The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application
Abstract
1. Aromatherapy and Essential oils
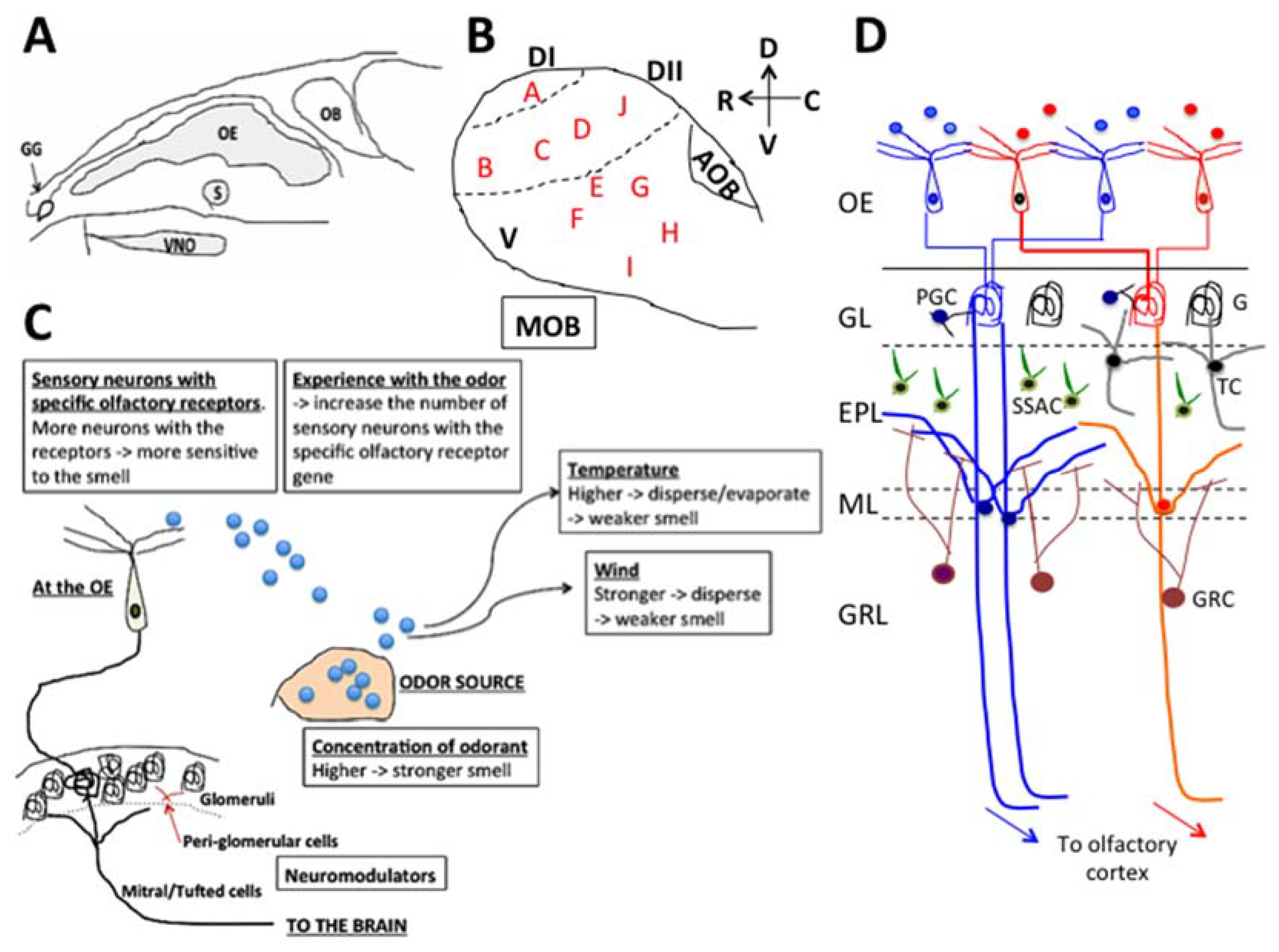
2. Olfactory System
3. The Effects of Essential Oils and Terpenes Through the Olfactory System
4. The Effects of Essential Oils and Terpenes Through the Olfactory Receptors Expressed in the Non-Olfactory System
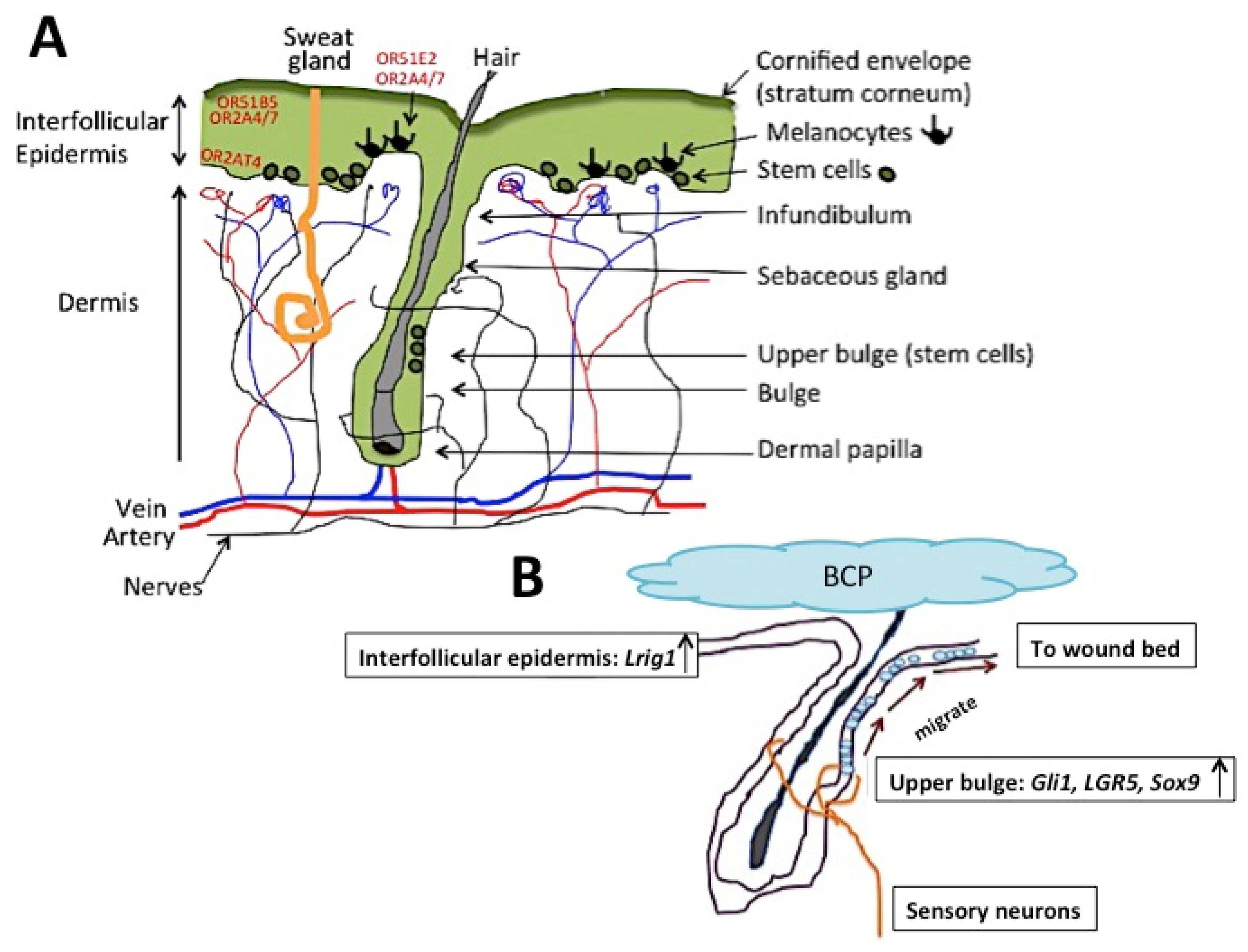
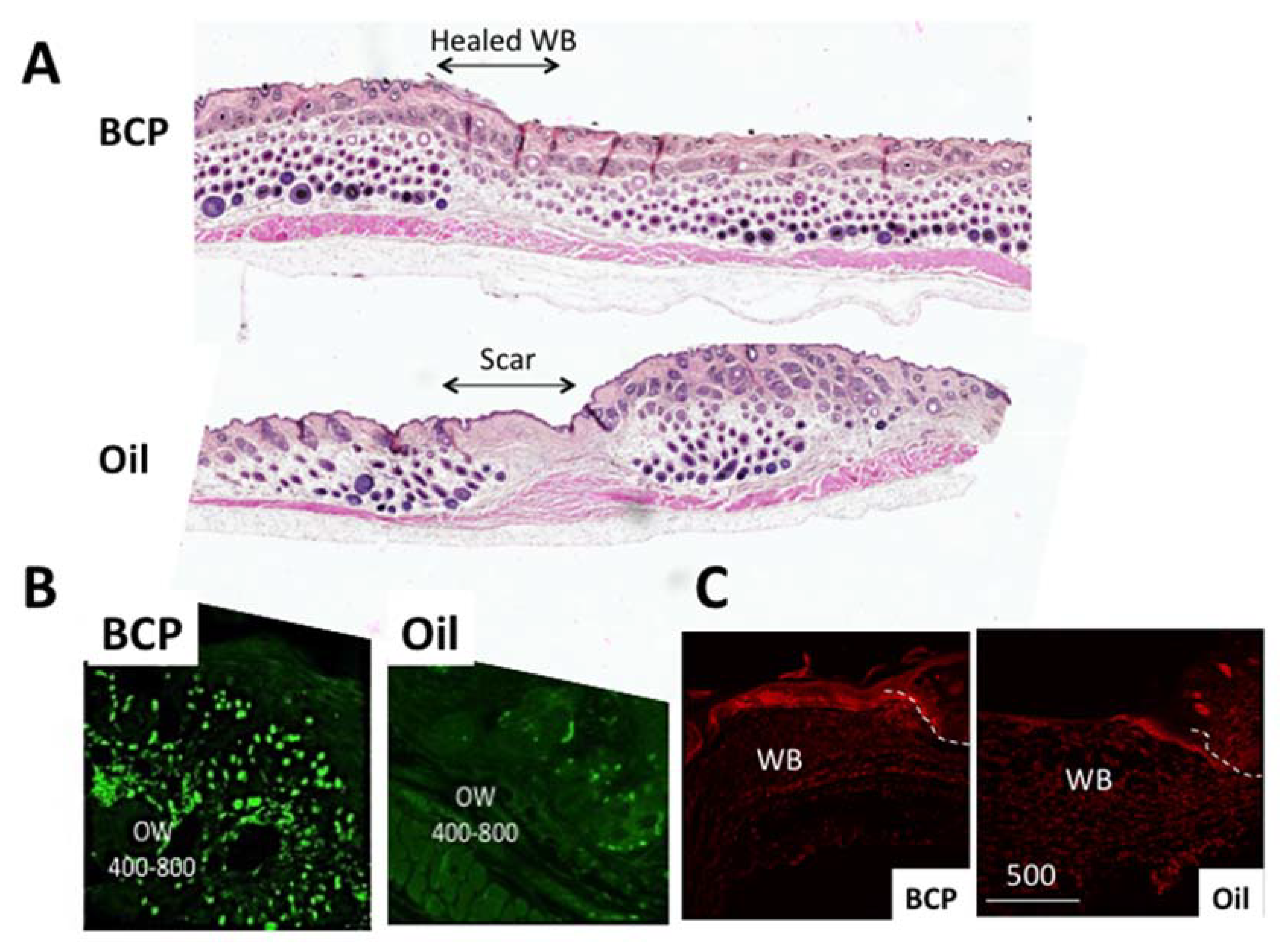
4.1. The Skin
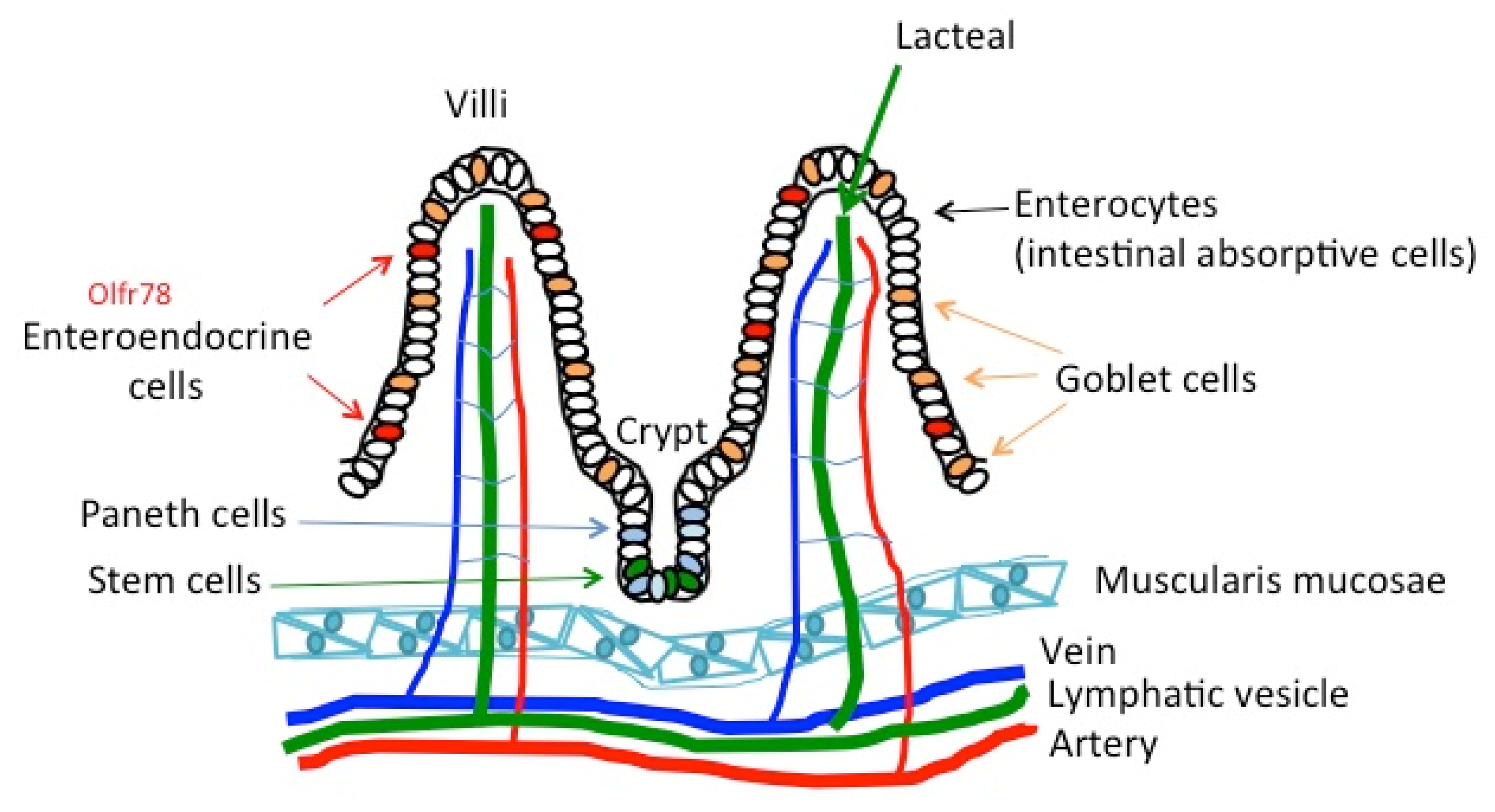
4.2. The Gut and Intestine
5. The Effects of Essential Oils and Terpenes through Non-Olfactory Receptors
6. Psychological and Physiological Impact of Essential Oils
7. The Receptors and Neuromodulators
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CAP | Compound action potentials |
| CB2 | Cannabinoid receptor 2 |
| GABA | Gamma aminobutyric acid |
| TRP | Transient receptor potential channels |
| TRPA | Transient receptor potential channels subfamily A |
| TRPM | Transient receptor potential channel metastatin |
| TRPV | Transient receptor potential channel vanilloid |
| MAPK | Mitogen-activated protein kinase |
| AKT | Protein kinase B |
| Chk-2 | Checkpoint kinase 2 |
| IL-1 | Interleukin 1 |
| IL-6 | Interleukin 6 |
| Hsp27 | Heat shock protein 27 |
| AMPK1 | Adenosine Monophosphate-activated protein kinase 1 |
| P38MAPK | protein kinases and the 38-kDa mitogen-activated protein kinase |
| Wnt | Wingless related integration site |
| Gli1 | Glioma associated oncogene |
| Adamts | Disintegrin and metalloproteinase with thrombospondin motifs |
| Bambi | bone morphogenetic protein and activin membrane-bound inhibitor homolog |
| Msx2 | Msh homeobox2 |
| Dlx3 | Distalless 3 |
| Padi1 | peptidyl arginine deiminase 1 |
| Hoxc13 | Homeobox C13 |
| S100a | a family of calcium-binding cytosolic proteins |
| BrdU | Bromodeoxyuridine |
| Lgr5 | Leucine-rich repeat-containing G-protein coupled receptor 5 |
| IP | Intra-peritoneal |
| CNS | Central nervous system |
| 5-HT | 5-hydroxytryptamine |
| TBPS | Tert-butylbicyclophosphorothionate; GABA receptor antagonist |
| EEG | electroencephalography |
| AOB | Accessory olfactory bulb |
| MOB | Main olfactory bulb |
| NG | Necklace glomeruli |
| OCAM | Olfactory cell adhesion molecule |
| GC-D | Receptor guanylyl cyclase |
| TAAR | Trace amine-associated receptors |
| TMT | Trimethyl-thiazoline |
| SBT | 2-sec-tutyl-dihydrothiazole |
| DHB | dehydro-exo-brevicomine |
References
- Gattefossé, R.-M. Gattefossé’s Aromatherapy; C. W. Daniell Co., Ltd.: Essex, England, 1993. [Google Scholar]
- Koyama, S.; Purk, A.; Kaur, M.; Soini, H.A.; Novotny, M.V.; Davis, K.; Kao, C.C.; Matsunami, H.; Mescher, A. Beta-caryophyllene enhances wound healing through multiple routes. PLoS ONE 2019, 14, e0216104. [Google Scholar] [CrossRef]
- Harada, H.; Kashiwadani, H.; Kanmura, Y.; Kuwaki, T. Linalool Odor-Induced Anxiolytic Effects in Mice. Front. Behav. Neurosci. 2018, 12, 241. [Google Scholar] [CrossRef] [PubMed]
- Maßberg, D.; Hatt, H. Human Olfactory Receptors: Novel Cellular Functions Outside of the Nose. Physiol. Rev. 2018, 98, 1739–1763. [Google Scholar] [CrossRef] [PubMed]
- Gertsch, J.; Leonti, M.; Raduner, S.; Rácz, I.; Chen, J.-Z.; Xie, X.-Q.; Altmann, K.-H.; Karsak, M.; Zimmer, A. Beta-caryophyllene is a dietary cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [PubMed]
- De Groot, A.C.; Schmidt, E. Essential Oils, Part III: Chemical Composition. Dermatitis 2016, 27, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Bushdid, C.; Magnasco, M.O.; Vosshall, L.B.; Keller, A. Humans Can Discriminate More than 1 Trillion Olfactory Stimuli. Science 2014, 343, 1370–1372. [Google Scholar] [CrossRef]
- Nagayama, S.; Homma, R.; Imamura, F. Neuronal organization of olfactory bulb circuits. Front. Neural Circuits 2014, 8. [Google Scholar] [CrossRef]
- Buck, L.; Axel, R. A novel multigene family may encode odorant receptors: A molecular basis for odor recognition. Cell 1991, 65, 175–187. [Google Scholar] [CrossRef]
- Mombaerts, P. Molecular Biology of Odorant Receptors In Vertebrates. Annu. Rev. Neurosci. 1999, 22, 487–509. [Google Scholar] [CrossRef]
- Mombaerts, P. Seven-Transmembrane Proteins as Odorant and Chemosensory Receptors. Science 1999, 286, 707–711. [Google Scholar] [CrossRef]
- Firestein, S. How the olfactory system makes sense of scents. Nature 2001, 413, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Ache, B.W.; Young, J.M. Olfaction: Diverse Species, Conserved Principles. Neuron 2005, 48, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Breer, H.; Fleischer, J.; Strotmann, J. The sense of smell: Multiple olfactory subsystems. Cell. Mol. Life Sci. 2006, 63, 1465–1475. [Google Scholar] [CrossRef]
- Ma, M. Multiple Olfactory Subsystems Convey Various Sensory Signals. In The Neurobiology of Olfaction; Menini, A., Ed.; CRC: Boca Raton, FL, USA, 2010. [Google Scholar]
- Fülle, H.J.; Vassar, R.; Foster, D.C.; Yang, R.-B.; Axel, R.; Garbers, D.L. A receptor guanylyl cyclase expressed specifically in olfactory sensory neurons. Proc. Natl. Acad. Sci. USA 1995, 92, 3571–3575. [Google Scholar] [CrossRef] [PubMed]
- Liberles, S.D.; Buck, L.B. A second class of chemosensory receptors in the olfactory epithelium. Nature 2006, 442, 645–650. [Google Scholar] [CrossRef] [PubMed]
- Liberles, S.D. Trace Amine-associated Receptors Are Olfactory Receptors in Vertebrates. Ann. N. Y. Acad. Sci. 2009, 1170, 168–172. [Google Scholar] [CrossRef]
- Ackels, T.; Von Der Weid, B.; Rodriguez, I.; Spehr, M.; Von Der Weid, B. Physiological characterization of formyl peptide receptor expressing cells in the mouse vomeronasal organ. Front. Neuroanat. 2014, 8, 134. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Von Campenhause, H.; Yoshihara, Y. Zonal organization of the mammalian main and accessory olfactory systems. Philos. Trans. R. Soc. B Boil. Sci. 2000, 355, 1801–1812. [Google Scholar] [CrossRef]
- Miyamichi, K.; Serizawa, S.; Kimura, H.M.; Sakano, H. Continuous and Overlapping Expression Domains of Odorant Receptor Genes in the Olfactory Epithelium Determine the Dorsal/Ventral Positioning of Glomeruli in the Olfactory Bulb. J. Neurosci. 2005, 25, 3586–3592. [Google Scholar] [CrossRef]
- Fuss, S.H.; Omura, M.; Mombaerts, P. The Grueneberg ganglion of the mouse projects axons to glomeruli in the olfactory bulb. Eur. J. Neurosci. 2005, 22, 2649–2654. [Google Scholar] [CrossRef]
- Koos, D.S.; Fraser, S.E. The Grueneberg ganglion projects to the olfactory bulb. NeuroReport 2005, 16, 1929–1932. [Google Scholar] [CrossRef] [PubMed]
- Zufall, F.; Munger, S.D. Receptor guanylyl cyclases in mammalian olfactory function. Mol. Cell. Biochem. 2009, 334, 191–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Levai, O.; Strotmann, J. Projection pattern of nerve fibers from the septal organ: DiI-tracing studies with transgenic OMP mice. Histochem. Cell Boil. 2003, 120, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Yoshihara, Y.; Kawasaki, M.; Tamada, A.; Fujita, H.; Hayashi, H.; Kagamiyama, H.; Mori, K. OCAM: A New Member of the Neural Cell Adhesion Molecule Family Related to Zone-to-Zone Projection of Olfactory and Vomeronasal Axons. J. Neurosci. 1997, 17, 5830–5842. [Google Scholar] [CrossRef] [PubMed]
- Tsuboi, A.; Miyazaki, T.; Imai, T.; Sakano, H. Olfactory sensory neurons expressing class I odorant receptors converge their axons on an antero-dorsal domain of the olfactory bulb in the mouse. Eur. J. Neurosci. 2006, 23, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Bozza, T.; Vassalli, A.; Fuss, S.H.; Zhang, J.-J.; Weiland, B.; Pacifico, R.; Feinstein, P.; Mombaerts, P. Mapping of Class I and Class II Odorant Receptors to Glomerular Domains by Two Distinct Types of Olfactory Sensory Neurons in the Mouse. Neuron 2009, 61, 220–233. [Google Scholar] [CrossRef] [PubMed]
- Kobayakawa, K.; Kobayakawa, R.; Matsumoto, H.; Oka, Y.; Imai, T.; Ikawa, M.; Okabe, M.; Ikeda, T.; Itohara, S.; Kikusui, T.; et al. Innate versus learned odour processing in the mouse olfactory bulb. Nature 2007, 450, 503–508. [Google Scholar] [CrossRef]
- Mori, K.; Takahashi, Y.K.; Igarashi, K.; Yamaguchi, M. Maps of Odorant Molecular Features in the Mammalian Olfactory Bulb. Physiol. Rev. 2006, 86, 409–433. [Google Scholar] [CrossRef]
- Mori, K.; Sakano, H. How is the olfactory map formed and interpreted in the mammalian brain? Annu. Rev. Neurosci. 2011, 34, 467–499. [Google Scholar] [CrossRef]
- Scott, J.W.; Brierley, T. A functional map in rat olfactory epithelium. Chem. Senses 1999, 24, 679–690. [Google Scholar] [CrossRef]
- Grosmaitre, X.; Fuss, S.H.; Lee, A.C.; Adipietro, K.A.; Matsunami, H.; Mombaerts, P.; Ma, M. SR1, a mouse odorant receptor with an unusually broad response profile. J. Neurosci. 2009, 29, 14545–14552. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Soria-Gomez, E.; Bellocchio, L.; Reguero, L.; Lepousez, G.; Martin, C.; Bendahmane, M.; Ruehle, S.; Remmers, F.; Desprez, T.; Matias, I.; et al. The endocannabinoid system controls food intake via olfactory processes. Nat. Neurosci. 2014, 17, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Koyama, S. Primer Effects by Murine Pheromone Signaling; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Tyler, W.; Petzold, G.C.; Pal, S.K.; Murthy, V. Experience-Dependent Modification of Primary Sensory Synapses in the Mammalian Olfactory Bulb. J. Neurosci. 2007, 27, 9427–9438. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dias, B.G.; Ressler, K.J. Parental olfactory experience influences behavior and neural structure in subsequent generations. Nat. Neurosci. 2013, 17, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Parma, V.; Ferraro, S.; Miller, S.S.; Åhs, F.; Lundström, J.N. Enhancement of Odor Sensitivity Following Repeated Odor and Visual Fear Conditioning. Chem. Senses 2015, 40, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.M.; Fletcher, M.L. Olfactory fear conditioning non-specifically enhances glomerular odor responses and increases representational overlap of learned and neutral odors. J. Neurosci. 2018, 38, 4623–4640. [Google Scholar] [CrossRef]
