Lactobacillus paracasei KW3110 Suppresses Inflammatory Stress-Induced Premature Cellular Senescence of Human Retinal Pigment Epithelium Cells and Reduces Ocular Disorders in Healthy Humans
Abstract
1. Introduction
2. Results
2.1. In Vitro Experiments
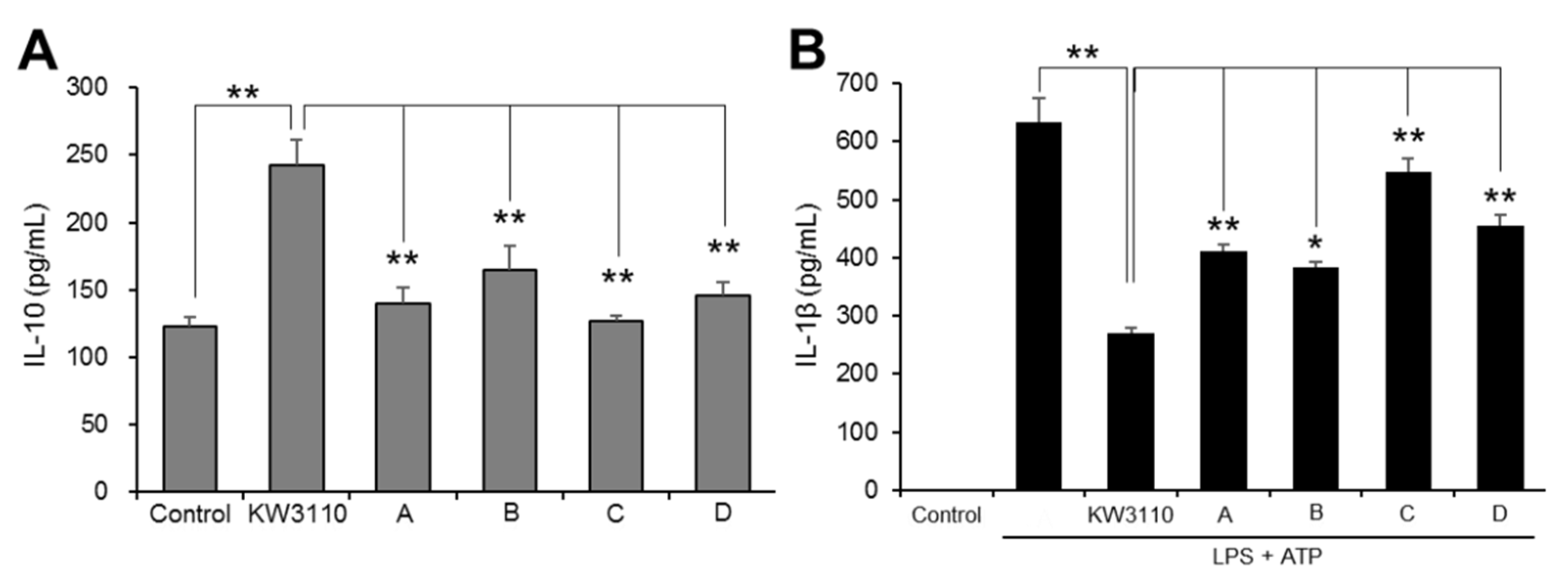
2.1.1. KW3110 Has a Stronger Anti-Inflammatory Effect than LAB from Commercial Products
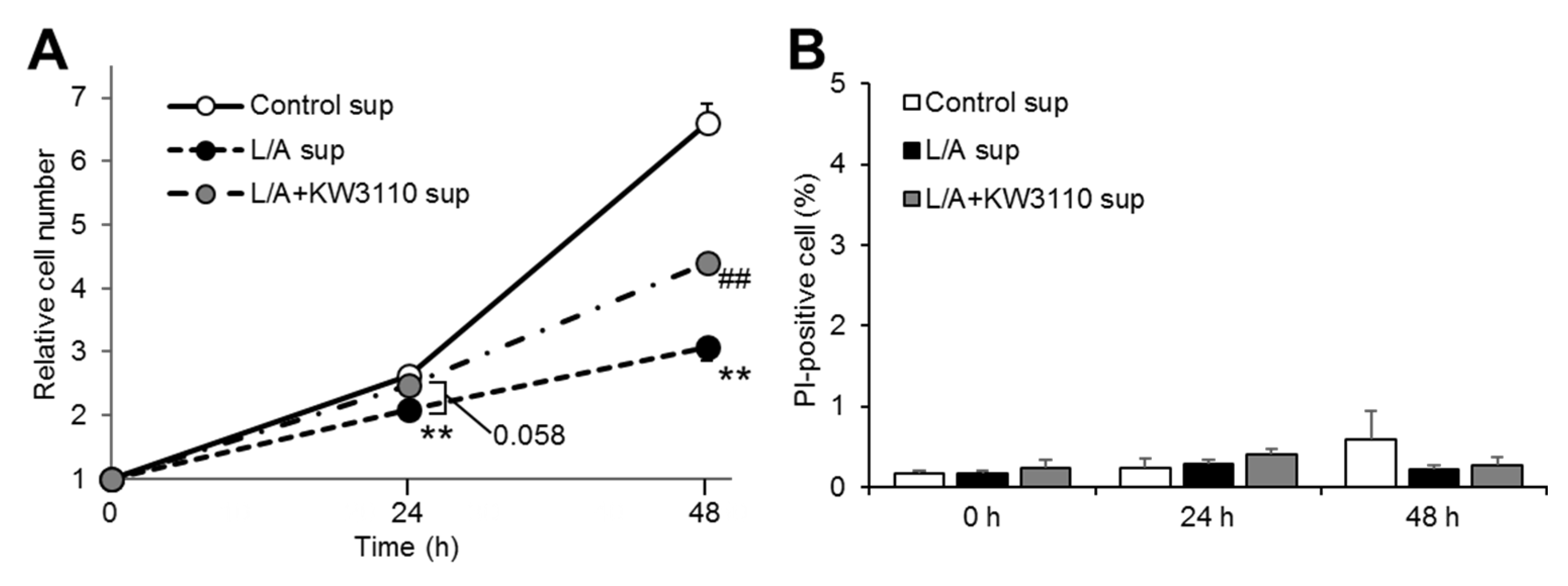
2.1.2. ARPE-19 Cell Proliferation, But Not Cell Death, Is Affected by KW3110 or LPS/ATP Stimulation of Macrophages
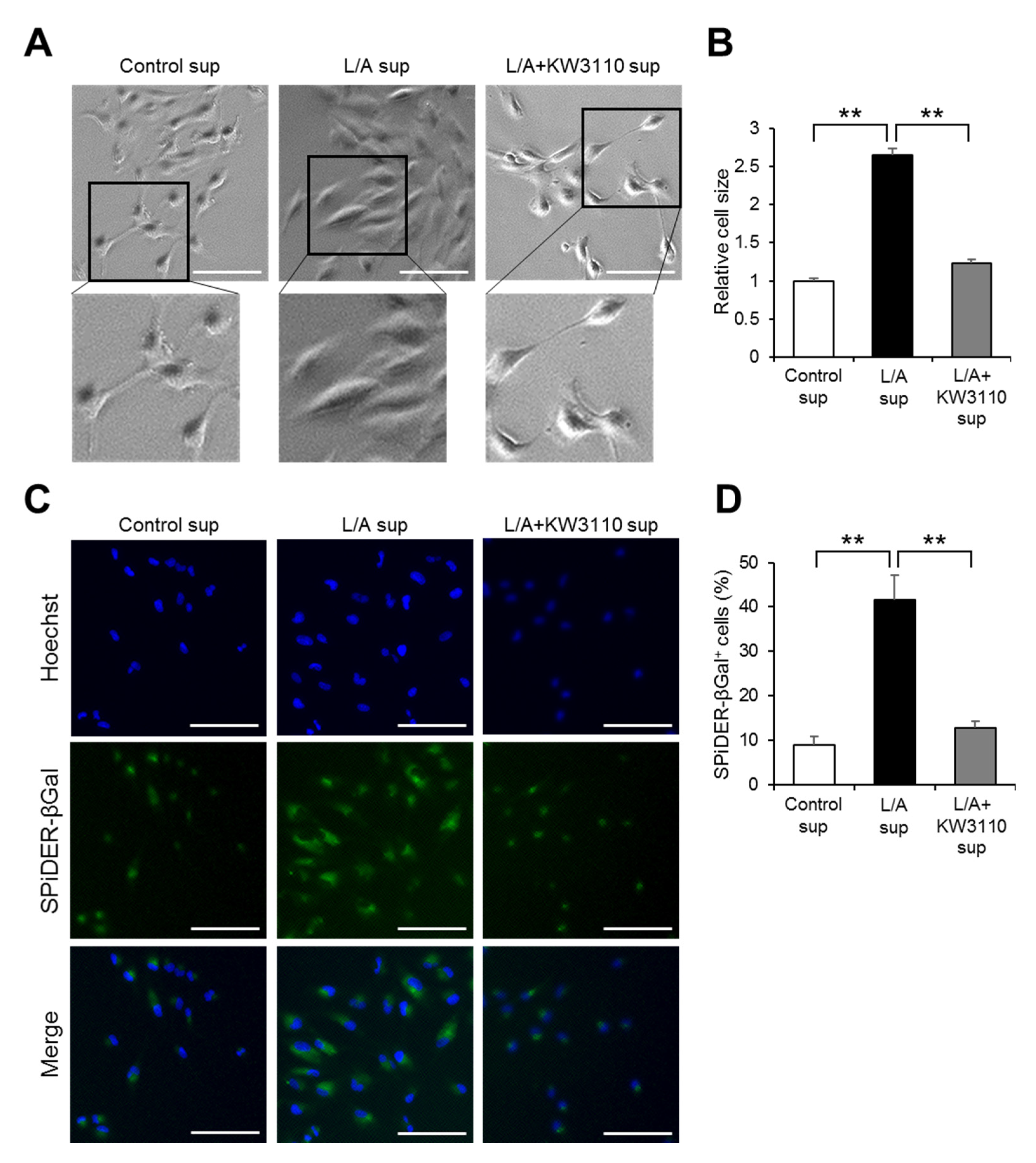
2.1.3. Effects of KW3110 on Prematurely Senescent Phenotypes of Inflammatory-Stressed ARPE-19 Cells
2.1.4. Effects of KW3110 on Cell Cycle Arrest and Senescence-Associated Secretory Phenotype (SASP)-Related Gene Upregulation in Inflammatory-Stressed ARPE-19 Cells
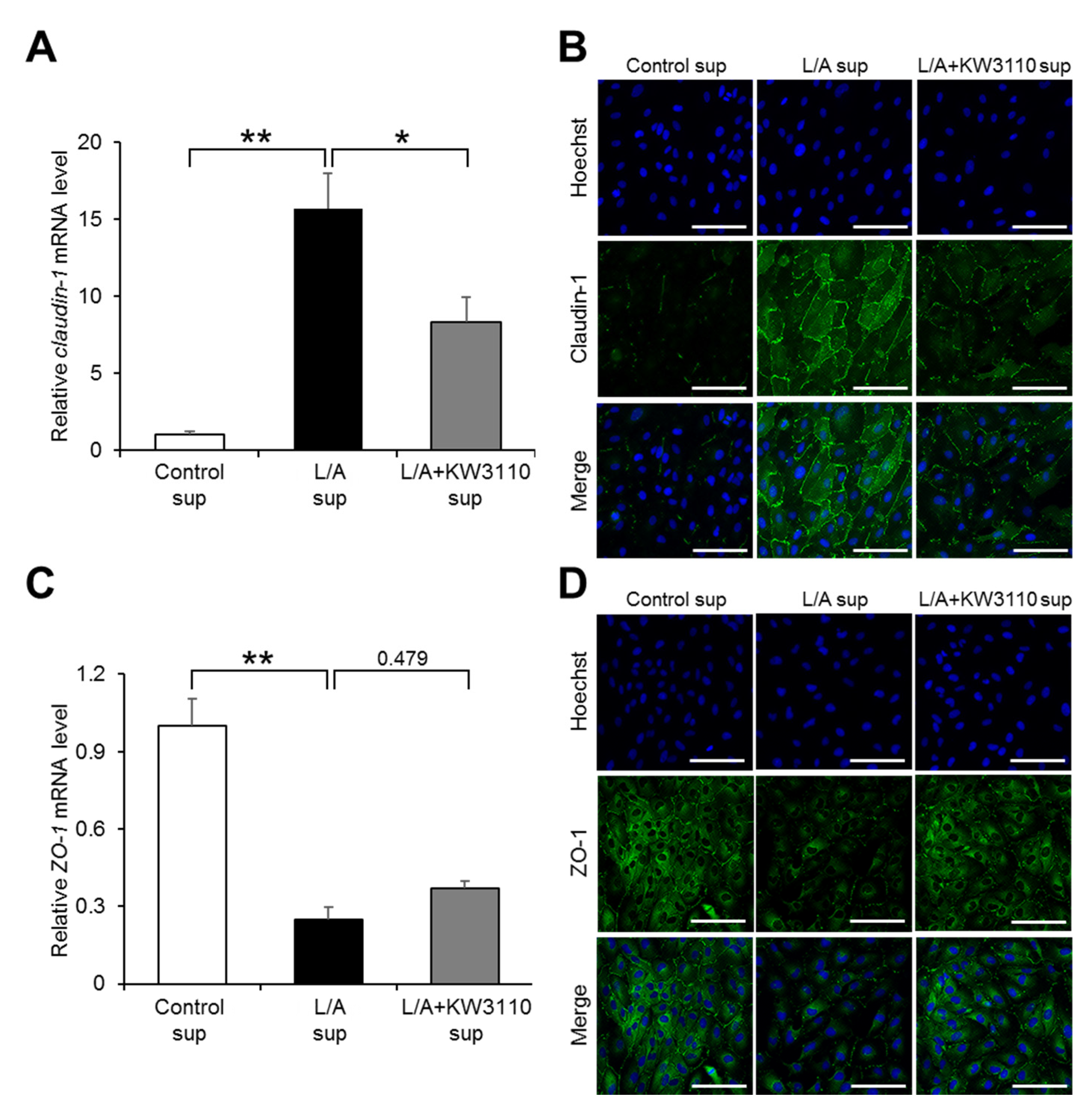
2.1.5. Effects of KW3110 on the Expression of Tight Junction Molecules in Inflammatory-Stressed ARPE-19 Cells
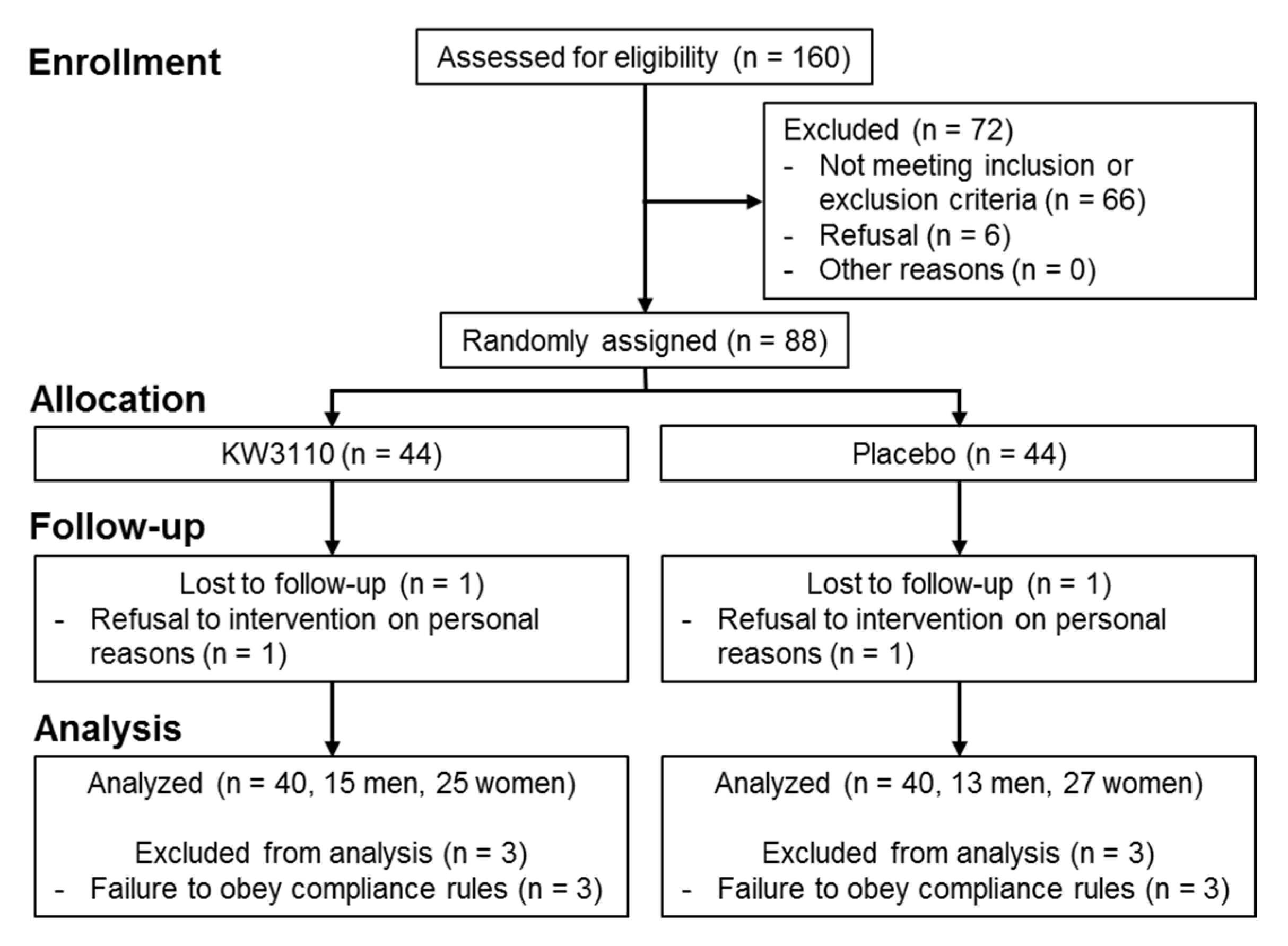
2.2. Clinical Study
2.2.1. Background Information and Baseline Characteristics of the Subjects
2.2.2. Primary Outcomes
2.2.3. Secondary Outcomes
2.2.4. Safety Endpoints
3. Discussion
4. Materials and Methods
4.1. In Vitro Experiments
4.1.1. Materials
4.1.2. Preparation of Human Monocytes
4.1.3. Cell Culture
4.1.4. Determination of Cytokine Production
4.1.5. Cell Counts and Cell Death Analysis
4.1.6. SPiDER-βGal Staining
4.1.7. Quantitative Real-Time RT-PCR
4.1.8. Cell Cycle Assay
4.1.9. Immunocytochemistry
4.1.10. Statistical Analysis
4.2. Clinical Study
4.2.1. Ethics
4.2.2. Subjects
4.2.3. Target Sample Size
4.2.4. Test Supplements
4.2.5. Study Design
4.2.6. Primary Outcomes
4.2.7. Secondary Outcomes
4.2.8. Safety Endpoints
