Hypothesis: Single Actomyosin Properties Account for Ensemble Behavior in Active Muscle Shortening and Isometric Contraction
Abstract
1. Introduction
2. Methods
3. Results and Discussion
3.1. Detailed Specifications of Hypothesis and Bottom-Up Method of Assigning Model Parameter Values
3.2. Existing Evidence in Favor of the Hypothesis
3.3. Evidence Against the Hypothesis
3.4. Future Tests of Hypothesis
3.5. Bottom-Up Assembled Experimental Systems
3.6. Top-Down Disassembled Systems
3.7. The Cross-Bridge Elasticity with an Intact Myofilament Lattice
4. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Geeves, M.A.; Holmes, K.C. The Molecular Mechanism of Muscle Contraction. Protein Fold. Cell 2005, 71, 161–193. [Google Scholar] [CrossRef]
- Månsson, A.; Rassier, D.; Tsiavaliaris, G. Poorly Understood Aspects of Striated Muscle Contraction. BioMed Res. Int. 2015, 2015, 245154. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, H.L.; Hammers, D.W. Muscle Contraction. Cold Spring Harb. Perspect. Biol. 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Reggiani, C. Fiber Types in Mammalian Skeletal Muscles. Physiol. Rev. 2011, 91, 1447–1531. [Google Scholar] [CrossRef] [PubMed]
- Steinmetz, P.R.H.; Kraus, J.E.M.; Larroux, C.; Hammel, J.U.; Amon-Hassenzahl, A.; Houliston, E.; Wörheide, G.; Nickel, M.; Degnan, B.M.; Technau, U. Independent evolution of striated muscles in cnidarians and bilaterians. Nat. Cell Biol. 2012, 487, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Gautel, M.; Djinović-Carugo, K. The sarcomeric cytoskeleton: From molecules to motion. J. Exp. Biol. 2016, 219, 135–145. [Google Scholar] [CrossRef]
- Frey, N.; Luedde, M.; Katus, H.A. Mechanisms of disease: Hypertrophic cardiomyopathy. Nat. Rev. Cardiol. 2011, 9, 91–100. [Google Scholar] [CrossRef]
- Spudich, J.A. Hypertrophic and Dilated Cardiomyopathy: Four Decades of Basic Research on Muscle Lead to Potential Therapeutic Approaches to These Devastating Genetic Diseases. Biophys. J. 2014, 106, 1236–1249. [Google Scholar] [CrossRef] [PubMed]
- McKillop, D.F.A.; Geeves, M.A. Regulation of the acto·myosin subfragment 1 interaction by troponin/tropomyosin. Evidence for control of a specific isomerization between two acto·myosin subfragment 1 states. Biochem. J. 1991, 279, 711–718. [Google Scholar] [CrossRef]
- Vibert, P.; Craig, R.; Lehman, W. Steric-model for activation of muscle thin filaments 1 1 Edited by P.E. Wright. J. Mol. Biol. 1997, 266, 8–14. [Google Scholar] [CrossRef]
- Heling, L.W.H.J.; Geeves, M.A.; Kad, N.M. MyBP-C: One protein to govern them all. J. Muscle Res. Cell Motil. 2020, 41, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Freundt, J.K.; Linke, W.A. Titin as a force-generating muscle protein under regulatory control. J. Appl. Physiol. 2019, 126, 1474–1482. [Google Scholar] [CrossRef] [PubMed]
- Millman, B.M. The Filament Lattice of Striated Muscle. Physiol. Rev. 1998, 78, 359–391. [Google Scholar] [CrossRef]
- Tanner, B.C.; Daniel, T.L.; Regnier, M. Sarcomere Lattice Geometry Influences Cooperative Myosin Binding in Muscle. PLoS Comput. Biol. 2007, 3, e115. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.D.; Regnier, M.; Daniel, T.L. Axial and Radial Forces of Cross-Bridges Depend on Lattice Spacing. PLoS Comput. Biol. 2010, 6, e1001018. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.D.; Salcedo, M.K.; Irving, T.C.; Regnier, M.; Daniel, T.L. The length–tension curve in muscle depends on lattice spacing. Proc. R. Soc. B: Boil. Sci. 2013, 280, 20130697. [Google Scholar] [CrossRef]
