Targeting Aging Pathways in Chronic Obstructive Pulmonary Disease
Abstract
1. Introduction
Impact and Pathophysiology of COPD in the Elderly
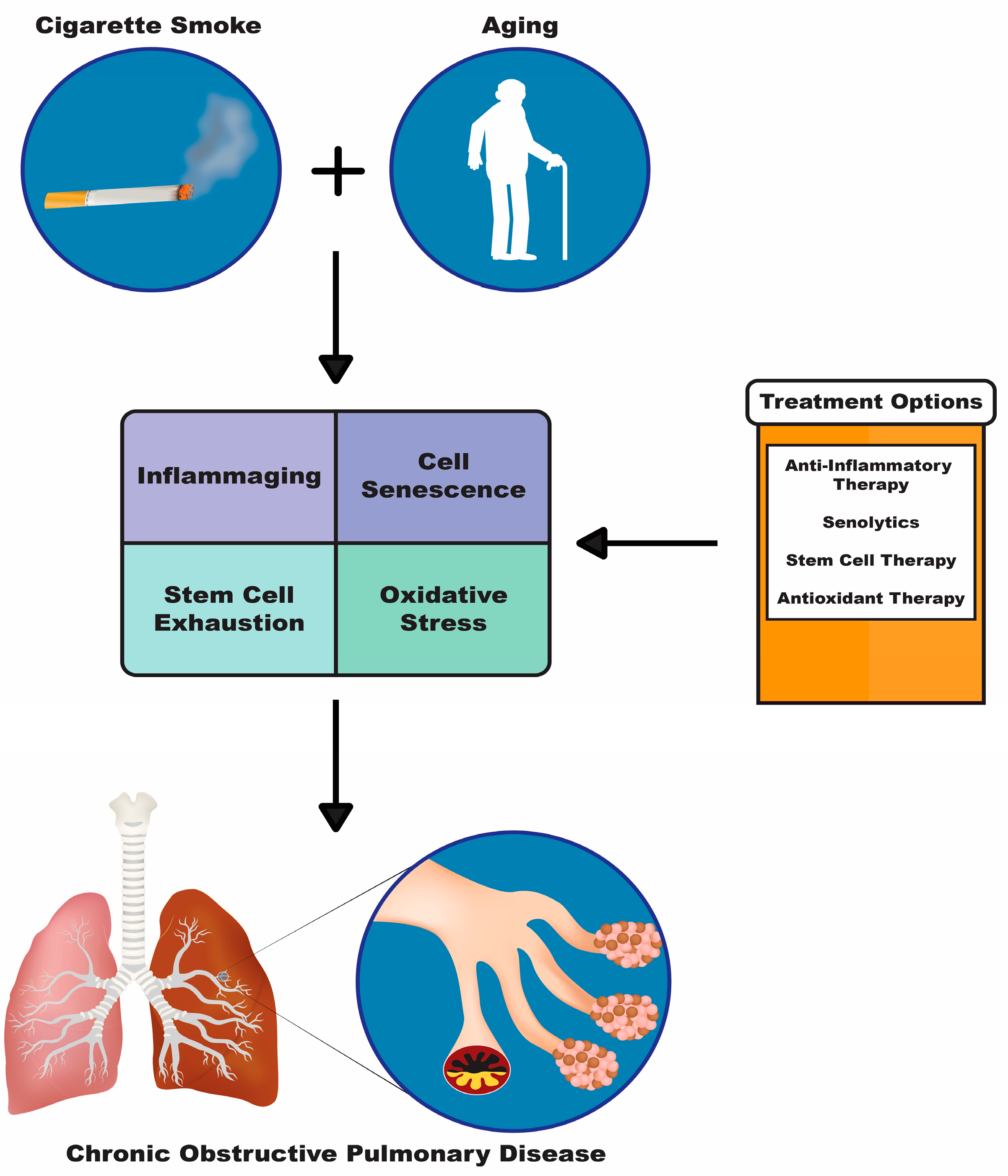
2. Aging-Associated Molecular Mechanisms in the Pathogenesis of COPD
2.1. Inflammaging
2.2. Cell Senescence
2.3. Stem Cell Exhaustion
2.4. Oxidative Stress
3. Current Treatment Options for Individuals with COPD
4. Anti-Aging Treatment Strategies in COPD
4.1. Repurposed Drugs as Potential Anti-Aging Therapies
4.1.1. Resveratrol
4.1.2. Metformin
4.1.3. Melatonin
4.2. The FGF23/Klotho Signaling Pathway
4.3. IL-6 as an Anti-Inflammatory Therapy in COPD
5. Potential Novel Anti-Aging Treatments for COPD
5.1. Senolytics
5.2. Stem Cell Therapies
5.3. Antioxidant Therapy
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- May, S.M.; Li, J.T. Burden of chronic obstructive pulmonary disease: Healthcare costs and beyond. Allergy Asthma Proc. 2015, 36, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Divo, M.J.; Martinez, C.H.; Mannino, D.M. Ageing and the epidemiology of multimorbidity. Eur. Respir. Soc. 2014, 44, 1055–1068. [Google Scholar] [CrossRef] [PubMed]
- Guilleminault, L.; Rolland, Y.; Didier, A. Characteristics of non-pharmacological interventions in the elderly with COPD. Smoking cessation, pulmonary rehabilitation, nutritional management and patient education. Rev. Mal. Respir. 2018, 35, 626–641. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.K.; Berger, S.L.; Brunet, A.; Campisi, J.; Cuervo, A.M.; Epel, E.S.; Franceschi, C.; Lithgow, G.J.; Morimoto, R.I.; Pessin, J.E.; et al. Geroscience: Linking aging to chronic disease. Cell 2014, 159, 709–713. [Google Scholar] [CrossRef]
- Fragoso, C.A.V.; Gill, T.M. Defining chronic obstructive pulmonary disease in an aging population. J. Am. Geriatr. Soc. 2010, 58, 2224. [Google Scholar] [CrossRef]
- Dall, T.M.; Gallo, P.D.; Chakrabarti, R.; West, T.; Semilla, A.P.; Storm, M.V. An aging population and growing disease burden will require alarge and specialized health care workforce by 2025. Health Aff. 2013, 32, 2013–2020. [Google Scholar] [CrossRef]
- Raherison, C.; Girodet, P.-O. Epidemiology of COPD. Eur. Respir. Rev. 2009, 18, 213–221. [Google Scholar] [CrossRef]
- Anecchino, C.; Rossi, E.; Fanizza, C.; De Rosa, M.; Tognoni, G.; Romero, M. Prevalence of chronic obstructive pulmonary disease and pattern of comorbidities in a general population. Int. J. Chronic Obs. Pulm. Dis. 2007, 2, 567–574. [Google Scholar]
- Bellia, M.; Benfante, A.; Menozzi, M.; Augugliaro, G.; Scichilone, N.; Cannizzaro, F.; Midiri, M.; Bellia, V. Validation of lung densitometry threshold at CT for the distinction between senile lung and emphysema in elderly subjects. Monaldi Arch. Chest Dis. 2011, 75, 162–166. [Google Scholar] [CrossRef][Green Version]
- Pistelli, R.; Ferrara, L.; Misuraca, C.; Bustacchini, S. Practical management problems of stable chronic obstructive pulmonary disease in the elderly. Curr. Opin. Pulm. Med. 2011, 17 (Suppl. S1), S43–S48. [Google Scholar] [CrossRef] [PubMed]
- Vestbo, J.; Hurd, S.S.; Agustí, A.G.; Jones, P.W.; Vogelmeier, C.; Anzueto, A.; Barnes, P.J.; Fabbri, L.M.; Martinez, F.J.; Nishimura, M.; et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef] [PubMed]
- Meiners, S.; Eickelberg, O.; Königshoff, M. Hallmarks of the ageing lung. Eur. Respir. J. 2015, 45, 807–827. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Barnes, P.J. COPD as a disease of accelerated lung aging. Chest 2009, 135, 173–180. [Google Scholar] [CrossRef]
- Mercado, N.; Ito, K.; Barnes, P.J. Accelerated ageing of the lung in COPD: New concepts. Thorax 2015, 70, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Chapman, K.; Mannino, D.; Soriano, J.; Vermeire, P.; Buist, A.S.; Thun, M.; Connell, C.; Jemal, A.; Lee, T.; Miravitlles, M.; et al. Epidemiology and costs of chronic obstructive pulmonary disease. Eur. Respir. J. 2006, 27, 188–207. [Google Scholar] [CrossRef]
- Ito, K.; Mercado, N. STOP accelerating lung aging for the treatment of COPD. Exp. Gerontol. 2014, 59, 21–27. [Google Scholar] [CrossRef]
- Hikichi, M.; Mizumura, K.; Maruoka, S.; Gon, Y. Pathogenesis of chronic obstructive pulmonary disease (COPD) induced by cigarette smoke. J. Thorac. Dis. 2019, 11, S2129–S2140. [Google Scholar] [CrossRef]
- Culpitt, S.V.; Rogers, D.F.; Fenwick, P.S.; Shah, P.; De Matos, C.; Russell, R.E.K.; Barnes, P.J.; Donnelly, L.E. Inhibition by red wine extract, resveratrol, of cytokine release by alveolar macrophages in COPD. Thorax 2003, 58, 942–946. [Google Scholar] [CrossRef]
- Hogg, J.C. Pathophysiology of airflow limitation in chronic obstructive pulmonary disease. Lancet 2004, 364, 709–721. [Google Scholar] [CrossRef]
- Sethi, S. Infectious etiology of acute exacerbations of chronic bronchitis. Chest 2000, 117, 380S–385S. [Google Scholar] [CrossRef]
- Marengoni, A.; Rizzuto, D.; Wang, H.X.; Winblad, B.; Fratiglioni, L. Patterns of chronic multimorbidity in the elderly population. J. Am. Geriatr. Soc. 2009, 57, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Soriano, J.B.; Visick, G.T.; Muellerova, H.; Payvandi, N.; Hansell, A.L. Patterns of comorbidities in newly diagnosed COPD and asthma in primary care. Chest 2005, 128, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Pedone, C. Comorbidities of COPD as a function of age: Evidence and practical recommendations. J. Gerontol. Geriatr. 2016, 64, 126–130. [Google Scholar]
