Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS
Abstract
1. Introduction
2. Data Collection and Methodology
3. Emerging Cardiometabolic Risk Biomarkers
4. Antiandrogen Therapy
4.1. Flutamide
4.2. Bicalutamide
4.3. Spironolactone
4.4. Ketoconazole
4.5. Finasteride
5. Antidiabetic Medications
5.1. Metformin
5.2. Inositols
5.3. Acarbose
5.4. Thiazolidinediones
5.5. Incretin Mimetics
5.6. SGLT2 Inhibitors
6. Metabolic Impact of Combined Oral Contraceptives
7. Statins
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABC | ATP-binding cassette transporter |
| AR | Androgen receptor |
| CEC | Cholesterol efflux capacity |
| CRP | C-reactive protein |
| CV | Cardiovascular |
| HDL | High-density lipoprotein |
| IR | Insulin resistance |
| LDL | Low-density lipoprotein |
| MR | Mineralocorticoid receptor |
| RAAS | Renin–angiotensin–aldosterone system |
| TG | Triglycerides |
References
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat. Rev. Dis. Primers 2016, 2, 16057. [Google Scholar] [CrossRef] [PubMed]
- Escobar-Morreale, H.F. Polycystic ovary syndrome: Definition, aetiology, diagnosis and treatment. Nat. Rev. Endocrinol. 2018, 14, 270–284. [Google Scholar] [CrossRef] [PubMed]
- McCartney, C.R.; Marshall, J.C. Clinical practice–polycystic ovary syndrome. N. Engl. J. Med. 2016, 375, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Solomon, C.G.; Hu, F.B.; Dunaif, A.; Rich-Edwards, J.E.; Stampfer, M.J.; Willett, W.C.; Speizer, F.E.; Manson, J.E. Menstrual cycle irregularity and risk for future cardiovascular disease. J. Clin. Endocrinol. Metab. 2002, 87, 2013–2017. [Google Scholar] [CrossRef]
- Osibogun, O.; Ogunmoroti, O.; Michos, E.D. Polycystic ovary syndrome and cardiometabolic risk: Opportunities for cardiovascular disease prevention. Trends Cardiovasc. Med. 2020, 30, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Kakoly, N.S.; Earnest, A.; Teede, H.J.; Moran, L.J.; Joham, A.E. The impact of obesity on the incidence of type 2 diabetes among women with polycystic ovary syndrome. Diabetes Care 2019, 42, 560–567. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Paterakis, T.; Alexandraki, K.; Piperi, C.; Aessopos, A.; Katsikis, I.; Katsilambros, N.; Kreatsas, G.; Panidis, D. Indices of low-grade chronic inflammation in polycystic ovary syndrome and the beneficial effect of metformin. Hum. Reprod. 2006, 21, 1426–1431. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Dunaif, A. Insulin resistance and the polycystic ovary syndrome revisited: An update on mechanisms and implications. Endocr. Rev. 2012, 33, 981–1030. [Google Scholar] [CrossRef]
- Teede, H.J.; Misso, M.L.; Costello, M.F.; Dokras, A.; Laven, J.; Moran, L.; Piltonen, T.; Norman, R.J. International PCOS Network. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil. Steril. 2018, 110, 364–379. [Google Scholar] [CrossRef]
- Agarwala, A.; Michos, E.D.; Samad, Z.; Ballantyne, C.M.; Virani, S.S. The use of sex-specific factors in the assessment of women’s cardiovascular risk. Circulation 2020, 141, 592–599. [Google Scholar] [CrossRef]
- Mulder, C.L.; Lassi, Z.S.; Grieger, J.A.; Ali, A.; Jankovic-Karasoulos, T.; Roberts, C.T.; Andraweera, P.H. Cardio-metabolic risk factors among young infertile women: A systematic review and meta-analysis. BJOG 2020, 127, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Bajuk Studen, K.; Pfeifer, M. Cardiometabolic risk in polycystic ovary syndrome. Endocr. Connect. 2018, 7, R238–R251. [Google Scholar] [CrossRef] [PubMed]
- Honold, L.; Nahrendorf, M. Resident and monocyte-derived macrophages in cardiovascular disease. Circ. Res. 2018, 122, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Bolego, C.; Cignarella, A.; Staels, B.; Chinetti-Gbaguidi, G. Macrophage function and polarization in cardiovascular disease: A role of estrogen signaling? Arterioscler. Thromb. Vasc. Biol. 2013, 33, 1127–1134. [Google Scholar] [CrossRef]
- Toniolo, A.; Fadini, G.P.; Tedesco, S.; Cappellari, R.; Vegeto, E.; Maggi, A.; Avogaro, A.; Bolego, C.; Cignarella, A. Alternative activation of human macrophages is rescued by estrogen treatment in vitro and impaired by menopausal status. J. Clin. Endocrinol. Metab. 2015, 100, E50–E58. [Google Scholar] [CrossRef]
- Rohatgi, A.; Khera, A.; Berry, J.D.; Givens, E.G.; Ayers, C.R.; Wedin, K.E.; Neeland, I.J.; Yuhanna, I.S.; Rader, D.R.; de Lemos, J.A.; et al. HDL cholesterol efflux capacity and incident cardiovascular events. N. Engl. J. Med. 2014, 371, 2383–2393. [Google Scholar] [CrossRef]
- Saleheen, D.; Scott, R.; Javad, S.; Zhao, W.; Rodrigues, A.; Picataggi, A.; Lukmanova, D.; Mucksavage, M.L.; Luben, R.; Billheimer, J.; et al. Association of HDL cholesterol efflux capacity with incident coronary heart disease events: A prospective case-control study. Lancet Diabetes Endocrinol. 2015, 3, 507–513. [Google Scholar] [CrossRef]
- Roe, A.; Hillman, J.; Butts, S.; Smith, M.; Rader, D.; Playford, M.; Mehta, N.N.; Dokras, A. Decreased cholesterol efflux capacity and atherogenic lipid profile in young women with PCOS. J. Clin. Endocrinol. Metab. 2014, 99, E841–E847. [Google Scholar] [CrossRef]
- Dokras, A.; Playford, M.; Kris-Etherton, P.M.; Kunselman, A.R.; Stetter, C.M.; Williams, N.I.; Gnatuk, C.L.; Estes, S.J.; Sarwer, D.B.; Allison, K.C.; et al. Impact of hormonal contraception and weight loss on high-density lipoprotein cholesterol efflux and lipoprotein particles in women with polycystic ovary syndrome. Clin. Endocrinol. 2017, 86, 739–746. [Google Scholar] [CrossRef]
- Tedesco, S.; Adorni, M.P.; Ronda, N.; Cappellari, R.; Mioni, R.; Barbot, M.; Pinelli, S.; Plebani, M.; Bolego, C.; Scaroni, C.; et al. Activation profiles of monocyte-macrophages and HDL function in healthy women in relation to menstrual cycle and in polycystic ovary syndrome patients. Endocrine 2019, 66, 360–369. [Google Scholar] [CrossRef]
- Gidwani, S.; Phelan, N.; McGill, J.; McGowan, A.; O’Connor, A.; Young, I.S.; Gibney, J.; McEneny, J. Polycystic ovary syndrome influences the level of serum amyloid A and activity of phospholipid transfer protein in HDL2 and HDL3. Hum. Reprod. 2014, 29, 1518–1525. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bonacina, F.; Da Dalt, L.; Catapano, A.L.; Norata, G.D. Metabolic adaptations of cells at the vascular-immune interface during atherosclerosis. Mol. Asp. Med. 2020, 100918. [Google Scholar] [CrossRef] [PubMed]