4.2.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATP | adenosine 5′-triphosphate |
| CFF | critical flicker frequency |
| DD | depression-dejection |
| HFC-1 | high frequency component |
| IL-1β | interleukin-1β |
| IL-10 | interleukin-10 |
| KW3110 | Lactobacillus paracasei KW3110 |
| LAB | lactic acid bacteria |
| LPS | ultrapure lipopolysaccharide from E. coli 0111:B4 strain |
| PI | propidium iodide |
| POMS2 | profile of mood states 2nd edition |
| SASP | senescence-associated secretory phenotype |
| VAS | visual analogue scale |
References
- Miller, A.H.; Raison, C.L. The role of inflammation in depression: From evolutionary imperative to modern treatment target. Nat. Rev. Immunol. 2016, 16, 22–34. [Google Scholar] [CrossRef]
- Calçada, D.; Vianello, D.; Giampieri, E.; Sala, C.; Castellani, G.; de Graaf, A.; Kremer, B.; van Ommen, B.; Feskens, E.; Santoro, A. The role of low-grade inflammation and metabolic flexibility in aging and nutritional modulation thereof: A systems biology approach. Mech. Ageing Dev. 2014, 136, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T.; Serrano, M.; Blasco, M.A. The common biology of cancer and ageing. Nature 2007, 448, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Deleidi, M.; Jäggle, M.; Rubino, G. Immune aging, dysmetabolism, and inflammation in neurological diseases. Front. Neurosci. 2015, 9, 172. [Google Scholar] [CrossRef]
- Fontana, L.; Bermudez-Brito, M.; Plaza-Diaz, J.; Munoz-Quezada, S.; Gil, A. Sources, isolation, characterisation and evaluation of probiotics. Br. J. Nutr. 2013, 109, S35–S50. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, S.; Fujii, R.; Fujiwara, D.; Komiyama, Y.; Kaisho, T.; Sakaguchi, M.; Konishi, Y. MyD88 but not TLR2, 4 or 9 is essential for IL-12 induction by lactic acid bacteria. Biosci. Biotechnol. Biochem. 