Preparation of Defined Chitosan Oligosaccharides Using Chitin Deacetylases
Abstract
:1. Why Using Defined Chitosans Is so All-Important in Bioactivity Studies
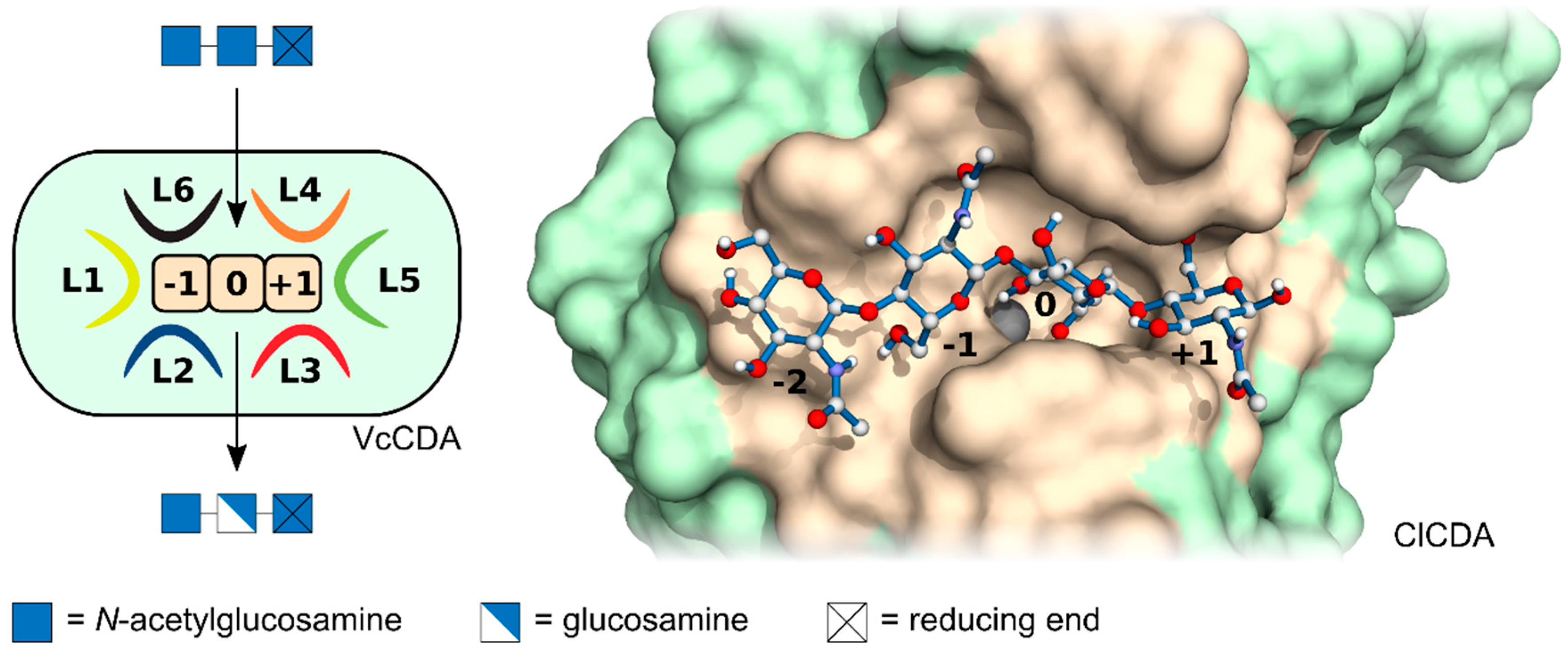
2. Regio-Selective Chitin de-N-Acteylases
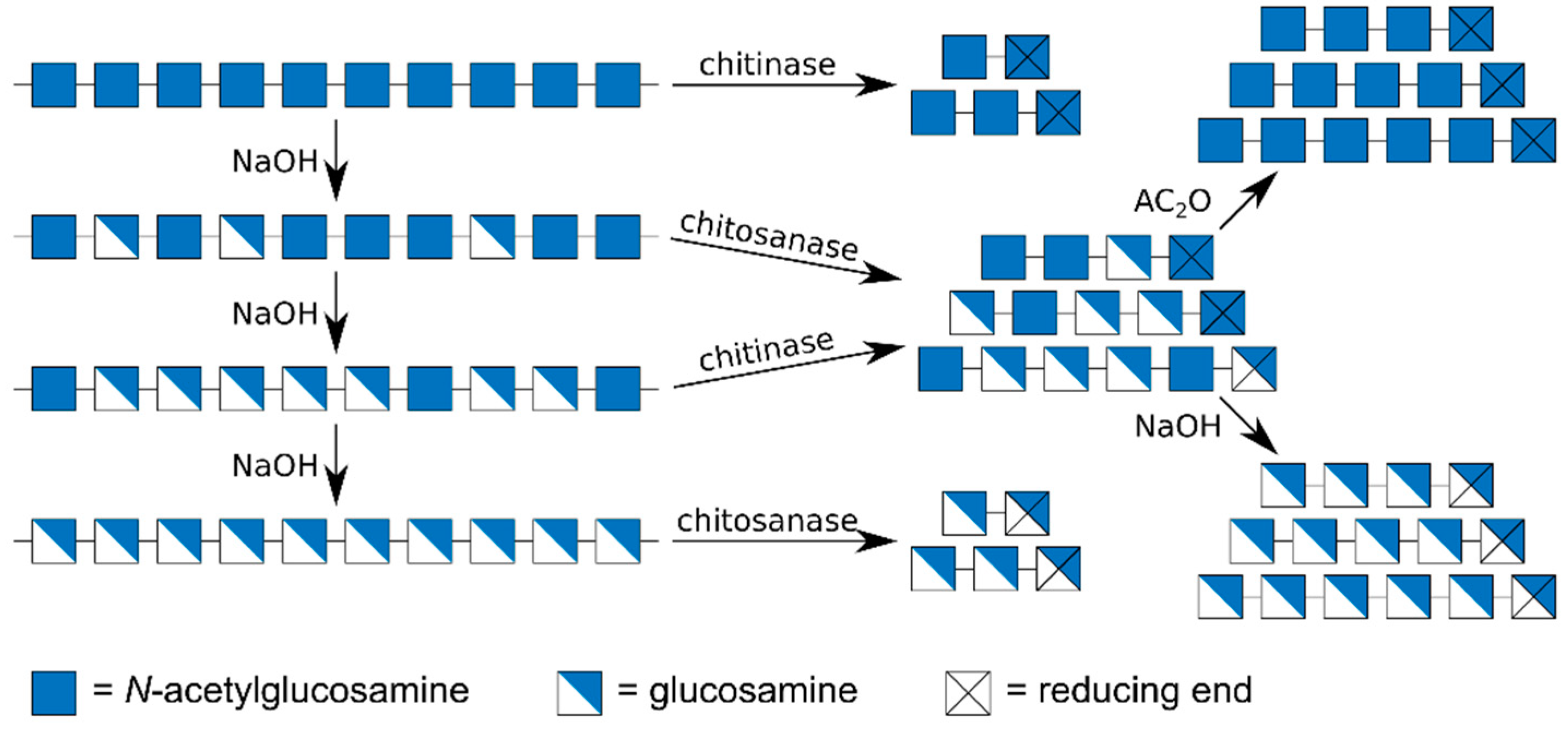
3. Preparation of the Substrates (An and Dn)
4. paCOS Nomenclature
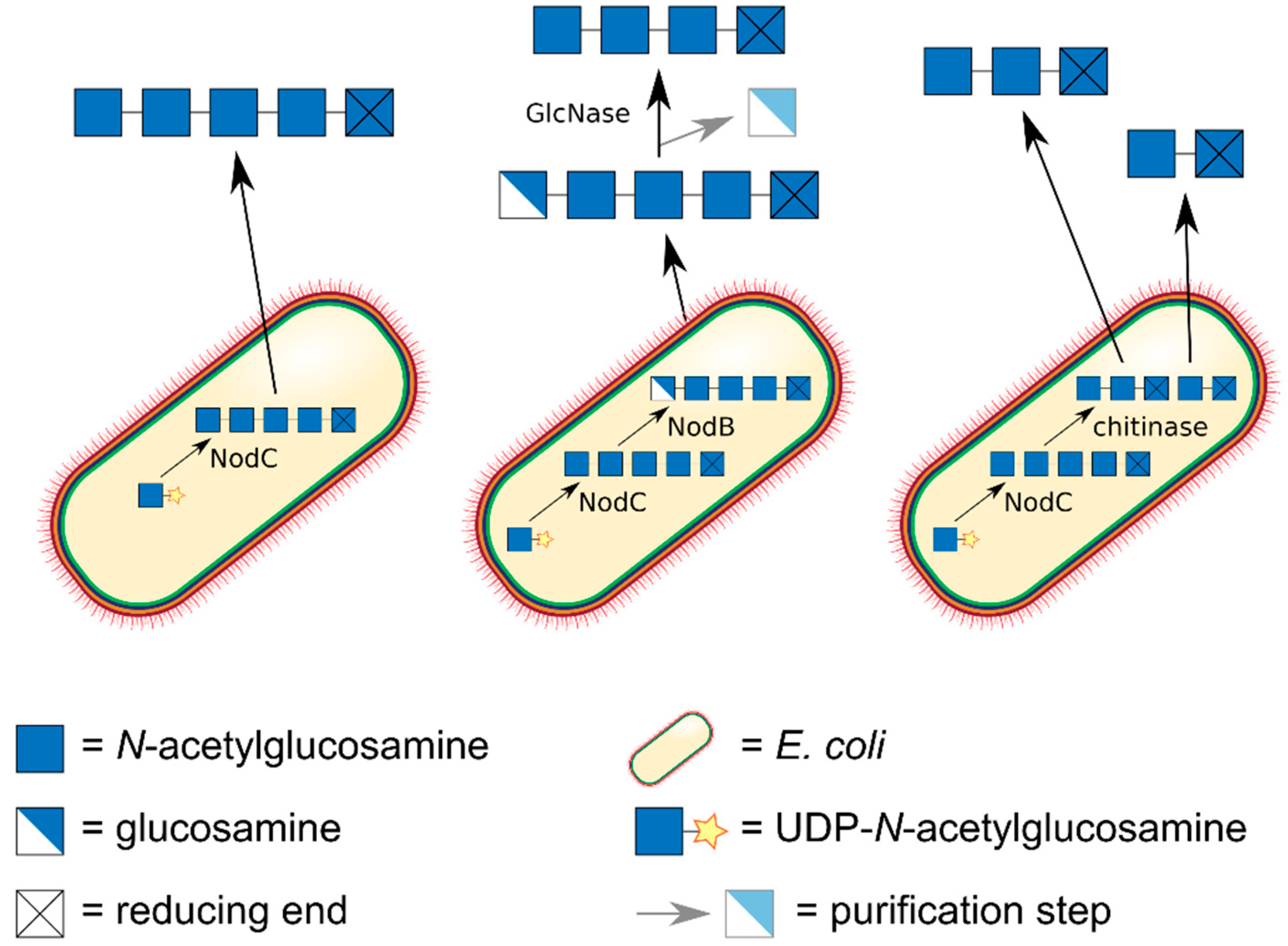
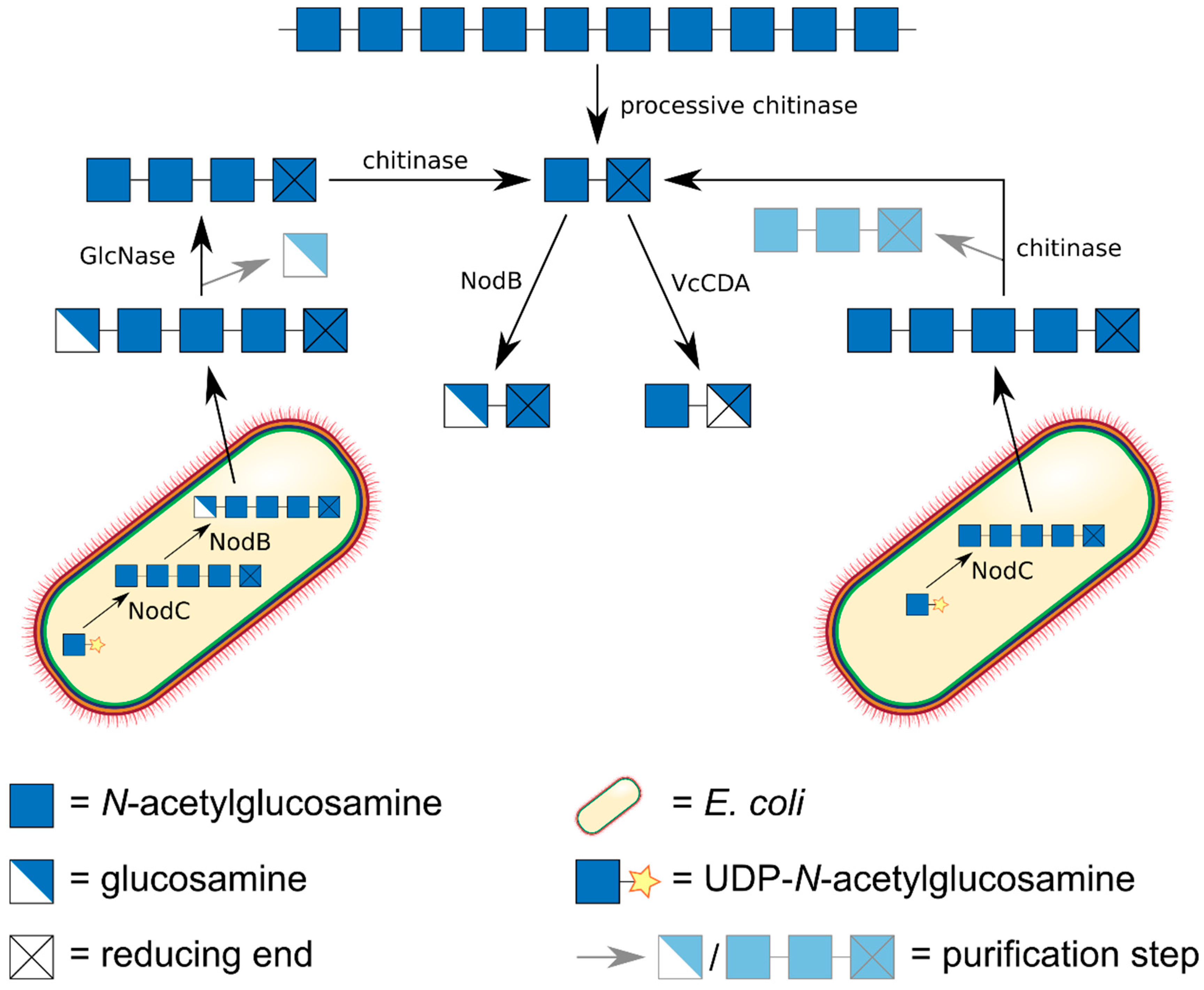
5. Preparation of paCOS Dimers
6. Preparation of paCOS Trimers
7. Preparation of paCOS Tetramers
8. Preparation of paCOS Pentamers and Hexamers
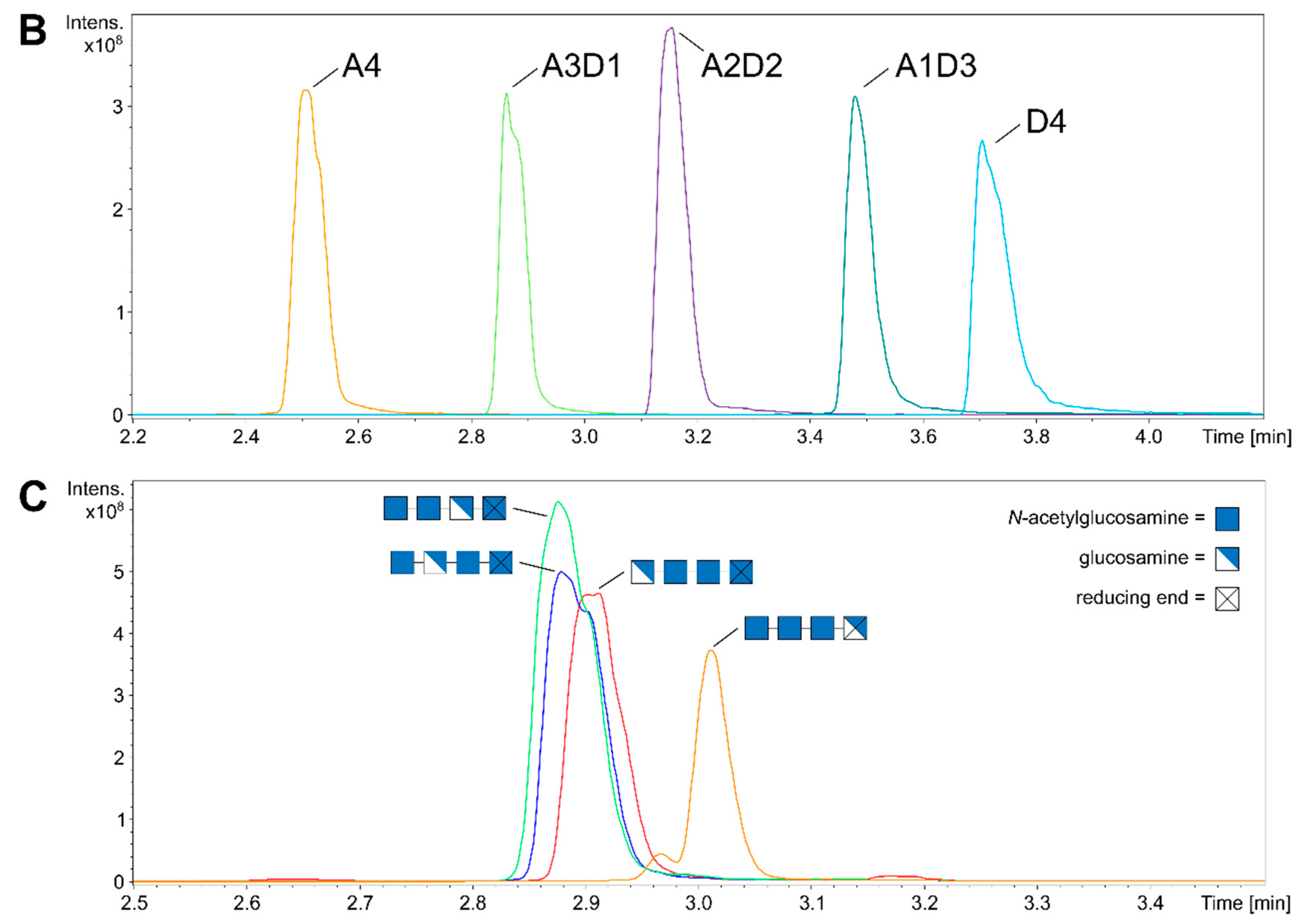
9. Mass Spectrometric Analysis of paCOS
10. Chromatographic Purification of paCOS
11. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| DP | Degree of Polymerisation |
| FA | Fraction of Acetylation |
| PA | Pattern of Acetylation |
| paCOS | partially acetylated Chitosan Oligosaccharides |
| CDA | Chitin Deacetylase |
References
- El Gueddari, N.E.; Moerschbacher, B.M. A bioactivity matrix for chitosans as elicitors of disease resistance reactions in wheat. Adv. Chitin Sci. 2004, VII, 56–59. [Google Scholar]
- El Gueddari, N.E.; Kolkenbrock, S.; Schaaf, A.; Chilukoti, N.; Brunel, F.; Gorzelanny, C.; Fehser, S.; Chachra, S.; Bernard, F.; Nampally, M.; et al. Chitin and chitosan modifying enzymes: Versatile novel tools for the analysis of structure-function relationship of partially acetylaed chitosans. Adv. Chitin Sci. 2014, XIV, 40–47. [Google Scholar]
- Kauss, H.; Jeblick, W.; Domard, A. The degrees of polymerization and N-acetylation of chitosan determine its ability to elicit callose formation in suspension cells and protoplasts of Catharanthus roseus. Planta 1989, 178, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Vander, P.; Vårum, K.M.; Domard, A.; El Gueddari, N.E.; Moerschbacher, B.M. Comparison of the Ability of Partially N-Acetylated Chitosans and Chitooligosaccharides to Elicit Resistance Reactions in Wheat Leaves. Plant Physiol. 1998, 118, 1353–1359. [Google Scholar] [CrossRef] [Green Version]
- Moerschbacher, B.M. Bio-activity matrices of chitosans in plant protection. In Emerging Trends in Plant-Microbe Interactions; Gnanamanickam, S.S., Balasubramanian, R., Anand, N., Eds.; University of Madras: Chennai, India, 2005; pp. 186–190. [Google Scholar]
- Wattjes, J.; Sreekumar, S.; Richter, C.; Cord-Landwehr, S.; Singh, R.; El Gueddari, N.E.; Moerschbacher, B.M. Patterns matter part 1: Chitosan polymers with non-random patterns of acetylation. React. Funct. Polym. 2020, 151, 104583. [Google Scholar] [CrossRef]
- Thadathil, N.; Suresh, P. Recent developments in chitosanase research and its biotechnological applications: A review. Food Chem. 2014, 150, 392–399. [Google Scholar] [CrossRef]
- Arnold, N.D.; Brück, W.M.; Garbe, D.; Brück, T.B. Enzymatic Modification of Native Chitin and Conversion to Specialty Chemical Products. Mar. Drugs 2020, 18, 93. [Google Scholar] [CrossRef] [Green Version]
- Júnior, E.N.; El Gueddari, N.E.; Moerschbacher, B.M.; Franco, T.T. Growth rate inhibition of phytopathogenic fungi by characterized chitosans. Braz. J. Microbiol. 2012, 43, 800–809. [Google Scholar] [CrossRef] [Green Version]