- De Miguel Díez, J.; Morgan, J.C.; García, R.J. The association between COPD and heart failure risk: A review. Int. J. Chronic Obstr. Pulm. Dis. 2013, 8, 305. [Google Scholar] [CrossRef] [PubMed]
- Incalzi, R.A.; Corsonello, A.; Pedone, C.; Battaglia, S.; Paglino, G.; Bellia, V. Extrapulmonary Consequences of COPD in the Elderly Study Investigators. S. Chronic renal failure: A neglected comorbidity of COPD. Chest 2010, 137, 831–837. [Google Scholar] [PubMed]
- Maurer, J.; Rebbapragada, V.; Borson, S.; Goldstein, R.; Kunik, M.E.; Yohannes, A.M.; Hanania, N.A. Anxiety and depression in COPD: Current understanding, unanswered questions, and research needs. Chest 2008, 134, 43S–56S. [Google Scholar] [CrossRef] [PubMed]
- Graat-Verboom, L.; Wouters, E.; Smeenk, F.; Van Den Borne, B.; Lunde, R.; Spruit, M. Current status of research on osteoporosis in COPD: A systematic review. Eur. Respir. J. 2009, 34, 209–218. [Google Scholar] [CrossRef]
- Barnes, P.J. Senescence in COPD and Its Comorbidities. Annu. Rev. Physiol. 2017, 79, 517–539. [Google Scholar] [CrossRef]
- Córdoba-Lanús, E.; Cazorla-Rivero, S.; Espinoza-Jiménez, A.; de-Torres, J.P.; Pajares, M.J.; Aguirre-Jaime, A.; Celli, B.; Casanova, C. Telomere shortening and accelerated aging in COPD: Findings from the BODE cohort. Respir. Res. 2017, 18, 59. [Google Scholar] [CrossRef]
- Churg, A.; Zhou, S.; Wright, J.L. Series “matrix metalloproteinases in lung health and disease”: Matrix metalloproteinases in COPD. Eur. Respir. J. 2012, 39, 197–209. [Google Scholar] [CrossRef]
- Shapiro, S.D.; Ingenito, E.P. The pathogenesis of chronic obstructive pulmonary disease: Advances in the past 100 years. Am. J. Respir. Cell Mol. Biol. 2005, 32, 367–372. [Google Scholar] [CrossRef] [PubMed]
- MacNee, W. Is Chronic Obstructive Pulmonary Disease an Accelerated Aging Disease? Ann. Am. Thorac. Soc. 2016, 13 (Suppl. S5), S429–S437. [Google Scholar] [CrossRef] [PubMed]
- Fragoso, C.A.V. Epidemiology of Chronic Obstructive Pulmonary Disease (COPD) in Aging Populations. COPD J. Chronic Obstr. Pulm. Dis. 2016, 13, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Teramoto, S.; Ishii, M. Aging, the aging lung, and senile emphysema are different. Am. J. Respir. Crit. Care Med. 2007, 175, 197–198. [Google Scholar] [CrossRef] [PubMed]
- Janssens, J.-P.; Pache, J.-C.; Nicod, L. Physiological changes in respiratory function associated with ageing. Eur. Respir. J. 1999, 13, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Brandsma, C.-A.; de Vries, M.; Costa, R.; Woldhuis, R.R.; Königshoff, M.; Timens, W. Lung ageing and COPD: Is there a role for ageing in abnormal tissue repair? Eur. Respir. Rev. 2017, 26, 170073. [Google Scholar] [CrossRef]
- Sato, A.; Hirai, T.; Imura, A.; Kita, N.; Iwano, A.; Muro, S.; Nabeshima, Y.; Suki, B.; Mishima, M. Morphological mechanism of the development of pulmonary emphysema in klotho mice. Proc. Natl. Acad. Sci. USA 2007, 104, 2361–2365. [Google Scholar] [CrossRef]
- Teramoto, S.; Fukuchi, Y.; Uejima, Y.; Teramoto, K.; Oka, T.; Orimo, H. A novel model of senile lung: Senescence-accelerated mouse (SAM). Am. J. Respir. Crit. Care Med. 1994, 150, 238–244. [Google Scholar] [CrossRef]
- Schulte, H.; Mühlfeld, C.; Brandenberger, C. Age-Related Structural and Functional Changes in the Mouse Lung. Front. Physiol. 2019, 10, 1466. [Google Scholar] [CrossRef]
- Vandivier, R.W.; Ghosh, M. Understanding the Relevance of the Mouse Cigarette Smoke Model of COPD: Peering through the Smoke. Am. J. Respir. Cell. Mol. Biol. 2017, 57, 3–4. [Google Scholar] [CrossRef]
- Franceschi, C.; Garagnani, P.; Vitale, G.; Capri, M.; Salvioli, S. Inflammaging and ‘Garb-aging’. Trends Endocrinol. Metab. 2017, 28, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Inflammatory mechanisms in patients with chronic obstructive pulmonary disease. J. Allergy Clin. Immunol. 2016, 138, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Garth, J.; Barnes, J.W.; Krick, S. Targeting cytokines as evolving treatment strategies in chronic inflammatory airway diseases. Int. J. Mol. Sci. 2018, 19, 3402. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Capri, M.; Monti, D.; Giunta, S.; Olivieri, F.; Sevini, F.; Panourgia, M.P.; Invidia, L.; Celani, L.; Scurti, M.; et al. Inflammaging and anti-inflammaging: A systemic perspective on aging and longevity emerged from studies in humans. Mech. Ageing Dev. 2007, 128, 92–105. [Google Scholar] [CrossRef]
- Remick, D.G.; Bolgos, G.; Copeland, S.; Siddiqui, J. Role of Interleukin-6 in Mortality from and Physiologic Response to Sepsis. Infect. Immun. 2005, 73, 2751–2757. [Google Scholar] [CrossRef]
- Damas, P.; Ledoux, D.; Nys, M.; Vrindts, Y.; De Groote, D.; Franchimont, P.; Lamy, M. Cytokine serum level during severe sepsis in human IL-6 as a marker of severity. Ann. Surg. 1992, 215, 356. [Google Scholar] [CrossRef]
