Curcumin as an Anticancer Agent in Malignant Mesothelioma: A Review
Abstract
1. Malignant Mesothelioma: A Growing Health Emergency
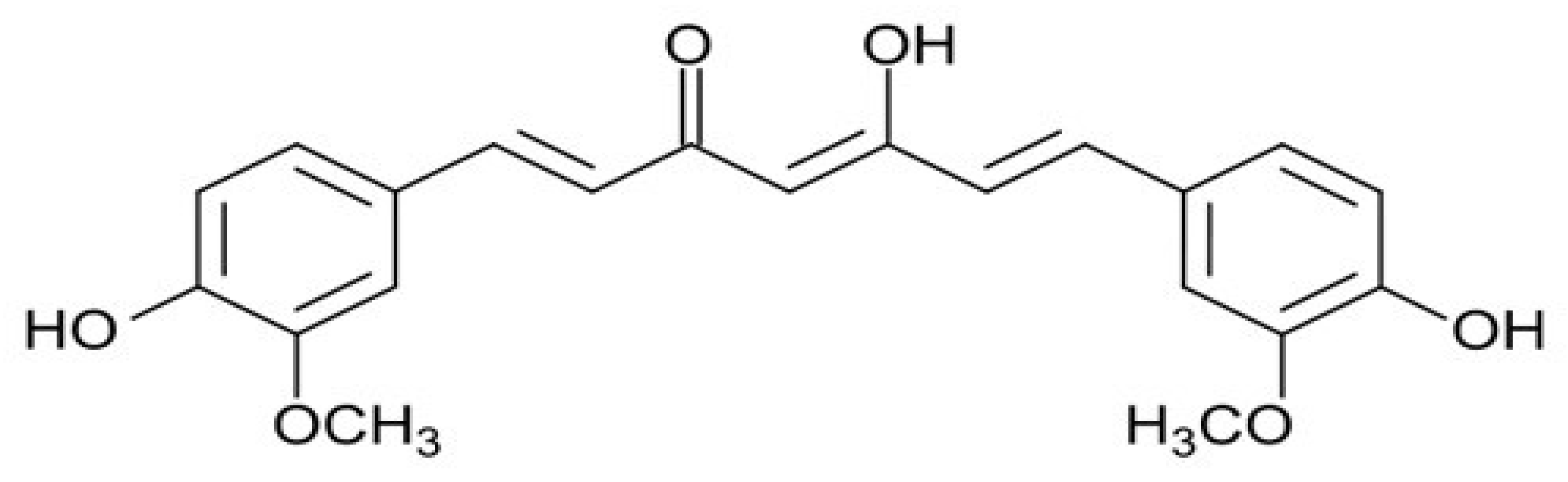
2. Phytochemicals as Anti-Cancer Molecules: The Example of Curcumin
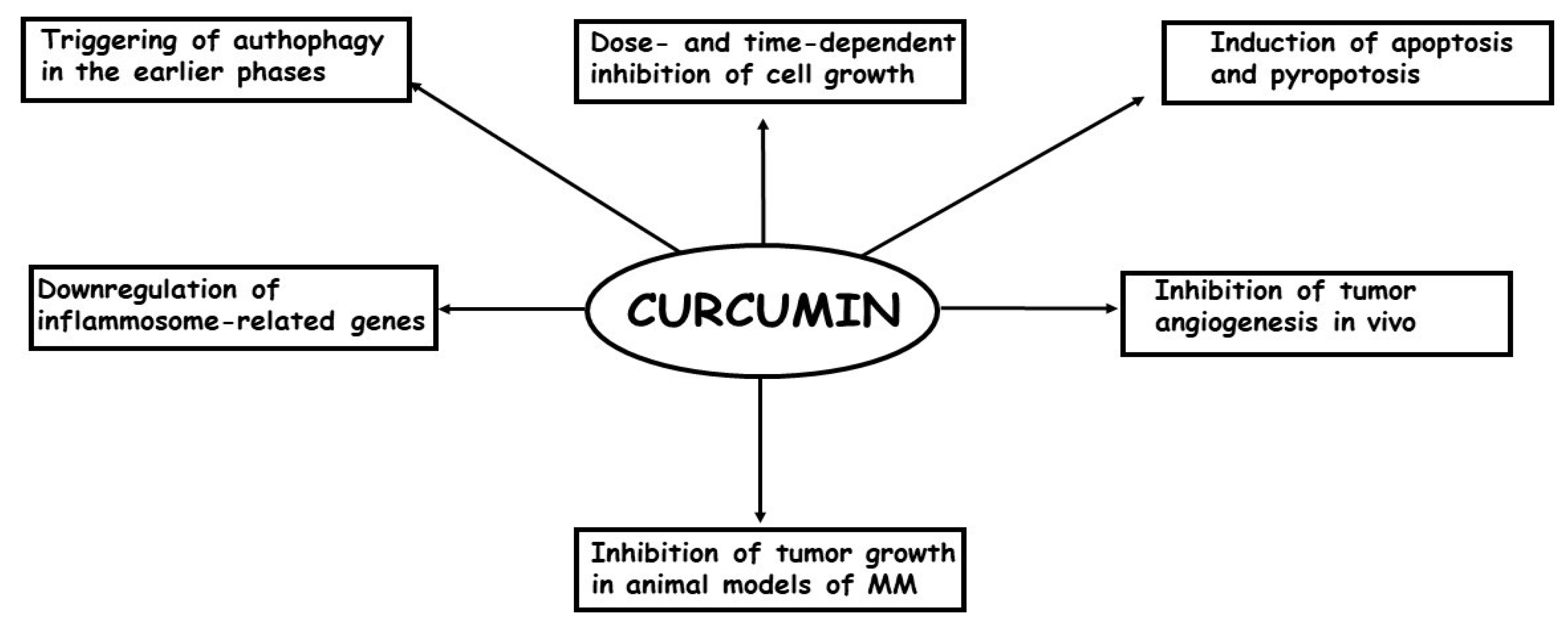
3. Laboratory and Clinical Studies Carried Out on the Effects of Curcumin in Malignant Mesothelioma
| First Author (Year) [ref] | Study Design | MM Cell Lines | Main Results |
|---|---|---|---|
| Yamauchi (2012) [63] | Laboratory study | ACC-MESO-1 | curcumin was effective in reducing in dose-dependent manner cell viability through to autophagy |
| Miller (2012) [64] | Laboratory study | HMESO, H2595 | growth-suppressive activity of H2461 curcumin on MM cells was through induction of pyroptosis and protection against inflammation |
| Serri (2017) [65] | Laboratory study | MSTO-211H | curcumin induced a persistent block in G0/G1 phase of the cell cycle up to 72 h, thus overwhelming the drug tolerance phenomenon |
| Masuelli (2017) [66] | Laboratory study | MM-B1, H-Meso-1, MM-F1 and #40a | curcumin inhibited cell survival in an earlier phase by triggering autophagic flux, but later on by activating apoptosis |
| Pouliquen (2017) [69] | Laboratory study | M5-T1 | intracavitary administration of curcumin was able to significantly decrease the tumor mass in a rat orthotopic model of sarcomatoid MM |
| Zhang (2018) [70] | Laboratory study | RN5 | curcumin was able to induce apoptosis via the mitochondrial pathway and caspase-independent and apoptosis-inducing factor dependent pathways |
| Di Meo (2019) [71] | Laboratory study | MSTO-221H, NCI-H2452 | curcumin-C3complex®/Bioperine® induced growth inhibition by apoptosis in all MM cell lines examined in a dose- and time-depended manner and reduced cell migration and cell invasive ability |
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MM | Malignant mesothelioma |
References
- Treasure, T. The learning curve. BMJ 2004, 329, 424. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Crispi, S.; Cardillo, I.; Spugnini, E.P.; Citro, G.; Menegozzo, S.; Baldi, A. Biological agents involved in malignant mesothelioma: Relevance as biomarkers or therapeutic targets. Curr. Cancer Drug Targets 2010, 10, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Sugarbaker, P.H. Update on the management of malignant peritoneal mesothelioma. Transl. Lung Cancer Res. 2018, 7, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Spugnini, E.P.; Cardillo, I.; Verdina, A.; Crispi, S.; Saviozzi, S.; Calogero, R.; Nebbioso, A.; Altucci, L.; Cortese, G.; Galati, R.; et al. Piroxicam and cisplatin in a mouse model of peritoneal mesothelioma. Clin. Cancer Res. 2006, 12, 6133–6143. [Google Scholar] [CrossRef]
