Conjugation with Methylsulfonylmethane Improves Hyaluronic Acid Anti-Inflammatory Activity in a Hydrogen Peroxide-Exposed Tenocyte Culture In Vitro Model
Abstract
:1. Introduction
2. Results
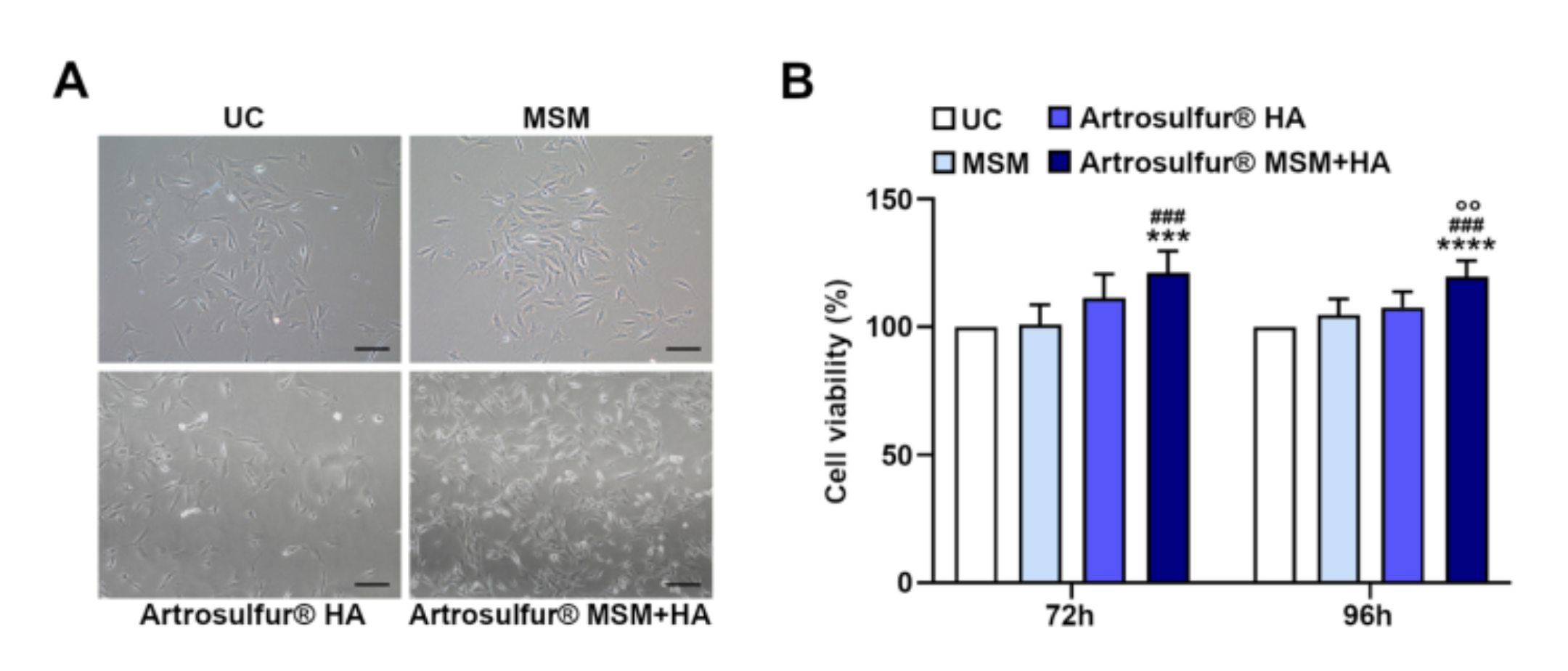
2.1. Cell Morphology and Proliferation
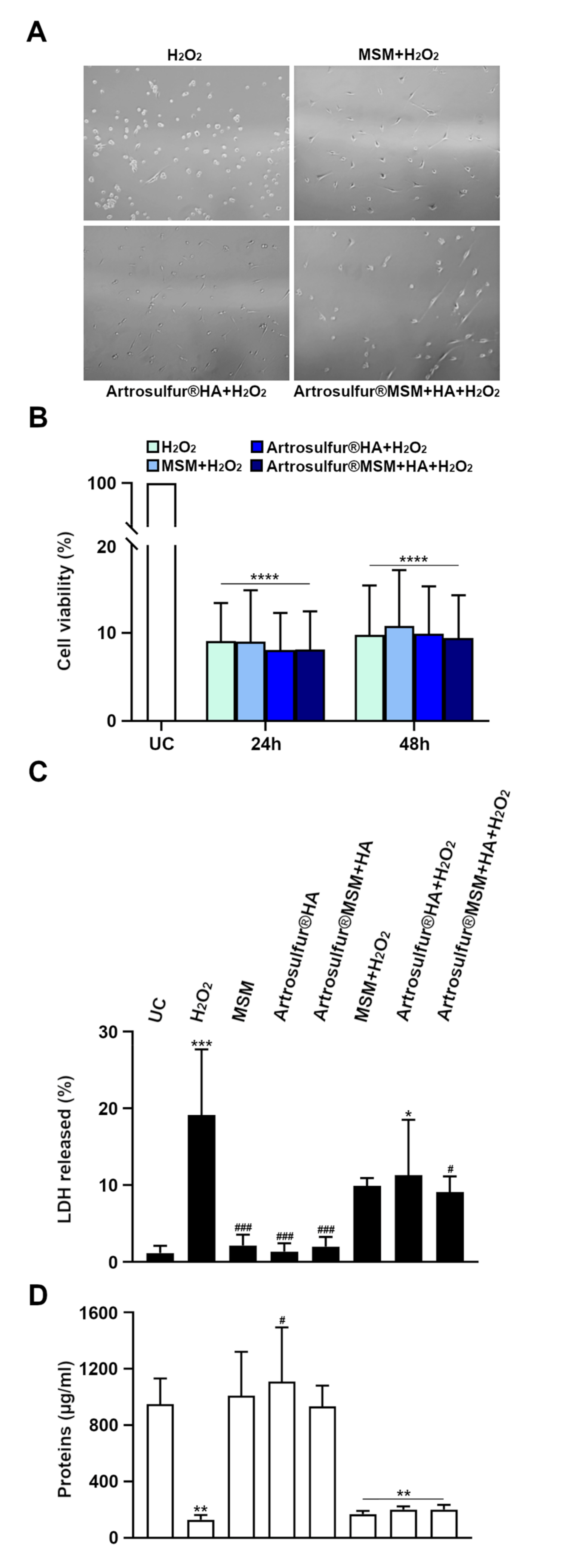
2.2. Lactate Dehydrogenase (LDH) Cytotoxicity Assay and Protein Quantification
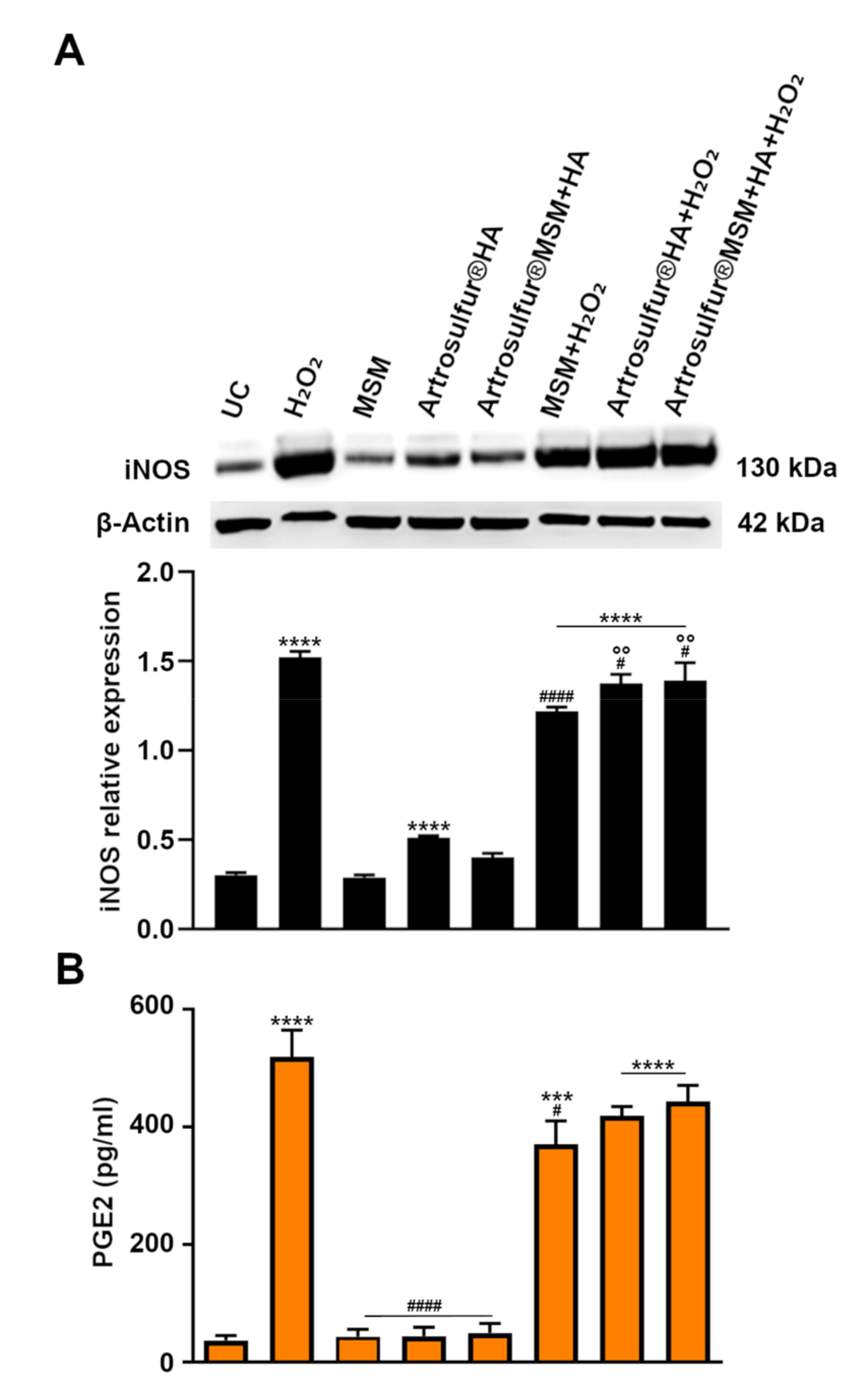
2.3. iNOS Expression
2.4. PGE2 Secretion
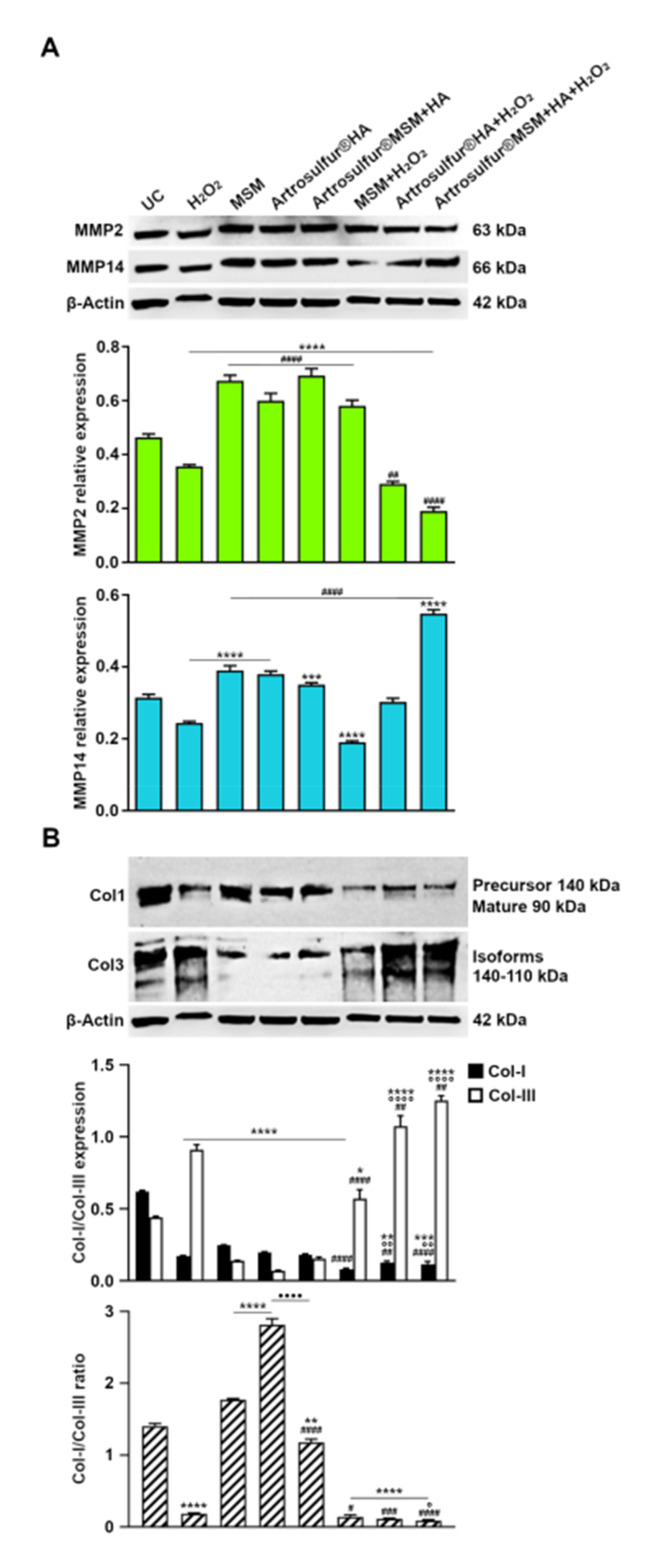
2.5. Metalloproteinases and Expression of Collagen Proteins
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Cell Treatment
4.3. H2O2 Treatment
4.4. MTT Assay for cell Proliferation
4.5. Lactate Dehydrogenase (LDH) Release Assay
4.6. Cell Lysis and Protein Extraction
4.7. Prostaglandin E2 Secretion
4.8. Western Blot Analysis
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oliva, F.; Osti, L.; Padulo, J.; Maffulli, N. Epidemiology of the rotator cuff tears: A new incidence related to thyroid disease. Muscles Ligaments Tendons J. 2014, 4, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Honda, H.; Gotoh, M.; Kanazawa, T.; Ohzono, H.; Nakamura, H.; Ohta, K.; Nakamura, K.I.; Fukuda, K.; Teramura, T.; Hashimoto, T.; et al. Hyaluronic Acid Accelerates Tendon-to-Bone Healing after Rotator Cuff Repair. Am. J. Sports Med. 2017, 45, 3322–3330. [Google Scholar] [CrossRef] [PubMed]
- Osti, L.; Buda, M.; Buono, A.D.; Osti, R.; Massari, L. Clinical evidence in the treatment of rotator cuff tears with hyaluronic acid. Muscles Ligaments Tendons J. 2016, 5, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Cipollaro, L.; Sahemey, R.; Oliva, F.; Maffulli, N. Immunohistochemical features of rotator cuff tendinopathy. Br. Med. Bull. 2019, 130, 105–123. [Google Scholar] [CrossRef]