2007, 71, 3026. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, H.; Nariai, C.; Takemura, F.; Nakao, W.; Fujiwara, D. Dietary supplementation with lactic acid bacteria attenuates the development of atopic dermatitis-like skin lesions in NC/Nga mice in a strain-dependent manner. Int. Arch. Allergy Immunol. 2008, 145, 141–151. [Google Scholar] [CrossRef]
- Fujiwara, D.; Inoue, S.; Wakabayashi, H.; Fujii, T. The anti-allergic effects of lactic acid bacteria are strain dependent and mediated by effects on both Th1/Th2 cytokine expression and balance. Int. Arch. Allergy Immunol. 2004, 135, 205–215. [Google Scholar] [CrossRef]
- Ichikawa, S.; Miyake, M.; Fujii, R.; Konishi, Y. Orally Administered Lactobacillus paracasei KW3110 induces in vivo IL-12 production. Biosci. Biotechnol. Biochem. 2009, 73, 1561–1565. [Google Scholar] [CrossRef]
- Morita, Y.; Miwa, Y.; Jounai, K.; Fujiwara, D.; Kurihara, T.; Kanauchi, O. Lactobacillus paracasei KW3110 prevents blue light-induced inflammation and degeneration in the retina. Nutrients 2018, 10, 1991. [Google Scholar] [CrossRef]
- Morita, Y.; Jounai, K.; Miyake, M.; Inaba, M.; Kanauchi, O. Effect of heat-killed Lactobacillus paracasei KW3110 ingestion on ocular disorders caused by visual display terminal (VDT) Loads: A randomized, double-blind, placebo-controlled parallel-group study. Nutrients 2018, 10, 1058. [Google Scholar] [CrossRef]
- Miceli, M.V.; Jazwinski, S.M. Nuclear gene expression changes due to mitochondrial dysfunction in ARPE-19 cells: Implications for age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2005, 46, 1765–1773. [Google Scholar] [CrossRef]
- Kumashiro, M.; Mikami, K.; Hasegawa, T. Effects of visual and mental strain on VDT performance. Jpn. J. Ind. Health 1984, 26, 105–111. [Google Scholar]