- Hammar, S.P. Macroscopic, Histologic, Immunohistochemical and Ultrastructural Features of Mesothelioma. Ultrastruct. Pathol. 2006, 30, 3–17. [Google Scholar] [CrossRef]
- Galateau-Salle, F.; Churg, A.; Roggli, V.; Travis, W.D. World Health Organization Committee for Tumors of the Pleura. The 2015 World Health Organization Classification of Tumors of the Pleura: Advances since the 2004 Classification. J. Thorac. Oncol. 2016, 11, 142–154. [Google Scholar] [CrossRef]
- Husain, A.N.; Colby, T.V.; Ordóñez, N.G.; Allen, T.C.; Attanoos, R.L.; Beasley, M.B.; Butnor, K.J.; Chirieac, L.R.; Churg, A.M.; Dacic, S.; et al. Guidelines for Pathologic Diagnosis of Malignant Mesothelioma 2017 Update of the Consensus Statement from the International Mesothelioma Interest Group. Arch. Pathol. Lab. Med. 2018, 142, 89–108. [Google Scholar] [CrossRef]
- Hotta, K.; Fujimoto, N. Current evidence and future perspectives of immune-checkpoint inhibitors in unresectable malignant pleural mesothelioma. J. Immunother. Cancer 2020, 8, e000461. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Bosari, S.; Citro, G.; Lorenzon, I.; Cognetti, F.; Baldi, A. Human malignant mesothelioma: Molecular mechanisms of pathogenesis and progression. Int. J. Biochem. Cell Biol. 2006, 38, 2000–2004. [Google Scholar] [CrossRef]
- Vogelzang, N.J.; Rusthoven, J.J.; Symanowski, J.; Denham, C.; Kaukel, E.; Ruffie, P.; Gatzemeier, U.; Boyer, M.; Emri, S.; Manegold, C.; et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J. Clin. Oncol. 2003, 21, 2636–2644. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Crispi, S.; Scarabello, A.; Caruso, G.; Citro, G.; Baldi, A. Piroxicam and intracavitary platinum-based chemotherapy for the treatment of advanced mesothelioma in pets: Preliminary observations. J. Exp. Clin. Cancer Res. 2008, 27, 6. [Google Scholar] [CrossRef]
- Zervos, M.D.; Bizekis, C.; Pass, H.I. Malignant mesothelioma 2008. Curr. Opin. Pulm. Med. 2008, 14, 303–309. [Google Scholar] [CrossRef]
- Behling, C.A.; Wolf, P.L.; Haghighi, P. AIDS and malignant mesothelioma--is there a connection? Chest 1993, 103, 1268–1269. [Google Scholar] [CrossRef]
- Stoppoloni, D.; Canino, C.; Cardillo, I.; Verdina, A.; Baldi, A.; Sacchi, A.; Galati, R. Synergistic effect of gefitinib and rofecoxib in mesothelioma cells. Mol. Cancer 2010, 9, 27. [Google Scholar] [CrossRef]
- Crispi, S.; Fagliarone, C.; Biroccio, A.; D’Angelo, C.; Galati, R.; Sacchi, A.; Vincenzi, B.; Baldi, A.; Verdina, A. Antiproliferative effect of Aurora kinase targeting in mesothelioma. Lung Cancer 2010, 70, 271–279. [Google Scholar] [CrossRef]