- Daniel, T.L.; Trimble, A.C.; Chase, P.B. Compliant Realignment of Binding Sites in Muscle: Transient Behavior and Mechanical Tuning. Biophys. J. 1998, 74, 1611–1621. [Google Scholar] [CrossRef]
- Homsher, E.; Wang, F.; Sellers, J.R. Factors affecting movement of F-actin filaments propelled by skeletal muscle heavy meromyosin. Am. J. Physiol. Physiol. 1992, 262, C714–C723. [Google Scholar] [CrossRef]
- Mijailovich, S.M.; Kayser-Herold, O.; Stojanovic, B.; Nedic, D.; Irving, T.C.; Geeves, M.A. Three-dimensional stochastic model of actin–myosin binding in the sarcomere lattice. J. Gen. Physiol. 2016, 148, 459–488. [Google Scholar] [CrossRef] [PubMed]
- Shalabi, N.; Persson, M.; Månsson, A.; Vengallatore, S.; Rassier, D.E. Sarcomere Stiffness during Stretching and Shortening of Rigor Skeletal Myofibrils. Biophys. J. 2017, 113, 2768–2776. [Google Scholar] [CrossRef]
- Trivedi, D.V.; Adhikari, A.S.; Sarkar, S.S.; Ruppel, K.M.; Spudich, J.A. Hypertrophic cardiomyopathy and the myosin mesa: Viewing an old disease in a new light. Biophys. Rev. 2018, 10, 27–48. [Google Scholar] [CrossRef]
- Cheng, Y.-S.; Leite, F.D.S.; Rassier, D.E. The load dependence and the force-velocity relation in intact myosin filaments from skeletal and smooth muscles. Am. J. Physiol. Physiol. 2020, 318, C103–C110. [Google Scholar] [CrossRef] [PubMed]
- Månsson, A.; Usaj, M.; Moretto, L.; Rassier, D.E. Do Actomyosin Single-Molecule Mechanics Data Predict Mechanics of Contracting Muscle? Int. J. Mol. Sci. 2018, 19, 1863. [Google Scholar] [CrossRef] [PubMed]
- Edman, K.A.P.; Reggiani, C. Redistribution of sarcomere length during isometric contraction of frog muscle fibres and its relation to tension creep. J. Physiol. 1984, 351, 169–198. [Google Scholar] [CrossRef]
- Edman, K.A.P.; Reggiani, C.; Kronnie, G.T. Differences in maximum velocity of shortening along single muscle fibres of the frog. J. Physiol. 1985, 365, 147–163. [Google Scholar] [CrossRef]
- Edman, K.A.P.; Reggiani, C.; Schiaffino, S.; Kronnie, G.T. Maximum velocity of shortening related to myosin isoform composition in frog skeletal muscle fibres. J. Physiol. 1988, 395, 679–694. [Google Scholar] [CrossRef]
- Edman, K.A.P.; Flitney, F.W. Laser diffraction studies of sarcomere dynamics during ‘isometric’ relaxation in isolated muscle fibres of the frog. J. Physiol. 1982, 329, 1–20. [Google Scholar] [CrossRef]
- Poggesi, C.; Tesi, C.; Stehle, R. Sarcomeric determinants of striated muscle relaxation kinetics. Pflügers Archiv 2005, 449, 505–517. [Google Scholar] [CrossRef]
- Månsson, A. Comparing models with one versus multiple myosin-binding sites per actin target zone: The power of simplicity. J. Gen. Physiol. 2019, 151, 578–592. [Google Scholar] [CrossRef]
- Månsson, A. The effects of inorganic phosphate on muscle force development and energetics: Challenges in modelling related to experimental uncertainties. J. Muscle Res. Cell Motil. 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Månsson, A. Actomyosin based contraction: One mechanokinetic model from single molecules to muscle? J. Muscle Res. Cell Motil. 2016, 37, 181–194. [Google Scholar] [CrossRef]