- Hoshino, K.; Watanabe, A.; Saito, M. Estimation of mental fatigue through analysis of pupillary light response. J. Inst. Telev. Eng. 1995, 49, 657–664. [Google Scholar]
- Geerlings, M.J.; de Jong, E.K.; den Hollander, A.I. The complement system in age-related macular degeneration: A review of rare genetic variants and implications for personalized treatment. Mol. Immunol. 2017, 84, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Kauppinen, A.; Paterno, J.J.; Blasiak, J.; Salminen, A.; Kaarniranta, K. Inflammation and its role in age-related macular degeneration. Cell. Mol. Life Sci. 2016, 73, 1765–1786. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.-H.; Pan, T.-M.; Wu, Y.-J.; Chang, S.-J.; Chang, M.-S.; Hu, C.-Y. Exopolysaccharide activities from probiotic bifidobacterium: Immunomodulatory effects (on J774A.1 macrophages) and antimicrobial properties. Int. J. Food Microbiol. 2010, 144, 104–110. [Google Scholar] [CrossRef]
- Hara, K.; Shirasuna, K.; Usui, F.; Karasawa, T.; Mizushina, Y.; Kimura, H.; Kawashima, A.; Ohkuchi, A.; Matsuyama, S.; Kimura, K.; et al. Interferon-tau attenuates uptake of nanoparticles and secretion of interleukin-1β in macrophages. PLoS ONE 2014, 9, e113974. [Google Scholar] [CrossRef]
- Ip, W.E.; Hoshi, N.; Shouval, D.S.; Snapper, S.; Medzhitov, R. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science 2017, 356, 513–519. [Google Scholar] [CrossRef]
- Serrano, M.; Lin, A.W.; McCurrach, M.E.; Beach, D.; Lowe, S.W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 1997, 88, 593–602. [Google Scholar] [CrossRef]