- Frizziero, A.; Vittadini, F.; Oliva, F.; Abatangelo, G.; Bacciu, S.; Berardi, A.; Bossa, M.; Buda, R.; Buonocore, D.; Calderazzi, F.; et al. Hyaluronic acid injections in musculoskeletal disorders guidelines. Muscles Ligaments Tendons J. 2018, 8, 364–398. [Google Scholar] [CrossRef] [Green Version]
- Osti, L.; Berardocco, M.; di Giacomo, V.; Di Bernardo, G.; Oliva, F.; Berardi, A.C. Hyaluronic acid increases tendon derived cell viability and collagen type I expression in vitro: Comparative study of four different Hyaluronic acid preparations by molecular weight. BMC Musculoskelet. Disord. 2015, 16, 284. [Google Scholar] [CrossRef] [Green Version]
- Gallorini, M.; Berardi, A.C.; Berardocco, M.; Gissi, C.; Maffulli, N.; Cataldi, A.; Oliva, F. Hyaluronic acid increases tendon derived cell viability and collagen type I expression in vitro: Comparative study of four different Hyaluronic acid preparations by molecular weight. Muscles Ligaments Tendons J. 2017, 7, 208–214. [Google Scholar] [CrossRef]
- Gallorini, M.; Berardi, A.C.; Gissi, C.; Cataldi, A.; Osti, L. Nrf2-mediated cytoprotective effect of four different hyaluronic acids by molecular weight in human tenocytes. J. Drug Target 2020, 28, 212–224. [Google Scholar] [CrossRef]
- Butawan, M.; Benjamin, R.L.; Bloomer, R.J. Methylsulfonylmethane: Applications and Safety of a Novel Dietary Supplement. Nutrients 2017, 9, 290. [Google Scholar] [CrossRef]
- Kamel, R.; El Morsy, E.M. Hepatoprotective effect of methylsulfonylmethane against carbon tetrachloride-induced acute liver injury in rats. Arch. Pharm. Res. 2013, 36, 1140–1148. [Google Scholar] [CrossRef]