- Rahman, M.A.; Ušaj, M.; Rassier, D.E.; Månsson, A. Blebbistatin Effects Expose Hidden Secrets in the Force-Generating Cycle of Actin and Myosin. Biophys. J. 2018, 115, 386–397. [Google Scholar] [CrossRef]
- Hill, T.L. Theoretical formalism for the sliding filament model of contraction of striated muscle Part I. Prog. Biophys. Mol. Biol. 1974, 28, 267–340. [Google Scholar] [CrossRef]
- Llinas, P.; Isabet, T.; Song, L.; Ropars, V.; Zong, B.; Benisty, H.; Sirigu, S.; Morris, C.; Kikuti, C.; Safer, D.; et al. How actin initiates the motor activity of Myosin. Dev. Cell 2015, 33, 401–412. [Google Scholar] [CrossRef]
- Walcott, S.; Warshaw, D.M.; DeBold, E.P. Mechanical Coupling between Myosin Molecules Causes Differences between Ensemble and Single-Molecule Measurements. Biophys. J. 2012, 103, 501–510. [Google Scholar] [CrossRef]
- Pertici, I.; Bongini, L.; Melli, L.; Bianchi, G.; Salvi, L.; Falorsi, G.; Squarci, C.; Bozó, T.; Cojoc, D.; Kellermayer, M.S.Z.; et al. A myosin II nanomachine mimicking the striated muscle. Nat. Commun. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kaya, M.; Tani, Y.; Washio, T.; Hisada, T.; Higuchi, H. Coordinated force generation of skeletal myosins in myofilaments through motor coupling. Nat. Commun. 2017, 8, 16036. [Google Scholar] [CrossRef]
- Månsson, A.; Mörner, J.; Edman, K.A.P. Effects of amrinone on twitch, tetanus and shortening kinetics in mammalian skeletal muscle. Acta Physiol. Scand. 1989, 136, 37–45. [Google Scholar] [CrossRef]
- Asmussen, G.; Beckers-Bleukx, G. The force-velocity relation of the rabbit inferior oblique muscle; influence of temperature. Pflügers Arch. Eur. J. Physiol. 1994, 426, 542–547. [Google Scholar] [CrossRef]
- Ranatunga, K.W. The force-velocity relation of rat fast- and slow-twitch muscles examined at different temperatures. J. Physiol. 1984, 351, 517–529. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, D.T. A general method for numerically simulating the stochastic time evolution of coupled chemical reactions. J. Comput. Phys. 1976, 22, 403–434. [Google Scholar] [CrossRef]
- Balaz, M.; Sundberg, M.; Persson, M.; Kvassman, J.; Månsson, A. Effects of Surface Adsorption on Catalytic Activity of Heavy Meromyosin Studied Using a Fluorescent ATP Analogue†. Biochemistry 2007, 46, 7233–7251. [Google Scholar] [CrossRef]
- Lard, M.; Siethoff, L.T.; Generosi, J.; Persson, M.; Linke, H.; Månsson, A. Nanowire-Imposed Geometrical Control in Studies of Actomyosin Motor Function. IEEE Trans. NanoBioscience 2015, 14, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Huxley, A. Muscle Structure and Theories of Contraction. Prog. Biophys. Biophys. Chem. 1957, 7, 255–318. [Google Scholar] [CrossRef]
- Eisenberg, E.; Hill, T.; Chen, Y. Cross-bridge model of muscle contraction. Quantitative analysis. Biophys. J. 1980, 29, 195–227. [Google Scholar] [CrossRef]
- Smith, D.; Geeves, M. Strain-dependent cross-bridge cycle for muscle. Biophys. J. 1995, 69, 524–537. [Google Scholar] [CrossRef]
- Smith, D.; Mijailovich, S.M. Toward a Unified Theory of Muscle Contraction. II: Predictions with the Mean-Field Approximation. Ann. Biomed. Eng. 2008, 36, 1353–1371. [Google Scholar] [CrossRef] [PubMed]
- Piazzesi, G.; Lombardi, V. A cross-bridge model that is able to explain mechanical and energetic properties of shortening muscle. Biophys. J. 1995, 68, 1966–1979. [Google Scholar] [CrossRef]