- Wedzicha, J.A.; Seemungal, T.A.; MacCallum, P.K.; Paul, E.A.; Donaldson, G.C.; Bhowmik, A.; Jeffries, D.J.; Meade, T.W. Acute exacerbations of chronic obstructive pulmonary disease are accompanied by elevations of plasma fibrinogen and serum IL-6 levels. Thromb. Haemost. 2000, 84, 210–215. [Google Scholar]
- Song, W.; Zhao, J.; Li, Z. Interleukin-6 in bronchoalveolar lavage fluid from patients with COPD. Chin. Med. J. 2001, 114, 1140–1142. [Google Scholar]
- Kirkland, J.L.; Tchkonia, T.; Zhu, Y.; Niedernhofer, L.J.; Robbins, P.D. The Clinical Potential of Senolytic Drugs. J. Am. Geriatr. Soc. 2017, 65, 2297–2301. [Google Scholar] [CrossRef]
- Freund, A.; Orjalo, A.V.; Desprez, P.Y.; Campisi, J. Inflammatory networks during cellular senescence: Causes and consequences. Trends Mol. Med. 2010, 16, 238–246. [Google Scholar] [CrossRef]
- Hayflick, L.; Moorhead, P.S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Wang, C.; Jurk, D.; Maddick, M.; Nelson, G.; Martin-Ruiz, C.; Von Zglinicki, T. DNA damage response and cellular senescence in tissues of aging mice. Aging Cell 2009, 8, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Herbig, U.; Ferreira, M.; Condel, L.; Carey, D.; Sedivy, J.M. Cellular senescence in aging primates. Science 2006, 311, 1257. [Google Scholar] [CrossRef] [PubMed]
- Passos, J.F.; Nelson, G.; Wang, C.; Richter, T.; Simillion, C.; Proctor, C.J.; Miwa, S.; Olijslagers, S.; Hallinan, J.; Wipat, A. Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol. Syst. Biol. 2010, 6, 347. [Google Scholar] [CrossRef] [PubMed]
- Chilosi, M.; Carloni, A.; Rossi, A.; Poletti, V. Premature lung aging and cellular senescence in the pathogenesis of idiopathic pulmonary fibrosis and COPD/emphysema. Transl. Res. 2013, 162, 156–173. [Google Scholar] [CrossRef]
- Tuder, R.M.; Kern, J.A.; Miller, Y.E. Senescence in Chronic Obstructive Pulmonary Disease. Proc. Am. Thorac. Soc. 2012, 9, 62–63. [Google Scholar] [CrossRef]
- Signer, R.A.; Morrison, S.J. Mechanisms that regulate stem cell aging and life span. Cell Stem Cell 2013, 12, 152–165. [Google Scholar] [CrossRef]
- Ruzankina, Y.; Asare, A.; Brown, E.J. Replicative stress, stem cells and aging. Mech. Ageing Dev. 2008, 129, 460–466. [Google Scholar] [CrossRef]
- Stripp, B.R.; Reynolds, S.D. Maintenance and repair of the bronchiolar epithelium. Proc. Am. Thorac. Soc. 2008, 5, 328–333. [Google Scholar] [CrossRef]
- Ghosh, M.; Miller, Y.E.; Nakachi, I.; Kwon, J.B.; Barón, A.E.; Brantley, A.E.; Merrick, D.T.; Franklin, W.A.; Keith, R.L.; Vandivier, R.W. Exhaustion of airway basal progenitor cells in early and established chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2018, 197, 885–896. [Google Scholar] [CrossRef]
- Staudt, M.R.; Buro-Auriemma, L.J.; Walters, M.S.; Salit, J.; Vincent, T.; Shaykhiev, R.; Mezey, J.G.; Tilley, A.E.; Kaner, R.J.; Ho, M.W.; et al. Airway Basal stem/progenitor cells have diminished capacity to regenerate airway epithelium in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2014, 190, 955–958. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Unnikrishnan, A.; Deepa, S.S.; Liu, Y.; Li, Y.; Ikeno, Y.; Sosnowska, D.; Van Remmen, H.; Richardson, A. A new role for oxidative stress in aging: The accelerated aging phenotype in Sod1−/− mice is correlated to increased cellular senescence. Redox Biol. 2017, 11, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, P.A.; Barnes, P.J. Oxidative stress in COPD. Chest 2013, 144, 266–273. [Google Scholar] [CrossRef]
- Pérez, V.I.; Bokov, A.; Van Remmen, H.; Mele, J.; Ran, Q.; Ikeno, Y.; Richardson, A. Is the oxidative stress theory of aging dead? Biochim. Biophys. Acta 2009, 1790, 1005–1014. [Google Scholar] [CrossRef] [PubMed]
- Lithgow, G.J.; White, T.M.; Melov, S.; Johnson, T.E. Thermotolerance and extended life-span conferred by single-gene mutations and induced by thermal stress. Proc. Natl. Acad. Sci. USA 1995, 92, 7540–7544. [Google Scholar] [CrossRef]
- Johnson, T.E.; Lithgow, G.J.; Murakami, S. Hypothesis: Interventions that increase the response to stress offer the potential for effective life prolongation and increased health. J. Gerontol. Ser. A Biol. Sci. Med Sci. 1996, 51, B392–B395. [Google Scholar] [CrossRef]
- Murakami, S.; Salmon, A.; Miller, R.A. Multiplex stress resistance in cells from long-lived dwarf mice. FASEB J. 2003, 17, 1565–1576. [Google Scholar] [CrossRef]
- Migliaccio, E.; Giorgio, M.; Mele, S.; Pelicci, G.; Reboldi, P.; Pandolfi, P.P.; Lanfrancone, L.; Pelicci, P.G. The p66 shc adaptor protein controls oxidative stress response and life span in mammals. Nature 1999, 402, 309–313. [Google Scholar] [CrossRef]
- Holzenberger, M.; Dupont, J.; Ducos, B.; Leneuve, P.; Géloën, A.; Even, P.C.; Cervera, P.; Le Bouc, Y. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature 2003, 421, 182–187. [Google Scholar] [CrossRef]
- Boukhenouna, S.; Wilson, M.A.; Bahmed, K.; Kosmider, B. Reactive Oxygen Species in Chronic Obstructive Pulmonary Disease. Oxid. Med. Cell Longev. 2018, 2018, 5730395. [Google Scholar] [CrossRef]
- Aoshiba, K.; Tsuji, T.; Yamaguchi, K.; Itoh, M.; Nakamura, H. The danger signal plus DNA damage two-hit hypothesis for chronic inflammation in COPD. Eur. Respir. J. 2013, 42, 1689–1695. [Google Scholar] [CrossRef] [PubMed]