- Johmura, Y.; Shimada, M.; Misaki, T.; Naiki-Ito, A.; Miyoshi, H.; Motoyama, N.; Ohtani, N.; Hara, E.; Nakamura, M.; Morita, A.; et al. Necessary and sufficient role for a mitosis skip in senescence induction. Mol. Cell 2014, 55, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.; Humphries, P. The blood-retina barrier: Tight junctions and barrier modulation. Adv. Exp. Med. Biol. 2012, 763, 70–84. [Google Scholar] [PubMed]
- Suzuki, H.; Yamazaki, T.; Ohshio, K.; Sugamata, M.; Yoshikawa, M.; Kanauchi, O.; Morita, Y. A specific strain of lactic acid bacteria, lactobacillus paracasei, inhibits inflammasome activation in vitro and prevents inflammation-related disorders. J. Immunol. 2020, 20, 1900657. [Google Scholar] [CrossRef] [PubMed]
- Underhill, D.M.; Ozinsky, A. Phagocytosis of microbes: Complexity in action. Annu. Rev. Immunol. 2002, 20, 825–852. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, S.; Miyake, M.; Fujii, R.; Konishi, Y. MyD88 associated ROS generation is crucial for Lactobacillus induced IL-12 production in macrophage. PLoS ONE 2012, 7, e35880. [Google Scholar] [CrossRef]
- Narimatsu, T.; Negishi, K.; Miyake, S.; Hirasawa, M.; Osada, H.; Kurihara, T.; Tsubota, K.; Ozawa, Y. Blue light-induced inflammatory marker expression in the retinal pigment epithelium-choroid of mice and the protective effect of a yellow intraocular lens material in vivo. Exp. Eye Res. 2015, 132, 48–51. [Google Scholar] [CrossRef] [PubMed]
- Kubota, S.; Kurihara, T.; Ebinuma, M.; Kubota, M.; Yuki, K.; Sasaki, M.; Noda, K.; Ozawa, Y.; Oike, Y.; Ishida, S. Resveratrol prevents light-induced retinal degeneration via suppressing activator protein-1 activation. Am. J. Pathol. 2010, 177, 1725–1731. [Google Scholar] [CrossRef]
- Glotin, A.L.; Debacq-Chainiaux, F.; Brossas, J.Y.; Faussat, A.M.; Treton, J.; Zubielewicz, A.; Toussaint, O.; Mascarelli, F. Prematurely senescent ARPE-19 cells display features of age-related macular degeneration. Free Radic. Biol. Med. 2008, 44, 1348–1361. [Google Scholar] [CrossRef]