- Lacombe-Harvey, M.È.; Fukamizo, T.; Gagnon, J.; Ghinet, M.G.; Dennhart, N.; Letzel, T.; Brzezinski, R. Accessory active site residues of Streptomyces sp. N174 chitosanase. FEBS J. 2009, 276, 857–869. [Google Scholar] [CrossRef]
- Ghinet, M.G.; Roy, S.; Poulin-Laprade, D.; Lacombe-Harvey, M.È.; Morosoli, R.; Brzezinski, R. Chitosanase from Streptomyces coelicolorA3(2): Biochemical properties and role in protection against antibacterial effect of chitosan. Biochem. Cell Biol. 2010, 88, 907–916. [Google Scholar] [CrossRef]
- Miya, A.; Albert, P.; Shinya, T.; Desaki, Y.; Ichimura, K.; Shirasu, K.; Narusaka, Y.; Kawakami, N.; Kaku, H.; Shibuya, N. CERK1, a LysM receptor kinase, is essential for chitin elicitor signaling in Arabidopsis. Proc. Natl. Acad. Sci. USA 2007, 104, 19613–19618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaku, H.; Nishizawa, Y.; Ishii-Minami, N.; Akimoto-Tomiyama, C.; Dohmae, N.; Takio, K.; Minami, E.; Shibuya, N. Plant cells recognize chitin fragments for defense signaling through a plasma membrane receptor. Proc. Natl. Acad. Sci. USA 2006, 103, 11086–11091. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gubaeva, E.; Gubaev, A.; Melcher, R.L.J.; Cord-Landwehr, S.; Singh, R.; El Gueddari, N.E.; Moerschbacher, B.M. Slipped Sandwich Model for Chitin and Chitosan Perception in Arabidopsis. Mol. Plant-Microbe Interact. 2018, 31, 1145–1153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuchs, K.; Gloria, Y.C.; Wolz, O.; Herster, F.; Sharma, L.; Dillen, C.A.; Täumer, C.; Dickhöfer, S.; Bittner, Z.; Dang, T.; et al. The fungal ligand chitin directly binds TLR 2 and triggers inflammation dependent on oligomer size. EMBO Rep. 2018, 19, e201846065. [Google Scholar] [CrossRef]
- Sørbotten, A.; Horn, S.J.; Eijsink, V.G.H.; Vårum, K.M. Degradation of chitosans with chitinase B from Serratia marcescens. FEBS J. 2004, 272, 538–549. [Google Scholar] [CrossRef]
- Weikert, T.; Niehues, A.; Cord-Landwehr, S.; Hellmann, M.J.; Moerschbacher, B.M. Reassessment of chitosanase substrate specificities and classification. Nat. Commun. 2017, 8, 1–11. [Google Scholar] [CrossRef]
- Cord-Landwehr, S.; Richter, C.; Wattjes, J.; Sreekumar, S.; Singh, R.; Basa, S.; El Gueddari, N.E.; Moerschbacher, B.M. Patterns matter part 2: Chitosan oligomers with defined patterns of acetylation. React. Funct. Polym. 2020, 151, 104577. [Google Scholar] [CrossRef]
- Aragunde, H.; Biarnés, X.; Planas, A. Substrate Recognition and Specificity of Chitin Deacetylases and Related Family 4 Carbohydrate Esterases. Int. J. Mol. Sci. 2018, 19, 412. [Google Scholar] [CrossRef] [Green Version]
- Grifoll-Romero, L.; Pascual, S.; Aragunde, H.; Biarnés, X.; Planas, A. Chitin Deacetylases: Structures, Specificities, and Biotech Applications. Polymers 2018, 10, 352. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Park, R.-D.; Muzzarelli, R.A.A. Chitin Deacetylases: Properties and Applications. Mar. Drugs 2010, 8, 24–46. [Google Scholar] [CrossRef]
