p53 Immunohistochemical Patterns in HPV-Independent Squamous Cell Carcinomas of the Vulva and the Associated Skin Lesions: A Study of 779 Cases
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Case Selection
4.2. Tissue Preparation, Nucleic-Acid Isolation, and Human Papillomavirus (HPV) DNA Detection
4.3. Histological Evaluation
4.4. p53 Immunohistochemical (IHC)
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- del Pino, M.; Rodriguez-Carunchio, L.; Ordi, J. Pathways of vulvar intraepithelial neoplasia and squamous cell carcinoma. Histopathology 2013, 62, 161–175. [Google Scholar] [CrossRef] [PubMed]
- Nitecki, R.; Feltmate, C.M. Human papillomavirus and nonhuman papillomavirus pathways to vulvar squamous cell carcinoma: A review. Curr. Probl. Cancer 2018, 42, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Tessier-Cloutier, B.; Pors, J.; Thompson, E.; Ho, J.; Prentice, L.; McConechy, M.; Aguirre-Hernandez, R.; Miller, R.; Leung, S.; Proctor, L.; et al. Molecular characterization of invasive and in situ squamous neoplasia of the vulva and implications for morphologic diagnosis and outcome. Mod. Pathol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Trietsch, M.D.; Nooij, L.S.; Gaarenstroom, K.N.; van Poelgeest, M.I.E. Genetic and epigenetic changes in vulvar squamous cell carcinoma and its precursor lesions: A review of the current literature. Gynecol. Oncol. 2015, 136, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Nooij, L.S.; Ter Haar, N.T.; Ruano, D.; Rakislova, N.; van Wezel, T.; Smit, V.T.; Trimbos, B.J.; Ordi, J.; Van Poelgeest, M.I.; Bosse, T. Genomic Characterization of Vulvar (Pre)cancers Identifies Distinct Molecular Subtypes with Prognostic Significance. Clin. Cancer Res. 2017, 23, 6781–6789. [Google Scholar] [CrossRef] [PubMed]
- Zięba, S.; Pouwer, A.F.W.; Kowalik, A.; Zalewski, K.; Rusetska, N.; Bakuła-Zalewska, E.; Kopczyński, J.; Pijnenborg, J.M.A.; de Hullu, J.A.; Kowalewska, M. Somatic mutation profiling in premalignant lesions of vulvar squamous cell carcinoma. Int. J. Mol. Sci. 2020, 21, 4880. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.P.; Miron, A.; Yassin, Y.; Monte, N.; Woo, T.Y.C.; Mehra, K.K.; Medeiros, F.; Crum, C.P. Differentiated vulvar intraepithelial neoplasia contains Tp53 mutations and is genetically linked to vulvar squamous cell carcinoma. Mod. Pathol. 2010, 23, 404–412. [Google Scholar] [CrossRef]