- Zhu, D.; Wu, J.; Spee, C.; Ryan, S.J.; Hinton, D.R. BMP4 mediates oxidative stress-induced retinal pigment epithelial cell senescence and is overexpressed in age-related macular degeneration. J. Biol. Chem. 2009, 284, 9529–9539. [Google Scholar] [CrossRef]
- Mishima, K.; Handa, J.T.; Aotaki-Keen, A.; Lutty, G.A.; Morse, L.S.; Hjelmeland, L.M. Senescence-associated beta-galactosidase histochemistry for the primate eye. Investig. Ophthalmol. Vis. Sci. 1999, 40, 1590–1593. [Google Scholar]
- Hirata, J.; Ko, J.-A.; Mochizuki, H.; Funaishi, K.; Yamane, K.; Sonoda, K.-H.; Kiuchi, Y. Oxidative stress regulates expression of claudin-1 in human RPE cells. Cent. Eur. J. Biol. 2014, 9, 461–468. [Google Scholar] [CrossRef]
- Abe, T.; Sugano, E.; Saigo, Y.; Tamai, M. Interleukin-1β and barrier function of retinal pigment epithelial cells (ARPE-19): Aberrant expression of junctional complex molecules. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4097–4104. [Google Scholar] [CrossRef] [PubMed]
- Narayan, S.; Prasanna, G.; Krishnamoorthy, R.R.; Zhang, X.; Yorio, T. Endothelin-1 synthesis and secretion in human retinal pigment epithelial cells (ARPE-19): Differential regulation by cholinergics and TNF-α. Investig. Ophthalmol. Vis. Sci. 2003, 44, 4885–4894. [Google Scholar] [CrossRef] [PubMed]
- Rizzolo, L.J.; Peng, S.; Luo, Y.; Xiao, W. Integration of tight junctions and claudins with the barrier functions of the retinal pigment epithelium. Prog. Retin. Eye Res. 2011, 30, 296–323. [Google Scholar] [CrossRef] [PubMed]
- Tonooka, T. Basic Text of Uchida-Kraepelin Test, 2nd ed.; Nihon Seishin Gijutsu Kenkyujo: Tokyo, Japan, 1973; pp. 1–16. Available online: https://ci.nii.ac.jp/ncid/BA36332323 (accessed on 18 July 2020).