- Vilfan, A.; Frey, E.; Schwabl, F. Force-velocity relations of a two-state crossbridge model for molecular motors. EPL (Europhys. Lett.) 1999, 45, 283–289. [Google Scholar] [CrossRef]
- Månsson, A. Cross-bridge movement and stiffness during the rise of tension in skeletal muscle—A theoretical analysis. J. Muscle Res. Cell Motil. 2000, 21, 383–403. [Google Scholar] [CrossRef]
- Linari, M.; Brunello, E.; Reconditi, M.; Fusi, L.; Caremani, M.; Narayanan, T.; Piazzesi, G.; Lombardi, V.; Irving, M. Force generation by skeletal muscle is controlled by mechanosensing in myosin filaments. Nat. Cell Biol. 2015, 528, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Tesi, C.; Piroddi, N.; Colomo, F.; Poggesi, C. Relaxation Kinetics Following Sudden Ca2+ Reduction in Single Myofibrils from Skeletal Muscle. Biophys. J. 2002, 83, 2142–2151. [Google Scholar] [CrossRef]
- Campbell, S.G.; Hatfield, P.C.; Campbell, K.S. A Mathematical Model of Muscle Containing Heterogeneous Half-Sarcomeres Exhibits Residual Force Enhancement. PLoS Comput. Biol. 2011, 7, e1002156. [Google Scholar] [CrossRef]
- Edman, K.A.P.; Tsuchiya, T. Strain of passive elements during force enhancement by stretch in frog muscle fibres. J. Physiol. 1996, 490, 191–205. [Google Scholar] [CrossRef]
- Cornachione, A.S.; Leite, F.D.S.; Bagni, M.A.; Rassier, D.E. The increase in non-cross-bridge forces after stretch of activated striated muscle is related to titin isoforms. Am. J. Physiol. Physiol. 2016, 310, C19–C26. [Google Scholar] [CrossRef]
- Shalabi, N.; Cornachione, A.; Leite, F.D.S.; Vengallatore, S.; Rassier, D.E. Residual force enhancement is regulated by titin in skeletal and cardiac myofibrils. J. Physiol. 2017, 595, 2085–2098. [Google Scholar] [CrossRef]
- Lombardi, V.; Piazzesi, G. The contractile response during steady lengthening of stimulated frog muscle fibres. J. Physiol. 1990, 431, 141–171. [Google Scholar] [CrossRef]
- Månsson, A. The tension response to stretch of intact skeletal muscle fibres of the frog at varied tonicity of the extracellular medium. J. Muscle Res. Cell Motil. 1994, 15, 145–157. [Google Scholar] [CrossRef]
- Brunello, E.; Reconditi, M.; Elangovan, R.; Linari, M.; Sun, Y.; Narayanan, T.; Panine, P.; Piazzesi, G.; Irving, M.; Lombardi, V. Skeletal muscle resists stretch by rapid binding of the second motor domain of myosin to actin. Proc. Natl. Acad. Sci. USA 2007, 104, 20114–20119. [Google Scholar] [CrossRef] [PubMed]
- Steffen, W.; Smith, D.; Simmons, R.; Sleep, J. Mapping the actin filament with myosin. Proc. Natl. Acad. Sci. USA 2001, 98, 14949–14954. [Google Scholar] [CrossRef]
- Månsson, A.; Persson, M.; Shalabi, N.; Rassier, D.E. Non-linear actomyosin elasticity in muscle? Biophys. J. 2019, 116, 330–346. [Google Scholar] [CrossRef] [PubMed]
- Linari, M.; Piazzesi, G.; Pertici, I.; Dantzig, J.A.; Goldman, Y.E.; Lombardi, V. Straightening Out the Elasticity of Myosin Cross-Bridges. Biophys. J. 2020, 118, 994–1002. [Google Scholar] [CrossRef]
- Debold, E.P.; Patlak, J.B.; Warshaw, D.M. Slip Sliding Away: Load-Dependence of Velocity Generated by Skeletal Muscle Myosin Molecules in the Laser Trap. Biophys. J. 2005, 89, L34–L36. [Google Scholar] [CrossRef]
- Pate, E.; Cooke, R. A model of crossbridge action: The effects of ATP, ADP and Pi. J. Muscle Res. Cell Motil. 1989, 10, 181–196. [Google Scholar] [CrossRef] [PubMed]