- Hanania, N.A.; Ambrosino, N.; Calverley, P.; Cazzola, M.; Donner, C.F.; Make, B. Treatments for COPD. Respir. Med. 2005, 99, S28–S40. [Google Scholar] [CrossRef] [PubMed]
- Mulhall, P.; Criner, G. Non-pharmacological treatments for COPD. Respirology 2016, 21, 791–809. [Google Scholar] [CrossRef] [PubMed]
- Thabut, G.; Dauriat, G.; Stern, J.B.; Logeart, D.; Levy, A.; Marrash-Chahla, R.; Mal, H. Pulmonary hemodynamics in advanced COPD candidates for lung volume reduction surgery or lung transplantation. Chest 2005, 127, 1531–1536. [Google Scholar] [CrossRef] [PubMed]
- Stoller, J.K.; Panos, R.J.; Krachman, S.; Doherty, D.E.; Make, B.; Group, Long-term Oxygen Treatment Trial Research Group. Oxygen therapy for patients with COPD: Current evidence and the long-term oxygen treatment trial. Chest 2010, 138, 179–187. [Google Scholar] [CrossRef]
- Willemse, B.W.; ten Hacken, N.H.; Rutgers, B.; Lesman-Leegte, I.G.; Postma, D.S.; Timens, W. Effect of 1-year smoking cessation on airway inflammation in COPD and asymptomatic smokers. Eur. Respir. J. 2005, 26, 835–845. [Google Scholar] [CrossRef]
- van Eerd, E.A.; van der Meer, R.M.; van Schayck, O.C.; Kotz, D. Smoking cessation for people with chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2016, 2016, Cd010744. [Google Scholar] [CrossRef]
- Frishman, W.H.; Mitta, W.; Kupersmith, A.; Ky, T. Nicotine and non-nicotine smoking cessation pharmacotherapies. Cardiol. Rev. 2006, 14, 57–73. [Google Scholar] [CrossRef]
- CADTH. CADTH Rapid Response Reports. In Nicotine Replacement Therapy for Smoking Cessation or Reduction: A Review of the Clinical Evidence; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2014. [Google Scholar]
- Aubin, H.J.; Luquiens, A.; Berlin, I. Pharmacotherapy for smoking cessation: Pharmacological principles and clinical practice. Br. J. Clin. Pharmacol. 2014, 77, 324–336. [Google Scholar] [CrossRef]
- Buels, K.S.; Fryer, A.D. Muscarinic receptor antagonists: Effects on pulmonary function. In Handbook of Experimental Pharmacology; Spinger: Berlin/Heidelberg, Germany, 2012; pp. 317–341. [Google Scholar]
- Lipson, D.A.; Barnacle, H.; Birk, R.; Brealey, N.; Locantore, N.; Lomas, D.A.; Ludwig-Sengpiel, A.; Mohindra, R.; Tabberer, M.; Zhu, C.-Q.; et al. FULFIL Trial: Once-Daily Triple Therapy for Patients with Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2017, 196, 438–446. [Google Scholar] [CrossRef]
- Rossi, A.; Khirani, S.; Cazzola, M. Long-acting β2-agonists (LABA) in chronic obstructive pulmonary disease: Efficacy and safety. Int. J. Chronic Obstr. Pulm. Dis. 2008, 3, 521. [Google Scholar] [CrossRef] [PubMed]
- Singh, D. New combination bronchodilators for chronic obstructive pulmonary disease: Current evidence and future perspectives. Br. J. Clin. Pharmacol. 2015, 79, 695–708. [Google Scholar] [CrossRef]
- Agusti, A.; Fabbri, L.M.; Singh, D.; Vestbo, J.; Celli, B.; Franssen, F.M.; Rabe, K.F.; Papi, A. Inhaled corticosteroids in COPD: Friend or foe? Eur. Respir. J. 2018, 52, 1801219. [Google Scholar] [CrossRef] [PubMed]
- Lipson, D.A.; Barnhart, F.; Brealey, N.; Brooks, J.; Criner, G.J.; Day, N.C.; Dransfield, M.T.; Halpin, D.M.G.; Han, M.K.; Jones, C.E.; et al. Once-Daily Single-Inhaler Triple versus Dual Therapy in Patients with COPD. N. Engl. J. Med. 2018, 378, 1671–1680. [Google Scholar] [CrossRef]
- McDonald, C.F. Oxygen therapy for COPD. J. Thorac. Dis. 2014, 6, 1632–1639. [Google Scholar] [PubMed]
- Reardon, J.; Casaburi, R.; Morgan, M.; Nici, L.; Rochester, C. Pulmonary rehabilitation for COPD. Respir. Med. 2005, 99, S19–S27. [Google Scholar] [CrossRef]
- Patel, N.; DeCamp, M.; Criner, G.J. Lung transplantation and lung volume reduction surgery versus transplantation in chronic obstructive pulmonary disease. Proc. Am. Thorac. Soc. 2008, 5, 447–453. [Google Scholar] [CrossRef]
- Subramanian, M.P.; Meyers, B.F. Bilateral versus single lung transplantation: Are two lungs better than one? J. Thorac. Dis. 2018, 10, 4588–4601. [Google Scholar] [CrossRef]
- National Emphysema Treatment Trial Research Group. A Randomized Trial Comparing Lung-Volume–Reduction Surgery with Medical Therapy for Severe Emphysema. N. Engl. J. Med. 2003, 348, 2059–2073. [Google Scholar] [CrossRef]
- Refaely, Y.; Dransfield, M.; Kramer, M.; Gotfried, M.; Leeds, W.; McLennan, G.; Tewari, S.; Krasna, M.; Criner, G. Biologic lung volume reduction therapy for advanced homogeneous emphysema. Eur. Respir. J. 2010, 36, 20–27. [Google Scholar] [CrossRef]
- Fishman, A.; Fessler, H.; Martinez, F.; McKenna, R.J., Jr.; Naunheim, K.; Piantadosi, S.; Weinmann, G.; Wise, R. Patients at high risk of death after lung-volume-reduction surgery. N. Engl. J. Med. 2001, 345, 1075–1083. [Google Scholar]
- Criner, G.J.; Delage, A.; Voelker, K.; Hogarth, D.K.; Majid, A.; Zgoda, M.; Lazarus, D.R.; Casal, R.; Benzaquen, S.B.; Holladay, R.C.; et al. Improving Lung Function in Severe Heterogenous Emphysema with the Spiration Valve System (EMPROVE). A Multicenter, Open-Label Randomized Controlled Clinical Trial. Am. J. Respir. Crit. Care Med. 2019, 200, 1354–1362. [Google Scholar] [CrossRef] [PubMed]