- Dey, S.; Chamero, P.; Pru, J.K.; Chien, M.-S.; Ibarra-Soria, X.; Spencer, K.R.; Logan, D.W.; Matsunami, H.; Peluso, J.J.; Stowers, L. Cyclic Regulation of Sensory Perception by a Female Hormone Alters Behavior. Cell 2015, 161, 1334–1344. [Google Scholar] [CrossRef]
- Harvey, J.D.; Heinbockel, T. Neuromodulation of Synaptic Transmission in the Main Olfactory Bulb. Int. J. Environ. Res. Public Heal. 2018, 15, 2194. [Google Scholar] [CrossRef]
- Price, J.L.; Powell, T.P. The morphology of the granule cells of the olfactory bulb. J. Cell Sci. 1970, 7, 91–123. [Google Scholar]
- Moss, M.; Cook, J.; Wesnes, K.; Duckett, P. Aromas of Rosemary and Lavender Essential Oils Differentially Affect Cognition and Mood in Healthy Adults. Int. J. Neurosci. 2003, 113, 15–38. [Google Scholar] [CrossRef]
- Sayorwan, W.; Ruangrungsi, N.; Piriyapunyporn, T.; Hongratanaworakit, T.; Kotchabhakdi, N.; Siripornpanich, V. Effects of Inhaled Rosemary Oil on Subjective Feelings and Activities of the Nervous System. Sci. Pharm. 2012, 81, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Moss, M.; Hewitt, S.; Moss, L.; Wesnes, K. Modulation of Cognitive Performance and Mood by Aromas of Peppermint and Ylang-Ylang. Int. J. Neurosci. 2008, 118, 59–77. [Google Scholar] [CrossRef] [PubMed]
- Miroddi, M.; Navarra, M.; Quattropani, M.C.; Calapai, F.; Gangemi, S.; Calapai, G. Systematic Review of Clinical Trials Assessing Pharmacological Properties of Salvia Species on Memory, Cognitive Impairment and Alzheimer’s Disease. CNS Neurosci. Ther. 2014, 20, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Villareal, M.O.; Ikeya, A.; Sasaki, K.; Ben Arfa, A.; Neffati, M.; Isoda, H. Anti-stress and neuronal cell differentiation induction effects of Rosmarinus officinalis L. essential oil. BMC Complement. Altern. Med. 2017, 17, 549. [Google Scholar] [CrossRef]
- Ueno, H.; Shimada, A.; Suemitsu, S.; Murakami, S.; Kitamura, N.; Wani, K.; Matsumoto, Y.; Okamoto, M.; Ishihara, T. Anti-depressive-like effect of 2-phenylethanol inhalation in mice. Biomed. Pharmacother. 2019, 111, 1499–1506. [Google Scholar] [CrossRef]
- Özcan, M.M.; Chalchat, J.-C. Chemical composition and antifungal activity of rosemary (Rosmarinus officinalis L.) oil from Turkey. Int. J. Food Sci. Nutr. 2008, 59, 691–698. [Google Scholar] [CrossRef]
- Angioni, A.; Barra, A.; Cereti, E.; Barile, D.; Coisson, J.D.; Arlorio, M.; Dessi, S.; Coroneo, V.; Cabras, P. Chemical Composition, Plant Genetic Differences, Antimicrobial and Antifungal Activity Investigation of the Essential Oil ofRosmarinus officinalisL. J. Agric. Food Chem. 2004, 52, 3530–3535. [Google Scholar] [CrossRef]
- Lakusic, D.; Ristić, M.; Slavkovska, V.; Lakusić, B. Seasonal variations in the composition of the essential oils of rosemary (Rosmarinus officinalis, Lamiaceae). Nat. Prod. Commun. 2013, 8, 131–134. [Google Scholar]
- Moss, M.; Oliver, L. Plasma 1,8-cineole correlates with cognitive performance following exposure to rosemary essential oil aroma. Ther. Adv. Psychopharmacol. 2012, 2, 103–113. [Google Scholar] [CrossRef]
- Parmentier, M.; Libert, F.; Schurmans, S.; Schiffmann, S.; Lefort, A.; Eggerickx, D.; Ledent, C.; Mollereau, C.; Gérard, C.; Perret, J.; et al. Expression of members of the putative olfactory receptor gene family in mammalian germ cells. Nature 1992, 355, 453–455. [Google Scholar] [CrossRef]
- Spehr, M.; Gisselmann, G.; Poplawski, A.; Riffell, J.A.; Wetzel, C.; Zimmer, R.K.; Hatt, H. Identification of a Testicular Odorant Receptor Mediating Human Sperm Chemotaxis. Science 2003, 299, 2054–2058. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Yomogida, K.; Okabe, M.; Touhara, K. Functional characterization of a mouse testicular olfacory receptor and its role in chemosensing and in regulatin of sperm motility. J. Cell Sci. 2004, 117, 5835–5845. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, N.; Touhara, K. Developmental expression patterns of testicular olfactoryreceptor genes during mouse spermatogenesis. Genes Cells 2006, 11, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-H.; Yoon, Y.C.; Lee, A.S.; Kang, N.; Koo, J.; Rhyu, M.-R.; Park, J.H. Expression of human olfactory receptor 10J5 in heart aorta, coronary artery, and endothelial cells and its functional role in angiogenesis. Biochem. Biophys. Res. Commun. 2015, 460, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Pluznick, J.L.; Zou, N.-J.; Zhang, X.; Yan, Q.; Rodriguez-Gil, D.J.; Eisner, C.; Wells, E.; Greer, C.A.; Wang, T.; Firestein, S.; et al. Functional expression of the olfactory signaling system in the kidney. Proc. Natl. Acad. Sci. USA 2009, 106, 2059–2064. [Google Scholar] [CrossRef] [PubMed]
- Busse, D.; Kudella, P.; Grüning, N.-M.; Gisselmann, G.; Ständer, S.; Luger, T.; Jacobsen, F.; Steinsträßer, L.; Paus, R.; Gkogkolou, P.; et al. A Synthetic Sandalwood Odorant Induces Wound-Healing Processes in Human Keratinocytes via the Olfactory Receptor OR2AT4. J. Investig. Dermatol. 2014, 134, 2823–2832. [Google Scholar] [CrossRef]
- Braun, T.; Voland, P.; Kunz, L.; Prinz, C.; Gratzl, M. Enterochromaffin Cells of the Human Gut: Sensors for Spices and Odorants. Gastroenterology 2007, 132, 1890–1901. [Google Scholar] [CrossRef]
- Fleischer, J.; Bumbalo, R.; Bautze, V.; Strotmann, J.; Breer, H. Expression of odorant receptor Olfr78 in enteroendocrine cells of the colon. Cell Tissue Res. 2015, 361, 697–710. [Google Scholar] [CrossRef]
- Kim, K.-S.; Lee, I.-S.; Kim, K.-H.; Park, J.; Kim, Y.; Choi, J.-H.; Choi, J.-S.; Jang, H.-J. Activation of intestinal olfactory receptor stimulates glucagon-like peptide-1 secretion in enteroendocrine cells and attenuates hyperglycemia in type 2 diabetic mice. Sci. Rep. 2017, 7, 13978. [Google Scholar] [CrossRef]
- Chéret, J.; Bertolini, M.; Ponce, L.; Lehmann, J.; Tsai, T.; Alam, M.; Hatt, H.; Paus, R. Olfactory receptor OR2AT4 regulates human hair growth. Nat. Commun. 2018, 9, 3624. [Google Scholar] [CrossRef]
- Ito, M.; Liu, Y.; Yang, Z.; Nguyen, J.; Liang, F.; Morris, R.J.; Cotsarelis, G. Stem cells in the hair follicle bulge contribute to wound repair but not to homeostasis of the epidermis. Nat. Med. 2005, 11, 1351–1354. [Google Scholar] [CrossRef] [PubMed]
- Gelis, L.; Jovancevic, N.; Veitinger, S.; Mandal, B.; Arndt, H.-D.; Neuhaus, E.; Hatt, H. Functional Characterization of the Odorant Receptor 51E2 in Human Melanocytes. J. Boil. Chem. 2016, 291, 17772–17786. [Google Scholar] [CrossRef] [PubMed]
- Pavan, B.; Dalpiaz, A. Odorants could elicit repair processes in melanized neuronal and skin cells. Neural Regen. Res. 2017, 12, 1401–1404. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.; Veitinger, S.; Peek, I.; Busse, D.; Eckardt, J.; Vladimirova, D.; Jovancevic, N.; Wojcik, S.; Gisselmann, G.; Altmuller, J.; et al. Two olfactory receptors-OR2A4/7 and OR51B5-differentially affect epidermal proliferation and differentiation. Exp. Dermatol. 2017, 26, 58–65. [Google Scholar] [CrossRef]
- Amorim, J.L.; Figueiredo, J.D.B.; Amaral, A.C.F.; Barros, E.G.D.O.; Palmero, C.; Mpalantinos, M.A.; Ramos, A.D.S.; Ferreira, J.L.P.; Silva, J.; Benjamim, C.; et al. Wound healing properties of Copaifera paupera in diabetic mice. PLoS ONE 2017, 12, e0187380. [Google Scholar] [CrossRef]
- Abe, Y.; Tanaka, N. Roles of the hedgehog signaling pathway in epidermal and hair fillicle development, homeostasis, and cancer. J. Dev. Biol. 2017, 5, 12. [Google Scholar] [CrossRef]
- Brownell, I.; Guevara, E.; Bai, C.B.; Loomis, C.A.; Joyner, A.L. Nerve-Derived Sonic Hedgehog Defines a Niche for Hair Follicle Stem Cells Capable of Becoming Epidermal Stem Cells. Cell Stem Cell 2011, 8, 552–565. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Fan, G.; Ren, J.; Zhang, L.; Pan, S. Effects of orange essential oil on intestinal microflora in mice. J. Sci. Food Agric. 2019, 99, 4019–4028. [Google Scholar] [CrossRef]
- Krautwurst, D.; Yau, K.W.; Reed, R.R. Identification of ligands for olfactory receptors by functional expression of a receptor library. Cell 1998, 95, 917–926. [Google Scholar] [CrossRef]
- Zhang, X.; Rodriguez, I.; Mombaerts, P.; Firestein, S. Odorant and vomeronasal receptor genes in two mouse genome assemblies. Genomics 2004, 83, 802–811. [Google Scholar] [CrossRef]
- Godfrey, P.A.; Malnic, B.; Buck, L.B. The mouse olfactory receptor gene family. Proc. Natl. Acad. Sci. USA 2004, 101, 2156–2161. [Google Scholar] [CrossRef] [PubMed]
- Malnic, B.; Godfrey, P.A.; Buck, L.B. The human olfactory receptor gene family. Proc. Natl. Acad. Sci. USA 2004, 101, 7205. [Google Scholar] [CrossRef] [PubMed]
- El Hage, R.; Hernandez-Sanabria, E.; Arroyo, M.C.; Props, R.; Van De Wiele, T. Propionate-Producing Consortium Restores Antibiotic-Induced Dysbiosis in a Dynamic in vitro Model of the Human Intestinal Microbial Ecosystem. Front. Microbiol. 2019, 10, 1206. [Google Scholar] [CrossRef] [PubMed]
- La Rocca, V.; Da Fonsêca, D.V.; Silva-Alves, K.S.; Ferreira-Da-Silva, F.W.; De Sousa, D.P.; Santos, P.L.; Quintans-Júnior, L.J.; Leal-Cardoso, J.H.; Almeida, R. Geraniol Induces Anti-nociceptive Effect in Mice Evaluated in Behavioural and Electrophysiological Models. Basic Clin. Pharmacol. Toxicol. 2016, 120, 22–29. [Google Scholar] [CrossRef]
- Deng, X.-Y.; Xue, J.; Li, H.-Y.; Ma, Z.-Q.; Fu, Q.; Qu, R.; Ma, S.-P. Geraniol produces antidepressant-like effects in a chronic unpredictable mild stress mice model. Physiol. Behav. 2015, 152, 264–271. [Google Scholar] [CrossRef]
