Potential Role of Lycopene in the Prevention of Postmenopausal Bone Loss: Evidence from Molecular to Clinical Studies
Abstract
1. Introduction
2. Bone Biology, Modelling, and Remodeling
3. Postmenopausal Osteoporosis: A Silent Disease
3.1. Risk Factors of Postmenopausal Osteoporosis
3.2. Pathophysiology of Postmenopausal Osteoporosis
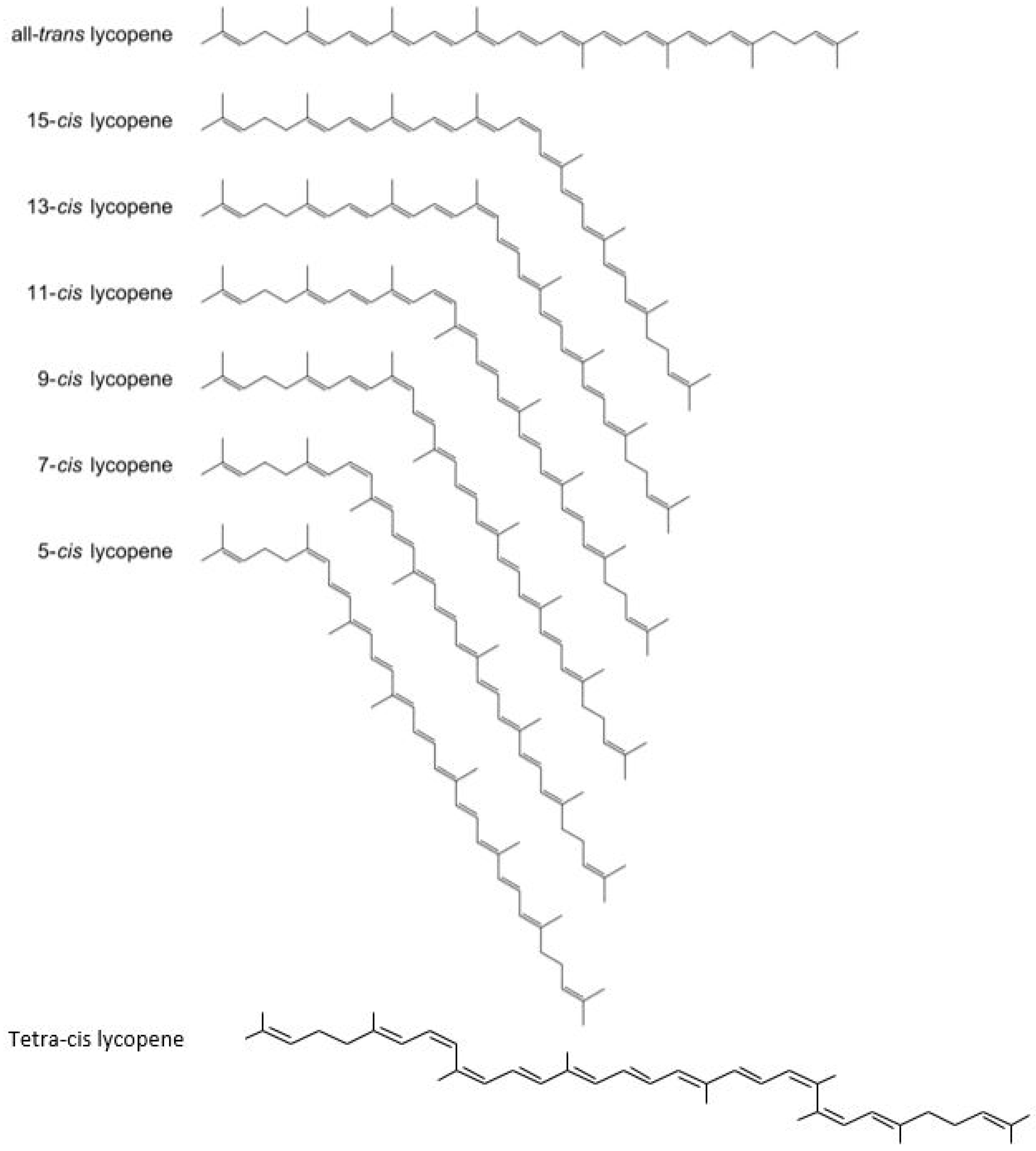
4. Carotenoid Lycopene: Chemistry and its Isomers
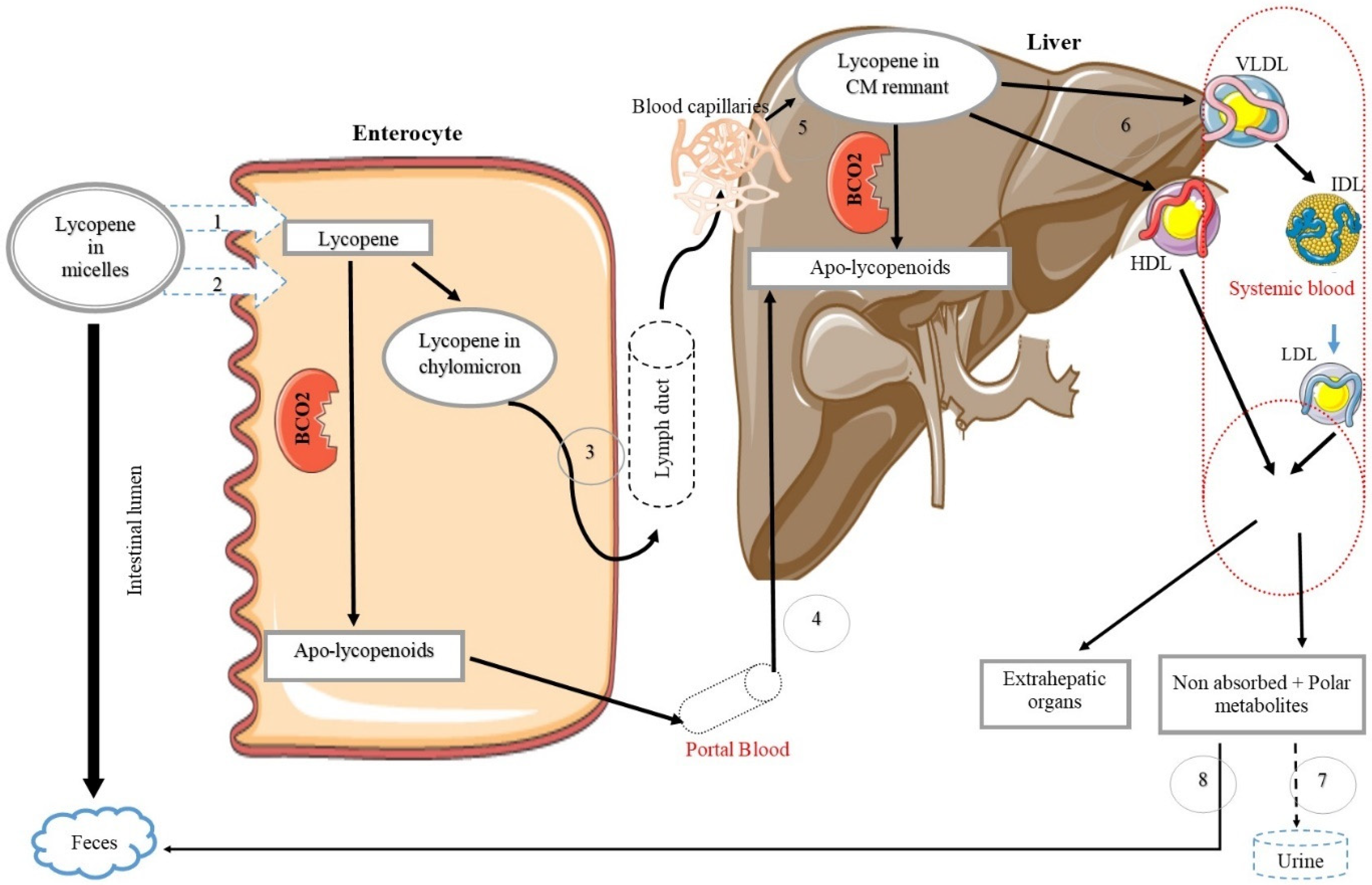
Lycopene Bioavailability, Absorption, and Metabolism
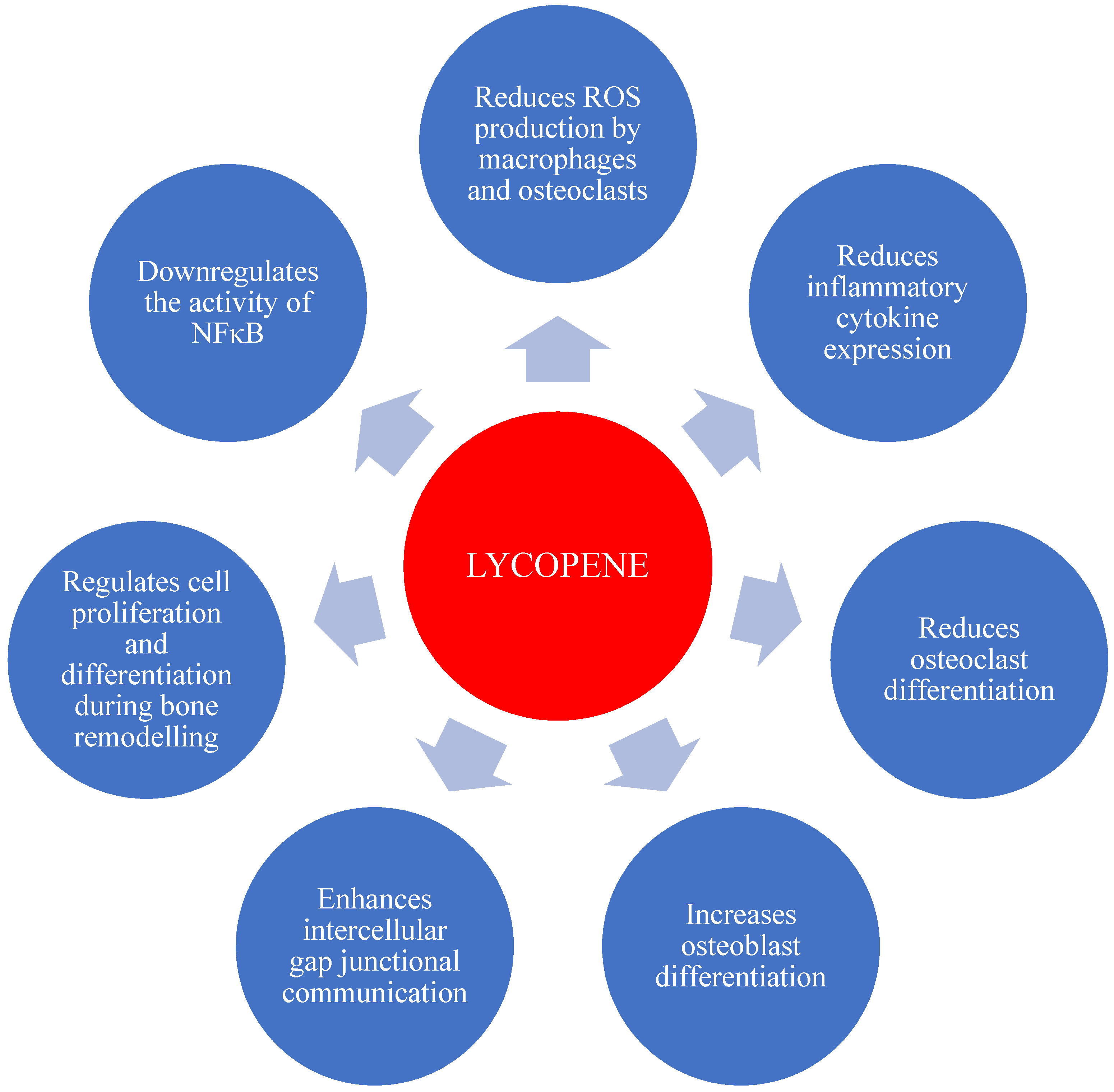
5. Evidence of the Effect of Lycopene on Bone Health
5.1. Epidemiological and Clinical Studies
5.2. Animal Trials
5.3. Bone Cell Culture Studies
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CKD | Chronic kidney disease |
| BMU | Basic multicellular unit |
| BMD | Bone mineral density |
| BMC | Bone mineral content |
| SD | Standard deviation |
| DEXA | Dual-energy X-ray absorptiometry |
| MRI | Magnetic resonance imaging |
| ROS | Reactive oxygen species |
| OH | Hydroxyl |
| O2− | Superoxide |
| H2O2 | Hydrogen peroxides |
| TNF | Tumor necrosis factor |
| OPG | Osteoprotegerin |
| M-CSF | Macrophage colony-stimulating factor |
| RANK | Receptor Activator of Nuclear Factor Kappa B |
| RANKL | Receptor Activator of Nuclear Factor Kappa B Ligand |
| IL | Interleukin |
| IGF | Insulin-like growth factor |
| IFN | Interferon |
| MHC | Major histocompatibility complex |
| PGE | Prostaglandin E2 |
| PTH | Parathyroid hormone |
| BCO2 | Β-carotene-9′,10′-oxygenase |
| VLDL | Very low-density lipoprotein |
| HDL | High-density lipoprotein |
| LDL | Low-density lipoprotein |
| IDL | Intermediate density lipoprotein |
| CM | Chylomicron |
| NTx | N-telopeptide of type 1 bone biomarker |
| SOD | Superoxide dismutase |
| OVX | Ovariectomy |
