Glutamatergic Signaling Along The Microbiota-Gut-Brain Axis
Abstract
1. Introduction
2. Gut Microbiota and Host Interaction
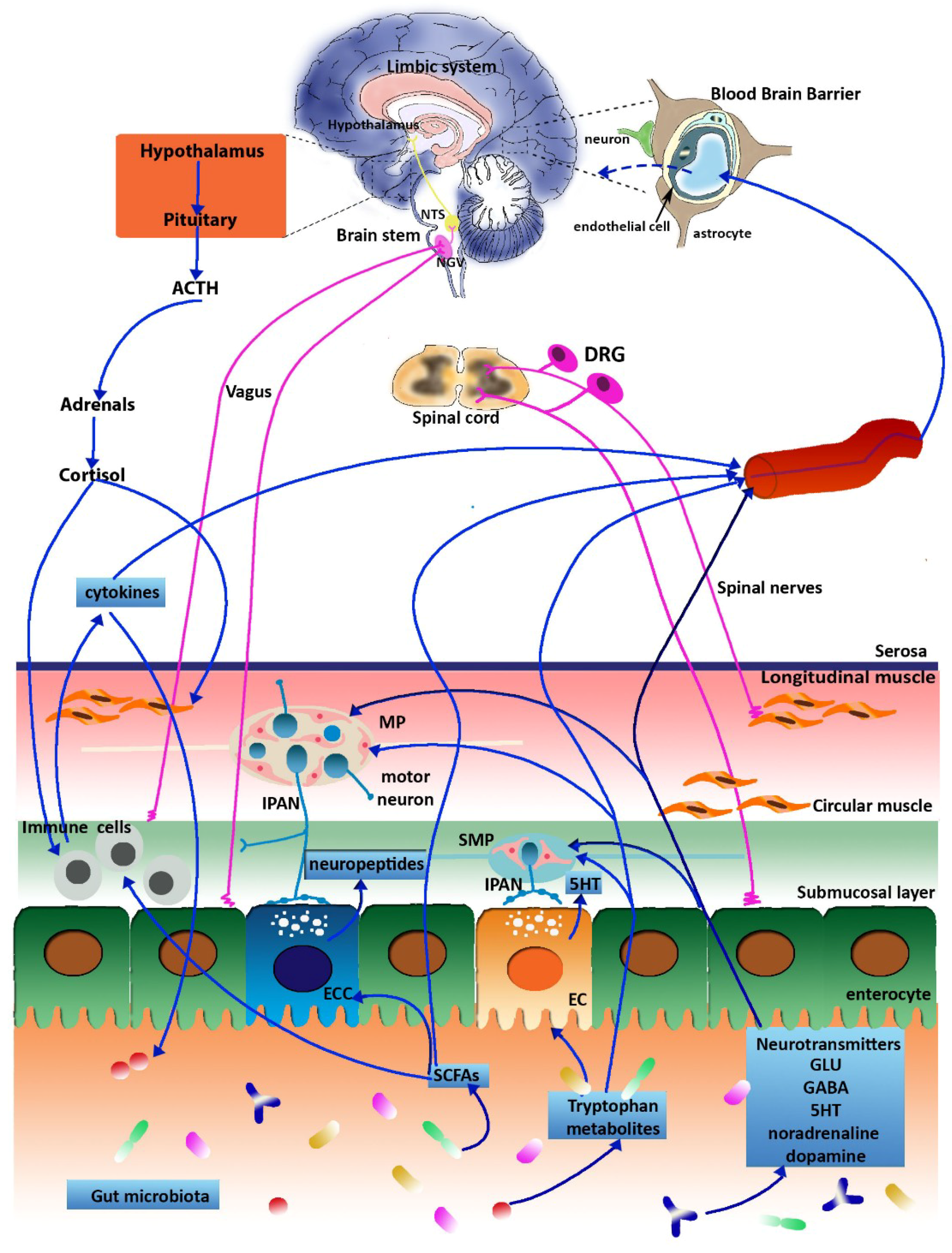
3. The Microbiota-Gut-Brain Axis
4. Sources of Glutamate along the Microbiota-Gut-Brain Axis
4.1. Dietary Sources of Glutamate
4.2. Bacterial Production of Glutamate
4.3. Glutamate as a Neurotransmitter in the CNS and ENS
4.3.1. Glutamate in the CNS
4.3.2. The Enteric Nervous System
4.3.3. Glutamatergic Enteric Neurons
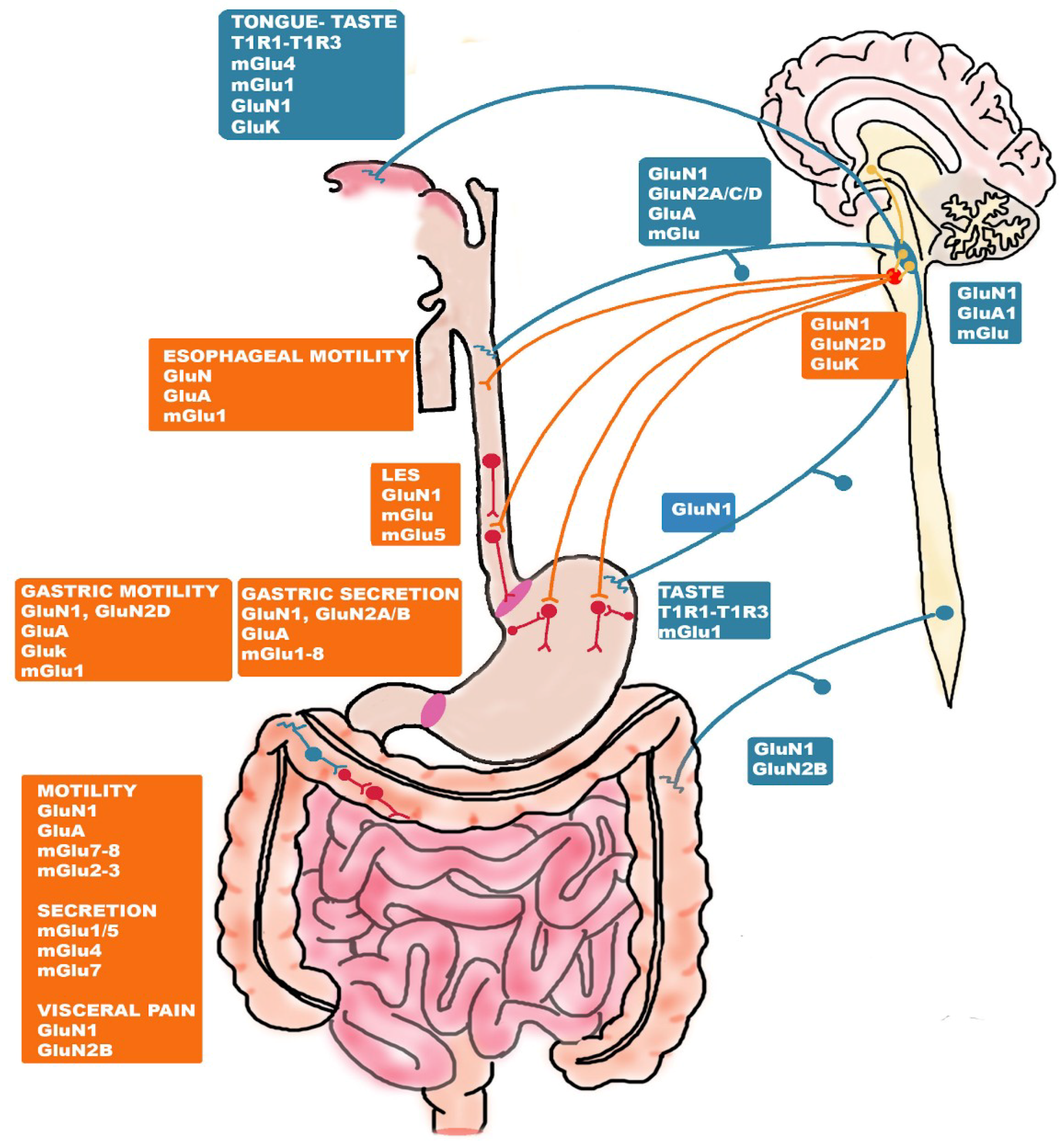
5. Glutamate Receptors: Distribution and Function along the Gut-Brain Axis
5.1. Glutamate Receptor-Mediated Taste and Savoriness
5.2. Glutamate-Receptor Mediated Control of Esophageal and Gastric Function
5.3. Glutamate-Receptor Mediated Control of the Intestinal Function
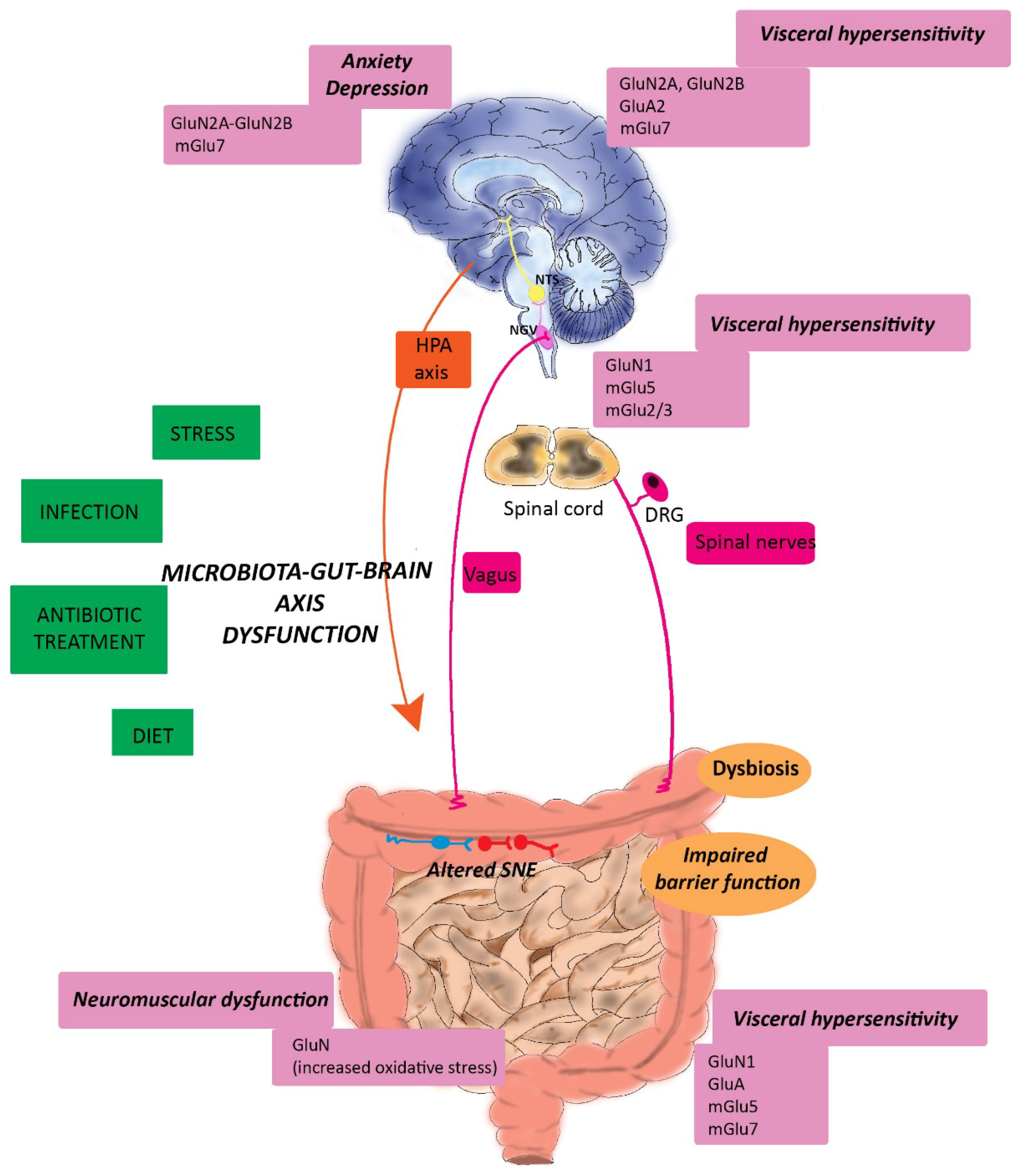
6. Glutamatergic Dysfunction along the Microbiota-Gut-Brain Axis: Relation to IBS and IBD
6.1. Irritable Bowel Syndrome
6.1.1. Glutamatergic Transmission and IBS-associated Visceral Pain
6.1.2. Glutamatergic Transmission and IBS-associated Psychiatric Disorders
6.2. Inflammatory Bowel Disorders
6.2.1. Glutamatergic Transmission and IBD-associated Neuromuscular Dysfunction
6.2.2. Glutamatergic Transmission and IBD-associated Psychiatric Disturbances
7. Perspective: Areas of Importance for Advancing the Field
8. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Rhee, S.H.; Pothoulakis, C.; Mayer, E.A. Principles and clinical implications of the brain-gut-enteric microbiota axis. Nat. Rev. Gastroenterol. Hepatol. 2009, 6, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209. [Google Scholar] [PubMed]
- Sekirov, I.; Russell, S.L.; Caetano, M.; Antunes, L.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef] [PubMed]
- Borre, Y.E.; O’Keeffe, G.W.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Microbiota and neurodevelopmental windows: Implications for brain disorders. Trends Mol. Med. 2014, 20, 509–518. [Google Scholar] [CrossRef]
- Kelly, J.R.; Minuto, C.; Cryan, J.F.; Clarke, G.; Dinan, T.G. Cross talk: The microbiota and neurodevelopmental disorders. Front. Neurosci. 2017, 11, 490. [Google Scholar] [CrossRef] [PubMed]
- Bastiaanssen, T.F.S.; Cowan, C.S.M.; Claesson, M.J.; Dinan, T.G.; Cryan, J.F. Making Sense of … the Microbiome in Psychiatry. Int. J. Neuropsychopharmacol. 2019, 22, 37–52. [Google Scholar] [CrossRef]
- Tomé, D. The roles of dietary glutamate in the intestine. Ann. Nutr. Metab. 2018, 73, 15–20. [Google Scholar] [CrossRef]
- Mazzoli, R.; Pessione, E. The neuro-endocrinological role of microbial glutamate and GABA signaling. Front. Microbiol. 2016, 7, 1934. [Google Scholar] [CrossRef] [PubMed]
- Miladinovic, T.; Nashed, M.G.; Singh, G. Overview of glutamatergic dysregulation in central pathologies. Biomolecules 2015, 5, 3112–3141. [Google Scholar] [CrossRef] [PubMed]
- Filpa, V.; Moro, E.; Protasoni, M.; Crema, F.; Frigo, G.; Giaroni, C. Role of glutamatergic neurotransmission in the enteric nervous system and brain-gut axis in health and disease. Neuropharmacology 2016, 111, 14–33. [Google Scholar] [CrossRef]
- Matsumoto, M.; Kibe, R.; Ooga, T.; Aiba, Y.; Sawaki, E.; Koga, Y.; Benno, Y. Cerebral low-molecular metabolites influenced by intestinal microbiota: A pilot study. Front. Syst. Neurosci. 2013, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Nagasawa, M.; Ikeda, H.; Yasuo, S. Gut microbiota of mice putatively modifies amino acid metabolism in the host brain. Br. J. Nutr. 2017, 117, 775–783. [Google Scholar] [CrossRef]
- Holtmann, G.J.; Ford, A.C.; Talley, N.J. Pathophysiology of irritable bowel syndrome. Lancet Gastroenterol. Hepatol. 2016, 1, 133–146. [Google Scholar] [CrossRef]
- Mikocka-Walus, A.; Knowles, S.R.; Keefer, L.; Graff, L. Controversies Revisited: A Systematic Review of the Comorbidity of Depression and Anxiety with Inflammatory Bowel Diseases. Inflamm. Bowel Dis. 2016, 22, 752–762. [Google Scholar] [CrossRef]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef]
- Deng, P.; Swanson, K.S. Gut microbiota of humans, dogs and cats: Current knowledge and future opportunities and challenges. Br. J. Nutr. 2015, 113, S6–S17. [Google Scholar] [CrossRef] [PubMed]
- Aagaard, K.; Ma, J.; Antony, K.M.; Ganu, R.; Petrosino, J.; Versalovic, J. The placenta harbors a unique microbiome. Sci. Transl. Med. 2014, 6, 237ra65. [Google Scholar] [CrossRef] [PubMed]
- Fernández, L.; Langa, S.; Martín, V.; Maldonado, A.; Jiménez, E.; Martín, R.; Rodríguez, J.M. The human milk microbiota: Origin and potential roles in health and disease. Pharmacol. Res. 2013, 69, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.M.; Murphy, K.; Stanton, C.; Ross, R.P.; Kober, O.I.; Juge, N.; Avershina, E.; Rudi, K.; Narbad, A.; Jenmalm, M.C.; et al. The composition of the gut microbiota throughout life, with an emphasis on early life. Microbes Ecol. Health Dis. 2015, 26, 26050. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, K.; Wu, W.; Giannoulatou, E.; Ho, J.W.K.; Li, L. Host and microbiome multi-omics integration: Applications and methodologies. Biophys. Rev. 2019, 11, 55–56. [Google Scholar] [CrossRef]
- Lloyd-Price, J.; Abu-Ali, G.; Huttenhower, C. The healthy human microbiome. Genome Med. 2016, 8, 51. [Google Scholar] [CrossRef]
- Fujio-Vejar, S.; Vasquez, Y.; Morales, P.; Magne, F.; Vera-Wolf, P.; Ugalde, J.A.; Navarrete, P.; Gotteland, M. The Gut Microbiota of Healthy Chilean Subjects Reveals a High Abundance of the Phylum Verrucomicrobia. Front. Microbiol. 2017, 8, 1221. [Google Scholar] [CrossRef]
- Urdaneta, V.; Casadesús, J. Interactions between Bacteria and Bile Salts in the gastrointestinal and Hepatobiliary Tracts. Front. Med. (Lausanne) 2017, 4, 163. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Bergman, E.N. Energy contributions of volatile fatty acids from the gastrointestinal tract in various species. Physiol. Rev. 1990, 70, 567–590. [Google Scholar] [CrossRef] [PubMed]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Kitamoto, S.; Nagao-Kitamoto, H.; Kuffa, P.; Kamada, N. Regulation of virulence: The rise and fall of gastrointestinal pathogens. J. Gastroenterol. 2016, 51, 195–205. [Google Scholar] [CrossRef]
- Martin, C.R.; Osadchiy, V.; Kalani, A.; Mayer, E.A. The Brain-Gut-Microbiome Axis. Cell. Mol. Gastroenterol. Hepatol. 2018, 6, 133–148. [Google Scholar] [CrossRef]
- Furness, J.B.; Callaghan, B.P.; Rivera, L.R.; Cho, H.-J. The enteric nervous system and gastrointestinal innervation: Integrated local and central control. Adv. Exp. Med. Biol. 2014, 817, 39–71. [Google Scholar]
- O’Mahony, S.M.; Hyland, N.P.; Dinan, T.G.; Cryan, J.F. Maternal separation as a model of brain-gut axis dysfunction. Psychopharmacology 2011, 214, 71–88. [Google Scholar] [CrossRef]
- Mulak, A.; Bonaz, B. Irritable bowel syndrome: A model of the brain-gut interactions. Med Sci. Monit. 2004, 10, RA55–RA62. [Google Scholar] [PubMed]
- Cani, P.D.; Knauf, C. How gut microbes talk to organs: The role of endocrine and nervous routes. Mol. Metab. 2016, 5, 743–752. [Google Scholar] [CrossRef]
- Holzer, P.; Farzi, A. Neuropeptides and the microbiota- Gut-brain axis. Adv. Exp. Med. Biol. 2014, 817, 195–219. [Google Scholar]
- Janik, R.; Thomason, L.A.M.; Stanisz, A.M.; Forsythe, P.; Bienenstock, J.; Stanisz, G.J. Magnetic resonance spectroscopy reveals oral Lactobacillus promotion of increases in brain GABA, N-acetyl aspartate and glutamate. Neuroimage 2016, 125, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Moloney, R.D.; Johnson, A.C.; O’Mahony, S.M.; Dinan, T.G.; Greenwood-Van Meerveld, B.; Cryan, J.F. Stress and the microbiota-gut-brain axis in visceral pain: Relevance to irritable bowel syndrome. CNS Neurosci. Ther. 2016, 22, 102–117. [Google Scholar] [CrossRef]
- Agus, A.; Planchais, J.; Sokol, H. Gut Microbiota Regulation of Tryptophan Metabolism in Health and Disease. Cell Host Microbe 2018, 23, 716–724. [Google Scholar] [CrossRef]
- Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Kynurenine pathway metabolism and the microbiota-gut-brain axis. Neuropharmacology 2017, 112, 399–412. [Google Scholar] [CrossRef]
- Lugo-Huitrón, R.; Ugalde Muñiz, P.; Pineda, B.; Pedraza-Chaverrí, J.; Ríos, C.; Pérez-De La Cruz, V. Quinolinic acid: An endogenous neurotoxin with multiple targets. Oxid. Med. Cell. Longev. 2013, 2013, 104024. [Google Scholar] [CrossRef]
- Ramos-Chávez, L.A.; Lugo Huitrón, R.; González Esquivel, D.; Pineda, B.; Ríos, C.; Silva-Adaya, D.; Sánchez-Chapul, L.; Roldán-Roldán, G.; Pérez de la Cruz, V. Relevance of Alternative Routes of Kynurenic Acid Production in the Brain. Oxid. Med. Cell. Longev. 2018, 2018, 5272741. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Xu, K.; Liu, H.; Liu, G.; Bai, M.; Peng, C.; Li, T.; Yin, Y. Impact of the gut microbiota on intestinal immunity mediated by tryptophan metabolism. Front. Cell. Infect. Microbiol. 2018, 8, 13. [Google Scholar] [CrossRef]
