Towards the Development of AgoKirs: New Pharmacological Activators to Study Kir2.x Channel and Target Cardiac Disease
Abstract
:1. Kir2.x Expression, Structure and Rectification
2. Kir2.x Disease Relationships
3. AgoKirs, Agonists of Kir2.x Function
3.1. Indirect Activators
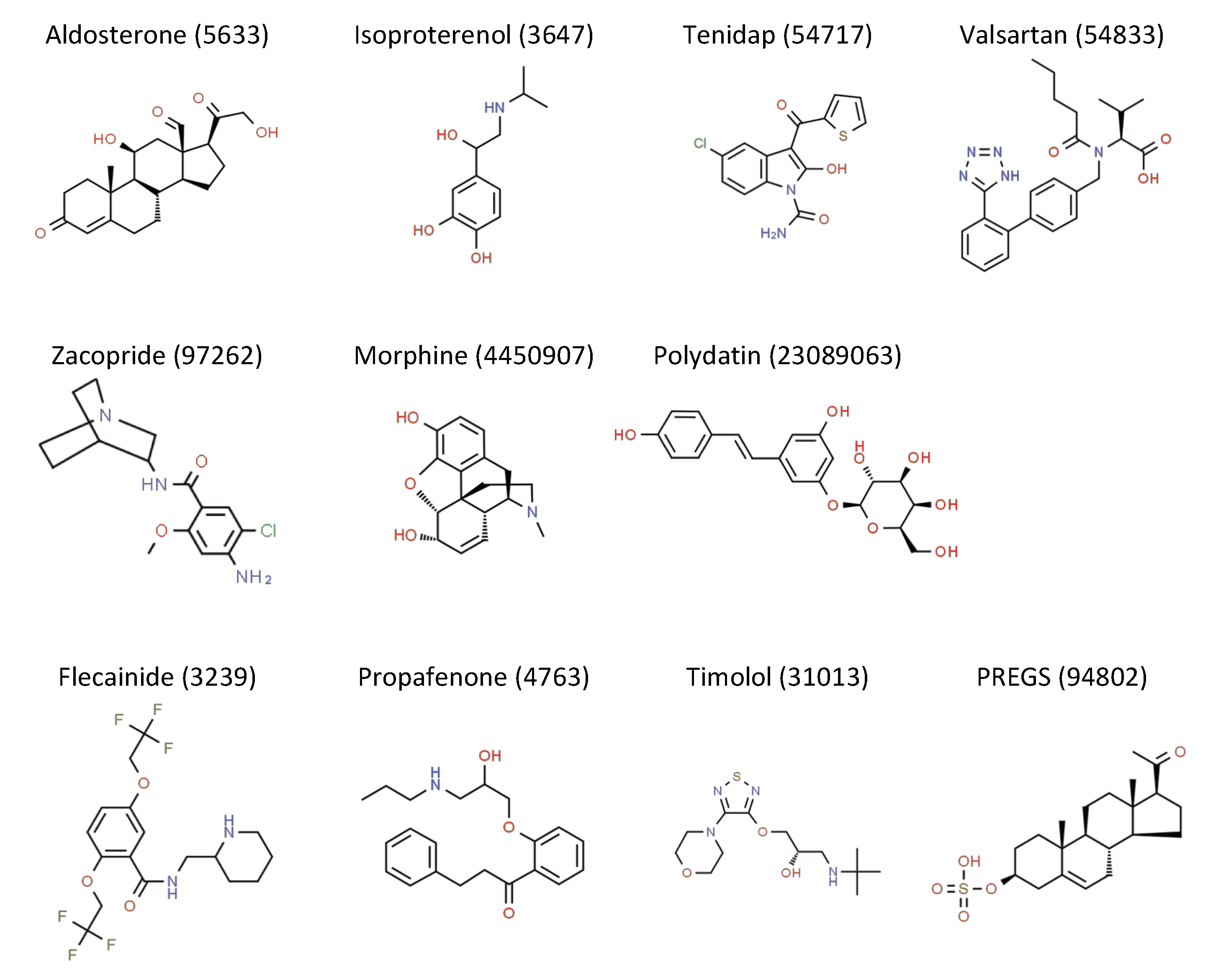
3.1.1. Aldosterone
3.1.2. Isoproterenol
3.1.3. Tenidap
3.1.4. Valsartan
3.1.5. Zacopride
3.2. Direct Activators
3.2.1. Flecainide
3.2.2. Propafenone
3.2.3. Timolol
3.2.4. Pregnenolone Sulfate
3.3. Unknown Mechanism of Activation
3.3.1. LPS (lipopolysaccharides)
3.3.2. Morphine
3.3.3. Polydatin
4. Lead Compounds and Clinical Perspective
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AF | Atrial Fibrillation |
| AgoKir | Agonist of Kir channel |
| AS | Andersen Syndrome |
| CFTR | Cystic Fibrosis Transmembrane Conductance Regulator |
| CHO | Chinese Hamster Ovary |
| EK | Potassium equilibrium potential |
| HEK | Human Embryonal Kidney |
| HF | Heart Failure |
| IK1 | Inward rectifier current |
| IKATP | ATP regulated inward rectifier current |
| ISO | Isoproterenol/isoprenaline |
| Kir2.x | Isoform x of the inward rectifier protein Kir2 family |
| LPS | Lipopolysacharides |
| MI | Myocardial Infarction |
| MR | Mineral corticoid Receptor |
| PD | Polydatin |
| PKA | Protein Kinase A |
| PKC | Protein Kinase C |
| PREGS | Pregnenolone Sulfate |
| RAAS | Renin-Angiotensin-Aldosterone-System |
| Vm | Membrane potential |
| ZAC | Zacopride |
References
- Hibino, H.; Inanobe, A.; Furutani, K.; Murakami, S.; Findlay, I.; Kurachi, Y. Inwardly rectifying potassium channels: Their structure, function, and physiological roles. Physiol. Rev. 2010, 90, 291–366. [Google Scholar] [CrossRef] [Green Version]
- Van der Heyden, M.A.G.; Jespersen, T. Pharmacological exploration of the resting membrane potential reserve: Impact on atrial fibrillation. Eur. J. Pharmacol. 2016, 771, 56–64. [Google Scholar] [CrossRef]
- Pini, J.; Rouleau, M.; Desnuelle, C.; Sacconi, S.; Bendahhou, S. Modeling Andersen’s Syndrome in Human Induced Pluripotent Stem Cells. Stem Cells Dev. 2016, 25, 151–159. [Google Scholar] [CrossRef]
- De Boer, T.P.; Houtman, M.J.C.; Compier, M.; Van der Heyden, M.A.G. The mammalian KIR2.x inward rectifier ion channel family: Expression pattern and pathophysiology. Acta Physiol. 2010, 199, 243–256. [Google Scholar] [CrossRef]
- Anumonwo, J.M.B.; Lopatin, A.N. Cardiac strong inward rectifier potassium channels. J. Mol. Cell. Cardiol. 2010, 48, 45–54. [Google Scholar] [CrossRef] [Green Version]
- Glowatzki, E.; Fakler, G.; Brändle, U.; Rexhausen, U.; Zenner, H.P.; Ruppersberg, J.P.; Fakler, B. Subunit-dependent assembly of inward-rectifier K+ channels. Proc. R. Soc. B Biol. Sci. 1995, 261, 251–261. [Google Scholar] [CrossRef]
- Preisig-Müller, R.; Schlichthörl, G.; Goerge, T.; Heinen, S.; Brüggemann, A.; Rajan, S.; Derst, C.; Veh, R.W.; Daut, J. Heteromerization of Kir2.x potassium channels contributes to the phenotype of Andersen’s syndrome. Proc. Natl. Acad. Sci. USA 2002, 99, 7774–7779. [Google Scholar] [CrossRef] [Green Version]