| CTX-1 | Carboxy terminal crosslinked telopeptides of type 1 collagen |
| DPD | Deoxypyridinoline |
| PINP | Procollagen type 1 N-terminal propeptide |
| NFκB | Nuclear factor kappa B |
| COL1A | Collagen type 1 alpha |
| CYP19A1 | Aromatase genes |
| ERα | Estrogen receptor alpha genes |
References
- Cooper, C.; Campion, G.; Melton, L.J., III. Hip fractures in the elderly: A world-wide projection. Osteoporos. Int. 1992, 2, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Szulc, P.; Bouxsein, M.L. Overview of osteoporosis: Epidemiology and clinical management. Vertebr. Fract. Initiat. Resour. Doc. 2011, 1, 1–65. [Google Scholar]
- Wright, N.C.; Looker, A.C.; Saag, K.G.; Curtis, J.R.; Delzell, E.S.; Randall, S.; Dawson-Hughes, B. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J. Bone Miner. Res. 2014, 29, 2520–2526. [Google Scholar] [CrossRef]
- Cauley, J.A.; Chalhoub, D.; Kassem, A.M.; Fuleihan, G.E.-H. Geographic and ethnic disparities in osteoporotic fractures. Nat. Rev. Endocrinol. 2014, 10, 338. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.K.; Gupta, S.C.; Karelia, D.; Gilhooley, P.J.; Shakibaei, M.; Aggarwal, B.B. Dietary nutraceuticals as backbone for bone health. Biotechnol. Adv. 2018, 36, 1633–1648. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Rao, A. Oxidative stress and antioxidants in the risk of osteoporosis—Role of phytochemical antioxidants lycopene and polyphenol-containing nutritional supplements. In Phytochemicals—Isolation, Characterisation and Role in Human Health; Venketeshwer Rao, L.R., Ed.; InTech Open Access Publisher: Rijeka, Croatia, 2015; pp. 247–260. [Google Scholar]
- International Osteoporosis Foundation. Facts and Statistics. Available online: https://www.iofbonehealth.org/facts-statistics (accessed on 5 May 2020).