- Stoppoloni, D.; Salvatori, L.; Biroccio, A.; D’Angelo, C.; Muti, P.; Verdina, A.; Sacchi, A.; Vincenzi, B.; Baldi, A.; Galati, R. Aromatase inhibitor exemestane has antiproliferative effects on human mesothelioma cells. J. Thorac. Oncol. 2011, 6, 583–591. [Google Scholar] [CrossRef]
- Baldi, A.; Piccolo, M.T.; Boccellino, M.R.; Donizetti, A.; Cardillo, I.; La Porta, R.; Quagliuolo, L.; Spugnini, E.P.; Cordero, F.; Citro, G.; et al. Apoptosis induced by piroxicam plus cisplatin combined treatment is triggered by p21 in mesothelioma. PLoS ONE 2011, 6, e23569. [Google Scholar] [CrossRef]
- Coppola, N.; Perna, A.; Lucariello, A.; Martini, S.; Macera, M.; Carleo, M.A.; Guerra, G.; Esposito, V.; De Luca, A. Effects of treatment with Maraviroc a CCR5 inhibitor on a human hepatic stellate cell line. J. Cell. Physiol. 2018, 233, 6224–6231. [Google Scholar] [CrossRef]
- Scherpereel, A.; Wallyn, F.; Albelda, S.M.; Munck, C. Novel therapies for malignant pleural mesothelioma. Lancet Oncol. 2018, 19, e161–e172. [Google Scholar] [CrossRef]
- Esposito, V.; Perna, A.; Lucariello, A.; Carleo, M.A.; Viglietti, R.; Sangiovanni, V.; Coppola, N.; Guerra, G.; De Luca, A.; Chirianni, A. Different impact of antiretroviral drugs on bone differentiation in an in vitro model. J. Cell. Biochem. 2015, 116, 2188–2194. [Google Scholar] [CrossRef]
- Zito Marino, F.; Ascierto, P.A.; Rossi, G.; Staibano, S.; Montella, M.; Russo, D.; Alfano, R.; Morabito, A.; Botti, G.; Franco, R. Are tumor-infiltrating lymphocytes protagonists or background actors in patient selection for cancer immunotherapy? Expert Opin. Biol. Ther. 2017, 17, 735–746. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Campioni, M.; D’Avino, A.; Caruso, G.; Citro, G.; Baldi, A. Cell-cycle molecules in mesothelioma: An overview. J. Exp. Clin. Cancer Res. 2007, 26, 443–449. [Google Scholar]
- Ambrogi, V.; Baldi, A.; Schillaci, O.; Mineo, T.C. Clinical impact of extrapleural pneumonectomy for malignant pleural mesothelioma. Ann. Surg. Oncol. 2012, 19, 1692–1699. [Google Scholar] [CrossRef]
- Piccolo, M.T.; Menale, C.; Crispi, S. Combined anticancer therapies: An overview of the latest applications. Anticancer Agents Med. Chem. 2015, 15, 408–422. [Google Scholar] [CrossRef]
- Damiano, S.; Lombari, P.; Salvi, E.; Papale, M.; Giordano, A.; Amenta, M.; Ballistreri, G.; Fabroni, S.; Rapisarda, P.; Capasso, G.; et al. A red orange and lemon by-products extract rich in anthocyanins inhibits the progression of diabetic nephropathy. J. Cell. Physiol. 2019, 234, 23268–23278. [Google Scholar] [CrossRef]
