18F-Fluorodeoxyglucose Positron Emission Tomography Tracks the Heterogeneous Brain Susceptibility to the Hyperglycemia-Related Redox Stress
Abstract
:1. Introduction
2. Results
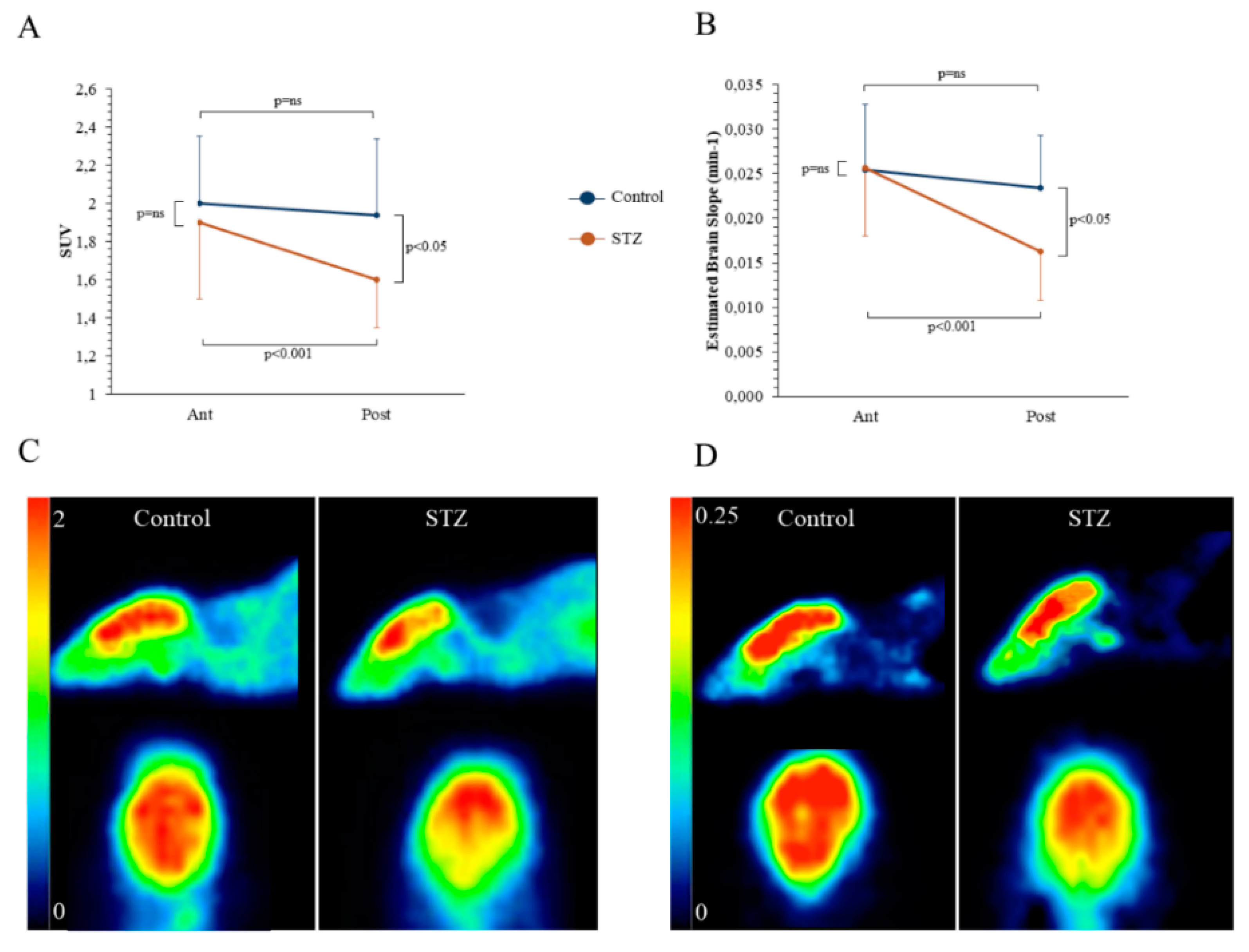
2.1. Effect of STZ on Serum Glucose Levels and Brain FDG Kinetics
2.2. Evaluation of Enzymatic Catalytic Functions
2.3. Regional Heterogeneity of the Hyperglycemia-Related Redox Damage
2.4. FDG Uptake Heterogeneity Tracks the Divergent ER-PPP Antioxidant Response to the Hyperglycemia-Related Redox Damage
3. Discussion
4. Materials and Methods
4.1. In Vivo Study Protocol
4.2. Micro-PET Imaging Protocol and Image Processing
4.3. Histochemical Analysis
4.4. Spectrophotometric Assays
4.5. Western Blot Analysis
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mergenthaler, P.; Lindauer, U.; Dienel, G.A.; Meisel, A. Sugar for the brain: The role of glucose in physiological and pathological brain function. Trends Neurosci. 2013, 36, 587–597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morbelli, S.; Bauckneht, M.; Scheltens, P. Imaging biomarkers in Alzheimer’s disease: Added value in the clinical setting. QJ Nucl. Med. Mol. Imaging 2017, 61, 360–371. [Google Scholar]
- Sarikaya, I.; Sarikaya, A.; Sharma, P. Assessing the Effect of Various Blood Glucose Levels on 18F-FDG Activity in the Brain, Liver, and Blood Pool. J. Nucl. Med. Technol. 2019, 47, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Varrone, A.; Asenbaum, S.; Borght, T.V.; Booij, J.; Nobili, F.; Någren, K.; Darcourt, J.; Kapucu, Ö.L.; Tatsch, K.; Bartenstein, P.; et al. EANM procedure guidelines for PET brain imaging using [18F]FDG, version 2. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 2103–2110. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, K.; Kameyama, M.; Ishii, K.; J-ADNI Study Group. Brain 18F-FDG distribution: Which region is most affected by increased plasma glucose levels? Ann. Nucl. Med. 2019, 33, 303–304. [Google Scholar] [CrossRef] [PubMed]
- Burns, C.M.; Chen, K.; Kaszniak, A.W.; Lee, W.; Alexander, G.E.; Bandy, D.; Fleisher, A.S.; Caselli, R.J.; Reiman, E.M. Higher serum glucose levels are associated with cerebral hypometabolism in Alzheimer regions. Neurology 2013, 80, 1557–1564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kawasaki, K.; Ishii, K.; Saito, Y.; Oda, K.; Kimura, Y.; Ishiwata, K. Influence of mild hyperglycemia on cerebral FDG distribution patterns calculated by statistical parametric mapping. Ann. Nucl. Med. 2008, 22, 191–200. [Google Scholar] [CrossRef]
