Thymoquinone Selectively Kills Hypoxic Renal Cancer Cells by Suppressing HIF-1α-Mediated Glycolysis
Abstract
:1. Introduction
2. Results and Discussion
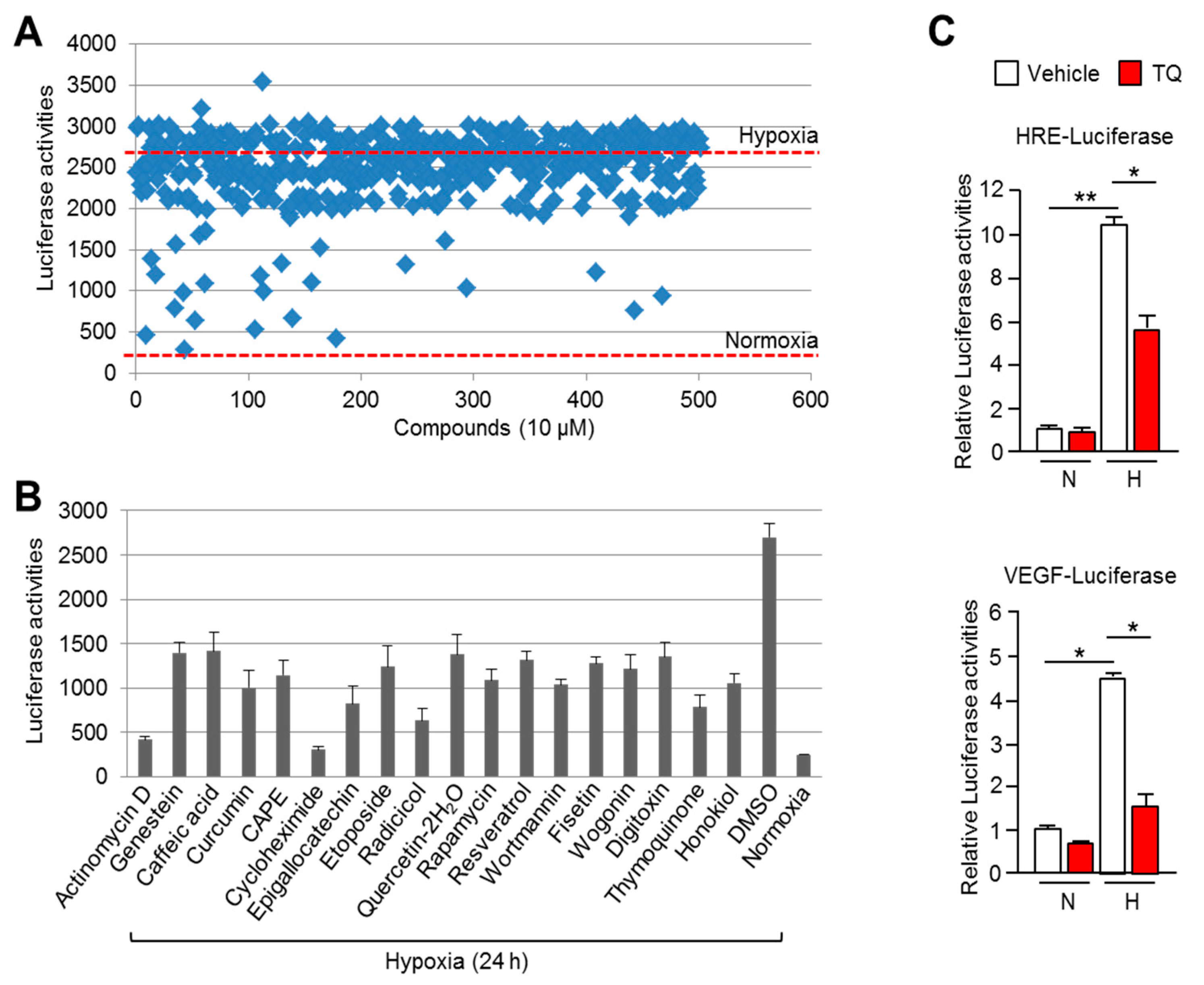
2.1. Thymoquinone (TQ) is a Potential HIF-1α Inhibitor
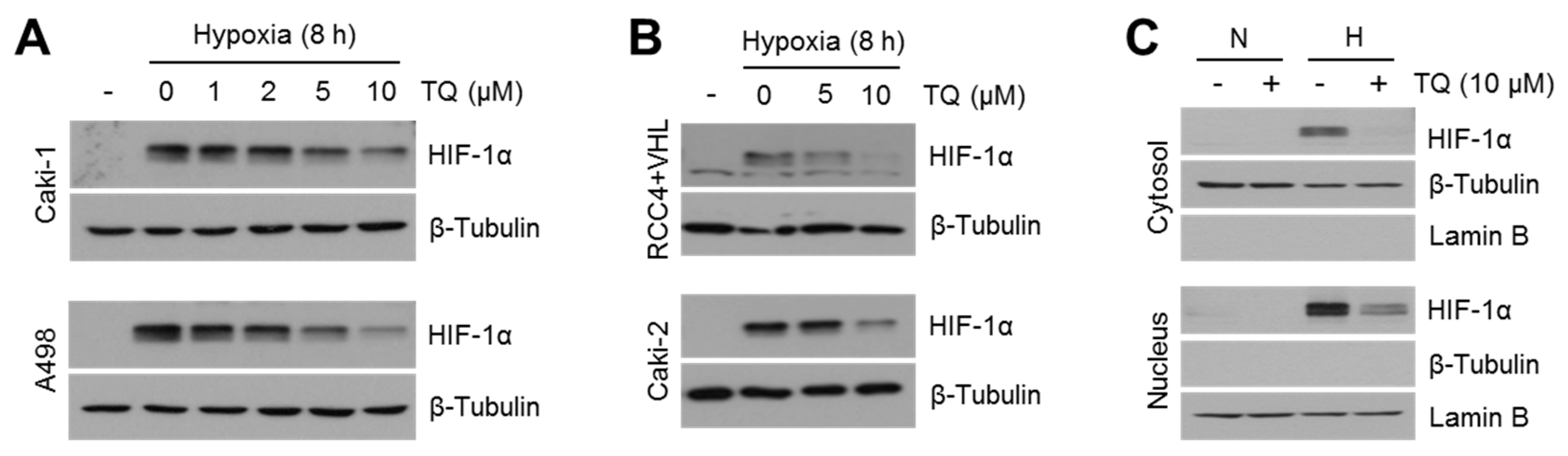
2.2. TQ Decreases HIF-1α in Hypoxic Renal Cancer Cells
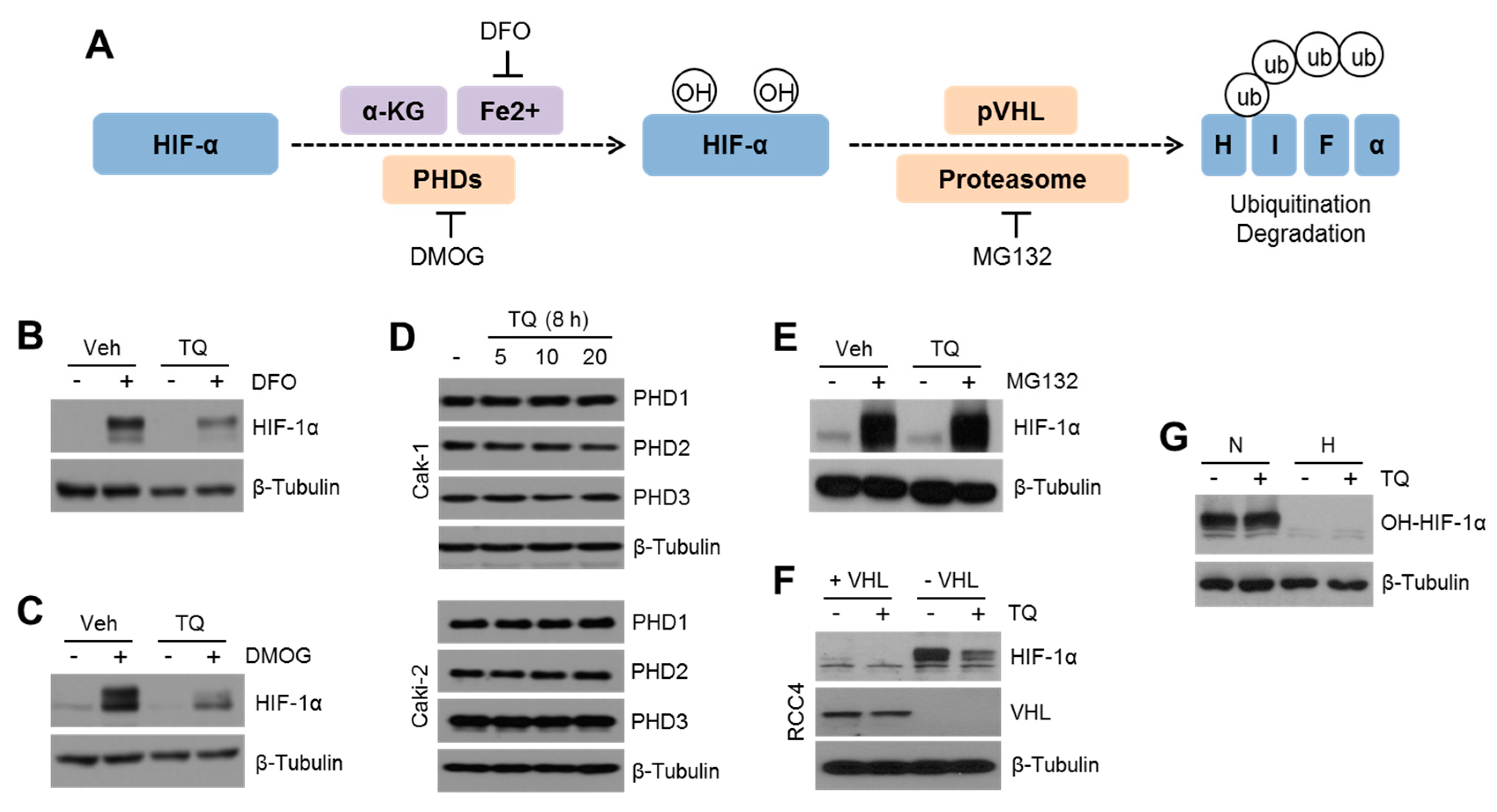
2.3. TQ Decreases HIF-1α Expression Independent of PHD-Mediated Hydroxylation but Dependent on Proteasomal Degradation Pathway
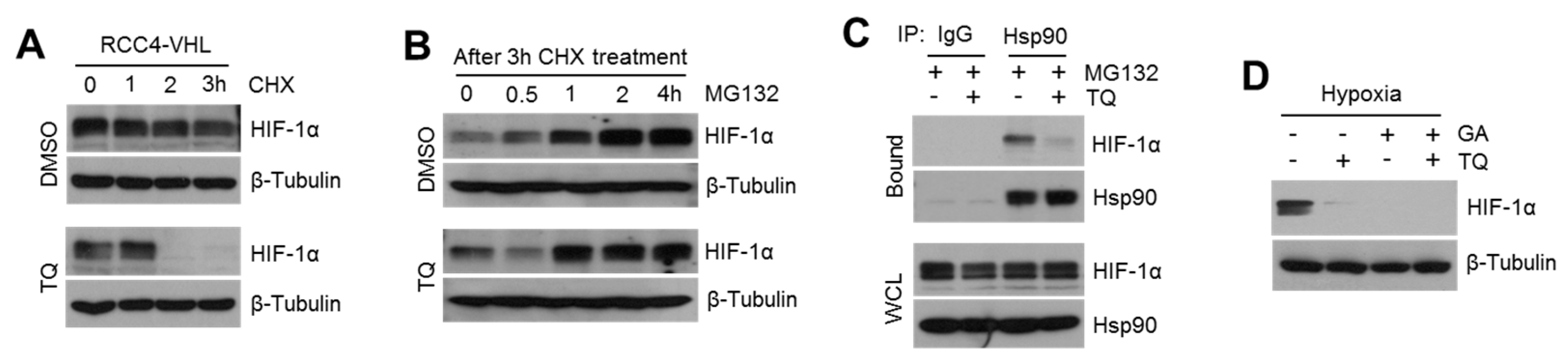
2.4. TQ Suppresses HSP90-Mediated HIF-1α Stabilization
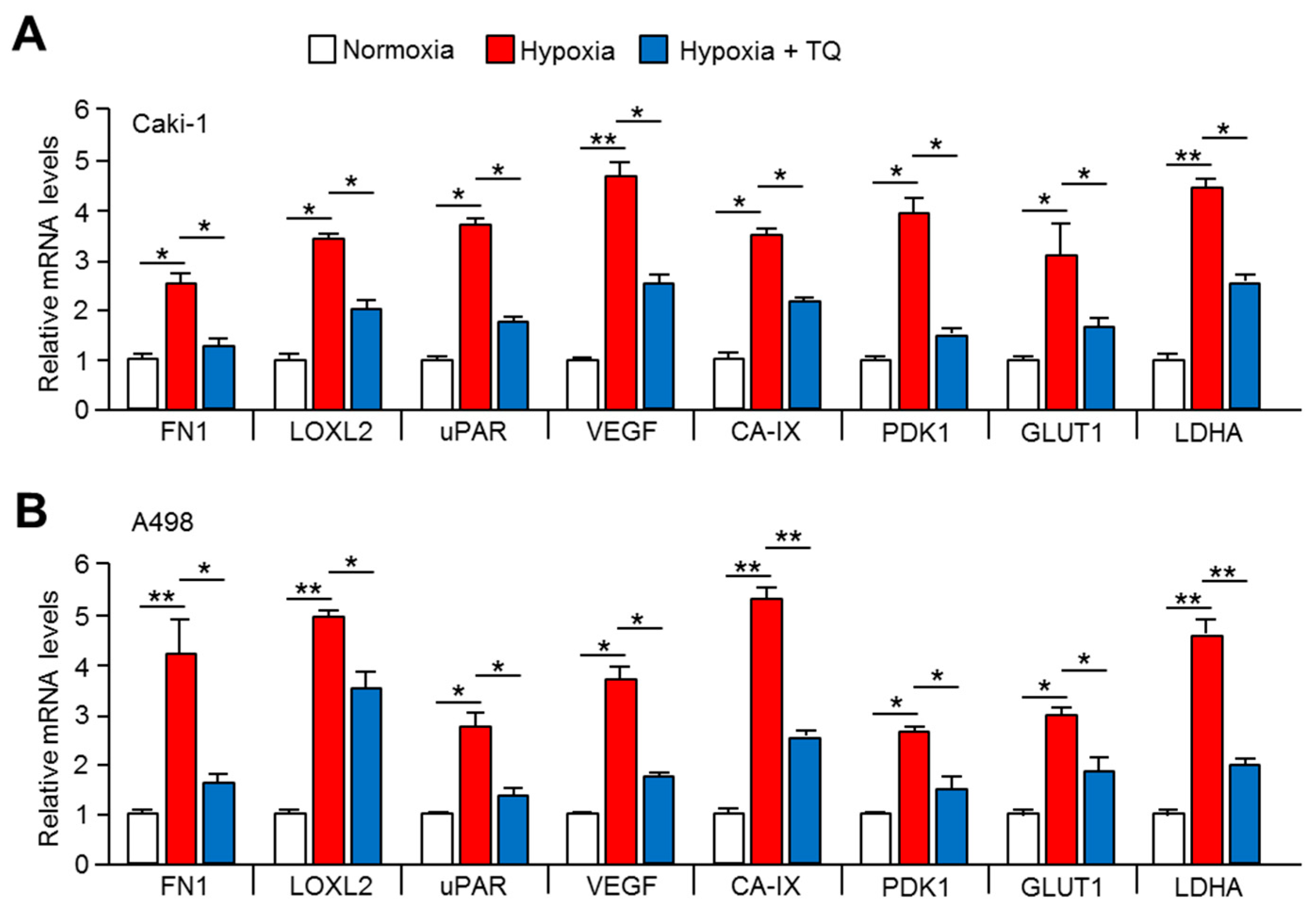
2.5. Expression of Tumor Promoting HIF-1α Target Genes is Downregulated by TQ Treatment
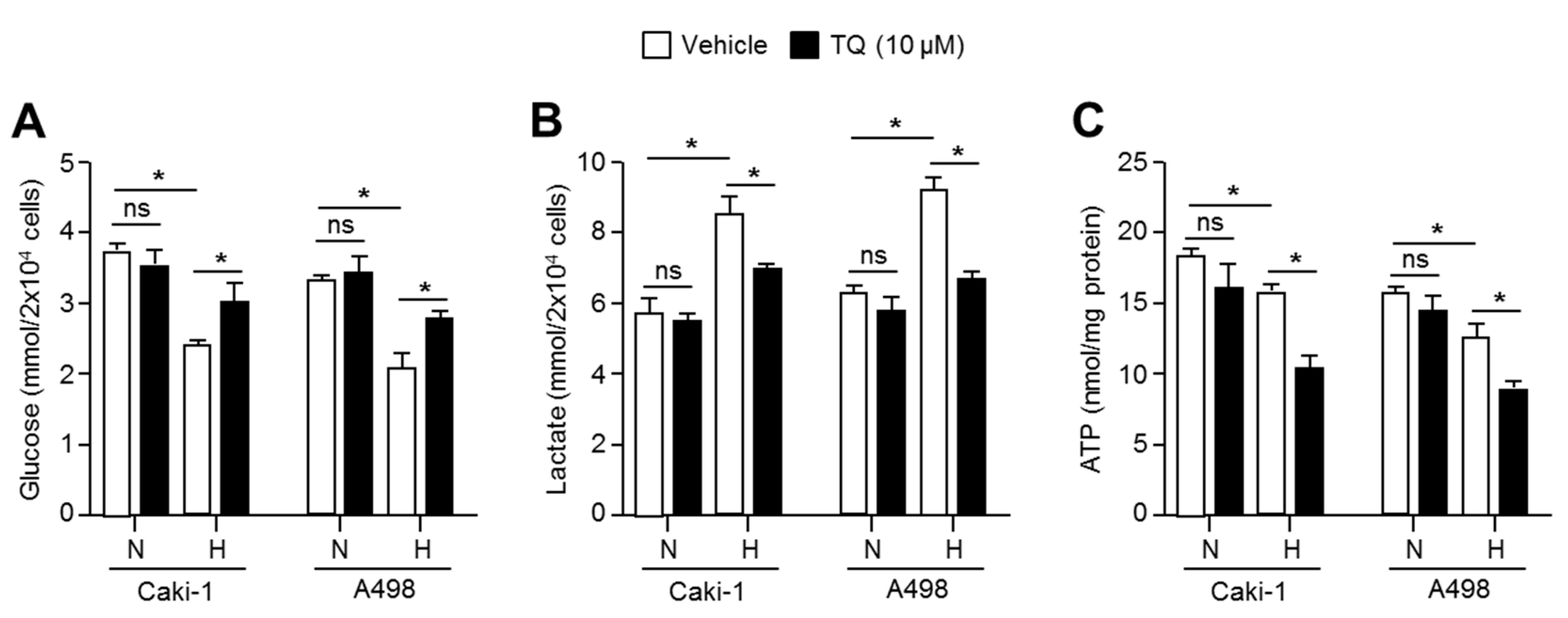
2.6. TQ Suppresses Anaerobic Glycolysis in Hypoxic Renal Cancer Cells
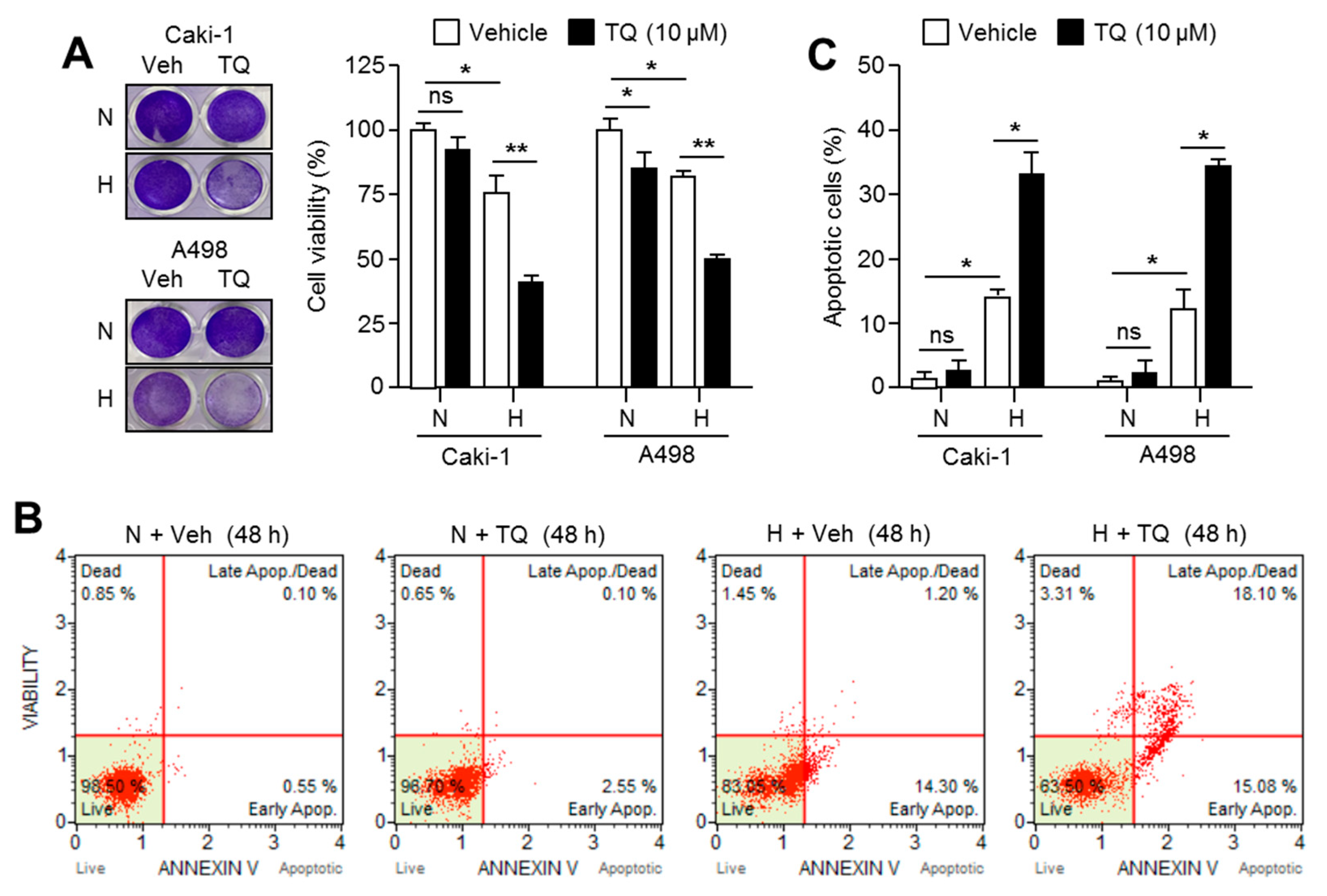
2.7. TQ Selectively Promotes Apoptosis in Hypoxic Renal Cancer Cells
3. Materials and Methods
3.1. Reagents and Antibodies
3.2. Cell Culture and Cell Viability Assay
3.3. Immunoprecipitation and Western Blotting
3.4. Quantitative Real-Time PCR
3.5. Apoptosis Assay
3.6. Luciferase Assay
3.7. Measurement of Glucose Consumption, ATP, and Lactate Production
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HIF-1α | Hypoxia-inducible factor-1alpha |
| HRE | Hypoxia response element |
| VEGFR | Vascular endothelial growth factor receptor |
| mTOR | Mammalian target of rapamycin |
| MAPK | Mitogen-activated protein kinase |
| NF-κB | Nuclear factor-κB |
| PHD | Prolyl hydroxylase |
| VHL | Von-hippel linadu |
| DMOG | Dimethyloxaloylglycine |
| DFO | Deferoxamine |
| HSP90 | Heat shock protein 90 |