- Damiano, S.; Iovane, V.; Squillacioti, C.; Mirabella, N.; Prisco, F.; Ariano, A.; Amenta, M.; Giordano, A.; Florio, S.; Ciarcia, R. Red orange and lemon extract prevents the renal toxicity induced by ochratoxin A in rats. J. Cell. Physiol. 2020, 235, 5386–5393. [Google Scholar] [CrossRef]
- Ho, J.W.; Cheung, M.W. Combination of phytochemicals as adjuvants for cancer therapy. Recent Pat. Anticancer Drug Discov. 2014, 9, 297–302. [Google Scholar] [CrossRef]
- Barone, D.; Cito, L.; Tommonaro, G.; Abate, A.A.; Penon, D.; De Prisco, R.; Penon, A.; Forte, I.M.; Benedetti, E.; Cimini, A.; et al. Antitumoral potential, antioxidant activity and carotenoid content of two Southern Italy tomato cultivars extracts: San Marzano and Corbarino. J. Cell. Physiol. 2018, 233, 1266–1277. [Google Scholar] [CrossRef]
- Nuvoli, B.; Santoro, R.; Catalani, S.; Battistelli, S.; Benedetti, S.; Canestrari, F.; Galati, R. CELLFOOD induces apoptosis in human mesothelioma and colorectal cancer cells by modulating p53, c-myc and pAkt signaling pathways. J. Exp. Clin. Cancer Res. 2014, 33, 24. [Google Scholar] [CrossRef]
- Priyadarsini, K.I. The chemistry of curcumin: From extraction to therapeutic agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef]
- Prasad, S.; Gupta, S.C.; Tyagi, A.K.; Aggarwal, B.B. curcumin, a component of golden spice: From bedside to bench and back. Biotechnol. Adv. 2014, 32, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Kocaadam, B.; Sanlier, N. Curcumin, an active component of turmeric (Curcuma longa) and its effects on health. Crit. Rev. Food Sci. Nutr. 2015, 57, 2889–2895. [Google Scholar] [CrossRef] [PubMed]
- Unlu, A.; Nayir, E.; Kalenderoglu, M.D.; Kirca, O.; Ozdogan, M. Curcumin (Turmeric) and cancer. J. BUON 2016, 21, 1050–1060. [Google Scholar] [PubMed]
- Singh, S. From exotic spice to modern drug? Cell 2007, 130, 765–768. [Google Scholar] [CrossRef] [PubMed]
- Hatcher, H.; Planalp, R.; Cho, J.; Torti, F.M.; Torti, S.V. Curcumin: From ancient medicine to current clinical trials. Cell. Mol. Life Sci. 2008, 65, 1631–1652. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Huang, X.; Zhang, H.M.; Ren, P. Pharmacokinetic comparison of puerarin after oral administration of Jiawei-Xiaoyao-San to healthy volunteers and patients with functional dyspepsia: Influence of disease state. J. Pharm. Pharmacol. 2009, 61, 125–129. [Google Scholar] [CrossRef]
- Esposito, T.; Lucariello, A.; Hay, E.; Contieri, M.; Tammaro, P.; Varriale, B.; Guerra, G.; De Luca, A.; Perna, A. Effects of curcumin and its adjuvant on TPC1 thyroid cell line. Chem. Biol. Interact. 2019, 305, 112–118. [Google Scholar] [CrossRef]
- Perna, A.; De Luca, A.; Adelfi, L.; Pasquale, T.; Varriale, B.; Esposito, T. Effects of different extracts of curcumin on TPC1 papillary thyroid cancer cell line. BMC Complement. Altern. Med. 2018, 18, 63. [Google Scholar] [CrossRef]