- Nichols, C.G.; Lee, S.J. Polyamines and potassium channels: A 25-year romance. J. Biol. Chem. 2018, 293, 18779–18788. [Google Scholar] [CrossRef] [Green Version]
- Bichet, D.; Haass, F.A.; Jan, L.Y. Merging functional studies with structures of inward-rectifier K+ channels. Nat. Rev. Neurosci. 2003, 4, 957–967. [Google Scholar] [CrossRef]
- Ji, Y.; Varkevisser, R.; Opacic, D.; Bossu, A.; Kuiper, M.; Beekman, J.D.M.; Yang, S.; Khan, A.P.; Dobrev, D.; Voigt, N.; et al. The inward rectifier current inhibitor PA-6 terminates atrial fibrillation and does not cause ventricular arrhythmias in goat and dog models. Br. J. Pharmacol. 2017, 174, 2576–2590. [Google Scholar] [CrossRef] [Green Version]
- Szatmári, V.; Ji, Y.; Herwijnen, B.V.; Feng, M.; Wang, M.Z.; Bossu, A.; Van der Heyden, M.A.G. Efficacy of pentamidine analogue 6 in dogs with chronic atrial fibrillation. J. Vet. Intern. Med. 2018, 32, 1549–1554. [Google Scholar] [CrossRef]
- Van der Heyden, M.A.G.; Sánchez-Chapula, J.A. Toward specific cardiac I(K1) modulators for in vivo application: Old drugs point the way. Heart Rhythm 2011, 8, 1076–1080. [Google Scholar] [CrossRef]
- Priori, S.G.; Pandit, S.V.; Rivolta, I.; Berenfeld, O.; Ronchetti, E.; Dhamoon, A.; Napolitano, C.; Anumonwo, J.; di Barletta, M.R.; Gudapakkam, S.; et al. A novel form of short QT syndrome (SQT3) is caused by a mutation in the KCNJ2 gene. Circ. Res. 2005, 96, 800–807. [Google Scholar] [CrossRef] [Green Version]
- Xia, M.; Jin, Q.; Bendahhou, S.; He, Y.; Larroque, M.M.; Chen, Y.; Zhou, Q.; Yang, Y.; Liu, Y.; Liu, B.; et al. A Kir2.1 gain-of-function mutation underlies familial atrial fibrillation. Biochem. Biophys. Res. Commun. 2005, 332, 1012–1019. [Google Scholar] [CrossRef]
- Veldhuis, M.G.; Ji, Y.; van der Heyden, M.A.G. A Little Too Much: Cardiac Electrophysiological Effects of Elevated Inward Rectifying Current Carried by the KIR2.1 Ion Channel Protein. Adapt. Med. 2015, 7, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Tristani-Firouzi, M.; Jensen, J.L.; Donaldson, M.R.; Sansone, V.; Meola, G.; Hahn, A.; Bendahhou, S.; Kwiecinski, H.; Fidzianska, A.; Plaster, N.; et al. Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J. Clin. Investig. 2002, 110, 381–388. [Google Scholar] [CrossRef]
- Andersen, E.D.; Krasilnikoff, P.A.; Overvad, H. Intermittent muscular weakness, extrasystoles, and multiple developmental anomalies: A new syndrome? Acta Paediatr. 1971, 60, 559–564. [Google Scholar] [CrossRef]
- Bosch, R.F.; Zeng, X.; Grammer, J.B.; Popovic, K.; Mewis, C.; Kühlkamp, V. Ionic mechanisms of electrical remodeling in human atrial fibrillation. Cardiovasc. Res. 1999, 44, 121–131. [Google Scholar] [CrossRef]
- Dobrev, D.; Graf, E.; Wettwer, E.; Himmel, H.M.; Hála, O.; Doerfel, C.; Christ, T.; Schüler, S.; Ravens, U. Molecular basis of downregulation of G-protein-coupled inward rectifying K(+) current (I(K,ACh) in chronic human atrial fibrillation: Decrease in GIRK4 mRNA correlates with reduced I(K,ACh) and muscarinic receptor-mediated shortening of action potentials. Circulation 2001, 104, 2551–2557. [Google Scholar] [CrossRef] [Green Version]
- Dobrev, D.; Wettwer, E.; Kortner, A.; Knaut, M.; Schüler, S.; Ravens, U. Human inward rectifier potassium channels in chronic and postoperative atrial fibrillation. Cardiovasc. Res. 2002, 54, 397–404. [Google Scholar] [CrossRef] [Green Version]