- Chirumbolo, S.; Bjørklund, G. The Antinociceptive Activity of Geraniol. Basic Clin. Pharmacol. Toxicol. 2016, 120, 105–107. [Google Scholar] [CrossRef][Green Version]
- Kennedy, D.; Okello, E.; Chazot, P.L.; Howes, M.R.; Ohiomokhare, S.; Jackson, P.; Stonehouse, W.; Khan, J.; Forster, J.; Wightman, E. Volatile Terpenes and Brain Function: Investigation of the Cognitive and Mood Effects of Mentha × Piperita L Essential Oil with In Vitro Properties Relevant to Central Nervous System Function. Nutrients 2018, 10, 1029. [Google Scholar] [CrossRef]
- Premkumar, L.S. Transient Receptor Potential Channels as Targets for Phytochemicals. ACS Chem. Neurosci. 2014, 5, 1117–1130. [Google Scholar] [CrossRef]
- Zheng, J.-L.; Yu, T.-S.; Li, X.-N.; Fan, Y.-Y.; Ma, W.-X.; Du, Y.; Zhao, R.; Guan, D.-W. Cannabinoid receptor type 2 is time-dependently expressed during skin wound healing in mice. Int. J. Leg. Med. 2012, 126, 807–814. [Google Scholar] [CrossRef]
- Tóth, K.F.; Ádám, D.; Bíró, T.; Oláh, A. Cannabinoid Signaling in the Skin: Therapeutic Potential of the “C(ut)annabinoid” System. Molecules 2019, 24, 918. [Google Scholar] [CrossRef]
- Ho, J.-C.; Lee, C.-H. TRP channels in skin: From physiological implications to clinical significances. Biophysics 2015, 11, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Candi, E.; Schmidt, R.; Melino, G. The cornified envelope: A model of cell death in the skin. Nat. Rev. Mol. Cell Boil. 2005, 6, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Watt, F.E. Mammalian skin cell biology: At the interface between laboratory and clinic. Science 2014, 346, 937–940. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Dwadasi, B.S.; Rai, B.; Mitragotri, S. Effect of Chemical Permeation Enhancers on Skin Permeability: In silico screening using Molecular Dynamics simulations. Sci. Rep. 2019, 9, 1456. [Google Scholar] [CrossRef]
- Aqil, M.; Ahad, A.; Sultana, Y.; Ali, A.; Aqil, M. Status of terpenes as skin penetration enhancers. Drug Discov. Today 2007, 12, 1061–1067. [Google Scholar] [CrossRef]
- Karande, P.; Mitragotri, S. Enhancement of transdermal drug delivery via synergistic action of chemicals. Biochim. Biophys. Acta-Biomembr. 2009, 1788, 2362–2373. [Google Scholar] [CrossRef]
- Williams, A.; Barry, B.W. Penetration enhancers. Adv. Drug Deliv. Rev. 2012, 64, 128–137. [Google Scholar] [CrossRef]
- Wang, Z.-J.; Heinbockel, T. Essential Oils and Their Constituents Targeting the GABAergic System and Sodium Channels as Treatment of Neurological Diseases. Molecules 2018, 23, 1061. [Google Scholar] [CrossRef]
- De Sousa, D.P.; Hocayen, P.D.A.S.; Andrade, L.N.; Andreatini, R. A Systematic Review of the Anxiolytic-Like Effects of Essential Oils in Animal Models. Molecule 2015, 20, 18620–18660. [Google Scholar] [CrossRef]
- Sowndhararajan, K.; Kim, S. Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Sci. Pharm. 2016, 84, 724–751. [Google Scholar] [CrossRef]
- Majnooni, M.B.; Mohammadi-Farani, A.; Gholivand, M.B.; Nikbakht, M.R.; Bahrami, G.R. Chemical composition and znxiolytic evaluation of Achillea Wilhelmsii, C. Koch essential oil in rat. Res. Pharm. Sci. 2013, 8, 269–275. [Google Scholar] [PubMed]
- Khazneh, E.; Hribova, P.; Hosek, J.; Suchy, P.; Kollar, P.; Prazanova, G.; Muselik, J.; Hanakova, Z.; Vaclavik, J.; Milek, M.; et al. The chemical composition of Achillea wilhelmsii C. Koch and its desirable effects on hyperglycemia, inflammatory meditors and hypercholesterolemia as risk factors for cardiometabolic diseases. Molecules 2016, 21, 404. [Google Scholar] [CrossRef] [PubMed]
- Koushki, M.; Yekta, R.F.; Amiri-Dashatan, N.; Dadpay, M.; Goshadrou, F. Therapeutic effects of hydro-alcoholic extract of Achillea wilhelmsii C. Koch on indomethacin-induced gastric ulcer in rats: A proteomic and metabolomic approach. BMC Complement. Altern. Med. 2019, 19, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.-S.; Park, K.-S.; Ha, J.-H.; Park, J.H.; Lim, J.-C.; Lee, D.-U. Inhibitory effects of the fragrance inhalation of essential oil from Acorus gramineus on central nervous system. Boil. Pharm. Bull. 2003, 26, 978–982. [Google Scholar] [CrossRef]
- Lee, H.-S. Fungicidal property of active component derived from Acorus gramineus rhizome against phytopathogenic fungi. Bioresour. Technol. 2007, 98, 1324–1328. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Li, W.-G.; Zhang, X.; Wang, L.; Xu, T.-L.; Wu, D.; Li, Y. Alpha-asarone from Acorus gramineus alleviates epilepsy by modulating A-Type GABA receptors. Neuropharmacology 2013, 65, 1–11. [Google Scholar] [CrossRef]
- Sun, Y.R.; Wang, X.Y.; Li, S.S.; Dong, H.Y.; Zhang, X.J. beta-asarone from Acorus gramineus alleviates depression by modulating MKP-1. Genet. Mol. Res. 2015, 14, 4495–4504. [Google Scholar] [CrossRef]
- Han, P.; Han, T.; Peng, W.; Wang, X.-R. Antidepressant-like effects of essential oil and asarone, a major essential oil component from the rhizome of Acorus tatarinowii. Pharm. Boil. 2013, 51, 589–594. [Google Scholar] [CrossRef]
- Zhang, F.-H.; Wang, Z.-M.; Liu, Y.-T.; Huang, J.-S.; Liang, S.; Wu, H.-H.; Xu, Y.-T. Bioactivities of serotonin transporter mediate antidepressant effects of Acorus tatarinowii Schott. J. Ethnopharmacol. 2019, 241, 111967. [Google Scholar] [CrossRef]
- Manzuoerh, R.; Farahpour, M.R.; Oryan, A.; Sonboli, A. Effectiveness of topical administration of Anethum graveolens essential oil on MRSA-infected wounds. Biomed. Pharmacother. 2019, 109, 1650–1658. [Google Scholar] [CrossRef]
- Kaur, N.; Chahal, K.K.; Kumar, A.; Singh, R.; Bhardwaj, U. Antioxidant activity of Anethum graveolens L. essential oil constituents and their chemical analogues. J. Food Biochem. 2019, 43, e12782. [Google Scholar] [CrossRef]
- Almeida, J.R.G.D.S.; Araújo, C.D.S.; De Oliveira, A.P.; Lima, R.N.; Alves, P.B.; Diniz, T.C. Chemical constituents and antioxidant activity of the essential oil from leaves of Annona vepretorum Mart. (Annonaceae). Pharmacogn. Mag. 2015, 11, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Diniz, T.C.; de Oliveira Junior, R.G.; Miranda Bezerra Medeiros, M.A.; Silva, M.G.; de Andrade Teles, R.E.; dos Passos Menezes, P.; de Sousa, B.M.; Frank, L.A.; de Souza Araujo, A.A.; Serafini, M.R.; et al. Anticonvulsant, sedative, anxiolytic and antidepessant activities of the essential oil of Annona vepretorum in mice: Involvement of GABAergic and serotonergic systems. Biomed. Pharmacother. 2019, 111, 1074–1087. [Google Scholar] [CrossRef] [PubMed]
- Abu-Darwish, M.S.; Cabral, C.; Gonçalves, M.; Cavaleiro, C.; Cruz, M.T.; Efferth, T.; Salgueiro, L. Artemisia herba-alba essential oil from Buseirah (South Jordan): Chemical characterization and assessment of safe antifungal and anti-inflammatory doses. J. Ethnopharmacol. 2015, 174, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Bourgou, S.; Rebey, I.B.; Mkadmini, K.; Isoda, H.; Ksouri, R.; Megdiche-Ksouri, W.; Bettaieb, I. LC-ESI-TOF-MS and GC-MS profiling of Artemisia herba-alba and evaluation of its bioactive properties. Food Res. Int. 2017, 99, 702–712. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, T.; El Aty, A.A.A.; Shahat, A.A.; Abdel-Azim, N.S.; Shams, K.; ElShamy, A.I.; Ahmed, M.M.; Youns, S.H.H.; El-Wassimy, T.M.; El-Toumy, S.A.; et al. New antimicrobial metabolites from the medicinal herb Artemisia herba-Alba. Nat. Prod. Res. 2019, 31, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Anaya-Eugenio, G.D.; Rivero-Cruz, I.; Bye, R.; Linares, E.; Mata, R. Antinociceptive activity of the essential oil from Artemisia ludoviciana. J. Ethnopharmacol. 2016, 179, 403–411. [Google Scholar] [CrossRef]
- Elmassry, K.; El-Ghorab, A.; Farouk, A. Antioxidant activity and volatile components of Egyptian Artemisia judaica L. Food Chem. 2002, 79, 331–336. [Google Scholar] [CrossRef]
- Abu-Darwish, M.S.; Cabral, C.; Gonçalves, M.; Cavaleiro, C.; Cruz, M.; Zulfiqar, A.; Khan, I.; Efferth, T.; Salgueiro, L. Chemical composition and biological activities of Artemisia judaica essential oil from southern desert of Jordan. J. Ethnopharmacol. 2016, 191, 161–168. [Google Scholar] [CrossRef]
- Sayyah, M.; Nadjafnia, L.; Kamalinejad, M. Anticonvulsant activity and chemical composition of Artemisia dracunculus L. essential oil. J. Ethnopharmacol. 2004, 94, 283–287. [Google Scholar] [CrossRef]
- Maham, M.; Moslemzadeh, H.; Jalilzadeh-Amin, G. Antinociceptive effect of the essential oil of tarragon (Artemisia dracunculus). Pharm. Boil. 2013, 52, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Eidi, A.; Oryan, S.; Zaringhalam, J.; Rad, M. Antinociceptive and anti-inflammatory effects of the aerial parts of Artemisia dracunculus in mice. Pharm. Boil. 2015, 54, 1–6. [Google Scholar]
- Park, H.J.; Lim, E.J.; Zhao, R.J.; Oh, S.R.; Jung, J.W.; Ahn, E.-M.; Lee, E.S.; Koo, J.S.; Kim, H.Y.; Chang, S.; et al. Effect of the fragrance inhalation of essential oil from Asarum heterotropoides on depression-ike behaviors in mice. BMC Complement. Altern. Med. 2015, 15, 43. [Google Scholar] [CrossRef] [PubMed]
- Haque, A.T.; Moon, J.N.; Saravana, P.; Tilahun, A.; Chun, B.-S. Composition of Asarum heterotropoides var. mandshuricum radix oil from different extraction methods and activities against human body odor-producing bacteria. J. Food Drug Anal. 2016, 24, 813–821. [Google Scholar] [CrossRef]
- Hossain, M.M.; Mahmood, S. In vitro studies on antibacterial, thrombolytic and antioxidant activities of green tea or Camellia sinensis. Am. J. Phytomed. Clinic. Therap. 2014, 2, 1200–1211. [Google Scholar]
- Lee, L.-S.; Kim, S.-H.; Kim, Y.-B.; Kim, Y.-C. Quantitative Analysis of Major Constituents in Green Tea with Different Plucking Periods and Their Antioxidant Activity. Molecules 2014, 19, 9173–9186. [Google Scholar] [CrossRef]