- Singh, N.; Leen, S.L.; Han, G.; Faruqi, A.; Kokka, F.; Rosenthal, A.; Jiang, X.R.; Kim, R.; McAlpine, J.N.; Gilks, C.B. Expanding the morphologic spectrum of differentiated VIN (dVIN) through detailed mapping of cases with p53 loss. Am. J. Surg. Pathol. 2015, 39, 52–60. [Google Scholar] [CrossRef]
- Rakislova, N.; Alemany, L.; Clavero, O.; Del Pino, M.; Saco, A.; Marimon, L.; Quirós, B.; Lloveras, B.; Ribera-Cortada, I.; Alejo, M.; et al. HPV-independent Precursors Mimicking High-grade Squamous Intraepithelial Lesions (HSIL) of the Vulva. Am. J. Surg. Pathol. 2020, 44, 1506–1514. [Google Scholar] [CrossRef]
- Ordi, J.; Alejo, M.; Fusté, V.; Lloveras, B.; Del Pino, M.; Alonso, I.; Torné, A. HPV-negative vulvar intraepithelial neoplasia (VIN) with basaloid histologic pattern: An unrecognized variant of simplex (differentiated) VIN. Am. J. Surg. Pathol. 2009, 33, 1659–1665. [Google Scholar] [CrossRef]
- Nascimento, A.F.; Granter, S.R.; Cviko, A.; Yuan, L.; Hecht, J.L.; Crum, C.P. Vulvar Acanthosis with Altered Differentiation: A Precursor to Verrucous Carcinoma? Am. J. Surg. Pathol. 2004, 28, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.C.; Howitt, B.E.; Horowitz, N.S.; Ritterhouse, L.L.; Dong, F.; Macconaill, L.E.; Garcia, E.; Lindeman, N.I.; Lee, L.J.; Berkowitz, R.S.; et al. Differentiated exophytic vulvar intraepithelial lesions are genetically distinct from keratinizing squamous cell carcinomas and contain mutations in PIK3CA. Mod. Pathol. 2017, 30, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Hoang, L.N.; Park, K.J.; Soslow, R.A.; Murali, R. Squamous precursor lesions of the vulva: Current classification and diagnostic challenges. Pathology 2016, 48, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Reutter, J.C.; Walters, R.A.; Selim, M.A. Differentiated Vulvar Intraepithelial Neoplasia: What Criteria Do We Use in Practice? J. Low. Genit. Tract Dis. 2016, 20, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Prieske, K.; Alawi, M.; Oliveira-Ferrer, L.; Jaeger, A.; Eylmann, K.; Burandt, E.; Schmalfeldt, B.; Joosse, S.A.; Woelber, L. Genomic characterization of vulvar squamous cell carcinoma. Gynecol. Oncol. 2020, 158, 547–554. [Google Scholar] [CrossRef]