- Kääb, S.; Nuss, H.B.; Chiamvimonvat, N.; O’Rourke, B.; Pak, P.H.; Kass, D.A.; Marban, E.; Tomaselli, G.F. Ionic mechanism of action potential prolongation in ventricular myocytes from dogs with pacing-induced heart failure. Circ. Res. 1996, 78, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.; Armoundas, A.A.; Tian, Y.; DiSilvestre, D.; Burysek, M.; Halperin, V.; O’Rourke, B.; Kass, D.A.; Marbán, E.; Tomaselli, G.F. Molecular correlates of altered expression of potassium currents in failing rabbit myocardium. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H2077–H2087. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, A.; Nishijima, Y.; Terentyev, D.; Khan, M.; Terentyeva, R.; Hamlin, R.L.; Nakayama, T.; Gyorke, S.; Cardounel, A.J.; Carnes, C.A. Chronic heart failure and the substrate for atrial fibrillation. Cardiovasc. Res. 2009, 84, 227–236. [Google Scholar] [CrossRef]
- Hegyi, B.; Bossuyt, J.; Griffiths, L.G.; Shimkunas, R.; Coulibaly, Z.; Jian, Z.; Grimsrud, K.N.; Sondergaard, S.S.; Ginsburg, K.S.; Chiamvimonvat, N.; et al. Complex electrophysiological remodeling in postinfarction ischemic heart failure. Proc. Natl. Acad. Sci. USA 2018, 115, E3036–E3044. [Google Scholar] [CrossRef] [Green Version]
- Beuckelmann, D.J.; Näbauer, M.; Erdmann, E. Alterations of K+ currents in isolated human ventricular myocytes from patients with terminal heart failure. Circ. Res. 1993, 73, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Ehrlich, J.R. Inward rectifier potassium currents as a target for atrial fibrillation therapy. J. Cardiovasc. Pharmacol. 2008, 52, 129–135. [Google Scholar] [CrossRef]
- Tobón, C.; Palacio, L.C.; Chidipi, B.; Slough, D.P.; Tran, T.; Tran, N.; Reiser, M.; Lin, Y.S.; Herweg, B.; Sayad, D.; et al. The Antimalarial Chloroquine Reduces the Burden of Persistent Atrial Fibrillation. Front. Pharmacol. 2019, 10, 1392. [Google Scholar] [CrossRef]
- Mundt, H.M.; Matenaer, M.; Lammert, A.; Göttmann, U.; Krämer, B.K.; Birck, R.; Benck, U. Minoxidil for Treatment of Resistant Hypertension in Chronic Kidney Disease--A Retrospective Cohort Analysis. J. Clin. Hypertens. 2016, 18, 1162–1167. [Google Scholar] [CrossRef] [Green Version]
- Ratjen, F.; Bell, S.C.; Rowe, S.M.; Goss, C.H.; Quittner, A.L.; Bush, A. Cystic fibrosis. Nat. Rev. Dis. Primers 2015, 1, 15010. [Google Scholar] [CrossRef]
- Varothai, S.; Bergfeld, W.F. Androgenetic alopecia: An evidence-based treatment update. Am. J. Clin. Dermatol. 2014, 15, 217–230. [Google Scholar] [CrossRef]
- Caballero, R.; Dolz-Gaitón, P.; Gómez, R.; Amorós, I.; Barana, A.; de la Fuente, M.G.; Osuna, L.; Duarte, J.; López-Izquierdo, A.; Moraleda, I.; et al. Flecainide increases Kir2.1 currents by interacting with cysteine 311, decreasing the polyamine-induced rectification. Proc. Natl. Acad. Sci. USA 2010, 107, 15631–15636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kobayashi, T.; Wahiyama, K.; Ikeda, K. Pregnenolone Sulfate Potentiates the Inwardly Rectifying K+ Channel Kir2.3. PLoS ONE 2009, 4, e6311. [Google Scholar] [CrossRef] [PubMed]