| CA-IX | Carbonic anhydrase 9 |
| PDK1 | pyruvate dehydrogenase kinase 1 |
| GLUT1 | Glucose transporter 1 |
| VEGF | Vascular endothelial growth factor A |
| LDHA | Lactate dehydrogenase A |
| FN1 | Fibronectin 1 |
| LOXL2 | Lysyl oxidase homolog 2 |
References
- Shinojima, T.; Oya, M.; Takayanagi, A.; Mizuno, R.; Shimizu, N.; Murai, M. Renal cancer cells lacking hypoxia inducible factor (HIF)-1alpha expression maintain vascular endothelial growth factor expression through HIF-2alpha. Carcinogenesis 2007, 3, 529–536. [Google Scholar] [CrossRef]
- Zhang, T.; Niu, X.; Liao, L.; Cho, E.A.; Yang, H. The contributions of HIF-target genes to tumor growth in RCC. PLoS ONE 2013, 8, e80544. [Google Scholar] [CrossRef] [PubMed]
- Darakhshan, S.; Bidmeshki Pour, A.; Hosseinzadeh Colagar, A.; Sisakhtnezhad, S. Thymoquinone and its therapeutic potentials. Pharmacol. Res. 2015, 95, 138–158. [Google Scholar] [CrossRef] [PubMed]
- Noorbakhsh, M.F.; Hayati, F.; Samarghandian, S.; Shaterzadeh-Yazdi, H.; Farkhondeh, T. An overview of hepatoprotective effects of thymoquinone. Recent Pat. Food Nutr. Agric. 2018, 1, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Farkhondeh, T.; Samarghandian, S.; Shahri, A.M.P.; Samini, F. The neuroprotective effects of thymoquinone: A review. Dose Response 2018, 16. [Google Scholar] [CrossRef] [PubMed]
- Samarghandian, S.; Farkhondeh, T.; Samini, F. A review on possible therapeutic effect of Nigella sativa and thymoquinone in neurodegenerative diseases. CNS Neurol. Disord. Drug Targets 2018, 17, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Rauf, A.; Khan, I.A.; Shahbaz, M.; Qaisrani, T.B.; Fatmawati, S.; Abu-Izneid, T.; Imranh, A.; Rahman, K.U.; Gondal, T.A. Thymoquinone: A novel strategy to combat cancer: A review. Biomed. Pharmacother. 2018, 106, 390–402. [Google Scholar] [CrossRef] [PubMed]
