Antimicrobial Peptides as New Combination Agents in Cancer Therapeutics: A Promising Protocol against HT-29 Tumoral Spheroids
Abstract
1. Introduction
2. Results
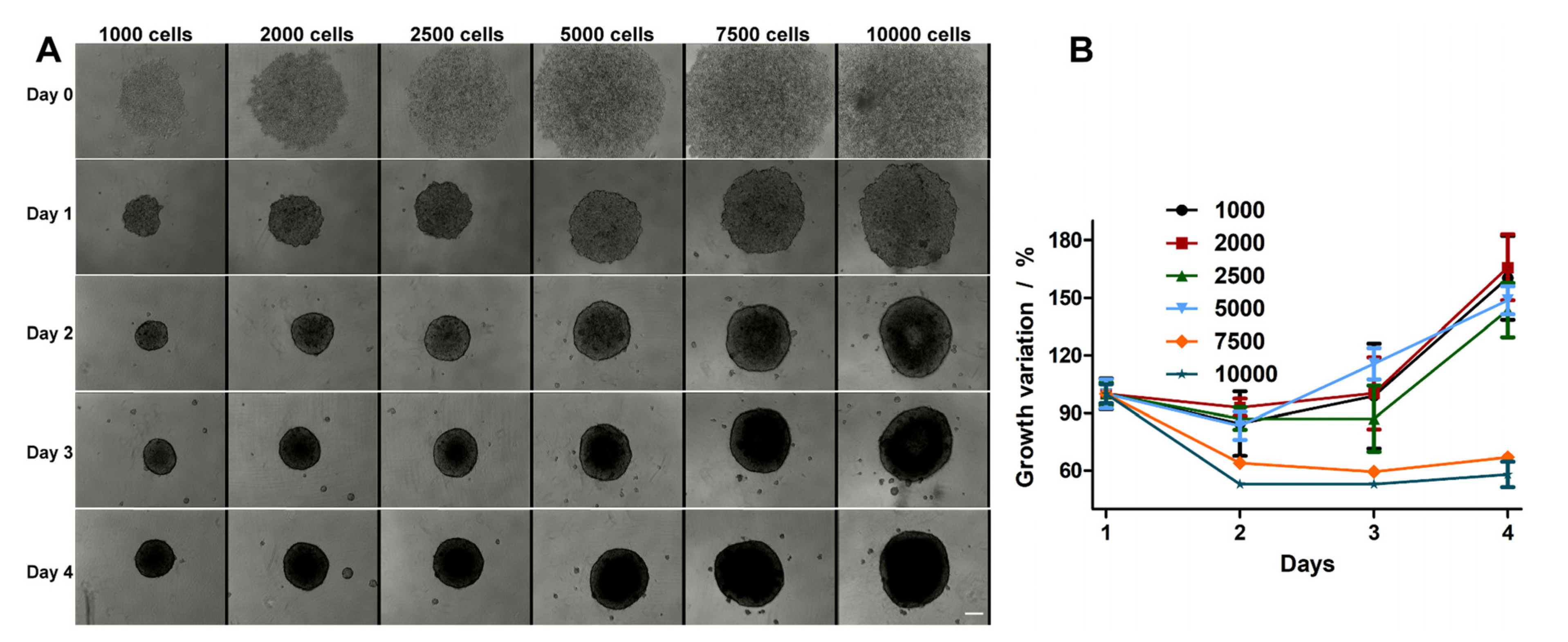
2.1. HT-29 Spheroids Evolution
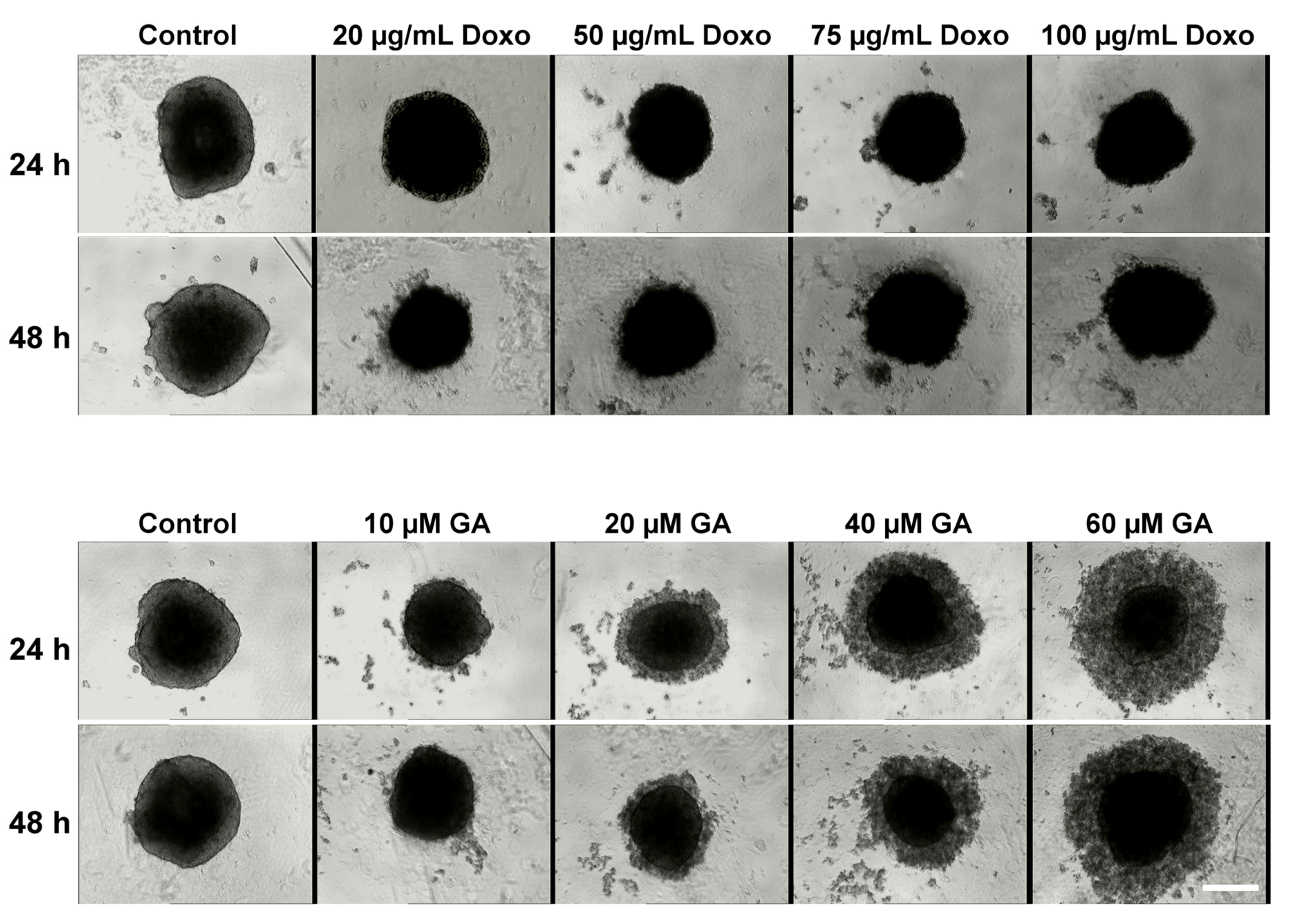
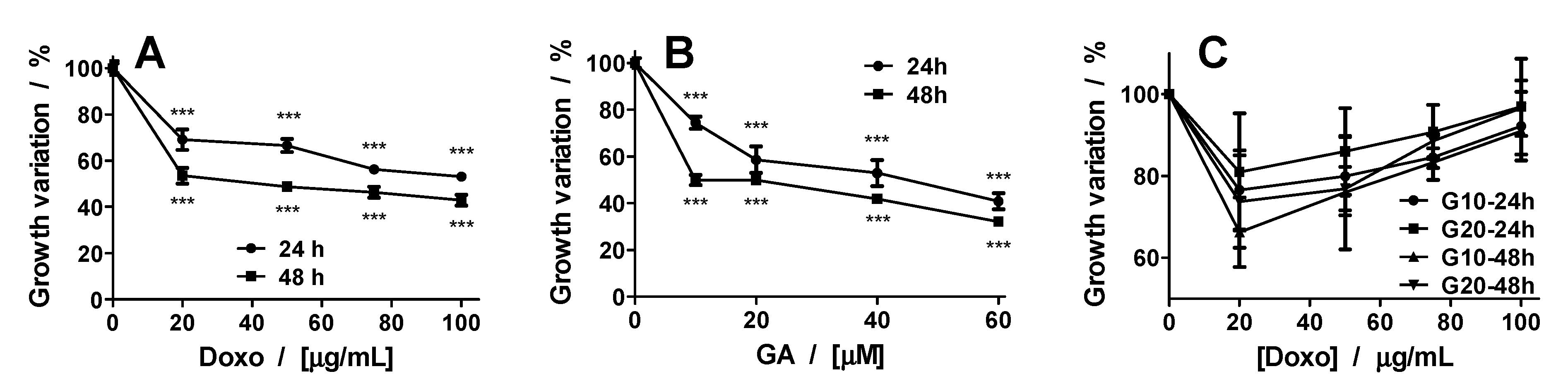
2.2. Treatment Effect on HT-29 Spheroid Morphology
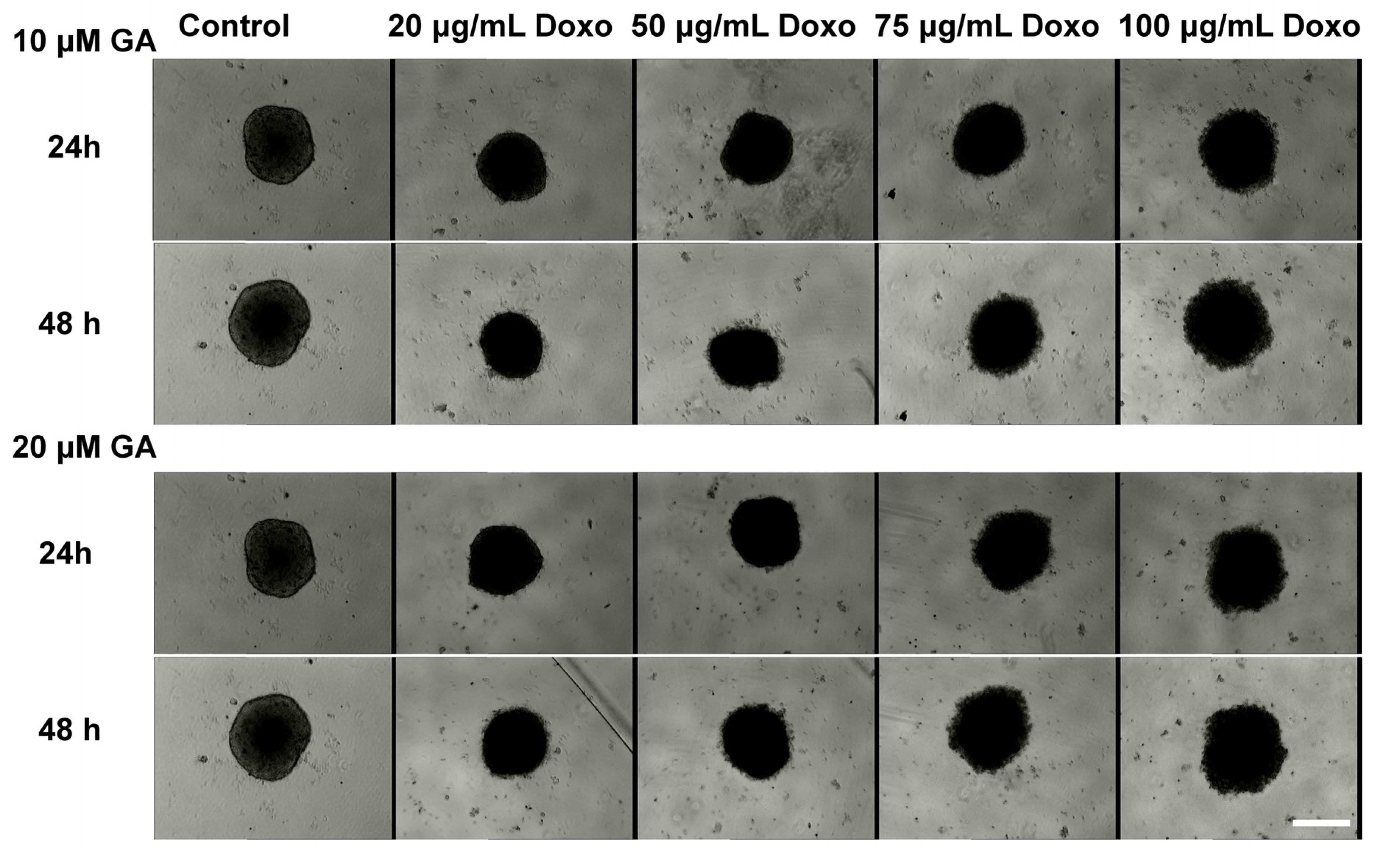
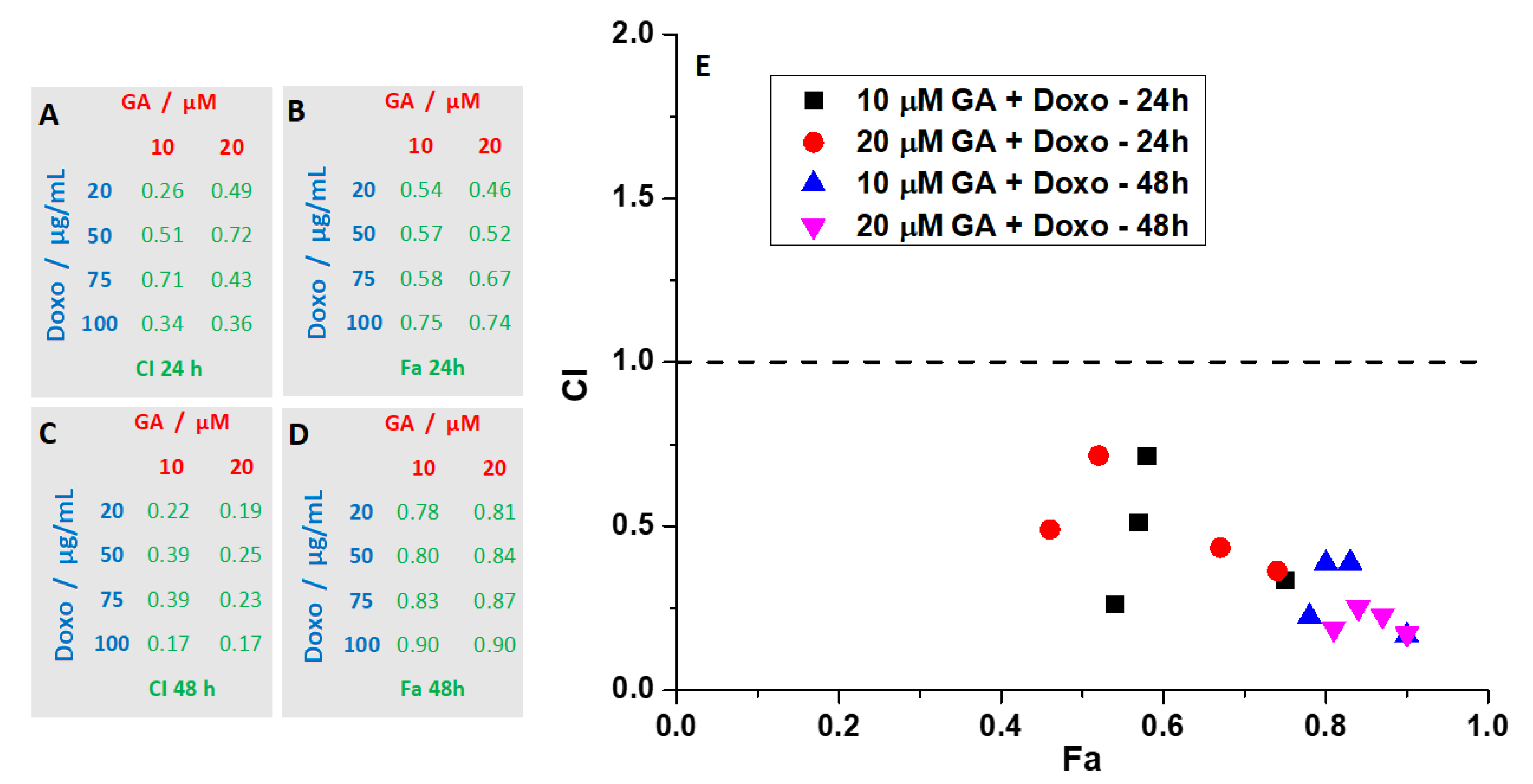
2.3. Synergetic Effect of Doxo and GA Treatments on HT-29 Spheroids
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Spheroid Formation and Analysis
4.4. Treatment of HT-29 Spheroids
4.5. Cell Viability Assays
4.6. ATP Assays
4.7. Analysis of Combination Index (CI)
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CI | Combination Index |
| Doxo | Doxorubicin |
| GA | Gramicidin A |
| Fa | Fraction affected |
References
- Tannock, I.F.; Lee, C.M.; Tunggal, J.K.; Cowan, D.S.; Egorin, M.J. Limited penetration of anticancer drugs through tumor tissue: A potential cause of resistance of solid tumors to chemotherapy. Clin. Cancer Res. 2002, 8, 878–884. [Google Scholar] [PubMed]
- McMillin, D.W.; Negri, J.M.; Mitsiades, C.S. The role of tumour-stromal interactions in modifying drug response: Challenges and opportunities. Nat. Rev. Drug Discov. 2013, 12, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Desoize, B.; Jardillier, J. Multicellular resistance: A paradigm for clinical resistance? Crit. Rev. Oncol. Hematol. 2000, 36, 193–207. [Google Scholar] [CrossRef]
- Tamburrino, A.; Piro, G.; Carbone, C.; Tortora, G.; Melisi, D. Mechanisms of resistance to chemotherapeutic and anti-angiogenic drugs as novel targets for pancreatic cancer therapy. Front. Pharmacol. 2013, 4, 56. [Google Scholar] [CrossRef]
- Mitchison, T.J. The proliferation rate paradox in antimitotic chemotherapy. Mol. Biol. Cell 2012, 23, 1–6. [Google Scholar] [CrossRef]
- Deslouches, B.; Di, Y.P. Antimicrobial peptides with selective antitumor mechanisms: Prospect for anticancer applications. Oncotarget 2017, 8, 46635–46651. [Google Scholar] [CrossRef]