- Gómez, R.; Caballero, R.; Barana, A.; Amorós, I.; De Palm, S.H.; Matamoros, M.; Núñez, M.; Pérez-Hernández, M.; Iriepa, I.; Tamargo, J.; et al. Structural basis of drugs that increase cardiac inward rectifier Kir2.1 currents. Cardiovasc. Res. 2014, 104, 337–346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexandre, J.; Hof, T.; Puddu, P.E.; Rouet, R.; Guinamard, R.; Manrique, A.; Beygui, F.; Sallé, L.; Milliez, P. Rapid and MR-Independent IK1 Activation by Aldosterone during Ischemia-Reperfusion. PLoS ONE 2015, 10, e0132592. [Google Scholar] [CrossRef]
- Lammers, C.; Dartsch, T.; Brandt, M.C.; Rottländer, D.; Halbach, M.; Peinkofer, G.; Ockenpoehler, S.; Weierbraeger, M.; Schneider, T.; Reuter, H.; et al. Spironolactone prevents aldosterone induced increased duration of atrial fibrillation in rat. Cell. Physiol. Biochem. 2012, 29, 833–840. [Google Scholar] [CrossRef]
- Scherer, D.; Kiesecker, C.; Kulzer, M.; Günth, M.; Scholz, E.P.; Kathöfer, S.; Thomas, D.; Maurer, M.; Kreuzer, J.; Bauer, A.; et al. Activation of inwardly rectifying Kir2.x potassium channels by β3-adrenoceptors is mediated via different signaling pathways with a predominant role of PKC for Kir2.1 and of PKA for Kir2.2. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2007, 375, 311–322. [Google Scholar] [CrossRef]
- Li, X.; Hu, H.; Wang, Y.; Xue, M.; Li, X.; Cheng, W.; Xuan, Y.; Yin, J.; Yang, N.; Yan, S. Valsartan Upregulates Kir2.1 in Rats Suffering from Myocardial Infarction via Casein Kinase 2. Cardiovasc. Drugs Ther. 2015, 29, 209–218. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Hu, H.; Wang, Y.; Xue, M.; Li, X.; Cheng, W.; Xuan, Y.; Yin, J.; Yang, N.; Yan, S. Valsartan Attenuates KIR2.1 by Downregulating the Th1 Immune Response in Rats Following Myocardial Infarction. J. Cardiovasc. Pharmacol. 2016, 67, 252–259. [Google Scholar] [CrossRef]
- Li, X.; Hu, H.; Wang, Y.; Xue, M.; Li, X.; Cheng, W.; Xuan, Y.; Yin, J.; Yang, N.; Yan, S. Valsartan ameliorates KIR2.1 in rats with myocardial infarction via the NF-κB-mir-16 pathway. Gene 2016, 590, 201–209. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Q.H.; Liu, C.F.; Zhai, X.W.; Feng, Q.L.; Xu, R.L.; Cui, X.L.; Zhao, Z.Q.; Cao, J.M.; Wu, B. Zacopride selectively activates the Kir2.1 channel via a PKA signaling pathway in rat cardiomyocytes. Sci. China Life Sci. 2013, 56, 788–796. [Google Scholar] [CrossRef] [Green Version]
- Fang, W.; Xia, L.; Sun, X.; Cai, S.; Bai, S.; Sun, Y.; Zhou, P.; Liu, X.; Zhao, R.; Shen, B. Lipopolysaccharides increase Kir2.1 expression in lung endothelial cells. Int. J. Clin. Exp. Pathol. 2018, 11, 2959–2967. [Google Scholar] [PubMed]
- Xiao, G.-S.; Zhou, J.-J.; Wang, G.-Y.; Cao, C.-M.; Li, G.-R.; Wong, T.-M. In vitro electrophysiologic effects of morphine in rabbit ventricular myocytes. Anesthesiology 2005, 103, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhou, J.-J.; Yang, J.; Zhang, J.; Zhang, L.-P.; Zhang, Y. Enhancement of polydatin on inward rectifier potassium channel current in rat ventricular myocytes. Acta Physiol. Sin. 2013, 65, 285–292. [Google Scholar]