- Kini, U.; Nandeesh, B. Physiology of bone formation, remodeling, and metabolism. In Radionuclide and Hybrid Bone Imaging; Springer: Berlin/Heidelberg, Germany, 2012; pp. 29–57. [Google Scholar]
- Duque, G.; Troen, B.R. Understanding the mechanisms of senile osteoporosis: New facts for a major geriatric syndrome. J. Am. Geriatr. Soc. 2008, 56, 935–941. [Google Scholar] [CrossRef]
- Sipos, W.; Pietschmann, P.; Rauner, M.; Kerschan-Schindl, K.; Patsch, J. Pathophysiology of osteoporosis. Wien. Med. Wochenschr. 2009, 159, 230–234. [Google Scholar] [CrossRef]
- Sugiura, M.; Nakamura, M.; Ogawa, K.; Ikoma, Y.; Ando, F.; Shimokata, H.; Yano, M. Dietary patterns of antioxidant vitamin and carotenoid intake associated with bone mineral density: Findings from post-menopausal Japanese female subjects. Osteoporos. Int. 2011, 22, 143–152. [Google Scholar] [CrossRef]
- Wattanapenpaiboon, N.; Lukito, W.; Wahlqvist, M.L.; Strauss, B.J. Dietary carotenoid intake as a predictor of bone mineral density. Asia Pac. J. Clin. Nutr. 2003, 12, 467–473. [Google Scholar]
- Iimura, Y.; Agata, U.; Takeda, S.; Kobayashi, Y.; Yoshida, S.; Ezawa, I.; Omi, N. Lycopene intake facilitates the increase of bone mineral density in growing female rats. J. Nutr. Sci. Vitaminol. 2014, 60, 101–107. [Google Scholar] [CrossRef]
- Iimura, Y.; Agata, U.; Takeda, S.; Kobayashi, Y.; Yoshida, S.; Ezawa, I.; Omi, N. The protective effect of lycopene intake on bone loss in ovariectomized rats. J. Bone Miner. Metab. 2015, 33, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Burri, B.J.; Chapman, M.H.; Neidlinger, T.R.; Seo, J.S.; Ishida, B.K. Tangerine tomatoes increase total and tetra-cis-lycopene isomer concentrations more than red tomatoes in healthy adult humans. Int. J. Food Sci. Nutr. 2009, 60, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Cooperstone, J.L.; Ralston, R.A.; Riedl, K.M.; Haufe, T.C.; Schweiggert, R.M.; King, S.A.; Timmers, C.D.; Francis, D.M.; Lesinski, G.B.; Clinton, S.K. Enhanced bioavailability of lycopene when consumed as cis-isomers from tangerine compared to red tomato juice, a randomized, cross-over clinical trial. Mol. Nutr. Food Res. 2015, 59, 658–669. [Google Scholar] [CrossRef] [PubMed]
- Ishida, B.; Roberts, J.; Chapman, M.; Burri, B. Processing tangerine tomatoes: Effects on lycopene-isomer concentrations and profile. J. Food Sci. 2007, 72, 307–312. [Google Scholar] [CrossRef]
- Shi, J.; Maguer, M.L. Lycopene in tomatoes: Chemical and physical properties affected by food processing. Crit. Rev. Food Sci. Nutr. 2000, 40, 1–42. [Google Scholar] [CrossRef]
- Rao, A.; Waseem, Z.; Agarwal, S. Lycopene content of tomatoes and tomato products and their contribution to dietary lycopene. Food Res. Int. 1998, 31, 737–741. [Google Scholar] [CrossRef]
- Cooperstone, J.L.; Francis, D.M.; Schwartz, S.J. Thermal processing differentially affects lycopene and other carotenoids in cis-lycopene containing, tangerine tomatoes. Food Chem. 2016, 210, 466–472. [Google Scholar] [CrossRef]
- Unlu, N.Z.; Bohn, T.; Francis, D.; Clinton, S.K.; Schwartz, S.J. Carotenoid absorption in humans consuming tomato sauces obtained from tangerine or high-β-carotene varieties of tomatoes. J. Agric. Food Chem. 2007, 55, 1597–1603. [Google Scholar] [CrossRef]
- Morgan, K.T. Nutritional determinants of bone health. J. Nutr. Elder. 2008, 27, 3–27. [Google Scholar] [CrossRef]
- Bartl, R.; Frisch, B. Osteoporosis: Diagnosis, Prevention, Therapy, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 7–22. [Google Scholar]
- Florencio-Silva, R.; Sasso, G.R.d.S.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of bone tissue: Structure, function, and factors that influence bone cells. Biomed Res. Int. 2015, 2015, 421746. [Google Scholar] [CrossRef]
- Burr, D.B.; Akkus, O. Chapter 1—Bone Morphology and Organization. In Basic and Applied Bone Biology; Burr, D.B., Allen, M.R., Eds.; Academic Press: San Diego, CA, USA, 2014; pp. 3–25. [Google Scholar]
- Alves, R. Osteoblast Differentiation and Bone: Relevant proteins, regulatory processes and the vascular connection. Ph.D. Thesis, Erasmus University, Rotterdam, The Netherlands, 2012. [Google Scholar]
- McCormick, R.K. Osteoporosis: Integrating biomarkers and other diagnostic correlates into the management of bone fragility. Altern. Med. Rev. 2007, 12, 113. [Google Scholar] [PubMed]
- Jilka, R.L.; Weinstein, R.S.; Bellido, T.; Parfitt, A.M.; Manolagas, S.C. Osteoblast programmed cell death (apoptosis): Modulation by growth factors and cytokines. J. Bone Miner. Res. 1998, 13, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, M.-N.; Offord, E. Naturally plant-derived compounds: Role in bone anabolism. Curr. Mol. Pharmacol. 2012, 5, 205–218. [Google Scholar] [CrossRef]
- Clarke, B. Normal bone anatomy and physiology. Clin. J. Am. Soc. Nephrol. 2008, 3, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Iñiguez-Ariza, N.M.; Clarke, B.L. Bone biology, signaling pathways, and therapeutic targets for osteoporosis. Maturitas 2015, 82, 245–255. [Google Scholar] [CrossRef]
- Langdahl, B.; Ferrari, S.; Dempster, D.W. Bone modeling and remodeling: Potential as therapeutic targets for the treatment of osteoporosis. Ther. Adv. Musculoskelet. Dis. 2016, 8, 225–235. [Google Scholar] [CrossRef]
- Baron, R.; Hesse, E. Update on bone anabolics in osteoporosis treatment: Rationale, current status, and perspectives. J. Clin. Endocrinol. Metab. 2012, 97, 311–325. [Google Scholar] [CrossRef]
- Rao, L.G.; Krishnadev, N.; Banasikowska, K.; Rao, A.V. Lycopene 1—Effect on osteoclasts: Lycopene inhibits basal and parathyroid hormone-stimulated osteoclast formation and mineral resorption mediated by reactive oxygen species in rat bone marrow cultures. J. Med. Food 2003, 6, 69–78. [Google Scholar] [CrossRef]
- Dempster, D.W.; Raisz, L.G. Bone physiology: Bone cells, modeling, and remodeling. In Nutrition and Bone Health; Holick, M., Nieves, J., Eds.; Humana Press: New York, NY, USA, 2015; pp. 37–56. [Google Scholar]
- Manolagas, S.C. Birth and death of bone cells: Basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr. Rev. 2000, 21, 115–137. [Google Scholar]
- Hadjidakis, D.J.; Androulakis, I.I. Bone remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef]
- Shetty, S.; Kapoor, N.; Bondu, J.D.; Thomas, N.; Paul, T.V. Bone turnover markers: Emerging tool in the management of osteoporosis. Indian J. Endocrinol. Metab. 2016, 20, 846. [Google Scholar] [PubMed]
- Siddiqui, J.A.; Partridge, N.C. Physiological bone remodeling: Systemic regulation and growth factor involvement. Physiology 2016, 31, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Rao, L.; Rao, A. Oxidative stress and antioxidants in the risk of osteoporosis—Role of the antioxidants lycopene and polyphenols. In Topics in Osteoporosis; Valdés-Flores, M., Ed.; InTech Open Access Publisher: Rijeka, Croatia, 2013; Volume 5, pp. 117–144. [Google Scholar]
- Ferdous, H.; Afsana, F.; Qureshi, N.K.; Rouf, R.S.B. Osteoporosis: A Review. BIRDEM Med. J. 2016, 5, 30–36. [Google Scholar] [CrossRef]
- Kruger, M.C.; Morel, P.C. Experimental control for the ovariectomized rat model: Use of sham versus nonmanipulated animal. J. Appl. Anim. Welf. Sci. 2016, 19, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Annapoorna, N.; Rao, G.V.; Reddy, N.; Rambabu, P.; Rao, K.S. An increased risk of osteoporosis during acquired immunodeficiency syndrome. Int. J. Med. Sci. 2004, 1, 152. [Google Scholar] [CrossRef] [PubMed]
- Siris, E.; Boonen, S.; Mitchell, P.; Bilezikian, J.; Silverman, S. What’s in a name? What constitutes the clinical diagnosis of osteoporosis? Osteoporos. Int. 2012, 23, 2093–2097. [Google Scholar] [CrossRef]
- The National Institute of Health. Bone Mass Measurement: What the Numbers Mean. Available online: https://www.bones.nih.gov/health-info/bone/bone-health/bone-mass-measure (accessed on 5 May 2020).