- Deslouches, B.; Steckbeck, J.D.; Craigo, J.K.; Doi, Y.; Miexztzner, T.A.; Montelaro, R.C. Rational design of engineered cationic antimicrobial peptides consisting exclusively of arginine and tryptophan, and their activity against multidrug-resistant pathogens. Antimicrob. Agents Chemother. 2013, 57, 2511–2521. [Google Scholar] [CrossRef]
- Dutta, P.; Das, S. Mammalian Antimicrobial Peptides: Promising Therapeutic Targets against Infection and Chronic Inflammation. Curr. Topics Med. Chem. 2016, 16, 99–129. [Google Scholar] [CrossRef]
- Nalbantsoy, A.; Hempel, B.F.; Petras, D.; Heiss, P.; Gocmen, B.; Igci, N.; Yildiz, M.Z.; Sussmuth, R.D. Combined venom profiling and cytotoxicity screening of the Radde’s mountain viper (Montivipera raddei) and Mount Bulgar Viper (Montivipera bulgardaghica) with potent cytotoxicity against human A549 lung carcinoma cells. Toxicon Off. J. Int. Soc. Toxinology 2017, 135, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Ghani, L.M.; Rahmy, T.R.; Tawfik, M.M.; Kaziri, I.; Al-Obaidi, A.; Rowan, E.G.; Plevin, R.; Abdel-Rahman, M.A. Cytotoxicity of Nubein6.8 peptide isolated from the snake venom of Naja nubiae on melanoma and ovarian carcinoma cell lines. Toxicon Off. J. Int. Soc. Toxinology 2019, 168, 22–31. [Google Scholar]
- Juvvadi, P.; Vunnam, S.; Merrifield, E.L.; Boman, H.G.; Merrifield, R.B. Hydrophobic effects on antibacterial and channel-forming properties of cecropin A-melittin hybrids. J. Pept. Sci. 1996, 2, 223–232. [Google Scholar] [PubMed]
- Kheirandish, M.H.; Jaliani, H.Z.; Rahmani, B.; Nikukar, H. Specific targeting of a pore-forming toxin (listeriolysin O) to LHRH-positive cancer cells using LHRH targeting peptide. Toxicon Off. J. Int. Soc. Toxinology 2019, 164, 82–86. [Google Scholar] [CrossRef]
- Teixeira, V.; Feio, M.J.; Bastos, M. Role of lipids in the interaction of antimicrobial peptides with membranes. Prog. Lipid Res. 2012, 51, 149–177. [Google Scholar] [CrossRef]
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 21. [Google Scholar] [CrossRef]