- Liu, Y.; Liu, D.; Printzenhoff, D.; Coghlan, M.J.; Harris, R.; Krafte, D.S. Tenidap, a novel anti-inflammatory agent, is an opener of the inwardly rectifying K+ channel hKir2.3. Eur. J. Pharmacol. 2002, 435, 153–160. [Google Scholar] [CrossRef]
- Dartsch, T.; Fischer, R.; Gapelyuk, A.; Weiergraeber, M.; Ladage, D.; Schneider, T.; Schirdewan, A.; Reuter, H.; Müeller-Ehmsen, J.; Zobel, C. Aldosterone induces electrical remodeling independent of hypertension. Int. J. Cardiol. 2013, 164, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Sterin-Borda, L.; Bernabeo, G.; Ganzinelli, S.; Joensen, L.; Borda, E. Role of nitric oxide/cyclic GMP and cyclic AMP in β3 adrenoceptor-chronotropic response. J. Mol. Cell. Cardiol. 2006, 40, 580–588. [Google Scholar] [CrossRef]
- Zitron, E.; Kiesecker, C.; Lück, S.; Kathöfer, S.; Thomas, D.; Kreye, V.A.W.; Kiehn, J.; Katus, H.A.; Schoels, W.; Karle, C.A. Human cardiac inwardly rectifying current IKir2.2 is upregulated by activation of protein kinase A. Cardiovasc. Res. 2004, 63, 520–527. [Google Scholar] [CrossRef] [Green Version]
- Hwang, S.H.; Wecksler, A.T.; Wagner, K.; Hammock, B.D. Rationally designed multitarget agents against inflammation and pain. Curr. Med. Chem. 2013, 20, 1783–1799. [Google Scholar] [CrossRef]
- Best, L.; Brown, P.D.; Sener, A.; Malaisse, W.J. Opposing effects of tenidap on the volume-regulated anion channel and K(ATP) channel activity in rat pancreatic beta-cells. Eur. J. Pharmacol. 2010, 629, 159–163. [Google Scholar] [CrossRef]
- Kızılirmak, P.; Üresin, A.Y. Hypertension and valsartan. Anadolu Kardiyol. Derg. 2014, 14, S20–S24. [Google Scholar] [CrossRef] [Green Version]
- Liao, S.-Y.; Tse, H.-F.; Chan, Y.-C.; Yip, P.M.-C.; Zhang, Y.; Liu, Y.; Li, R.A. Overexpression of Kir2.1 channel in embryonic stem cell-derived cardiomyocytes attenuates posttransplantation proarrhythmic risk in myocardial infarction. Heart Rhythm 2013, 10, 273–282. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, H.; Contesse, V.; Delarue, C.; Soubrane, C.; Legrand, A.; Kuhn, J.M.; Wolf, L.M.; Vaudry, H. Effect of the serotonin-4 receptor agonist zacopride on aldosterone secretion from the human adrenal cortex: In vivo and in vitro studies. J. Clin. Endocrinol. Metab. 1993, 77, 1662–1666. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.-H.; Qiao, X.; Zhang, L.-J.; Wang, J.; Zhang, L.; Zhai, X.-W.; Re, X.-Z.; Li, Y.; Cao, X.-N.; Feng, Q.-L.; et al. IK1 Channel Agonist Zacopride Alleviates Cardiac Hypertrophy and Failure via Alterations in Calcium Dyshomeostasis and Electrical Remodeling in Rats. Front. Pharmacol. 2019, 10, 929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.; Liu, E.; Luo, T.; Zhang, W.; He, R. Opening of the inward rectifier potassium channel alleviates maladaptive tissue repair following myocardial infarction. Acta Biochim. Biophys. Sin. 2016, 48, 687–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korte, S.M.; Van der Heyden, M.A.G. Preventing publication of falsified and fabricated data: Roles of scientists, editors, reviewers, and readers. J. Cardiovasc. Pharmacol. 2017, 69, 65–70. [Google Scholar] [CrossRef]