- Albu, A.; Radian, S.; Fica, S.; Barbu, C.G. Biochemical hyperandrogenism is associated with metabolic syndrome independently of adiposity and insulin resistance in Romanian polycystic ovary syndrome patients. Endocrine 2015, 48, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Mioni, R.; Dalla Cà, A.; Turra, J.; Azzolini, S.; Xamin, N.; Bleve, L.; Maffei, P.; Vettor, R.; Fallo, F. Hyperinsulinemia and obese phenotype differently influence blood pressure in young normotensive patients with polycystic ovary syndrome. Endocrine 2017, 55, 625–634. [Google Scholar] [CrossRef]
- Scarpitta, A.M.; Sinagra, D. Polycystic ovary syndrome: An endocrine and metabolic disease. Gynecol. Endocrinol. 2000, 14, 392–395. [Google Scholar] [CrossRef]
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 2004, 81, 19–25. [Google Scholar] [CrossRef]
- Wild, R.A.; Carmina, E.; Diamanti-Kandarakis, E.; Dokras, A.; Escobar-Morreale, H.F.; Futterweit, W.; Lobo, R.; Norman, R.J.; Talbott, E.; Dumesic, D.A. Assessment of cardiovascular risk and prevention of cardiovascular disease in women with the polycystic ovary syndrome: A consensus statement by the Androgen Excess and Polycystic Ovary Syndrome (AE-PCOS) Society. J. Clin. Endocrinol. Metab. 2010, 95, 2038–2049. [Google Scholar] [CrossRef]
- Goodman, N.F.; Cobin, R.H.; Futterweit, W.; Glueck, J.S.; Legro, R.S.; Carmina, E.; American Association of Clinical Endocrinologists (AACE); American College of Endocrinology (ACE); Androgen Excess and PCOS Society. American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society Disease State Clinical Review: Guide to the best practices in the evaluation and treatment of polycystic ovary syndrome-Part 2. Endocr. Pract. 2015, 21, 1415–1426. [Google Scholar] [CrossRef]
- Saslow, L.R.; Aikens, J.E. Lifestyle interventions for polycystic ovary syndrome: Cross-sectional survey to assess women’s treatment and outcome preferences. JMIR Form. Res. 2020, 4, e17126. [Google Scholar] [CrossRef]
- De Niet, J.E.; de Koning, C.M.; Pastoor, H.; Duivenvoorden, H.J.; Valkenburg, O.; Ramakers, M.J.; Passchier, J.; de Klerk, C.; Laven, J.S. Psychological well-being and sexarche in women with polycystic ovary syndrome. Hum. Reprod. 2010, 25, 1497–1503. [Google Scholar] [CrossRef]
- Hahn, S.; Janssen, O.E.; Tan, S.; Pleger, K.; Mann, K.; Schedlowski, M.; Kimmig, R.; Benson, S.; Balamitsa, E.; Elsenbruch, S. Clinical and psychological correlates of quality-of-life in polycystic ovary syndrome. Eur. J. Endocrinol. 2005, 153, 853–860. [Google Scholar] [CrossRef]
- Shet, M.S.; Mc Phaul, M.; Fisher, C.W.; Stallings, N.R.; Estabrook, R.W. Metabolism of the antiandrogenic drug (flutamide) by human CYP1A2. Drug Metab. Dispos. 1997, 25, 1298–1303. [Google Scholar]
- Cusan, L.; Dupont, A.; Gomez, J.L.; Tremblay, R.R.; Labrie, F. Comparison of flutamide and spironolactone in the treatment of hirsutism: A randomized controlled trial. Fertil. Steril. 1994, 61, 281–287. [Google Scholar] [CrossRef]
- Marcondes, J.A.; Minnani, S.L.; Luthold, W.W.; Wajchenberg, B.L.; Samojlik, E.; Kirschner, M.A. Treatment of hirsutism in women with flutamide. Fertil. Steril. 1992, 57, 543–547. [Google Scholar] [CrossRef]
- Brahm, J.; Brahm, M.; Segovia, R.; Latorre, R.; Zapata, R.; Poniachik, J.; Buckel, E.; Contreras, L. Acute and fulminant hepatitis induced by flutamide: Case series report and review of the literature. Ann. Hepatol. 2011, 10, 93–98. [Google Scholar]
- Giorgetti, R.; di Muzio, M.; Giorgetti, A.; Girolami, D.; Borgia, L.; Tagliabracci, A. Flutamide-induced hepatotoxicity: Ethical and scientific issues. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 69–77. [Google Scholar] [PubMed]
- Generali, J.A.; Cada, D.J. Flutamide: Hirsutism in women. Hosp. Pharm. 2014, 49, 517–520. [Google Scholar] [CrossRef]
- De Zegher, F.; Ibáñez, L. Therapy: Low-dose flutamide for hirsutism: Into the limelight, at last. Nat. Rev. Endocrinol. 2010, 6, 421–422. [Google Scholar] [CrossRef]
- Paradisi, R.; Venturoli, S. Retrospective observational study on the effects and tolerability of flutamide in a large population of patients with various kinds of hirsutism over a 15-year period. Eur. J. Endocrinol. 2010, 163, 139–147. [Google Scholar] [CrossRef]
- Castelo-Branco, C.; Moyano, D.; Gómez, O.; Balasch, J. Long-term safety and tolerability of flutamide for the treatment of hirsutism. Fertil. Steril. 2009, 91, 1183–1188. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Mitrakou, A.; Raptis, S.; Tolis, G.; Duleba, A.J. The effect of a pure antiandrogen receptor blocker, flutamide, on the lipid profile in the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1998, 83, 2699–2705. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sahin, I.; Serter, R.; Karakurt, F.; Demirbas, B.; Culha, C.; Taskapan, C.; Kosar, F.; Aral, Y. Metformin versus flutamide in the treatment of metabolic consequences of non-obese young women with polycystic ovary syndrome: A randomized prospective study. Gynecol. Endocrinol. 2004, 19, 115–124. [Google Scholar] [CrossRef]
- Gambineri, A.; Pelusi, C.; Genghini, S.; Morselli-Labate, A.M.; Cacciari, M.; Pagotto, U.; Pasquali, R. Effect of flutamide and metformin administered alone or in combination in dieting obese women with polycystic ovary syndrome. Clin. Endocrinol. 2004, 60, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, L.; de Zegher, F. Low-dose flutamide-metformin therapy for hyperinsulinemic hyperandrogenism in non-obese adolescents and women. Hum. Reprod. Update 2006, 12, 243–252. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Birdsall, M.A.; Farquhar, C.M.; White, H.D. Association between polycystic ovaries and extent of coronary artery disease in women having cardiac catheterization. Ann. Int. Med. 1997, 126, 32–35. [Google Scholar] [CrossRef] [PubMed]
- McCredie, R.J.; McCrohon, J.A.; Turner, L.; Griffiths, K.A.; Handelsman, D.J.; Celermajer, D.S. Vascular reactivity is impaired in genetic females taking high-dose androgens. J. Am. Coll. Cardiol. 1998, 32, 1331–1335. [Google Scholar] [CrossRef]