- Persson, M.; Bengtsson, E.; Siethoff, L.T.; Månsson, A. Nonlinear Cross-Bridge Elasticity and Post-Power-Stroke Events in Fast Skeletal Muscle Actomyosin. Biophys. J. 2013, 105, 1871–1881. [Google Scholar] [CrossRef]
- Homsher, E.; Nili, M.; Chen, I.Y.; Tobacman, L.S. Regulatory Proteins Alter Nucleotide Binding to Acto-Myosin of Sliding Filaments in Motility Assays. Biophys. J. 2003, 85, 1046–1052. [Google Scholar] [CrossRef]
- Marston, S.B. Random walks with thin filaments: Application of in vitro motility assay to the study of actomyosin regulation. J. Muscle Res. Cell Motil. 2003, 24, 149–156. [Google Scholar] [CrossRef]
- Capitanio, M.; Canepari, M.; Maffei, M.; Beneventi, D.; Monico, C.; Vanzi, F.; Bottinelli, R.; Pavone, F.S. Ultrafast force-clamp spectroscopy of single molecules reveals load dependence of myosin working stroke. Nat. Methods 2012, 9, 1013–1019. [Google Scholar] [CrossRef]
- Sung, J.; Nag, S.; Mortensen, K.I.; Vestergaard, C.L.; Sutton, S.; Ruppel, K.; Flyvbjerg, H.; Spudich, J.A. Harmonic force spectroscopy measures load-dependent kinetics of individual human beta-cardiac myosin molecules. Nat. Commun. 2015, 6, 7931. [Google Scholar] [CrossRef]
- Duke, T.A.J. Molecular model of muscle contraction. Proc. Natl. Acad. Sci. USA 1999, 96, 2770–2775. [Google Scholar] [CrossRef]
- Marcucci, L.; Reggiani, C. Mechanosensing in Myosin Filament Solves a 60 Years Old Conflict in Skeletal Muscle Modeling between High Power Output and Slow Rise in Tension. Front. Physiol. 2016, 7, 427. [Google Scholar] [CrossRef]
- Månsson, A. Actomyosin-ADP states, inter-head cooperativity and the force-velocity relation of skeletal muscle. Biophys. J. 2010, 98, 1237–1246. [Google Scholar] [CrossRef]
- Edman, K.A.P.; Mansson, A.; Caputo, C. The biphasic force-velocity relationship in frog muscle fibres and its evaluation in terms of cross-bridge function. J. Physiol. (Lond.) 1997, 503, 141–156. [Google Scholar] [CrossRef]
- Bunk, R.; Carlberg, P.; Månsson1, A.; Nicholls1, I.A.; Omling, P.; Sundberg1, M.; Tågerud1, S.; Montelius, L. Guiding molecular motors by nanoimprinted structures. Jap. J. Appl. Phys. 2005, 44, 3337–3340. [Google Scholar] [CrossRef]
- Huxley, A.F.; Tideswell, S. Rapid regeneration of power stroke in contracting muscle by attachment of second myosin head. J. Muscle Res. Cell Motil. 1997, 18, 111–114. [Google Scholar] [CrossRef]
- Caremani, M.; Melli, L.; Dolfi, M.; Lombardi, V.; Linari, M. The working stroke of the myosin II motor in muscle is not tightly coupled to release of orthophosphate from its active site. J. Physiol. 2013, 591, 5187–5205. [Google Scholar] [CrossRef]
- Kaya, M.; Higuchi, H. Nonlinear Elasticity and an 8-nm Working Stroke of Single Myosin Molecules in Myofilaments. Science 2010, 329, 686–689. [Google Scholar] [CrossRef]
- Geeves, M.A.; Lehrer, S.S.; Lehman, W. The mechanism of thin filament regulation: Models in conflict? J. Gen. Physiol. 2019, 151, 1265–1271. [Google Scholar] [CrossRef]
- Gordon, A.M.; Homsher, E.; Regnier, M. Regulation of Contraction in Striated Muscle. Physiol. Rev. 2000, 80, 853–924. [Google Scholar] [CrossRef]
- Bing, W.; Knott, A.; Marston, S.B. A simple method for measuring the relative force exerted by myosin on actin filaments in the in vitro motility assay: Evidence that tropomyosin and troponin increase force in single thin filaments. Biochem. J. 2000, 350 (Pt 3), 693–699. [Google Scholar] [CrossRef]