- Kaszaki, J.; Érces, D.; Varga, G.; Szabó, A.; Vécsei, L.; Boros, M. Kynurenines and intestinal neurotransmission: The role of N-methyl-d-aspartate receptors. J. Neural Transm. 2012, 119, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Briguglio, M.; Dell’Osso, B.; Panzica, G.; Malgaroli, A.; Banfi, G.; Dina, C.Z.; Galentino, R.; Porta, M. Dietary neurotransmitters: A narrative review on current knowledge. Nutrients 2018, 10, 591. [Google Scholar] [CrossRef]
- Burrin, D.G.; Stoll, B. Metabolic fate and function of dietary glutamate in the gut. Am. J. Clin. Nutr. 2009, 90, 850S–856S. [Google Scholar] [CrossRef] [PubMed]
- Blachier, F.; Boutry, C.; Bos, C.; Tomé, D. Metabolism and functions of L-glutamate in the epithelial cells of the small and large intestines. Am. J. Clin. Nutr. 2009, 90, 814S–821S. [Google Scholar] [CrossRef]
- Janeczko, M.J.; Stoll, B.; Chang, X.; Guan, X.; Burrin, D.G. Extensive gut metabolism limits the intestinal absorption of excessive supplemental dietary glutamate loads in infant pigs. J. Nutr. 2007, 137, 2384–2390. [Google Scholar] [CrossRef] [PubMed]
- Haÿs, S.P.; Ordonez, J.M.; Burrin, D.G.; Sunehag, A.L. Dietary glutamate is almost entirely removed in its first pass through the splanchnic bed in premature infants. Pediatr. Res. 2007, 62, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Reeds, P.J.; Burrin, D.G.; Stoll, B.; Jahoor, F. Intestinal glutamate metabolism. J. Nutr. 2000, 130 (Suppl. 4S), 978S–982S. [Google Scholar] [CrossRef]
- Brosnan, J.T.; Drewnowski, A.; Friedman, M.I. Is there a relationship between dietary MSG obesity in animals or humans? Amino Acids 2014, 46, 2075–2087. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.R.; Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G.; Hyland, N.P. Breaking down the barriers: The gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front. Cell. Neurosci. 2015, 9, 392. [Google Scholar] [CrossRef]
- Braniste, V.; Al-Asmakh, M.; Kowal, C.; Anuar, F.; Abbaspour, A.; Tóth, M.; Korecka, A.; Bakocevic, N.; Guan, N.L.; Kundu, P.; et al. The gut microbiota influences blood-brain barrier permeability in mice. Sci. Transl. Med. 2014, 6, 263ra158. [Google Scholar] [CrossRef]
- Nakayama, Y.; Hashimoto, K.-I.; Sawada, Y.; Sokabe, M.; Kawasaki, H.; Martinac, B. Corynebacterium glutamicum mechanosensitive channels: Towards unpuzzling “glutamate efflux” for amino acid production. Biophys. Rev. 2018, 10, 1359–1369. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, S.; Rodríguez-Sanoja, R.; Ramos, A.; Demain, A.L. Our microbes not only produce antibiotics, they also overproduce amino acids. J. Antibiot. 2018, 71, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Zareian, M.; Ebrahimpour, A.; Bakar, F.A.; Mohamed, A.K.S.; Forghani, B.; Ab-Kadir, M.S.B.; Saari, N. A glutamic acid-producing lactic acid bacteria isolated from malaysian fermented foods. Int. J. Mol. Sci. 2012, 13, 5482–5497. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Cul, C.; Mayer, M.L.; Gouaux, E. Functional characterization of a potassium-selective prokaryotic glutamate receptor. Nature 1999, 402, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Ger, M.-F.; Rendon, G.; Tilson, J.L.; Jakobsson, E. Domain-based identification and analysis of glutamate receptor ion channels and their relatives in prokaryotes. PLoS ONE 2010, 5, e12827. [Google Scholar] [CrossRef]
- Pessione, E. Lactic acid bacteria contribution to gut microbiota complexity: Lights and shadows. Front. Cell. Infect. Microbiol. 2012, 2, 86. [Google Scholar] [CrossRef]
- Tsai, M.-F.; Miller, C. Substrate selectivity in arginine-dependent acid resistance in enteric bacteria. Proc. Natl. Acad. Sci. USA 2013, 110, 5893–5897. [Google Scholar] [CrossRef] [PubMed]
- Nand, K.; Rao, D.V. Arthrobacter mysorens—A new species excreting L-glutamic acid. Zentralblatt fur Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene. Zweite naturwissenschaftliche Abt. Allgemeine landwirtschaftliche und technische Mikrobiologie 1972, 127, 324–331. [Google Scholar]
- Nadeem, S.; Niaz, B.; Rajoka, M.I.; Al-Ghanim, K.A.; Muzammil, H.M.; Mahboob, S.; Nadeem, A.; Yousaf, S.; Shakoori, A.R. Optimizing different vitamins for L-glutamic acid production by Brevibacterium strain NIAB SS59. Pak. J. Zool. 2014, 46, 733–739. [Google Scholar]
- Yugandhar, N.M.; Kiran Babu, U.; Raju, C.A.I.; Jaya Raju, K.; Sri Rami Reddy, D. Optimization of glutamic acid production by brevibacterium roseum. Res. J. Microbiol. 2010, 5, 1150–1154. [Google Scholar]
- Yang, J.; Yang, S. Comparative analysis of Corynebacterium glutamicum genomes: A new perspective for the industrial production of amino acids. BMC Genom. 2017, 18, 240. [Google Scholar] [CrossRef]
- Persicke, M.; Albersmeier, A.; Bednarz, H.; Niehaus, K.; Kalinowski, J.; Rückert, C. Genome sequence of the soil bacterium Corynebacterium callunae type strain DSM 20147. Stand. Genom. Sci. 2015, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, H.; Anazawa, H.; Katsumata, R.; Araki, K.; Teshiba, S. Amino Acid Production from Methanol by Methylobacillus Glycogenes Mutants: Isolation of L-Glutamic Acid Hyper-producing Mutants from M. glycogenes Strains, and Derivation of l-Threonine and l-Lysine-producing Mutants from Them. Biosci. Biotechnol. Biochem. 1993, 57, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Niciu, M.J.; Kelmendi, B.; Sanacora, G. Overview of glutamatergic neurotransmission in the nervous system. Pharmacol. Biochem. Behav. 2012, 100, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, R.A. The blood-brain barrier and glutamate. Am. J. Clin. Nutr. 2009, 90, 867S–874S. [Google Scholar] [CrossRef] [PubMed]
- Bai, W.; Zhou, Y.-G. Homeostasis of the intraparenchymal-blood glutamate concentration gradient: Maintenance, imbalance, and regulation. Front. Mol. Neurosci. 2017, 10, 400. [Google Scholar] [CrossRef]
- McKenna, M.C. Glutamate pays its own way in astrocytes. Front. Endocrinol. 2013, 4, 191. [Google Scholar] [CrossRef]
- Zhou, Y.; Danbolt, N.C. Glutamate as a neurotransmitter in the healthy brain. J. Neural Transm. 2014, 121, 799–817. [Google Scholar] [CrossRef] [PubMed]
- Reimer, R.J. SLC17: A functionally diverse family of organic anion transporters. Mol. Asp. Med. 2013, 34, 350–359. [Google Scholar] [CrossRef]
- Zhou, Y.; Danbolt, N.C. GABA and glutamate transporters in brain. Front. Endocrinol. 2013, 4, 165. [Google Scholar] [CrossRef]
- Danbolt, N.C.; Furness, D.N.; Zhou, Y. Neuronal vs glial glutamate uptake: Resolving the conundrum. Neurochem. Int. 2016, 98, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Pál, B. Involvement of extrasynaptic glutamate in physiological and pathophysiological changes of neuronal excitability. Cell. Mol. Life Sci. 2018, 75, 2917–2949. [Google Scholar] [CrossRef] [PubMed]
- Furness, J.B. The enteric nervous system and neurogastroenterology. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D. Cellular Neurophysiology of Enteric Neurons. In Physiology of the Gastrointestinal Tract, 5th ed.; Johnson, L.R., Kaunitz, J.D., Ghishan, F.K., Merchant, J.L., Said, H.M., Wood, J.D., Eds.; Elsevier: San Diego, CA, USA, 2012; pp. 629–669. [Google Scholar]
- Vermeulen, W.; De Man, J.G.; Pelckmans, P.A.; De Winter, B.Y. Neuroanatomy of lower gastrointestinal pain disorders. World J. Gastroenterol. 2014, 20, 1005–1020. [Google Scholar] [CrossRef] [PubMed]
- Clerc, N.; Furness, J.B. Intrinsic primary afferent neurons of the digestive tract. Neurogastroenterol. Motil. 2004, 16, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Ceccotti, C.; Giaroni, C.; Bistoletti, M.; Viola, M.; Crema, F.; Terova, G. Neurochemical characterization of myenteric neurons in the juvenile gilthead sea bream (Sparus aurata) intestine. PLoS ONE 2018, 13, e0201760. [Google Scholar] [CrossRef] [PubMed]
- Giaroni, C. Purinergic signaling and development of the autonomic nervous system. Auton. Neurosci. Basic Clin. 2015, 191, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Filpa, V.; Carpanese, E.; Marchet, S.; Pirrone, C.; Conti, A.; Rainero, A.; Moro, E.; Chiaravalli, A.M.; Zucchi, I.; Moriondo, A.; et al. Nitric oxide regulates homeoprotein OTX1 and OTX2 expression in the rat myenteric plexus after intestinal ischemia-reperfusion injury. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 312, G374–G389. [Google Scholar] [CrossRef] [PubMed]
- Giaroni, C.; Zanetti, E.; Pascale, A.; Oldrini, R.; Canciani, L.; Giuliani, D.; Amadio, M.; Chiaravalli, A.; Lecchini, S.; Frigo, G. Involvement of C2+-dependent PKCs in the adaptive changes of μ-opioid pathways to sympathetic denervation in the guinea pig colon. Biochem. Pharmacol. 2009, 78, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- Caputi, V.; Marsilio, I.; Filpa, V.; Cerantola, S.; Orso, G.; Bistoletti, M.; Paccagnella, N.; De Martin, S.; Montopoli, M.; Dall’Acqua, S.; et al. Antibiotic-induced dysbiosis of the microbiota impairs gut neuromuscular function in juvenile mice. Br. J. Pharmacol. 2017, 174, 3623–3639. [Google Scholar] [CrossRef]