- Tan, L.T.H.; Lee, L.H.; Yin, W.F.; Chan, C.-K.; Kadir, H.A.; Chan, K.-G.; Goh, B.-H. Traditional Uses, Phytochemistry, and Bioactivities of Cananga odorata (Ylang-Ylang). Evid.-Based Complement. Altern. Med. 2015, 2015, 1–30. [Google Scholar]
- Ahmadi, S.G.S.; Farahpour, M.R.; Hamishehkar, H. Topical application of Cinnamon verum essential oil accelerates infected wound healing process by increasing tissue antioxidant capacity and keratin biosynthesis. Kaohsiung J. Med Sci. 2019, 35, 686–694. [Google Scholar] [CrossRef]
- Pultrini, A.D.M.; Galindo, L.A.; Costa, M. Effects of the essential oil from Citrus aurantium L. in experimental anxiety models in mice. Life Sci. 2006, 78, 1720–1725. [Google Scholar] [CrossRef]
- Costa, C.A.R.A.; Cury, T.C.; Cassettari, B.; Takahira, R.; Florio, J.C.; Costa, M. Citrus aurantium L. essential oil exhibits anxiolytic-like activity mediated by 5-HT1A-receptors and reduces cholesterol after repeated oral treatment. BMC Complement. Altern. Med. 2013, 13, 42. [Google Scholar] [CrossRef]
- Sanei-Dehkordi, A.; Sedaghat, M.M.; Vatandoost, H.; Abai, M.R. Chemical compositions of the peel essential oil of Citrus aurantium and its natural larvicidal activity against the malaria vector Anopheles stephensi (Dpitra: Cullidae) in comparison with Citrus paradisi. J. Arthropod-Borne Dis. 2016, 10, 577–585. [Google Scholar] [PubMed]
- Bagetta, G.; Morrone, L.A.; Rombolà, L.; Amantea, D.; Russo, R.; Berliocchi, L.; Sakurada, S.; Sakurada, T.; Rotiroti, D.; Corasaniti, M.T. Neuropharmacology of the essential oil of bergamot. Fitoterapia 2010, 81, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Rombolà, L.; Amantea, D.; Russo, R.; Adornetto, A.; Berliocchi, L.; Tridico, L.; Corasaniti, M.T.; Sakurada, S.; Sakurada, T.; Bagetta, G.; et al. Rational Basis for the Use of Bergamot Essential Oil in Complementary Medicine to Treat Chronic Pain. Mini-Rev. Med. Chem. 2016, 16, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Rombolà, L.; Tridico, L.; Scuteri, D.; Sakurada, T.; Sakurada, S.; Mizoguchi, H.; Avato, P.; Corasaniti, M.T.; Bagetta, G.; Morrone, L.A. Bergamot Essential Oil Attenuates Anxiety-Like Behaviour in Rats. Molecules 2017, 22, 614. [Google Scholar] [CrossRef]
- Rombolà, L.; Scuteri, D.; Adornetto, A.; Straface, M.; Sakurada, T.; Sakurada, S.; Mizoguchi, H.; Corasaniti, M.T.; Bagetta, G.; Tonin, P.; et al. Anxiolytic-Like Effects of Bergamot Essential Oil Are Insensitive to Flumazenil in Rats. Evid. -Based Complement. Altern. Med. 2019, 2019. [Google Scholar] [CrossRef]
- Faturi, C.B.; Leite, J.R.; Alves, P.B.; Canton, A.C.; Teixeira-Silva, F. Anxiolytic effect of sweet orange aroma in Wister rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 2010, 34, 605–609. [Google Scholar] [CrossRef]
- Goes, T.C.; Antunes, F.D.; Alves, P.B.; Teixeira-Silva, F. Effect of Sweet Orange Aroma on Experimental Anxiety in Humans. J. Altern. Complement. Med. 2012, 18, 798–804. [Google Scholar] [CrossRef]
- Hocayen, P.D.A.; Wendler, E.; Vecchia, D.D.; Kanazawa, L.K.; Issy, A.C.; Del Bel, E.; Andreatini, R. The nitrergic neurotransmission contributes to the anxiolytic-like effect of Citrus sinensis essential oil in animal models. Phytother. Res. 2019, 33, 901–909. [Google Scholar] [CrossRef]
- Rodrigues, I.A.; Ramos, A.d.S.; Falcao, D.Q.; Ferreira, J.L.P.; Basso, S.L.; Silva, J.R.; Amarat, A.C.F. Development of nanoemulsions to enhace the antileishmanial activity of Copaifera paupera oleoresins. BioMed Res. Int. 2018, 2018. [Google Scholar] [CrossRef]
- Cioanca, O.; Hritcu, L.; Mihasan, M.; Hancianu, M. Cognitive-enhancing and antioxidant activities of inhaled coriander oil in amyloid beta(1-42) rat model of Alzheimer’s disease. Physiol. Behav. 2013, 120, 193–202. [Google Scholar] [CrossRef]
- Cioanca, O.; Hritcu, L.; Mihasan, M.; Trifan, A.; Hancianu, M. Inhalation of coriander volatile oil increased anxiolytic-antidepressant-like behaviors and decreased oxidative status in beta-amyloid (1-42) rat model of Alzheimer’s disease. Physiol. Behav. 2014, 131, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Mandal, M. Coriander (Coriandrum sativum L.) essential oil: Chemistry and biological activity. Asian Pac. J. Trop. Biomed. 2015, 5, 421–428. [Google Scholar] [CrossRef]
- Blanco, M.; Costa, C.; Freire, A.; Santos, J.; Costa, M. Neurobehavioral effect of essential oil of Cymbopogon citratus in mice. Phytomedicine 2009, 16, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.R.; Ximenes, R.M.; Da Costa, J.G.M.; Leal, L.K.; De Lopes, A.A.; Viana, G.D.-B. Comparative anticonvulsant activities of the essential oils (EOs) from Cymbopogon winterianus Jowitt and Cymbopogon citratus (DC) Stapf. in mice. Naunyn-Schmiedebergs Arch. Pharmacol. 2010, 381, 415–426. [Google Scholar] [CrossRef]
- Costa, C.A.; Kohn, D.O.; de Lima, V.M.; Gargano, A.C.; Florio, J.C.; Costa, M. The GABAergic system contributes to the anxiolytic-like effect of essential oil from Cymbopogon citratus (lemon grass). J. Ethnopharmacol. 2011, 137, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.S.; Bidinotto, L.T.; Takahira, R.K.; Salvadori, D.M.; Barbisan, L.F.; Costa, M. Cholesterol reduction and lack of genotoxic or toxic effects in mice after repeated 21-day oral intake of lemon grass (Cymbopogon citratus) essential oil. Food Chem. Toxicol. 2011, 49, 2268–2272. [Google Scholar] [CrossRef]
- Shah, G.; Shri, R.; Panchal, V.; Sharma, N.; Singh, B.; Mann, A.S. Scientific basis for the therapeutic use of Cymbopogon citratus, stapf (Lemon grass). J. Adv. Pharm. Technol. Res. 2011, 2, 3–8. [Google Scholar] [CrossRef]
- Quintans-Junior, L.J.; Souza, T.T.; Leite, B.S.; Lessa, N.M.; Bonjardim, L.R.; Santos, M.R.V.; Alves, P.B.; Blank, A.F.; Antoniolli, A.R. Phytochemical screening and anticonvulsant activity of Cymbopogon winterianus Jowitt (Poaceae) leaf essential oil in rodents. Phytomedicine 2008, 15, 619–624. [Google Scholar] [CrossRef]
- Rodrigues, K.A.F.; Dias, C.N.; do Amaral, F.M.M.; Moraes, D.F.C.; Filho, V.E.M.; Andrade, E.H.A.; Maia, J.G.S. Molluschicidal and larvicidal activities and essential oil composition of Cymbopogon winterianus. Pharm. Biol. 2013, 51, 1293–1297. [Google Scholar] [CrossRef]
- Déciga-Campos, M.; Mata, R.; Rivero-Cruz, I. Antinociceptive pharmacological profile of Dysphania graveolens in mouse. Biomed. Pharmacother. 2017, 89, 933–938. [Google Scholar] [CrossRef]
- Silva, L.; Garlet, Q.; Benovit, S.; Dolci, G.; Mallmann, C.A.; Bürger, M.; Baldisserotto, B.; Longhi, S.; Heinzmann, B.M. Sedative and anesthetic activities of the essential oils of Hyptis mutabilis (Rich.) Briq. and their isolated components in silver catfish (Rhamdia quelen). Braz. J. Med. Boil. Res. 2013, 46, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Abuhamdah, S.; Howes, M.-J.R.; Dixon, C.L.; Elliot, M.S.J.; Ballard, C.; Holmes, C.; Burns, A.; Perry, E.; Francis, P.; et al. Pharmacological profile of essential oils derived from Lavandula angustifolia and Melissa officinalis with anti-agitation properties: Focus on ligand-gated channels. J. Pharm. Pharmacol. 2008, 60, 1515–1522. [Google Scholar] [CrossRef] [PubMed]
- Chioca, L.R.; Antunes, V.D.; Ferro, M.M.; Losso, E.M.; Andreatini, R. Anosmia does not impair the anxiolytic-like effect of lavender essential oil inhalation in mice. Life Sci. 2013, 92, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Chioca, L.R.; Ferro, M.M.; Baretta, I.P.; Oliveira, S.M.; Silva, C.R.; Ferreira, J.; Losso, E.M.; Andreatini, R. Anxiolytic-like effect of lavender essential oil inhalation in mice: Participation of serotonergic but not GABAA/benzodiazepine neurotransmission. J. Ethnopharmacol. 2013, 147, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.H.; Seol, G.H. Lavandula angustifolia Mill. Oil and Its Active Constituent Linalyl Acetate Alleviate Pain and Urinary Residual Sense after Colorectal Cancer Surgery: A Randomised Controlled Trial. Evid.-Based Complement. Altern. Med. 2017, 2017, 1–7. [Google Scholar] [CrossRef]
- Watson, K.; Hatcher, D.; Good, A. A randomised controlled trial of Lavender (Lavandula angustifolia) and Lemon Balm (Melissa officinalis) essential oils for the treatment of agitated behaviour in older people with and without dementia. Complement. Ther. Med. 2019, 42, 366–373. [Google Scholar] [CrossRef]
- Heldwein, C.G.; Silva, L.L.; Reckziegel, P.; Barros, F.M.; Burger, M.E.; Baldisserotto, B.; Mallmann, C.A.; Schmidt, D.; Caron, B.O.; Heinzmann, B.M. Participation of the GABAergic system in the anesthetic effect of Lippia alba (Mill.) N.E. Brown essential oil. Braz. J. Med. Biol. Res. 2012, 45, 436–443. [Google Scholar] [CrossRef]
- Heldwein, C.G.; Silva, L.D.L.; Gai, E.Z.; Roman, C.; Parodi, T.V.; E Bürger, M.; Baldisserotto, B.; Flores, E.M.M.; Heinzmann, B.M. S -(+)-Linalool from Lippia alba: Sedative and anesthetic for silver catfish (Rhamdia quelen). Vet. Anaesth. Analg. 2014, 41, 621–629. [Google Scholar] [CrossRef]
- Souza, C.D.F.; Baldissera, M.D.; Bianchini, A.E.; Da Silva, E.G.; Mourão, R.H.V.; Da Silva, L.V.F.; Schmidt, D.; Heinzmann, B.M.; Baldisserotto, B. Citral and linalool chemotypes of Lippia alba essential oil as anesthetics for fish: A detailed physiological analysis of side effects during anesthetic recovery in silver catfish (Rhamdia quelen). Fish Physiol. Biochem. 2017, 44, 21–34. [Google Scholar] [CrossRef]
- Souza, C.D.F.; Descovi, S.; Baldissera, M.D.; Bertolin, K.; Bianchini, A.E.; Mourão, R.H.V.; Schmidt, D.; Heinzmann, B.M.; Antoniazzi, A.; Baldisserotto, B.; et al. Involvement of HPI-axis in anesthesia with Lippia alba essential oil citral and linalool chemotypes: Gene expression in the secondary responses in silver catfish. Fish Physiol. Biochem. 2018, 45, 155–166. [Google Scholar] [CrossRef]