- Adler, R.A. Update on osteoporosis in men. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 759–772. [Google Scholar] [CrossRef]
- Feng, X.; McDonald, J.M. Disorders of bone remodeling. Annu. Rev. Pathol. 2011, 6, 121–145. [Google Scholar] [CrossRef]
- Nieves, J.W.; Cosman, F. Impact of Nutrition on Medications for Osteoporosis. In Nutrition and Bone Health; Springer: Berlin/Heidelberg, Germany, 2015; pp. 681–686. [Google Scholar]
- International Osteoporosis Foundation. Treatment. Available online: https://www.osteoporosis.foundation/health-professionals/treatment (accessed on 2 August 2020).
- Ferrari, S.; Karasik, D. Gene–Diet Interactions on Bone. In Nutrition and Bone Health; Holick, M., Nieves, J., Eds.; Humana Press: New York, NY, USA, 2015; pp. 21–36. [Google Scholar]
- Ralston, S.H. Osteoporosis as an hereditary disease. Clin. Rev. Bone Miner. Metab. 2010, 8, 68–76. [Google Scholar] [CrossRef]
- Spotila, L.D.; Constantinou, C.D.; Sereda, L.; Ganguly, A.; Riggs, B.L.; Prockop, D.J. Mutation in a gene for type I procollagen (COL1A2) in a woman with postmenopausal osteoporosis: Evidence for phenotypic and genotypic overlap with mild osteogenesis imperfecta. Proc. Natl. Acad. Sci. USA 1991, 88, 5423–5427. [Google Scholar] [CrossRef]
- Prockop, D.J.; Colige, A.; Helminen, H.; Khillan, J.; Pereira, R.; Vandenberg, P. Mutations in type 1 procollagen that cause osteogenesis imperfecta: Effects of the mutations on the assembly of collagen into fibrils, the basis of phenotypic variations, and potential antisense therapies. J. Bone Miner. Res. 1993, 8, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Marini, J.C.; Forlino, A.; Cabral, W.A.; Barnes, A.M.; San Antonio, J.D.; Milgrom, S.; Hyland, J.C.; Körkkö, J.; Prockop, D.J.; De Paepe, A. Consortium for osteogenesis imperfecta mutations in the helical domain of type I collagen: Regions rich in lethal mutations align with collagen binding sites for integrins and proteoglycans. Hum. Mutat. 2007, 28, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Morishima, A.; Grumbach, M.M.; Simpson, E.R.; Fisher, C.; Qin, K. Aromatase deficiency in male and female siblings caused by a novel mutation and the physiological role of estrogens. J. Clin. Endocrinol. Metab. 1995, 80, 3689–3698. [Google Scholar] [PubMed]
- Smith, E.P.; Boyd, J.; Frank, G.R.; Takahashi, H.; Cohen, R.M.; Specker, B.; Williams, T.C.; Lubahn, D.B.; Korach, K.S. Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. N. Engl. J. Med. 1994, 331, 1056–1061. [Google Scholar] [CrossRef]
- Sullivan, S.D.; Lehman, A.; Nathan, N.K.; Thomson, C.A.; Howard, B.V. Age of Menopause and Fracture Risk in Post-Menopausal Women Randomized to Calcium+ Vitamin D, Hormone Therapy, or the combination: Results from the Women’s Health Initiative Clinical Trials. Menopause 2017, 24, 371. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Heaney, R.P.; Holick, M.F.; Lips, P.; Meunier, P.J.; Vieth, R. Estimates of optimal vitamin D status. Osteoporos. Int. 2005, 16, 713–716. [Google Scholar] [CrossRef]
- Joint FAO/WHO Committee on Food Additives (JECFA). Evaluation of certain food additives and contaminants: Sixty-eighth report of the Joint FAO/WHO Expert Committee on Food Additives. In WHO Technical Repport Series; World Helath Organization: Geneva, Switserland, 2007; Volume 68. [Google Scholar]
- Melton, L.J., III; Achenbach, S.J.; Gebhart, J.B.; Babalola, E.O.; Atkinson, E.J.; Bharucha, A.E. Influence of hysterectomy on long-term fracture risk. Fertil. Steril. 2007, 88, 156–162. [Google Scholar] [CrossRef]
- Kanis, J.A.; Johansson, H.; Johnell, O.; Oden, A.; De Laet, C.; Eisman, J.A.; Pols, H.; Tenenhouse, A. Alcohol intake as a risk factor for fracture. Osteoporos. Int. 2005, 16, 737–742. [Google Scholar] [CrossRef]
- Fazeli, P.K.; Klibanski, A. Effects of anorexia nervosa on bone metabolism. Endocr. Rev. 2018, 39, 895–910. [Google Scholar] [CrossRef]
- Kanis, J.A.; Johnell, O.; Odén, A.; Johansson, H.; De Laet, C.; Eisman, J.; Fujiwara, S.; Kroger, H.; McCloskey, E.; Mellstrom, D. Smoking and fracture risk: A meta-analysis. Osteoporos. Int. 2005, 16, 155–162. [Google Scholar] [CrossRef]
- Kanis, J.; Johansson, H.; Odén, A.; Johnell, O.; De Laet, C.; Eisman, J.; McCloskey, E.; Mellstrom, D.; Melton III, L.; Pols, H. A family history of fracture and fracture risk: A meta-analysis. Bone 2004, 35, 1029–1037. [Google Scholar] [CrossRef]
- Seeman, E.; Hopper, J.L.; Bach, L.A.; Cooper, M.E.; Parkinson, E.; McKay, J.; Jerums, G. Reduced bone mass in daughters of women with osteoporosis. N. Engl. J. Med. 1989, 320, 554–558. [Google Scholar] [CrossRef] [PubMed]
- De Laet, C.; Kanis, J.; Odén, A.; Johanson, H.; Johnell, O.; Delmas, P.; Eisman, J.; Kroger, H.; Fujiwara, S.; Garnero, P. Body mass index as a predictor of fracture risk: A meta-analysis. Osteoporos. Int. 2005, 16, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.; Johnell, O.; De Laet, C.; Johansson, H.; Odén, A.; Delmas, P.; Eisman, J.; Fujiwara, S.; Garnero, P.; Kroger, H. A meta-analysis of previous fracture and subsequent fracture risk. Bone 2004, 35, 375–382. [Google Scholar] [CrossRef]
- Misra, M.; Golden, N.H.; Katzman, D.K. State of the art systematic review of bone disease in anorexia nervosa. Int. J. Eat. Disord. 2016, 49, 276–292. [Google Scholar] [CrossRef]
- Moayyeri, A.; Luben, R.N.; Bingham, S.A.; Welch, A.A.; Wareham, N.J.; Khaw, K.T. Measured height loss predicts fractures in middle-aged and older men and women: The EPIC-Norfolk prospective population study. J. Bone Miner. Res. 2008, 23, 425–432. [Google Scholar] [CrossRef]
- Bass, S.L.; Saxon, L.; Daly, R.; Turner, C.H.; Robling, A.G.; Seeman, E.; Stuckey, S. The effect of mechanical loading on the size and shape of bone in pre-, peri-, and postpubertal girls: A study in tennis players. J. Bone Miner. Res. 2002, 17, 2274–2280. [Google Scholar] [CrossRef]
- Kemmler, W.; Lauber, D.; Weineck, J.; Hensen, J.; Kalender, W.; Engelke, K. Benefits of 2 years of intense exercise on bone density, physical fitness, and blood lipids in early postmenopausal osteopenic women: Results of the Erlangen Fitness Osteoporosis Prevention Study (EFOPS). Arch. Intern. Med. 2004, 164, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- International Osteoporosis Foundation. Fixed Risk. Available online: https://www.osteoporosis.foundation/health-professionals/about-osteoporosis/risk-factors/fixed-risks (accessed on 25 August 2020).