- Hay, E.; Lucariello, A.; Contieri, M.; Esposito, T.; De Luca, A.; Guerra, G.; Perna, A. Therapeutic effects of turmeric in several diseases: An overview. Chem. Biol. Interact. 2019, 310, 108729. [Google Scholar] [CrossRef]
- Zhou, J.; Miao, H.; Li, X.; Hu, Y.; Sun, H.; Hou, Y. Curcumin inhibits placental inflammation to ameliorate LPS-induced adverse pregnancy outcomes in mice via upregulation of phosphorylated Akt. Inflamm. Res. 2017, 66, 177–185. [Google Scholar] [CrossRef]
- Lucariello, A.; Trabucco, E.; Boccia, O.; Perna, A.; Sellitto, C.; Castaldi, M.A.; De Falco, M.; De Luca, A.; Cobellis, L. Small leucine rich proteoglycans are differently distributed in normal and pathological endometrium. In Vivo 2015, 29, 217–222. [Google Scholar] [PubMed]
- Hongyu, Z.; Beevers, C.S.; Huang, S. Targets of curcumin. Curr. Drug Targets 2011, 12, 332–347. [Google Scholar]
- Kuttan, R.; Bhanumathy, P.; Nirmala, K.; George, M.C. Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett. 1985, 29, 197–202. [Google Scholar] [CrossRef]
- Jessay, G.D.; Ifeanyi, D.N.; Peter, J.H.J. curcumin and cancer: Barriers to obtaining a health claim. Nutr. Rev. 2015, 73, 155–165. [Google Scholar]
- Bhattacharyya, S.; Mandal, D.; Saha, B.; Sen, G.S.; Das, T.; Sa, G. curcumin prevents tumor-induced T cell apoptosis through Stat-5a-mediated Bcl-2 induction. J. Biol. Chem. 2007, 282, 15954–15964. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Mandal, D.; Sen, G.S.; Pal, S.; Banerjee, S.; Lahiry, L.; Finke, J.H.; Tannenbaum, C.S.; Das, T.; Sa, G. Tumor-induced oxidative stress perturbs nuclear factor-kappaB activity-augmenting tumor necrosis factor-alpha-mediated T-cell death: Protection by curcumin. Cancer Res. 2007, 67, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.G.; Lu, J.; Holmgren, A. Thioredoxin reductase is irreversibly modified by curcumin—A novel molecular mechanism for its anticancer activity. J. Biol. Chem. 2005, 280, 25284–25290. [Google Scholar] [CrossRef]
- Limtrakul, P.; Chearwae, W.; Shukla, S.; Phisalphong, C.; Ambudkar, S.V. Modulation of function of three ABC drug transporters, P-glycoprotein (ABCB1), mitoxantrone resistance protein (ABCG2) and multidrug resistance protein 1 (ABCC1) by tetrahydrocurcumin, a major metabolite of curcumin. Mol. Cell. Biochem. 2007, 296, 85–95. [Google Scholar] [CrossRef]
- Howells, L.M.; Sale, S.; Sriramareddy, S.N.; Irving, G.R.; Jones, D.J.; Ottley, C.J.; Pearson, D.G.; Mann, C.D.; Manson, M.M.; Berry, D.P.; et al. Curcumin ameliorates oxaliplatin-induced chemoresistance in HCT116 colorectal cancer cells in vitro and in vivo. Int. J. Cancer 2011, 129, 476–486. [Google Scholar] [CrossRef]