- McKay, D.L.; Blumberg, J. A Review of the bioactivity and potential health benefits of chamomile tea (Matricaria recutita L.). Phytotherapy Res. 2006, 20, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Keefe, J.; Mao, J.J.; Soeller, I.; Li, Q.S.; Amsterdam, J.D. Short-term open-label chamomile (Matricaria chamomilla L.) therapy of moderate to severe generalized anxiety disorder. Phytomedicine 2016, 23, 1699–1705. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.J.; Xie, S.X.; Keefe, J.R.; Soeller, I.; Li, Q.S.; Amsterdam, J.D. Long-term chamomile (Matricaria chamomilla L.) treatment for generalized anxiety disorder: A randomized clinical trial. Phytomedicine 2016, 23, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- Abuhamdah, S.; Huang, L.; Elliott, M.S.J.; Howes, M.-J.R.; Ballard, C.; Holmes, C.; Burns, A.; Perry, E.K.; Francis, P.; Lees, G.; et al. Pharmacological profile of an essential oil derived from Melissa officinalis with anti-agitation properties: Focus on ligand-gated channels. J. Pharm. Pharmacol. 2008, 60, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Abdellatif, F.; Boudjella, H.; Zitouni, A.; Hassani, A. Chemical composition and antimicrobial activity of the essential oil from leaves of Algerian Melissa officinalis L. EXCLI J. 2014, 13, 772–781. [Google Scholar] [PubMed]
- Schmidt, E.; Bail, S.; Buchbauer, G.; Stoilova, I.; Atanasova, T.; Stoyanova, A.; Krastanov, A.; Jirovetz, L. Chemical composition, olfactory evaluation and antioxidant effects of essential oil from Mentha x piperita. Nat. Prod. Commun. 2009, 4, 1107–1112. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.; Alghasham, A.A.; Alorainy, M.S.; El-Hadiyah, T.M. Potentiation of Valproate-induced Anticonvulsant Response by Nigella sativa Seed Constituents: The Role of GABA Receptors. Int. J. Health Sci. 2008, 2, 15–25. [Google Scholar]
- Tankam, J.M.; Ito, M. Inhalation of the Essential Oil of Piper guineense from Cameroon Shows Sedative and Anxiolytic-Like Effects in Mice. Boil. Pharm. Bull. 2013, 36, 1608–1614. [Google Scholar] [CrossRef]
- Oyemitan, I.A.; Olayera, O.A.; Alabi, A.; Abass, L.A.; Elusiyan, C.A.; Oyedji, A.O.; Akanmu, M.A. Psychoneuropharmacological activities and chemcal composition of essential oil of fresh fruits of Piper guineense (Piperaceae) in mice. J. Ethnopharmacol. 2015, 166, 240–249. [Google Scholar] [CrossRef]
- Khezri, K.; Farahpour, M.R.; Rad, S.M. Accelerated infected wound healing by topical application of encapsulated Rosemary essential oil into nanostructured lipid carriers. Artif. Cells Nanomed. Biotechnol. 2019, 47, 980–988. [Google Scholar] [CrossRef]
- Seol, G.H.; Shim, H.S.; Kim, P.-J.; Moon, H.K.; Lee, K.H.; Shim, I.; Suh, S.H.; Min, S.S. Antidepressant-like effect of Salvia sclarea is explained by modulation of dopamine activities in rats. J. Ethnopharmacol. 2010, 130, 187–190. [Google Scholar] [CrossRef] [PubMed]
- Nassar, M.I.; Gaara, A.H.; El-Ghorab, A.H.; Farrag, A.-R.H.; Shen, H.; Huq, E.; Mabry, T.J. Chemical constituents of clove (Syzygium aromaticum, Fam. Myrtaceae) and their antioxidant activity. Rev. Latinoam. Quim. 2007, 35, 47–57. [Google Scholar]
- Nirmala, M.J.; Durai, L.; Gopakumar, V.; Nagarajan, R. Anticancer and antibacterial effects of a clove bud essential oil-based nanoscale emulsion system. Int. J. Nanomed. 2019, 14, 6439–6450. [Google Scholar] [CrossRef] [PubMed]
- Marin, R.; Garcia, D.; Martijena, I.D.; A Zygadlo, J.; Arce, A.; Perillo, A.M. Anxiogenic-like effects of Tagetes minuta L essential oil on T-maze and tonic immobility behaviour in domestic chicks. Fundam. Clin. Pharmacol. 1998, 12, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, J.C.R.; De Meneses, D.A.; De Vasconcelos, A.P.; Piauilino, C.A.; Almeida, F.R.D.C.; Napoli, E.M.; Ruberto, G.; Araujo, D. Essential oil composition and antinociceptive activity of Thymus capitatus. Pharm. Boil. 2017, 55, 782–786. [Google Scholar] [CrossRef]
- El-Jalel, L.F.A.; Elkady, W.M.; Gonaid, M.H.; Gareeb, K.A. Difference in chemical compositionand antimicrobial activity of Thymus capitatus L. essential oil at different altitudes. Fut. J. Pharm. Sci. 2018, 4, 156–160. [Google Scholar]
- Houghton, P.J. The scientific basis for the reputed activity of Valerian. J. Pharm. Pharmacol. 1999, 51, 505–512. [Google Scholar] [CrossRef]
- Kazemi, M. Phenolic profile, antioxidant capacity and anti-inflammatory activity of Anethum graveolens L. essential oil. Nat. Prod. Res. 2014, 29, 551–553. [Google Scholar] [CrossRef]
- Rao, B.R.R.; Rajput, D.K.; Bhattacharya, A.K. Essential oil composition of petiole of Cinnamomum verum Bercht. & Presl. J. Spices Arom. Crops. 2007, 16, 38–41. [Google Scholar]
- Dar, S.; Lawrence, R.; Khan, A.N. Evaluation of the chemical constituents and the antibacterial activity of essential oil of Citrus karna fruit peel. Int. J. Pharm. Sci. Res. 2016, 7, 1240–1250. [Google Scholar]
- Rivero-Cruz, I.; Ospina, H.A.; Mata, R. Chemical Composition of Dysphania graveolens and Dysphania ambrosioides Essential Oils. Planta Med. 2012, 78, 97. [Google Scholar] [CrossRef]
- Hennebelle, T.; Sahpaz, S.; Joseph, H.; Bailleul, F. Ethnopharmacology of Lippia alba. J. Ethnopharmacol. 2008, 116, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Jirovetz, L.; Buchbauer, G.; Denkova, Z.; Slavchev, A.; Stoyanova, A.; Schmidt, E. Chemical composition, antimicrobial activities and odor descriptions of various Salvia sp. and Thuja sp. essential oils. Nutrition 2006, 30, 152–159. [Google Scholar]
- Acimovic, M.; Kiprovski, B.; Rat, M.; Sikora, V.; Popovic, V. Salvia sclarea: Chemical composition and biological activity. J. Agron. Technol. Eng. Manag. 2018, 1, 18–28. [Google Scholar]
- Chamorro, E.R.; Ballerini, G.; Sequeira, A.F.; Velasco, G.A.; Zalazar, M.F. Chemical composition of essential oil from Tagetes minuta L. leaves and flowers. J. Argent. Chem. Soc. 2008, 96, 80–86. [Google Scholar]
- Chen, H.-W.; Wei, B.-J.; He, X.-H.; Liu, Y.; Wang, J. Chemical Components and Cardiovascular Activities of Valeriana spp. Evid.-Based Complement. Altern. Med. 2015, 2015, 1–11. [Google Scholar]
- Petrović, J.; Stojković, D.; Sokovic, M. Terpene core in selected aromatic and edible plants: Natural health improving agents. Adv. Food Nutr. Res. 2019, 90, 423–451. [Google Scholar]
- Guimarães, A.C.; Meireles, L.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial Activity of Terpenes and Terpenoids Present in Essential Oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Bioactivity and protective effects of natural carotenoids. Biochim. Biophys. Acta-Mol. Basis Dis. 2005, 1740, 101–107. [Google Scholar] [CrossRef]
- Ince, B.; Dadaci, M.; Kilinc, I.; Oltulu, P.; Yarar, S.; Uyar, M. Effect of cineole, alpha-pinene, and camphor on survivability of skin flaps. Turk. J. Med. Sci. 2018, 48, 644–652. [Google Scholar]
- Sá, R.D.C.D.S.E.; Lima, T.C.; Da Nóbrega, F.R.; De Brito, A.E.M.; De Sousa, D.P. Analgesic-Like Activity of Essential Oil Constituents: An Update. Int. J. Mol. Sci. 2017, 18, 2392. [Google Scholar] [CrossRef]
- Chen, H.; Yuan, J.; Hao, J.; Wen, Y.; Lv, Y. Alpha-humulene inhibits hepatocellular carcinoma cell proliferation and induces apopptosis through the inhibition of Akt signaling. Food Chem. Toxicol. 2019, 134, 110830. [Google Scholar] [CrossRef] [PubMed]
- Boehm, U.; Zou, Z.; Buck, L.B. Feedback Loops Link Odor and Pheromone Signaling with Reproduction. Cell 2005, 123, 683–695. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Fukuoka, T.; Obata, K.; Yamanaka, H.; Dai, Y.; Tokunaga, A.; Noguchi, K. Distinct expression of TRPM8, TRPA1, and TRPV1 mRNAs in rat primary afferent neurons with Adelta/C-fibers and colocalization with Trk receptors. J. Comp. Neurol. 2005, 493, 596–606. [Google Scholar] [CrossRef]
- Ferreira-Da-Silva, F.W.; Da Silva-Alves, K.S.; Alves-Fernandes, T.A.; Coelho-De-Souza, A.N.; Leal-Cardoso, J.H. Effects of 1,8-cineole on Na+ currents of dissociated superior cervical ganglia neurons. Neurosci. Lett. 2015, 595, 45–49. [Google Scholar] [CrossRef]
- Takaishi, M.; Fujita, F.; Uchida, K.; Yamamoto, S.; Shimizu, M.S.; Uotsu, C.H.; Shimizu, M.; Tominaga, M. 1,8-cineole, a TRPM8 agonist, is a novel natural antagonist of human TRPA1. Mol. Pain 2012, 8, 86. [Google Scholar] [CrossRef]
- Kawasaki, H.; Mizuta, K.; Fujita, T.; Kumamoto, E. Inhibition by menthol and its related chemicals of compound action potentials in frog sciatic nerves. Life Sci. 2013, 92, 359–367. [Google Scholar] [CrossRef]
- Hanack, C.; Moroni, M.; Lima, W.C.; Wende, H.; Kirchner, M.; Adelfinger, L.; Schrenk-Siemens, K.; Tappe-Theodor, A.; Wetzel, C.; Kuich, P.H.; et al. GABA Blocks Pathological but Not Acute TRPV1 Pain Signals. Cell 2015, 160, 759–770. [Google Scholar] [CrossRef]
- Storozhuk, M.; Moroz, O.; Zholos, A. Multifunctional TRPV1 Ion Channels in Physiology and Pathology with Focus on the Brain, Vasculature, and Some Visceral Systems. BioMed Res. Int. 2019, 2019. [Google Scholar] [CrossRef]
- Ngo, D.H.; Vo, T.S. An Updated Review on Pharmaceutical Properties of Gamma-Aminobutyric Acid. Molecules 2019, 24, 2678. [Google Scholar] [CrossRef]
- Ramesh, S.; Tyerman, S.; Gilliham, M.; Xu, B. γ-Aminobutyric acid (GABA) signalling in plants. Cell. Mol. Life Sci. 2016, 74, 1577–1603. [Google Scholar] [CrossRef] [PubMed]
- Fait, A.; Yellin, A.; Fromm, H. GABA and GHB Neurotransmitters in Plants and Animals. In Communication in Plants; Springer: Berlin/Heidelberg, Germany, 2006; pp. 171–185. [Google Scholar]
- Johnston, G.A.; Hanrahan, J.; Chebib, M.; Duke, R.K.; Mewett, K.N. Modulation of ionotropic GABA receptors by natural products of plant origin. Adv. Pharmacol. 2006, 54, 285–316. [Google Scholar] [PubMed]