- Sciurba, F.C.; Criner, G.J.; Strange, C.; Shah, P.L.; Michaud, G.; Connolly, T.A.; Deslée, G.; Tillis, W.P.; Delage, A.; Marquette, C.H.; et al. Effect of Endobronchial Coils vs Usual Care on Exercise Tolerance in Patients With Severe Emphysema: The RENEW Randomized Clinical Trial. JAMA 2016, 315, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- Dransfield, M.T.; Garner, J.L.; Bhatt, S.P.; Slebos, D.J.; Klooster, K.; Sciurba, F.C.; Shah, P.L.; Marchetti, N.T.; Sue, R.D.; Wright, S.; et al. Effect of Zephyr Endobronchial Valves on Dyspnea, Activity Levels, and Quality of Life at One Year. Results from a Randomized Clinical Trial. Ann. Am. Thorac. Soc. 2020, 17, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Knutson, M.D.; Leeuwenburgh, C. Resveratrol and novel potent activators of Sirt1: Effects on aging and age-related diseases. Nutr. Rev. 2008, 66, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I. Antioxidant therapies in COPD. Int. J. Chronic Obs. Pulm. Dis. 2006, 1, 15–29. [Google Scholar] [CrossRef]
- Zou, S.; Carey, J.R.; Liedo, P.; Ingram, D.K.; Müller, H.G.; Wang, J.L.; Yao, F.; Yu, B.; Zhou, A. The prolongevity effect of resveratrol depends on dietary composition and calorie intake in a tephritid fruit fly. Exp. Gerontol. 2009, 44, 472–476. [Google Scholar] [CrossRef]
- Tatar, M. Diet restriction in Drosophila melanogaster. Design and analysis. Interdiscip. Top. Gerontol. 2007, 35, 115–136. [Google Scholar]
- Bo, S.; Ciccone, G.; Castiglione, A.; Gambino, R.; De Michieli, F.; Villois, P.; Durazzo, M.; Cavallo-Perin, P.; Cassader, M. Anti-inflammatory and antioxidant effects of resveratrol in healthy smokers a randomized, double-blind, placebo-controlled, cross-over trial. Curr. Med. Chem. 2013, 20, 1323–1331. [Google Scholar] [CrossRef]
- Beijers, R.; Gosker, H.R.; Schols, A. Resveratrol for patients with chronic obstructive pulmonary disease: Hype or hope? Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 138–144. [Google Scholar] [CrossRef]
- Wang, X.-L.; Li, T.; Li, J.-H.; Miao, S.-Y.; Xiao, X.-Z. The effects of resveratrol on inflammation and oxidative stress in a rat model of chronic obstructive pulmonary disease. Molecules 2017, 22, 1529. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Colley, T.; Mercado, N. Geroprotectors as a novel therapeutic strategy for COPD, an accelerating aging disease. Int. J. Chronic Obstr. Pulm. Dis. 2012, 7, 641. [Google Scholar] [CrossRef]
- Ghosh, H.S. The anti-aging, metabolism potential of SIRT1. Curr. Opin. Investig. Drugs 2008, 9, 1095–1102. [Google Scholar] [PubMed]
- Grabowska, W.; Sikora, E.; Bielak-Zmijewska, A. Sirtuins, a promising target in slowing down the ageing process. Biogerontology 2017, 18, 447–476. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yang, X.; Zhang, W.; Peng, D.; Xia, Y.; Lu, Y.; Han, X.; Song, G.; Zhu, J.; Liu, R. Therapeutic effects of resveratrol in a mouse model of LPS and cigarette smoke-induced COPD. Inflammation 2016, 39, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Berstein, L.M.; Popovich, I.G.; Zabezhinski, M.A.; Egormin, P.A.; Piskunova, T.S.; Semenchenko, A.V.; Tyndyk, M.L.; Yurova, M.N.; Kovalenko, I.G.; et al. If started early in life, metformin treatment increases life span and postpones tumors in female SHR mice. Aging 2011, 3, 148–157. [Google Scholar] [CrossRef]
- Rena, G.; Hardie, D.G.; Pearson, E.R. The mechanisms of action of metformin. Diabetologia 2017, 60, 1577–1585. [Google Scholar] [CrossRef]
- Piskovatska, V.; Stefanyshyn, N.; Storey, K.B.; Vaiserman, A.M.; Lushchak, O. Metformin as a geroprotector: Experimental and clinical evidence. Biogerontology 2019, 20, 33–48. [Google Scholar] [CrossRef]
- Ho, T.W.; Huang, C.T.; Ruan, S.Y.; Tsai, Y.J.; Lai, F.; Yu, C.J. Diabetes mellitus in patients with chronic obstructive pulmonary disease-The impact on mortality. PLoS ONE 2017, 12, e0175794. [Google Scholar] [CrossRef]
- Hitchings, A.W.; Lai, D.; Jones, P.W.; Baker, E.H. Metformin in severe exacerbations of chronic obstructive pulmonary disease: A randomised controlled trial. Thorax 2016, 71, 587–593. [Google Scholar] [CrossRef]
- Bishwakarma, R.; Zhang, W.; Lin, Y.-L.; Kuo, Y.-F.; Cardenas, V.J.; Sharma, G. Metformin use and health care utilization in patients with coexisting chronic obstructive pulmonary disease and diabetes mellitus. Int. J. Chronic Obstr. Pulm. Dis. 2018, 13, 793. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.-X.; Poeggeler, B.; Menendez-Pelaez, A.; Chen, L.-D.; Saarela, S. Melatonin As a Free Radical Scavenger: Implications for Aging and Age-Related Diseasesa. Ann. N. Y. Acad. Sci. 1994, 719, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Poeggeler, B.; Thuermann, S.; Dose, A.; Schoenke, M.; Burkhardt, S.; Hardeland, R. Melatonin’s unique radical scavenging properties—Roles of its functional substituents as revealed by a comparison with its structural analogs. J. Pineal Res. 2002, 33, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Popovich, I.G.; Zabezhinski, M.A.; Anisimov, S.V.; Vesnushkin, G.M.; Vinogradova, I.A. Melatonin as antioxidant, geroprotector and anticarcinogen. Biochim. Biophys. Acta 2006, 1757, 573–589. [Google Scholar] [CrossRef]