- Gangula, P.R.; Reed, L.; Yallampalli, C. Antihypertensive effects of flutamide in rats that are exposed to a low-protein diet in utero. Am. J. Obstet. Gynecol. 2005, 192, 952–960. [Google Scholar] [CrossRef]
- Hurliman, A.; Keller Brown, J.; Maille, N.; Mandala, M.; Casson, P.; Osol, G. Hyperandrogenism and insulin resistance, not changes in body weight, mediate the development of endothelial dysfunction in a female rat model of polycystic ovary syndrome (PCOS). Endocrinology 2015, 156, 4071–4080. [Google Scholar] [CrossRef]
- Ajossa, S.; Guerriero, S.; Paoletti, A.M.; Orrù, M.; Melis, G.B. The antiandrogenic effect of flutamide improves uterine perfusion in women with polycystic ovary syndrome. Fertil. Steril. 2002, 77, 1136–1140. [Google Scholar] [CrossRef]
- Ricci, F.; Buzzatti, G.; Rubagotti, A.; Boccardo, F. Safety of antiandrogen therapy for treating prostate cancer. Expert Opin. Drug Saf. 2014, 13, 1483–1499. [Google Scholar] [CrossRef]
- Ismail, F.F.; Meah, N.; Trindade de Carvalho, L.; Bhoyrul, B.; Wall, D.; Sinclair, R. Safety of oral bicalutamide in female pattern hair loss: A retrospective review of 316 patients. J. Am. Acad. Dermatol. 2020, 83, 1478–1479. [Google Scholar] [CrossRef] [PubMed]
- Bahceci, M.; Tuzcu, A.; Canoruc, N.; Tuzun, Y.; Kidir, V.; Aslan, C. Serum C-reactive protein (CRP) levels and insulin resistance in non-obese women with polycystic ovarian syndrome, and effect of bicalutamide on hirsutism, CRP levels and insulin resistance. Horm. Res. 2004, 62, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Moretti, C.; Guccione, L.; Di Giacinto, P.; Simonelli, I.; Exacoustos, C.; Toscano, V.; Motta, C.; De Leo, V.; Petraglia, F.; Lenzi, A. Combined oral contraception and bicalutamide in polycystic ovary syndrome and severe hirsutism: A double-blind randomized controlled trial. J. Clin. Endocrinol. Metab. 2018, 103, 824–838. [Google Scholar] [CrossRef] [PubMed]
- Christy, N.A.; Franks, A.S.; Cross, L.B. Spironolactone for hirsutism in polycystic ovary syndrome. Ann. Pharmacother. 2005, 39, 1517–1521. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Farquhar, C.; Lee, O.; Toomath, R.; Jepson, R.G. Spironolactone versus placebo or in combination with steroids for hirsutism and/or acne. Cochrane Database Syst. Rev. 2009, 15, CD000194. [Google Scholar] [CrossRef] [PubMed]
- Moghetti, P.; Tosi, F.; Tosti, A.; Negri, C.; Misciali, C.; Perrone, F.; Caputo, M.; Muggeo, M.; Castello, R. Comparison of spironolactone, flutamide, and finasteride efficacy in the treatment of hirsutism: A randomized, double blind, placebo-controlled trial. J. Clin. Endocrinol. Metab. 2000, 85, 89–94. [Google Scholar] [CrossRef]
- Spritzer, P.M.; Lisboa, K.O.; Mattiello, S.; Lhullier, F. Spironolactone as a single agent for long-term therapy of hirsute patients. Clin. Endocrinol. 2000, 52, 587–594. [Google Scholar] [CrossRef]
- Armanini, D.; Andrisani, A.; Bordin, L.; Sabbadin, C. Spironolactone in the treatment of polycystic ovary syndrome. Expert Opin. Pharmacother. 2016, 17, 1713–1715. [Google Scholar] [CrossRef]
- Studen, K.B.; Sebestjen, M.; Pfeifer, M.; Prezelj, J. Influence of spironolactone treatment on endothelial function in non-obese women with polycystic ovary syndrome. Eur. J. Endocrinol. 2011, 164, 389–395. [Google Scholar] [CrossRef]
- Armeni, E.; Stamatelopoulos, K.; Rizos, D.; Georgiopoulos, G.; Kazani, M.; Kazani, A.; Kolyviras, A.; Stellos, K.; Panoulis, K.; Alexandrou, A.; et al. Arterial stiffness is increased in asymptomatic nondiabetic postmenopausal women with a polycystic ovary syndrome phenotype. J. Hypertens 2013, 31, 1998–2004. [Google Scholar] [CrossRef]
- Zieman, S.J.; Melenovsky, V.; Kass, D.A. Mechanisms, pathophysiology, and therapy of arterial stiffness. Arterioscler Thromb. Vasc Biol. 2005, 25, 932–943. [Google Scholar] [CrossRef] [PubMed]
- Muneyyirci-Delale, O.; Co, S.; Winer, N. Vascular compliance in women with polycystic ovary syndrome treated with spironolactone. J. Clin. Hypertens 2018, 20, 1536–1540. [Google Scholar] [CrossRef] [PubMed]
- Sonino, N. The use of ketoconazole as an inhibitor of steroid production. N. Engl. J. Med. 1987, 317, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Sonino, N.; Boscaro, M.; Merola, G.; Mantero, F. Prolonged treatment of Cushing’s disease by ketoconazole. J. Clin. Endocrinol. Metab. 1985, 61, 718–722. [Google Scholar] [CrossRef]
- Cedeno, J.; Mendoza, S.G.; Velazquez, E.; Nucete, H.; Speirs, J.; Glueck, C.J. Effect of ketoconazole on plasma sex hormones, lipids, lipoproteins, and apolipoproteins in hyperandrogenic women. Metabolism 1990, 39, 511–517. [Google Scholar] [CrossRef]
- Lewis, J.H.; Zimmerman, H.J.; Benson, G.D.; Ishak, K.G. Hepatic injury associated with ketoconazole therapy–analysis of 33 cases. Gastroenterology 1984, 86, 503–513. [Google Scholar] [CrossRef]
- Hu, A.C.; Chapman, L.W.; Mesinkovska, N.A. The efficacy and use of finasteride in women: A systematic review. Int. J. Dermatol. 2019, 58, 759–776. [Google Scholar] [CrossRef]
- Kohler, C.; Tschumi, K.; Bodmer, C.; Schneiter, M.; Birkhaeuser, M. Effect of finasteride 5 mg (Proscar) on acne and alopecia in female patients with normal serum levels of free testosterone. Gynecol. Endocrinol. 2007, 23, 142–145. [Google Scholar] [CrossRef]
- Falsetti, L.; De Fusco, D.; Eleftheriou, G.; Rosina, B. Treatment of hirsutism by finasteride and flutamide in women with polycystic ovary syndrome. Gynecol. Endocrinol. 1997, 11, 251–257. [Google Scholar] [CrossRef]
- Lakryc, E.M.; Motta, E.L.; Soares, J.M., Jr.; Haidar, M.A.; de Lima, G.R.; Baracat, E.C. The benefits of finasteride for hirsute women with polycystic ovary syndrome or idiopathic hirsutism. Gynecol. Endocrinol. 2003, 17, 57–63. [Google Scholar] [CrossRef]
- Zwadlo, C.; Schmidtmann, E.; Szaroszyk, M.; Kattih, B.; Froese, N.; Hinz, H.; Schmitto, J.D.; Widder, J.; Batkai, S.; Bähre, H.; et al. Antiandrogenic therapy with finasteride attenuates cardiac hypertrophy and left ventricular dysfunction. Circulation 2015, 131, 1071–1081. [Google Scholar] [CrossRef] [PubMed]