- Kang, H.K.; Choi, M.C.; Seo, C.H.; Park, Y. Therapeutic Properties and Biological Benefits of Marine-Derived Anticancer Peptides. Int. J. Mol. Sci. 2018, 19, 919. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.M.; Cukierman, E. Modeling tissue morphogenesis and cancer in 3D. Cell 2007, 130, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, G. Multicellular spheroids as an in vitro tumor model. Cancer Lett. 1998, 131, 29–34. [Google Scholar] [CrossRef]
- Bray, F.; Moller, B. Predicting the future burden of cancer. Nat. Rev. Cancer 2006, 6, 63–74. [Google Scholar] [CrossRef]
- Edmondson, R.; Broglie, J.J.; Adcock, A.F.; Yang, L. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. Assay Drug Dev. Technol. 2014, 12, 207–218. [Google Scholar] [CrossRef]
- Imamura, Y.; Mukohara, T.; Shimono, Y.; Funakoshi, Y.; Chayahara, N.; Toyoda, M.; Kiyota, N.; Takao, S.; Kono, S.; Nakatsura, T.; et al. Comparison of 2D- and 3D-culture models as drug-testing platforms in breast cancer. Oncol. Rep. 2015, 33, 1837–1843. [Google Scholar] [CrossRef]
- Mellor, H.R.; Callaghan, R. Accumulation and distribution of doxorubicin in tumour spheroids: The influence of acidity and expression of P-glycoprotein. Cancer Chemother. Pharmacol. 2011, 68, 1179–1190. [Google Scholar] [CrossRef] [PubMed]
- Frassineti, G.L.; Zoli, W.; Silvestro, L.; Serra, P.; Milandri, C.; Tienghi, A.; Gianni, L.; Gentile, A.; Salzano, E.; Amadori, D. Paclitaxel plus doxorubicin in breast cancer: An Italian experience. Semin. Oncol. 1997, 24, S17–S19. [Google Scholar] [PubMed]
- Kelkar, D.A.; Chattopadhyay, A. The gramicidin ion channel: A model membrane protein. Biochimica et Biophysica Acta (BBA)-Biomembranes 2007, 1768, 2011–2025. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Qin, L.; Pace, C.J.; Wong, P.; Malonis, R.; Gao, J. Solubilized gramicidin A as potential systemic antibiotics. Chembiochem A Eur. J. Chem. Biol. 2012, 13, 51–55. [Google Scholar] [CrossRef]
- Otten-Kuipers, M.A.; Beumer, T.L.; Kronenburg, N.A.; Roelofsen, B.; Op den Kamp, J.A. Effects of gramicidin and tryptophan-N-formylated gramicidin on the sodium and potassium content of human erythrocytes. Mol. Membr. Biol. 1996, 13, 225–232. [Google Scholar] [CrossRef]
