Gemfibrozil Induces Anemia, Leukopenia and Reduces Hematopoietic Stem Cells via PPAR-α in Mice
Abstract
1. Introduction
2. Results
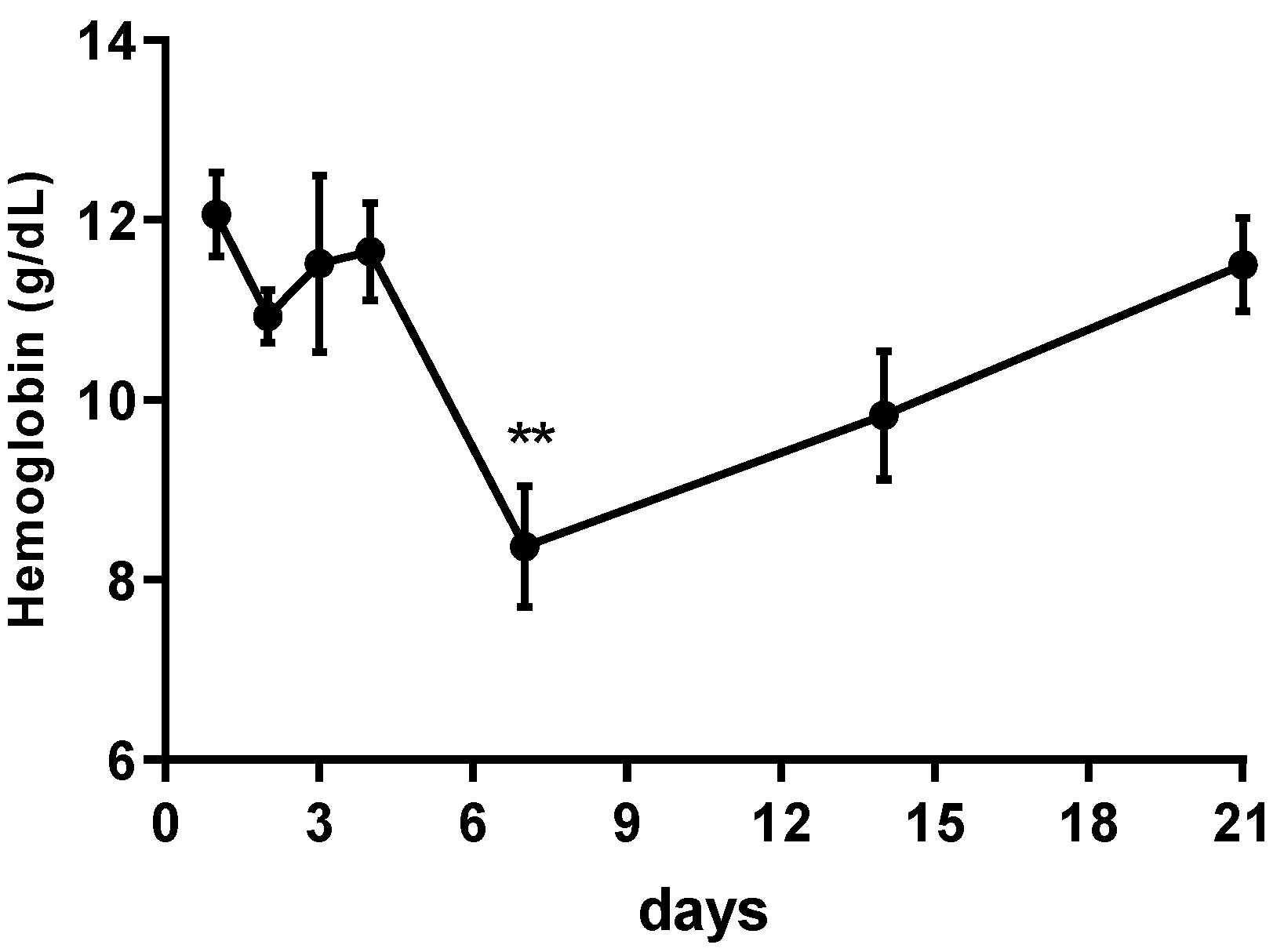
2.1. PPAR-α Deletion Prevents Gemfibrozil-Induced Anemia
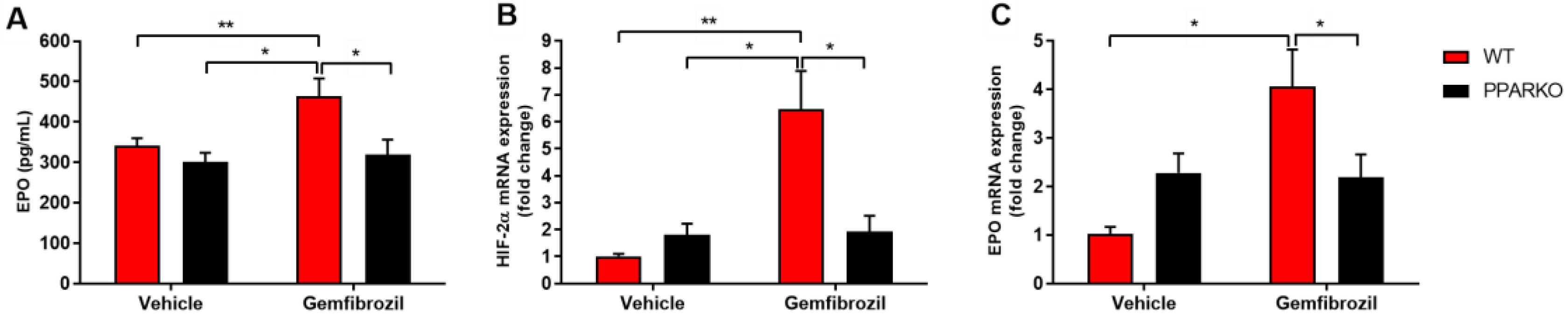
2.2. PPAR-α Deletion Blunts Gemfibrozil-Induced Increase in Serum Erythropoietin
2.3. PPAR-α-Knockout Mice Avoided the Increase in HIF-2α and Erythropoietin mRNA Levels Induced by Gemfibrozil in Renal Tissue
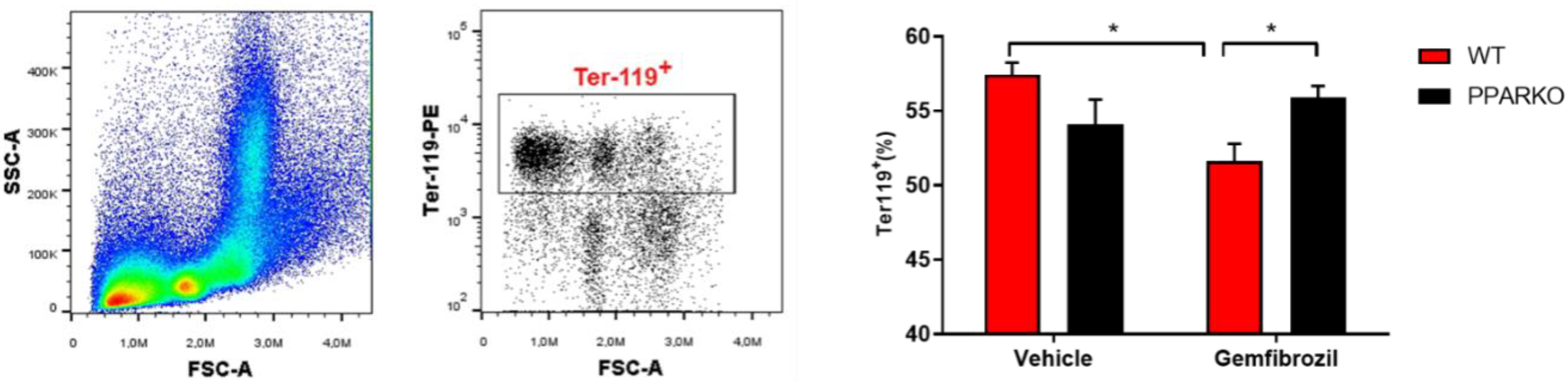
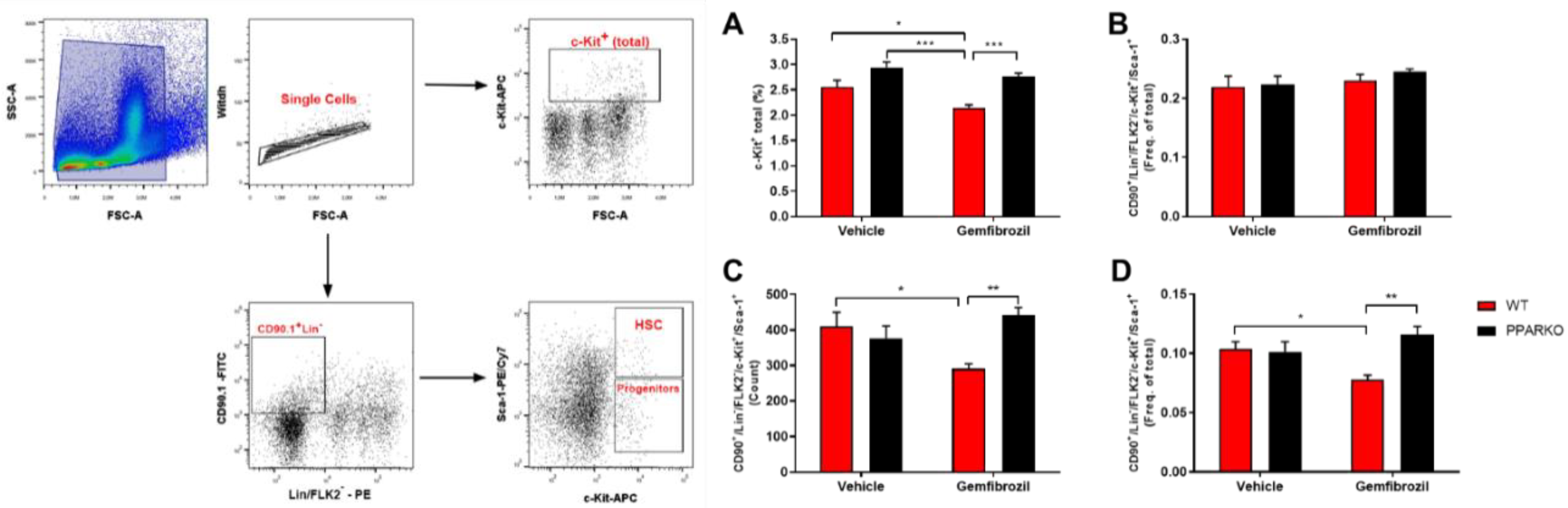
2.4. PPAR-α Ablation Prevents Gemfibrozil-Induced Decreases in Erythroid and Hematopoietic Stem Cells in the Bone Marrow
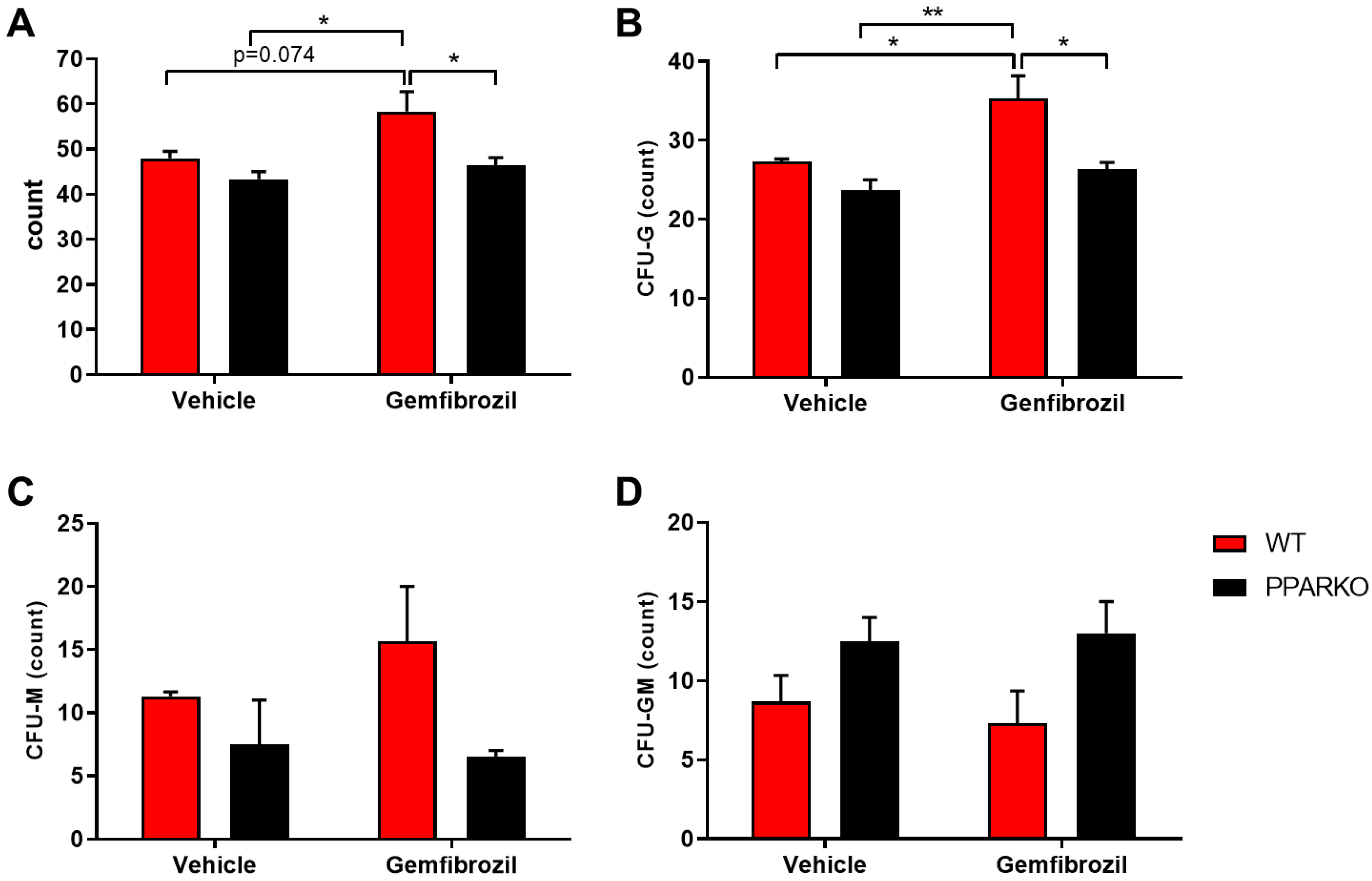
2.5. PPAR-α Deletion Blunted the Increased Levels of Colony Forming Units that Generate Myeloid Cells Induced by Gemfibrozil
3. Discussion
4. Methods
4.1. Animals
4.2. Experimental Protocol
4.3. Drug Treatment
4.4. Blood Sampling and Tissue Collection
4.5. Blood Count Test
4.6. Hemoglobin Measurement
4.7. Real-Time PCR
4.8. Serum Erythropoietin Assay
4.9. Colony-Forming-Unit Assay
4.10. Immunolabeling
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CFU | colony-forming unit |
| EPO | erythropoietin |
| HDL | high-density lipoprotein |
| HIF-2α | hypoxia-inducible factor 2 alpha |
| HSC | hematopoietic stem cell |
| LDL | low-density lipoprotein |
| PPAR-α | peroxisome proliferator-activated receptor alpha |
| RBC | red blood cells |
| WBC | white blood cells |
References