- Santiago, M.J.; Sachdev, S.; Arnold, J.C.; McGregor, I.S.; Connor, M. Absence of Entourage: Terpenoids Commonly Found inCannabis sativaDo Not Modulate the Functional Activity of Δ9-THC at Human CB1and CB2Receptors. Cannabis Cannabinoid Res. 2019, 4, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Klauke, A.-L.; Rácz, I.; Pradier, B.; Markert, A.; Zimmer, A.; Gertsch, J.; Zimmer, A. The cannabinoid CB2 receptor-selective phytocannabinoid beta-caryophyllene exerts analgesic effects in mouse models of inflammatory and neuropathic pain. Eur. Neuropsychopharmacol. 2014, 24, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.M.; Rousseau, M.A.; Wanas, A.S.; Radwan, M.M.; Caldwell, S.; Sufka, K.J.; ElSohly, M.A. Role of Cannabinoids and Terpenes in Cannabis-Mediated Analgesia in Rats. Cannabis Cannabinoid Res. 2019, 4, 177–182. [Google Scholar] [CrossRef]
- Mercier, B.; Prost, J.; Prost, M. The essential oil of turpentine and its major volatile fraction (alpha- and beta-pinenes): A review. Int. J. Occup. Med. Environ. Health 2009, 22, 331–342. [Google Scholar] [CrossRef]
- Graßmann, J.; Hippeli, S.; Vollmann, R.; Elstner, E.F. Antioxidative Properties of the Essential Oil fromPinus mugo. J. Agric. Food Chem. 2003, 51, 7576–7582. [Google Scholar] [CrossRef]
- Muller, C.; Morales, P.; Reggio, P.H. Cannabinoid Ligands Targeting TRP Channels. Front. Mol. Neurosci. 2019, 11, 487. [Google Scholar] [CrossRef] [PubMed]
- Spehr, M.; Spehr, J.; Ukhanov, K.; Kelliher, K.R.; Leinders-Zufall, T.; Zufall, F. Signaling in the Chemosensory Systems. Cell. Mol. Life Sci. 2006, 63, 1476–1484. [Google Scholar] [CrossRef]
- Canavan, S.V.; Mayes, L.C.; Treloar, H.B. Changes in Maternal Gene Expression in Olfactory Circuits in the Immediate Postpartum Period. Front. Psychol. 2011, 2. [Google Scholar] [CrossRef]
| Domain | Cluster | Examples of the Odorous Chemical Compounds, which Activated the Cluster | Odor Quality |
|---|---|---|---|
| Dorsal (DI) | A | Fatty acids and alkylamines [31]. Aliphatic acids, aliphatic aldehydes as well as “the subsets of these odorants with a similar carbon chain length” [30]. Diketones, benzaldehyde, and benzoic acid; common features are “a carboxyl group (-COOH), a diketone group (-(CO)(CO)-), or an ester group (-COO-)”. | Fatty Rancid sour pungent |
| Dorsal (DII) | B | Aliphatic alcohols, aliphatic ketones, and phenyl ethers [31]. “Aliphatic alcohols with relatively long carbon chain and to a wide range of aliphatic ketones”. Subsets of esters. Anisole and its derivatives with a methoxy group (-O-CH3). Combination “of elongated carbon chain structures with a hydroxyl group (-OH), an alkoxyl group (-O-R), or a carbonyl group (>C=O)”. | Floral Fruity Green and grassy Anisic |
| Dorsal DII | C | Phenols and phenyl ethers [31]. “Phenol family odorants, molecules having a hydroxyl group attached to the benzene ring”. Phenyl ethers; “molecular features of a benzene ring with hydroxyl group, a methoxy group, or an ethoxy group”. | Phenolic Medicinal Spicy Ethereal |
| D | Aliphatic and aromatic ketones [31]; “responses to a variety of ketones: aliphatic ketones, aliphatic-aromatic ketones, diketones, and cyclic ketones”. | Spicy Minty | |
| Lateral/ventral | E | Not specified in Mori et al. [30] nor in Mori and Sakano [31]; weak responses to ethers [30]. | |
| F | Aliphatic “ketones, phenyl ethers, diketones, aliphatic-aromatic ketones, and cyclic ketones”. | ||
| G | Phenyl “ethers, diketones, aliphatic ketones with relatively short side chains, aliphatic-aromatic ketones, cyclic ketones and ethers”. | ||
| H | “Benzene-family odorants”. No response “or only a weak response to cyclic terpene hydrocarbons”. Some “respond to open-chain hydrocarbons”. “benzene derivatives with a polar functional group, such as anisole”, phenol, benzaldehyde, or acetophenone. | Gassy and kerosene-like odor | |
| I | “respond to at least one of the cyclic terpene hydrocarbons”. Subset of benzene-family odorants; cyclic terpene ketones, cyclic terpene alcohols | Citrusy Woody | |
| Dorsal DII | J | Thiazoles and thiazolines [31]. |
| Latin Name | Plants Common Name | Species | Routes Used in Experiments | Results | References |
|---|---|---|---|---|---|
| Achillea wilhelmsii | Achillea | Rat | IP; gavage | Anxiolytic effect; improved healing of gastric ulcer; anti-hypercholesterolemic | [94,95,96] |
| Acorus gramineus | Grass-leaf sweet flag | Mice; rats; bacteria | Inhalation; gavage; in vitro | Anti-convulsive; sedative effect; act on central nervous system via GABA system; anti-depression; anti-epilepsy through GABA system; anti-fungal | [97,98,99,100] |
| Acorus tatarinowii | Acorus | Mice | IP; gavage | Anti-depressant; enhanced serotonin transporter | [101,102] |
| Anethum graveolens | Dill | Mice | Topical | Improved infected wound healing; anti-oxidant | [103,104] |
| Annona vepretorum | Anaticum | Mice | Gavage | Anti-oxidant; Anti-convulsant; sedative; anxiolytic; anti-depressant | [105,106] |
| Artemisia herba-alba | White wormwood | Bacteria | In vitro | Anti-bacterial activity; anti-fungal; anti-oxidant, anti-cancer, anti-bacterial; anti-inflammatory | [107,108,109] |
| Artemisia ludoviciana | Silver wormwood | Mice | IP | Anti-nociceptive | [110] |
| Artemisia judaica | Judean wormwood | Yeasts | In vitro | Anti-oxidant; Anti-fungal; anti-biofilm; anti-inflammatory | [111,112] |
| Artemisia dracunculus | Tarragon | Rats; Mice | IP | Anti-convulsant; Anti-nociceptive effect; anti-inflammatory effect | [113,114,115] |
| Asarum heterotropoides | Asarum | Mice | Inhalation | Anti-depressant; anti-bacterial | [116,117] |
| Camellia sinensis | Tea tree; tea shrub | Mice | Inhalation | GABA-A enhanced using oolong tea; anti-oxidant using green tea | [118,119] |
| Cananga odorata | Ylang-ylang | Human | Inhalation | Decreased arousal level; effective in treating depression, high blood pressure, and anxiety. Anti-microbial, anti-biofilm, anti-inflammatory, anti-vector, insect-repellent, anti-diabetic, anti-fertility, anti-melanogenesis | [45,120] |
| Cinnamon verum | Cinnamon | Mice | Topical | Improved infected wound healing | [121] |
| Citrus aurantium, L. | Bitter orange | Mice | Gavage | Anxiolytic effect mediated by 5-HT (1A) receptors; reduce cholesterol; larvicidal effect | [122,123,124] |
| Citrus bergamia | Bergamot orange | Mice; rat | Decreased stress-induced anxiety; anxiolytic, sedative; suppress pain | [125,126,127,128] | |
| Citrus sinensis | Sweet orange | Mice; rat; human | Anxiolytic effect; nitrogenic neurotransmission plays a role | [129,130,131] | |
| Copaifera paupera | Oleoresin | Mice | Topical application | Improved wound healing; anti-leishmanial | [68,132] |
| Coriandrum sativum, L. | Coriander | Rat | Inhalation | Anxiolytic effect; cognitive enhancement; anti-bacterial, anti-fungal, anti-oxidative effect | [133,134,135] |
| Cymbopogon citratus | Lemon grass | Mice | Gavage; IP | Anxiolytic effect through GABA system; increased sleeping time; anti-convulsive effect at least partially mediated by GABA system; cholesterol reduction; anti-amoebic, anti-bacterial, anti-diarrheal, anti-filarial, anti-fungal, anti-inflammatory, anti-malarial, anti-mutagenicity, anti-mycobacterial, anti-oxidants, and hypoglycemic and neurobehavioral effects | [136,137,138,139,140] |
| Cymbopogon winterianus | Citronella grass | Mice | IP | Anti-convulsant; Molluschicidal and larvicidal activity | [137,141,142] |
| Dysphania graveolens | Fetid goosefoot | Mice | Gavage | Anti-nociceptive, analgesic effect not through 5-HT2A/2C | [143] |
| Hyptis Mutabilis | Tropical bushmint | Silver catfish | In water | Sedative effect not through GABA system | [144] |
| Lavandula angustifolia | English lavender | Human; Mice; in vitro study with rat cells | Inhalation | Decreased cognitive function, decreased arousal level; inhibited TBPS binding to rat GABA-A receptor; anxiolytic-like effect not through GABA system; anosmia does not impair anxiolytic effect; pain suppression; reduced agitation and physical non-aggressive behavior in older people with dementia | [43,144,145,146,147,148,149] |
| Lippia alba | Bushy lippia | Silver catfish | In water | Anesthetic effect through GABA system; S-(+)-linalool in lippia has sedative and anesthetic effect; citral and linalool in it have anesthetic effect | [150,151,152,153] |
| Matricaria chamomilla | Chamomile | Human; review of animal studies | Inhalation; capsule consumption | Anxiety suppression; anti-inflammatory effect, anti-mutagenic effect, cholesterol-reduction effect, anti-spasmotic effect, anxiolytic effect | [154,155,156] |
| Melissa officinalis, L. | Common balm; lemon balm | Rat; human | In vitro; inhalation | Anti-agitation; anti-microbial | [144,149,157,158] |
| Mentha x piperita | Peppermint | Human | Inhalation; digested capsules | Increased arousal level Showed less fatigue to cognitively demanding tasks and showed higher cognitive function; anti-oxidant | [45,80,159] |
| Nigella sativa, L. | Black cumin | Mice | Inhalation | Anti-convulsant effect | [160] |
| Piper guineense | Pepper | Mice | Inhalation; IP | Sedative effect; anxiolytic effect; central nervous system depressant; rectal temperature suppressed, muscle relaxant, induced catalepsy; anti-psychotic effect, anti-convulsant effect | [161,162] |
| Rosmarinus officinalis | Rosemary | Mice; human | Inhalation; topical application | Suppressed serum corticosterone, brain dopamine increased; anxiolytic effect, Increased arousal level More active and fresher feeling; blood pressure enhanced, heart rate enhanced, improved infected wound healing; enhanced cognitive function in humans | [43,44,47,163] |