- Tsigos, I.; Martinou, A.; Kafetzopoulos, D.; Bouriotis, V. Chitin deacetylases: New, versatile tools in biotechnology. Trends Biotechnol. 2000, 18, 305–312. [Google Scholar] [CrossRef]
- Horn, S.J.; Sorbotten, A.; Synstad, B.; Sikorski, P.; Sorlie, M.; Varum, K.M.; Eijsink, V.G.H. Endo/exo mechanism and processivity of family 18 chitinases produced by Serratia marcescens. FEBS J. 2006, 273, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Fukamizo, T.; Ohkawa, T.; Ikeda, Y.; Goto, S. Specificity of chitosanase from Bacillus pumilus. Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzym. 1994, 1205, 183–188. [Google Scholar] [CrossRef]
- Hirano, K.; Watanabe, M.; Seki, K.; Ando, A.; Saito, A.; Mitsutomi, M. Classification of Chitosanases by Hydrolytic Specificity toward N1, N4-Diacetylchitohexaose. Biosci. Biotechnol. Biochem. 2012, 76, 1932–1937. [Google Scholar] [CrossRef] [Green Version]
- Tokuyasu, K.; Mitsutomi, M.; Yamaguchi, I.; Hayashi, K.; Mori, Y. Recognition of Chitooligosaccharides and TheirN-Acetyl Groups by Putative Subsites of Chitin Deacetylase from a Deuteromycete, Colletotrichum lindemuthianum. Biochemistry 2000, 39, 8837–8843. [Google Scholar] [CrossRef]
- Hembach, L.; Bonin, M.; Gorzelanny, C.; Moerschbacher, B.M. Unique subsite specificity and potential natural function of a chitosan deacetylase from the human pathogen Cryptococcus neoformans. Proc. Natl. Acad. Sci. USA 2020, 117, 3551–3559. [Google Scholar] [CrossRef]
- Blair, D.E.; Hekmat, O.; Schüttelkopf, A.W.; Shrestha, B.; Tokuyasu, K.; Withers, S.G.; Van Aalten, D.M.F. Structure and Mechanism of Chitin Deacetylase from the Fungal Pathogen Colletotrichum lindemuthianum. Biochemistry 2006, 45, 9416–9426. [Google Scholar] [CrossRef] [Green Version]
- Andrés, E.; Albesa-Jové, D.; Biarnés, X.; Moerschbacher, B.M.; Guerin, M.E.; Planas, A. Structural Basis of Chitin Oligosaccharide Deacetylation. Angew. Chem. Int. Ed. 2014, 53, 6882–6887. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, Y.; Qu, M.; Qiu, Y.; Guo, X.; Zhang, Y.; Liu, T.; Yang, J.; Yang, Q. Structural and biochemical insights into the catalytic mechanisms of two insect chitin deacetylases of the carbohydrate esterase 4 family. J. Biol. Chem. 2019, 294, 5774–5783. [Google Scholar] [CrossRef] [Green Version]
- Naqvi, S.; Cord-Landwehr, S.; Singh, R.; Bernard, F.; Kolkenbrock, S.; El Gueddari, N.E.; Moerschbacher, B.M. A Recombinant Fungal Chitin Deacetylase Produces Fully Defined Chitosan Oligomers with Novel Patterns of Acetylation. Appl. Environ. Microbiol. 2016, 82, 6645–6655. [Google Scholar] [CrossRef] [Green Version]
- Cord-Landwehr, S.; Ihmor, P.; Niehues, A.; Luftmann, H.; Moerschbacher, B.M.; Mormann, M. Quantitative Mass-Spectrometric Sequencing of Chitosan Oligomers Revealing Cleavage Sites of Chitosan Hydrolases. Anal. Chem. 2017, 89, 2893–2900. [Google Scholar] [CrossRef] [PubMed]
- Tokuyasu, K.; Ono, H.; Hayashi, K.; Mori, Y. Reverse hydrolysis reaction of chitin deacetylase and enzymatic synthesis of β-d-GlcNAc-(1→4)-GlcN from chitobiose. Carbohydr. Res. 1999, 322, 26–31. [Google Scholar] [CrossRef]
