Exosomal Expression of CXCR4 Targets Cardioprotective Vesicles to Myocardial Infarction and Improves Outcome after Systemic Administration
Abstract
1. Introduction
2. Results
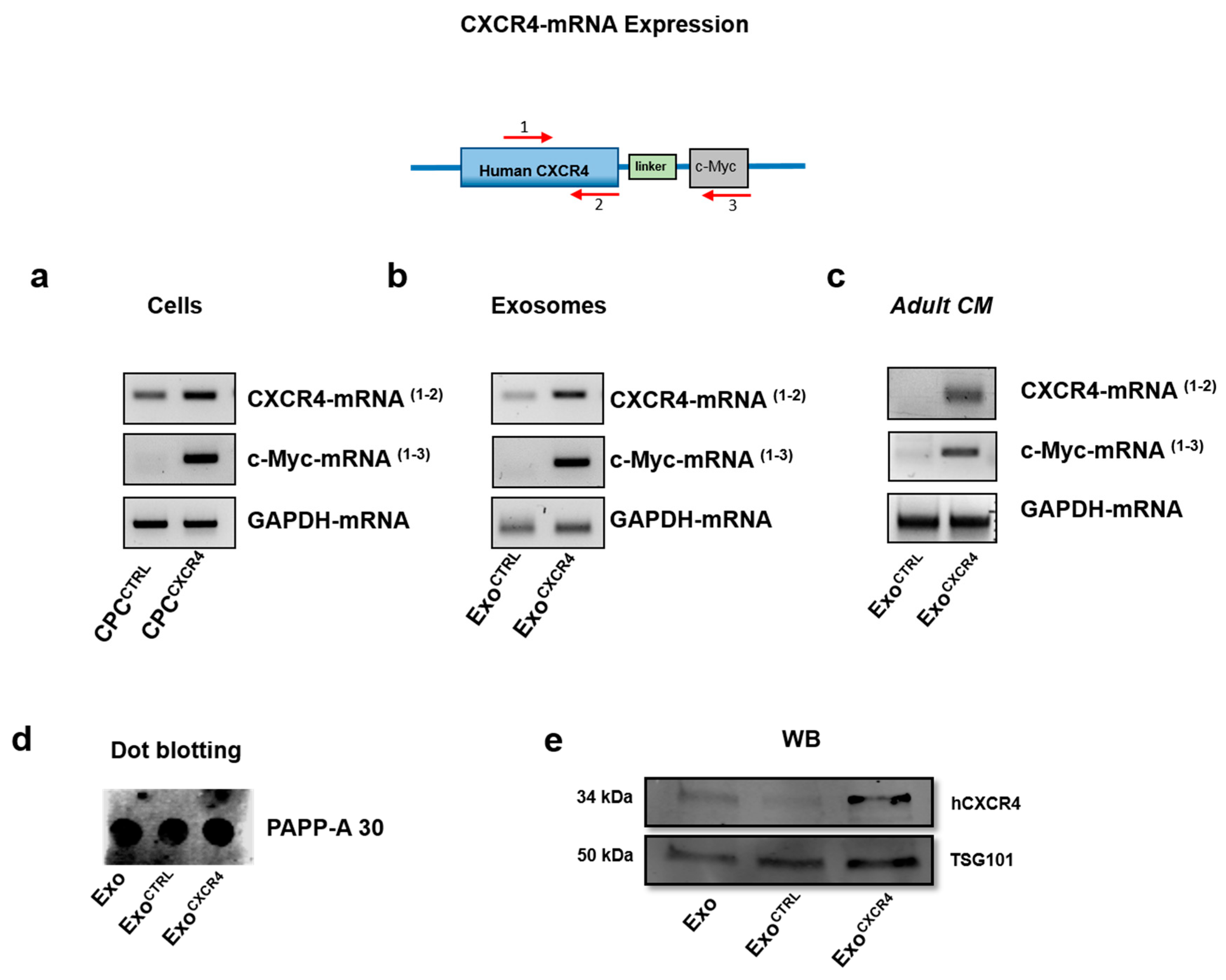
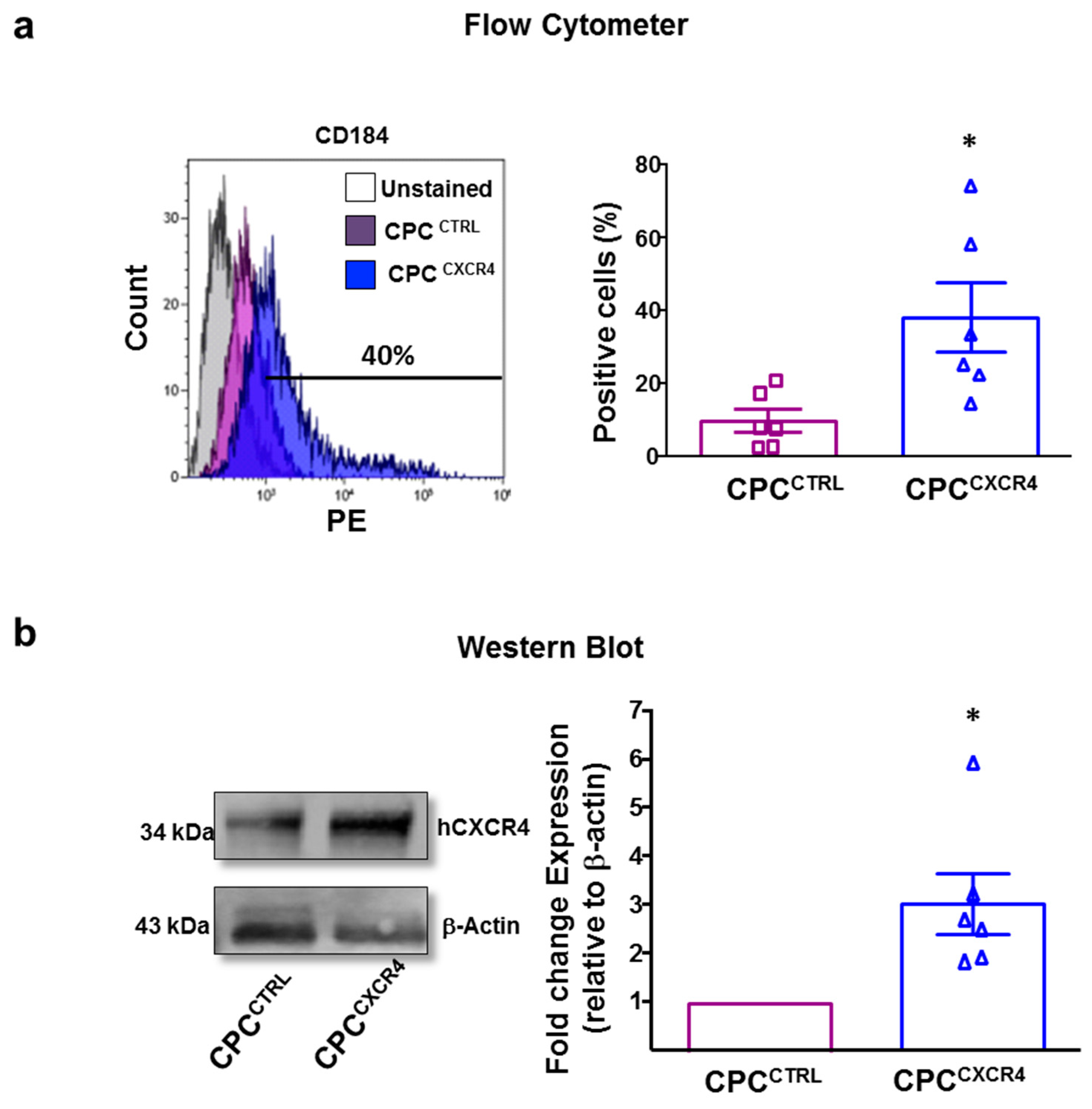
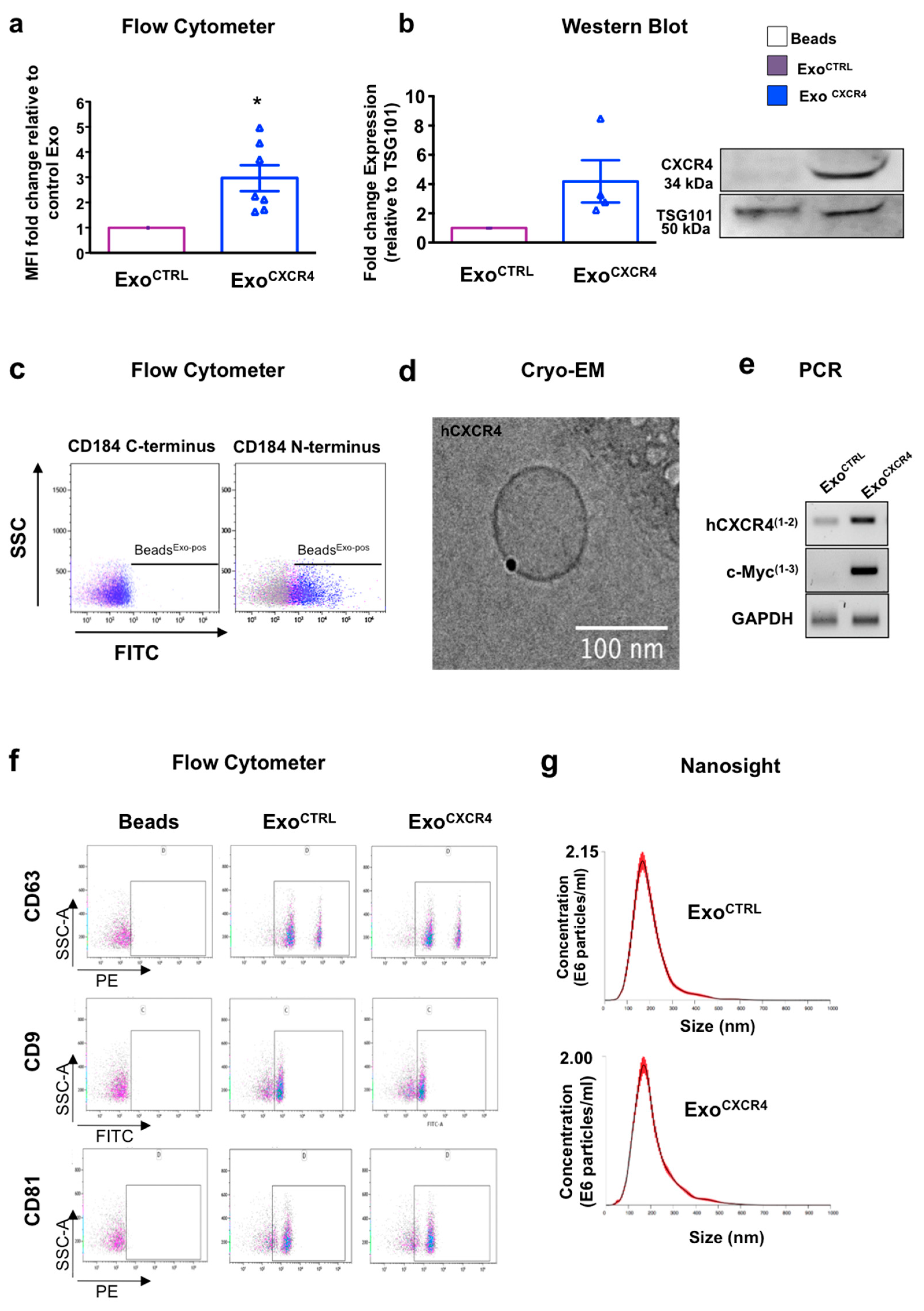
2.1. CPCCXCR4 Load Exogenous Receptor into Exo at Protein and mRNA Levels
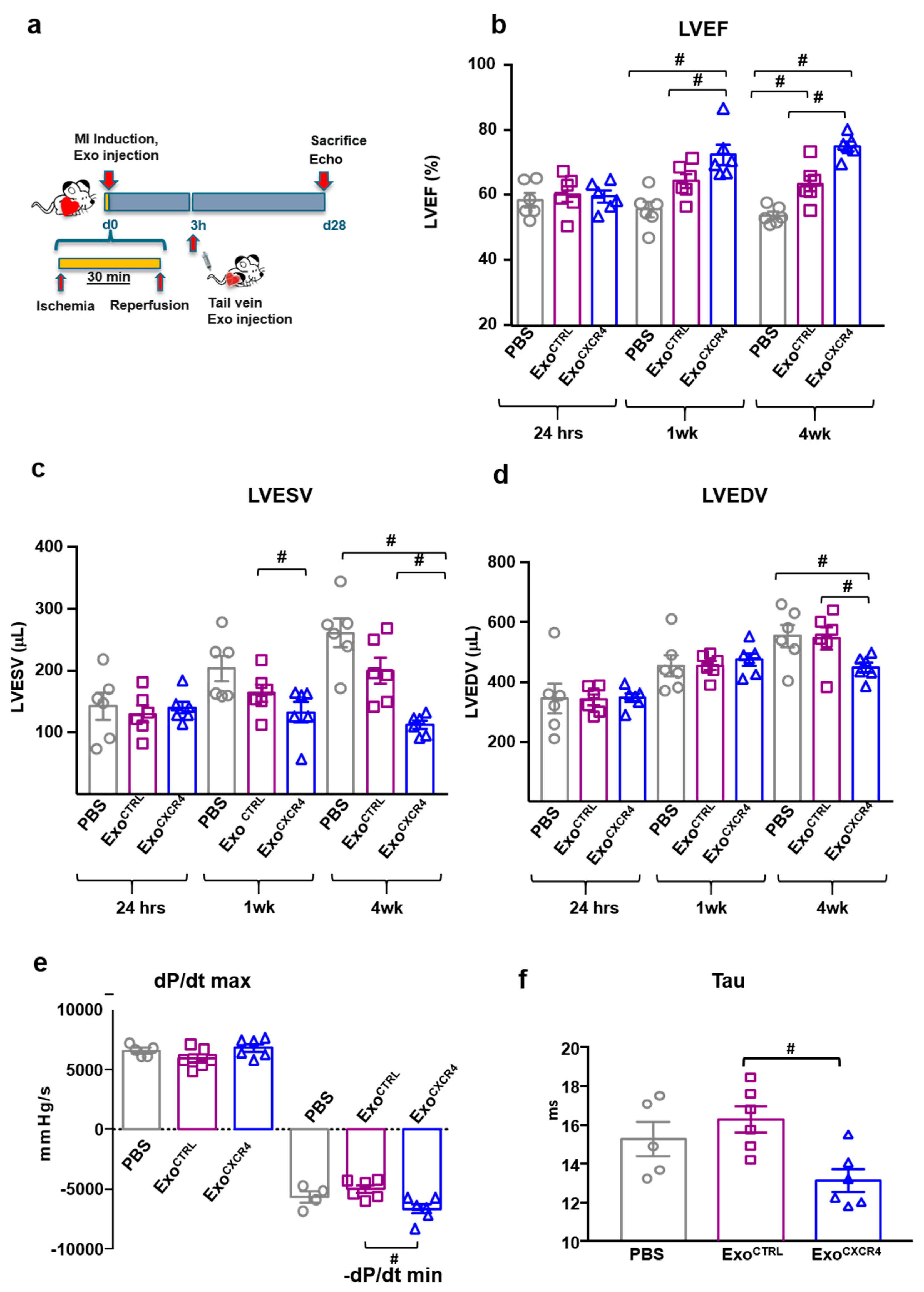
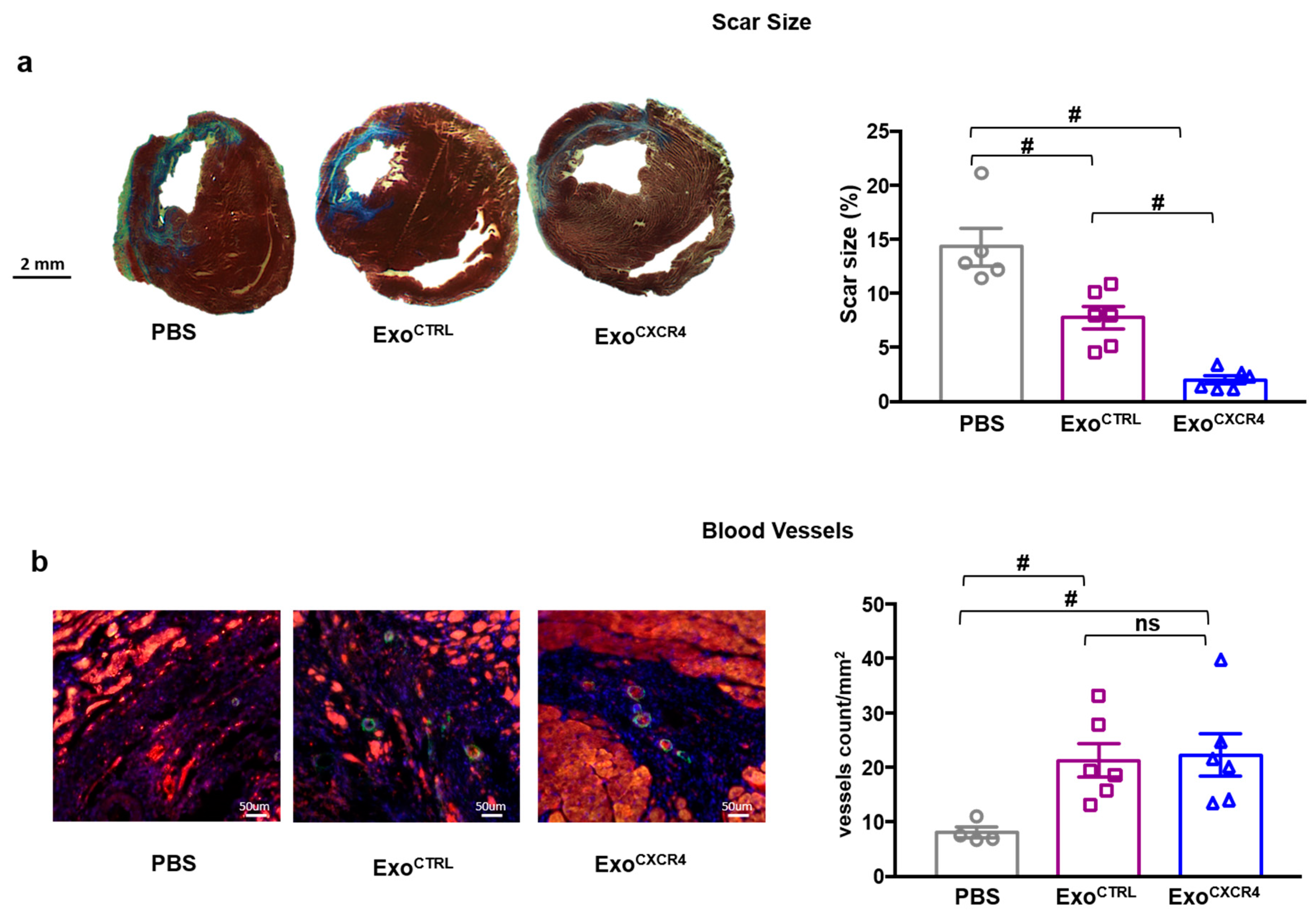
2.2. Intravenous Injection of ExoCXCR4, but not ExoCTRL, Is Protective after In Vivo I/R Injury
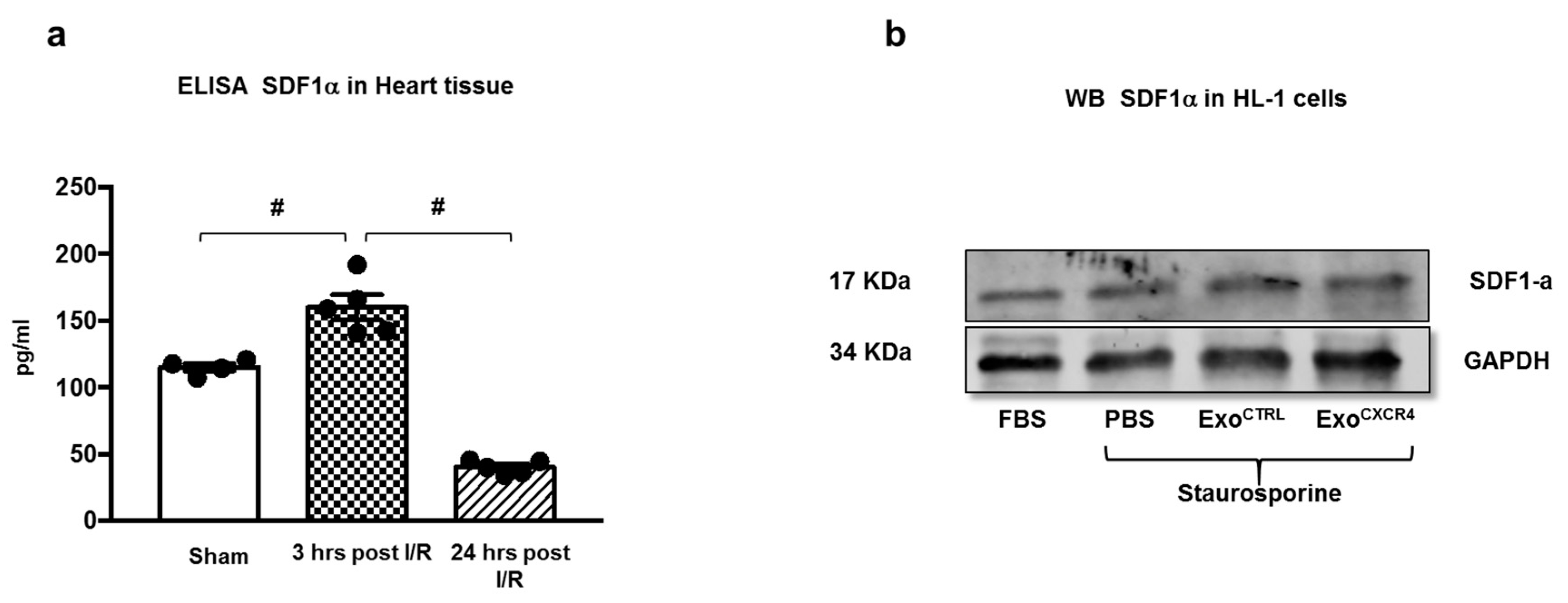