- Mercado, C.; DeSimone, A.K.; Odom, E.; Gillespie, C.; Ayala, C.; Loustalot, F. Prevalence of cholesterol treatment eligibility and medication use among adults—United States, 2005–2012. MMWR Morb. Mortal. Wkly Rep. 2015, 64, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Jakob, T.; Nordmann, A.J.; Schandelmaier, S.; Ferreira-González, I.; Briel, M. Fibrates for primary prevention of cardiovascular disease events. Cochrane Database Syst. Rev. 2016, 11, CD009753. [Google Scholar] [CrossRef] [PubMed]
- Hankey, G.J. Role of lipid-modifying therapy in the prevention of initial and recurrent stroke. Curr. Opin. Lipidol. 2002, 13, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, R.H.; Davenport, J.; Babikian, V.; Brass, L.M.; Collins, D.; Wexler, L.; Wagner, S.; Papademetriou, V.; Rutan, G.; Robins, S.J.; et al. Reduction in stroke with gemfibrozil in men with coronary heart disease and low HDL cholesterol: The Veterans Affairs HDL Intervention Trial (VA-HIT). Circulation 2001, 103, 2828–2833. [Google Scholar] [CrossRef]
- Rubins, H.B.; Robins, S.J.; Collins, D.; Fye, C.L.; Anderson, J.W.; Elam, M.B.; Faas, F.H.; Linares, E.; Schaefer, E.J.; Schectman, G.; et al. Gemfibrozil for the secondary prevention of coronary heart disease in men with low levels of high-density lipoprotein cholesterol. Veterans affairs high-density lipoprotein cholesterol intervention trial study group. N. Engl. J. Med. 1999, 341, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Athyros, V.G.; Papageorgiou, A.A.; Avramidis, M.J.; Kontopoulos, A.G. Long-term effect of gemfibrozil on coronary heart disease risk profile of patients with primary combined hyperlipidaemia. Coron. Artery Dis. 1995, 6, 251–256. [Google Scholar] [PubMed]
- Bell, H.; Dittmeier, G.; Martinez, L. Gemfibrozil therapy in patients with coronary heart disease. Mo. Med. 1988, 85, 27–30. [Google Scholar]