- Apostolova, I.; Initiative, F.T.A.D.N.; Lange, C.; Suppa, P.; Spies, L.; Klutmann, S.; Adam, G.; Grothe, M.J.; Buchert, R. Impact of plasma glucose level on the pattern of brain FDG uptake and the predictive power of FDG PET in mild cognitive impairment. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1417–1422. [Google Scholar] [CrossRef]
- Reivich, M.; Kuhl, D.; Wolf, A.; Greenberg, J.; Phelps, M.; Ido, T.; Casella, V.; Fowler, J.; Hoffman, E.; Alavi, A.; et al. The [18F] fluorodeoxyglucose method for the measurement of local cerebral glucose utilization in man. Circ. Res. 1979, 44, 127–137. [Google Scholar] [CrossRef] [Green Version]
- Kanazawa, Y.; Yamane, H.; Shinohara, S.; Kuribayashi, S.; Momozono, Y.; Yamato, Y.; Kojima, M.; Masuda, K. 2-Deoxy-2-fluoro-D-glucose as a functional probe for NMR: The unique metabolism beyond its 6-phosphate. J. Neurochem. 1996, 66, 2113–2120. [Google Scholar] [CrossRef]
- Dienel, G.A.; Cruz, N.F. Synthesis of Deoxyglucose-1-Phosphate, Deoxyglucose-1,6-Bisphosphate, and Other Metabolites of 2-Deoxy-D-[14C] Glucose in Rat Brain In Vivo: Influence of Time and Tissue Glucose Level. J. Neurochem. 1993, 60, 2217–2231. [Google Scholar] [CrossRef] [PubMed]
- Southworth, R.; Parry, C.R.; Parkes, H.G.; Medina, R.A.; Garlick, P.B. Tissue-specific differences in 2-fluoro-2-deoxyglucose metabolism beyond FDG-6-P: A 19F NMR spectroscopy study in the rat. NMR Biomed. 2003, 16, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Marini, C.; Bianchi, G.; Buschiazzo, A.; Ravera, S.; Martella, R.; Bottoni, G.; Petretto, A.; Emionite, L.; Monteverde, E.; Capitanio, S.; et al. Divergent targets of glycolysis and oxidative phosphorylation result in additive effects of metformin and starvation in colon and breast cancer. Sci. Rep. 2016, 6, 19569. [Google Scholar] [CrossRef]
- Cossu, V.; Marini, C.; Piccioli, P.; Rocchi, A.; Bruno, S.; Orengo, A.M.; Emionite, L.; Bauckneht, M.; Grillo, F.; Capitanio, S.; et al. Obligatory role of endoplasmic reticulum in brain FDG uptake. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1184–1196. [Google Scholar] [CrossRef] [PubMed]
- Buschiazzo, A.; Cossu, V.; Bauckneht, M.; Orengo, A.; Piccioli, P.; Emionite, L.; Bianchi, G.; Grillo, F.; Rocchi, A.; Di Giulio, F.; et al. Effect of starvation on brain glucose metabolism and 18F-2-fluoro-2-deoxyglucose uptake: An experimental in-vivo and ex-vivo study. EJNMMI Res. 2018, 11, 44. [Google Scholar] [CrossRef] [PubMed]
- Bauckneht, M.; Pastorino, F.; Castellani, P.; Cossu, V.; Orengo, A.M.; Piccioli, P.; Emionite, L.; Capitanio, S.; Yosifov, N.; Bruno, S.; et al. Increased myocardial 18F-FDG uptake as a marker of Doxorubicin-induced oxidative stress. J. Nucl. Cardiol. 2019, 8, 1–12. [Google Scholar] [CrossRef]