- Fujita, H.; Sasaki, D.; Ishiwata, S.; Kawai, M. Elementary Steps of the Cross-Bridge Cycle in Bovine Myocardium with and without Regulatory Proteins. Biophys. J. 2002, 82, 915–928. [Google Scholar] [CrossRef]
- Ishii, S.; Kawai, M.; Ishiwata, S.; Suzuki, M. Estimation of actomyosin active force maintained by tropomyosin and troponin complex under vertical forces in the in vitro motility assay system. PLoS ONE 2018, 13, e0192558. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Ishiwata, S. Use of thin filament reconstituted muscle fibres to probe the mechanism of force generation. J. Muscle Res. Cell Motil. 2006, 27, 455–468. [Google Scholar] [CrossRef]
- VanBuren, P.; Palmiter, K.A.; Warshaw, D.M. Tropomyosin directly modulates actomyosin mechanical performance at the level of a single actin filament. Proc. Natl. Acad. Sci. USA 1999, 96, 12488–12493. [Google Scholar] [CrossRef]
- Reconditi, M.; Brunello, E.; Fusi, L.; Linari, M.; Martínez, M.F.; Lombardi, V.; Irving, M.; Piazzesi, G. Sarcomere-length dependence of myosin filament structure in skeletal muscle fibres of the frog. J. Physiol. 2014, 592, 1119–1137. [Google Scholar] [CrossRef]
- Kampourakis, T.; Yan, Z.; Gautel, M.; Sun, Y.-B.; Irving, M. Myosin binding protein-C activates thin filaments and inhibits thick filaments in heart muscle cells. Proc. Natl. Acad. Sci. USA 2014, 111, 18763–18768. [Google Scholar] [CrossRef]
- Li, A.; Nelson, S.R.; Rahmanseresht, S.; Braet, F.; Cornachione, A.S.; Previs, S.B.; O’Leary, T.S.; McNamara, J.W.; Rassier, D.E.; Sadayappan, S.; et al. Skeletal MyBP-C isoforms tune the molecular contractility of divergent skeletal muscle systems. Proc. Natl. Acad. Sci. USA 2019, 116, 21882–21892. [Google Scholar] [CrossRef] [PubMed]
- Mun, J.Y.; Previs, M.J.; Yu, H.Y.; Gulick, J.; Tobacman, L.S.; Previs, S.B.; Robbins, J.; Warshaw, D.M.; Craig, R. Myosin-binding protein C displaces tropomyosin to activate cardiac thin filaments and governs their speed by an independent mechanism. Proc. Natl. Acad. Sci. USA 2014, 111, 2170–2175. [Google Scholar] [CrossRef]
- Inchingolo, A.V.; Previs, S.B.; Previs, M.J.; Warshaw, D.M.; Kad, N.M. Revealing the mechanism of how cardiac myosin-binding protein C N-terminal fragments sensitize thin filaments for myosin binding. Proc. Natl. Acad. Sci. USA 2019, 116, 6828–6835. [Google Scholar] [CrossRef]
- Previs, M.J.; Previs, S.B.; Gulick, J.; Robbins, J.; Warshaw, D.M. Molecular Mechanics of Cardiac Myosin-Binding Protein C in Native Thick Filaments. Science 2012, 337, 1215–1218. [Google Scholar] [CrossRef]
- Robinett, J.C.; Hanft, L.M.; Geist, J.; Kontrogianni-Konstantopoulos, A.; McDonald, K.S. Regulation of myofilament force and loaded shortening by skeletal myosin binding protein C. J. Gen. Physiol. 2019, 151, 645–659. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.D.; Regnier, M.; Daniel, T.L. Elastic Energy Storage and Radial Forces in the Myofilament Lattice Depend on Sarcomere Length. PLoS Comput. Biol. 2012, 8, e1002770. [Google Scholar] [CrossRef]
- Chase, P.B.; MacPherson, J.M.; Daniel, T.L. A Spatially Explicit Nanomechanical Model of the Half-Sarcomere: Myofilament Compliance Affects Ca2+-Activation. Ann. Biomed. Eng. 2004, 32, 1559–1568. [Google Scholar] [CrossRef]