- Sugimoto, K.; Kanai, A.; Shoji, N. The effectiveness of the Uchida-Kraepelin test for psychological stress: An analysis of plasma and salivary stress substances. Biopsychosoc. Med. 2009, 3, 5. [Google Scholar] [CrossRef]
- Ito, M.; Nakamura, T.; Yoshida, Y. Utility of TriIRIS C9000 in diagnosis and treatment for patients with asthenopia. J. Jpn. Assoc. Certif. Orthoptists 2007, 36, 73–80. [Google Scholar]
- Osaka, N. Effects of VDT color, eccentricity, and adaptation upon visual fatigue. Hum. Factors Ergon. 1985, 21, 89–95. [Google Scholar]
- Kogi, K.; Kawamura, H. On the variation of flicker fusion frequencies of visual pathway with special reference to activating system of the brain. Sci. Labour 1960, 36, 459–473. [Google Scholar]
- Nishimura, T.; Morimoto, K. Measurement method and condition of CFF as a reflection of psychic fatigue-relating to prolonged VDT work. Hum. Factors Ergon. 1986, 22, 203–210. [Google Scholar] [CrossRef]
- Standards, T.I.C.o.L. The Handbook of Industrial Fatigue; Editorial Committee of Japan Society of Occupational Health and Working Group for Occupational Fatigue: Tokyo, Japan, 1995. [Google Scholar]
- Tsukada, T. Evaluation of eye strain relaxation of progressive power lenses for the young adjustment by accommodative fluctuation. J. Jpn. Assoc. Certif. Orthoptists 2016, 45, 25–37. [Google Scholar]
- Ishikawa, H. Near reflex and its abnormalities. Rinsho Ganka 2010, 64, 1670–1674. [Google Scholar]
- Fujiwara, A.; Tabuchi, A.; Fujiwara, M.; Morita, M. The normal value of pupil response and convergent eye movement during near accommodative stimulation by TriIRIS C9000. J. Jpn. Assoc. Certif. Orthoptists 2007, 36, 67–72. [Google Scholar]
- Kajita, M.; Takahashi, N.; Takahashi, F. Dry-eye and accommodative stress: Possibility of their relationship. Jpn. J. Vis. Sci. 2004, 25, 40–45. [Google Scholar]
- McCormack, H.M.; David, J.d.L.; Sheather, S. Clinical applications of visual analogue scales: A critical review. Psychol. Med. 1988, 18, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, K.; Ishikawa, H. Decline of accommodation and asthenopia. Ganka 2009, 51, 409–413. [Google Scholar]
- Provine, R.R.; Cabrera, M.O.; Nave-Blodgett, J. Binocular symmetry/asymmetry of scleral redness as a cue for sadness, healthiness, and attractiveness in humans. Evol. Psychol. 2013, 11, 147470491301100411. [Google Scholar] [CrossRef]
- Sunaga, S. White blood cell count and differential. Rinsho Kensa 2015, 59, 166–172. [Google Scholar]
- Fujiwara, D.; Wakabayashi, H.; Watanabe, H.; Nishida, S.; Iino, H. A Double-blind trial of Lactobacillus paracasei strain KW3110 administration for immunomodulation in patients with pollen allergy. Allergol. Int. 2005, 54, 143–149. [Google Scholar] [CrossRef]
- Ralph, P.; Nakoinz, I. Phagocytosis and cytolysis by a macrophage tumour and its cloned cell line. Nature 1975, 257, 393–394. [Google Scholar] [CrossRef]
- Dunn, K.C.; Aotaki-Keen, A.E.; Putkey, F.R.; Hjelmeland, L.M. ARPE-19, A Human Retinal Pigment Epithelial Cell Line with Differentiated Properties. Exp. Eye Res. 1996, 62, 155–170. [Google Scholar] [CrossRef]
- Iwasaki, T. Measurements and evaluation of asthenopia by means of CFF and AA-1. Ganka 2009, 51, 387–395. [Google Scholar]
- Mukuno, K.; Ichibe, Y. Theory and practical skill of ophthalmic test (adjustment and convergence test). Rinsho Ganka 2017, 71, 79–84. [Google Scholar]
- Japanese Society of Fatigue Science. The Guideline of Clinical Evaluation of Anti-Fatigue, 5th ed.; Japanese Society of Fatigue Science: Tokyo, Japan, 2011; Available online: https://www.hirougakkai.com/guideline.pdf (accessed on 18 July 2020).