- Mostofa, A.G.M.; Hossain, M.K.; Basak, D.; Bin Sayeed, M.S. Thymoquinone as a potential adjuvant therapy for cancer treatment: Evidence from preclinical studies. Front. Pharmacol. 2017, 8, 295. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Hypoxia-inducible factors: Mediators of cancer progression and targets for cancer therapy. Trends Pharmacol. Sci. 2012, 4, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Powis, G.; Kirkpatrick, L. Hypoxia inducible factor-1alpha as a cancer drug target. Mol. Cancer Ther. 2004, 5, 647–654. [Google Scholar]
- Majkundar, A.J.; Wong, W.J.; Simon, M.C. Hypoxia-inducible factors and the response to hypoxic stress. Mol. Cell 2010, 2, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Isaacs, J.S.; Jung, Y.J.; Mimnaugh, E.G.; Martinez, A.; Cuttitta, F.; Neckers, L.M. Hsp90 regulates a von Hippel Lindau-independent hypoxia-inducible factor-1 alpha-degradative pathway. J. Biol. Chem. 2002, 33, 29936–29944. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.O.; Hahn, T.; Franke, C.; Stiehl, D.P.; Wirthner, R.; Wenger, R.H.; Katschinski, D.M. Induction of the hypoxia-inducible factor system by low levels of heat shock protein 90 inhibitors. Cancer Res. 2005, 23, 11094–11100. [Google Scholar] [CrossRef] [PubMed]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 5930, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Simon, M.C. Oxygen availability and metabolic reprogramming in cancer. J. Biol. Chem. 2017, 41, 16825–16832. [Google Scholar] [CrossRef] [PubMed]