- Anisimov, V.N.; Alimova, I.N.; Baturin, D.A.; Popovich, I.G.; Zabezhinski, M.A.; Rosenfeld, S.V.; Manton, K.G.; Semenchenko, A.V.; Yashin, A.I. Dose-dependent effect of melatonin on life span and spontaneous tumor incidence in female SHR mice. Exp. Gerontol. 2003, 38, 449–461. [Google Scholar] [CrossRef]
- Bonilla, E.; Medina-Leendertz, S.; Díaz, S. Extension of life span and stress resistance of Drosophila melanogaster by long-term supplementation with melatonin. Exp. Gerontol. 2002, 37, 629–638. [Google Scholar] [CrossRef]
- Gumral, N.; Nazıroglu, M.; Ongel, K.; Beydilli, E.D.; Ozguner, F.; Sutcu, R.; Calıskan, S.; Akkaya, A. Antioxidant enzymes and melatonin levels in patients with bronchial asthma and chronic obstructive pulmonary disease during stable and exacerbation periods. Cell Biochem. Funct. 2009, 27, 276–283. [Google Scholar] [CrossRef]
- de Matos Cavalcante, A.G.; de Bruin, P.F.C.; de Bruin, V.M.S.; Nunes, D.M.; Pereira, E.D.B.; Cavalcante, M.M.; Andrade, G.M. Melatonin reduces lung oxidative stress in patients with chronic obstructive pulmonary disease: A randomized, double-blind, placebo-controlled study. J. Pineal Res. 2012, 53, 238–244. [Google Scholar] [CrossRef]
- Kesimer, M.; Ford, A.A.; Ceppe, A.; Radicioni, G.; Cao, R.; Davis, C.W.; Doerschuk, C.M.; Alexis, N.E.; Anderson, W.H.; Henderson, A.G.; et al. Airway Mucin Concentration as a Marker of Chronic Bronchitis. N. Engl. J. Med. 2017, 377, 911–922. [Google Scholar] [CrossRef]
- Shin, I.-S.; Park, J.-W.; Shin, N.-R.; Jeon, C.-M.; Kwon, O.-K.; Lee, M.-Y.; Kim, H.-S.; Kim, J.-C.; Oh, S.-R.; Ahn, K.-S. Melatonin inhibits MUC5AC production via suppression of MAPK signaling in human airway epithelial cells. J. Pineal Res. 2014, 56, 398–407. [Google Scholar] [CrossRef]
- Shin, N.-R.; Park, J.-W.; Lee, I.-C.; Ko, J.-W.; Park, S.-H.; Kim, J.-S.; Kim, J.-C.; Ahn, K.-S.; Shin, I.-S. Melatonin suppresses fibrotic responses induced by cigarette smoke via downregulation of TGF-β1. Oncotarget 2017, 8, 95692–95703. [Google Scholar] [CrossRef] [PubMed]
- Kuro-o, M.; Matsumura, Y.; Aizawa, H.; Kawaguchi, H.; Suga, T.; Utsugi, T.; Ohyama, Y.; Kurabayashi, M.; Kaname, T.; Kume, E.; et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 1997, 390, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Kurosu, H.; Yamamoto, M.; Clark, J.D.; Pastor, J.V.; Nandi, A.; Gurnani, P.; McGuinness, O.P.; Chikuda, H.; Yamaguchi, M.; Kawaguchi, H.; et al. Suppression of aging in mice by the hormone Klotho. Science 2005, 309, 1829–1833. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.H.; Kuro, O.M.; Chen, C.H.; Sue, Y.M.; Chen, Y.C.; Wu, H.H.; Cheng, C.Y. The secreted Klotho protein restores phosphate retention and suppresses accelerated aging in Klotho mutant mice. Eur. J. Pharmacol. 2013, 698, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Sun, Z. Molecular basis of Klotho: From gene to function in aging. Endocr. Rev. 2015, 36, 174–193. [Google Scholar] [CrossRef]
- Torbus-Paluszczak, M.; Bartman, W.; Adamczyk-Sowa, M. Klotho protein in neurodegenerative disorders. Neurol. Sci. 2018, 39, 1677–1682. [Google Scholar] [CrossRef]
- Erben, R.G. Update on FGF23 and Klotho signaling. Mol. Cell Endocrinol. 2016, 432, 56–65. [Google Scholar] [CrossRef]
- Grabner, A.; Amaral, A.P.; Schramm, K.; Singh, S.; Sloan, A.; Yanucil, C.; Li, J.; Shehadeh, L.A.; Hare, J.M.; David, V.; et al. Activation of Cardiac Fibroblast Growth Factor Receptor 4 Causes Left Ventricular Hypertrophy. Cell Metab. 2015, 22, 1020–1032. [Google Scholar] [CrossRef]
- Lu, X.; Hu, M.C. Klotho/FGF23 Axis in Chronic Kidney Disease and Cardiovascular Disease. Kidney Dis. 2017, 3, 15–23. [Google Scholar] [CrossRef]
- Krick, S.; Grabner, A.; Baumlin, N.; Yanucil, C.; Helton, S.; Grosche, A.; Sailland, J.; Geraghty, P.; Viera, L.; Russell, D.W.; et al. Fibroblast growth factor 23 and Klotho contribute to airway inflammation. Eur. Respir. J. 2018, 52, 1800236. [Google Scholar] [CrossRef]
- Singh, S.; Grabner, A.; Yanucil, C.; Schramm, K.; Czaya, B.; Krick, S.; Czaja, M.J.; Bartz, R.; Abraham, R.; Di Marco, G.S.; et al. Fibroblast growth factor 23 directly targets hepatocytes to promote inflammation in chronic kidney disease. Kidney Int. 2016, 90, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Krick, S.; Baumlin, N.; Aller, S.P.; Aguiar, C.; Grabner, A.; Sailland, J.; Mendes, E.; Schmid, A.; Qi, L.; David, N.V.; et al. Klotho Inhibits Interleukin-8 Secretion from Cystic Fibrosis Airway Epithelia. Sci. Rep. 2017, 7, 14388. [Google Scholar] [CrossRef] [PubMed]
- Barnes, J.W.; Duncan, D.; Helton, S.; Hutcheson, S.; Kurundkar, D.; Logsdon, N.J.; Locy, M.; Garth, J.; Denson, R.; Farver, C.; et al. Role of fibroblast growth factor 23 and klotho cross talk in idiopathic pulmonary fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 317, L141–L154. [Google Scholar] [CrossRef] [PubMed]
- Gulati, S.; Wells, J.M.; Urdaneta, G.P.; Balestrini, K.; Vital, I.; Tovar, K.; Barnes, J.W.; Bhatt, S.P.; Campos, M.; Krick, S. Fibroblast Growth Factor 23 is Associated with a Frequent Exacerbator Phenotype in COPD: A Cross-Sectional Pilot Study. Int. J. Mol. Sci. 2019, 20, 2292. [Google Scholar] [CrossRef] [PubMed]