- Kim, L.S.; Axelrod, L.J.; Howard, P.; Buratovich, N.; Waters, R.F. Efficacy of methylsulfonylmethane (MSM) in osteoarthritis pain of the knee: A pilot clinical trial. Osteoarthr. Cartil. 2006, 14, 286–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bokhari, A.R.; Murrell, G.A. The role of nitric oxide in tendon healing. J. Shoulder Elb. Surg. 2012, 21, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Murrell, G.A. Using nitric oxide to treat tendinopathy. Br. J. Sports Med. 2007, 41, 227–231. [Google Scholar] [CrossRef] [Green Version]
- Bergqvist, F.; Carr, A.J.; Wheway, K.; Watkins, B.; Oppermann, U.; Jakobsson, P.J.; Dakin, S.G. Divergent roles of prostacyclin and PGE(2) in human tendinopathy. Arthritis Res. Ther. 2019, 21, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dakin, S.G.; Dudhia, J.; Werling, N.J.; Werling, D.; Abayasekara, D.R.E.; Smith, R.K. Inflamm-aging and arachadonic acid metabolite differences with stage of tendon disease. PLoS ONE 2012, 7, e48978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thankam, F.G.; Dilisio, M.F.; Gross, R.M.; Agrawal, D.K. Collagen I: A kingpin for rotator cuff tendon pathology. Am. J. Transl. Res. 2018, 15, 3291–3309. [Google Scholar]
- Del Buono, A.; Oliva, F.; Longo, U.G.; Rodeo, S.A.; Orchard, J.; Denaro, V.; Maffulli, N. Metalloproteases and rotator cuff disease. J. Shoulder Elb. Surg. 2012, 21, 200–208. [Google Scholar] [CrossRef]
- Davis, M.E.; Gumucio, J.P.; Sugg, K.B.; Bedi, A.; Mendias, C.L. MMP inhibition as a potential method to augment the healing of skeletal muscle and tendon extracellular matrix. J. Appl. Physiol. 2013, 115, 884–891. [Google Scholar] [CrossRef] [Green Version]
- Michele, A.; Cosima, S.; Vincenzo, S. The Use of Hyaluronic Acid after Tendon Surgery and in Tendinopathies. BioMed Res. Int. 2014, 2014, 783632. [Google Scholar] [CrossRef] [Green Version]
- Pereira, H.; Sousa, D.A.; Cunha, A.; Andrade, R.; Espregueira-Mendes, J.; Oliveira, J.M.; Reis, R.L. Hyaluronic Acid. Adv. Exp. Med. Biol. 2018, 1059, 137–153. [Google Scholar] [CrossRef]