- Toden, S.; Okugawa, Y.; Jascur, T.; Wodarz, D.; Komarova, N.L.; Buhrmann, C.; Shakibaei, M.; Boland, C.R.; Goel, A. curcumin mediates chemosensitization to 5-fluorouracil through miRNA-induced suppression of epithelial-to-mesenchymal transition in chemoresistant colorectal cancer. Carcinogenesis 2015, 36, 355–367. [Google Scholar] [CrossRef]
- Zhang, P.; Lai, Z.L.; Chen, H.F.; Zhang, M.; Wang, A.; Jia, T.; Sun, W.Q.; Zhu, X.M.; Chen, X.F.; Zhao, Z.; et al. Curcumin synergizes with 5-fluorouracil by impairing AMPK/ULK1-dependent autophagy, AKT activity and enhancing apoptosis in colon cancer cells with tumor growth inhibition in xenograft mice. J. Exp. Clin. Cancer Res. 2017, 36, 190. [Google Scholar] [CrossRef] [PubMed]
- Fetoni, A.R.; Paciello, F.; Mezzogori, D.; Rolesi, R.; Eramo, S.L.; Paludetti, G.; Troiani, D. Molecular targets for anticancer redox chemotherapy and cisplatin-induced ototoxicity: The role of curcumin on pSTAT3 and Nrf-2 signalling. Br. J. Cancer 2015, 113, 1434–1444. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Wang, D.; Zou, D.; Wang, C.; Lopes-Bastos, B.; Jiang, W.G.; Chester, J.; Zhou, Q.; Cai, J. Re-purposing of curcumin as an anti-metastatic agent for the treatment of epithelial ovarian cancer: In vitro model using cancer stem cell enriched ovarian cancer spheroids. Oncotarget 2016, 7, 86374–86387. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Sundaram, C.; Jhurani, S.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin and cancer: An “old-age” disease with an “age-old” solution. Cancer Lett. 2008, 267, 133–164. [Google Scholar] [CrossRef]
- Park, W.; Amin, A.R.; Chen, Z.G.; Shin, D.M. New perspectives of curcumin in cancer prevention. Cancer Prev. Res. 2013, 6, 387–400. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, W.H.; Chen, B.L.; Fan, L.; Han, Y.; Wang, G.; Hu, D.L.; Tan, Z.R.; Zhou, G.; Cao, S.; et al. Plant polyphenol curcumin significantly affects CYP1A2 and CYP2A6 activity in healthy, male Chinese volunteers. Ann. Pharmacother. 2010, 44, 1038–1045. [Google Scholar] [CrossRef]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X. A Review of curcumin and Its Derivatives as Anticancer Agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef]
- Hussain, Z.; Thu, H.E.; Ng, S.F.; Khan, S.; Katas, H. Nanoencapsulation, an efficient and promising approach to maximize wound healing efficacy of curcumin: A review of new trends and state-of-the-art. Colloids Surf. B Biointerfaces 2017, 150, 223–241. [Google Scholar] [CrossRef]