- David, J.M.; Owens, T.A.; Barwe, S.P.; Rajasekaran, A.K. Gramicidin A induces metabolic dysfunction and energy depletion leading to cell death in renal cell carcinoma cells. Mol. Cancer Ther. 2013, 12, 2296–2307. [Google Scholar] [CrossRef]
- David, J.M.; Owens, T.A.; Inge, L.J.; Bremner, R.M.; Rajasekaran, A.K. Gramicidin A blocks tumor growth and angiogenesis through inhibition of hypoxia-inducible factor in renal cell carcinoma. Mol. Cancer Ther. 2014, 13, 788–799. [Google Scholar] [CrossRef]
- Chen, T.; Wang, Y.; Yang, Y.; Yu, K.; Cao, X.; Su, F.; Xu, H.; Peng, Y.; Hu, Y.; Qian, F.; et al. Gramicidin inhibits human gastric cancer cell proliferation, cell cycle and induced apoptosis. Biol. Res. 2019, 52, 57. [Google Scholar] [CrossRef]
- Wang, R.Q.; Geng, J.; Sheng, W.J.; Liu, X.J.; Jiang, M.; Zhen, Y.S. The ionophore antibiotic gramicidin A inhibits pancreatic cancer stem cells associated with CD47 down-regulation. Cancer Cell Int. 2019, 19, 145. [Google Scholar] [CrossRef]
- Rao, D.K.; Liu, H.; Ambudkar, S.V.; Mayer, M. A combination of curcumin with either gramicidin or ouabain selectively kills cells that express the multidrug resistance-linked ABCG2 transporter. J. Biol. Chem. 2014, 289, 31397–31410. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.X.; Ye, L.; Gong, J.W.; Ren, H.H.; Ding, Y.F.; Chen, X.Y.; Liu, X.N.; Lu, P.; Wei, F.; Xu, W.J.; et al. Alteronol Enhances the Anti-tumor Activity and Reduces the Toxicity of High-Dose Adriamycin in Breast Cancer. Front. Pharmacol. 2019, 10, 285. [Google Scholar] [CrossRef] [PubMed]
- Zheng, N.; Liu, W.Q.; Li, B.F.; Nie, H.F.; Liu, J.; Cheng, Y.L.; Wang, J.C.; Dong, H.Y.; Jia, L. Co-delivery of sorafenib and metapristone encapsulated by CXCR4-targeted PLGA-PEG nanoparticles overcomes hepatocellular carcinoma resistance to sorafenib. J. Exp. Clin. Cancer Res. 2019, 38, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Heidler, C.L.; Roth, E.K.; Thiemann, M.; Blattmann, C.; Perez, R.L.; Huber, P.E.; Kovac, M.; Amthor, B.; Neu-Yilik, G.; Kulozik, A.E. Prexasertib (LY2606368) reduces clonogenic survival by inducing apoptosis in primary patient-derived osteosarcoma cells and synergizes with cisplatin and talazoparib. Int. J. Cancer 2020, 147, 1059–1070. [Google Scholar] [CrossRef]