- Kortekaas, K.E.; Bastiaannet, E.; van Doorn, H.C.; van Steenwijk, P.J.D.V.; Ewing-Graham, P.C.; Creutzberg, C.L.; Akdeniz, K.; Nooij, L.S.; Van Der Burg, S.H.; Bosse, T.; et al. Vulvar cancer subclassification by HPV and p53 status results in three clinically distinct subtypes. Gynecol. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ji, J.; Almadani, N.; Crawford, R.; Gilks, C.; Kinloch, M.; Hoang, L. Comparison of p53 Immunohistochemical Staining in Differentiated Vulvar Intraepithelial Neoplasia (dVIN) to Inflammatory Dermatoses and Benign Squamous Lesions in the Vulva. Histopathology 2020, 14238, his.14238. [Google Scholar] [CrossRef] [PubMed]
- Tessier-Cloutier, B.; Kortekaas, K.E.; Thompson, E.; Pors, J.; Chen, J.; Ho, J.; Prentice, L.M.; McConechy, M.K.; Chow, C.; Proctor, L.; et al. Major p53 immunohistochemical patterns in in situ and invasive squamous cell carcinomas of the vulva and correlation with TP53 mutation status. Mod. Pathol. 2020, 33, 1595–1605. [Google Scholar] [CrossRef] [PubMed]
- Kortekaas, K.E.; Solleveld-Westerink, N.; Tessier-Cloutier, B.; Rutten, T.A.; I E Van Poelgeest, M.; Gilks, C.B.; Hoang, L.N.; Bosse, T. Performance of the Pattern Based Interpretation of p53 Immunohistochemistry as a Surrogate for TP53 Mutations in Vulvar Squamous Cell Carcinoma. Histopathology 2020, 77, 92–99. [Google Scholar] [CrossRef]
- Thompson, E.F.; Chen, J.; Huvila, J.; Pors, J.; Ren, H.; Ho, J.; Chow, C.; Ta, M.; Proctor, L.; McAlpine, J.N.; et al. p53 Immunohistochemical patterns in HPV-related neoplasms of the female lower genital tract can be mistaken for TP53 null or missense mutational patterns. Mod. Pathol. 2020, 33, 1649–1659. [Google Scholar] [CrossRef]
- Santos, M.; Montagut, C.; Mellado, B.; Garcia, A.; Cajal, S.; Cardesa, A.; Puig-Tintoré, L.M.; Ordi, J. Immunohistochemical staining for p16 and p53 in premalignant and malignant epithelial lesions of the vulva. Int. J. Gynecol. Pathol. 2004, 23, 206–214. [Google Scholar] [CrossRef]
- Barlow, E.L.; Lambie, N.; Donoghoe, M.W.; Naing, Z.; Hacker, N.F. The Clinical Relevance of p16 and p53 Status in Patients with Squamous Cell Carcinoma of the Vulva. J. Oncol. 2020, 3739075. [Google Scholar] [CrossRef] [PubMed]
- Hay, C.M.; Lachance, J.A.; Lucas, F.L.; Smith, K.A.; Jones, M.A. Biomarkers p16, HPV, and p53 Predict Recurrence and Survival in Early Stage Squamous Cell Carcinoma of the Vulva. J. Low. Genit. Tract Dis. 2016, 20, 252–256. [Google Scholar] [CrossRef]
- Dong, F.; Kojiro, S.; Borger, D.R.; Growdon, W.B.; Oliva, E. Squamous Cell Carcinoma of the Vulva: A Subclassification of 97 Cases by Clinicopathologic, Immunohistochemical, and Molecular Features (p16, p53, and EGFR). Am. J. Surg. Pathol. 2015, 39, 1045–1053. [Google Scholar] [CrossRef]