- Mayol, L.; Serri, C.; Menale, C.; Crispi, S.; Piccolo, M.T.; Mita, L.; Giarra, S.; Forte, M.; Saija, A.; Biondi, M.; et al. Curcumin loaded PLGA-poloxamer blend nanoparticles induce cell cycle arrest in mesothelioma cells. Eur. J. Pharm. Biopharm. 2015, 93, 37–45. [Google Scholar] [CrossRef]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Rishi, A.K.; Wu, W.; Polin, L.; Sharma, S.; Levi, E.; Albelda, S.; Pass, H.I.; Wali, A. Curcumin suppresses growth of mesothelioma cells in vitro and in vivo, in part, by stimulating apoptosis. Mol. Cell. Biochem. 2011, 357, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, Y.; Izumi, Y.; Asakura, K.; Hayashi, Y.; Nomori, H. Curcumin induces autophagy in ACC-MESO-1 cells. Phytother. Res. 2012, 26, 1779–1783. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.M.; Thompson, J.K.; MacPherson, M.B.; Beuschel, S.L.; Westbom, C.M.; Sayan, M.; Shukla, A. Curcumin: A double hit on malignant mesothelioma. Cancer Prev. Res. 2014, 7, 330–340. [Google Scholar] [CrossRef]
- Serri, C.; Argiro, M.; Piras, L.; Mita, D.G.; Saija, A.; Mita, L.; Forte, M.; Giarra, S.; Biondi, M.; Crispi, S.; et al. Nano-precipitated curcumin loaded particles: Effect of carrier size and drug complexation with (2-hydroxypropyl)-beta-cyclodextrin on their biological performances. Int. J. Pharm. 2017, 520, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Masuelli, L.; Benvenuto, M.; Di Stefano, E.; Mattera, R.; Fantini, M.; De Feudis, G.; De Smaele, E.; Tresioldi, I.; Giganti, M.G.; Modesti, A.; et al. Curcumin blocks autophagy and activates apoptosis of malignant mesothelioma cell lines and increases the survival of mice intraperitoneally transplanted with a malignant mesothelioma cell line. Oncotarget 2017, 8, 34405–34422. [Google Scholar] [CrossRef]
- Mariño, G.; Niso-Santano, M.; Baehrecke, E.H.; Kroemer, G. Self-consumption: The interplay of autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2014, 15, 81–94. [Google Scholar] [CrossRef]
- Maiuri, M.C.; Zalckvar, E.; Kimchi, A.; Kroemer, G. Self-eating and self-killing: Crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2007, 8, 741–752. [Google Scholar] [CrossRef]
- Pouliquen, D.L.; Nawrocki-Raby, B.; Nader, J.; Blandin, S.; Robard, M.; Birembaut, P.; Gregoire, M. Evaluation of intracavitary administration of curcumin for the treatment of sarcomatoid mesothelioma. Oncotarget 2017, 8, 57552–57573. [Google Scholar] [CrossRef][Green Version]
- Zhang, C.; Hao, Y.; Wu, L.; Dong, X.; Jiang, N.; Cong, B.; Liu, J.; Zhang, W.; Tang, D.; De Perrot, M.; et al. Curcumin induces apoptosis and inhibits angiogenesis in murine malignant mesothelioma. Int. J. Oncol. 2018, 53, 2531–2541. [Google Scholar] [CrossRef]