- Hembach, L.; Cord-Landwehr, S.; Moerschbacher, B.M. Enzymatic production of all fourteen partially acetylated chitosan tetramers using different chitin deacetylases acting in forward or reverse mode. Sci. Rep. 2017, 7, 17692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- John, M.; Rohrig, H.; Schmidt, J.; Wieneke, U.; Schell, J. Rhizobium NodB protein involved in nodulation signal synthesis is a chitooligosaccharide deacetylase. Proc. Natl. Acad. Sci. USA 1993, 90, 625–629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Wang, L.-X.; Wang, X.; Roseman, S. The Chitin Catabolic Cascade in the Marine Bacterium Vibrio Cholerae: Characterization of a Unique Chitin Oligosaccharide Deacetylase. Glycobiology 2007, 17, 1377–1387. [Google Scholar] [CrossRef]
- Cord-Landwehr, S.; Melcher, R.L.J.; Kolkenbrock, S.; Moerschbacher, S.; Moerschbacher, B.M. A chitin deacetylase from the endophytic fungus Pestalotiopsis sp. efficiently inactivates the elicitor activity of chitin oligomers in rice cells. Sci. Rep. 2016, 6, 38018. [Google Scholar] [CrossRef]
- Aranda-Martinez, A.; Grifoll-Romero, L.; Aragunde, H.; Sancho-Vaello, E.; Biarnés, X.; Lopez-Llorca, L.V.; Planas, A. Expression and specificity of a chitin deacetylase from the nematophagous fungus Pochonia chlamydosporia potentially involved in pathogenicity. Sci. Rep. 2018, 8, 2170. [Google Scholar] [CrossRef]
- Grifoll-Romero, L.; Sainz-Polo, M.A.; Albesa-Jové, D.; Guerin, M.E.; Biarnés, X.; Planas, A. Structure-function relationships underlying the dual N-acetylmuramic and N-acetylglucosamine specificities of the bacterial peptidoglycan deacetylase PdaC. J. Biol. Chem. 2019, 294, 19066–19080. [Google Scholar] [CrossRef]
- No, H.K.; Meyers, S.P. Preperation of chitin and chitosan. In Chitin Handbook; Muzzarelli, R.A.A., Peter, M.G., Eds.; Euopean Chitin Society: Grottammare, Italy, 1997; pp. 475–489. ISBN 88-86889-01-1. [Google Scholar]
- Roberts, G.A.F. Chitosan production routes and their role in determining the structure and properties of the product. Adv. Chitin Sci. 1998, II, 22–31. [Google Scholar]
- Varum, K. Acid hydrolysis of chitosans. Carbohydr. Polym. 2001, 46, 89–98. [Google Scholar] [CrossRef]
- Einbu, A.; Vårum, K.M. Depolymerization and De-N-acetylation of Chitin Oligomers in Hydrochloric Acid. Biomacromolecules 2007, 8, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Einbu, A.; Grasdalen, H.; Vårum, K.M. Kinetics of hydrolysis of chitin/chitosan oligomers in concentrated hydrochloric acid. Carbohydr. Res. 2007, 342, 1055–1062. [Google Scholar] [CrossRef]
- Vaaje-Kolstad, G.; Horn, S.J.; Sørlie, M.; Eijsink, V.G.H. The chitinolytic machinery of Serratia marcescens—A model system for enzymatic degradation of recalcitrant polysaccharides. FEBS J. 2013, 280, 3028–3049. [Google Scholar] [CrossRef] [PubMed]
- Kidibule, P.E.; Santos-Moriano, P.; Jiménez-Ortega, E.; Ramírez-Escudero, M.; Limón, M.C.; Remacha, M.; Plou, F.J.; Sanz-Aparicio, J.; Fernández-Lobato, M. Use of chitin and chitosan to produce new chitooligosaccharides by chitinase Chit42: Enzymatic activity and structural basis of protein specificity. Microb. Cell Factories 2018, 17, 47. [Google Scholar] [CrossRef] [PubMed]
