann, H. M. (2021). Shiftwork and Light at Night Negatively Impact Molecular and Endocrine Timekeeping in the Female Reproductive Axis in Humans and Rodents. International Journal of Molecular Sciences, 22(1), 324. https://doi.org/10.3390/ijms22010324