- Di Meo, F.; Filosa, S.; Madonna, M.; Giello, G.; Di Pardo, A.; Maglione, V.; Baldi, A.; Crispi, S. curcumin C3 complex®/Bioperine® has antineoplastic activity in mesothelioma: An in vitro and in vivo analysis. J. Exp. Clin. Cancer Res. 2019, 38, 360. [Google Scholar] [CrossRef] [PubMed]
- Panahi, Y.; Khalili, N.; Hosseini, M.S.; Abbasinazari, M.; Sahebkar, A. Lipid-modifying effects of adjunctive therapy with curcuminoids-piperine combination in patients with metabolic syndrome: Results of a randomized controlled trial. Complement. Ther. Med. 2014, 22, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Giordano, A.; Tommonaro, G. Curcumin and Cancer. Nutrients 2019, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Spugnini, E.P.; Fais, S.; Azzarito, T.; Baldi, A. Novel Instruments for the Implementation of Electrochemotherapy Protocols: From Bench Side to Veterinary Clinic. J. Cell. Physiol. 2017, 232, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Spugnini, E.P.; Melillo, A.; Quagliuolo, L.; Boccellino, M.; Vincenzi, B.; Pasquali, P.; Baldi, A. Definition of novel electrochemotherapy parameters and validation of their in vitro and in vivo effectiveness. J. Cell. Physiol. 2014, 229, 1177–1181. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Baldi, A. Electrochemotherapy in Veterinary Oncology: State-of-the-Art and Perspectives. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 967–979. [Google Scholar] [CrossRef]
- Spugnini, E.P.; Azzarito, T.; Fais, S.; Fanciulli, M.; Baldi, A. Electrochemotherapy as First Line Cancer Treatment: Experiences from Veterinary Medicine in Developing Novel Protocols. Curr. Cancer Drug Targets 2016, 16, 43–52. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldi, A.; De Luca, A.; Maiorano, P.; D’Angelo, C.; Giordano, A. Curcumin as an Anticancer Agent in Malignant Mesothelioma: A Review. Int. J. Mol. Sci. 2020, 21, 1839. https://doi.org/10.3390/ijms21051839
Baldi A, De Luca A, Maiorano P, D’Angelo C, Giordano A. Curcumin as an Anticancer Agent in Malignant Mesothelioma: A Review. International Journal of Molecular Sciences. 2020; 21(5):1839. https://doi.org/10.3390/ijms21051839
Chicago/Turabian StyleBaldi, Alfonso, Antonio De Luca, Patrizia Maiorano, Costantino D’Angelo, and Antonio Giordano. 2020. "Curcumin as an Anticancer Agent in Malignant Mesothelioma: A Review" International Journal of Molecular Sciences 21, no. 5: 1839. https://doi.org/10.3390/ijms21051839
APA StyleBaldi, A., De Luca, A., Maiorano, P., D’Angelo, C., & Giordano, A. (2020). Curcumin as an Anticancer Agent in Malignant Mesothelioma: A Review. International Journal of Molecular Sciences, 21(5), 1839. https://doi.org/10.3390/ijms21051839