- Kumirska, J.; Weinhold, M.X.; Steudte, S.; Thöming, J.; Brzozowski, K.; Stepnowski, P. Determination of the pattern of acetylation of chitosan samples: Comparison of evaluation methods and some validation parameters. Int. J. Biol. Macromol. 2009, 45, 56–60. [Google Scholar] [CrossRef]
- Vårum, K.M.; Anthonsen, M.W.; Grasdalen, H.; Smidsrød, O. 13C-N.m.r. studies of the acetylation sequences in partially N-deacetylated chitins (chitosans). Carbohydr. Res. 1991, 217, 19–27. [Google Scholar] [CrossRef]
- Cord-Landwehr, S.; Niehues, A.; Wattjes, J.; Moerschbacher, B.M. New developments in the analysis of partially acetylated chitosan polymers and oligomers. In Chitin and Chitosan; van den Broek, L.A.M., Boeriu, C.G., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2019; pp. 81–95. ISBN 9781119450467. [Google Scholar]
- Van Aalten, D.M.F.; Komander, D.; Synstad, B.; Gaseidnes, S.; Peter, M.G.; Eijsink, V.G.H. Structural insights into the catalytic mechanism of a family 18 exo-chitinase. Proc. Natl. Acad. Sci. USA 2001, 98, 8979–8984. [Google Scholar] [CrossRef] [Green Version]
- Ohno, T.; Armand, S.; Hata, T.; Nikaidou, N.; Henrissat, B.; Mitsutomi, M.; Watanabe, T. A modular family 19 chitinase found in the prokaryotic organism Streptomyces griseus HUT 6037. J. Bacteriol. 1996, 178, 5065–5070. [Google Scholar] [CrossRef] [Green Version]
- Fukamizo, T.; Koga, D.; Goto, S. Comparative Biochemistry of Chitinases—Anomeric Form of the Reaction Products. Biosci. Biotechnol. Biochem. 1995, 59, 311–313. [Google Scholar] [CrossRef]
- Sasaki, C.; Vårum, K.M.; Itoh, Y.; Tamoi, M.; Fukamizo, T. Rice chitinases: Sugar recognition specificities of the individual subsites. Glycobiology 2006, 16, 1242–1250. [Google Scholar] [CrossRef] [Green Version]
- Regel, E.K.; Weikert, T.; Niehues, A.; Moerschbacher, B.M.; Singh, R. Protein-engineering of chitosanase from Bacillus sp. MN to alter its substrate specificity. Biotechnol. Bioeng. 2018, 115, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Bußwinkel, F.; Goñi, O.; Cord-Landwehr, S.; O’Connell, S.; Moerschbacher, B.M. Endochitinase 1 (Tv-ECH1) from Trichoderma virens has high subsite specificities for acetylated units when acting on chitosans. Int. J. Biol. Macromol. 2018, 114, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Aam, B.B.; Heggset, E.B.; Norberg, A.L.; Sørlie, M.; Vårum, K.M.; Eijsink, V.G.H. Production of Chitooligosaccharides and Their Potential Applications in Medicine. Mar. Drugs 2010, 8, 1482–1517. [Google Scholar] [CrossRef] [Green Version]
- Naqvi, S.; Moerschbacher, B.M. The cell factory approach toward biotechnological production of high-value chitosan oligomers and their derivatives: An update. Crit. Rev. Biotechnol. 2015, 37, 11–25. [Google Scholar] [CrossRef]
- Geremia, R.A.; Mergaert, P.; Geelen, D.; Van Montagu, M.; Holsters, M. The NodC protein of Azorhizobium caulinodans is an N-acetylglucosaminyltransferase. Proc. Natl. Acad. Sci. USA 1994, 91, 2669–2673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samain, E.; Drouillard, S.; Heyraud, A.; Driguez, H.; Geremia, R.A. Gram-scale synthesis of recombinant chitooligosaccharides in Escherichia coli. Carbohydr. Res. 1997, 302, 35–42. [Google Scholar] [CrossRef]