- Liu, Q.-H.; Li, X.-L.; Xu, Y.-W.; Lin, Y.-Y.; Cao, J.-M.; Wu, B.-W. A novel discovery of IK1 channel agonist: Zacopride selectively enhances IK1 current and suppresses triggered arrhythmias in the rat. J. Cardiovasc. Pharmacol. 2012, 59, 37–48. [Google Scholar] [CrossRef]
- Zhai, X.-W.; Zhang, L.; Guo, Y.-F.; Yang, Y.; Wang, D.-M.; Zhang, Y.; Li, P.; Niu, Y.-F.; Feng, Q.-L.; Wu, B.-W.; et al. The IK1/Kir2.1 channel agonist zacopride prevents and cures acute ischemic arrhythmias in the rat. PLoS ONE 2017, 12, e0177600. [Google Scholar] [CrossRef] [Green Version]
- Katritsis, D.; Rowland, E.; O’Nunain, S.; Shakespeare, C.F.; Poloniecki, J.; Camm, A.J. Effect of flecainide on atrial and ventricular refractoriness and conduction in patients with normal left ventricle. Implications for possible antiarrhythmic and proarrhythmic mechanisms. Eur. Heart J. 1995, 16, 1930–1935. [Google Scholar] [CrossRef]
- Amorós, I.; Dolz-Gaitón, P.; Gómez, R.; Matamoros, M.; Barana, A.; de la Fuente, M.G.; Núñez, M.; Pérez-Hernández, M.; Moraleda, I.; Gálvez, E.; et al. Propafenone blocks human cardiac Kir2.x channels by decreasing the negative electrostatic charge in the cytoplasmic pore. Biochem. Pharmacol. 2013, 86, 267–278. [Google Scholar] [CrossRef]
- Pegan, S.; Arrabit, C.; Zhou, W.; Kwiatkowski, W.; Collins, A.; Slesinger, P.A.; Choe, S. Cytoplasmic domain structures of Kir2.1 and Kir3.1 show sites for modulating gating and rectification. Nat. Neurosci. 2005, 8, 279–287. [Google Scholar] [CrossRef]
- Schumacher, M.; Liere, P.; Akwa, Y.; Rajkowski, K.; Griffiths, W.; Bodin, K.; Sjövall, J.; Baulieu, E.-E. Pregnenolone sulfate in the brain: A controversial neurosteroid. Neurochem. Int. 2008, 52, 522–540. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, G.W. Pharmacological mechanisms of opioid analgesics. Clin. NeuroPharmacol. 1993, 16, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.F.; Tsai, C.H.; Su, M.J. Opioid receptor independent effects of morphine on membrane currents in single cardiac myocytes. Br. J. Anaesth. 1998, 81, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chen, P.; Deng, J.; Lv, J.; Liu, J. Resveratrol and polydatin as modulators of Ca2+ mobilization in the cardiovascular system. Ann. N. Y. Acad. Sci. 2017, 1403, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Ramos, E.; O’leary, M.E. State-dependent trapping of flecainide in the cardiac sodium channel. J. Physiol. 2004, 560, 37–49. [Google Scholar] [CrossRef]
- Walsh, K.B. Screening Technologies for Inward Rectifier Potassium Channels: Discovery of New Blockers and Activators. SLAS Discov. 2020, 25, 420–433. [Google Scholar] [CrossRef]
- Heijerman, H.G.M.; McKone, E.F.; Downey, D.G.; Van Braeckel, E.; Rowe, S.M.; Tullis, E.; Mall, M.A.; Welter, J.J.; Ramsey, B.W.; McKee, C.M.; et al. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: A double-blind, randomised, phase 3 trial. Lancet 2019, 394, 1940–1948. [Google Scholar] [CrossRef]
- Middleton, P.G.; Mall, M.A.; Dřevínek, P.; Lands, L.C.; McKone, E.F.; Polineni, D.; Ramsey, B.W.; Taylor-Cousar, J.L.; Tullis, E.; Vermeulen, F.; et al. Elexacaftor-Tezacaftor-Ivacaftor for Cystic Fibrosis with a Single Phe508del Allele. N. Engl. J. Med. 2019, 381, 1809–1819. [Google Scholar] [CrossRef]