- Aljohani, H.; Senbanjo, L.T.; Chellaiah, M.A. Methylsulfonylmethane increases osteogenesis and regulates the mineralization of the matrix by transglutaminase 2 in SHED cells. PLoS ONE 2019, 5, e0225598. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.R.; Nguyen, T.B.; Min, Y.K.; Le, B.T. In vitro and in vivo studies of BMP-2-loaded PCL-gelatin-BCP electrospun scaffolds. Tissue Eng. Part A 2014, 20, 3279–3289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.H.; Sp, N.; Kang, D.Y.; Jo, E.S.; Rugamba, A.; Jang, K.J.; Yang, Y.M. Effect of Methylsulfonylmethane on Proliferation and Apoptosis of A549 Lung Cancer Cells Through G(2)/M Cell-cycle Arrest and Intrinsic Cell Death Pathway. Anticancer Res. 2020, 40, 1905–1913. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Park, J.W.; Yang, Y.M.; Song, K.D.; Cho, B.W. Methylsulfonylmethane affects oxidative stress and CYP3A93 expression in fetal horse liver cells. Asian Australas. J. Anim. Sci. 2020, 24. [Google Scholar] [CrossRef]
- Karabay, A.Z.; Aktan, F.; Sunguroğlu, A.; Buyukbingol, Z. Methylsulfonylmethane modulates apoptosis of LPS/IFN-γ-activated RAW 264.7 macrophage-like cells by targeting p53, Bax, Bcl-2, cytochrome c and PARP proteins. Immunopharmacol. Immunotoxicol. 2014, 36, 379–389. [Google Scholar] [CrossRef]
- Xia, W.; Szomor, Z.; Wang, Y.; Murrell, G.A.C. Nitric oxide enhances collagen synthesis in cultured human tendon cells. J. Orthop. Res. 2006, 24, 159–172. [Google Scholar] [CrossRef]
- Millar, N.L.; Murrell, G.A.C.; McInnes, I.B. Inflammatory mechanisms in tendinopathy—Towards translation. Nat. Rev. Rheumatol. 2017, 13, 110–122. [Google Scholar] [CrossRef]