- Cottaz, S.; Samain, E. Genetic engineering of Escherichia coli for the production of NI,NII-diacetylchitobiose (chitinbiose) and its utilization as a primer for the synthesis of complex carbohydrates. Metab. Eng. 2005, 7, 311–317. [Google Scholar] [CrossRef]
- Tanaka, T.; Fukui, T.; Atomi, H.; Imanaka, T. Characterization of an Exo-β-d-Glucosaminidase Involved in a Novel Chitinolytic Pathway from the Hyperthermophilic Archaeon Thermococcus kodakaraensis KOD1. J. Bacteriol. 2003, 185, 5175–5181. [Google Scholar] [CrossRef] [Green Version]
- Hamer, S.N.; Moerschbacher, B.M.; Kolkenbrock, S. (Stephan) Enzymatic sequencing of partially acetylated chitosan oligomers. Carbohydr. Res. 2014, 392, 16–20. [Google Scholar] [CrossRef]
- Ajandouz, E.H.; Tchiakpe, L.; Ore, F.D.; Benajiba, A.; Puigserver, A. Effects of pH on Caramelization and Maillard Reaction Kinetics in Fructose-Lysine Model Systems. J. Food Sci. 2001, 66, 926–931. [Google Scholar] [CrossRef]
- Chung, Y.C.; Tsai, C.-F.; Li, C.F. Preparation and characterization of water-soluble chitosan produced by Maillard reaction. Fish. Sci. 2006, 72, 1096–1103. [Google Scholar] [CrossRef]
- Waghmare, S.R.; Ghosh, J.S. Chitobiose production by using a novel thermostable chitinase from Bacillus licheniformis strain JS isolated from a mushroom bed. Carbohydr. Res. 2010, 345, 2630–2635. [Google Scholar] [CrossRef] [PubMed]
- Kadokura, K.; Sakamoto, Y.; Saito, K.; Ikegami, T.; Hirano, T.; Hakamata, W.; Oku, T.; Nishio, T. Production of a recombinant chitin oligosaccharide deacetylase from Vibrio parahaemolyticus in the culture medium of Escherichia coli cells. Biotechnol. Lett. 2007, 29, 1209–1215. [Google Scholar] [CrossRef] [PubMed]
- Kurakake, M.; Yo-U, S.; Nakagawa, K.; Sugihara, M.; Komaki, T. Properties of chitosanase from Bacillus cereus S1. Curr. Microbiol. 2000, 40, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Jung, W.; Park, R.-D. Bioproduction of Chitooligosaccharides: Present and Perspectives. Mar. Drugs 2014, 12, 5328–5356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamer, S.N.; Cord-Landwehr, S.; Biarnés, X.; Planas, A.; Waegeman, H.; Moerschbacher, B.; Kolkenbrock, S. Enzymatic production of defined chitosan oligomers with a specific pattern of acetylation using a combination of chitin oligosaccharide deacetylases. Sci. Rep. 2015, 5, 8716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gercke, D.; Regel, E.; Singh, R.; Moerschbacher, B.M. Rational protein design of Bacillus sp. MN chitosanase for altered substrate binding and production of specific chitosan oligomers. J. Biol. Eng. 2019, 13, 23. [Google Scholar] [CrossRef]
- Pascual, S.; Planas, A. Screening Assay for Directed Evolution of Chitin Deacetylases: Application to Vibrio cholerae Deacetylase Mutant Libraries for Engineered Specificity. Anal. Chem. 2018, 90, 10654–10658. [Google Scholar] [CrossRef] [Green Version]
- Haebel, S.; Bahrke, A.S.; Peter†, M.G. Quantitative Sequencing of Complex Mixtures of Heterochitooligosaccharides by vMALDI-Linear Ion Trap Mass Spectrometry. Anal. Chem. 2007, 79, 5557–5566. [Google Scholar] [CrossRef]