- Bauckneht, M.; Cossu, V.; Castellani, P.; Piccioli, P.; Orengo, A.M.; Emionite, L.; Di Giulio, F.; Donegani, M.I.; Miceli, A.; Raffa, S.; et al. FDG uptake tracks the oxidative damage in diabetic skeletal muscle: An experimental study. Mol. Metab. 2020, 31, 98–108. [Google Scholar] [CrossRef]
- Marini, C.; Cossu, V.; Bonifacino, T.; Bauckneht, M.; Torazza, C.; Bruno, S.; Castellani, P.; Ravera, S.; Milanese, M.; Venturi, C.; et al. Mechanisms underlying the predictive power of high skeletal muscle uptake of FDG in amyotrophic lateral sclerosis. EJNMMI Res. 2020, 10, 76. [Google Scholar] [CrossRef]
- Marini, C.; Ravera, S.; Buschiazzo, A.; Bianchi, G.; Orengo, A.M.; Bruno, S.; Bottoni, G.; Emionite, L.; Pastorino, F.; Monteverde, E.; et al. Discovery of a novel glucose metabolism in cancer: The role of endoplasmic reticulum beyond glycolysis and pentose phosphate shunt. Sci. Rep. 2016, 6, 25092. [Google Scholar] [CrossRef] [Green Version]
- Scussolini, M.; Bauckneht, M.; Cossu, V.; Bruno, S.; Orengo, A.M.; Piccioli, P.; Capitanio, S.; Yosifov, N.; Ravera, S.; Morbelli, S.; et al. G6Pase location in the endoplasmic reticulum: Implications on compartmental analysis of FDG uptake in cancer cells. Sci. Rep. 2019, 9, 2794. [Google Scholar] [CrossRef] [Green Version]
- Cossu, V.; Bauckneht, M.; Bruno, S.; Orengo, A.M.; Emionite, L.; Balza, E.; Castellani, P.; Piccioli, P.; Miceli, A.; Raffa, S.; et al. The Elusive Link Between Cancer FDG Uptake and Glycolytic Flux Explains the Preserved Diagnostic Accuracy of PET/CT in Diabetes. Transl. Oncol. 2020, 13, 100752. [Google Scholar] [CrossRef] [PubMed]
- Olsen, B.B.; Gjedde, A.; Vilstrup, M.H.; Johnsen, I.B.G.; Neumann, G.; Torigian, D.A.; Alavi, A.; Høilund-Carlsen, P.F. Linked Hexokinase and Glucose-6-Phosphatase Activities Reflect Grade of Ovarian Malignancy. Mol. Imaging Biol. 2019, 21, 375–381. [Google Scholar] [CrossRef]
- Bouter, C.; Henniges, P.; Franke, T.N.; Irwin, C.; Sahlmann, C.O.; Sichler, M.E.; Beindorff, N.; Bayer, T.A.; Bouter, Y. 18F-FDG-PET Detects Drastic Changes in Brain Metabolism in the Tg4–42 Model of Alzheimer’s Disease. Front. Aging Neurosci. 2019, 10, 425. [Google Scholar] [CrossRef] [Green Version]