| Salvia lavandulifolia | Spanish sage | Human | Drink | Enhanced cognitive function | [46] |
| Salvia officinalis | Common sage | Human | Drink | Enhanced cognitive function | [46] |
| Salvia sclarea | Clary sage | Rat | IP | Anti-depressant; anti-stressor; modulation of dopamine | [164] |
| Syzygium aromaticum | Clove | In vitro | Anti-oxidant; anti-bacterial effect; anti-cancer effect | [165,166] | |
| Tagetes minuta, L. | Black mint | Chicks | SC | Anxiolytic | [167] |
| Thymus capitatus | Spanish oregano | Mice; rats | Gavage | Anti-nociception; anti-microbial | [168,169] |
| Valeriana officinalis, L. | Valerian | Review | Sedative effect; suppressed GABA breakdown | [170] |
| Latin Name | Plants Common Name | Major constituents | References |
|---|---|---|---|
| Achillea wilhelmsii | Achillea | Luteolin, apigenin, caffeic acid, ferulic acid, leucodin, 1β-epoxydesacetoxymatricarin, 10β-epoxydesacetoxymatricarin, 2-(3,4-dimethoxyphenyl)-5-hydroxy-6,7-dimethoxychromen-4-one, 2-(3,4-dimethoxyphenyl)-5,6,7-dimethoxychromen-4-one, salvigenin | [95] |
| Acorus gramineus | Grass-leaf sweet flag | From rhizomes: α-asarone (17.7%), cis-asarone (7.29%), asaronaldehyde (5.35%), borneol (2.18%), γ-cadinene (2.56%), calarene (1.64%), camphene (0.73%), camphor (3.63%), elemicin (1.98%), euasarone (12.7%), α-gurjunene (1.21%), 1,2,4-methenoazulene (0.82%), methyleugenol (34.18%), methyl isoeugenol (4.90%), and α-muurolene (0.76%). | [98] |
| Acorus tatarinowii | Acorus | β-asarone, α-asarone, veratric acid, anisic acid, 3,4,5-trimethoxybenzoic acid, trans-isoferulic acid, 2,4,5-trimethoxybenzoic acid, 4-hydroxybenzoic acid, syringic acid | [102] |
| Anethum graveolens | Dill | Kazemi: α-phellandrene (19.12%), limonene (26.34%), dill ether (15.23%), sabinene (11.34%), n-nonadecane (1%), n-eicosane (0.78%), n-heneicosane (0.67%), β-myrcene (0.23%), α-thujene (0.21%); Kaur et al.: Carvone, limonene, camphor, carveol, perillyl alcohol | [104,171] |
| Annona vepretorum | Anaticum | Spathulenol (43.7%), limonene (20.5%), caryophyllene oxide (8.1%), α-pinene (5.5%) | [105] |
| Artemisia herba-alba | White wormwood | Bourgou et al.: c-glycosylated and methylated flavones, quinic acid derivatives, coumarins, sesquiterpenes lactones, terpenoids, fatty acids, carbohydrates, organic acids and alkaloids, di-O-caffeoylquinic acids, artemisinic acid, menthol, α-ketoglutaric acid, scopolin, isoschaftoside and sucrose; Mohamed et al.: 1,3,8-trihydroxyeudesm-4-en-7α,11βH-12,6α-olide, 5-β-D-glucopyranosyloxy-7-methoxy-6H-benzopyran-2-one, 3α,8β-dihydroxygermacr-4,9(10)-dien-7β,11αH,12,6α-olide, 1β,8α-dihydroxy-11α,13-dihydrobalchanin, 11-epiartapshin, tomenin benzoic acid, p-(β-D-glucopyranosyloxy)-methyl ester | [108,109] |
| Artemisia ludoviciana | Silver wormwood | (±)-camphor, γ-terpineol, 1,8-cineole, and borneol | [110] |
| Artemisia judaica | Piperitone (45.0%), trans-ethyl cinnamate (20.8%), ethyl-3-phenyl propionate (11.0%), spathulenol (6.27%), cis-ethyl cinnamate (5.64%), 2,6-dimethyl phenole (1.39%), methyl cinnamate (1.06%), 2,6-dimethyl phenol (1.39%), camphor (0.38%) | [111] | |
| Artemisia dracunculus | Tarragon | trans-anethole (21.1%), α-trans-ocimene (20.6%), limonene (12.4%), α-pinene (5.1%), allo ocimene (4.8%), methyl eugenol (2.2%), β-pinene (0.8%), α-terpinolene (0.5%), bornyl acetate (0.5%), and bicyclogermacrene (0.5%) | [113] |
| Asarum heterotropoids | Asarum | Methyl eugenol (37.6%), sesamin (22.1%), safrole (14.7%), N-isobutyl-(2E,4Z,8Z,10E)-dodecatetraenamide (8.6%), linoleic acid (3.2%), trans-isocroweacin (5.5%), pentadecane (4.2%), 3,4-benzocyclodec-3-ene-1,5-diyn-7-one (3%), myristicin (1%) | [117] |
| Camellia sineusis | Green tea | Theanine, theobromine, caffeine, gallic acid, (+)-gallocatechin, caffeine, (-)-epigallocatechin, (+)-catechin, (-)-epicatechin, (-)-epigallocatechin gallate, (+)-gallocatechin gallate, (-)-epicatechin gallate, (+)-catechin gallate | [119] |
| Cananga odorata | Ylang-ylang | p-methylanisole, methyl benzoate, benzyl benzoate, benzyl acetate, geranyl acetate, cinnamyl acetate, (E,E)-farnesyl acetate, linalool, geraniol, benzyl salicylate, germacrene D, β-caryophyllene, gamma-muurolene, (E,E)-farnesyl acetate | [120] |
| Linalool, germacrene D, benzyl acetate, p-cresyl methyl ether, (E,E)-α-farnesene, geranyl acetate, methyl benzoate, β-caryophyllene | [6] | ||
| Cinnamomum verum | Cinnamon | Eugenol (17.32%), Benzyl benzoate (0.22%), (E)-Cinnamyl alcohol (0.09%), eugenyl acetate (1.29%), (E)-cinnamyl acetate (11.78%), (Z)-cinnamyl acetate (0.01%), (E)-cinnamaldehyde (33.04%), safrole (0.01%), geraniol (0.05%), piperitone (0.16%), α-terpineol (0.61%), terpinen-4-ol (0.13%), linalyl acetate (2.25%), linalool (16.85%), p-cymene (0.92%), trans- β-ocimene (0.05%), cis-β-ocimene (0.03%), 1,8-coneole (0.46%), limonene (0.41%), α-terminene (0.09%), myrcene (1.17%), sabinene (0.06%), β-pinene (0.19%), camphene (0.09%) | [172] |
| Citrus aurantium, L. | Bitter orange | DI-limonene (94.81%), α-pinene (0.30%), β-pinene (0.65%), β-myrcene (1.00%), trans-ocimene (0.19%), γ-terpinene (0.01%), linalool oxide (0.04%), 1-octanol (0.13%), trans-linalool oxie (0.02%), isoterpinolene (0.01%), nonanal (0.02%), α-terminolene (0.44%), linalyl acetate (0.32%), sabinene hydrate acetate (0.93%), | [124] |
| Citrus bergamia | Bergamot orange | Limonene (25.62 – 52.19%), linalool (1.75–20.26%), linalyl acetate (15.61–40.37%), | [127] |
| Citrus sinensis | Sweet orange | Limonene (93.5%), β-pinene (2.979%), α-pinene (0.792%), cis-ocimene (0.235%), linalook (0.206%), α-terpineol (0.142%), (E)-citral (geranial) (0.150%), α-bergamotene (0.448%), β-bisabolene (0.365%) | [173] |
| Copaifera paupera | Oleoresin | α-copaene (A 21.8%, B 38.8%), trans-caryophyllene (A 4.1%, B 21.4%), γ-cadinene (B 7.7%), caryophyllene oxide (A 12.5%), diterpene kaurene (A 33.2%, B 2.4%) | [132] |
| Coriandrum sativum, L. | Coriander | Linalool (60–80%), geraniol (1.2%-4.6%), terpinen-4-ol (3%), α-terpineol (0.5%), γ-terpinene (1–8%), r-cymene (3.5%), limonene (0.5%-4.0%), α-pinene (0.2%-8.5%), camphene (1.4%), myrcene (0.2%-2.0%), camphor (0.9%-4.9%), geranyl acetate (0.1%-4.7%), linalyl acetate (0%-2.7%) | [135] |
| Cymbopogon citratus | Lemon grass | citral α (40.8%), citral β (32%), nerol (4.18%), geraniol (3.04%), citronellal (2.10%), terpinolene (1.23%), geranyl acetate (0.83%), myrecene (0.72%), terpinol (0.45%), methylheptenone (0.2%), borneol (0.1–0.4%), linalyl acetate (0.1%), α-pinene (0.07%), β-pinene (0.04%), limonene (traces), linalool (traces) | [140] |
| Cymbopogon winterianus | Citronella grass | Myrcene (3.3%), limonene (2.2%), (E)-β-ocimene 0.7%), allo-ocimene (0.2%), citronellal (26.5%), (E)-isocitral (0.2%), citronellol (7.3%), nerol (0.4%), neral (0.5%), geraniol (16.2%), geranial (0.7%), citronelyl acetate (2.5%), neryl acetate (0.1%), geranyl acetate (3.4%), β-elemene (4.4%), β-ylangene (0.3%), β-gurjunene (0.2%), aromadendrene (0.1%), α-humulene (0.1%), cis-cadina-1,(6),4-diene (0.1%), cis-muurola-4,(14),5-diene (0.1%), γ-muurolene (0.1%), germacrene D (1.1%), trans-muurola-4,(14),5-diene (0.1%), viridiflorene (0.1%), α-muurolene (0.4%), γ-cadinene(0.4%), δ-cadinene (2.5%), zonarene (0.1%), α-cadinene (0.1%), elemol (14.5%), 10-epi-γ-eudesmol (0.1%), 1-epi-cubenol (0.1%), γ-eudesmol (0.8%) epi-o-cadinol (0.5%), epi-o-muurolol (0.7%), β-eudesmol (0.2%), α-eudesmol (0.2%), α-cadinol (2.7%) | [142] |
| Dysphania graveolens | Fetid goosefoot | (Z,Z)-farnesol (16.01%), γ-terpineol (11.11%), cadine-4,11-dien-15-ol (8/34%), phytol (31.30%), ascaridol (32.21%), carvacrol (13.42%), carvacrol (17.45%), ascaridol (13.15%), p-cymene (12.79%) | [174] |
| Hyptis mutabilis | Tropical bushmint | α-thujene (1.6–6.0%), α-pinene (1.0–7.2%), 4(10)-thujene (2.027%), β-pinene (1.915–7.906%), 1-octen-3-ol (0.774–1.27%), limonene (1.34–1.731%), terpinene (0.199–0.715%), (+)-1-terpinen-4-ol (0.395–0.886%), α-copaene, (1.694–2.647%), β-bourbonene (0.787–1.532%), β-cubebene (0.693–1.172%), (-)-β-elemene (0.552–0.789%), E-caryophyllene (10.839–13.948%), α-caryophyllene (2.434–3.95%), germacrene D (6.936–14.968%), bicyclogermacrene (7.845–10.895%), germacrene A (0.591–0.906%), γ-cadinene (0.994%), cubebol (1.085–2.06%), (+)-δ-dadinene (1.188–2.166%), spathulenol (1.401%), germacrene D-4-ol (0.875%), (-)-globulol (11.604–26.61%), (-)-globulol (11.604–26.61%) | [144] |
| Lavandula angustifolia | English lavender | Linalool (33.3%), linalyl acetate (38.5%), caryophyllene (3.9%), myrcene (3.9%), trans-ocimene (2.4%), lavandulyl acetate (2.2%), terpinen-4-ol (2.1%) | [148] |
| Linalyl acetate, linalool, (Z) β—ocimene, lavandulyl acetate, terpinen-4-ol | [6] | ||
| Lippia alba | Bushy lippia | Citral, linalool, β-caryophyllene; tagetenone; limonene, carvone, dihydrocarvone, piperitone, piperitenone; myrcene; γ-terpinene; camphor-1,8-cineole; estragole | [175] |
| Matricaria chamomilla | Chamomile | Isobutyl angelate, isoamyl angelate, methallyl angelate, isobutyl isobutyrate, methylpentyl angelate, trans-pinocarveol, pinocarvone | [6] |
| Melissa officinalis, L. | Common balm; lemon balm | Geranial (44.20%), neral (30.20%), citronellal (6.30%) | [158] |
| Mentha x piperita | Peppermint | Menthol, menthone, (+/-)-menthyl acetate, 1,8-cineole, limonene, β-pinene, β-caryophyllene | [159] |
| Nigella sativa, L. | Black cumin | Thymoquinone (63%), p-cymene (23%), α-pinene (<14%) | [169] |
| Piper guineense | Pepper | Linalool (41.8%), 3,5-dimethoxytoluene (10.9%) | [161] |
| Rosmarinus officinalis | Rosemary | p-Cymene (44.02%), linalool (20.5%), γ-terpinene (16.62%), thymol (1.81%), β-pinene (3.61%), α-pinene (2,83%), eucalyptol (2.64%), β-caryophyllene (0.11%) | [49] |