- Gulbransen, B.D.; Sharkey, K.A. Novel functional roles for enteric glia in the gastrointestinal tract. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 625–632. [Google Scholar] [CrossRef]
- Bin, A.; Caputi, V.; Bistoletti, M.; Montopoli, M.; Colucci, R.; Antonioli, L.; De Martin, S.; Castagliuolo, I.; Orso, G.; Giaroni, C.; et al. The ecto-enzymes CD73 and adenosine deaminase modulate 5′-AMP-derived adenosine in myofibroblasts of the rat small intestine. Purinergic Signal. 2018, 14, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, M.; Cerdà-Cuéllar, M.; Martínez, V. Antibiotic-induced dysbiosis alters host-bacterial interactions and leads to colonic sensory and motor changes in mice. Gut Microbes 2015, 6, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Husebye, E.; Hellström, P.M.; Sundler, F.; Chen, J.; Midtvedt, T. Influence of microbial species on small intestinal myoelectric activity and transit in germ-free rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 280, G368–G380. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.-K.; Kasper, D.L.; Wang, B.; Forsythe, P.; Bienenstock, J.; Kunze, W.A. Bacteroides fragilis polysaccharide A is necessary and sufficient for acute activation of intestinal sensory neurons. Nat. Commun. 2013, 4, 1465. [Google Scholar] [CrossRef]
- Kunze, W.A.; Mao, Y.-K.; Wang, B.; Huizinga, J.D.; Ma, X.; Forsythe, P.; Bienenstock, J. Lactobacillus reuteri enhances excitability of colonic AH neurons by inhibiting calcium-dependent potassium channel opening. J. Cell. Mol. Med. 2009, 13, 2261–2270. [Google Scholar] [CrossRef]
- Mcvey Neufeld, K.-A.; Mao, Y.K.; Bienenstock, J.; Foster, J.A.; Kunze, W.A. The microbiome is essential for normal gut intrinsic primary afferent neuron excitability in the mouse. Neurogastroenterol. Motil. 2013, 25, 183-e88. [Google Scholar] [CrossRef]
- Mcvey Neufeld, K.A.; Perez-Burgos, A.; Mao, Y.K.; Bienenstock, J.; Kunze, W.A. The gut microbiome restores intrinsic and extrinsic nerve function in germ-free mice accompanied by changes in calbindin. Neurogastroenterol. Motil. 2015, 27, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Perez-Burgos, A.; Wang, B.; Mao, Y.-K.; Mistry, B.; Neufeld, K.-A.M.; Bienenstock, J.; Kunze, W. Psychoactive bacteria Lactobacillus rhamnosus (JB-1) elicits rapid frequency facilitation in vagal afferents. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G211–G220. [Google Scholar] [CrossRef] [PubMed]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef]
- Wang, G.-D.; Wang, X.-Y.; Xia, Y.; Wood, J.D. Dietary glutamate: Interactions with the Enteric Nervous System. J. Neurogastroenterol. Motil. 2014, 20, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Gershon, M.D.; Bursztajn, S. Properties of the enteric nervous system: Limitation of access of intravascular macromolecules to the myenteric plexus and muscularis externa. J. Comp. Neurol. 1978, 180, 467–487. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-T.; Rothstein, J.D.; Gershon, M.D.; Kirchgessner, A.L. Glutamatergic enteric neurons. J. Neurosci. 1997, 17, 4764–4784. [Google Scholar] [CrossRef]
- Giaroni, C.; Zanetti, E.; Chiaravalli, A.M.; Albarello, L.; Dominioni, L.; Capella, C.; Lecchini, S.; Frigo, G. Evidence for a glutamatergic modulation of the cholinergic function in the human enteric nervous system via NMDA receptors. Eur. J. Pharmacol. 2003, 476, 63–69. [Google Scholar] [CrossRef]
- Wiley, J.W.; Lu, Y.; Owyang, C. Evidence for a glutamatergic neural pathway in the myenteric plexus. Am. J. Physiol. Gastrointest. Liver Physiol. 1991, 261, G693–G700. [Google Scholar] [CrossRef]
- Tsai, L.H.; Tsai, W.; Wu, J.Y. Effect of L-glutamic acid on acid secretion and immunohistochemical localization of glutamatergic neurons in the rat stomach. J. Neurosci. Res. 1994, 38, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Reis, H.J.; Massensini, A.R.; Prado, M.A.M.; Gomez, R.S.; Gomez, M.V.; Romano-Silva, M.A. Calcium channels coupled to depolarization-evoked glutamate release in the myenteric plexus of guinea-pig ileum. Neuroscience 2000, 101, 237–242. [Google Scholar] [CrossRef]
- Giuliani, D.; Giaroni, C.; Zanetti, E.; Canciani, L.; Borroni, P.; Lecchini, S.; Frigo, G. Involvement of glutamate receptors of the NMDA type in the modulation of acetylcholine and glutamate overflow from the guinea pig ileum during in vitro hypoxia and hypoglycaemia. Neurochem. Int. 2006, 48, 191–200. [Google Scholar] [CrossRef]
- Tsai, H.T. Function of GABAergic and glutamatergic neurons in the stomach. J. Biomed. Sci. 2005, 12, 255–266. [Google Scholar] [CrossRef]
- Neunlist, M.; Michel, K.; Reiche, D.; Dobreva, G.; Huber, K.; Schemann, M. Glycine activates myenteric neurons in adult guinea-pigs. J. Physiol. 2001, 536, 727–739. [Google Scholar] [CrossRef]
- Seifi, M.; Swinny, J.D. Immunolocalization of AMPA receptor subunits within the enteric nervous system of the mouse colon and the effect of their activation on spontaneous colonic contractions. Neurogastroenterol. Motil. 2016, 28, 705–720. [Google Scholar] [CrossRef]
- Schousboe, A.; Waagepetersen, H.S. Role of astrocytes in glutamate homeostasis: Implications for excitotoxicity. Neurotox. Res. 2005, 8, 221–225. [Google Scholar] [CrossRef]
- Jessen, K.R.; Mirsky, R. Astrocyte-like glia in the peripheral nervous system: An immunohistochemical study of enteric glia. J. Neurosci. 1983, 3, 2206–2218. [Google Scholar] [CrossRef]
- Carpanese, E.; Moretto, P.; Filpa, V.; Marchet, S.; Moro, E.; Crema, F.; Frigo, G.; Giaroni, C. Antagonism of ionotropic glutamate receptors attenuates chemical ischemia-induced injury in rat primary cultured myenteric ganglia. PLoS ONE 2014, 9, e113613. [Google Scholar] [CrossRef] [PubMed]
- Von Boyen, G.; Steinkamp, M.; Adler, G.; Kirsch, J. Glutamate receptor subunit expression in primary enteric glia cultures. J. Recept. Signal Transduct. 2006, 26, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Iwanaga, T.; Goto, M.; Watanabe, M. Cellular distribution of glutamate transporters in the gastrointestinal tract of mice. An immunohistochemical and in situ hybridization approach. Biomed. Res. 2006, 26, 271–278. [Google Scholar] [CrossRef]
- Tong, Q.; Ma, J.; Kirchgessner, A.L. Vesicular glutamate transporter 2 in the brain-gut axis. NeuroReport 2001, 12, 3929–3934. [Google Scholar] [CrossRef]
- Brumovsky, P.R.; Robinson, D.R.; La, J.-H.; Seroogy, K.B.; Lundgren, K.H.; Albers, K.M.; Kiyatkin, M.E.; Seal, R.P.; Edwards, R.H.; Watanabe, M.; et al. Expression of vesicular glutamate transporters type 1 and 2 in sensory and autonomic neurons innervating the mouse colorectum. J. Comp. Neurol. 2011, 519, 3346–3366. [Google Scholar] [CrossRef]
- Traynelis, S.F.; Wollmuth, L.P.; McBain, C.J.; Menniti, F.S.; Vance, K.M.; Ogden, K.K.; Hansen, K.B.; Yuan, H.; Myers, S.J.; Dingledine, R. Glutamate receptor ion channels: Structure, regulation, and function. Pharmacol. Rev. 2010, 62, 405–496. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.B.; Yi, F.; Perszyk, R.E.; Furukawa, H.; Wollmuth, L.P.; Gibb, A.J.; Traynelis, S.F. Structure, function, and allosteric modulation of NMDA receptors. J. Gen. Physiol. 2018, 150, 1081–1105. [Google Scholar]
- Nicoletti, F.; Bockaert, J.; Collingridge, G.L.; Conn, P.J.; Ferraguti, F.; Schoepp, D.D.; Wroblewski, J.T.; Pin, J.P. Metabotropic glutamate receptors: From the workbench to the bedside. Neuropharmacology 2011, 60, 1017–1041. [Google Scholar] [CrossRef]
- Julio-Pieper, M.; Flor, P.J.; Dinan, T.G.; Cryan, J.F. Exciting times beyond the brain: Metabotropic glutamate receptors in peripheral and non-neural tissues. Pharmacol. Rev. 2011, 63, 35–58. [Google Scholar] [CrossRef]
- Kinnamon, S.C. Umami taste transduction mechanisms. Am. J. Clin. Nutr. 2009, 90, 753S–755S. [Google Scholar] [CrossRef]
- Tsurugizawa, T.; Uneyama, H.; Torii, K. Brain amino acid sensing. Diabetes Obes. Metab. 2014, S1, 41–48. [Google Scholar] [CrossRef] [PubMed]
- De Araujo, I.E.; Simon, S.A. The gustatory cortex and multisensory integration. Int. J. Obes. 2009, 33 (Suppl. 2), S34–S43. [Google Scholar] [CrossRef]
- Vandenbeuch, A.; Kinnamon, S.C. Glutamate: Tastant and Neuromodulator in Taste Buds. Adv. Nutr. 2016, 7, 823S–827S. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.Q.; Zhang, Y.; Hoon, M.A.; Chandrashekar, J.; Erlenbach, I.; Ryba, N.J.P.; Zuker, C.S. The receptors for mammalian sweet and umami taste. Cell 2003, 115, 255–266. [Google Scholar] [CrossRef]