- Rosenthal, A.N.; Hopster, D.; Ryan, A.; Jacobs, I.J. Immunohistochemical analysis of p53 in vulval intraepithelial neoplasia and vulval squamous cell carcinoma. Br. J. Cancer 2003, 88, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Choschzick, M.; Hantaredja, W.; Tennstedt, P.; Gieseking, F.; Wölber, L.; Simon, R. Role of TP53 mutations in vulvar carcinomas. Int. J. Gynecol. Pathol. 2011, 30, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Emanuels, A.G.; Koudstaal, J.; Burger, M.P.M.; Hollema, H. In squamous cell carcinoma of the vulva, overexpression of p53 is a late event and neither p53 nor mdm2 expression is a useful marker to predict lymph node metastases. Br. J. Cancer 1999, 80, 38–43. [Google Scholar] [CrossRef]
- Jeffreys, M.; Jeffus, S.K.; Herfs, M.; Quick, C.M. Accentuated p53 staining in usual type vulvar dysplasia-A potential diagnostic pitfall. Pathol. Res. Pract. 2018, 214, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Watkins, J.C.; Yang, E.; Crum, C.P.; Herfs, M.; Gheit, T.; Tommasino, M.; Nucci, M.R. Classic Vulvar Intraepithelial Neoplasia with Superimposed Lichen Simplex Chronicus: A Unique Variant Mimicking Differentiated Vulvar Intraepithelial Neoplasia. Int. J. Gynecol. Pathol. 2019, 38, 175–182. [Google Scholar] [CrossRef]
- Akbari, A.; Pinto, A.; Amemiya, Y.; Seth, A.; Mirkovic, J.; Parra-Herran, C. Differentiated exophytic vulvar intraepithelial lesion: Clinicopathologic and molecular analysis documenting its relationship with verrucous carcinoma of the vulva. Mod. Pathol. 2020, 33, 2011–2018. [Google Scholar] [CrossRef]
- Liegl, B.; Regauer, S. p53 immunostaining in lichen sclerosus is related to ischaemic stress and is not a marker of differentiated vulvar intraepithelial neoplasia (d-VIN). Histopathology 2006, 48, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Zięba, S.; Kowalik, A.; Zalewski, K.; Rusetska, N.; Goryca, K.; Piaścik, A.; Misiek, M.; Bakuła-Zalewska, E.; Kopczyński, J.; Kowalski, K.; et al. Somatic mutation profiling of vulvar cancer: Exploring therapeutic targets. Gynecol. Oncol. 2018, 150, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Mulvany, N.J.; Allen, D.G. Differentiated intraepithelial neoplasia of the vulva. Int. J. Gynecol. Pathol. 2008, 27, 125–135. [Google Scholar] [CrossRef]