- Garth, J.; Easter, M.; Skylar Harris, E.; Sailland, J.; Kuenzi, L.; Chung, S.; Dennis, J.S.; Baumlin, N.; Adewale, A.T.; Rowe, S.M.; et al. The Effects of the Anti-aging Protein Klotho on Mucociliary Clearance. Front. Med. (Lausanne) 2019, 6, 339. [Google Scholar] [CrossRef] [PubMed]
- Doi, S.; Zou, Y.; Togao, O.; Pastor, J.V.; John, G.B.; Wang, L.; Shiizaki, K.; Gotschall, R.; Schiavi, S.; Yorioka, N.; et al. Klotho inhibits transforming growth factor-beta1 (TGF-beta1) signaling and suppresses renal fibrosis and cancer metastasis in mice. J. Biol. Chem. 2011, 286, 8655–8665. [Google Scholar] [CrossRef]
- Raja, A.; Park, I.; Haq, F.; Ahn, S.-M. FGF19-FGFR4 Signaling in Hepatocellular Carcinoma. Cells 2019, 8, 536. [Google Scholar] [CrossRef]
- Choy, E.; Isenberg, D.; Garrood, T.; Farrow, S.; Ioannou, Y.; Bird, H.; Cheung, N.; Williams, B.; Hazleman, B.; Price, R.; et al. Therapeutic benefit of blocking interleukin-6 activity with an anti–interleukin-6 receptor monoclonal antibody in rheumatoid arthritis: A randomized, double-blind, placebo-controlled, dose-escalation trial. Arthritis Rheum. 2002, 46, 3143–3150. [Google Scholar] [CrossRef]
- Saha, S.; Brightling, C.E. Eosinophilic airway inflammation in COPD. Int. J. Chronic Obs. Pulm. Dis. 2006, 1, 39–47. [Google Scholar] [CrossRef]
- Narendra, D.K.; Hanania, N.A. Targeting IL-5 in COPD. Int. J. Chronic Obs. Pulm. Dis. 2019, 14, 1045–1051. [Google Scholar] [CrossRef]
- Singh, D.; Kolsum, U.; Brightling, C.E.; Locantore, N.; Agusti, A.; Tal-Singer, R. Eosinophilic inflammation in COPD: Prevalence and clinical characteristics. Eur. Respir. J. 2014, 44, 1697–1700. [Google Scholar] [CrossRef] [PubMed]
- Zeiger, R.S.; Schatz, M.; Li, Q.; Chen, W.; Khatry, D.B.; Gossage, D.; Tran, T.N. High Blood Eosinophil Count Is a Risk Factor for Future Asthma Exacerbations in Adult Persistent Asthma. J. Allergy Clin. Immunol. Pract. 2014, 2, 741–750. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.N.; Khatry, D.B.; Ke, X.; Ward, C.K.; Gossage, D. High blood eosinophil count is associated with more frequent asthma attacks in asthma patients. Ann. Allergy Asthma Immunol. 2014, 113, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Fala, L. Nucala (Mepolizumab): First IL-5 Antagonist Monoclonal Antibody FDA Approved for Maintenance Treatment of Patients with Severe Asthma. Am. Health Drug Benefits 2016, 9, 106–110. [Google Scholar]
- Hom, S.; Pisano, M. Reslizumab (Cinqair): An Interleukin-5 Antagonist for Severe Asthma of the Eosinophilic Phenotype. Pharm. Ther. 2017, 42, 564–568. [Google Scholar]
- Mathur, S.K.; Schwantes, E.A.; Jarjour, N.N.; Busse, W.W. Age-related changes in eosinophil function in human subjects. Chest 2008, 133, 412–419. [Google Scholar] [CrossRef]
- Busse, P.J.; Zhang, T.F.; Srivastava, K.; Schofield, B.; Li, X.M. Effect of ageing on pulmonary inflammation, airway hyperresponsiveness and T and B cell responses in antigen-sensitized and -challenged mice. Clin. Exp. Allergy 2007, 37, 1392–1403. [Google Scholar] [CrossRef] [PubMed]
- Cazzola, M.; Matera, M.G.; Rogliani, P.; Calzetta, L. Senolytic drugs in respiratory medicine: Is it an appropriate therapeutic approach? Expert Opin. Investig. Drugs 2018, 27, 573–581. [Google Scholar] [CrossRef]
- Baker, D.J.; Wijshake, T.; Tchkonia, T.; LeBrasseur, N.K.; Childs, B.G.; van de Sluis, B.; Kirkland, J.L.; van Deursen, J.M. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 2011, 479, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Tchkonia, T.; Pirtskhalava, T.; Gower, A.C.; Ding, H.; Giorgadze, N.; Palmer, A.K.; Ikeno, Y.; Hubbard, G.B.; Lenburg, M.; et al. The Achilles’ heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell 2015, 14, 644–658. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.R.; Donnelly, L.E.; Barnes, P.J. Senotherapy: A New Horizon for COPD Therapy. Chest 2020, 158, 562–570. [Google Scholar] [CrossRef]
- Roos, C.M.; Zhang, B.; Palmer, A.K.; Ogrodnik, M.B.; Pirtskhalava, T.; Thalji, N.M.; Hagler, M.; Jurk, D.; Smith, L.A.; Casaclang-Verzosa, G.; et al. Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell 2016, 15, 973–977. [Google Scholar] [CrossRef] [PubMed]
- Justice, J.N.; Nambiar, A.M.; Tchkonia, T.; LeBrasseur, N.K.; Pascual, R.; Hashmi, S.K.; Prata, L.; Masternak, M.M.; Kritchevsky, S.B.; Musi, N.; et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine 2019, 40, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Hickson, L.J.; Prata, L.G.L.; Bobart, S.A.; Evans, T.K.; Giorgadze, N.; Hashmi, S.K.; Herrmann, S.M.; Jensen, M.D.; Jia, Q.; Jordan, K.L.; et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine 2019, 47, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, A.; Herranz, N.; Sun, B.; Wagner, V.; Gallage, S.; Guiho, R.; Wolter, K.; Pombo, J.; Irvine, E.E.; Innes, A.J.; et al. Cardiac glycosides are broad-spectrum senolytics. Nat. Metab. 2019, 1, 1074–1088. [Google Scholar] [CrossRef]
- Broekman, W.; Khedoe, P.P.; Schepers, K.; Roelofs, H.; Stolk, J.; Hiemstra, P.S. Mesenchymal stromal cells: A novel therapy for the treatment of chronic obstructive pulmonary disease? Thorax 2018, 73, 565–574. [Google Scholar] [CrossRef]