- Froese, N.; Wang, H.; Zwadlo, C.; Wang, Y.; Grund, A.; Gigina, A.; Hofmann, M.; Kilian, K.; Scharf, G.; Korf-Klingebiel, M.; et al. Anti-androgenic therapy with finasteride improves cardiac function, attenuates remodeling and reverts pathologic gene-expression after myocardial infarction in mice. J. Mol. Cell Cardiol. 2018, 122, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Venturoli, S.; Marescalchi, O.; Colombo, F.M.; Macrelli, S.; Ravaioli, B.; Bagnoli, A.; Paradisi, R.; Flamigni, C. A prospective randomized trial comparing low dose flutamide, finasteride, ketoconazole, and cyproterone acetate-estrogen regimens in the treatment of hirsutism. J. Clin. Endocrinol. Metab. 1999, 84, 1304–1310. [Google Scholar] [CrossRef] [PubMed]
- Diri, H.; Bayram, F.; Simsek, Y.; Caliskan, Z.; Kocer, D. Comparison of finasteride, metformin, and finasteride plus metformin in PCOS. Acta Endocrinol. 2017, 13, 84–89. [Google Scholar] [CrossRef]
- Legro, R.S.; Arslanian, S.A.; Ehrmann, D.A.; Hoeger, K.M.; Murad, M.H.; Pasquali, R.; Welt, C.K. Endocrine Society. Diagnosis and treatment of polycystic ovary syndrome: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2013, 98, 4565–4592. [Google Scholar] [CrossRef]
- Pasquali, R. Metformin in women with PCOS, pros. Endocrine 2015, 48, 422–426. [Google Scholar] [CrossRef]
- Abdalla, M.A.; Deshmukh, H.; Atkin, S.; Sathyapalan, T. A review of therapeutic options for managing the metabolic aspects of polycystic ovary syndrome. Ther. Adv. Endocrinol. Metab. 2020, 11, 2042018820938305. [Google Scholar] [CrossRef]
- Pasquali, R.; Gambineri, A.; Biscotti, D.; Vicennati, V.; Gagliardi, L.; Colitta, D.; Fiorini, S.; Cognigni, G.E.; Filicori, M.; Morselli-Labate, A.M. Effect of long-term treatment with metformin added to hypocaloric diet on body composition, fat distribution, and androgen and insulin levels in abdominally obese women with and without the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2000, 85, 2767–2774. [Google Scholar] [CrossRef]
- Naderpoor, N.; Shorakae, S.; de Courten, B.; Misso, M.L.; Moran, L.J.; Teede, H.J. Metformin and lifestyle modification in polycystic ovary syndrome: Systematic review and meta-analysis. Hum. Reprod. Update 2015, 21, 560–574. [Google Scholar] [CrossRef]
- Domecq, J.P.; Prutsky, G.; Mullan, R.J.; Hazem, A.; Sundaresh, V.; Elamin, M.B.; Phung, O.J.; Wang, A.; Hoeger, K.; Pasquali, R.; et al. Lifestyle modification programs in polycystic ovary syndrome: Systematic review and meta- analysis. J. Clin. Endocrinol. Metab. 2013, 98, 4655–4663. [Google Scholar] [CrossRef]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M. Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Palomba, S.; Falbo, A.; Russo, T.; Manguso, F.; Tolino, A.; Zullo, F.; De Feo, P.; Orio, F., Jr. Insulin sensitivity after metformin suspension in normal-weight women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2007, 92, 3128–3135. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, R.; Gambineri, A. Targeting insulin sensitivity in the treatment of polycystic ovary syndrome. Expert Opin. Ther. Targets 2009, 13, 1205–1226. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, A.; Morley, L.C.; Tang, T.; Norman, R.J.; Balen, A.H. Metformin for ovulation induction (excluding gonadotrophins) in women with polycystic ovary syndrome. Cochrane Database Syst. Rev. 2019, 12, CD01350. [Google Scholar] [CrossRef] [PubMed]
- Viollet, B.; Guigas, B.; Sanz Garcia, N.; Leclerc, J.; Foretz, M.; Andreelli, F. Cellular and molecular mechanisms of metformin: An overview. Clin. Sci. 2012, 122, 253–270. [Google Scholar] [CrossRef] [PubMed]
- Barbot, M.; Ceccato, F.; Scaroni, C. Diabetes mellitus secondary to Cushing’s disease. Front. Endocrinol. 2018, 9, 284. [Google Scholar] [CrossRef] [PubMed]
- Bizzarri, M.; Fuso, A.; Dinicola, S.; Cucina, A.; Bevilacqua, A. Pharmacodynamics and pharmacokinetics of inositol(s) in health and disease. Expert Opin. Drug Metab. Toxicol. 2016, 12, 1181–1196. [Google Scholar] [CrossRef]
- Romero, G.; Larner, J. Insulin mediators and the mechanism of insulin action. Adv. Pharmacol. 1993, 24, 21–50. [Google Scholar] [CrossRef]
- Nestler, J.E.; Unfer, V. Reflections on inositol(s) for PCOS therapy: Steps toward success. Gynecol. Endocrinol. 2015, 31, 501–505. [Google Scholar] [CrossRef]
- Gateva, A.; Unfer, V.; Kamenov, Z. The use of inositol(s) isomers in the management of polycystic ovary syndrome: A comprehensive review. Gynecol. Endocrinol. 2018, 34, 545–550. [Google Scholar] [CrossRef]
- Baillargeon, J.P.; Diamanti-Kandarakis, E.; Ostlund, R.E., Jr.; Apridonidze, T.; Iuorno, M.J.; Nestler, J.E. Altered D-chiro-inositol urinary clearance in women with polycystic ovary syndrome. Diabetes Care. 2006, 29, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Baillargeon, J.P.; Nestler, J.E.; Ostlund, R.E.; Apridonidze, T.; Diamanti-Kandarakis, E. Greek hyperinsulinemic women, with or without polycystic ovary syndrome, display altered inositols metabolism. Hum. Reprod. 2008, 23, 1439–1446. [Google Scholar] [CrossRef] [PubMed]
- Larner, J. D-chiro-inositol–its functional role in insulin action and its deficit in insulin resistance. Int. J. Exp. Diabetes Res. 2002, 3, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Nestler, J.E.; Jakubowicz, D.J.; Reamer, P.; Gunn, R.D.; Allan, G. Ovulatory and metabolic effects of D-chiro-inositol in the polycystic ovary syndrome. N. Engl. J. Med. 1999, 340, 1314–1320. [Google Scholar] [CrossRef]
- Iuorno, M.J.; Jakubowicz, D.J.; Baillargeon, J.P.; Dillon, P.; Gunn, R.D.; Allan, G.; Nestler, J.E. Effects of d-chiro-inositol in lean women with the polycystic ovary syndrome. Endocr. Pract. 2002, 8, 417–423. [Google Scholar] [CrossRef]
- Gerli, S.; Papaleo, E.; Ferrari, A.; Di Renzo, G.C. Randomized, double blind placebo-controlled trial: Effects of myo-inositol on ovarian function and metabolic factors in women with PCOS. Eur. Rev. Med. Pharmacol. Sci. 2007, 11, 347–354. [Google Scholar]
- Genazzani, A.D.; Lanzoni, C.; Ricchieri, F.; Jasonni, V.M. Myo-inositol administration positively affects hyperinsulinemia and hormonal parameters in overweight patients with polycystic ovary syndrome. Gynecol. Endocrinol. 2008, 24, 139–144. [Google Scholar] [CrossRef]