- Vega, G.L.; Grundy, S.M. Gemfibrozil therapy in primary hypertriglyceridemia associated with coronary heart disease. Effects on metabolism of low-density lipoproteins. JAMA 1985, 253, 2398–2403. [Google Scholar] [CrossRef]
- Kersten, S. Peroxisome proliferator activated receptors and lipoprotein metabolism. PPAR Res. 2008, 2008, 132960. [Google Scholar] [CrossRef]
- Strauss, V.; Mellert, W.; Wiemer, J.; Leibold, E.; Kamp, H.; Walk, T.; Looser, R.; Prokoudine, A.; Fabian, E.; Krennrich, G.; et al. Increased toxicity when fibrates and statins are administered in combination—A metabolomics approach with rats. Toxicol. Lett. 2012, 211, 187–200. [Google Scholar] [CrossRef]
- Kim, K.; Kleinman, H.K.; Lee, H.J.; Pahan, K. Safety and potential efficacy of gemfibrozil as a supportive treatment for children with late infantile neuronal ceroid lipofuscinosis and other lipid storage disorders. Orphanet J. Rare Dis. 2017, 12, 113. [Google Scholar] [CrossRef]
- Chaparro, C.M.; Suchdev, P.S. Anemia epidemiology, pathophysiology, and etiology in low- and middle-income countries. Ann. N. Y. Acad Sci. 2019, 1450, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Janz, T.G.; Johnson, R.L.; Rubenstein, S.D. Anemia in the emergency department: Evaluation and treatment. Emerg. Med. Pract. 2013, 15, 1–15. [Google Scholar] [PubMed]
- Smith, R.E. The clinical and economic burden of anemia. Am. J. Manag. Care 2010, 16, S59–S66. [Google Scholar] [PubMed]
- Ing, V.W. The etiology and management of leukopenia. Can. Fam. Physician 1984, 30, 1835–1839. [Google Scholar]
- Smith, T.G.; Robbins, P.A.; Ratcliffe, P.J. The human side of hypoxia-inducible factor. Br. J. Haematol. 2008, 141, 325–334. [Google Scholar] [CrossRef]