- Tinetti, M.E. Preventing falls in elderly persons. N. Engl. J. Med. 2003, 348, 42–49. [Google Scholar] [CrossRef]
- International Osteoporosis Foundation. About Osteoporosis. Available online: https://www.osteoporosis.foundation/health-professionals/about-osteoporosis (accessed on 24 July 2020).
- Becker, C. Pathophysiology and clinical manifestations of osteoporosis. Clin. Cornerstone 2006, 8, 19–27. [Google Scholar] [CrossRef]
- Jahanian, E.; Karimifar, M.; Rafieian-Kopaei, M. Antioxidants as a novel way to alleviate the adverse effects of oxidative stress in osteoporosis. J. Parathyr. Dis. 2016, 4, 61. [Google Scholar]
- Kim, N.; Choi, H.-R.; Kim, S.-W.; Kim, B.-S.; Won, C.-W.; Kim, S.-Y. Association between bone mineral density and sleep duration in the Korean elderly population. Korean J. Fam. Med. 2014, 35, 90. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, D.; Takahashi, O.; Deshpande, G.A.; Shimbo, T.; Fukui, T. Association between osteoporosis and sleep duration in healthy middle-aged and elderly adults: A large-scale, cross-sectional study in Japan. Sleep Breath 2012, 16, 579–583. [Google Scholar] [CrossRef]
- Hill, S.M.; Spriggs, L.L.; Simon, M.A.; Muraoka, H.; Blask, D.E. The growth inhibitory action of melatonin on human breast cancer cells is linked to the estrogen response system. Cancer Lett. 1992, 64, 249–256. [Google Scholar] [CrossRef]
- Ochs-Balcom, H.M.; Hovey, K.M.; Andrews, C.; Cauley, J.A.; Hale, L.; Li, W.; Bea, J.W.; Sarto, G.E.; Stefanick, M.L.; Stone, K.L. Short sleep is associated with low bone mineral density and osteoporosis in the Women’s Health Initiative. J. Bone Miner. Res. 2020, 35, 261–268. [Google Scholar] [CrossRef]
- Fu, X.; Zhao, X.; Lu, H.; Jiang, F.; Ma, X.; Zhu, S. Association between sleep duration and bone mineral density in Chinese women. Bone 2011, 49, 1062–1066. [Google Scholar] [CrossRef]
- Wang, K.; Wu, Y.; Yang, Y.; Chen, J.; Zhang, D.; Hu, Y.; Liu, Z.; Xu, J.; Shen, Q.; Zhang, N. The associations of bedtime, nocturnal, and daytime sleep duration with bone mineral density in pre-and post-menopausal women. Endocrine 2015, 49, 538–548. [Google Scholar] [CrossRef]
- Niu, J.; Sahni, S.; Liao, S.; Tucker, K.L.; Dawson-Hughes, B.; Gao, X. Association between sleep duration, insomnia symptoms and bone mineral density in older Boston Puerto Rican adults. PLoS ONE 2015, 10, e0132342. [Google Scholar] [CrossRef]
- Khosla, S.; Oursler, M.J.; Monroe, D.G. Estrogen and the skeleton. Trends Endocrinol. Metab. 2012, 23, 576–581. [Google Scholar] [CrossRef]
- Okman-Kilic, T. Estrogen deficiency and osteoporosis. In Advances in Osteoporosis; Dionyssiotis, Y., Ed.; InTech Open Access Publisher: Rijeka, Croatia, 2015; pp. 7–17. [Google Scholar]
- Weitzmann, M.N.; Pacifici, R. Estrogen deficiency and bone loss: An inflammatory tale. J. Clin. Investig. 2006, 116, 1186–1194. [Google Scholar] [CrossRef]
- Charatcharoenwitthaya, N.; Khosla, S.; Atkinson, E.J.; McCready, L.K.; Riggs, B.L. Effect of blockade of TNF-α and interleukin-1 action on bone resorption in early postmenopausal women. J. Bone Miner. Res. 2007, 22, 724–729. [Google Scholar] [CrossRef] [PubMed]
- Garnero, P.; Sornay-Rendu, E.; Chapuy, M.C.; Delmas, P.D. Increased bone turnover in late postmenopausal women is a major determinant of osteoporosis. J. Bone Miner. Res. 1996, 11, 337–349. [Google Scholar] [CrossRef] [PubMed]
- Dresner-Pollak, R.; Parker, R.A.; Poku, M.; Thompson, J.; Seibel, M.J.; Greenspan, S.L. Biochemical Markers of Bone Turnover Reflect Femoral Bone Loss in Elderly Women. Calcif. Tissue Int. 1996, 59, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.-Y.; Li, H.-L.; Xie, H.; Luo, X.-H.; Peng, Y.-Q.; Yuan, L.-Q.; Sheng, Z.-F.; Dai, R.-C.; Wu, X.-P.; Liao, E.-Y. Age-related bone turnover markers and osteoporotic risk in native Chinese women. BMC Endocr. Disord. 2014, 14, 8. [Google Scholar] [CrossRef]
- Iki, M.; Akiba, T.; Matsumoto, T.; Nishino, H.; Kagamimori, S.; Kagawa, Y.; Yoneshima, H.; Group, J.S. Reference database of biochemical markers of bone turnover for the Japanese female population. Japanese Population-based Osteoporosis (JPOS) Study. Osteoporos. Int. 2004, 15, 981–991. [Google Scholar] [CrossRef]
- Yasuda, H.; Shima, N.; Nakagawa, N.; Yamaguchi, K.; Kinosaki, M.; Mochizuki, S.-i.; Tomoyasu, A.; Yano, K.; Goto, M.; Murakami, A. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proc. Natl. Acad. Sci. USA 1998, 95, 3597–3602. [Google Scholar] [CrossRef]
- Jagtap, V.R.; Ganu, J.V.; Nagane, N.S. BMD and serum intact osteocalcin in postmenopausal osteoporosis women. Indian J. Clin. Biochem. 2011, 26, 70–73. [Google Scholar] [CrossRef]
- Kalaiselvi, V.; Prabhu, K.; Mani Ramesh, V.V. The association of serum osteocalcin with the bone mineral density in postmenopausal women. J. Clin. Diagn. Res. 2013, 7, 814. [Google Scholar]
- Tariq, S.; Tariq, S.; Lone, K.P.; Khaliq, S. Alkaline phosphatase is a predictor of Bone Mineral Density in postmenopausal females. Pak. J. Med. Sci. 2019, 35, 749. [Google Scholar] [CrossRef]
- Zoch, M.L.; Clemens, T.L.; Riddle, R.C. New insights into the biology of osteocalcin. Bone 2016, 82, 42–49. [Google Scholar] [CrossRef]
- Wauquier, F.; Leotoing, L.; Coxam, V.; Guicheux, J.; Wittrant, Y. Oxidative stress in bone remodelling and disease. Trends Mol. Med. 2009, 15, 468–477. [Google Scholar] [CrossRef] [PubMed]
- Domazetovic, V.; Marcucci, G.; Iantomasi, T.; Brandi, M.L.; Vincenzini, M.T. Oxidative stress in bone remodeling: Role of antioxidants. Clin. Cases Miner. Bone. Metab. 2017, 14, 209–216. [Google Scholar] [CrossRef]
- Ardawi, M.-S.M.; Badawoud, M.H.; Hassan, S.M.; Rouzi, A.A.; Ardawi, J.M.; AlNosani, N.M.; Qari, M.H.; Mousa, S.A. Lycopene treatment against loss of bone mass, microarchitecture and strength in relation to regulatory mechanisms in a postmenopausal osteoporosis model. Bone 2016, 83, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Callaway, D.A.; Jiang, J.X. Reactive oxygen species and oxidative stress in osteoclastogenesis, skeletal aging and bone diseases. J. Bone Miner. Metab. 2015, 33, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, C.C.C.; Zamarripa, C.A.J. Menopause Induces Oxidative Stress in Oxidative Stress and Chronic Degenerative Diseases-A Role for Antioxidants; Morales-González, J.A., Ed.; InTech Open Access Publisher: Rijeka, Croatia, 2013; pp. 289–307. [Google Scholar]
- Britton, G.; Liaaen-Jensen, S.; Pfander, H. Carotenoids: Handbook; Birkhäuser: Basel, Switzerland, 2012. [Google Scholar]