- Vandenberg, J.I.; Perry, M.D.; Perrin, M.J.; Mann, S.A.; Ke, Y.; Hill, A.P. hERG K+ channels: Structure, function, and clinical significance. Physiol. Rev. 2012, 92, 1393–1478. [Google Scholar] [CrossRef] [Green Version]
- Perry, M.; Sanguinetti, M.; Mitcheson, J. Revealing the structural basis of action of hERG potassium channel activators and blockers. J. Physiol. 2010, 588, 3157–3167. [Google Scholar] [CrossRef]
- Meng, J.; Shi, C.; Li, L.; Du, Y.; Xu, Y. Compound ICA-105574 prevents arrhythmias induced by cardiac delayed repolarization. Eur. J. Pharmacol. 2013, 718, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Sanguinetti, M.C. HERG1 channel agonists and cardiac arrhythmia. Curr. Opin. Pharmacol. 2014, 15, 22–27. [Google Scholar] [CrossRef] [Green Version]
- Qile, M.; Beekman, H.D.M.; Sprenkeler, D.J.; Houtman, M.J.C.; van Ham, W.B.; Stary-Weinzinger, A.; Beyl, S.; Hering, S.; van den Berg, D.-J.; de Lange, E.C.M.; et al. LUF7244, an allosteric modulator/activator of Kv11.1 channels, counteracts dofetilide-induced torsades de pointes arrhythmia in the chronic atrioventricular block dog model. Br. J. Pharmacol. 2019, 176, 3871–3885. [Google Scholar] [CrossRef] [Green Version]
- Anderson, C.L.; Delisle, B.P.; Anson, B.D.; Kilby, J.A.; Will, M.L.; Tester, D.J.; Gong, Q.; Zhou, Z.; Ackerman, M.J.; January, C.T. Most LQT2 mutations reduce Kv11.1 (hERG) current by a class 2 (trafficking-deficient) mechanism. Circulation 2016, 113, 365–373. [Google Scholar] [CrossRef] [Green Version]
- Anderson, C.L.; Kuzmicki, C.E.; Childs, R.R.; Hintz, C.J.; Delisle, B.P.; January, C.T. Large-scale mutational analysis of Kv11.1 reveals molecular insights into type 2 long QT syndrome. Nat. Commun. 2014, 5, 5535. [Google Scholar] [CrossRef] [PubMed]
- Wible, B.A.; Hawryluk, P.; Ficker, E.; Kuryshev, Y.A.; Kirsch, G.; Brown, A.M. HERG-Lite: A novel comprehensive high-throughput screen for drug-induced hERG risk. J. Pharmacol. Toxicol. Methods 2005, 52, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Varkevisser, R.; Houtman, M.J.C.; Linder, T.; de Git, K.C.G.; Beekman, H.D.M.; Tidwell, R.R.; IJzerman, A.P.; Star-Weinziger, A.; Vos, M.A.; Van der Heyden, M.A.G. Structure-activity relationships of pentamidine-affected ion channel trafficking and dofetilide mediated rescue. Br. J. Pharmacol. 2013, 169, 1322–1334. [Google Scholar] [CrossRef]
- Qile, M.; Ji, Y.; Golden, T.D.; Houtman, M.J.C.; Romunde, F.; Fransen, D.; van Ham, W.B.; IJzerman, A.P.; January, C.T.; Heitman, L.H.; et al. LUF7244 plus dofetilide rescues aberrant Kv11.1 trafficking and produces functional IKv11.1. Mol. Pharmacol. 2020, 97, 355–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Compound | Readout | Test System | Dose-Effect Relation | Mechanism of Action | Ref. |
|---|---|---|---|---|---|
| Direct activators | |||||
| Flecainide | C | CHO cells | IKir2.1 EC50/Emax (−50 mV) = 0.4 ± 0.01 μM/53.9 ± 3.6% IKir2.1 EC50/Emax (−120 mV) = 0.8 ± 0.01 μM/22.0 ± 1.96% | Interaction with Cys311 | [31] |
| C | guinea pig ventricular cmc | IK1 1 μM: 19.5 ± 3.2% (−120 mV); 38.0 ± 9.5% (−40 mV) | Interaction with Cys311 | [31] | |