- Haase, V.H. Regulation of erythropoiesis by hypoxia-inducible factors. Blood Rev. 2013, 27, 41–53. [Google Scholar] [CrossRef]
- Barminko, J.; Reinholt, B.; Baron, M.H. Development and differentiation of the erythroid lineage in mammals. Dev. Comp. Immunol. 2016, 58, 18–29. [Google Scholar] [CrossRef]
- Barbosa, C.M.; Leon, C.M.; Nogueira-Pedro, A.; Wasinsk, F.; Araújo, R.C.; Miranda, A.; Ferreira, A.T.; Paredes-Gamero, E.J. Differentiation of hematopoietic stem cell and myeloid populations by ATP is modulated by cytokines. Cell Death Dis. 2011, 2, e165. [Google Scholar] [CrossRef]
- Fruchart, J.C.; Duriez, P.; Staels, B. Molecular mechanism of action of the fibrates. J. Soc. Biol. 1999, 193, 67–75. [Google Scholar] [CrossRef]
- Schoonjans, K.; Staels, B.; Auwerx, J. Role of the peroxisome proliferator-activated receptor (PPAR) in mediating the effects of fibrates and fatty acids on gene expression. J. Lipid Res. 1996, 37, 907–925. [Google Scholar] [PubMed]
- Paliege, A.; Rosenberger, C.; Bondke, A.; Sciesielski, L.; Shina, A.; Heyman, S.N.; Flippin, L.A.; Arend, M.; Klaus, S.J.; Bachmann, S. Hypoxia-inducible factor-2alpha-expressing interstitial fibroblasts are the only renal cells that express erythropoietin under hypoxia-inducible factor stabilization. Kidney Int. 2010, 77, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Scortegagna, M.; Ding, K.; Zhang, Q.; Oktay, Y.; Bennett, M.J.; Bennett, M.; Shelton, J.M.; Richardson, J.A.; Moe, O.; Garcia, J.A. HIF-2alpha regulates murine hematopoietic development in an erythropoietin-dependent manner. Blood 2005, 105, 3133–3140. [Google Scholar] [CrossRef] [PubMed]