- Pasquier, E.; Ciccolini, J.; Carre, M.; Giacometti, S.; Fanciullino, R.; Pouchy, C.; Montero, M.P.; Serdjebi, C.; Kavallaris, M.; Andre, N. Propranolol potentiates the anti-angiogenic effects and antitumor efficacy of chemotherapy agents: Implication in breast cancer treatment. Oncotarget 2011, 2, 797–809. [Google Scholar] [CrossRef] [PubMed]
- Joyce, J.A.; Pollard, J.W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 2009, 9, 239–252. [Google Scholar] [CrossRef]
- Johnson, J.I.; Decker, S.; Zaharevitz, D.; Rubinstein, L.V.; Venditti, J.; Schepartz, S.; Kalyandrug, S.; Christian, M.; Arbuck, S.; Hollingshead, M.; et al. Relationships between drug activity in NCI preclinical in vitro and in vivo models and early clinical trials. Brit. J. Cancer 2001, 84, 1424–1431. [Google Scholar] [CrossRef]
- Siegel, R.; Desantis, C.; Jemal, A. Colorectal cancer statistics, 2014. CA A Cancer J. Clin. 2014, 64, 104–117. [Google Scholar] [CrossRef]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef]
- Redondo-Blanco, S.; Fernandez, J.; Gutierrez-del-Rio, I.; Villar, C.J.; Lombo, F. New Insights toward Colorectal Cancer Chemotherapy Using Natural Bioactive Compounds. Front. Pharmacol. 2017, 8, 109. [Google Scholar] [CrossRef]
- Van der Jeught, K.; Xu, H.C.; Li, Y.J.; Lu, X.B.; Ji, G. Drug resistance and new therapies in colorectal cancer. World J. Gastroenterol. 2018, 24, 3834–3848. [Google Scholar] [CrossRef]
- Ohashi, W.; Yamamine, N.; Imura, J.; Hattori, Y. SKL2001 suppresses colon cancer spheroid growth through regulation of the E-cadherin/beta-Catenin complex. Biochem. Biophys. Res. Commun. 2017, 493, 1342–1348. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Jiang, L.; Xia, Q.; Zhong, L. Synergistic inhibitory effects of curcumin and 5-fluorouracil on the growth of the human colon cancer cell line HT-29. Chemotherapy 2006, 52, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, A.; Canu, B.; Ali, G.; Orlandi, P.; Allegrini, G.; Di Desidero, T.; Emmenegger, U.; Fontanini, G.; Danesi, R.; Del Tacca, M.; et al. Metronomic 5-fluorouracil, oxaliplatin and irinotecan in colorectal cancer. Eur. J. Pharmacol. 2009, 619, 8–14. [Google Scholar] [CrossRef]