| Characteristic | KW3110 | Placebo | p Values |
|---|---|---|---|
| Mean ± SD | Mean ± SD | ||
| Number of subjects (male/female) | 40 (15/25) | 40 (13/27) | 0.6487 |
| Age (years) | 42.5 ± 3.9 | 42.4 ± 4.4 | 0.9150 |
| Height (cm) | 165.0 ± 7.6 | 161.7 ± 8.9 | 0.0787 |
| Body weight (kg) | 60.8 ± 10.2 | 59.5 ± 12.2 | 0.6059 |
| BMI (kg/m2) | 22.3 ± 2.9 | 22.7 ± 4.0 | 0.5604 |
| Body fat ratio (%) | 24.8 ± 6.1 | 25.9 ± 8.5 | 0.4973 |
| Body temperature (°C) | 36.3 ± 0.3 | 36.4 ± 0.3 | 0.5964 |
| Nonspecific IgE (IU/mL) | 105.1 ± 129.9 | 116.4 ± 177.9 | 0.7476 |
| Dominant eye | Right: 27 Left: 13 | Right: 26 Left: 14 | 1.0000 |
| CFF (Hz) | KW3110 | Placebo | ||||
|---|---|---|---|---|---|---|
| Week 0 | Week 4 | Week 8 | Week 0 | Week 4 | Week 8 | |
| 34.32 ± 1.79 | 35.51 ± 1.80 ## | 34.41 ± 2.10 | 35.33 ± 3.14 | 35.65 ± 2.43 | 34.43 ± 2.88 # | |
| Change from the week-0 value $ | 1.19 ± 1.40 *,## | 0.09 ± 1.45 * | 0.32 ± 2.24 | −0.90 ± 2.54 # | ||
| 1. | Subjects who were under treatment or had experienced malignancy, cardiac insufficiency, or heart attack |
| 2. | Subjects who were under treatment or had experienced chronic diseases (e.g., arrhythmia, hepatopathy, nephropathy, cerebrovascular disorder, rheumatism, diabetes, dyslipidemia, hypertension) |
| 3. | Subjects who were diagnosed with presbyopia or who were aware of having presbyopia previously |
| 4. | Subjects with eye disease, entropion, trichiasis, or color blindness |
| 5. | Subjects who were diagnosed with asthenopia |
| 6. | Subjects who were taking eye drops for eye diseases |
| 7. | Subjects with an uncorrected refractive error |
| 8. | Subjects who had undergone laser in situ keratomileusis |
| 9. | Subjects with severe astigmatism |
| 10. | Subjects with amblyopia or strabismus |
| 11. | Subjects with best-corrected visual acuity <1.0 for the dominant eye |
| 12. | Subjects with a diagnosis of eye fatigue caused by something other than failed neurological function or ocular adjustment function |
| 13. | Subjects who worked in a company developing or manufacturing foods with functional claims |
| 14. | Subjects with excessive alcohol-drinking behavior |
| 15. | Subjects who could not stop drinking alcoholic beverages for 2 days until the check-up |
| 16. | Subjects who regularly took drugs or health foods with potential effects on the eyes or were expecting to use them during the study |
| 17. | Subjects who could not stop taking drugs or health foods that might have effects on immune functions |
| 18. | Subjects who could not stop eating foods similar to the test foods and/or were taking drugs or health foods including lactic acid bacteria or Bifidobacterium |
| 19. | Subjects who had a tendency to get diarrhea after eating dairy products |
| 20. | Subjects with the possibility of drug and/or food allergies |
| 21. | Subjects with drug or alcohol dependence |
| 22. | Subjects who were diagnosed with pollinosis |
| 23. | Subjects who could not execute a work load test |
| 24. | Subjects who were judged as unsuitable for the study from the background questionnaire |
| 25. | Subjects who were pregnant, breastfeeding, or planning to get pregnant during the study |
| 26. | Subjects who revoked the agreement/acquisition day and/or participated in other clinical studies within 3 months or were planning to participate in other clinical studies during this study |
| 27. | Subjects who were judged as unsuitable for other reasons by the supervising physician |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamazaki, T.; Suzuki, H.; Yamada, S.; Ohshio, K.; Sugamata, M.; Yamada, T.; Morita, Y. Lactobacillus paracasei KW3110 Suppresses Inflammatory Stress-Induced Premature Cellular Senescence of Human Retinal Pigment Epithelium Cells and Reduces Ocular Disorders in Healthy Humans. Int. J. Mol. Sci. 2020, 21, 5091. https://doi.org/10.3390/ijms21145091
Yamazaki T, Suzuki H, Yamada S, Ohshio K, Sugamata M, Yamada T, Morita Y. Lactobacillus paracasei KW3110 Suppresses Inflammatory Stress-Induced Premature Cellular Senescence of Human Retinal Pigment Epithelium Cells and Reduces Ocular Disorders in Healthy Humans. International Journal of Molecular Sciences. 2020; 21(14):5091. https://doi.org/10.3390/ijms21145091
Chicago/Turabian StyleYamazaki, Takahiro, Hiroaki Suzuki, Sayuri Yamada, Konomi Ohshio, Miho Sugamata, Takahiro Yamada, and Yuji Morita. 2020. "Lactobacillus paracasei KW3110 Suppresses Inflammatory Stress-Induced Premature Cellular Senescence of Human Retinal Pigment Epithelium Cells and Reduces Ocular Disorders in Healthy Humans" International Journal of Molecular Sciences 21, no. 14: 5091. https://doi.org/10.3390/ijms21145091
APA StyleYamazaki, T., Suzuki, H., Yamada, S., Ohshio, K., Sugamata, M., Yamada, T., & Morita, Y. (2020). Lactobacillus paracasei KW3110 Suppresses Inflammatory Stress-Induced Premature Cellular Senescence of Human Retinal Pigment Epithelium Cells and Reduces Ocular Disorders in Healthy Humans. International Journal of Molecular Sciences, 21(14), 5091. https://doi.org/10.3390/ijms21145091