- Loiacono, C.; Palermi, S.; Massa, B.; Belviso, I.; Romano, V.; Gregorio, A.D.; Sirico, F.; Sacco, A.M. Tendinopathy: Pathophysiology, therapeutic options, and role of nutraceutics. A narrative literature review. Medicina 2019, 55, 447. [Google Scholar] [CrossRef] [Green Version]
- Kim, Y.H.; Kim, D.H.; Lim, H.; Baek, D.Y.; Shin, H.K.; Kim, J.K. The anti-inflammatory effects of methylsulfonylmethane on lipopolysaccharide-induced inflammatory responses in murine macrophages. Biol. Pharm. Bull. 2009, 32, 651–656. [Google Scholar] [CrossRef] [Green Version]
- Grzanna, M.W.; Au, R.Y.; Au, A.Y.; Rashmir, A.M.; Frondoza, C.G. Avocado/Soybean Unsaponifiables, Glucosamine and Chondroitin Sulfate Combination Inhibits Proinflammatory COX-2 Expression and Prostaglandin E2 Production in Tendon-Derived Cells. J. Med. Food 2020, 23, 139–146. [Google Scholar] [CrossRef]
- Caley, M.P.; Martins, V.L.; O’Toole, E.A. Metalloproteinases and Wound Healing. Adv. Wound Care (New Rochelle) 2015, 4, 225–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, S.H.; Yeung, C.Y.; Kalson, N.S.; Lu, Y.; Zigrino, P.; Starborg, T.; Warwood, S.; Holmes, D.F.; Canty-Laird, E.G.; Mauch, C.; et al. Matrix metalloproteinase 14 is required for fibrous tissue expansion. eLife 2015, 4, e09345. [Google Scholar] [CrossRef] [PubMed]
- Del Buono, A.; Oliva, F.; Osti, L.; Maffulli, N. Metalloproteases and tendinopathy. Muscles Ligaments Tendons J. 2013, 3, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Attia, M.; Huet, E.; Gossard, C.; Menashi, S.; Tassoni, M.C.; Martelly, I. Early events of overused supraspinatus tendons involve matrix metalloproteinases and EMMPRIN/CD147 in the absence of inflammation. Am. J. Sports Med. 2013, 41, 908–917. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, D.; Itoigawa, Y.; Nojiri, H.; Sano, H.; Itoi, E.; Saijo, Y.; Kaneko, K.; Shimizu, T. Contribution of oxidative stress to the degeneration of rotator cuff entheses. J. Shoulder Elb. Surg. 2014, 23, 628–635. [Google Scholar] [CrossRef]