- Zhu, H.; Zhao, F.; Yu, S.; He, J.; Deng, L.; Yi, C.; Huang, Y. The synergistic effects of low-dose irinotecan and TRAIL on TRAIL-resistant HT-29 colon carcinoma in vitro and in vivo. Int. J. Mol. Med. 2012, 30, 1087–1094. [Google Scholar] [CrossRef]
- Hashim, F.; Ismail, W.I.; Ali, A.M. Combinatorial Cytotoxic Effects of Gelam Honey and 5-Fluorouracil against Human Adenocarcinoma Colon Cancer HT-29 Cells in Vitro. Int. J. Cell Biol. 2019, 2019, 3059687. [Google Scholar]
- Jang, H.J.; Hong, E.M.; Jang, J.; Choi, J.E.; Park, S.W.; Byun, H.W.; Koh, D.H.; Choi, M.H.; Kae, S.H.; Lee, J. Synergistic Effects of Simvastatin and Irinotecan against Colon Cancer Cells with or without Irinotecan Resistance. Gastroenterol. Res. Pract. 2016, 2016, 7891374. [Google Scholar] [CrossRef]
- Bacalum, M.; Zorila, B.; Radu, M. Investigating the Anticancer Activity of Some Cationic Antimicrobial Peptides in Epithelial Tumor Cells. Rom. Rep. Phys. 2016, 68, 1159–1169. [Google Scholar]
- Maraming, P.; Maijaroen, S.; Klaynongsruang, S.; Boonsiri, P.; Daduang, S.; Chung, J.G.; Daduang, J. Antitumor Ability of KT2 Peptide Derived from Leukocyte Peptide of Crocodile Against Human HCT116 Colon Cancer Xenografts. In Vivo 2018, 32, 1137–1144. [Google Scholar] [CrossRef]
- Freire, K.A.; Torres, M.T.; Lima, D.B.; Monteiro, M.L.; de Menezes, R.B.; Martins, A.M.C.; Oliveira, V.X., Jr. Wasp venom peptide as a new antichagasic agent. Toxicon Off. J. Int. Soc. Toxinology 2020, 181, 71–78. [Google Scholar] [CrossRef]
- Soliman, C.; Eastwood, S.; Truong, V.K.; Ramsland, P.A.; Elbourne, A. The membrane effects of melittin on gastric and colorectal cancer. PLoS ONE 2019, 14, e0224028. [Google Scholar] [CrossRef]