- Kitamura, A.; Sato, W.; Uneyama, H.; Torii, K.; Niijima, A. Effects of intragastric infusion of inosine monophosphate and l-glutamate on vagal gastric afferent activity and subsequent autonomic reflexes. J. Physiol. Sci. 2011, 61, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Pal Choudhuri, S.; Delay, R.J.; Delay, E.R. Metabotropic glutamate receptors are involved in the detection of IMP and L-amino acids by mouse taste sensory cells. Neuroscience 2016, 316, 94–108. [Google Scholar] [CrossRef]
- Roper, S.D.; Chaudhari, N. Taste buds: Cells, signals and synapses. Nat. Rev. Neurosci. 2017, 18, 485–497. [Google Scholar] [CrossRef]
- Kusuhara, Y.; Yoshida, R.; Ohkuri, T.; Yasumatsu, K.; Voigt, A.; Hübner, S.; Maeda, K.; Boehm, U.; Meyerhof, W.; Ninomiya, Y. Taste responses in mice lacking taste receptor subunit T1R1. J. Physiol. 2013, 591, 1967–1985. [Google Scholar] [CrossRef]
- Yasumatsu, K.; Manabe, T.; Yoshida, R.; Iwatsuki, K.; Uneyama, H.; Takahashi, I.; Ninomiya, Y. Involvement of multiple taste receptors in umami taste: Analysis of gustatory nerve responses in metabotropic glutamate receptor 4 knockout mice. J. Physiol. 2015, 593, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, A.S.; Maekawa, T.; Uneyama, H.; Torii, K. Metabotropic glutamate receptor type 1 in taste tissue. Am. J. Clin. Nutr. 2009, 90, 743S–746S. [Google Scholar] [CrossRef] [PubMed]
- Danilova, V.; Hellekant, G. Comparison of the responses of the chorda tympani and glossopharyngeal nerves to taste stimuli in C57BL/6J mice. BMC Neurosci. 2003, 4, 5. [Google Scholar] [CrossRef]
- Caicedo, A.; Kim, K.-N.; Roper, S.D. Glutamate-induced cobalt uptake reveals non-NMDA receptors in rat taste cells. J. Comp. Neurol. 2000, 417, 315–324. [Google Scholar] [CrossRef]
- Chung, K.-M.; Lee, S.-B.; Heur, R.; Cho, Y.-K.; Lee, C.-H.; Jung, H.-Y.; Chung, S.-H.; Lee, S.-P.; Kim, K.-N. Glutamate-induced cobalt uptake elicited by kainate receptors in rat taste bud cells. Chem. Sens. 2005, 30, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Höfer, D.; Püschel, B.; Drenckhahn, D. Taste receptor-like cells in the rat gut identified by expression of α-gustducin. Proc. Natl. Acad. Sci. USA 1996, 93, 6631–6634. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, E.; Uneyama, H.; Torii, K. Gastrointestinal nutrient chemosensing and the gut-brain axis: Significance of glutamate signaling for normal digestion. J. Gastroenterol. Hepatol. 2013, 28, 2–8. [Google Scholar] [CrossRef]
- Tsurugizawa, T.; Uematsu, A.; Nakamura, M.; Hirota, M.; Kondoh, T.; Uneyama, H.; Torii, K. Mechanisms of neural response to gastrointestinal nutritive stimuli: The gut-brain axis. Gastroenterology 2009, 137, 262–273. [Google Scholar] [CrossRef]
- Lu, W.Y.; Bieger, D. Vagal afferent transmission in the NTS mediating reflex responses of the rat esophagus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1998, 274, R1436–R1445. [Google Scholar] [CrossRef]
- Broussard, D.L.; Bao, X.; Li, X.; Altschuler, S.M. Co-localization of NOS and NMDA receptor in esophageal premotor neurons of the rat. Neuroreport 1995, 6, 2073–2076. [Google Scholar] [CrossRef]
- Rossiter, C.D.; Norman, W.P.; Jain, M.; Hornby, P.J.; Benjamin, S.; Gillis, R.A. Control of lower esophageal sphincter pressure by two sites in dorsal motor nucleus of the vagus. Am. J. Physiol. 1990, 259, G899–G906. [Google Scholar] [CrossRef]
- Kohjitani, A.; Funahashi, M.; Miyawaki, T.; Hanazaki, M.; Matsuo, R.; Shimada, M. Peripheral N-methtl-d-aspartate receptors modulate nonadrenergic noncholinergic lower esophageal sphincter relaxation in rabbits. Anesth. Analg. 2005, 101, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Page, A.J.; Young, R.L.; Martin, C.M.; Umaerus, M.; O’Donnell, T.A.; Cooper, N.J.; Coldwell, J.R.; Hulander, M.; Mattsson, J.P.; Lehmann, A.; et al. Metabotropic glutamate receptors inhibit mechanosensitivity in vagal sensory neurons. Gastroenterology 2005, 128, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Slattery, J.A.; Page, A.J.; Dorian, C.L.; Brierley, S.M.; Blackshaw, L.A. Potentiation of mouse vagal afferent machanosensitivity by ionotropic and metabotropic glutamate receptors. J. Physiol. 2006, 577, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Akiba, Y.; Watanabe, C.; Mizumori, M.; Kaunitz, J.D. Luminal Glutamate enhances duodenal mucosal defense mechanisms via multiple glutamate receptors in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 297, G781–G791. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.J.; Yoo, B.C.; Lim, S.B.; Jeong, S.Y.; Kim, W.H.; Park, J.G. Metabotropic glutamate receptor 4 expression in colorectal carcinoma and its prognostic significance. Clin. Cancer Res. 2005, 11, 3288–3295. [Google Scholar] [CrossRef]
- Hirsch, D.P.; Tytgat, G.N.; Boeckxstaens, G.E. Is glutamate involved in transient lower esophageal sphincter relaxations? Digestive Disease and Sciences 2002, 47, 661–666. [Google Scholar] [CrossRef]
- Boeckxstaens, G.E. Alterations confined to the gastrooesophageal junction: The relationship between low LOSP, TLOSRs, hiatus hernia and acid pocket. Best Pract. Res. Clin. Gastroenterol. 2010, 24, 821–829. [Google Scholar] [CrossRef]
- Frisby, C.L.; Mattsson, J.P.; Jensen, J.M.; Lehmann, A.; Dent, J.; Blackshaw, L.A. Inhibition of transient lower esophageal sphincter relaxation and gastroesophageal reflux by metabotropic glutamate receptor ligands. Gastroenterology 2005, 129, 995–1004. [Google Scholar] [CrossRef]
- Jensen, J.; Lehmann, A.; Uvebrant, A.; Carlsson, A.; Jerndal, G.; Nilsson, K.; Frisby, C.; Blackshaw, L.A.; Mattsson, J.P. Transient lower esophageal sphincter relaxations in dogs are inhibited by a metabotropic glutamate receptor 5 antagonist. Eur. J. Pharmacol. 2005, 519, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Keywood, C.; Wakefield, M.; Tack, J. A proof-of-concept study evaluating the effect of ADX10059, a metabotropic glutamate receptor-5 negative allosteric modulator, on acid exposure and symptoms in gastro-oesophageal reflux disease. Gut 2009, 58, 1192–1199. [Google Scholar] [CrossRef] [PubMed]
- Zerbib, F.; Bruley des Varannes, S.; Roman, S.; Tutuian, R.; Galmiche, J.P.; Mion, F.; Tack, J.; Malfertheiner, P.; Keywood, C. Randomised clinical trial: Effects of monotherapy with ADX10059, a mGluR5 inhibitor, on symptoms and reflux events in patients with gastro-oesophageal reflux disease. Aliment. Pharmacol. Ther. 2011, 33, 911–921. [Google Scholar] [CrossRef]
- Banerjee, B.; Medda, B.K.; Zheng, Y.; Miller, H.; Miranda, A.; Sengupta, J.N.; Shaker, R. Alterations in N-methyl-D-aspartate receptor subunits in primary sensory neurons following acid-induced esophagitis in cats. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G66–G77. [Google Scholar] [CrossRef]
- Raybould, H.E.; Jacobsen, L.J.; Novin, D.; Tachè, Y. TRh stimulation and L-glutamic acid inhibition of proximal gastric motor activity in the rat dorsal vagal complex. Brain Res. 1989, 495, 319–328. [Google Scholar] [CrossRef]
- Zhang, X.; Fogel, R. Involvement of glutamate in gastrointestinal vago-vagal reflexes initiated by gastrointestinal distension in the rat. Auton. Neurosci. 2003, 103, 19–37. [Google Scholar] [CrossRef]
- Sengupta, J.N.; Petersen, J.; Peles, S.; Shaker, R. Response properties of antral mechanosensitive afferent fibers and effects of ionotropic glutamate receptor antagonists. Neuroscience 2004, 125, 711–723. [Google Scholar] [CrossRef]
- Partosoedarso, E.R.; Blackshaw, L.A. Roles of central glutamate, acetylcholine and CGRP receptors in gastrointestinal afferent inputs to vagal preganglionic neurons. Auton. Neurosci. Basic Clin. 2000, 83, 37–48. [Google Scholar] [CrossRef]
- Czaja, K.; Ritter, R.C.; Burns, G.A. Vagal afferent neurons projecting to the stomach and small intestine exhibit multiple N-methyl-d-aspartate receptor subunit phenotypes. Brain Res. 2006, 1119, 86–93. [Google Scholar] [CrossRef]
- Watanabe, K.; Kanno, T.; Oshima, T.; Miwa, H.; Tashiro, C.; Nishizaki, T. Vagotomy upregulates expression of the N-methyl-d-aspartate receptor NR2D subunit in the stomach. J. Gastroenterol. 2008, 43, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Janković, S.M.; Milovanović, D.; Matović, M.; Irić-Cupić, V. The effects of excitatory amino acids on isolated gut segments of the rat. Pharmacol. Res. 1999, 39, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Tsai, L.H.; Lee, Y.J.; Wu, J.Y. Role of N-methyl-d-aspartate receptors in gastric mucosal blood flow induced by histamine. J. Neurosci. Res. 2004, 77, 730–738. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, E.; Hasumura, M.; San Gabriel, A.; Uneyama, H.; Torii, K. New frontiers in gut nutrient sensor research: Luminal glutamate-sensing cells in rat gastric mucosa. J. Pharmacol. Sci. 2010, 112, 13–18. [Google Scholar] [CrossRef] [PubMed]