2.3. ExoCXCR4 Is Delivered More Efficiently than ExoCTRL to CM in Ischemic/Reperfused Rat Hearts Ex Vivo
2.4. ExoCXCR4 Shows Superior Ex Vivo Cardioprotective Effects Compared with ExoCTRL
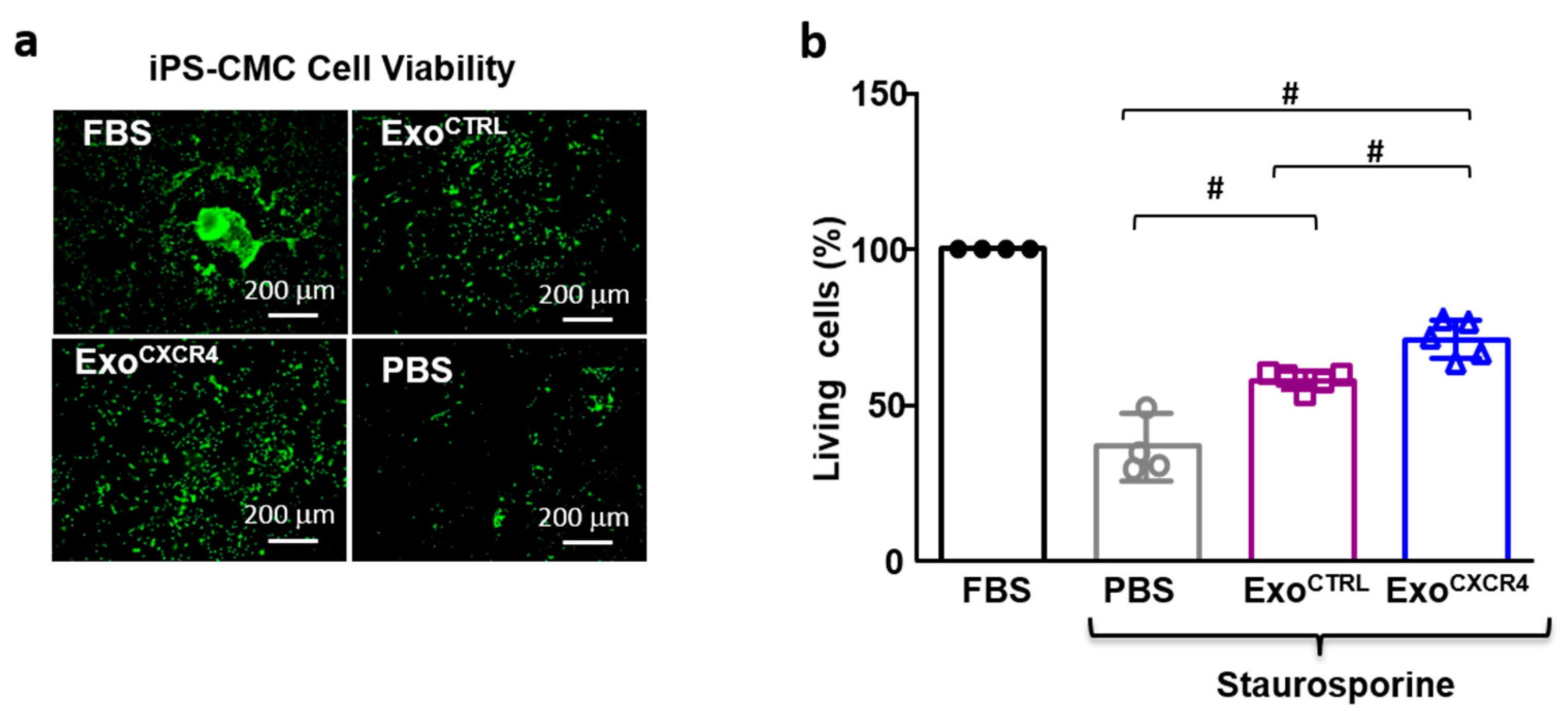
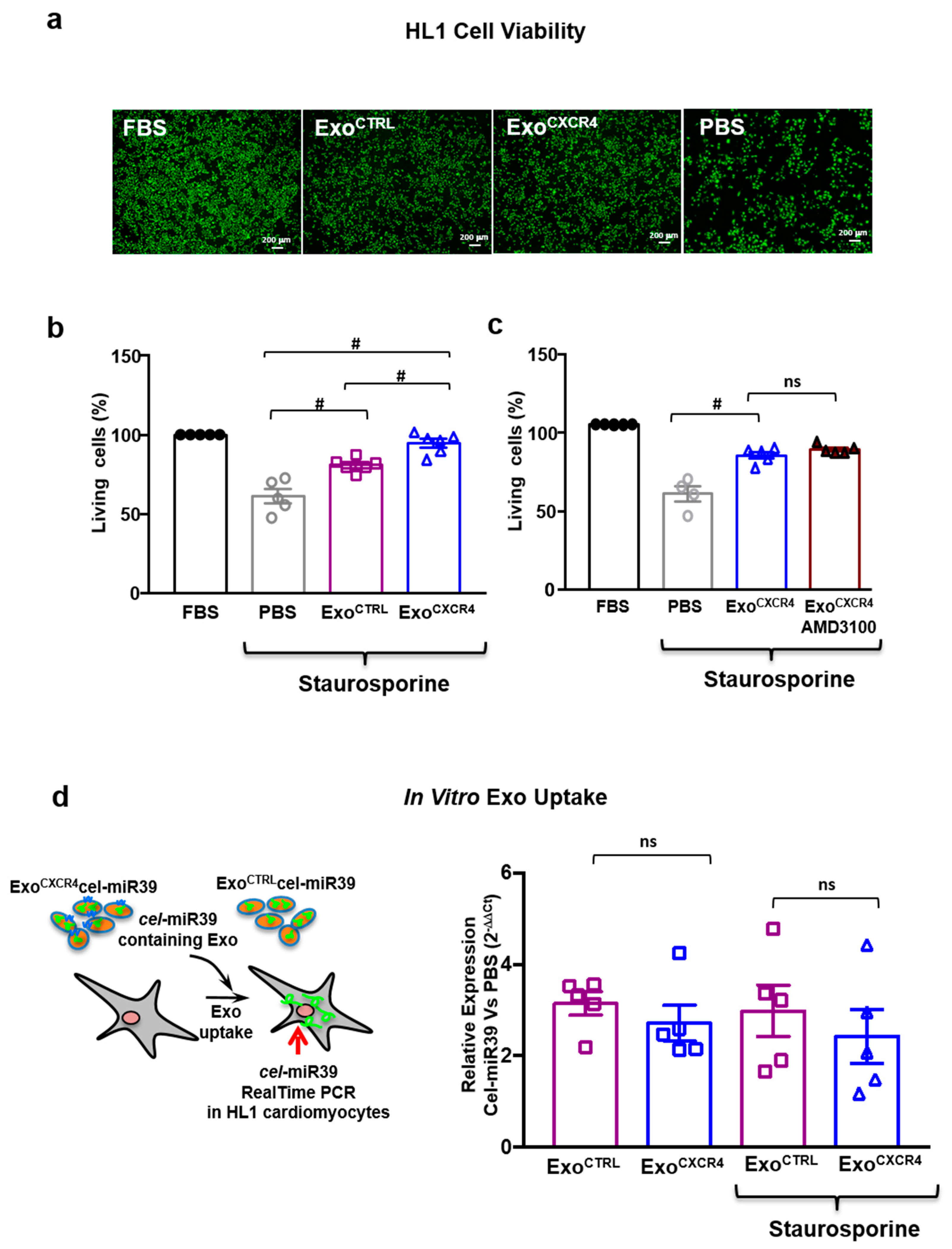
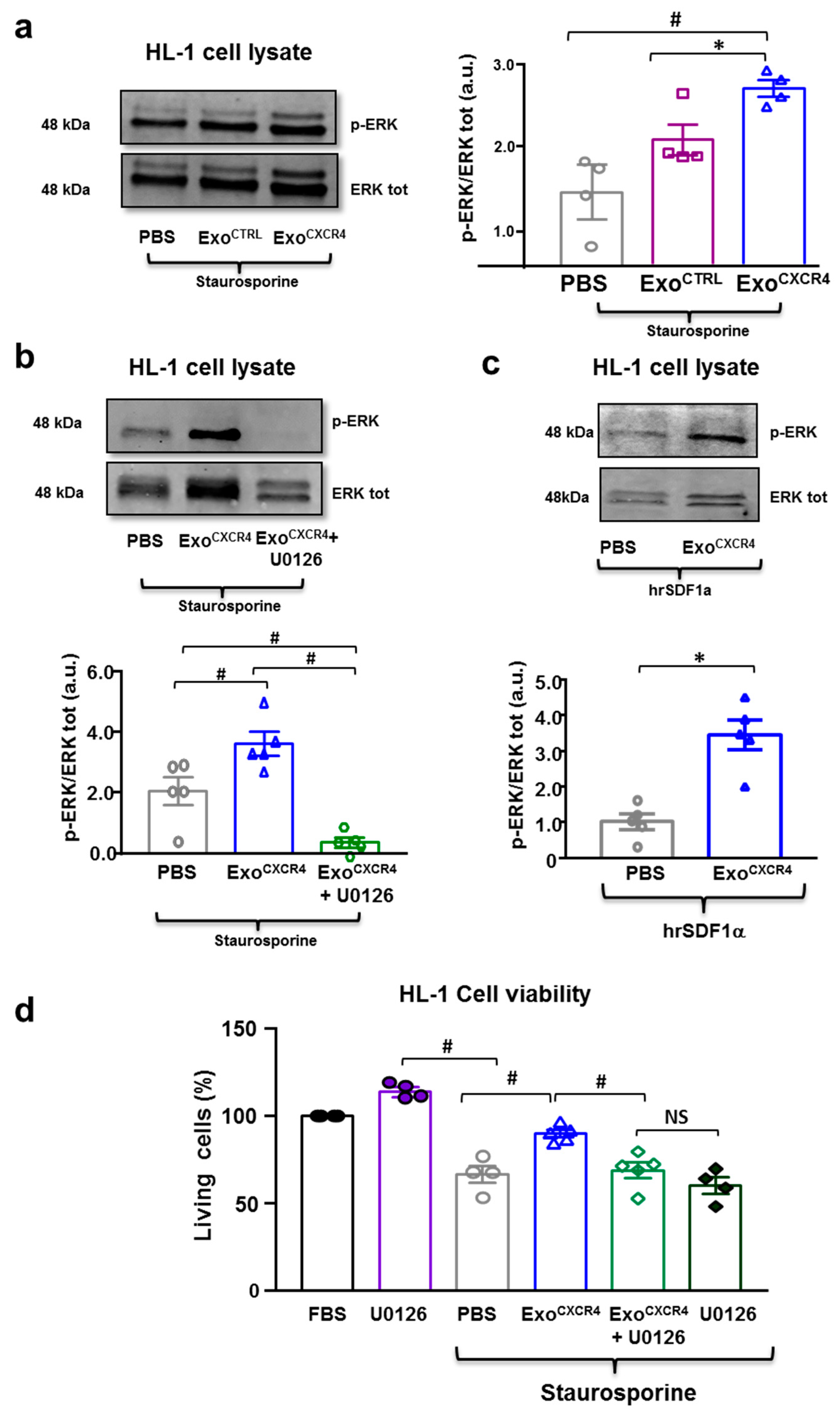
2.5. ExoCXCR4 Shows Superior In Vitro Cardioprotective Effects Compared with ExoCTRL
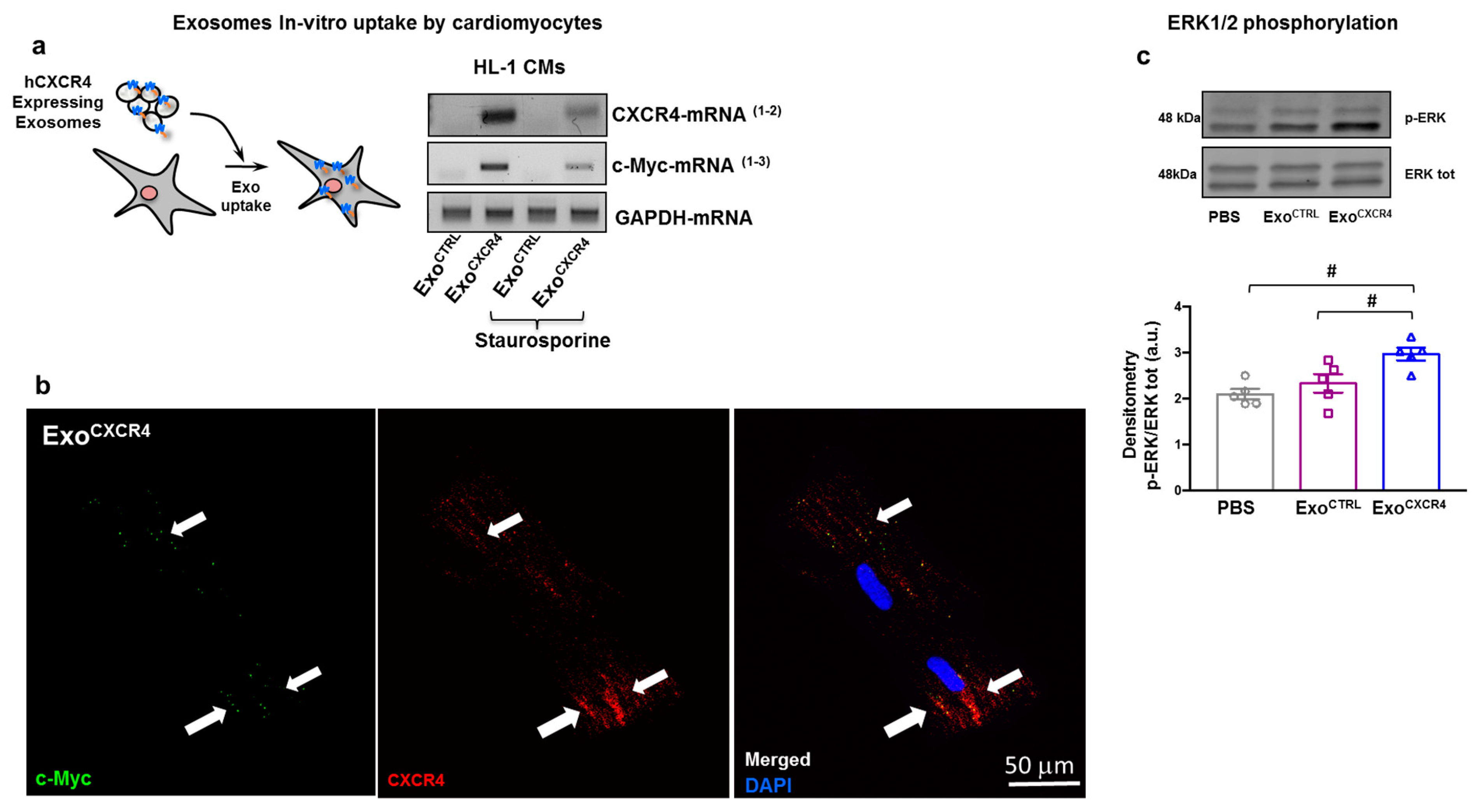
2.6. ExoCXCR4 Transfers CXCR4 mRNA and Protein into CM In Vitro
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. CPC Engineering to Overexpress CXCR4