- Li, X.-Y.; Men, W.-W.; Zhu, H.; Lei, J.-F.; Zuo, F.-X.; Wang, Z.-J.; Zhu, Z.; Bao, X.-J.; Wang, R. Age- and Brain Region-Specific Changes of Glucose Metabolic Disorder, Learning, and Memory Dysfunction in Early Alzheimer’s Disease Assessed in APP/PS1 Transgenic Mice Using 18F-FDG-PET. Int. J. Mol. Sci. 2016, 17, 1707. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, K.; Kawasaki, K.; Ishiwata, K. Reduced uptake of 18F-FDG and 15O-H2O in Alzheimer’s disease-related regions after glucose loading. J. Cereb. Blood Flow Metab. 2015, 35, 1380–1385. [Google Scholar] [CrossRef] [Green Version]
- Viglianti, B.L.; Wale, D.J.; Ma, T.; Johnson, T.D.; Bohnen, N.I.; Wong, K.K.; Ky, C.; Frey, K.A.; Townsend, D.M.; Rubello, D.; et al. Effects of plasma glucose levels on regional cerebral 18F-fluorodeoxyglucose uptake: Implications for dementia evaluation with brain PET imaging. Biomed. Pharmacother. 2019, 112, 108628. [Google Scholar] [CrossRef]
- Puskas, F.; Gergely, P., Jr.; Banki, K. Stimulation of the pentose phosphate pathway and glutathione levels by dehydroascorbate, the oxidized form of vitamin C. FASEB J. 2000, 14, 1352–1361. [Google Scholar] [CrossRef]
- Ge, T.; Yang, J.; Zhou, S.; Wang, Y.; Li, Y.; Tong, X. The Role of the Pentose Phosphate Pathway in Diabetes and Cancer. Front. Endocrinol. 2020, 11, 365. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.L.; Goldfine, I.D.; Maddux, B.A.; Grodsky, G.M. Oxidative Stress and Stress-Activated Signaling Pathways: A Unifying Hypothesis of Type 2 Diabetes. Endocr. Rev. 2002, 23, 599–622. [Google Scholar] [CrossRef] [Green Version]
- Marbet, P.; Klusonova, P.; Birk, J.; Kratschmar, D.V.; Odermatt, A. Absence of hexose-6-phosphate dehydrogenase results in reduced overall glucose consumption but does not prevent 11β-hydroxysteroid dehydrogenase-1-dependent glucocorticoid activation. FEBS J. 2018, 285, 3993–4004. [Google Scholar] [CrossRef]
- Wang, B.; Miao, Y.; Zhao, Z.; Zhong, Y. Inflammatory Macrophages Promotes Development of Diabetic Encephalopathy. Cell. Physiol. Biochem. 2015, 36, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Bathina, S.; Srinivas, N.; Das, U.N. Streptozotocin produces oxidative stress, inflammation and decreases BDNF concentrations to induce apoptosis of RIN5F cells and type 2 diabetes mellitus in Wistar rats. Biochem. Biophys. Res. Commun. 2017, 486, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Wanrooy, B.J.; Kumar, K.P.; Wen, S.W.; Qin, C.X.; Ritchie, R.H.; Wong, C.H.Y. Distinct contributions of hyperglycemia and high-fat feeding in metabolic syndrome-induced neuroinflammation. J. Neuroinflamm. 2018, 15, 293. [Google Scholar] [CrossRef]
- National Institutes of Health. Guide for the Care and Use of Laboratory Animals; NIH Publication: Bethesda, MD, USA, 1985; pp. 23–85. [Google Scholar]