- San Gabriel, A.M.; Maekawa, T.; Uneyama, H.; Yoshie, S.; Torii, K. mGluR1 in the fundic glands of rat stomach. FEBS Lett. 2007, 581, 1119–1123. [Google Scholar] [CrossRef]
- Gill, S.S.; Pulido, O.M. Glutamate receptors in peripheral tissues: Current knowledge, future research, and implications for toxicology. Toxicol. Pathol. 2001, 29, 208–223. [Google Scholar] [CrossRef]
- Milusheva, E.A.; Kuneva, V.I.; Itzev, D.E.; Kortezova, N.I.; Sperlagh, B.; Mizhorkova, Z.N. Glutamate stimulation of acetylcholine release from myenteric plexus is mediated by endogenous nitric oxide. Brain Res. Bull. 2005, 66, 229–234. [Google Scholar] [CrossRef]
- Filpa, V.; Carpanese, E.; Marchet, S.; Prandoni, V.; Moro, E.; Lecchini, S.; Frigo, G.; Giaroni, C.; Crema, F. Interaction between NMDA glutamatergic and nitrergic enteric pathways during in vitro ischemia and reperfusion. Eur. J. Pharmacol. 2015, 750, 123–131. [Google Scholar] [CrossRef]
- Tong, Q.; Kirchgessner, A.L. Localization and function of metabotropic glutamate receptor 8 in the enteric nervous system. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 285, G992–G1003. [Google Scholar] [CrossRef]
- Julio-Pieper, M.; O’Connor, R.M.; Dinan, T.G.; Cryan, J.F. Regulation of the brain-gut axis by group III metabotropic glutamate receptors. Eur. J. Pharmacol. 2013, 698, 19–30. [Google Scholar] [CrossRef]
- Julio-Pieper, M.; Hyland, N.P.; Bravo, J.A.; Dinan, T.G.; Cryan, J.F. A novel role for the metabotropic glutamate receptor-7: Modulation of faecal water content and colonic electrolyte transport in the mouse. Br. J. Pharmacol. 2010, 160, 367–375. [Google Scholar] [CrossRef]
- McRoberts, J.A.; Coutinho, S.V.; Marvizón, J.C.; Grady, E.F.; Tognetto, M.; Sengupta, J.N.; Ennes, H.S.; Chaban, V.V.; Amadesi, S.; Creminon, C.; et al. Role of peripheral N-methyl-d-aspartate (NMDA) receptors in visceral nociception in rats. Gastroenterology 2001, 120, 1737–1748. [Google Scholar] [CrossRef]
- Gaudreau, G.A.; Plourde, V. Involvement of N-methyl-d-aspartate (NMDA) receptors in a rat model of visceral hypersensitivity. Behav. Brain Res. 2004, 150, 185–189. [Google Scholar] [CrossRef]
- Brassai, A.; Suvanjeiev, R.-G.; Bán, E.-G.; Lakatos, M. Role of synaptic and nonsynaptic glutamate receptors in ischaemia induced neurotoxicity. Brain Res. Bull. 2015, 112, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Laskaratos, F.-M.; Goodkin, O.; Thoua, N.M.; Murray, C.D. Irritable bowel syndrome. Medicine 2015, 43, 266–270. [Google Scholar] [CrossRef]
- Kennedy, P.J.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Irritable bowel syndrome: A microbiome-gut-brain axis disorder? World J. Gastroenterol. 2014, 20, 14105–14125. [Google Scholar] [CrossRef] [PubMed]
- De Palma, G.; Collins, S.M.; Bercik, P. The microbiota-gut-brain axis in functional gastrointestinal disorders. Gut Microbes 2014, 5, 419–429. [Google Scholar] [CrossRef]
- Hyland, N.P.; Quigley, E.M.M.; Brint, E. Microbiota-host interactions in irritable bowel syndrome: Epithelial barrier, immune regulation and brain-gut interactions. World J. Gastroenterol. 2014, 20, 8859–8866. [Google Scholar]
- Marshall, J.K.; Thabane, M.; Garg, A.X.; Clark, W.F.; Salvadori, M.; Collins, S.M. Incidence and Epidemiology of Irritable Bowel Syndrome After a Large Waterborne Outbreak of Bacterial Dysentery. Gastroenterology 2006, 131, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Guilarte, M.; Santos, J.; De Torres, I.; Alonso, C.; Vicario, M.; Ramos, L.; Martinez, C.; Casellas, F.; Saperas, E.; Malagelada, J.R. Diarrhoea-predominant IBS patients show mast cell activation and hyperplasia in the jejunum. Gut 2007, 56, 203–209. [Google Scholar] [CrossRef]
- Dinan, T.G.; Quigley, E.M.M.; Ahmed, S.M.M.; Scully, P.; O’Brien, S.; O’Mahony, L.; O’Mahony, S.; Shanahan, F.; Keeling, P.W.N. Hypothalamic-pituitary-gut axis dysregulation in irritable bowel syndrome: Plasma cytokines as a potential biomarker? Gastroenterology 2006, 130, 304–311. [Google Scholar] [CrossRef]
- De Schepper, H.U.; De Man, J.G.; Moreels, T.G.; Pelckmans, P.A.; De Winter, B.Y. Review article: Gastrointestinal sensory and motor disturbances in inflammatory bowel disease - Clinical relevance and pathophysiological mechanisms. Aliment. Pharmacol. Ther. 2008, 27, 621–637. [Google Scholar] [CrossRef]
- Belmonte, L.; Beutheu Youmba, S.; Bertiaux-Vandaële, N.; Antonietti, M.; Lecleire, S.; Zalar, A.; Gourcerol, G.; Leroi, A.-M.; Déchelotte, P.; Coëffier, M.; et al. Role of toll like receptors in irritable bowel syndrome: Differential mucosal immune activation according to the disease subtype. PLoS ONE 2012, 7, e42777. [Google Scholar] [CrossRef]
- Koçak, E.; Akbal, E.; Köklü, S.; Ergül, B.; Can, M. The colonic tissue levels of TLR2, TLR4 and nitric oxide in patients with irritable bowel syndrome. Intern. Med. 2016, 55, 1043–1048. [Google Scholar]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef]
- Jeffery, I.B.; O’Toole, P.W.; Öhman, L.; Claesson, M.J.; Deane, J.; Quigley, E.M.M.; Simrén, M. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut 2012, 61, 997–1006. [Google Scholar] [CrossRef]
- Krogius-Kurikka, L.; Lyra, A.; Malinen, E.; Aarnikunnas, J.; Tuimala, J.; Paulin, L.; Mäkivuokko, H.; Kajander, K.; Palva, A. Microbial community analysis reveals high level phylogenetic alterations in the overall gastrointestinal microbiota of diarrhoea-predominant irritable bowel syndrome sufferers. BMC Gastroenterol. 2009, 9, 95. [Google Scholar] [CrossRef]
- Perez-Burgos, A.; Wang, L.; Mcvey Neufeld, K.-A.; Mao, Y.-K.; Ahmadzai, M.; Janssen, L.J.; Stanisz, A.M.; Bienenstock, J.; Kunze, W.A. The TRPV1 channel in rodents is a major target for antinociceptive effect of the probiotic Lactobacillus reuteri DSM 17938. J. Physiol. 2015, 593, 3943–3957. [Google Scholar] [CrossRef]
- Saulnier, D.M.; Riehle, K.; Mistretta, T.-A.; Diaz, M.-A.; Mandal, D.; Raza, S.; Weidler, E.M.; Qin, X.; Coarfa, C.; Milosavljevic, A.; et al. Gastrointestinal microbiome signatures of pediatric patients with irritable bowel syndrome. Gastroenterology 2011, 141, 1782–1791. [Google Scholar] [CrossRef] [PubMed]
- Crouzet, L.; Gaultier, E.; Del’Homme, C.; Cartier, C.; Delmas, E.; Dapoigny, M.; Fioramonti, J.; Bernalier-Donadille, A. The hypersensitivity to colonic distension of IBS patients can be transferred to rats through their fecal microbiota. Neurogastroenterol. Motil. 2013, 25, e272–e282. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.M.; Felice, V.D.; Nally, K.; Savignac, H.M.; Claesson, M.J.; Scully, P.; Woznicki, J.; Hyland, N.P.; Shanahan, F.; Quigley, E.M.; et al. Disturbance of the gut microbiota in early-life selectively affects visceral pain in adulthood without impacting cognitive or anxiety-related behaviors in male rats. Neuroscience 2014, 277, 885–901. [Google Scholar] [CrossRef] [PubMed]
- McKernan, D.P.; Fitzgerald, P.; Dinan, T.G.; Cryan, J.F. The probiotic Bifidobacterium infantis 35624 displays visceral antinociceptive effects in the rat. Neurogastroenterol. Motil. 2010, 22, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Verdú, E.F.; Bercik, P.; Verma-Gandhu, M.; Huang, X.-X.; Blennerhassett, P.; Jackson, W.; Mao, Y.; Wang, L.; Rochat, F.; Collins, S.M. Specific probiotic therapy attenuates antibiotic induced visceral hypersensitivity in mice. Gut 2006, 55, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, M.; Melgar, S. Microbial neuro-immune interactions and the pathophysiology of IBD. In New Insights into Inflammatory Bowel Disease; Huber, S., Ed.; Intechopen: London, UK, 2016; pp. 181–215. [Google Scholar]
- Blackshaw, L.A.; Gebhart, G.F. The pharmacology of gastrointestinal nociceptive pathways. Curr. Opin. Pharmacol. 2002, 2, 642–649. [Google Scholar] [CrossRef]
- Kolhekar, R.; Gebhart, G.F. Modulation of spinal visceral nociceptive neurotransmission by NMDA receptor activation in the rat. J. Neurophysiol. 1996, 75, 2344–2353. [Google Scholar] [CrossRef] [PubMed]
- Kolhekar, R.; Gebhart, G.F. NMDA and quisqualate modulation of visceral nociception in the rat. Brain Res. 1994, 651, 215–226. [Google Scholar] [CrossRef]
- Seybold, V.S. The role of peptides in central sensitization. Hand. Exp. Pharmacol. 2009, 194, 451–491. [Google Scholar]
- Raddant, A.C.; Russo, A.F. Calcitonin gene-related peptide in migraine: Intersection of peripheral inflammation and central modulation. Expert Rev. Mol. Med. 2011, 13, e36. [Google Scholar] [CrossRef]
- Onaga, T. Tachykinin: Recent developments and novel roles in health and disease. Biomol. Concepts 2014, 5, 225–243. [Google Scholar] [CrossRef]
- Li, J.; McRoberts, J.A.; Ennes, H.S.; Trevisani, M.; Nicoletti, P.; Mittal, Y.; Mayer, E.A. Experimental colitis modulates the functional properties of NMDA receptors in dorsal root ganglia neurons. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 29, G219–G228. [Google Scholar] [CrossRef]