- Docheva, D.; Müller, S.A.; Majewski, M.; Evans, C.H. Biologics for tendon repair. Adv. Drug Deliv. Rev. 2015, 84, 222–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliva, F.; Berardi, A.C.; Misiti, S.; Falzacappa, C.V.; Iacone, A.; Maffulli, N. Thyroid hormones enhance growth and counteract apoptosis in human tenocytes isolated from rotator cuff tendons. Cell Death Dis. 2013, 4, e705. [Google Scholar] [CrossRef] [Green Version]
- Yao, L.; Bestwick, C.S.; Bestwick, L.A.; Maffulli, N.; Aspden, R.M. Phenotypic drift in human tenocyte culture. Tissue Eng. 2006, 12, 1843–1849. [Google Scholar] [CrossRef]
- Rapino, M.; Di Valerio, V.; Zara, S.; Gallorini, M.; Marconi, G.D.; Sancilio, S.; Marsich, E.; Ghinassi, B.; di Giacomo, V.; Cataldi, A. Chitlac-coated Thermosets Enhance Osteogenesis and Angiogenesis in a Co-culture of Dental Pulp Stem Cells and Endothelial Cells. Nanomaterials 2019, 9, 928. [Google Scholar] [CrossRef] [Green Version]
- Frizziero, A.; Oliva, F.; Vittadini, F.; Vetrano, M.; Bernetti, A.; Giordan, N.; Vulpiani, M.C.; Santilli, V.; Masiero, S.; Maffulli, N. Efficacy of ultrasound-guided hyaluronic acid injections in achilles and patellar tendinopathies: A prospective multicentric clinical trial. MLTJ 2019, 9, 305–313. [Google Scholar] [CrossRef] [Green Version]
| Commercial Name | Active Substance | Molecular Weight | Source | Doses Tested | Manufacturer |
|---|---|---|---|---|---|
| Artrosulfur® HA | Linear Sodium Hyaluronate | 1200 KDa | Bacterial Fermentation | 1000 ug/mL | Alfakjn S.r.l., Garlasco (PV) Italy |
| MSM | Methylsulfonylmethane | / | Oxidation of DMSO with hydrogen (H2O2) and purified by distillation | 3125 ug/mL | Barentz, Paderno Dugnano (MI), Italy |