- Rosenberger, C.; Mandriota, S.; Jürgensen, J.S.; Wiesener, M.S.; Hörstrup, J.H.; Frei, U.; Ratcliffe, P.J.; Maxwell, P.H.; Bachmann, S.; Eckardt, K.-U. Expression of hypoxia-inducible factor-1alpha and -2alpha in hypoxic and ischemic rat kidneys. J. Am. Soc. Nephrol. 2002, 13, 1721–1732. [Google Scholar] [CrossRef]
- Scatena, R.; Nocca, G.; Sole, P.D.; Rumi, C.; Puggioni, P.; Remiddi, F.; Bottoni, P.; Ficarra, S.; Giardina, B. Bezafibrate as differentiating factor of human myeloid leukemia cells. Cell Death Differ. 1999, 6, 781–787. [Google Scholar] [CrossRef]
- Hua, H.; Yang, J.; Lin, H.; Xi, Y.; Dai, M.; Xu, G.; Wang, F.; Liu, L.; Zhao, T.; Huang, J.; et al. PPARα-independent action against metabolic syndrome development by fibrates is mediated by inhibition of STAT3 signalling. J. Pharm. Pharmacol. 2018, 70, 1630–1642. [Google Scholar] [CrossRef]
- Blednov, Y.A.; Black, M.; Benavidez, J.M.; Stamatakis, E.E.; Harris, R.A. PPAR agonists: II. fenofibrate and tesaglitazar alter behaviors related to voluntary alcohol consumption. Alcohol. Clin. Exp. Res. 2016, 40, 563–571. [Google Scholar] [CrossRef]
- Calkin, A.C.; Giunti, S.; Jandeleit-Dahm, K.A.; Allen, T.J.; Cooper, M.E.; Thomas, M.C. PPAR-alpha and -gamma agonists attenuate diabetic kidney disease in the apolipoprotein E knockout mouse. Nephrol. Dial. Transplant. 2006, 21, 2399–2405. [Google Scholar] [CrossRef]
- Benke, K.; Mátyás, C.; Sayour, A.A.; Oláh, A.; Németh, B.T.; Ruppert, M.; Szabó, G.; Kökény, G.; Horváth, E.M.; Hartyánszky, I.; et al. Pharmacological preconditioning with gemfibrozil preserves cardiac function after heart transplantation. Sci. Rep. 2017, 7, 14232. [Google Scholar] [CrossRef]