- Zhou, L.; Huang, J.; Gao, J.; Zhang, G.; Jiang, J. NMDA and AMPA receptors in the anterior cingulate cortex mediates visceral pain in visceral hypersensitivity rats. Cell. Immunol. 2014, 287, 86–90. [Google Scholar] [CrossRef]
- Zhang, X.-M.; Luo, J.-H. GluN2A versus GluN2B: Twins, but quite different. Neurosci. Bull. 2013, 29, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Sheng, L.; Guan, Y.; Qian, W.; Hou, X. Synaptic plasticity: The new explanation of visceral hypersensitivity in rats with Trichinella spiralis infection? Dig. Dis. Sci. 2009, 54, 937–946. [Google Scholar] [CrossRef]
- Chen, A.; Chen, Y.; Tang, Y.; Bao, C.; Cui, Z.; Xiao, M.; Lin, C. Hippocampal AMPARs involve the central sensitization of rats with irritable bowel syndrome. Brain Behav. 2017, 7, e00650. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Price, D.D.; Caudle, R.M.; Verne, G.N. Visceral and somatic hypersensitivity in TNBS-induced colitis in rats. Dig. Dis. Sci. 2007, 53, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Caudle, R.M.; Price, D.D.; Del Valle-Pinero, A.Y.; Verne, G.N. Selective up-regulation of NMDA-NR1 receptor expression in myenteric plexus after TNBS induced colitis in rats. Mol. Pain 2006, 2, 3. [Google Scholar] [CrossRef]
- Zhou, Q.; Price, D.D.; Caudle, R.M.; Verne, G.N. Spinal NMDA NR1 subunit expression following transient TNBS colitis. Brain Res. 2009, 1279, 109–120. [Google Scholar] [CrossRef]
- Lindström, E.; Brusberg, M.; Hughes, P.A.; Martin, C.M.; Brierley, S.M.; Phillis, B.D.; Martinsson, R.; Abrahamsson, C.; Larsson, H.; Martinez, V.; et al. Involvement of metabotropic glutamate 5 receptor in visceral pain. Pain 2008, 137, 295–305. [Google Scholar] [CrossRef]
- Bianchi, R.; Rezzani, R.; Borsani, E.; Rodella, L. mGlu5 receptor antagonist decreases Fos expression in spinal neurons after noxious visceral stimulation. Brain Res. 2003, 960, 263–266. [Google Scholar] [CrossRef]
- Nasser, Y.; Keenan, C.M.; Ma, A.C.; McCafferty, D.M.; Sharkey, K.A. Expression of a functional metabotropic glutamate receptor 5 on enteric glia is altered in states of inflammation. Glia 2007, 55, 859–872. [Google Scholar] [CrossRef]
- Johnson, M.P.; Muhlhauser, M.A.; Nisenbaum, E.S.; Simmons, R.M.A.; Forster, B.M.; Knopp, K.L.; Yang, L.; Morrow, D.; Li, D.L.; Kennedy, J.D.; et al. Broad spectrum efficacy with LY2969822, an oral prodrug of metabotropic glutamate 2/3 receptor agonist LY2934747, in rodent pain models. Br. J. Pharmacol. 2017, 174, 822–835. [Google Scholar] [CrossRef]
- Moloney, R.D.; Golubeva, A.V.; O’Connor, R.M.; Kalinichev, M.; Dinan, T.G.; Cryan, J.F. Negative allosteric modulation of the mGlu7 receptor reduces visceral hypersensitivity in a stress-sensitive rat strain. Neurobiol. Stress 2015, 2, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Gosselin, R.D.; O’Connor, R.M.; Tramullas, M.; Julio-Pieper, M.; Dinan, T.G.; Cryan, J.F. Riluzole normalizes early-life stress-induced visceral hypersensitivity in rats: role of spinal glutamate reuptake mechanisms. Gastroenterology 2010, 138, 2418–2425. [Google Scholar] [CrossRef] [PubMed]
- Bradesi, S. Role of spinal cord glia in the central processing of peripheral pain perception. Neurogastroenterol. Motil. 2010, 22, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.P.; Shukla, S.K.; Pandey, B.L. A preliminary evaluation of comparative effectiveness of riluzole in therapeutic regimen for irritable bowel syndrome. Asian Pac. J. Trop. Biomed. 2014, 4, S335–S340. [Google Scholar] [CrossRef]
- Lin, Y.; Tian, G.; Roman, K.; Handy, C.; Travers, J.B.; Lin, C.L.; Stephens, R.L., Jr. Increased glial glutamate transporter EAAT2 expression reduces visceral nociceptive response in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G129–G134. [Google Scholar] [CrossRef]
- Lin, Y.; Roman, K.; Foust, K.D.; Kaspar, B.K.; Bailey, M.T.; Stephens, R.L., Jr. Glutamate transporter GLT-1 uperegulation attenuates visceral nociception and hyperalgesia via spinal mechanisms not related to anti-inflammatory or probiotic effect. Pain Res. Treat. 2011, 2011, 507029. [Google Scholar]
- Lee, C.; Doo, E.; Choi, J.M.; Jang, S.; Ryu, H.-S.; Lee, J.Y.; Oh, J.H.; Park, J.H.; Kim, Y.S. The increased level of depression and anxiety in irritable bowel syndrome patients compared with healthy controls: Systematic review and meta-analysis. J. Neurogastroenterol. Motil. 2017, 23, 349–362. [Google Scholar] [CrossRef]
- Smoller, J.W. The genetic of stress-related disorders: PTSD, depression, and anxiety disorders. Neuropsychopharmacology 2016, 41, 297–319. [Google Scholar] [CrossRef]
- Holzer, P.; Farzi, A.; Hassan, A.M.; Zenz, G.; Jacan, A.; Reichmann, F. Visceral inflammation and immune activation stress the brain. Front. Immunol. 2017, 8, 1613. [Google Scholar] [CrossRef]
- Foster, J.A.; Rinaman, L.; Cryan, J.F. Stress & the gut-brain axis: Regulation by the microbiome. Neurobiol. Stress 2017, 7, 124–136. [Google Scholar]
- Sudo, N.; Chida, Y.; Aiba, Y.; Sonoda, J.; Oyama, N.; Yu, X.-N.; Kubo, C.; Koga, Y. Postnatal microbial colonization programs the hypothalamic-pituitary-adrenal system for stress response in mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Gareau, M.G.; Jury, J.; MacQueen, G. Probiotic treatment of rat pups normalises corticosterone release and ameliorates colonic dysfunction induced by maternal separation. Gut 2007, 56, 1522–1528. [Google Scholar] [CrossRef]
- Scott, L.V.; Clarke, G.; Dinan, T.G. The Brain-Gut Axis: A Target for Treating Stress-Related Disorders. Mod Trends Pharmacopsychiatry 2013, 28, 90–99. [Google Scholar]
- Farzi, A.; Fröhlich, E.E.; Holzer, P. Gut Microbiota and the Neuroendocrine System. Neurotherapeutics 2018, 15, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Gareau, M.G.; Silva, M.A.; Perdue, M.H. Pathophysiological mechanisms of stress-induced intestinal damage. Curr. Mol. Med. 2008, 8, 274–281. [Google Scholar] [CrossRef]
- Lafuse, W.P.; Gearinger, R.; Fisher, S.; Nealer, C.; Mackos, A.R.; Bailey, M.T. Exposure to a social stressor induces translocation of commensal lactobacilli to the spleen and priming of the innate immune system. J. Immunol. 2017, 198, 2383–2393. [Google Scholar] [CrossRef] [PubMed]
- Campos-Rodríguez, R.; Godínez-Victoria, M.; Abarca-Rojano, E.; Pacheco-Yépez, J.; Reyna-Garfias, H.; Barbosa-Cabrera, R.E.; Drago-Serrano, M.E. Stress modulates intestinal secretory immunoglobulin A. Front. Integr. Neurosci. 2013, 7, 86. [Google Scholar] [CrossRef]
- Lyte, M.; Vulchanova, L.; Brown, D.R. Stress at the intestinal surface: Catecholamines and mucosa-bacteria interactions. Cell Tissue Res. 2011, 343, 23–32. [Google Scholar] [CrossRef]
- Gárate, I.; Garcia-Bueno, B.; Madrigal, J.L.M.; Caso, J.R.; Alou, L.; Gomez-Lus, M.L.; Micó, J.A.; Leza, J.C. Stress-induced neuroinflammation: Role of the toll-like receptor-4 pathway. Biol. Psychiatry 2013, 73, 32–43. [Google Scholar] [CrossRef]
- Mumtaz, F.; Khan, M.I.; Zubair, M.; Dehpour, A.R. Neurobiology and consequences of social isolation stress in animal model—A comprehensive review. Biomed. Pharmacother. 2018, 105, 1205–1222. [Google Scholar] [CrossRef]
- Joffe, M.E.; Santiago, C.I.; Engers, J.L.; Lindsley, C.W.; Conn, P.J. Metabotropic glutamate receptor subtype 3 gates acute stress-induced dysregulation of amygdalo-cortical function. Mol. Psychiatry 2017. [Google Scholar] [CrossRef] [PubMed]
- Joffe, M.E.; Santiago, C.I.; Stansley, B.J.; Maksymetz, J.; Gogliotti, R.G.; Engers, J.L.; Nicoletti, F.; Lindsley, C.W.; Conn, P.J. Mechanisms underlying prelimbic prefrontal cortex mGlu3/mGlu5-dependent plasticity and reversal learning deficits following acute stress. Neuropharmacology 2019, 144, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-H.; Hsiao, Y.-H.; Chen, Y.-W.; Yu, Y.-J.; Gean, P.-W. Social isolation-induced increase in NMDA receptors in the hippocampus exacerbates emotional dysregulation in mice. Hippocampus 2015, 25, 474–485. [Google Scholar] [CrossRef]
- Ghasemi, M.; Phillips, C.; Trillo, L.; De Miguel, Z.; Das, D.; Salehi, A. The role of NMDA receptors in the pathophysiology and treatment of mood disorders. Neurosci. Biobehav. Rev. 2014, 47, 336–358. [Google Scholar] [CrossRef]
- Cao, B.; Zhu, J.; Zuckerman, H.; Rosenblat, J.D.; Brietzke, E.; Pan, Z.; Subramanieapillai, M.; Park, C.; Lee, Y.; McIntyre, R.S. Pharmacological interventions targeting anhedonia in patients with major depressive disorder: A systemic review. Prog. Neuropsychopharmaocl. Biol. Psychiatry 2019, 92, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Lepack, A.E.; Bang, E.; Lee, B.; Dwyer, J.M.; Duman, R.S. Fast-acting antidepressants rapidly stimulate ERK signaling and BDNF release in primary neuronal cultures. Neuropharmacology 2016, 111, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Maqsood, R.; Stone, T.W. The Gut-Brain Axis, BDNF, NMDA and CNS Disorders. Neurochem. Res. 2016, 41, 2819–2835. [Google Scholar] [CrossRef]