- de Sanjosé, S.; Alemany, L.; Ordi, J.; Tous, S.; Alejo, M.; Bigby, S.M.; Joura, E.A.; Maldonado, P.; Laco, J.; Bravo, I.G.; et al. Worldwide human papillomavirus genotype attribution in over 2000 cases of intraepithelial and invasive lesions of the vulva. Eur. J. Cancer 2013, 49, 3450–3461. [Google Scholar] [CrossRef]
- Rakislova, N.; Clavero, O.; Alemany, L.; Saco, A.; Quirós, B.; Lloveras, B.; Alejo, M.; Pawlita, M.; Quint, W.; Del Pino, M.; et al. “Histological characteristics of HPV-associated and -independent squamous cell carcinomas of the vulva: A study of 1,594 cases”. Int. J. Cancer 2017, 141, 2517–2527. [Google Scholar] [CrossRef]
- Rakislova, N.; Alemany, L.; Clavero, O.; Del Pino, M.; Saco, A.; Quirós, B.; Lloveras, B.; Alejo, M.; Halec, G.; Quint, W.; et al. Differentiated Vulvar Intraepithelial Neoplasia-like and Lichen Sclerosus-like Lesions in HPV-associated Squamous Cell Carcinomas of the Vulva. Am. J. Surg. Pathol. 2018, 42, 828–835. [Google Scholar] [CrossRef]
- Scurry, J.; Whitehead, J.; Healey, M. Histology of lichen sclerosus varies according to site and proximity to carcinoma. Am. J. Dermatopathol. 2001, 23, 413–418. [Google Scholar] [CrossRef] [PubMed]
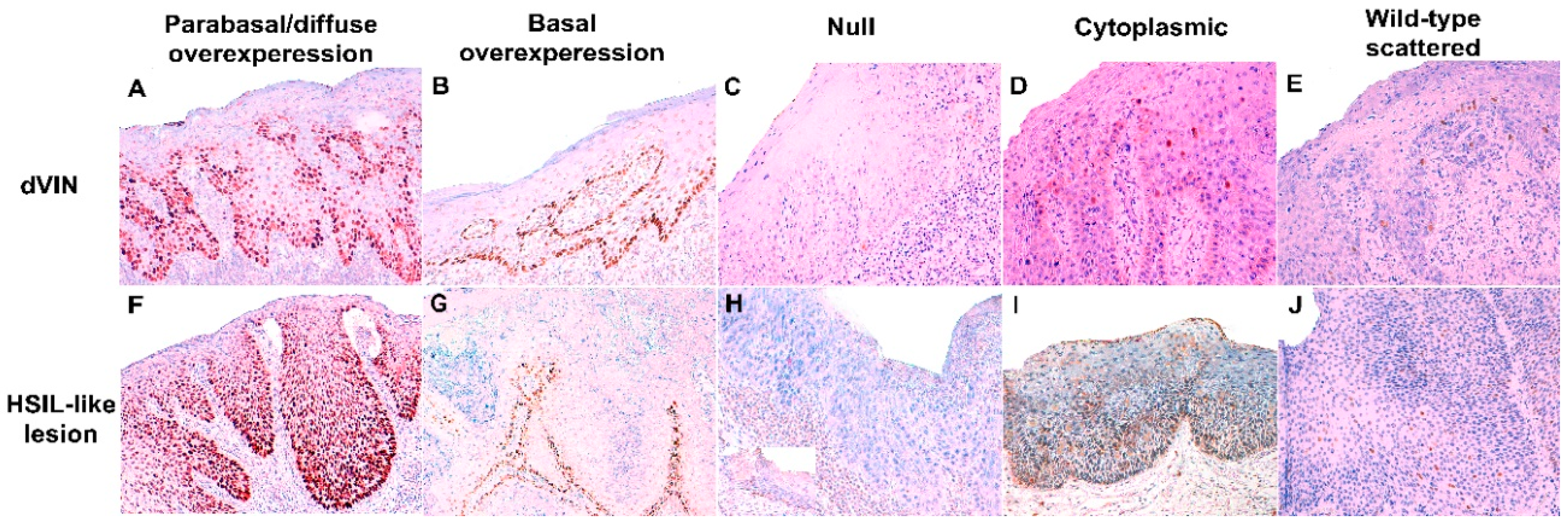


| Pattern of p53 Expression | ||||||
|---|---|---|---|---|---|---|
| Abnormal Patterns | Wild-Type Patterns | |||||
| Adjacent Skin Lesion | Basal Overexpression | Parabasal/Diffuse Overexpression | Null | Cytoplasmic | Scattered | Mid-Epithelial |
| Intraepithelial precursors | ||||||
| dVIN (n = 186) | 48 (25.8%) | 67 (36.0%) | 13 (7.0%) | 1 (0.5%) | 57 (30.7%) | 0 (0%) |
| HSIL-like lesion (n = 46) | 2 (4.3%) | 27 (58.7%) | 4 (8.7%) | 1 (2.2%) | 11 (23.9%) | 1 (2.2%) |
| VAAD/DEVIL (n = 22) | 5 (22.7%) | 4 (18.2%) | 0 (0%) | 0 (0%) | 13 (59.1%) | 0 (0%) |
| Inflammatory lesions | ||||||
| Lichen sclerosus (n = 36) | 6 (16.7%) | 0 0%) | 3 (8.3%) | 0 (0%) | 27 (75.0%) | 0 (0%) |
| Other inflammatory/reactive lesions (n = 160) | 14 (8.7%) | 15 (9.4%) | 0 (0%) | 0 (0%) | 131 (81.9%) | 0 (0%) |
| Total | 75 | 113 | 20 | 2 | 239 | 1 |
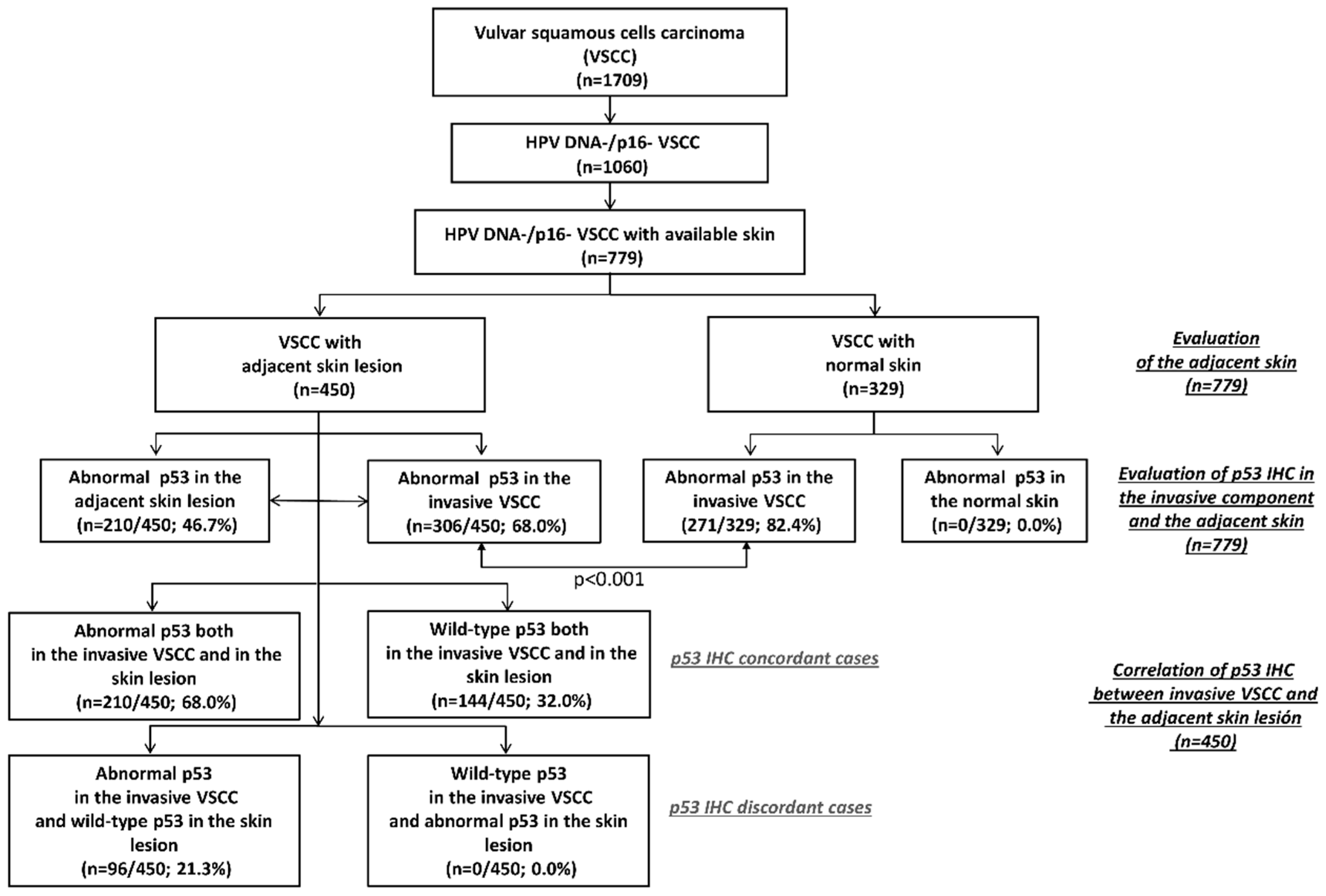
| Pattern of p53 Expression in the Adjacent Skin Lesion | ||||||
|---|---|---|---|---|---|---|
| Abnormal Patterns (n = 210) | Wild-Type Patterns (n = 240) | |||||
| Pattern of p53 Expression in the Invasive Carcinoma | Basal Overexpression | Parabasal/Diffuse Overexpression | Null | Cytoplasmic | Scattered | Mid-Epithelial |