- Donà, G.; Sabbadin, C.; Fiore, C.; Bragadin, M.; Giorgino, F.L.; Ragazzi, E.; Clari, G.; Bordin, L.; Armanini, D. Inositol administration reduces oxidative stress in erythrocytes of patients with polycystic ovary syndrome. Eur. J. Endocrinol. 2012, 166, 703–710. [Google Scholar] [CrossRef]
- Chiu, T.T.; Rogers, M.S.; Law, E.L.; Briton-Jones, C.M.; Cheung, L.P.; Haines, C.J. Follicular fluid and serum concentrations of myo-inositol in patients undergoing IVF: Relationship with oocyte quality. Hum. Reprod. 2002, 17, 1591–1596. [Google Scholar] [CrossRef]
- Papaleo, E.; Unfer, V.; Baillargeon, J.P.; Fusi, F.; Occhi, F.; De Santis, L. Myo-inositol may improve oocyte quality in intracytoplasmic sperm injection cycles–a prospective, controlled, randomized trial. Fertil. Steril. 2009, 91, 1750–1754. [Google Scholar] [CrossRef]
- Unfer, V.; Carlomagno, G.; Rizzo, P.; Raffone, E.; Roseff, S. Myo-inositol rather than D-chiro-inositol is able to improve oocyte quality in intracytoplasmic sperm injection cycles–a prospective, controlled, randomized trial. Eur. Rev. Med. Pharmacol. Sci. 2011, 15, 452–457. [Google Scholar] [PubMed]
- Isabella, R.; Raffone, E. CONCERN: Does ovary need D-chiro-inositol? J. Ovarian Res. 2012, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.; Christoforidis, N.; Gadd, C.; Nikolaou, D.; Seyani, L.; Donaldson, A.; Margara, R.; Hardy, K.; Franks, S. Impaired insulin-dependent glucose metabolism in granulosa-lutein cells from anovulatory women with polycystic ovaries. Hum. Reprod 2005, 20, 373–381. [Google Scholar] [CrossRef]
- Carlomagno, G.; Unfer, V.; Roseff, S. The D-chiro-inositol paradox in the ovary. Fertil. Steril. 2011, 95, 2515–2516. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, F.; Unfer, V.; Dewailly, D.; Kamenov, Z.A.; Diamanti-Kandarakis, E.; Laganà, A.S.; Nestler, J.E.; Soulage, C.O. Group of ‘Inositol in PCOS and Reproduction’. Inositols in polycystic ovary syndrome: An overview on the advances. Trends Endocrinol. Metab. 2020, 31, 435–447. [Google Scholar] [CrossRef]
- Scambia, G.; Lello, S.; Salomone, S.; Fedele, L.; Capozzi, A.; Fruzzetti, F.; Di Carlo, C.; Sortino, M.A.; Drago, F. The role of inositols in PCOS–opinion paper. Ital. J. Gynecol. Obstet. 2019, 31, 87–92. [Google Scholar] [CrossRef]
- Paul, C.; Laganà, A.S.; Maniglio, P.; Triolo, O.; Brady, D.M. Inositol’s and other nutraceuticals’ synergistic actions counteract insulin resistance in polycystic ovarian syndrome and metabolic syndrome: State-of-the-art and future perspectives. Gynecol. Endocrinol. 2016, 32, 431–438. [Google Scholar] [CrossRef]
- Facchinetti, F.; Orrù, B.; Grandi, G.; Unfer, V. Short-term effects of metformin and myo-inositol in women with polycystic ovarian syndrome (PCOS): A meta-analysis of randomized clinical trials. Gynecol. Endocrinol. 2019, 35, 198–206. [Google Scholar] [CrossRef]
- Agrawal, A.; Mahey, R.; Kachhawa, G.; Khadgawat, R.; Vanamail, P.; Kriplani, A. Comparison of metformin plus myoinositol vs metformin alone in PCOS women undergoing ovulation induction cycles: Randomized controlled trial. Gynecol. Endocrinol. 2019, 35, 511–514. [Google Scholar] [CrossRef]
- Pkhaladze, L.; Barbakadze, L.; Kvashilava, N. Myo-inositol in the treatment of teenagers affected by PCOS. Int. J. Endocrinol. 2016, 2016, 1473612. [Google Scholar] [CrossRef]
- Romualdi, D.; Versace, V.; Lanzone, A. What is new in the landscape of insulin-sensitizing agents for polycystic ovary syndrome treatment. Ther. Adv. Reprod. Health 2020, 14, 2633494120908709. [Google Scholar] [CrossRef] [PubMed]
- Fruzzetti, F.; Capozzi, A.; Canu, A.; Lello, S. Treatment with d-chiro-inositol and alpha lipoic acid in the management of polycystic ovary syndrome. Gynecol. Endocrinol. 2019, 35, 506–510. [Google Scholar] [CrossRef] [PubMed]
- Genazzani, A.D.; Prati, A.; Marchini, F.; Petrillo, T.; Napolitano, A.; Simoncini, T. Differential insulin response to oral glucose tolerance test (OGTT) in overweight/obese polycystic ovary syndrome patients undergoing to myo-inositol (MYO), alpha lipoic acid (ALA), or combination of both. Gynecol. Endocrinol. 2019, 35, 1088–1093. [Google Scholar] [CrossRef] [PubMed]
- Esmaeilzadeh, S.; Gholinezhad-Chari, M.; Ghadimi, R. The effect of metformin treatment on the serum levels of homocysteine, folic acid, and vitamin B12 in patients with polycystic ovary syndrome. J. Hum. Reprod. Sci. 2017, 10, 95–101. [Google Scholar] [CrossRef]
- Holman, R.R.; Coleman, R.L.; Chan, J.C.N.; Chiasson, J.L.; Feng, H.; Ge, J.; Gerstein, H.C.; Gray, R.; Huo, Y.; Lang, Z.; et al. Effects of acarbose on cardiovascular and diabetes outcomes in patients with coronary heart disease and impaired glucose tolerance (ACE): A randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2017, 5, 877–886. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Hou, L.Q.; Zhao, T.Y. Effects of acarbose on polycystic ovary syndrome: A meta-analysis. Exp. Clin. Endocrinol. Diabetes 2014, 122, 373–378. [Google Scholar] [CrossRef]
- Araujo Penna, I.; Canella, P.R.; Vieira, C.S.; Silva de Sá, M.F.; dos Reis, R.M.; Ferriani, R.A. Cardiovascular risk factors are reduced with a low dose of acarbose in obese patients with polycystic ovary syndrome. Fertil. Steril. 2007, 88, 519–522. [Google Scholar] [CrossRef]
- Tuğrul, S.; Kutlu, T.; Pekin, O.; Bağlam, E.; Kiyak, H.; Oral, O. Clinical, endocrine, and metabolic effects of acarbose, a alpha-glucosidase inhibitor, in overweight and nonoverweight patients with polycystic ovarian syndrome. Fertil. Steril. 2008, 90, 1144–1148. [Google Scholar] [CrossRef]
- Penna, I.A.; Canella, P.R.; Reis, R.M.; Silva de Sá, M.F.; Ferriani, R.A. Acarbose in obese patients with polycystic ovarian syndrome: A double-blind, randomized, placebo-controlled study. Hum. Reprod. 2005, 20, 2396–2401. [Google Scholar] [CrossRef]
- Ciotta, L.; Calogero, A.E.; Farina, M.; De Leo, V.; La Marca, A.; Cianci, A. Clinical, endocrine and metabolic effects of acarbose, an alpha-glucosidase inhibitor, in PCOS patients with increased insulin response and normal glucose tolerance. Hum. Reprod. 2001, 16, 2066–2072. [Google Scholar] [CrossRef][Green Version]