- Deeds, M.C.; Anderson, J.M.; Armstrong, A.S.; Gastineau, A.D.; Hiddinga, H.J.; Jahangir, A.; Eberhardt, N.L.; Kudva, Y.C. Single dose streptozotocin-induced diabetes: Considerations for study design in islet transplantation models. Lab. Anim. 2011, 45, 131–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayashi, K.; Kojima, R.; Ito, M. Strain Differences in the Diabetogenic Activity of Streptozotocin in Mice. Biol. Pharm. Bull. 2006, 29, 1110–1119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costes, S.V.; Daelemans, D.; Cho, E.H.; Dobbin, Z.; Pavlakis, G.; Lockett, S. Automatic and Quantitative Measurement of Protein-Protein Colocalization in Live Cells. Biophys. J. 2004, 86, 3993–4003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cappelli, E.; Degan, P.; Bruno, S.; Pierri, F.; Miano, M.; Raggi, F.; Farruggia, P.; Mecucci, C.; Crescenzi, B.; Naim, V.; et al. The passage from bone marrow niche to bloodstream triggers the metabolic impairment in Fanconi Anemia mononuclear cells. Redox Biol. 2020, 36, 101618. [Google Scholar] [CrossRef]
- Signorello, M.G.; Ravera, S.; Leoncini, G. Lectin-induced oxidative stress in human platelets. Redox Biol. 2020, 32, 101456. [Google Scholar] [CrossRef]
- Slinker, B.K.; Glantz, S.A. Multiple linear regression is a useful alternative to traditional analyses of variance. Am. J. Physiol. Integr. Comp. Physiol. 1988, 255, R353–R367. [Google Scholar] [CrossRef]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miceli, A.; Cossu, V.; Marini, C.; Castellani, P.; Raffa, S.; Donegani, M.I.; Bruno, S.; Ravera, S.; Emionite, L.; Orengo, A.M.; et al. 18F-Fluorodeoxyglucose Positron Emission Tomography Tracks the Heterogeneous Brain Susceptibility to the Hyperglycemia-Related Redox Stress. Int. J. Mol. Sci. 2020, 21, 8154. https://doi.org/10.3390/ijms21218154
Miceli A, Cossu V, Marini C, Castellani P, Raffa S, Donegani MI, Bruno S, Ravera S, Emionite L, Orengo AM, et al. 18F-Fluorodeoxyglucose Positron Emission Tomography Tracks the Heterogeneous Brain Susceptibility to the Hyperglycemia-Related Redox Stress. International Journal of Molecular Sciences. 2020; 21(21):8154. https://doi.org/10.3390/ijms21218154
Chicago/Turabian StyleMiceli, Alberto, Vanessa Cossu, Cecilia Marini, Patrizia Castellani, Stefano Raffa, Maria Isabella Donegani, Silvia Bruno, Silvia Ravera, Laura Emionite, Anna Maria Orengo, and et al. 2020. "18F-Fluorodeoxyglucose Positron Emission Tomography Tracks the Heterogeneous Brain Susceptibility to the Hyperglycemia-Related Redox Stress" International Journal of Molecular Sciences 21, no. 21: 8154. https://doi.org/10.3390/ijms21218154