- Tang, M.-C.; Nisole, A.; Dupont, C.; Pelletier, J.N.; Waldron, K.C. Chemical profiling of the deacetylase activity of acetyl xylan esterase A (AxeA) variants on chitooligosaccharides using hydrophilic interaction chromatography–mass spectrometry. J. Biotechnol. 2011, 155, 257–265. [Google Scholar] [CrossRef]
- Domon, B.; Costello, C.E. A systematic nomenclature for carbohydrate fragmentations in FAB-MS/MS spectra of glycoconjugates. Glycoconj. J. 1988, 5, 397–409. [Google Scholar] [CrossRef]
- Lopatin, S.; Ilyin, M.; Pustobaev, V.; Bezchetnikova, Z.; Varlamov, V.; Davankov, V. Mass-Spectrometric Analysis of N-Acetylchitooligosaccharides Prepared through Enzymatic Hydrolysis of Chitosan. Anal. Biochem. 1995, 227, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Li, X.J.; Piao, X.S.; Kim, S.W.; Liu, P.; Wang, L.; Shen, Y.B.; Jung, S.C.; Lee, H.S. Effects of Chito-Oligosaccharide Supplementation on Performance, Nutrient Digestibility, and Serum Composition in Broiler Chickens. Poult. Sci. 2007, 86, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Sashiwa, H.; Fujishima, S.; Yamano, N.; Kawasaki, N.; Nakayama, A.; Muraki, E.; Sukwattanasinitt, M.; Pichyangkura, R.; Aiba, S.-I. Enzymatic production of N-acetyl-d-glucosamine from chitin. Degradation study of N-acetylchitooligosaccharide and the effect of mixing of crude enzymes. Carbohydr. Polym. 2003, 51, 391–395. [Google Scholar] [CrossRef]
- Li, K.; Liu, S.; Xing, R.; Yu, H.; Qin, Y.; Li, R.; Li, P. High-resolution separation of homogeneous chitooligomers series from 2-mers to 7-mers by ion-exchange chromatography. J. Sep. Sci. 2013, 36, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Basa, S.; Nampally, M.; Honorato, T.; Das, S.N.; Podile, A.R.; El Gueddari, N.E.; Moerschbacher, B.M. The Pattern of Acetylation Defines the Priming Activity of Chitosan Tetramers. J. Am. Chem. Soc. 2020, 142, 1975–1986. [Google Scholar] [CrossRef] [PubMed]
- DelBianco, M.; Kononov, A.; Poveda, A.; Yu, Y.; Diercks, T.; Jiménez-Barbero, J.; Seeberger, P.H. Well-Defined Oligo- and Polysaccharides as Ideal Probes for Structural Studies. J. Am. Chem. Soc. 2018, 140, 5421–5426. [Google Scholar] [CrossRef] [PubMed]
- Darvill, A.G.; Albersheim, P. Phytoalexins and their Elicitors—A Defense Against Microbial Infection in Plants. Annu. Rev. Plant Physiol. 1984, 35, 243–275. [Google Scholar] [CrossRef]
- Ossowski, P.; Garegg, P.J.; Lindberg, B. Syntheses of a Branched Hepta and an Octassaccharide with Phytoalexin-Elicitor Activity. Angew. Chem. Int. Ed. 1983, 22, 793–794. [Google Scholar] [CrossRef]
- Aly, M.R.; Ibrahim, E.-S.I.; El Ashry, E.S.H.; Schmidt, R.R. Synthesis of chitotetraose and chitohexaose based on dimethylmaleoyl protection. Carbohydr. Res. 2001, 331, 129–142. [Google Scholar] [CrossRef]
- Kuyama, H.; Nukada, T.; Ito, Y.; Nakahara, Y.; Ogawa, T.; Nakahara, Y.; Nakahara, Y. Stereocontrolled synthesis of chitosan dodecamer. Carbohydr. Res. 1993, 243, C1–C7. [Google Scholar] [CrossRef]