- Khachik, F. Distribution and metabolism of dietary carotenoids in humans as a criterion for development of nutritional supplements. Pure Appl. Chem. 2006, 78, 1551–1557. [Google Scholar] [CrossRef]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef]
- Fiedor, J.; Burda, K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients 2014, 6, 466–488. [Google Scholar] [CrossRef]
- Bacanli, M.; BaŞAran, N.; BaŞAran, A.A. Lycopene: Is it Beneficial to Human Health as an Antioxidant? Turk. J. Pharm. Sci. 2017, 14, 311–318. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Sanchez-Zapata, E.; Sayas-Barberá, E.; Sendra, E.; Pérez-Álvarez, J.; Fernández-López, J. Tomato and tomato byproducts. Human health benefits of lycopene and its application to meat products: A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1032–1049. [Google Scholar] [CrossRef]
- Schieber, A.; Carle, R. Occurrence of carotenoid cis-isomers in food: Technological, analytical, and nutritional implications. Trends Food Sci. Technol. 2005, 16, 416–422. [Google Scholar] [CrossRef]
- Hunter, D.C.; Skinner, M.A.; Lister, C.E. Impact of phytochemicals on maintaining bone and joint health. Nutrition 2008, 24, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.; Wang, R.; Ang, L.W.; Low, Y.L.; Yuan, J.M.; Koh, W.P. Protective effects of dietary carotenoids on risk of hip fracture in men: The Singapore Chinese Health Study. J. Bone Miner. Res. 2014, 29, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Castenmiller, J.J.; West, C.E. Bioavailability and bioconversion of carotenoids. Annu. Rev. Nutr. 1998, 18, 19–38. [Google Scholar] [CrossRef]
- Unlu, N.Z.; Bohn, T.; Francis, D.M.; Nagaraja, H.N.; Clinton, S.K.; Schwartz, S.J. Lycopene from heat-induced cis-isomer-rich tomato sauce is more bioavailable than from all-trans-rich tomato sauce in human subjects. Br. J. Nutr. 2007, 98, 140–146. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.J.; Paulino, M.; Stinco, C.M.; Mapelli-Brahm, P.; Wang, X.-D. Study of the time-course of cis/trans (Z/E) isomerization of lycopene, phytoene, and phytofluene from tomato. J. Agric. Food Chem. 2014, 62, 12399–12406. [Google Scholar] [CrossRef]
- Boileau, A.C.; Merchen, N.R.; Wasson, K.; Atkinson, C.A.; Erdman, J.W., Jr. Cis-lycopene is more bioavailable than trans-lycopene in vitro and in vivo in lymph-cannulated ferrets. Nutr. J. 1999, 129, 1176–1181. [Google Scholar] [CrossRef] [PubMed]
- Kachanovsky, D.E.; Filler, S.; Isaacson, T.; Hirschberg, J. Epistasis in tomato color mutations involves regulation of phytoene synthase 1 expression by cis-carotenoids. Proc. Natl. Acad. Sci. USA 2012, 109, 19021–19026. [Google Scholar] [CrossRef] [PubMed]
- Englert, G.; Brown, B.O.; Moss, G.P.; Weedon, B.C.; Britton, G.; Goodwin, T.W.; Simpson, K.L.; Williams, R.J. Prolycopene, a tetra-cis-carotene with two hindered cis-double bonds. J. Chem. Soc. Chem. Comm. 1979, 12, 545–547. [Google Scholar] [CrossRef]
- Wang, X.-D. Lycopene metabolism and its biological significance. Am. J. Clin. Nutr. 2012, 96, 1214–1222. [Google Scholar] [CrossRef]
- Furr, H.C.; Clark, R.M. Intestinal absorption and tissue distribution of carotenoids. J. Nutr. Biochem. 1997, 8, 364–377. [Google Scholar] [CrossRef]
- Boileau, T.W.-M.; Boileau, A.C.; Erdman, J.W., Jr. Bioavailability of all-trans and cis-Isomers of Lycopene. Exp. Biol. Med. 2002, 227, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Anese, M.; Mirolo, G.; Fabbro, A.; Lippe, G. Lycopene bioaccessibility and bioavailability from processed foods. J. Sci. Ind. Res. 2013, 72, 543–547. [Google Scholar]
- Bramley, P.M. Is lycopene beneficial to human health? Phytochemistry 2000, 54, 233–236. [Google Scholar] [CrossRef]
- Schierle, J.; Bretzel, W.; Bühler, I.; Faccin, N.; Hess, D.; Steiner, K.; Schüep, W. Content and isomeric ratio of lycopene in food and human blood plasma. Food Chem. 1997, 59, 459–465. [Google Scholar] [CrossRef]
- Marković, K.; Hruškar, M.; Vahčić, N. Lycopene content of tomato products and their contribution to the lycopene intake of Croatians. Nutr. Res. 2006, 26, 556–560. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Lycopene: A biologically important carotenoid for humans? Arch. Biochem. Biophys. 1996, 336, 1–9. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Uptake of lycopene and its geometrical isomers is greater from heat-processed than from unprocessed tomato juice in humans. Nutr. J. 1992, 122, 2161–2166. [Google Scholar] [CrossRef]
- Ross, A.B.; Vuong, L.T.; Ruckle, J.; Synal, H.A.; Schulze-Koenig, T.; Wertz, K.; Ruembeli, R.; Liberman, R.G.; Skipper, P.L.; Tannenbaum, S.R. Lycopene bioavailability and metabolism in humans: An accelerator mass spectrometry study. Am. J. Clin. Nutr. 2011, 93, 1263–1273. [Google Scholar] [CrossRef]
- Kopec, R.E.; Riedl, K.M.; Harrison, E.H.; Curley Jr, R.W.; Hruszkewycz, D.P.; Clinton, S.K.; Schwartz, S.J. Identification and quantification of apo-lycopenals in fruits, vegetables, and human plasma. J. Agric. Food Chem. 2010, 58, 3290–3296. [Google Scholar] [CrossRef]
- Sicilia, T.; Bub, A.; Rechkemmer, G.; Kraemer, K.; Hoppe, P.P.; Kulling, S.E. Novel lycopene metabolites are detectable in plasma of preruminant calves after lycopene supplementation. Nutr. J. 2005, 135, 2616–2621. [Google Scholar] [CrossRef]
- Cichon, M.J.; Moran, N.E.; Riedl, K.M.; Schwartz, S.J.; Clinton, S.K. Identification of an Epoxide Metabolite of Lycopene in Human Plasma Using 13C-Labeling and QTOF-MS. Metabolites 2018, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Gajic, M.; Zaripheh, S.; Sun, F.; Erdman, J.W., Jr. Apo-8′-lycopenal and apo-12′-lycopenal are metabolic products of lycopene in rat liver. Nutr. J. 2006, 136, 1552–1557. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.; Koo, K.; Lian, F.; Hu, K.Q.; Ernst, H.; Wang, X.-D. Apo-10′-lycopenoic acid, a lycopene metabolite, increases sirtuin 1 mRNA and protein levels and decreases hepatic fat accumulation in ob/ob mice. Nutr. J. 2012, 142, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Ip, B.C.; Hu, K.-Q.; Liu, C.; Smith, D.E.; Obin, M.S.; Ausman, L.M.; Wang, X.-D. Lycopene metabolite, apo-10′-lycopenoic acid, inhibits diethylnitrosamine-initiated, high fat diet–promoted hepatic inflammation and tumorigenesis in mice. Cancer Prev. Res. 2013, 6, 1304–1316. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.A. Absorption, transport and metabolism of carotenoids in humans. Pure Appl. Chem. 1994, 66, 1011–1016. [Google Scholar] [CrossRef]