- Pelicano, H.; Martin, D.S.; Xu, R.H.; Huang, P. Glycolysis inhibition for anticancer treatment. Oncogene 2006, 34, 4633–4646. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Lee, G.; Oh, T.I.; Kim, B.M.; Shim, D.W.; Lee, K.H.; Jun, Y.; Lim, K.B.O.; Lim, J.H. Inhibition of glutamine utilization sensitizes lung cancer cells to apigenin-induced apoptosis resulting from metabolic and oxidative stress. Int. J. Oncol. 2016, 1, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.L.; Jiang, B.H.; Semenza, G.L. Effect of protein kinase and phosphatase inhibitors on expression of hypoxia-inducible factor 1. Biochem. Biophys. Res. Commun. 1995, 2, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.E.; Kim, H.S.; Lee, C.S.; Park, D.H.; Kim, Y.N.; Lee, M.J.; Park, J.W.; Kim, M.S.; Ye, S.K.; Chung, M.H. Caffeic acid and its synthetic derivative CADPE suppress tumor angiogenesis by blocking STAT3-mediated VEGF expression in human renal carcinoma cells. Carcinogenesis 2007, 8, 1780–1787. [Google Scholar] [CrossRef] [PubMed]
- Bae, M.K.; Kim, S.H.; Jeong, J.W.; Lee, Y.M.; Kim, H.S.; Kim, S.R.; Yun, I.; Bae, S.K.; Kim, K.W. Curcumin inhibits hypoxia-induced angiogenesis via down-regulation of HIF-1. Oncol. Rep. 2006, 6, 1557–1562. [Google Scholar] [CrossRef]
- Choi, D.; Han, J.; Lee, Y.; Chio, J.; Han, S.; Hong, S.; Jeon, H.; Kim, Y.M.; Jung, Y. Caffeic acid phenethyl ester is a potent inhibitor of HIF prolyl hydroxylase: Structural analysis and pharmacological implication. J. Nutr. Biochem. 2010, 9, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Kim, M.H. Epigallocatechin gallate inhibits HIF-1alpha degradation in prostate cancer cells. Biochem. Biophys. Res. Commun. 