- Liu, X.; Fang, Q.; Kim, H. Preclinical studies of mesenchymal stem cell (MSC) administration in chronic obstructive pulmonary disease (COPD): A systematic review and meta-analysis. PLoS ONE 2016, 11, e0157099. [Google Scholar] [CrossRef]
- Zhang, W.-G.; He, L.; Shi, X.-M.; Wu, S.-S.; Zhang, B.; Mei, L.; Xu, Y.-J.; Zhang, Z.-X.; Zhao, J.-P.; Zhang, H.-L. Regulation of transplanted mesenchymal stem cells by the lung progenitor niche in rats with chronic obstructive pulmonary disease. Respir. Res. 2014, 15, 33. [Google Scholar] [CrossRef]
- Gu, W.; Song, L.; Li, X.-M.; Wang, D.; Guo, X.-J.; Xu, W.-G. Mesenchymal stem cells alleviate airway inflammation and emphysema in COPD through down-regulation of cyclooxygenase-2 via p38 and ERK MAPK pathways. Sci. Rep. 2015, 5, 8733. [Google Scholar] [CrossRef]
- Zhen, G.; Xue, Z.; Zhao, J.; Gu, N.; Tang, Z.; Xu, Y.; Zhang, Z. Mesenchymal stem cell transplantation increases expression of vascular endothelial growth factor in papain-induced emphysematous lungs and inhibits apoptosis of lung cells. Cytotherapy 2010, 12, 605–614. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Kim, J.-Y.; Huh, J.W.; Lee, S.W.; Choi, S.J.; Oh, Y.-M. The therapeutic effects of optimal dose of mesenchymal stem cells in a murine model of an elastase induced-emphysema. Tuberc. Respir. Dis. 2015, 78, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.J.; Song, L.; Han, F.F.; Cui, Z.L.; Chen, X.; Guo, X.J.; Xu, W.G. Mesenchymal stem cells protect cigarette smoke-damaged lung and pulmonary function partly via VEGF–VEGF receptors. J. Cell. Biochem. 2013, 114, 323–335. [Google Scholar] [CrossRef] [PubMed]
- Weiss, D.J. Cell-based Therapy for Chronic Obstructive Pulmonary Disease. Rebuilding the Lung. Ann. Am. Thorac. Soc. 2018, 15 (Suppl. S4), S253–S259. [Google Scholar] [CrossRef] [PubMed]
- Wagner, D.E.; Bonvillain, R.W.; Jensen, T.; Girard, E.D.; Bunnell, B.A.; Finck, C.M.; Hoffman, A.M.; Weiss, D.J. Can stem cells be used to generate new lungs? Ex vivo lung bioengineering with decellularized whole lung scaffolds. Respirology 2013, 18, 895–911. [Google Scholar] [CrossRef] [PubMed]
- Horváth, L.; Umehara, Y.; Jud, C.; Blank, F.; Petri-Fink, A.; Rothen-Rutishauser, B. Engineering an in vitro air-blood barrier by 3D bioprinting. Sci. Rep. 2015, 5, 7974. [Google Scholar] [CrossRef] [PubMed]
- Beeh, K.M.; Beier, J.; Koppenhoefer, N.; Buhl, R. Increased glutathione disulfide and nitrosothiols in sputum supernatant of patients with stable COPD. Chest 2004, 126, 1116–1122. [Google Scholar] [CrossRef]
- Ciofu, O.; Smith, S.; Lykkesfeldt, J. Antioxidant supplementation for lung disease in cystic fibrosis. Cochrane Database Syst. Rev. 2019, 10, Cd007020. [Google Scholar] [CrossRef]
- Griese, M.; Kappler, M.; Eismann, C.; Ballmann, M.; Junge, S.; Rietschel, E.; van Koningsbruggen-Rietschel, S.; Staab, D.; Rolinck-Werninghaus, C.; Mellies, U.; et al. Inhalation treatment with glutathione in patients with cystic fibrosis. A randomized clinical trial. Am. J. Respir. Crit. Care Med. 2013, 188, 83–89. [Google Scholar] [CrossRef]
- Lamson, D.W.; Brignall, M.S. The use of nebulized glutathione in the treatment of emphysema: A case report. Altern. Med. Rev. 2000, 5, 429–431. [Google Scholar]
- Dal Negro, R.W.; Wedzicha, J.A.; Iversen, M.; Fontana, G.; Page, C.; Cicero, A.F.; Pozzi, E.; Calverley, P.M. Effect of erdosteine on the rate and duration of COPD exacerbations: The RESTORE study. Eur. Respir. J. 2017, 50, 1700711. [Google Scholar] [CrossRef]
- Moretti, M.; Bottrighi, P.; Dallari, R.; Da Porto, R.; Dolcetti, A.; Grandi, P.; Garuti, G.; Guffanti, E.; Roversi, P.; De Gugliemo, M.; et al. The effect of long-term treatment with erdosteine on chronic obstructive pulmonary disease: The EQUALIFE Study. Drugs Exp. Clin. Res. 2004, 30, 143–152. [Google Scholar] [PubMed]
- Meyer, K.C. Lung infections and aging. Ageing Res. Rev. 2004, 3, 55–67. [Google Scholar] [CrossRef] [PubMed]
| Senolytics | Target | Model |
|---|---|---|
| Dasatinib | Tyrosine Kinases | Phase 2 Clincal Trial |
| Quercetin | Tyrosine Kinases | Phase 2 Clincal Trial |
| Navitovlax | BCL–2 Family | Animal Models |
| Cardiag Glycosides | BCL–2 Family | Animal Models |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Easter, M.; Bollenbecker, S.; Barnes, J.W.; Krick, S. Targeting Aging Pathways in Chronic Obstructive Pulmonary Disease. Int. J. Mol. Sci. 2020, 21, 6924. https://doi.org/10.3390/ijms21186924
Easter M, Bollenbecker S, Barnes JW, Krick S. Targeting Aging Pathways in Chronic Obstructive Pulmonary Disease. International Journal of Molecular Sciences. 2020; 21(18):6924. https://doi.org/10.3390/ijms21186924
Chicago/Turabian StyleEaster, Molly, Seth Bollenbecker, Jarrod W. Barnes, and Stefanie Krick. 2020. "Targeting Aging Pathways in Chronic Obstructive Pulmonary Disease" International Journal of Molecular Sciences 21, no. 18: 6924. https://doi.org/10.3390/ijms21186924
APA StyleEaster, M., Bollenbecker, S., Barnes, J. W., & Krick, S. (2020). Targeting Aging Pathways in Chronic Obstructive Pulmonary Disease. International Journal of Molecular Sciences, 21(18), 6924. https://doi.org/10.3390/ijms21186924