| Salvia officinalis | Common sage | 1,8-cineole (7.45–9.69%), camphor (18.08–25.11%), linalool 0.31–0.51%), α-thujone (21.48–22.19%), β-thujone (8.78–17.70%), α-pinene (0.20–6.48%), β-pinene (1.55–3.74%), camphene (3.45–7.09%), myrcene (1.13–1.97%), limonene (1.87–1.91%), γ-terpinene (0.13–1.66%), terpinolene (0.34–0.87%), bormeol (1.60–2.88%), bormyl acetate (0.41–0.74%), β-caryophyllene (2.70–6.18%), α-humulene (2.53–4.21%), caryophyllene oxide (0.08–0.62%), viridiflorol (0.13–0.19%) | [176] |
| Salvia sclarea | Clary sage | Linalyl acetate (31.07%), linalool (23.11%), α-terpineol (7.03%), geranyl acetate (3.53%), sclareol (3.34%), germacrene D (2.92%), spathulenol (2.16%), caryophyllene oxide (2.01%), neryl acetate (1.87%), trans-β-caryophyllene (1.72%), β-myrcene (1.65%), geraniol (1.21%), nerol (1.02%), α-eudesmol (0.78%), limonene (0.58%), manool (0.57%), cis-β-ocimene (0.55%), α- and β-thujone (0.09%) | [177] |
| Syzygium aromaticum | Clove | Eugenol (71.56%), eugenol acetate (8.99%), caryophyllene oxide (1.67%), nootkatin (1.05%), phenol-4-(2,3-dihydro-7-methoxy-3-methyl-5-(1-propenyl)-2-benzofurane (0.98%), p-cymene (0.9%), guaiol (0.90%), thymol (0.87%), isolongifolanone (trans) (0.86%), 5-hexene-2-one (0.67%), benzene-1-butylheptyl (0.55%), hexadecanoic acid (0.50%), 9,17-octadeca-dienal (0.24%), octadecanoic acid butyl ester (0.33%), dodecatrienoic acid-3,7,11-trimethylethyl ester (0.38%), vitamin E acetate (0.43%) | [165] |
| Tagetes minuta, L. | Black mint | β-phelandrene, limonene, β-cimene, dihydrotagetone, tagetone, tagetenone | [178] |
| Thymus capitatus | Spanish oregano | α-thujene (1.64–7.92%), anisole (1.24%), p-cymene (3.71%), trans-β-ocimene (0.07–1.03%), γ-terpinene (0.76–16.18%), α-terpinolene (0.25–0.5%), 2-ethyl-4-methyl anisole (2.63%), thymol (0.07–1.77%), carvacrol (24.28%-58.56%), phenol,2,3,5,6-tetra-methyl (2.18–5.54%), hexanoic acid, hexyl ester (0.84%), β-caryophyllene (7.41–8.59%), aromadendrene (0.78%), α-humulene (1.82–5.20%), ledene/viridiflorene (1.63–6.57%), cis-α bisabolene (0.71–1.07%), γ-cadinene (0.69–1.93%), δ-cadinene (0.79%), (-)-spathulenol (1.03%), caryophyllene oxide (6.26–10.43%), iso aromadendrene epoxide (0.82–2.27%), α cadinol (0.25%), vulgarol B (1.11%) | [169] |
| Valeriana officinalis, L. | Valerian | Carene (0.29%), α-thujene (4.18%), 6-isopropyl-1-methyl bicycle[3,1,0]hexane (14.19%), sabinene (2.55%), p-cymene (0.43%), limonene (1.26%), camphor (0.19%), borneol (3.54%), L-myrtanol (0.81%), α-methyl 4(1′,1′-methyl ethyl) phenol (2.49%), bornyl acetate (23.93%), sabinol (1.70%), α-terpineol (1.20%), β-caryophyllene (0.82%), β-gurjunene (1.16%), humulene (0.40%), trans-caryophyllene (0.28%), nerolidol (0.78%), elemene (0.45%), bornyl isovalerianate (0.36%), azulene furan (0.58%), stereoisomer of ramie enol (1.46%), 4a,8-dimethyl-α-isopropyl naphthyl ketone (2.77%), tetramethyl-4-hydroxyl cyclopropane naphthalene (1.26%), ledol (1.22%), guaiol (4.73%), valerone (1.14%) | [179] |
| Chemical Compound | Classification | Species Tested | Application and Results | References |
|---|---|---|---|---|
| 13-acetyl solstitialin | Sesquiterpene | Review | Anti-cancer | [180] |
| 8α-Acetoxyzaluzanin C | Sesquiterpene | Review | Anti-microbial, anti-viral | |
| Angeloylenolin | Sesquiterpene | Review | Anti-proliferative | |
| Arglabin | Sesquiterpene | Review | Anti-cancer, anti-malarial | |
| Arguerin B | Sesquiterpene | Review | Anti-cancer | |
| Artemisin | Sesquiterpene | Review | Anti-cancer, anti-malarial | |
| α-asarone | Rats | Anxiolytic like effect; alleviated epilepsy; anti-depressant | [92,99,101] | |
| β-asarone | Anti-depressant | [100,101] | ||
| (-)-Borneol | Monoterpene | Review | Anti-bacterial | [181] |
| (+)-Borneol | Monoterpene | Review | Anti-bacterial | [181] |
| Camphor | Monoterpene | Rats | Anti-bacterial | [181] |
| Carotenoids | Tetraterpene | Review | Anti-oxidant, photoprotectant | [182] |
| Carvacrol | Monoterpene | Mice | Anxiolytic like effect; anti-microbial, pro- and anti-apoptotic | [92,180] |
| L-Carveol | Monoterpene | Review | Anti-bacterial; anti-oxidant | [104,181] |
| L-Carvone | Monoterpene | Review | Anti-bacterial | [181] |
| β–Caryophyllene | Sesquiterpene | Mouse | Improved re-epithelialization of cutaneous wound, stimulated cell proliferation and migration; Anti-inflammatory, analgesic; ligand of CB2; Anti-carcinogenic; Anxiolytic-like effect; allelopathic | [2,5,92,180] |
| Centaurepensin A | Sesquiterpene | Review | Anti-microbial, anti-viral | [180] |
| Chlorocyanerin | Sesquiterpene | Review | Anti-microbial, anti-viral | [180] |
| 1,4-Cineole | Oxides | Review | Anxiolytic-like effect | [92] |
| 1,8-Cineole | Oxides | Humans; rat | Improved cognitive function; increased skin flap survival | [52,183] |
| Citral | Monoterpene | Mouse, silver catfish | Enhanced microbiota, especially of Lactobacillus, in the gut; anxiolytic-like effect; anti-microbial, anti-inflammatory, anti-cancer; anti-bacterial; anesthetic | [71,92,152,153,180,181] |
| Citronellal | Monoterpene | Mouse | Stimulated glucagon-like peptide 1 secretion in mouse intestinal tissues and in cultured enteroendocrine cells; anti-bacterial | [62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179,180,181] |
| Cnicin | Sesquiterpene | Review | Anti-microbial | [180] |
| Coronopilin | Sesquiterpene | Humans | Anti-cancer | [180] |
| Cyclohexyl salicylate | Stimulated cytokinesis, cell migration, regeneration of keratinocytes, phosphorylation of AKT and Chk-2, and secretion of IL-1; ligand of OR2A4/7 | [67] | ||
| m-Cymene | Monoterpene | Review | Anti-bacterial | [181] |
| p-Cymene | Monoterpene | Review | Analgesic, anti-inflammatory, anti-nociceptive | [184] |
| Cynaropicrin | Sesquiterpene | Review | Anti-cancer | [180] |
| Eugenol | Monoterpene | Review | Anti-bacterial | [181] |
| Eupalinin | Sesquiterpene | Review | Anti-cancer | [180] |
| β-cis-farnesene | Sesquiterpene | Review | Allelopathic | [180] |
| Geraniol | Monoterpene | Mouse | Stimulated glucagon-like peptide 1 secretion in mouse intestinal tissues and in cultured enteroendocrine cells; Strong analgesic impact as morphine, measured by the latency to show writhing after intraperitoneal injection of acetic acid; IP injection; anti-depressant impact, measured by the immobility time in forced swimming tests and tail suspension tests, lower corticosterone level; gavage; anti-tumor, anti-microbial, anti-oxidant, anti-inflammatory | [62,77,78,79,180,181] |
| Helenalin | Sesquiterpene | Review | Anti-cancer | [180] |
| Humulene | Sesquiterpene | Suppressd hepatocellular carcinoma cell proliferation | [185] | |
| β –ionone | Humans | Melanin synthesis; ligand of OR51E2 | [66] | |
| Isomontanolide | Sesquiterpene | Review | Anti-microbial, anti-biofilm | [180] |
| Isononyl alcohol | Humans | Stimulated cell migration, phosphorylation of Hsp27, AMPK1, and p38MAPK, and secretion of IL-6; ligand of OR51B5 | [67] | |
| Linalool | Monoterpene | Mouse Silver catfish | Anxyolitic effect; inhalation enhanced the microbiota, especially of Lactobacillus, in the gut; anti-bacterial; sedative, anesthetic effect | [3,71,151,152,153,181] |
| Limonene | Monoterpene | Mouse; review | Enhanced the microbiota, especially of Lactobacillus, in the gut, Ligand of Olfr56 (human homolog, OR2V1); anti-depressant, anti-nociceptive, anti-diabetic, anti-ulcerogenic | [71,180] |
| Menthol | Monoterpene | Review | Anti-microbial, radioprotective, anti-oxidant, analgesic; | [180] |
| Montanolide | Sesquiterpene | Review | Anti-microbial, anti-biofilm | [180] |
| Myrcene | Monoterpene | Mouse | Anxiolytic-like effect; analgesic, anti-inflammatory, anti-oxidant, anti-bacterial | [92,180] |
| Ocimene | Monoterpene | Review | Wound healing, anti-inflammatory, abolished or reduced edema, hyperemia, laceration, hemorrhage; anti-microbial | [180,181] |
| Parthenolide | Sesquiterpene | Review | Anti-inflammatory | [180] |
| Perillyl alcohol | In vitro | Anti-oxidant | [104] | |
| 2-Phenylethanol | Mouse | Decreased immobility time in tail suspension test | [48] | |
| α–Pinene | Monoterpene | Rat; review | Increased survival of skin flap; anxiolytic-like effect; anti-microbial | [92,180,181,183] |
| β–Pinene | Monoterpene | Review | Anti-microbial | [181] |
| Sandalore | Synthetic sandalwood odorant | Humans | Stimulated chemotaxis in human keratinocyte cells; ligand of OR2AT4; Suppressed apoptosis and enhanced longevity of hair; ligand of OR2AT4 | [59,63] |
| Terpineol | Monoterpene | Review | Anti-bacterial | [181] |
| Tanachin | Sesquiterpene | Review | Anti-bacterial | [180] |
| Tavulin | Sesquiterpene | Review | Anti-bacterial | [180] |
| Tourneforine | Sesquiterpene | Review | Cytotoxic | [180] |
| Thymol | Monoterpene | Review | Anti-fungal, anti-parasitic, anti-septic; anti-bacterial | [180,181] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koyama, S.; Heinbockel, T. The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application. Int. J. Mol. Sci. 2020, 21, 1558. https://doi.org/10.3390/ijms21051558
Koyama S, Heinbockel T. The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application. International Journal of Molecular Sciences. 2020; 21(5):1558. https://doi.org/10.3390/ijms21051558
Chicago/Turabian StyleKoyama, Sachiko, and Thomas Heinbockel. 2020. "The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application" International Journal of Molecular Sciences 21, no. 5: 1558. https://doi.org/10.3390/ijms21051558
APA StyleKoyama, S., & Heinbockel, T. (2020). The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application. International Journal of Molecular Sciences, 21(5), 1558. https://doi.org/10.3390/ijms21051558