- Raimundo, L.; Espadinha, M.; Soares, J.; Loureiro, J.B.; Alves, M.G.; Santos, M.M.M.; Saraiva, L. Improving anticancer activity towards colon cancer cells with a new p53-activating agent. Br. J. Pharmacol. 2018, 175, 3947–3962. [Google Scholar] [CrossRef] [PubMed]
- Sousa, M.L.; Ribeiro, T.; Vasconcelos, V.; Linder, S.; Urbatzka, R. Portoamides A and B are mitochondrial toxins and induce cytotoxicity on the proliferative cell layer of in vitro microtumours. Toxicon Off. J. Int. Soc. Toxinology 2020, 175, 49–56. [Google Scholar] [CrossRef]
- Fernandez-Tome, S.; Xu, F.; Han, Y.; Hernandez-Ledesma, B.; Xiao, H. Inhibitory Effects of Peptide Lunasin in Colorectal Cancer HCT-116 Cells and Their Tumorsphere-Derived Subpopulation. Int. J. Mol. Sci. 2020, 21, 537. [Google Scholar] [CrossRef] [PubMed]
- David, J.M.; Rajasekaran, A.K. Gramicidin A: A New Mission for an Old Antibiotic. J. Kidney Cancer VHL 2015, 2, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez-Boussard, T.; McLeod, H.; Klein, T.E.; Altman, R.B. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharm. Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Sokolova, E.; Kutova, O.; Grishina, A.; Pospelov, A.; Guryev, E.; Schulga, A.; Deyev, S.; Balalaeva, I. Penetration Efficiency of Antitumor Agents in Ovarian Cancer Spheroids: The Case of Recombinant Targeted Toxin DARPin-LoPE and the Chemotherapy Drug, Doxorubicin. Pharmaceutics 2019, 11, 219. [Google Scholar] [CrossRef]
- Lupertz, R.; Watjen, W.; Kahl, R.; Chovolou, Y. Dose- and time-dependent effects of doxorubicin on cytotoxicity, cell cycle and apoptotic cell death in human colon cancer cells. Toxicology 2010, 271, 115–121. [Google Scholar] [CrossRef]
- Chen, K.; Xu, X.M.; Kobayashi, S.; Timm, D.; Jepperson, T.; Liang, Q.R. Caloric Restriction Mimetic 2-Deoxyglucose Antagonizes Doxorubicin-induced Cardiomyocyte Death by Multiple Mechanisms. J. Biol. Chem. 2011, 286, 21993–22006. [Google Scholar] [CrossRef]
- Tao, Z.M.; Withers, H.G.; Penefsky, H.S.; Goodisman, J.; Souid, A.K. Inhibition of cellular respiration by doxorubicin. Chem. Res. Toxicol. 2006, 19, 1051–1058. [Google Scholar] [CrossRef]