2005, 2, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.J.; Chua, Y.L.; Chew, E.H.; Gao, J.; Bushell, M.; Hagen, T. Inhibition of hypoxia-inducible factor-1alpha (HIF-1alpha) protein synthesis by DNA damage inducing agents. PLoS ONE 2010, 5, e10522. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Chiles, K.; Feldser, D.; Laughner, E.; Hanrahan, C.; Georgescu, M.M.; Simons, J.W.; Semenza, G.L. Modulation of hypoxia-inducible factor 1ahpha expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: Implications for tumor angiogenesis and therapeutics. Cancer Res. 2000, 6, 1541–1545. [Google Scholar]
- Wilson, W.J.; Poellinger, L. The dietary flavonoid quercetin modulates HIF-1 alpha activity in endothelial cells. Biochem. Biophys. Res. Commun. 2002, 1, 446–450. [Google Scholar] [CrossRef]
- Zhang, Q.; Tang, X.; Lu, Q.Y.; Zhang, Z.F.; Brown, J.; Le, A.D. Resveratrol inhibits hypoxia-induced accumulation of hypoxia-inducible factor-1alpha and VEGF expression in human tongue squamous cell carcinoma and hepatoma cells. Mol. Cancer Ther. 2005, 10, 1465–1474. [Google Scholar] [CrossRef] [PubMed]
- Triantafyllou, A.; Mylonis, I.; Simos, G.; Bonanou, S.; Tsakalof, A. Flavonoids induce HIF-1alpha but impair its nuclear accumulation and activity. Free Radic. Biol. Med. 2008, 4, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Hur, E.; Kim, H.H.; Choi, S.M.; Kim, J.H.; Yim, S.; Kwon, H.J.; Choi, Y.; Kim, D.K.; Lee, M.O.; Park, H. Reduction of hypoxia-induced transcription through the repression of hypoxia-inducible factor-1alpha/aryl hydrocarbon receptor nuclear translocator DNA binding by the 90-kDa heat-shock protein inhibitor radicicol. Mol. Pharmacol. 2002, 5, 975–982. [Google Scholar] [CrossRef]