- Glueck, C.J.; Moreira, A.; Goldenberg, N.; Sieve, L.; Wang, P. Pioglitazone and metformin in obese women with polycystic ovary syndrome not optimally responsive to metformin. Hum. Reprod. 2003, 18, 1618–1625. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lord, J.M.; Flight, I.H.; Norman, R.J. Insulin-sensitising drugs (metformin, troglitazone, rosiglitazone, pioglitazone, D-chiro-inositol) for polycystic ovary syndrome. Cochrane Database Syst. Rev. 2003, 3, CD003053. [Google Scholar] [CrossRef]
- Brettenthaler, N.; De Geyter, C.; Huber, P.R.; Keller, U. Effect of the insulin sensitizer pioglitazone on insulin resistance, hyperandrogenism, and ovulatory dysfunction in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2004, 89, 3835–3840. [Google Scholar] [CrossRef] [PubMed]
- Ortega-González, C.; Cardoza, L.; Coutiño, B.; Hidalgo, R.; Arteaga-Troncoso, G.; Parra, A. Insulin sensitizing drugs increase the endogenous dopaminergic tone in obese insulin-resistant women with polycystic ovary syndrome. J. Endocrinol. 2005, 184, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wu, Y.; Huang, Q. Comparison of the effect between pioglitazone and metformin in treating patients with PCOS: A meta-analysis. Arch. Gynecol. Obstet. 2017, 296, 661–677. [Google Scholar] [CrossRef] [PubMed]
- Stout, D.L.; Fugate, S.E. Thiazolidinediones for treatment of polycystic ovary syndrome. Pharmacotherapy 2005, 25, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Arlt, W.; Auchus, R.J.; Miller, W.L. Thiazolidinediones but not metformin directly inhibit the steroidogenic enzymes P450c17 and 3β-hydroxysteroid dehydrogenase. J. Biol. Chem. 2001, 276, 16767–16771. [Google Scholar] [CrossRef] [PubMed]
- Regazzo, D.; Barbot, M.; Scaroni, C.; Albiger, N.; Occhi, G. The pathogenic role of the GIP/GIPR axis in human endocrine tumors: Emerging clinical mechanisms beyond diabetes. Rev. Endocr. Metab. Disord. 2020, 21, 165–183. [Google Scholar] [CrossRef]
- Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; Poulter, N.R.; Ravn, L.S.; et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef]
- Jensterle Sever, M.; Kocjan, T.; Pfeifer, M.; Kravos, N.A.; Janez, A. Short-term combined treatment with liraglutide and metformin leads to significant weight loss in obese women with polycystic ovary syndrome and previous poor response to metformin. Eur. J. Endocrinol. 2014, 170, 451–459. [Google Scholar] [CrossRef]
- Cena, H.; Chiovato, L.; Nappi, R.E. Obesity, polycystic ovary syndrome, and infertility: A new avenue for GLP-1 receptor agonists. J. Clin. Endocrinol. Metab. 2020, 105, e2695–e2709. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, C.B.; Lindenberg, S. The effect of liraglutide on weight loss in women with polycystic ovary syndrome: An observational study. Front. Endocrinol. 2014, 5, 140. [Google Scholar] [CrossRef] [PubMed]
- Jensterle, M.; Goricar, K.; Janez, A. Metformin as an initial adjunct to low-dose liraglutide enhances the weight-decreasing potential of liraglutide in obese polycystic ovary syndrome: Randomized control study. Exp. Ther. Med. 2016, 11, 1194–1200. [Google Scholar] [CrossRef] [PubMed]
- Kahal, H.; Aburima, A.; Ungvari, T.; Rigby, A.S.; Coady, A.M.; Vince, R.V.; Ajjan, R.A.; Kilpatrick, E.S.; Naseem, K.M.; Atkin, S.L. The effects of treatment with liraglutide on atherothrombotic risk in obese young women with polycystic ovary syndrome and controls. BMC Endocr. Disord. 2015, 15, 14. [Google Scholar] [CrossRef]
- Nylander, M.; Frøssing, S.; Kistorp, C.; Faber, J.; Skouby, S.O. Liraglutide in polycystic ovary syndrome: A randomized trial, investigating effects on thrombogenic potential. Endocr. Connect. 2017, 6, 89–99. [Google Scholar] [CrossRef]
- Jensterle, M.; Kravos, N.A.; Goričar, K.; Janez, A. Short-term effectiveness of low dose liraglutide in combination with metformin versus high dose liraglutide alone in treatment of obese PCOS: Randomized trial. BMC Endocr. Disord. 2017, 17, 5. [Google Scholar] [CrossRef]
- Elkind-Hirsch, K.; Marrioneaux, O.; Bhushan, M.; Vernor, D.; Bhushan, R. Comparison of single and combined treatment with exenatide and metformin on menstrual cyclicity in overweight women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2008, 93, 2670–2678. [Google Scholar] [CrossRef]
- Dawson, A.J.; Sathyapalan, T.; Vince, R.; Coady, A.M.; Ajjan, R.A.; Kilpatrick, E.S.; Atkin, S.L. The effect of exenatide on cardiovascular risk markers in women with polycystic ovary syndrome. Front. Endocrinol. 2019, 10, 189. [Google Scholar] [CrossRef]
- Tang, L.; Yuan, L.; Yang, G.; Wang, F.; Fu, M.; Chen, M.; Liu, D. Changes in whole metabolites after exenatide treatment in overweight/obese polycystic ovary syndrome patients. Clin. Endocrinol. 2019, 91, 508–516. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Zheng, S.Y.; Lin, R.; Xie, Y.J.; Chen, H.; Zheng, Y.X.; Liu, E.; Chen, L.; Yan, J.H.; et al. Efficacy of exenatide on weight loss, metabolic parameters and pregnancy in overweight/obese polycystic ovary syndrome. Clin. Endocrinol. 2017, 87, 767–774. [Google Scholar] [CrossRef]
- Gerstein, H.C.; Colhoun, H.M.; Dagenais, G.R.; Diaz, R.; Lakshmanan, M.; Pais, P.; Probstfield, J.; Riesmeyer, J.S.; Riddle, M.C.; Rydén, L.; et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): A double-blind, randomized placebo-controlled trial. Lancet 2019, 394, 121–130. [Google Scholar] [CrossRef]
- Tao, T.; Wu, P.; Wang, Y.; Liu, W. Comparison of glycemic control and β-cell function in new onset T2DM patients with PCOS of metformin and saxagliptin monotherapy or combination treatment. BMC Endocr. Disord. 2018, 18, 14. [Google Scholar] [CrossRef] [PubMed]
- Ferjan, S.; Janez, A.; Jensterle, M. Dipeptidyl peptidase-4 inhibitor sitagliptin prevented weight regain in obese women with polycystic ovary syndrome previously treated with liraglutide: A pilot randomized study. Metab. Syndr. Relat. Disord. 2017, 15, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Nauck, M.A.; Meier, J.J.; Cavender, M.A.; Abd El Aziz, M.; Drucker, D.J. Cardiovascular actions and clinical outcomes with glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Circulation 2017, 136, 849–870. [Google Scholar] [CrossRef]