- Nesfield, S.R.; Clarke, C.J.; Hoivik, D.J.; Miller, R.T.; Allen, J.S.; Selinger, K.; Santostefano, M.J. Evaluation of the carcinogenic potential of clofibrate in the rasH2 mouse. Int. J. Toxicol. 2005, 24, 301–311. [Google Scholar] [CrossRef]
- Nogueira-Pedro, A.; Dias, C.C.; Regina, H.; Segreto, C.; Addios, P.C.; Lungato, L.; D’Almeida, V.; Barros, C.C.; Higa, E.M.; Buri, M.V.; et al. Nitric oxide-induced murine hematopoietic stem cell fate involves multiple signaling proteins, gene expression, and redox modulation. Stem Cells 2014, 32, 2949–2960. [Google Scholar] [CrossRef] [PubMed]
- Nogueira-Pedro, A.; Barbosa, C.M.; Segreto, H.R.; Lungato, L.; D’Almeida, V.; Moraes, A.A.; Miranda, A.; Paredes-Gamero, E.J.; Ferreira, A.T. α-Tocopherol induces hematopoietic stem/progenitor cell expansion and ERK1/2-mediated differentiation. J. Leukoc. Biol. 2011, 90, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Leon, C.M.; Barbosa, C.M.; Justo, G.Z.; Borelli, P.; Resende, J.D.; de Oliveira, J.S.; Ferreira, A.T.; Paredes-Gamero, E.J. Requirement for PLCγ2 in IL-3 and GM-CSF-stimulated MEK/ERK phosphorylation in murine and human hematopoietic stem/progenitor cells. J. Cell Physiol. 2011, 226, 1780–1792. [Google Scholar] [CrossRef] [PubMed]
| WT | PPARKO | WT | PPARKO | |
|---|---|---|---|---|
| Parameters | Vehicle | Vehicle | Gemfibrozil | Gemfibrozil |
| Hemoglobin (g/dL) | 12.55 ± 0.36 | 12.37 ± 0.91 | 09.52 ± 0.89 ac | 12.64 ± 0.43 |
| Hematocrit (%) | 33.18 ± 2.05 | 32.37 ± 1.41 | 23.30 ± 0.60 ac | 28.00 ± 1.00 |
| RBC (x106/mm3) | 06.39 ± 0.42 | 06.38 ± 0.29 | 04.46 ± 0.11 abc | 05.49 ± 0.19 |