| Abnormal patterns (n = 306) | ||||||
| Basal overexpression (n = 84) | 39 (46.4%) | 6 (7.2%) | 0 (0%) | 0 (0%) | 39 (46.4%) | 0 (0%) |
| Parabasal/Diffuse overexpression (n = 193) | 36 (18.7%) | 107 (55.4%) | 0 (0%) | 0 (0%) | 50 (25.9%) | 0 (0%) |
| Null (n = 27) | 0 (0%) | 0 (0%) | 20 (74.1%) | 0 (0%) | 7 (25.9%) | 0 (0%) |
| Cytoplasmic (n = 2) | 0 (0%) | 0 (0%) | 0 (0%) | 2 (100%) | 0 (0%) | 0 (0%) |
| Wild-type patterns (n = 144) | ||||||
| Scattered (n = 143) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 143 (100%) | 0 (0%) |
| Mid-epithelial (n = 1) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 1 (100%) |
| Total | 75 | 113 | 20 | 2 | 239 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rakislova, N.; Alemany, L.; Clavero, O.; Saco, A.; Torné, A.; del Pino, M.; Munmany, M.; Rodrigo-Calvo, M.T.; Guerrero, J.; Marimon, L.; et al. p53 Immunohistochemical Patterns in HPV-Independent Squamous Cell Carcinomas of the Vulva and the Associated Skin Lesions: A Study of 779 Cases. Int. J. Mol. Sci. 2020, 21, 8091. https://doi.org/10.3390/ijms21218091
Rakislova N, Alemany L, Clavero O, Saco A, Torné A, del Pino M, Munmany M, Rodrigo-Calvo MT, Guerrero J, Marimon L, et al. p53 Immunohistochemical Patterns in HPV-Independent Squamous Cell Carcinomas of the Vulva and the Associated Skin Lesions: A Study of 779 Cases. International Journal of Molecular Sciences. 2020; 21(21):8091. https://doi.org/10.3390/ijms21218091
Chicago/Turabian StyleRakislova, Natalia, Laia Alemany, Omar Clavero, Adela Saco, Aureli Torné, Marta del Pino, Meritxell Munmany, Maria Teresa Rodrigo-Calvo, José Guerrero, Lorena Marimon, and et al. 2020. "p53 Immunohistochemical Patterns in HPV-Independent Squamous Cell Carcinomas of the Vulva and the Associated Skin Lesions: A Study of 779 Cases" International Journal of Molecular Sciences 21, no. 21: 8091. https://doi.org/10.3390/ijms21218091
APA StyleRakislova, N., Alemany, L., Clavero, O., Saco, A., Torné, A., del Pino, M., Munmany, M., Rodrigo-Calvo, M. T., Guerrero, J., Marimon, L., Vega, N., Quirós, B., Lloveras, B., Ribera-Cortada, I., Alejo, M., Pawlita, M., Quint, W., de Sanjose, S., Ordi, J., & VVAP Study Group. (2020). p53 Immunohistochemical Patterns in HPV-Independent Squamous Cell Carcinomas of the Vulva and the Associated Skin Lesions: A Study of 779 Cases. International Journal of Molecular Sciences, 21(21), 8091. https://doi.org/10.3390/ijms21218091