- Parker, R.S. Absorption, metabolism, and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar] [CrossRef]
- Papachristodoulou, D.; Snape, A.; Elliott, W.H.; Elliott, D.C. Biochemistry and Molecular Biology; Oxford University Press: Oxford, UK, 2014; pp. 163–190. [Google Scholar]
- Wang, X.-D.; Krinsky, N.I.; Marini, R.P.; Tang, G.; Yu, J.; Hurley, R.; Fox, J.G.; Russell, R.M. Intestinal uptake and lymphatic absorption of beta-carotene in ferrets: A model for human beta-carotene metabolism. Am. J. Physiol. Gastrointest. Liver Physiol. 1992, 263, 480–486. [Google Scholar] [CrossRef]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef]
- Sahni, S.; Hannan, M.T.; Blumberg, J.; Cupples, L.A.; Kiel, D.P.; Tucker, K.L. Inverse association of carotenoid intakes with 4-y change in bone mineral density in elderly men and women: The Framingham Osteoporosis Study. Am. J. Clin. Nutr. 2009, 89, 416–424. [Google Scholar] [CrossRef]
- Sahni, S.; Hannan, M.T.; Blumberg, J.; Cupples, L.A.; Kiel, D.P.; Tucker, K.L. Protective effect of total carotenoid and lycopene intake on the risk of hip fracture: A 17-year follow-up from the Framingham Osteoporosis Study. J. Bone Miner. Res. 2009, 24, 1086–1094. [Google Scholar] [CrossRef]
- Mackinnon, E.; Venket Rao, A.; Rao, L. Dietary restriction of lycopene for a period of one month resulted in significantly increased biomarkers of oxidative stress and bone resorption in postmenopausal women. J. Nutr. Health Aging 2011, 15, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, E.; Rao, A.; Josse, R.; Rao, L. Supplementation with the antioxidant lycopene significantly decreases oxidative stress parameters and the bone resorption marker N-telopeptide of type 1 collagen in postmenopausal women. Osteoporos. Int. 2011, 22, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Yang; Zhang; Penniston; Binkley; Tanumihardjo. Serum carotenoid concentrations in postmenopausal women from the United States with and without osteoporosis. Int. J. Vitam. Nutr. Res. 2008, 78, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Russo, C.; Ferro, Y.; Maurotti, S.; Salvati, M.A.; Mazza, E.; Pujia, R.; Terracciano, R.; Maggisano, G.; Mare, R.; Giannini, S. Lycopene and bone: An in vitro investigation and a pilot prospective clinical study. J. Transl. Med. 2020, 18, 43. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, E.S. The Role of the Carotenoid Lycopene as an Antioxidant to Decrease Osteoporosis Risk in Women: Clinical and In Vitro Studies. Ph.D. Thesis, University of Toronto, Ontario, Canada, 2010. [Google Scholar]
- Rao, L.; Mackinnon, E.; Josse, R.; Murray, T.; Strauss, A.; Rao, A. Lycopene consumption decreases oxidative stress and bone resorption markers in postmenopausal women. Osteoporos. Int. 2007, 18, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Calciolari, E.; Donos, N.; Mardas, N. Osteoporotic animal models of bone healing: Advantages and pitfalls. J. Invest. Surg. 2017, 30, 342–350. [Google Scholar] [CrossRef]
- Lelovas, P.P.; Xanthos, T.T.; Thoma, S.E.; Lyritis, G.P.; Dontas, I.A. The laboratory rat as an animal model for osteoporosis research. Comp. Med. 2008, 58, 424–430. [Google Scholar]
- Ozsahin, E.T.; Çam, B.; Dere, F.; Kurkcu, M.; Evruke, C.; Soames, R.; Oguz, O. The effect of alendronate sodium on trabecular bone structure in an osteoporotic rat model. Turk. J. Phys. Med. Rehabil. 2017, 63, 165–174. [Google Scholar] [CrossRef]
- Gao, X.; Ma, W.; Dong, H.; Yong, Z.; Su, R. Establishing a rapid animal model of osteoporosis with ovariectomy plus low calcium diet in rats. Int. J. Clin. Exp. Pathol. 2014, 7, 5123. [Google Scholar]
- Thompson, D.; Simmons, H.; Pirie, C.; Ke, H. FDA Guidelines and animal models for osteoporosis. Bone 1995, 17, 125–133. [Google Scholar] [CrossRef]
- Liang, H.; Yu, F.; Tong, Z.; Zeng, W. Lycopene effects on serum mineral elements and bone strength in rats. Molecules 2012, 17, 7093–7102. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.R.; Vargas-Sanchez, P.K.; Fernandes, R.R.; Ricoldi, M.S.T.; Semeghini, M.S.; Pitol, D.L.; de Sousa, L.G.; Siessere, S.; Bombonato-Prado, K.F. Lycopene influences osteoblast functional activity and prevents femur bone loss in female rats submitted to an experimental model of osteoporosis. J. Bone Miner. Metab. 2019, 37, 658–667. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xue, W.; Cao, Y.; Long, Y.; Xie, M. Effect of lycopene on titanium implant osseointegration in ovariectomized rats. J. Orthop. Surg. Res. 2018, 13, 237. [Google Scholar] [CrossRef] [PubMed]
- Ishimi, Y.; Ohmura, M.; Wang, X.; Yamaguchi, M.; Ikegami, S. Inhibition by carotenoids and retinoic acid of osteoclast-like cell formation induced by bone-resorbing agents in vitro. J. Clin. Biochem. Nutr. 1999, 27, 113–122. [Google Scholar] [CrossRef]
- Park, C.-K.; Ishimi, Y.; Ohmura, M.; Yamaguchi, M.; Ikegami, S. Vitamin A and carotenoids stimulate differentiation of mouse osteoblastic cells. J. Nutr. Sci. Vitaminol. 1997, 43, 281–296. [Google Scholar] [CrossRef]
- Costa-Rodrigues, J.; Fernandes, M.H.; Pinho, O.; Monteiro, P.R.R. Modulation of human osteoclastogenesis and osteoblastogenesis by lycopene. J. Nutr. Biochem. 2018, 57, 26–34. [Google Scholar] [CrossRef]
- Boyce, B.F.; Yao, Z.; Xing, L. Functions of NF-κB in Bone. Ann. N. Y. Acad. Sci. 2010, 1192, 367. [Google Scholar] [CrossRef]
- Linnewiel-Hermoni, K.; Motro, Y.; Miller, Y.; Levy, J.; Sharoni, Y. Carotenoid derivatives inhibit nuclear factor kappa B activity in bone and cancer cells by targeting key thiol groups. Free Radic. Biol. Med. 2014, 75, 105–120. [Google Scholar] [CrossRef]
- Marcotorchino, J.; Romier, B.; Gouranton, E.; Riollet, C.; Gleize, B.; Malezet-Desmoulins, C.; Landrier, J.F. Lycopene attenuates LPS-induced TNF-α secretion in macrophages and inflammatory markers in adipocytes exposed to macrophage-conditioned media. Mol. Nutr. Food Res. 2012, 56, 725–732. [Google Scholar] [CrossRef]
- Feng, D.; Ling, W.-H.; Duan, R.-D. Lycopene suppresses LPS-induced NO and IL-6 production by inhibiting the activation of ERK, p38MAPK, and NF-κB in macrophages. Inflamm. Res. 2010, 59, 115–121. [Google Scholar] [CrossRef]
- De Stefano, D.; Maiuri, M.C.; Simeon, V.; Grassia, G.; Soscia, A.; Cinelli, M.P.; Carnuccio, R. Lycopene, quercetin and tyrosol prevent macrophage activation induced by gliadin and IFN-γ. Eur. J. Pharmacol. 2007, 566, 192–199. [Google Scholar] [CrossRef] [PubMed]