| WBC (/mm3) | 1885 ± 212 | 1680 ± 213 | 1133 ± 92.3 a | 1365 ± 88.0 |
| PLT (/mm3) | 404.0 ± 15.6 | 405.3 ± 23.3 | 351.0 ± 08.9 | 395.8 ± 15.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estrela, G.R.; Arruda, A.C.; Torquato, H.F.V.; Freitas-Lima, L.C.; Perilhão, M.S.; Wasinski, F.; Budu, A.; Fock, R.A.; Paredes-Gamero, E.J.; Araujo, R.C. Gemfibrozil Induces Anemia, Leukopenia and Reduces Hematopoietic Stem Cells via PPAR-α in Mice. Int. J. Mol. Sci. 2020, 21, 5050. https://doi.org/10.3390/ijms21145050
Estrela GR, Arruda AC, Torquato HFV, Freitas-Lima LC, Perilhão MS, Wasinski F, Budu A, Fock RA, Paredes-Gamero EJ, Araujo RC. Gemfibrozil Induces Anemia, Leukopenia and Reduces Hematopoietic Stem Cells via PPAR-α in Mice. International Journal of Molecular Sciences. 2020; 21(14):5050. https://doi.org/10.3390/ijms21145050
Chicago/Turabian StyleEstrela, Gabriel Rufino, Adriano Cleis Arruda, Heron Fernandes Vieira Torquato, Leandro Ceotto Freitas-Lima, Mauro Sérgio Perilhão, Frederick Wasinski, Alexandre Budu, Ricardo Ambrósio Fock, Edgar Julian Paredes-Gamero, and Ronaldo Carvalho Araujo. 2020. "Gemfibrozil Induces Anemia, Leukopenia and Reduces Hematopoietic Stem Cells via PPAR-α in Mice" International Journal of Molecular Sciences 21, no. 14: 5050. https://doi.org/10.3390/ijms21145050
APA StyleEstrela, G. R., Arruda, A. C., Torquato, H. F. V., Freitas-Lima, L. C., Perilhão, M. S., Wasinski, F., Budu, A., Fock, R. A., Paredes-Gamero, E. J., & Araujo, R. C. (2020). Gemfibrozil Induces Anemia, Leukopenia and Reduces Hematopoietic Stem Cells via PPAR-α in Mice. International Journal of Molecular Sciences, 21(14), 5050. https://doi.org/10.3390/ijms21145050