- Du, B.Y.; Song, W.; Bai, L.; Shen, Y.; Miao, S.Y.; Wang, L.F. Synergistic Effects of Combination Treatment with Bortezomib and Doxorubicin in Human Neuroblastoma Cell Lines. Chemotherapy 2012, 58, 44–51. [Google Scholar] [CrossRef]
- Arianingrum, R.; Arty, I.S.; Atun, S. Synergistic Combination of Fluoro Chalcone and Doxorubicin on HeLa Cervical Cancer cells by Inducing Apoptosis. In Proceedings of the AIP Conference, Yogyakarta, Indonesia, 15–16 November 2017; p. 1823. [Google Scholar]
- Vogus, D.R.; Pusuluri, A.; Chen, R.W.; Mitragotri, S. Schedule dependent synergy of gemcitabine and doxorubicin: Improvement of in vitro efficacy and lack of in vitro-in vivo correlation. Bioeng. Transl. Med. 2018, 3, 49–57. [Google Scholar] [CrossRef]
- Tun, J.O.; Salvador-Reyes, L.A.; Velarde, M.C.; Saito, N.; Suwanborirux, K.; Concepcion, G.P. Synergistic Cytotoxicity of Renieramycin M and Doxorubicin in MCF-7 Breast Cancer Cells. Mar. Drugs 2019, 17, 536. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010, 70, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raileanu, M.; Popescu, A.; Bacalum, M. Antimicrobial Peptides as New Combination Agents in Cancer Therapeutics: A Promising Protocol against HT-29 Tumoral Spheroids. Int. J. Mol. Sci. 2020, 21, 6964. https://doi.org/10.3390/ijms21186964
Raileanu M, Popescu A, Bacalum M. Antimicrobial Peptides as New Combination Agents in Cancer Therapeutics: A Promising Protocol against HT-29 Tumoral Spheroids. International Journal of Molecular Sciences. 2020; 21(18):6964. https://doi.org/10.3390/ijms21186964
Chicago/Turabian StyleRaileanu, Mina, Aurel Popescu, and Mihaela Bacalum. 2020. "Antimicrobial Peptides as New Combination Agents in Cancer Therapeutics: A Promising Protocol against HT-29 Tumoral Spheroids" International Journal of Molecular Sciences 21, no. 18: 6964. https://doi.org/10.3390/ijms21186964
APA StyleRaileanu, M., Popescu, A., & Bacalum, M. (2020). Antimicrobial Peptides as New Combination Agents in Cancer Therapeutics: A Promising Protocol against HT-29 Tumoral Spheroids. International Journal of Molecular Sciences, 21(18), 6964. https://doi.org/10.3390/ijms21186964