- Klug, M.; Hill, R.A.; Choy, K.H.C.; Kyrios, M.; Hannan, A.J.; van den Buuse, M. Long-term behavioral and NMDA receptor effects of young-adult corticosterone treatment in BDNF heterozygous mice. Neurobiol. Dis. 2012, 46, 722–731. [Google Scholar] [CrossRef]
- Suen, P.-C.; Wu, K.; Levine, E.S.; Mount, H.T.J.; Xu, J.-L.; Lin, S.-Y.; Black, I.B. Brain-derived neurotrophic factor rapidly enhances phosphorylation of the postsynaptic N-methyl-D-aspartate receptor subunit 1. Proc. Natl. Acad. Sci. USA 1997, 94, 8191–8195. [Google Scholar] [CrossRef]
- Jiang, C.; Lin, W.-J.; Salton, S.R. Role of a VGF/BDNF/TrkB Autoregulatory Feedback Loop in Rapid-Acting Antidepressant Efficacy. J. Mol. Neurosci. 2018. [Google Scholar] [CrossRef]
- Clarke, G.; Grenham, S.; Scully, P.; Fitzgerald, P.; Moloney, R.D.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol. Psychiatry 2013, 18, 666–673. [Google Scholar] [CrossRef]
- Neufeld, K.M.; Kang, N.; Bienenstock, J.; Foster, J.A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 2011, 23, 255–264. [Google Scholar] [CrossRef]
- Bistoletti, M.; Caputi, V.; Baranzini, N.; Marchesi, N.; Filpa, V.; Marsilio, I.; Cerantola, S.; Terova, G.; Baj, A.; Grimaldi, A. Antibiotic treatment-induced dysbiosis differently affects BDNF and TrkB expression in the brain and in the gut of juvenile mice. PLoS ONE 2019, 14, e0212856. [Google Scholar] [CrossRef] [PubMed]
- Heijtz, R.D.; Wang, S.; Anuar, F.; Qian, Y.; Björkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar] [CrossRef]
- Kelly, J.R.; Allen, A.P.; Temko, A.; Hutch, W.; Kennedy, P.J.; Farid, N.; Murphy, E.; Boylan, G.; Bienenstock, J.; Cryan, J.F.; et al. Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects. Brain Behav. Immun. 2017, 61, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Savignac, H.M.; Corona, G.; Mills, H.; Chen, L.; Spencer, J.P.E.; Tzortzis, G.; Burnet, P.W.J. Prebiotic feeding elevates central brain derived neurotrophic factor, N-methyl-d-aspartate receptor subunits and d-serine. Neurochem. Int. 2013, 63, 756–764. [Google Scholar] [CrossRef]
- Burokas, A.; Arboleya, S.; Moloney, R.D.; Peterson, V.L.; Murphy, K.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Targeting the Microbiota-Gut-Brain Axis: Prebiotics Have Anxiolytic and Antidepressant-like Effects and Reverse the Impact of Chronic Stress in Mice. Biol. Psychiatry 2017, 82, 472–487. [Google Scholar] [CrossRef]
- Gronier, B.; Savignac, H.M.; Di Miceli, M.; Idriss, S.M.; Tzortzis, G.; Anthony, D.; Burnet, P.W.J. Increased cortical neuronal responses to NMDA and improved attentional set-shifting performance in rats following prebiotic (B-GOS®) ingestion. Eur. Neuropsychopharmacol. 2018, 28, 211–224. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.; Zeng, B.; Zhou, C.; Liu, M.; Fang, Z.; Xu, X.; Zeng, L.; Chen, J.; Fan, S.; Du, X.; et al. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Mol. Psychiatry 2016, 21, 786–796. [Google Scholar] [CrossRef]
- Jiang, H.; Ling, Z.; Zhang, Y.; Mao, H.; Ma, Z.; Yin, Y.; Wang, W.; Tang, W.; Tan, Z.; Shi, J.; et al. Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav. Immun. 2015, 48, 186–194. [Google Scholar] [CrossRef]
- Naseribafrouei, A.; Hestad, K.; Avershina, E.; Sekelja, M.; Linløkken, A.; Wilson, R.; Rudi, K. Correlation between the human fecal microbiota and depression. Neurogastroenterol. Motil. 2014, 26, 1155–1162. [Google Scholar] [CrossRef]
- Kelly, J.R.; Borre, Y.; O’ Brien, C.; Patterson, E.; El Aidy, S.; Deane, J.; Kennedy, P.J.; Beers, S.; Scott, K.; Moloney, G.; et al. Transferring the blues: Depression-associated gut microbiota induces neurobehavioural changes in the rat. J. Psychiatr. Res. 2016, 82, 109–118. [Google Scholar] [CrossRef]
- Keszthelyi, D.; Troost, F.J.; Jonkers, D.M.; Kruimel, J.W.; Leue, C.; Masclee, A.A.M. Decreased levels of kynurenic acid in the intestinal mucosa of IBS patients: Relation to serotonin and psychological state. J. Psychosom. Res. 2013, 74, 501–504. [Google Scholar] [CrossRef]
- Clarke, G.; Quigley, E.M.M.; Cryan, J.F.; Dinan, T.G. Irritable bowel syndrome: Towards biomarker identification. Trends Mol. Med. 2009, 15, 478–489. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.C.; Shi, H.Y.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Wu, J.C.Y.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: A systematic review of population-based studies. Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- Ni, J.; Wu, G.D.; Albenberg, L.; Tomov, V.T. Gut microbiota and IBD: Causation or correlation? Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Lomax, A.E.; Fernández, E.; Sharkey, K.A. Plasticity of the enteric nervous system during intestinal inflammation. Neurogastroenterol. Motil. 2005, 17, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Filpa, V.; Bistoletti, M.; Caon, I.; Moro, E.; Grimaldi, A.; Moretto, P.; Baj, A.; Giron, M.C.; Karousou, E.; Viola, M.; et al. Changes in hyaluronan deposition in the rat myenteric plexus after experimentally-induced colitis. Sci. Rep. 2017, 7, 17644. [Google Scholar] [CrossRef]
- Vasina, V.; Barbara, G.; Talamonti, L.; Stanghellini, V.; Corinaldesi, R.; Tonini, M.; De Ponti, F.; De Giorgio, R. Enteric neuroplasticity evoked by inflammation. Auton. Neurosci. 2006, 126–127, 264–272. [Google Scholar] [CrossRef]
- Brierley, S.M.; Linden, D.R. Neuroplasticity and dysfunction after gastrointestinal inflammation. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 611–627. [Google Scholar] [CrossRef]
- Marchesi, J.R.; Adams, D.H.; Fava, F.; Hermes, G.D.A.; Hirschfield, G.M.; Hold, G.; Quraishi, M.N.; Kinross, J.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef]
- Hansen, R.; Russell, R.K.; Reiff, C.; Louis, P.; McIntosh, F.; Berry, S.H.; Mukhopadhya, I.; Bisset, W.M.; Barclay, A.R.; Bishop, J.; et al. Microbiota of de-novo pediatric IBD: Increased faecalibacterium prausnitzii and reduced bacterial diversity in Crohn’s but not in ulcerative colitis. Am. J. Gastroenterol. 2012, 107, 1913–1922. [Google Scholar] [CrossRef]
- Buttó, L.F.; Schaubeck, M.; Haller, D. Mechanisms of microbe-host interaction in Crohn’s disease: Dysbiosis vs. Pathobiont Selection. Front. Immunol. 2015, 6, 555. [Google Scholar] [CrossRef]
- Hoshi, N.; Schenten, D.; Nish, S.A.; Walther, Z.; Gagliani, N.; Flavell, R.A.; Reizis, B.; Shen, Z.; Fox, J.G.; Iwasaki, A.; et al. MyD88 signaling in colonic mononuclear phagocytes drives colitis in IL-10-deficient mice. Nat. Commun. 2012, 3, 1120. [Google Scholar] [CrossRef] [PubMed]
- De Jager, P.L.; Franchimont, D.; Waliszewska, A.; Bitton, A.; Cohen, A.; Langelier, D.; Belaiche, J.; Vermeire, S.; Farwell, L.; Goris, A.; et al. The role of the Toll receptor pathway in susceptibility to inflammatory bowel diseases. Genes Immun. 2007, 8, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Pierik, M.; Joossens, S.; Van Steen, K.; Van Schuerbeek, N.; Vlietinck, R.; Rutgeerts, P.; Vermeire, S. Toll-like receptor-1,-2, and -6 polymorphisms influence disease extension in inflammatory bowel diseases. Inflamm. Bowel Dis. 2006, 12, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Malvin, N.P.; Seno, H.; Stappenbeck, T.S. Colonic epithelial response to injury requires Myd88 signaling in myeloid cells. Mucosal Immunol. 2012, 5, 194–206. [Google Scholar] [CrossRef] [PubMed]
- Cario, E.; Gerken, G.; Podolsky, D.K. Toll-Like Receptor 2 Controls Mucosal Inflammation by Regulating Epithelial Barrier Function. Gastroenterology 2007, 132, 1359–1374. [Google Scholar] [CrossRef]
- Brun, P.; Giron, M.C.; Qesari, M.; Porzionato, A.; Caputi, V.; Zoppellaro, C.; Banzato, S.; Grillo, A.R.; Spagnol, L.; De Caro, R.; et al. Toll-like receptor 2 regulates intestinal inflammation by controlling integrity of the enteric nervous system. Gastroenterology 2013, 145, 1323–1333. [Google Scholar] [CrossRef]
- Caputi, V.; Marsilio, I.; Cerantola, S.; Roozfarakh, M.; Lante, I.; Galuppini, F.; Rugge, M.; Napoli, E.; Giulivi, C.; Orso, G.; et al. Toll-like receptor 4 modulates small intestine neuromuscular function through nitrergic and purinergic pathways. Front. Pharmacol. 2017, 8, 350. [Google Scholar] [CrossRef]
- Chang, Y.C.; Kim, H.-W.; Rapoport, S.I.; Rao, J.S. Chronic NMDA administration increases neuroinflammatory markers in rat frontal cortex: Cross-talk between excitotoxicity and neuroinflammation. Neurochem. Res. 2008, 33, 2318–2323. [Google Scholar] [CrossRef]
- Kalff, J.C.; Türler, A.; Schwarz, N.T.; Schraut, W.H.; Lee, K.K.; Tweardy, D.J.; Billiar, T.R.; Simmons, R.L.; Bauer, A.J. Intra-abdominal activation of a local inflammatory response within the human muscularis externa during laparotomy. Ann. Surg. 2003, 3, 301–315. [Google Scholar] [CrossRef] [PubMed]