- Song, X.; Yao, J.; Wang, F.; Zhou, M.; Zhou, Y.; Wang, H.; Wei, L.; Zhao, L.; Li, Z.; Lu, N.; et al. Wogonin inhibits tumor angiogenesis via degradation of HIF-1α protein. Toxicol. Appl. Pharmacol. 2013, 2, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Vavilala, D.T.; Ponnaluri, V.K.; Vadlapatla, R.K.; Pal, D.; Mitra, A.K.; Mukherji, M. Honokiol inhibits HIF pathway and hypoxia-induced expression of histone lysine demethylases. Biochem. Biophys. Res. Commun. 2012, 3, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Liu, A.; Shen, Y.; Xu, H.Z.; Yang, S.Z.; Ying, X.Z.; Liao, W.; Liu, H.X.; Lin, Z.Q.; Chen, Q.Y.; et al. Antitumor and anti-angiogenesis effects of thymoquinone on osteosarcoma through the NF-kB pathway. Oncol. Rep. 2013, 2, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Cho, S.G.; Yi, Z.; Pang, X.; Rodriguez, M.; Wang, Y.; Sethi, G.; Aggarwal, B.B.; Liu, M. Thymoquinone inhibits tumor angiogenesis and tumor growth through suppressing AKT and extracellular signal-regulated kinase signaling pathways. Mol. Cancer Ther. 2008, 7, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Mabjeesh, N.J.; Post, D.E.; Willard, M.T.; Kaur, B.; Van Meir, E.G.; Simons, J.W.; Zhong, H. Geldanamycin induces degradation of hypoxia-inducible factor 1alpha protein via the proteasome pathway in prostate cancer cells. Cancer Res. 2002, 9, 2478–2482. [Google Scholar]