- Gordon, A.M.; Huxley, A.F.; Julian, F.J. The variation in isometric tension with sarcomere length in vertebrate muscle fibres. J. Physiol. 1966, 184, 170–192. [Google Scholar] [CrossRef]
- Edman, K.A.P. The velocity of unloaded shortening and its relation to sarcomere length and isometric force in vertebrate muscle fibres. J. Physiol. 1979, 291, 143–159. [Google Scholar] [CrossRef] [PubMed]
- Edman, K.A.P.; Reggiani, C. The sarcomere length-tension relation determined in short segments of intact muscle fibres of the frog. J. Physiol. 1987, 385, 709–732. [Google Scholar] [CrossRef]
- Sundberg, M.; Rosengren, J.P.; Bunk, R.; Lindahl, J.; A Nicholls, I.; Tågerud, S.; Omling, P.; Montelius, L.; Månsson, A. Silanized surfaces for in vitro studies of actomyosin function and nanotechnology applications. Anal. Biochem. 2003, 323, 127–138. [Google Scholar] [CrossRef]
- Månsson, A. Translational actomyosin research: Fundamental insights and applications hand in hand. J. Muscle Res. Cell Motil. 2012, 33, 219–233. [Google Scholar] [CrossRef]
- Kishino, A.; Yanagida, T. Force measurements by micromanipulation of a single actin filament by glass needles. Nat. Cell Biol. 1988, 334, 74–76. [Google Scholar] [CrossRef]
- Fraser, I.D.C.; Marston, S.B. In Vitro Motility Analysis of Actin-Tropomyosin Regulation by Troponin and Calcium: The thin filament is switched as a single cooperative unit. J. Biol. Chem. 1995, 270, 7836–7841. [Google Scholar] [CrossRef]
- Homsher, E.; Kim, B.; Bobkova, A.; Tobacman, L. Calcium regulation of thin filament movement in an in vitro motility assay. Biophys. J. 1996, 70, 1881–1892. [Google Scholar] [CrossRef][Green Version]
- Saber, W.; Begin, K.J.; Warshaw, D.M.; VanBuren, P. Cardiac myosin binding protein-C modulates actomyosin binding and kinetics in the in vitro motility assay. J. Mol. Cell. Cardiol. 2008, 44, 1053–1061. [Google Scholar] [CrossRef]
- Tesi, C.; Colomo, F.; Piroddi, N.; Poggesi, C. Characterization of the cross-bridge force-generating step using inorganic phosphate and BDM in myofibrils from rabbit skeletal muscles. J. Physiol. 2002, 541, 187–199. [Google Scholar] [CrossRef]
- A Dantzig, J.; E Goldman, Y.; Millar, N.C.; Lacktis, J.; Homsher, E. Reversal of the cross-bridge force-generating transition by photogeneration of phosphate in rabbit psoas muscle fibres. J. Physiol. 1992, 451, 247–278. [Google Scholar] [CrossRef]
- Dantzig, J.A.; Hibberd, M.G.; Trentham, D.R.; Goldman, Y.E. Cross-bridge kinetics in the presence of MgADP investigated by photolysis of caged ATP in rabbit psoas muscle fibres. J. Physiol. 1991, 432, 639–680. [Google Scholar] [CrossRef] [PubMed]
- Funatsu, T.; Harada, Y.; Tokunaga, M.; Saito, K.; Yanagida, T. Imaging of single fluorescent molecules and individual ATP turnovers by single myosin molecules in aqueous solution. Nat. Cell Biol. 1995, 374, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Usaj, M.; Moretto, L.; Vemula, V.; Salhotra, A.; Månsson, A. Optimized single molecule fluorescence sheds light on elusive enzymatic mechanisms. bioRxiv 2020. [Google Scholar] [CrossRef]
- Hariadi, R.F.; Sommese, R.F.; Adhikari, A.S.; Taylor, R.; Sutton, S.; Spudich, J.A.; Sivaramakrishnan, S. Mechanical coordination in motor ensembles revealed using engineered artificial myosin filaments. Nat. Nanotechnol. 2015, 10, 696–700. [Google Scholar] [CrossRef]