- Kaszaki, J.; Palásthy, Z.; Erczes, D.; Rácz, A.; Torday, C.; Varga, G.; Vécsei, L.; Boros, M. Kynurenic acid inhibits intestinal hypermotility and xanthine oxidase activity during experimental colon obstruction in dogs. Neurogastroenterol. Motil. 2008, 20, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Varga, G.; Erces, D.; Fazekas, B.; Fülöp, M.; Kovács, T.; Kaszaki, J.; Fülöp, F.; Vécsei, L.; Boros, M. N-Methyl-d-aspartate receptor antagonism decreases motility and inflammatory activation in the early phase of acute experimental colitis in the rat. Neurogastroenterol. Motil. 2010, 22, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Forrest, C.M.; Youd, P.; Kennedy, A.; Gould, S.R.; Darlington, L.G.; Stone, T.W. Purine, kynurenine, neopterin and lipid peroxidation levels in inflammatory bowel disease. J. Biomed. Sci. 2002, 9, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Christmas, D.M.; Badawy, A.A.; Hince, D.; Davies, S.J.; Probert, C.; Creed, T.; Smithson, J.; Afzal, M.; Nutt, D.J.; Potokar, J.P. Increased serum free tryptophan in patients with diarrhea-predominant irritable bowel syndrome. Nutr. Res. 2010, 30, 678–688. [Google Scholar] [CrossRef] [PubMed]
- Jürgens, B.; Hainz, U.; Fuchs, D.; Felzmann, T.; Heitger, A. Interferon-γ-triggered indoleamine 2,3-dioxygenase competence in human monocyte-derived dendritic cells induces regulatory activity in allogeneic T cells. Blood 2009, 114, 3235–3243. [Google Scholar] [CrossRef]
- Yeung, A.W.S.; Terentis, A.C.; King, N.J.C.; Thomas, S.R. Role of indoleamine 2,3-dioxygenase in health and disease. Clin. Sci. 2015, 129, 601–672. [Google Scholar] [CrossRef]
- Brooks, A.K.; Lawson, M.A.; Smith, R.A.; Janda, T.M.; Kelley, K.W.; McCusker, R.H. Interactions between inflammatory mediators and corticosteroids regulate transcription of genes within the Kynurenine Pathway in the mouse hippocampus. J. Neuroinflam. 2016, 13, 98. [Google Scholar] [CrossRef]
- Gyires, K.; Tóth, É.V.; Zádori, S.Z. Gut inflammation: Current update on pathophysiology, molecular mechanism and pharmacological treatment modalities. Curr. Pharm. Des. 2014, 20, 1063–1081. [Google Scholar] [CrossRef]
- Érces, D.; Varga, G.; Fazekas, B.; Kovács, T.; Tőkés, T.; Tiszlavicz, L.; Fülöp, F.; Vécsei, L.; Boros, M.; Kaszaki, J. N-methyl-d-aspartate receptor antagonist therapy suppresses colon motility and inflammatory activation six days after the onset of experimental colitis in rats. Eur. J. Pharmacol. 2012, 691, 225–234. [Google Scholar]
- Viviani, B.; Boraso, M.; Marchetti, N.; Marinovich, M. Perspectives on neuroinflammation and excitotoxicity: A neurotoxic conspiracy? NeuroToxicology 2014, 43, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Narula, N.; Pinto-Sanchez, M.I.; Calo, N.C.; Ford, A.C.; Bercik, P.; Reinisch, W.; Moayyedi, P. Anxiety but Not Depression Predicts Poor Outcomes in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Emge, J.R.; Huynh, K.; Miller, E.N.; Kaur, M.; Reardon, C.; Barrett, K.E.; Gareau, M.G. Modulation of the microbiota-gut-brain axis by probiotics in a murine model of inflammatory bowel disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 310, G989–G998. [Google Scholar] [CrossRef]
- Bercik, P.; Park, A.J.; Sinclair, D.; Khoshdel, A.; Lu, J.; Huang, X.; Deng, Y.; Blennerhassett, P.A.; Fahnestock, M.; Moine, D.; et al. The anxiolytic effect of Bifidobacterium longum NCC3001 involves vagal pathways for gut-brain communication. Neurogastroenterol. Motil. 2011, 23, 1132–1139. [Google Scholar] [CrossRef]
- Bercik, P.; Verdu, E.F.; Foster, J.A.; MacRi, J.; Potter, M.; Huang, X.; Malinowski, P.; Jackson, W.; Blennerhassett, P.; Neufeld, K.A.; et al. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 2010, 139, 2102–2112. [Google Scholar] [CrossRef]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef]
- Waclawiková, B.; El Aidy, S. Role of microbiota and tryptophan metabolites in the remote effect of intestinal inflammation on brain and depression. Pharmaceuticals 2018, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Savitz, J.; Dantzer, R.; Meier, T.B.; Wurfel, B.E.; Victor, T.A.; McIntosh, S.A.; Ford, B.N.; Morris, H.M.; Bodurka, J.; Teague, T.K.; et al. Activation of the kynurenine pathway is associated with striatal volume in major depressive disorder. Psychoneuroendocrinology 2015, 62, 54–58. [Google Scholar] [CrossRef]
- Dantzer, R.; Walker, A.K. Is there a role for glutamate-mediated excitotoxicity in inflammation-induced depression? J. Neural Transm. 2014, 121, 925–932. [Google Scholar] [CrossRef]
- Kostandy, B.B. The role of glutamate in neuronal ischemic injury: The role of spark in fire. Neurol. Sci. 2012, 33, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Mony, L.; Kew, J.N.C.; Gunthorpe, M.J.; Paoletti, P. Allosteric modulators of NR2B-containing NMDA receptors: Molecular mechanisms and therapeutic potential. Br. J. Pharmacol. 2009, 157, 1301–1317. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Low, C.-M.; Moody, O.A.; Jenkins, A.; Traynelis, S.F. Ionotropic GABA and glutamate receptor mutations and human neurologic diseases. Mol. Pharmacol. 2015, 88, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A.; Knight, R.; Mazmanian, S.K.; Cryan, J.F.; Tillisch, K. Gut microbes and the brain: Paradigm shift in neuroscience. J. Neurosci. Off. J. Soc. Neurosci. 2014, 34, 15490–15496. [Google Scholar] [CrossRef] [PubMed]
- Burnet, P.W.J.; Cowen, P.J. Psychobiotics highlight the pathways to happiness. Biol. Psychiatry 2013, 74, 708–709. [Google Scholar] [CrossRef]
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the Manipulation of Bacteria–Gut–Brain Signals. Trends Neurosci. 2016, 39, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Bambury, A.; Sandhu, K.; Cryan, J.F.; Dinan, T.G. Finding the needle in the haystack: Systematic identification of psychobiotics. Br. J. Pharmacol. 2018, 175, 4430–4438. [Google Scholar] [CrossRef]
| BACTERIA | DESCRIPTION | GLU PRODUCTION | REFERENCES |
|---|---|---|---|
| Arthrobacter mysorens | Gram-positive soil bacterium | used for industrial production of l-glutamate | [58] |
| Brevibacterium spp. | Gram-positive soil bacteria | glutamic acid producer | [59,60] |
| Corynebacterium glutamicum | Gram-positive soil bacterium | used industrially for large-scale production of l-glutamic acid | [61] |
| Corynebacterium callunae | Gram-positive soil bacterium | used industrially for large-scale production of l-glutamic acid | [62] |
| Lactobacillus plantarum | Gram-positive bacterium commonly found in many fermented food products, as well as in saliva. The high levels of this organism in food make it an ideal candidate as a probiotic | glutamic acid producer | [53] |
| Lactococcus lactis | Gram-positive used for centuries for fermentation of food. Other than its important function in food, L. lactis has become the model LAB when it comes to genetic engineering | glutamic acid producer | [51] |
| Methylobacillus sp. | Group of Gram-negative methylotrophic aerobic bacteria, found in marine and fresh-water ecosystems | glutamic acid producer | [63] |
| Hugo Name | Aliases | REFERENCES | |
|---|---|---|---|
| Plasma membrane transporters | |||
| Excitatory amino acid transporter 1 (EAAT1; slc1 a3) | GLAST | [66,70] | |
| Excitatory amino acid transporter 2 (EAAT2; slc1 a2) | GLT-1 | [66,70] | |
| Excitatory amino acid transporter 3 (EAAT3; slc1 a1) | EAAC1 | [66,70] | |
| Excitatory amino acid transporter 4 (EAAT4; slc1 a6) | [66,70] | ||
| Excitatory amino acid transporter 5 (EAAT5; slc1 a7) | [66,70] | ||
| Vesicular transporters | |||
| Vesicular glutamate transporter 1 (VGLUT1; slc17 a7) | ______ | [68,69] | |
| Vesicular glutamate transporter 2 (VGLUT2; slc17 a6) | ______ | [68,69] | |
| Vesicular glutamate transporter 3 (VGLUT1; slc17 a8) | ______ | [68,69] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baj, A.; Moro, E.; Bistoletti, M.; Orlandi, V.; Crema, F.; Giaroni, C. Glutamatergic Signaling Along The Microbiota-Gut-Brain Axis. Int. J. Mol. Sci. 2019, 20, 1482. https://doi.org/10.3390/ijms20061482
Baj A, Moro E, Bistoletti M, Orlandi V, Crema F, Giaroni C. Glutamatergic Signaling Along The Microbiota-Gut-Brain Axis. International Journal of Molecular Sciences. 2019; 20(6):1482. https://doi.org/10.3390/ijms20061482
Chicago/Turabian StyleBaj, Andreina, Elisabetta Moro, Michela Bistoletti, Viviana Orlandi, Francesca Crema, and Cristina Giaroni. 2019. "Glutamatergic Signaling Along The Microbiota-Gut-Brain Axis" International Journal of Molecular Sciences 20, no. 6: 1482. https://doi.org/10.3390/ijms20061482
APA StyleBaj, A., Moro, E., Bistoletti, M., Orlandi, V., Crema, F., & Giaroni, C. (2019). Glutamatergic Signaling Along The Microbiota-Gut-Brain Axis. International Journal of Molecular Sciences, 20(6), 1482. https://doi.org/10.3390/ijms20061482