- Altman, B.J.; Rathmell, J.C. Metabolic stress in autophagy and cell death pathways. Cold Spring Harb. Perspect. Biol. 2012, 9, a008763. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Zhao, S.; Han, J.; Zheng, L.; Yang, Z.; Zhao, L. Hypoxia-inducible factor-1α induces multidrug resistance protein in colon cancer. Onco Targets Ther. 2015, 8, 1941–1948. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, R.; Pare, G.C.; Frederiksen, L.J.; Semenza, G.L.; Graham, C.H. Hypoxia-induced resistance to anticancer drugs is associated with decreased senescence and requires hypoxia-inducible factor-1 activity. Mol. Cancer Ther. 2008, 7, 1961–1973. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Wu, X.; Dai, L.; Ge, Z.; Gao, C.; Zhang, H.; Wang, F.; Zhang, X.; Chen, B. Gambogenic acid exerts antitumor activity in hypoxic multiple myeloma cells by regulation of miR-21. J. Cancer 2017, 16, 3278–3286. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Shin, H.W.; Chun, Y.S.; Leutou, A.S.; Son, B.W.; Park, J.W. Diacetoxyscirpenol as a new anticancer agent to target hypoxia-inducible factor 1. Oncotarget 2016, 38, 62107–62122. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Lee, Y.M.; Oh, T.I.; Shin, D.H.; Kim, G.H.; Kan, S.Y.; Kang, H.; Kim, J.H.; Kim, B.M.; Yim, W.J.; et al. Emodin sensitizes hepatocellular carcinoma cells to the anti-cancer effect of sorafenib through suppression of cholesterol metabolism. Int. J. Mol. Sci. 2018, 10, 3127. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Won, H.S.; Lee, Y.M.; Choi, J.W.; Oh, T.I.; Jang, J.H.; Choi, D.K.; Lim, B.O.; Kim, Y.J.; Park, J.W.; et al. Oxidative dimerization of PHD2 is responsible for its inactivation and contributes to metabolic reprogramming via HIF-1α activation. Sci. Rep. 2016, 6, 18928. [Google Scholar] [CrossRef] [PubMed]
| Gene | Forward Primer | Reverse Primer |
|---|---|---|