- Sundberg, M.; Bunk, R.; Albet-Torres, N.; Kvennefors, A.; Persson, F.; Montelius, L.; Nicholls, I.A.; Ghatnekar-Nilsson, S.; Omling, P.; Tågerud, S.; et al. Actin Filament Guidance on a Chip: Toward High-Throughput Assays and Lab-on-a-Chip Applications. Langmuir 2006, 22, 7286–7295. [Google Scholar] [CrossRef]
- Srikakulam, R.; Winkelmann, D.A. Myosin II Folding Is Mediated by a Molecular Chaperonin. J. Biol. Chem. 1999, 274, 27265–27273. [Google Scholar] [CrossRef]
- Heald, R.W.; Hitchcock-DeGregori, S.E. The structure of the amino terminus of tropomyosin is critical for binding to actin in the absence and presence of troponin. J. Biol. Chem. 1988, 263, 5254–5259. [Google Scholar]
- Mendoza, A.C.; Rassier, D.E. Extraction of Thick Filaments in Individual Sarcomeres Affects Force Production by Single Myofibrils. Biophys. J. 2020, 118, 1921–1929. [Google Scholar] [CrossRef]
- Leite, F.D.S.; Minozzo, F.C.; Altman, D.; Rassier, D.E. Microfluidic perfusion shows intersarcomere dynamics within single skeletal muscle myofibrils. Proc. Natl. Acad. Sci. USA 2017, 114, 8794–8799. [Google Scholar] [CrossRef] [PubMed]
- Swartz, D.; Moss, R.; Greaser, M. Characteristics of troponin C binding to the myofibrillar thin filament: Extraction of troponin C is not random along the length of the thin filament. Biophys. J. 1997, 73, 293–305. [Google Scholar] [CrossRef][Green Version]
- Ishiwata, S.; Funatsu, T.; Fujita, H. Contractile properties of thin (actin) filament-reconstituted muscle fibers. Adv. Exp. Med. Biol. 1998, 453, 319–329. [Google Scholar] [CrossRef]
- Lu, X.; Heeley, D.H.; Smillie, L.B.; Kawai, M. The role of tropomyosin isoforms and phosphorylation in force generation in thin-filament reconstituted bovine cardiac muscle fibres. J. Muscle Res. Cell Motil. 2010, 31, 93–109. [Google Scholar] [CrossRef]
- Matusovsky, O.S.; Månsson, A.; Persson, M.; Cheng, Y.-S.; Rassier, D.E. High-speed AFM reveals subsecond dynamics of cardiac thin filaments upon Ca2+ activation and heavy meromyosin binding. Proc. Natl. Acad. Sci. USA 2019, 116, 16384–16393. [Google Scholar] [CrossRef]
- Thompson, R.E.; Larson, D.R.; Webb, W.W. Precise Nanometer Localization Analysis for Individual Fluorescent Probes. Biophys. J. 2002, 82, 2775–2783. [Google Scholar] [CrossRef]
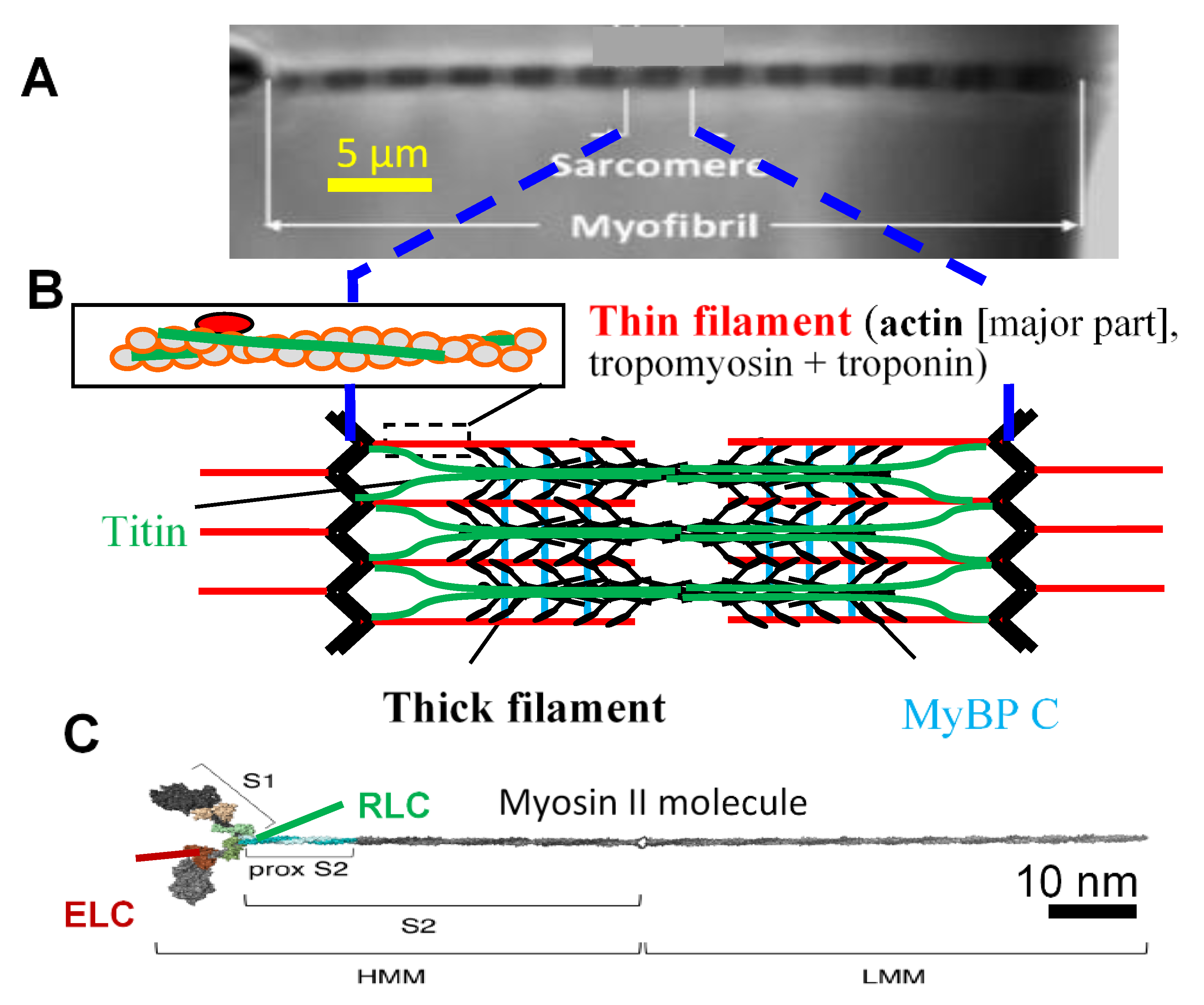
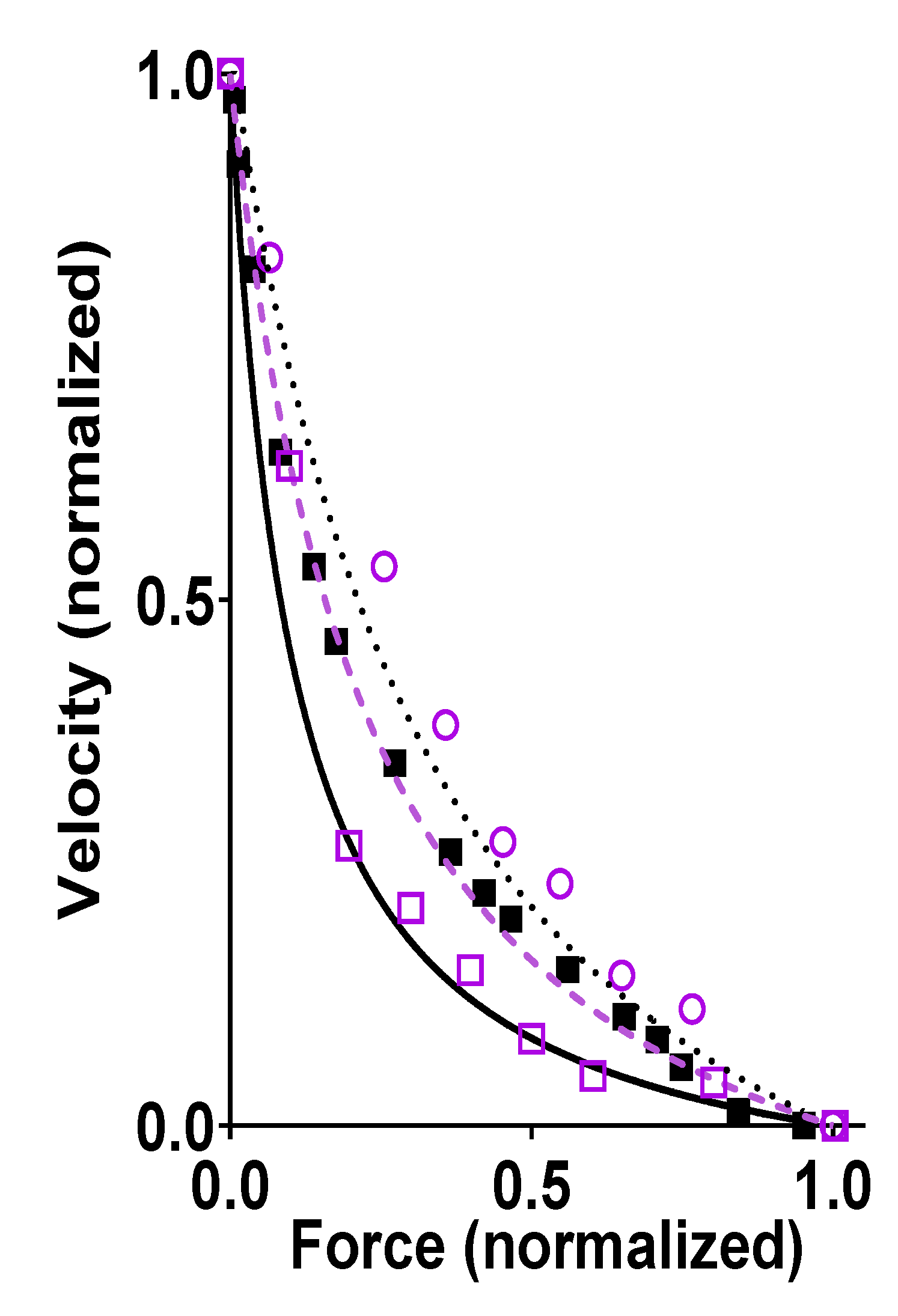




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Månsson, A. Hypothesis: Single Actomyosin Properties Account for Ensemble Behavior in Active Muscle Shortening and Isometric Contraction. Int. J. Mol. Sci. 2020, 21, 8399. https://doi.org/10.3390/ijms21218399
Månsson A. Hypothesis: Single Actomyosin Properties Account for Ensemble Behavior in Active Muscle Shortening and Isometric Contraction. International Journal of Molecular Sciences. 2020; 21(21):8399. https://doi.org/10.3390/ijms21218399
Chicago/Turabian StyleMånsson, Alf. 2020. "Hypothesis: Single Actomyosin Properties Account for Ensemble Behavior in Active Muscle Shortening and Isometric Contraction" International Journal of Molecular Sciences 21, no. 21: 8399. https://doi.org/10.3390/ijms21218399
APA StyleMånsson, A. (2020). Hypothesis: Single Actomyosin Properties Account for Ensemble Behavior in Active Muscle Shortening and Isometric Contraction. International Journal of Molecular Sciences, 21(21), 8399. https://doi.org/10.3390/ijms21218399