| PREGS | C | Xenopus oocytes | IKir2.3 EC50 (−70 mV) = 15.6 ± 0.9 μM | Binding extracellular site | [32] |
| Propafenone | C | CHO cells | IKir2.1 EC50/Emax (−50 mV) = 12.0 ± 3.0 nM/42.0 ± 2.6% | Interaction with Cys311 | [33] |
| C | guinea pig ventricular cmc | IK1 0.5 μM: approx 45 ± 5% (−40 mV) | Interaction with Cys311 | [33] | |
| Timolol | C | CHO cells | IKir2.1 EC50 (−50 mV) = 3.2 ± 0.3 nM | Interaction with Cys311 | [33] |
| Indirect activators | |||||
| Aldosterone | C | rabbit ventricular cmc | IK1 10 nM: 1.6-fold increase NPo of 30 pS current | MR-independent activation | [34] |
| rat ventricle | Kir2.1 2.24 mg/h/kg 4 wks: approx. 1.57 ± 0.14 fold Kir2.3 2.24 mg/h/kg 4 wks: approx. 1.26 ± 0.15 fold (ns) | Unknown Unknown | [35] | ||
| Isoproterenol | C | Xenopus oocytes | IKir2.1 EC50 = 27.4 nM * IKir2.2 EC50 = 17.8 nM * | PKC dependent via β3-AR PKA dependent via β3-AR | [36] |
| Valsartan | R,P | rat ventricle | 10 mg/kg/day for 7 days prevented Kir2.1 downregulation | Casein Kinase 2 inhibition and/or Th1 immune response inhibition and/or NF-κB-miR-16 pathway | [37,38,39] |
| R,P | ventricular cmc, H9c2 cells | 20 μM (48 h) prevented Kir2.1 downregulation | Casein Kinase 2 inhibition and/or Th1 immune response inhibition and/or NF-κB-miR-16 pathway | [37,38,39] | |
| C | rat ventricular cmc | 20 μM (48 h) prevented IK1 inward current downregulation | Th1 immune response inhibition | [38] | |
| Zacopride | C | rat atrial cmc, HEK-293 cells | IKir2.1 EC50 (−50 mV) = 30.7 nM IKir2.1 (−50 mV) = 40.7 ± 9.7% | PKA dependent | [40] |
| LPS | P,C | mouse pulmonary micro-vascular endothelial cells | 10 ng/mL (up to 24 h): 1.5-fold increase Kir2.1 10 ng/mL IKir2.1 (inward/outward) = approx. 1.7-fold | Unknown | [41] |
| Morphine | C | rabbit ventricular cmc | IK1 (−60 mV) = 25 ± 9% (0.1 μM); 32 ± 11% (1 μM) | Opioid-receptor pathway independent | [42] |
| Polydatin | C | rat ventricular cmc | IK1 10 μM: approx. 40% (−100 mV) | Unknown | [43] |
| Tenidap | C | CHO cells | IKir2.3 EC50 = 1.3 μM * | Extracellularly, unaffected by pA2, PKC, and AA secondary pathways | [44] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van der Schoor, L.; van Hattum, E.J.; de Wilde, S.M.; Harlianto, N.I.; van Weert, A.-J.; Bloothooft, M.; van der Heyden, M.A.G. Towards the Development of AgoKirs: New Pharmacological Activators to Study Kir2.x Channel and Target Cardiac Disease. Int. J. Mol. Sci. 2020, 21, 5746. https://doi.org/10.3390/ijms21165746
van der Schoor L, van Hattum EJ, de Wilde SM, Harlianto NI, van Weert A-J, Bloothooft M, van der Heyden MAG. Towards the Development of AgoKirs: New Pharmacological Activators to Study Kir2.x Channel and Target Cardiac Disease. International Journal of Molecular Sciences. 2020; 21(16):5746. https://doi.org/10.3390/ijms21165746
Chicago/Turabian Stylevan der Schoor, Laura, Emma J. van Hattum, Sophie M. de Wilde, Netanja I. Harlianto, Aart-Jan van Weert, Meye Bloothooft, and Marcel A. G. van der Heyden. 2020. "Towards the Development of AgoKirs: New Pharmacological Activators to Study Kir2.x Channel and Target Cardiac Disease" International Journal of Molecular Sciences 21, no. 16: 5746. https://doi.org/10.3390/ijms21165746