4.3. Particle Purification
4.4. Flow Cytometry
4.5. Immuno-Gold Labeling and Cryo-Electron Microscopy(Cryo-EM)
4.6. Protein Isolation and Western Blot Analyses
4.7. In Vitro and Ex Vivo Exo Uptake by CM
4.8. Levels of Exogenous mRNA into CM
4.9. In Vitro CM Viability Assay
4.10. RNA Extraction, Reverse Transcription and Real-Time PCR
4.11. Immunofluorescence and Histology
4.12. SDF-1α ELISA
4.13. TUNEL Assay
4.14. Animal Experiments
4.15. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| hDF-1 | Human Recombinant SDF-1 |
| Cryo-EM | Cryo Electron Microscopy |
| IMDM | Iscove’s Modified Dulbecco’s Medium |
| Exo-CPC | Exosomes Produced by Cardiac-Resident Progenitor Cells |
| HIF-1 | Hypoxia-Induced Factor 1 |
| Exo | Exosomes |
| CPC | Cardiac-Resident Progenitor Cells |
| MSC | Bone Marrow-Derived Mesenchymal Stem Cells |
| Exo CXCR4 | Exosomes Expressing CXCR4 |
| CPC CXCR4 | Cardiac-Resident Progenitor Cells that Overexpress CXCR4 |
| FBS | Fetal Bovine Serum |
| CPC CTRL | Control Cardiac-Resident Progenitor Cells |
| Exo CTRL | Exosomes Produced by Control Cardiac-Resident Progenitor Cells |
| SMA | Smooth Muscle Actin |
| TnI | Troponin I |
| MI | Myocardial Infarction |