- Slámová, K.; Krejzová, J.; Marhol, P.; Kalachova, L.; Kulik, N.; Pelantová, H.; Cvačka, J.; Křen, V. Synthesis of Derivatized Chitooligomers using Transglycosidases Engineered from the Fungal GH20 β-N-Acetylhexosaminidase. Adv. Synth. Catal. 2015, 357, 1941–1950. [Google Scholar] [CrossRef]
- Kobayashi, S.; Kiyosada, A.T.; Shoda, S.-I. Synthesis of Artificial Chitin: Irreversible Catalytic Behavior of a Glycosyl Hydrolase through a Transition State Analogue Substrate. J. Am. Chem. Soc. 1996, 118, 13113–13114. [Google Scholar] [CrossRef]
- Honda, Y.; Fushinobu, S.; Hidaka, M.; Wakagi, T.; Tsuruta, T.; Taniguchi, H.; Kitaoka, M. Alternative strategy for converting an inverting glycoside hydrolase into a glycosynthase. Glycobiology 2008, 18, 325–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alsina, C.; Faijes, M.; Planas, A. Glycosynthase-type GH18 mutant chitinases at the assisting catalytic residue for polymerization of chitooligosaccharides. Carbohydr. Res. 2019, 478, 1–9. [Google Scholar] [CrossRef]


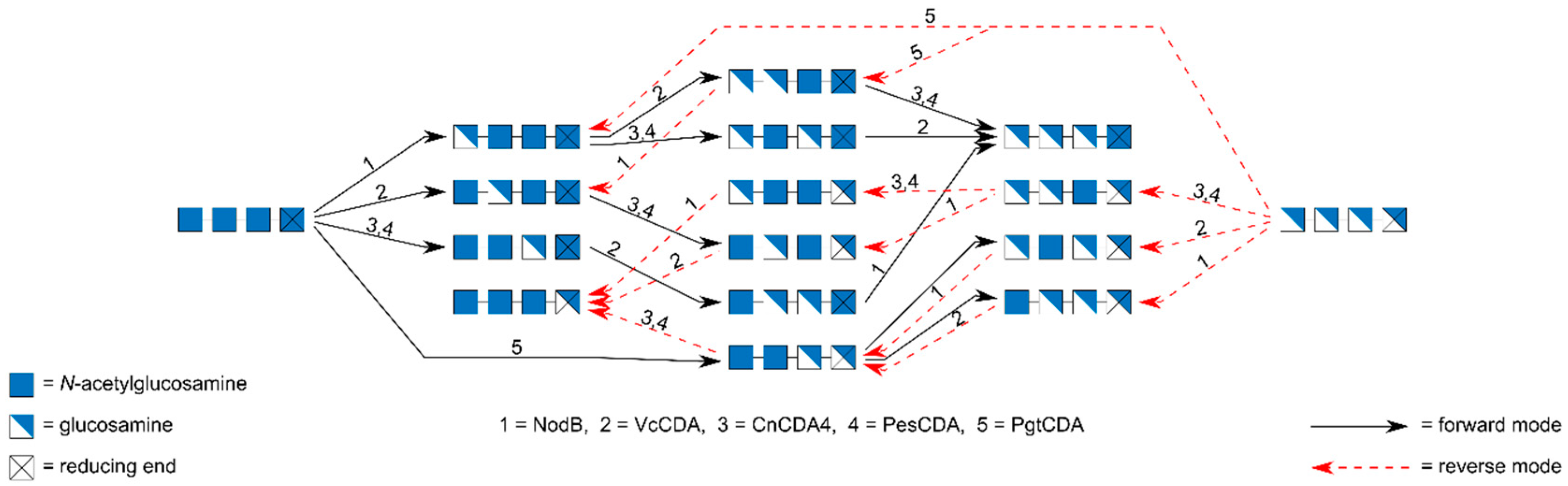
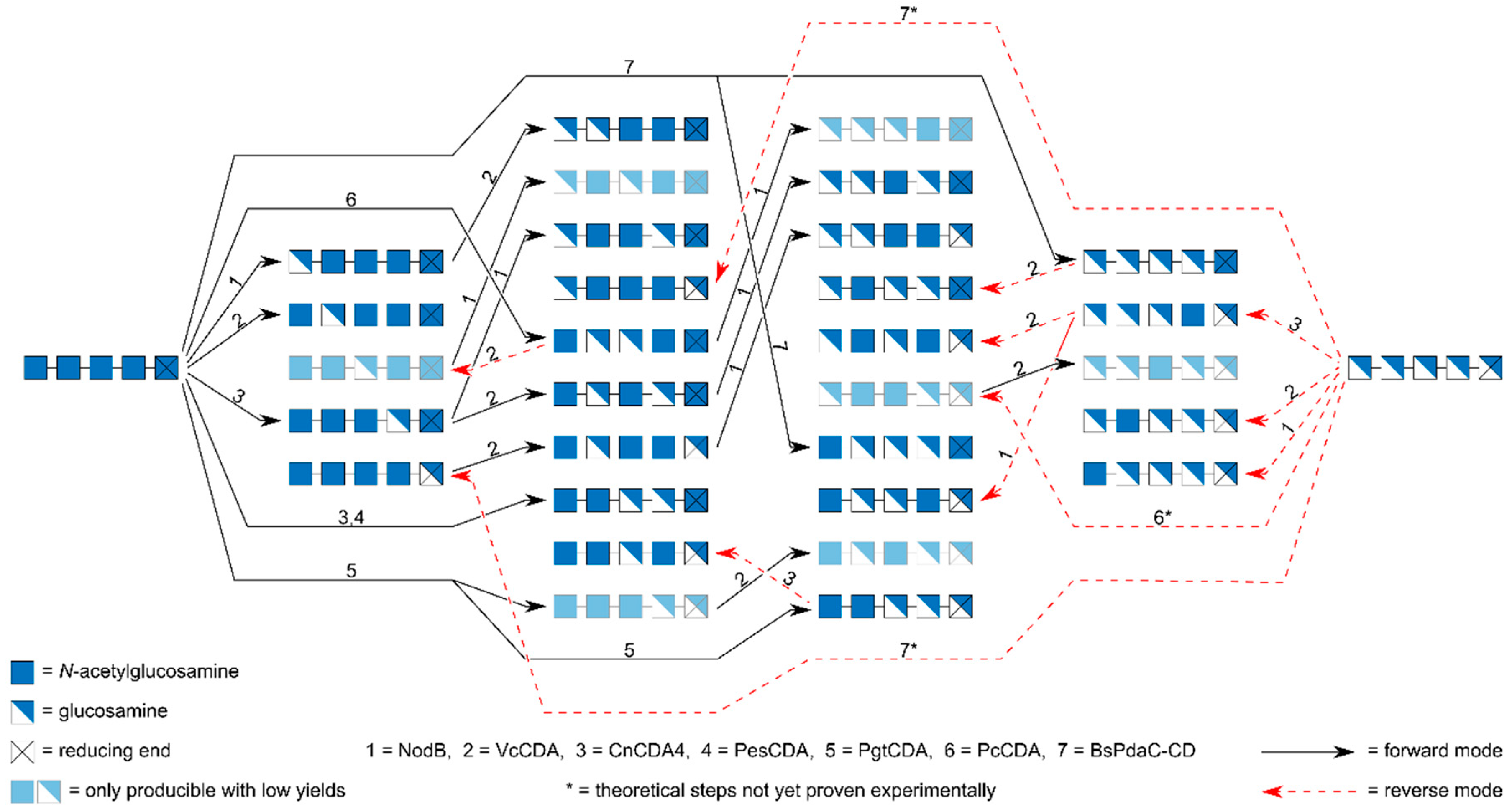
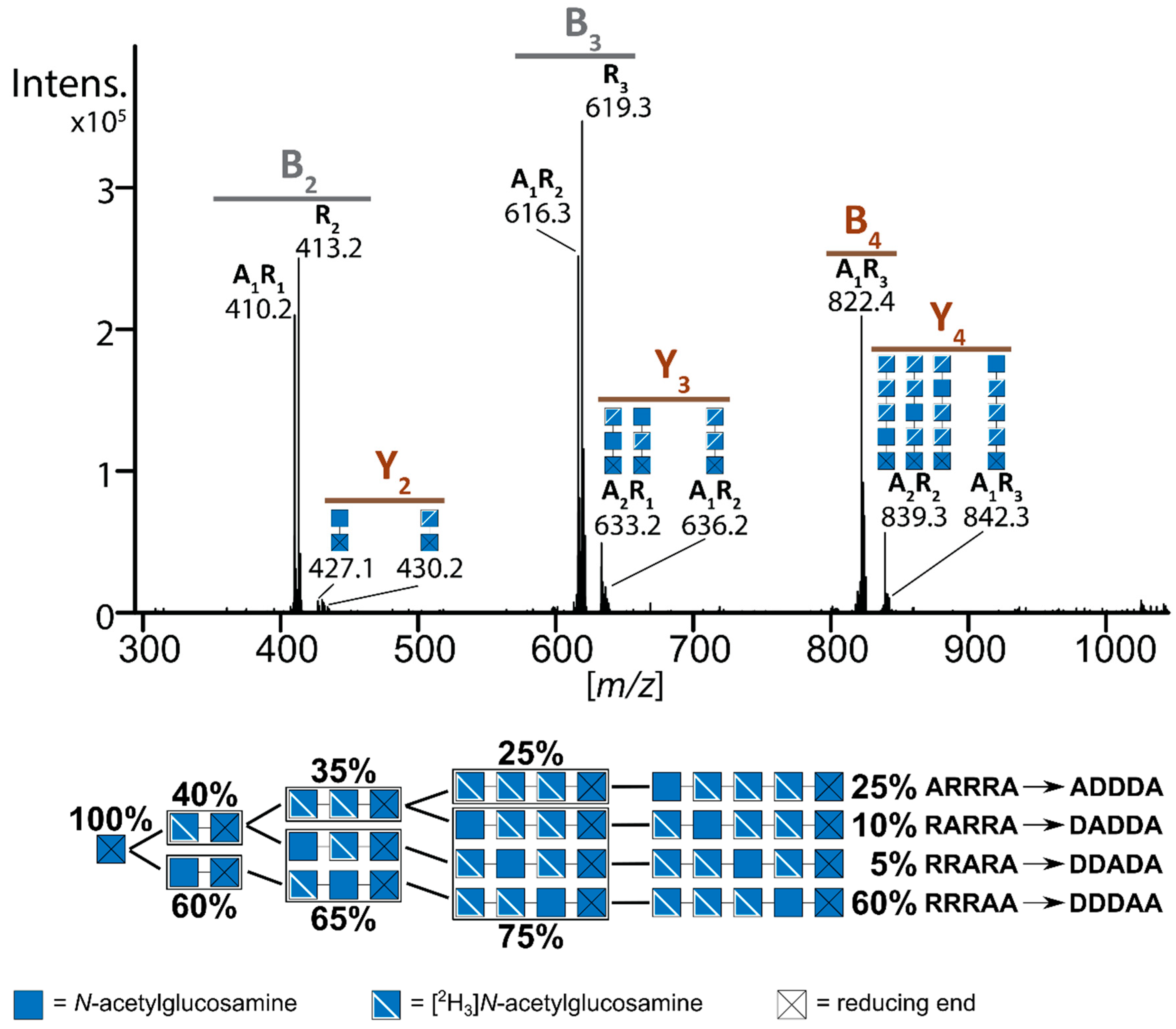
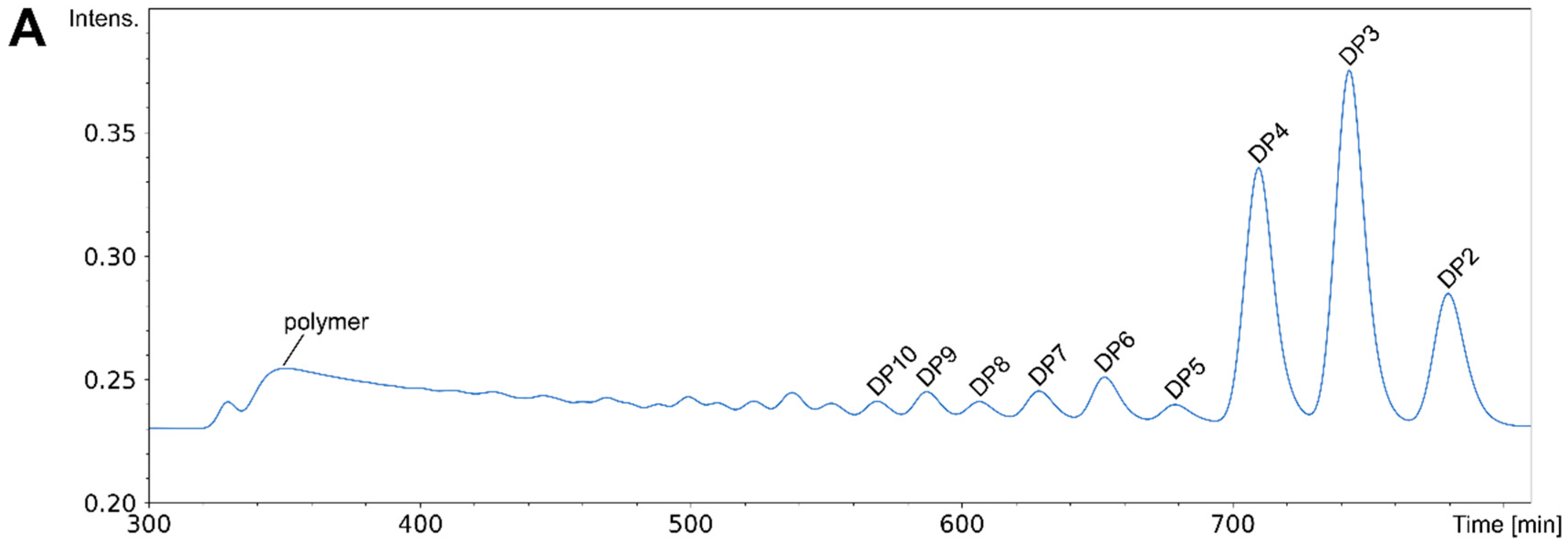
| Enzyme | Step | Substrate | Reference | ||||
|---|---|---|---|---|---|---|---|
| AA | AAA | AAAA | AAAAA | AAAAAA | |||
| NodB | 1 | DA | DAA | DAAA | DAAAA | DAAAAA | [35] |
| VcCDA | 1 | AD | ADA | ADAA | ADAAA | ADAAAA | [36] |
| CnCDA4 | 1 | - | (ADA) | AADA | AAADA | AAAADA | [27] |
| 2 | - | AADD | AADDA | AAADDA | |||
| 3 | - | - | AADDDA | ||||
| PesCDA | 1 | - | - | AADA | AA[AD]A | AA[A2D1]A | [37] |
| 2 | - | AADDA | AA[A1D2]A | ||||
| 3 | - | AADDDA | |||||
| PgtCDA | 1 | p.n.d. | p.n.d. | AADA | AAADA (AADAA) | AAADAA (AADAAA) | [31] |
| 2 | AADD | AADDA (AAADD) | AAADDA (AADDAA) | ||||
| 3 | - | AADDD | AADDDA (AAADDD) | ||||
| 4 | - | AADDDD | |||||
| PcCDA | 1 | a.n.d. | - | A3D1 | ADAAA | a.n.d. | [38] |
| 2 | A2D2 | ADDAA | |||||
| BsPdaC-CD | 1 | - | p.n.d. | ADAA AADA | AAADA (AADAA) | a.n.d. | [39] |
| 2 | ADDA (DADA) | AADDA (ADADA) | |||||
| 3 | DDDA | ADDDA | |||||
| 4 | - | DDDDA | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonin, M.; Sreekumar, S.; Cord-Landwehr, S.; Moerschbacher, B.M. Preparation of Defined Chitosan Oligosaccharides Using Chitin Deacetylases. Int. J. Mol. Sci. 2020, 21, 7835. https://doi.org/10.3390/ijms21217835
Bonin M, Sreekumar S, Cord-Landwehr S, Moerschbacher BM. Preparation of Defined Chitosan Oligosaccharides Using Chitin Deacetylases. International Journal of Molecular Sciences. 2020; 21(21):7835. https://doi.org/10.3390/ijms21217835
Chicago/Turabian StyleBonin, Martin, Sruthi Sreekumar, Stefan Cord-Landwehr, and Bruno M. Moerschbacher. 2020. "Preparation of Defined Chitosan Oligosaccharides Using Chitin Deacetylases" International Journal of Molecular Sciences 21, no. 21: 7835. https://doi.org/10.3390/ijms21217835
APA StyleBonin, M., Sreekumar, S., Cord-Landwehr, S., & Moerschbacher, B. M. (2020). Preparation of Defined Chitosan Oligosaccharides Using Chitin Deacetylases. International Journal of Molecular Sciences, 21(21), 7835. https://doi.org/10.3390/ijms21217835