- Devin, J.K.; Nian, H.; Celedonio, J.E.; Wright, P.; Brown, N.J. Sitagliptin decreases visceral fat and blood glucose in women with polycystic ovarian syndrome. J. Clin. Endocrinol. Metab. 2020, 105, dgz028. [Google Scholar] [CrossRef]
- Ferjan, S.; Janez, A.; Jensterle, M. DPP4 inhibitor sitagliptin as a potential treatment option in metformin-intolerant obese women with polycystic ovary syndrome: A pilot randomized study. Endocr. Pract. 2018, 24, 69–77. [Google Scholar] [CrossRef]
- Kalra, S. Sodium glucose co-transporter-2 (SGLT2) inhibitors: A review of their basic and clinical pharmacology. Diabetes Ther. 2014, 5, 355–366. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; de Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R.; et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef]
- Javed, Z.; Papageorgiou, M.; Madden, L.A.; Rigby, A.S.; Kilpatrick, E.S.; Atkin, S.L.; Sathyapalan, T. The effects of empagliflozin vs metformin on endothelial microparticles in overweight/obese women with polycystic ovary syndrome. Endocr. Connect. 2020, 9, 563–569. [Google Scholar] [CrossRef]
- Javed, Z.; Papageorgiou, M.; Deshmukh, H.; Rigby, A.S.; Qamar, U.; Abbas, J.; Khan, A.Y.; Kilpatrick, E.S.; Atkin, S.L.; Sathyapalan, T. Effects of empagliflozin on metabolic parameters in polycystic ovary syndrome: A randomized controlled study. Clin. Endocrinol. 2019, 90, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Dokras, A. Noncontraceptive use of oral combined hormonal contraceptives in polycystic ovary syndrome-risks versus benefits. Fertil. Steril. 2016, 106, 1572–1579. [Google Scholar] [CrossRef] [PubMed]
- De Medeiros, S.F. Risks, benefits size and clinical implications of combined oral contraceptive use in women with polycystic ovary syndrome. Reprod. Biol. Endocrinol. 2017, 15, 93. [Google Scholar] [CrossRef] [PubMed]
- Dokras, A. Cardiovascular disease risk in women with PCOS. Steroids 2013, 78, 773–776. [Google Scholar] [CrossRef]
- Dumesic, D.; Akopians, A.; Madrigal, V.; Ramirez, E.; Margolis, D.; Sarma, M.; Thomas, A.; Grogan, T.; Haykal, R.; Schooler, T.; et al. Hyperandrogenism accompanies increased intra-abdominal fat storage in normal weight polycystic ovary syndrome women. J. Clin. Endocrinol. Metab. 2016, 101, 4178–4188. [Google Scholar] [CrossRef]
- De Bastos, M.; Stegeman, B.H.; Rosendaal, F.R.; Van Hylckama Vlieg, A.; Helmerhorst, F.M.; Stijnen, T.; Dekkers, O.M. Combined oral contraceptives: Venous thrombosis. Cochrane Database Syst. Rev. 2014, 3, CD010813. [Google Scholar] [CrossRef]
- Sitruk-Ware, R. Hormonal contraception and thrombosis. Fertil. Steril. 2016, 106, 1289–1294. [Google Scholar] [CrossRef]
- Godsland, I.F. The influence of female sex steroids on glucose metabolism and insulin action. J. Int. Med. Suppl. 1996, 738, 1–60. [Google Scholar]
- Morin-Papunen, L.C.; Vauhkonen, I.; Koivunen, R.M.; Ruokonen, A.; Martikainen, H.K.; Tapanainen, J.S. Endocrine and metabolic effects of metformin versus ethinyl estradiol-cyproterone acetate in obese women with polycystic ovary syndrome: A randomized study. J. Clin. Endocrinol. Metab. 2000, 85, 3161–3168. [Google Scholar] [CrossRef]
- Halperin, I.J.; Kumar, S.S.; Stroup, D.F.; Laredo, S.E. The association between the combined oral contraceptive pill and insulin resistance, dysglycemia and dyslipidemia in women with polycystic ovary syndrome: A systematic review and meta-analysis of observational studies. Hum. Reprod. 2011, 26, 191–201. [Google Scholar] [CrossRef]
- Legro, R.S.; Dodson, W.C.; Kris-Etherton, P.M.; Kunselman, A.R.; Stetter, C.M.; Williams, N.I.; Gnatuk, C.L.; Estes, S.J.; Fleming, J.; Allison, K.C.; et al. Randomized controlled trial of preconception interventions in infertile women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2015, 100, 4048–4058. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, L.; De Zegher, F. Flutamide-metformin plus an oral contraceptive (OC) for young women with polycystic ovary syndrome: Switch from third- to fourth-generation OC reduces body adiposity. Hum. Reprod. 2004, 19, 1725–1727. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fuhrmann, U.; Krattenmacher, R.; Slater, E.P.; Fritzemeier, K.H. The novel progestin drospirenone and its natural counterpart progesterone: Biochemical profile and antiandrogenic potential. Contraception 1996, 54, 243–251. [Google Scholar] [CrossRef]
- Yildizhan, R.; Gokce, A.I.; Yildizhan, B.; Cim, N. Comparison of the effects of chlormadinone acetate versus drospirenone containing oral contraceptives on metabolic and hormonal parameters in women with PCOS for a period of two-year follow-up. Gynecol. Endocrinol. 2015, 31, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Pavicic Baldani, D.; Skrgatic, L.; Ougouag, R.; Kasum, M. The cardiometabolic effect of current management of polycystic ovary syndrome: Strategies of prevention and treatment. Gynecol. Endocrinol. 2018, 34, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Sabbadin, C.; Andrisani, A.; Ambrosini, G.; Bordin, L.; Donà, G.; Manso, J.; Ceccato, F.; Scaroni, C.; Armanini, D. Aldosterone in gynecology and its involvement on the risk of hypertension in pregnancy. Front. Endocrinol. 2019, 10, 575. [Google Scholar] [CrossRef]
- Luque-Ramírez, M.; Nattero-Chávez, L.; Ortiz Flores, A.E.; Escobar-Morreale, H.F. Combined oral contraceptives and/or antiandrogens versus insulin sensitizers for polycystic ovary syndrome: A systematic review and meta-analysis. Hum. Reprod. Update 2018, 24, 225–241. [Google Scholar] [CrossRef]
- Armanini, D.; Bordin, L.; Donà, G.; Sabbadin, C.; Bakdounes, L.; Ragazzi, E.; Giorgino, F.L.; Fiore, C. Polycystic ovary syndrome: Implications of measurement of plasma aldosterone, renin activity and progesterone. Steroids 2012, 77, 655–658. [Google Scholar] [CrossRef]
- Cascella, T.; Palomba, S.; Tauchmanovà, L.; Manguso, F.; Di Biase, S.; Labella, D.; Giallauria, F.; Vigorito, C.; Colao, A.; Lombardi, G.; et al. Serum aldosterone concentration and cardiovascular risk in women with polycystic ovarian syndrome. J. Clin. Endocrinol. Metab. 2006, 91, 4395–4400. [Google Scholar] [CrossRef]