| Artrosulfur® HA + HA | Linear Sodium Hyaluronate + Methylsulfonylmethane (5% w/v) | 1200 KDa | Bacterial Fermentation | 1000 ug/mL + 3125 ug/mL | Alfakjn S.r.l., Garlasco (PV) Italy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliva, F.; Gallorini, M.; Antonetti Lamorgese Passeri, C.; Gissi, C.; Ricci, A.; Cataldi, A.; Colosimo, A.; Berardi, A.C. Conjugation with Methylsulfonylmethane Improves Hyaluronic Acid Anti-Inflammatory Activity in a Hydrogen Peroxide-Exposed Tenocyte Culture In Vitro Model. Int. J. Mol. Sci. 2020, 21, 7956. https://doi.org/10.3390/ijms21217956
Oliva F, Gallorini M, Antonetti Lamorgese Passeri C, Gissi C, Ricci A, Cataldi A, Colosimo A, Berardi AC. Conjugation with Methylsulfonylmethane Improves Hyaluronic Acid Anti-Inflammatory Activity in a Hydrogen Peroxide-Exposed Tenocyte Culture In Vitro Model. International Journal of Molecular Sciences. 2020; 21(21):7956. https://doi.org/10.3390/ijms21217956
Chicago/Turabian StyleOliva, Francesco, Marialucia Gallorini, Cristina Antonetti Lamorgese Passeri, Clarissa Gissi, Alessia Ricci, Amelia Cataldi, Alessia Colosimo, and Anna Concetta Berardi. 2020. "Conjugation with Methylsulfonylmethane Improves Hyaluronic Acid Anti-Inflammatory Activity in a Hydrogen Peroxide-Exposed Tenocyte Culture In Vitro Model" International Journal of Molecular Sciences 21, no. 21: 7956. https://doi.org/10.3390/ijms21217956
APA StyleOliva, F., Gallorini, M., Antonetti Lamorgese Passeri, C., Gissi, C., Ricci, A., Cataldi, A., Colosimo, A., & Berardi, A. C. (2020). Conjugation with Methylsulfonylmethane Improves Hyaluronic Acid Anti-Inflammatory Activity in a Hydrogen Peroxide-Exposed Tenocyte Culture In Vitro Model. International Journal of Molecular Sciences, 21(21), 7956. https://doi.org/10.3390/ijms21217956