| Fixed Risk Factors | Modifiable Risk Factors |
|---|---|
| |
| |
|
|
| |
|
|
| |
|
|
| Author and Year | Cohort | Lycopene Formulation and Study Duration | Outcome |
|---|---|---|---|
| Russo et al. (2020) [143] | Postmenopausal women (n = 39) Age: 63 ± 7 years | 3.9 mg/day as tomato sauce 3 months | Patients who consumed tomato sauce did not show a significant loss of BMD compared to control group |
| Mackinnon et al. (2011) [141] | Postmenopausal women (n = 60) Age: 50–60 years | 30 mg/day (regular tomato juice), 70 mg/day (lycopene-rich tomato juice), 30 mg/day (Lyc-O-Mato capsules) 4 months | Lycopene intervention in capsule or juice form supplying at least 30 mg/day led to decreased oxidative stress and bone resorption markers |
| Mackinnon (2010) [144] | Postmenopausal women (n = 45) Age: 55 years | 43.33 mg/day supplementation 4 months | Lycopene supplemented group showed significantly lower levels of bone resorption marker (NTx) |
| Mackinnon et al. (2011) [140] | Postmenopausal women (n = 23) Age: 50–60 years | Lycopene intake at baseline and after one month of lycopene restriction was 3.5 mg/d and 0.13 mg/d, respectively (using 7-day dietary records) | Bone resorption marker (NTx) was increased after a month of lycopene restriction Endogenous antioxidant enzymes (SOD and catalase) were decreased after a month of lycopene restriction |
| Rao et al. (2007) [145] | Postmenopausal women (n = 33) Age: 50–60 years | Lycopene intake categorized into four groups as ranged from 1.76 to 7.35 mg/day (using 7-day dietary records) | Serum NTx values dose-dependently decreased Postmenopausal women who consumed 7.35 mg lycopene/day had lower serum NTx compared to the other three groups No difference in bone formation markers |
| Author and Year | Animal Strain | Lycopene Dose and Study Duration | Outcome |
|---|---|---|---|
| Oliveira et al. (2019) [152] | Female Wistar rats | 10 mg/kg BW/day 4 weeks pre-OVX and 8 weeks post-OVX | Decreased bone loss in femur epiphysis in the OVX + lycopene group compared to the OVX control group |
| Li et al. (2018) [153] | Female Sprague-Dawley rats | 50 mg/kg BW/day 12 weeks | Higher bone volume and trabecular thickness with low trabecular spaces in the OVX + lycopene group compared to the OVX control group Increased bone contact and bone area around the implant were in the lycopene-treated group compared to controls |
| Ardawi et al. (2016) [99] | Female Wistar rats | 15, 30, 45 mg/kg BW per day 12 weeks | Lycopene treatment dose-dependently enhanced BMD and BMC at the lumbar spine and humerus compared to OVX control group Lycopene (30 and 45 mg/kg BW) increased bone formation markers (serum-OC and serum PINP) while bone resorption markers (serum-CTX-1 and urine-DPD) were decreased |
| Iimura et al. (2015) [14] | Female Sprague-Dawley 6-week-old | 0, 50, 100, 200 mg lycopene/kg diet/day 9 weeks | Lycopene (100 mg/kg) increased lumbar spine BMD and femoral-breaking force compared to OVX control group Bone resorption markers were low in all lycopene-treated groups |
| Iimura et al. (2014) [13] | Female Sprague–Dawley 6-week-old | 0, 50, 100 mg/kg diet lycopene 9 weeks | Lycopene (100 mg/kg) increased BMD of the lumbar spine and the tibial proximal metaphysis compared to OVX control group |
| Liang et al. (2012) [151] | Female Wistar rats 8-week-old | 20, 30, 40 mg/kg BW/day 8 weeks | Lycopene (30 and 40 mg/kg BW) dose-dependently increased BMD and BMC in OVX rats compared to OVX control group |
| Author and Year | Cell Line | Lycopene Concentration | Outcome |
|---|---|---|---|
| Russo et al. (2020) [143] | Human osteoblast-like cell line Saos-2 | 5 and 10 μM | Lycopene suppressed RANKL expression indicating the reduction of bone resorption Lycopene reduced the stimulatory effect of ALP within 24 h indicating possible role in mineralization |
| Oliveira et al. (2019) [152] | Osteoblastic cells from femur medullary canals of ovariectomized female rats | 1 μM | Lycopene upregulated the genes associated with bone metabolism of osteoblastic cells within 3–10 days |
| Costa-Rodrigues et al. (2018) [156] | Osteoblastic cells (human mesenchymal stem cells bone-marrow-derived) Osteoclastic cells (human peripheral blood mononuclear cells) | 5 nM−50 μM | Lycopene (≥500 nM) increased osteoblastic cell proliferation and differentiation Lycopene (≥500 nM) significantly decreased osteoclast differentiation |
| Marcotorchino et al. (2012) [159] | RAW 264.7 cells | 0.5, 1, 2 μM | Lycopene dose-dependently reduced the lipopolysaccharides (LPS) mediated activation of inflammatory cytokine (TNF-α) produced by macrophages |
| Feng et al. (2010) [160] | RAW 264.7 cells | 1–10 μM | Lycopene dose-dependently inhibited the increase of nitric oxide production and the secretion of IL-6 when RAW cells were stimulated by LPS |
| Stefano et al. (2007) [161] | RAW 264.7 cells | 5, 10, 20 μM | Lycopene (20 μM) significantly inhibited the ROS accumulated due to addition of gliadin Lycopene (20 μM) significantly inhibited increase in nitric oxide synthase levels |
| Rao et al. (2003) [34] | Osteoclast were generated from bone marrow cells | 0.01, 0.1, 1, 10 μM | Lycopene (10 μM) significantly inhibited PTH stimulated resorption by osteoclasts |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walallawita, U.S.; Wolber, F.M.; Ziv-Gal, A.; Kruger, M.C.; Heyes, J.A. Potential Role of Lycopene in the Prevention of Postmenopausal Bone Loss: Evidence from Molecular to Clinical Studies. Int. J. Mol. Sci. 2020, 21, 7119. https://doi.org/10.3390/ijms21197119
Walallawita US, Wolber FM, Ziv-Gal A, Kruger MC, Heyes JA. Potential Role of Lycopene in the Prevention of Postmenopausal Bone Loss: Evidence from Molecular to Clinical Studies. International Journal of Molecular Sciences. 2020; 21(19):7119. https://doi.org/10.3390/ijms21197119
Chicago/Turabian StyleWalallawita, Umani S., Frances M. Wolber, Ayelet Ziv-Gal, Marlena C. Kruger, and Julian A. Heyes. 2020. "Potential Role of Lycopene in the Prevention of Postmenopausal Bone Loss: Evidence from Molecular to Clinical Studies" International Journal of Molecular Sciences 21, no. 19: 7119. https://doi.org/10.3390/ijms21197119
APA StyleWalallawita, U. S., Wolber, F. M., Ziv-Gal, A., Kruger, M. C., & Heyes, J. A. (2020). Potential Role of Lycopene in the Prevention of Postmenopausal Bone Loss: Evidence from Molecular to Clinical Studies. International Journal of Molecular Sciences, 21(19), 7119. https://doi.org/10.3390/ijms21197119