- Macut, D.; Mladenović, V.; Bjekić-Macut, J.; Livadas, S.; Stanojlović, O.; Hrnčić, D.; Rašić-Marković, A.; Milutinović, D.V.; Andrić, Z. Hypertension in polycystic ovary syndrome: Novel insights. Curr. Hypertens Rev. 2020, 16, 55–60. [Google Scholar] [CrossRef]
- Bastianelli, C.; Farris, M.; Rosato, E.; Brosens, I.; Benagiano, G. Pharmacodynamics of combined estrogen-progestin oral contraceptives: 1. Effects on metabolism. Expert Rev. Clin. Pharmacol. 2017, 10, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Grandi, G.; Napolitano, A.; Cagnacci, A. Metabolic impact of combined hormonal contraceptives containing estradiol. Expert Opin. Drug Metab. Toxicol. 2016, 12, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Di Carlo, C.; Gargano, V.; Sparice, S.; Tommaselli, G.A.; Bifulco, G.; Nappi, C. Effects of an oral contraceptive containing estradiol valerate and dienogest on circulating androgen levels and acne in young patients with PCOS: An observational preliminary study. Gynecol. Endocrinol. 2013, 29, 1048–1050. [Google Scholar] [CrossRef]
- De Leo, V.; Fruzzetti, F.; Musacchio, M.C.; Scolaro, V.; Di Sabatino, A.; Morgante, G. Effect of a new oral contraceptive with estradiol valerate/dienogest on carbohydrate metabolism. Contraception 2013, 88, 364–368. [Google Scholar] [CrossRef]
- Martin, K.A.; Anderson, R.R.; Chang, R.J.; Ehrmann, D.A.; Lobo, R.A.; Murad, M.H.; Pugeat, M.M.; Rosenfield, R.L. Evaluation and treatment of hirsutism in premenopausal women: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1233–1257. [Google Scholar] [CrossRef] [PubMed]
- Seyam, E.; Al Gelany, S.; Abd Al Ghaney, A.; Mohamed, M.A.A.; Youseff, A.M.; Ibrahim, E.M.; Khalifa, E.M.; Hefzy, E. Evaluation of prolonged use of statins on the clinical and biochemical abnormalities and ovulation dysfunction in single young women with polycystic ovary syndrome. Gynecol. Endocrinol. 2018, 34, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Sathyapalan, T.; Coady, A.M.; Kilpatrick, E.S.; Atkin, S.L. The effect of atorvastatin on pancreatic beta cell requirement in women with polycystic ovary syndrome. Endocr. Connect. 2017, 6, 811–816. [Google Scholar] [CrossRef]
- Cignarella, A. The gender-specific clinical pharmacology of statins: An update. Ital. J. Gend. Specif. Med. 2018, 4, 47–52. [Google Scholar] [CrossRef]
- Almalki, H.H.; Alshibani, T.M.; Alhifany, A.A.; Almohammed, O.A. Comparative efficacy of statins, metformin, spironolactone and combined oral contraceptives in reducing testosterone levels in women with polycystic ovary syndrome: A network meta-analysis of randomized clinical trials. BMC Womens Health 2020, 20, 68. [Google Scholar] [CrossRef]
- Banaszewska, B.; Pawelczyk, L.; Spaczynski, R. Current and future aspects of several adjunctive treatment strategies in polycystic ovary syndrome. Reprod. Biol. 2019, 19, 309–315. [Google Scholar] [CrossRef]
- Sathyapalan, T.; Hobkirk, J.P.; Javed, Z.; Carroll, S.; Coady, A.M.; Pemberton, P.; Smith, A.; Cianflone, K.; Atkin, S.L. The effect of atorvastatin (and subsequent metformin) on adipose tissue acylation-stimulatory-protein concentration and inflammatory biomarkers in overweight/obese women with polycystic ovary syndrome. Front Endocrinol. 2019, 10, 394. [Google Scholar] [CrossRef] [PubMed]
- Fruzzetti, F.; Cagnacci, A. Venous thrombosis and hormonal contraception: What’s new with estradiol-based hormonal contraceptives? Open Access J. Contracept. 2018, 9, 75–79. [Google Scholar] [CrossRef] [PubMed]
- De Leo, V.; Musacchio, M.C.; Cappelli, V.; Piomboni, P.; Morgante, G. Hormonal contraceptives: Pharmacology tailored to women’s health. Hum. Reprod. Update 2016, 22, 634–646. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Berneis, K.; Spinas, G.; Rini, G.B.; Carmina, E. Long-term consequences of polycystic ovary syndrome on cardiovascular risk. Fertil. Steril. 2009, 91 (Suppl. 4), 1563–1567. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pierpoint, T.; McKeigue, P.M.; Isaacs, A.J.; Wild, S.H.; Jacobs, H.S. Mortality of women with polycystic ovary syndrome at long-term follow-up. J. Clin. Epidemiol. 1998, 51, 581–586. [Google Scholar] [CrossRef]
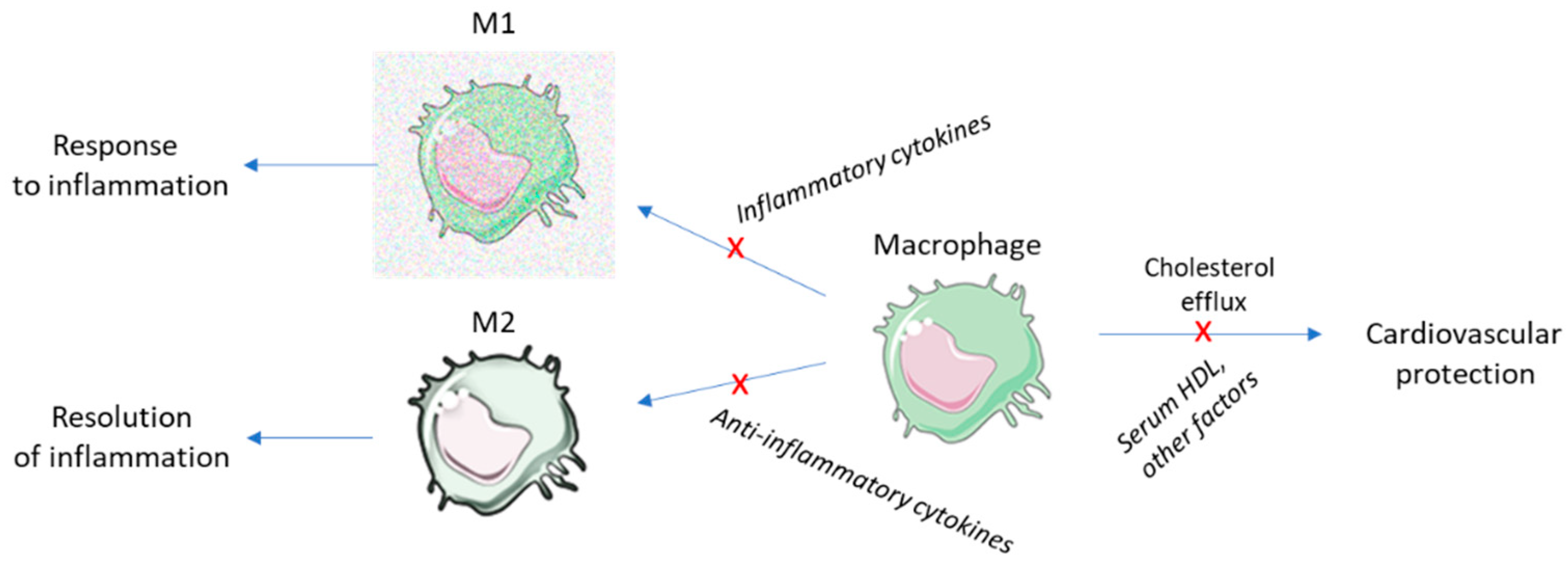
| Drug Class | Mechanism | Endpoints | Side Effects |
|---|---|---|---|
| Antiandrogens | Androgen receptor antagonists 5 alpha reductase inhibitors | Improved lipid profile and insulin resistance Vascular compliance | Dyslipidemia (BCL) possibly due to concomitant medication |
| Oral hypoglycemics | Insulin sensitization | Weight loss Improvement in insulin sensitivity resulting in lower circulating androgen concentration | Weight gain (TZD) |
| Combined oral contraceptives | Steroid hormone receptor modulation | Lower androgen levels | Dyslipidemia Thrombogenic risk (less with antiandrogenic progestins) |
| Statins | Cholesterol biosynthesis inhibition | Reduced testosterone levels and inflammatory markers |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cignarella, A.; Mioni, R.; Sabbadin, C.; Dassie, F.; Parolin, M.; Vettor, R.; Barbot, M.; Scaroni, C. Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS. Int. J. Mol. Sci. 2020, 21, 9554. https://doi.org/10.3390/ijms21249554
Cignarella A, Mioni R, Sabbadin C, Dassie F, Parolin M, Vettor R, Barbot M, Scaroni C. Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS. International Journal of Molecular Sciences. 2020; 21(24):9554. https://doi.org/10.3390/ijms21249554
Chicago/Turabian StyleCignarella, Andrea, Roberto Mioni, Chiara Sabbadin, Francesca Dassie, Matteo Parolin, Roberto Vettor, Mattia Barbot, and Carla Scaroni. 2020. "Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS" International Journal of Molecular Sciences 21, no. 24: 9554. https://doi.org/10.3390/ijms21249554
APA StyleCignarella, A., Mioni, R., Sabbadin, C., Dassie, F., Parolin, M., Vettor, R., Barbot, M., & Scaroni, C. (2020). Pharmacological Approaches to Controlling Cardiometabolic Risk in Women with PCOS. International Journal of Molecular Sciences, 21(24), 9554. https://doi.org/10.3390/ijms21249554