| FN1 | GGTGACACTTATGAGCGTCCTAAA | AACATGTAACCACCAGTCTCATGTG |
| uPAR | GGTGACGCCTTCAGCATGA | CCCACTGCGGTACTGGACAT |
| VEGFA | AGCTGCGCTGATAGACATCC | CTACCTCCACCATGCCAAGT |
| CA-IX | GCGACGCAGCCTTTGAAT | CCACTCCAGCAGGGAAGGA |
| GLUT1 | GGCATTGATGACTCCAGTGTT | ATGGAGCCCAGCAGCAA |
| PDK1 | ATGATGTCATTCCCACAATGGCCC | TGAACATTCTGGCTGGTGACAGGA |
| LDHA | ACCCAGTTTCCACCATGATT | CCCAAAATGCAAGGAACACT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-M.; Kim, G.-H.; Park, E.-J.; Oh, T.-I.; Lee, S.; Kan, S.-Y.; Kang, H.; Kim, B.M.; Kim, J.H.; Lim, J.-H. Thymoquinone Selectively Kills Hypoxic Renal Cancer Cells by Suppressing HIF-1α-Mediated Glycolysis. Int. J. Mol. Sci. 2019, 20, 1092. https://doi.org/10.3390/ijms20051092
Lee Y-M, Kim G-H, Park E-J, Oh T-I, Lee S, Kan S-Y, Kang H, Kim BM, Kim JH, Lim J-H. Thymoquinone Selectively Kills Hypoxic Renal Cancer Cells by Suppressing HIF-1α-Mediated Glycolysis. International Journal of Molecular Sciences. 2019; 20(5):1092. https://doi.org/10.3390/ijms20051092
Chicago/Turabian StyleLee, Yoon-Mi, Geon-Hee Kim, Eun-Ji Park, Taek-In Oh, Sujin Lee, Sang-Yeon Kan, Hyeji Kang, Byeong Mo Kim, Ji Hyung Kim, and Ji-Hong Lim. 2019. "Thymoquinone Selectively Kills Hypoxic Renal Cancer Cells by Suppressing HIF-1α-Mediated Glycolysis" International Journal of Molecular Sciences 20, no. 5: 1092. https://doi.org/10.3390/ijms20051092
APA StyleLee, Y.-M., Kim, G.-H., Park, E.-J., Oh, T.-I., Lee, S., Kan, S.-Y., Kang, H., Kim, B. M., Kim, J. H., & Lim, J.-H. (2019). Thymoquinone Selectively Kills Hypoxic Renal Cancer Cells by Suppressing HIF-1α-Mediated Glycolysis. International Journal of Molecular Sciences, 20(5), 1092. https://doi.org/10.3390/ijms20051092