| CM | Cardiomyocyte |
| EV | Extracellular Vesicle |
| IC | Intracoronary |
| IM | Intramyocardial |
| I/R | Ischemia/Reperfusion |
| FC | Flow Citometry |
| WB | Western Blot |
| PE | Phycoerythrin |
Appendix A
References
- Eschenhagen, T.; Bolli, R.; Braun, T.; Field, L.J.; Fleischmann, B.K.; Frisen, J.; Giacca, M.; Hare, J.M.; Houser, S.; Lee, R.T.; et al. Cardiomyocyte Regeneration: A Consensus Statement. Circulation 2017, 136, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.; Shim, W. Cardiac stem cell therapy: Stemness or commitment? Cell Transplant. 2013, 22, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Gaetani, R.; Barile, L.; Forte, E.; Chimenti, I.; Ionta, V.; Di Consiglio, A.; Miraldi, F.; Frati, G.; Messina, E.; Giacomello, A. New perspectives to repair a broken heart. Cardiovasc. Hematol. Agents Med. Chem. 2009, 7, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Lefer, D.J.; Marban, E. Is Cardioprotection Dead? Circulation 2017, 136, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Serebruany, V.L.; Atar, D. Assessment of bleeding events in clinical trials—Proposal of a new classification. Am. J. Cardiol. 2007, 99, 288–290. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Cervio, E.; Lionetti, V.; Milano, G.; Ciullo, A.; Biemmi, V.; Bolis, S.; Altomare, C.; Matteucci, M.; Disilvestre, D.; et al. Cardioprotection by cardiac progenitor cell-secreted exosomes: Role of pregnancy-associated plasma protein-A. Cardiovasc. Res. 2018, 114, 992–1005. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Lionetti, V.; Cervio, E.; Matteucci, M.; Gherghiceanu, M.; Popescu, L.M.; Torre, T.; Siclari, F.; Moccetti, T.; Vassalli, G. Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc. Res. 2014, 103, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.G.; Cheng, K.; Marban, E. Exosomes as critical agents of cardiac regeneration triggered by cell therapy. Stem Cell Rep. 2014, 2, 606–619. [Google Scholar] [CrossRef]
- Lai, R.C.; Arslan, F.; Lee, M.M.; Sze, N.S.; Choo, A.; Chen, T.S.; Salto-Tellez, M.; Timmers, L.; Lee, C.N.; El Oakley, R.M.; et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010, 4, 214–222. [Google Scholar] [CrossRef]
- Barile, L.; Milano, G.; Vassalli, G. Beneficial effects of exosomes secreted by cardiac-derived progenitor cells and other cell types in myocardial ischemia. Stem Cell Investig. 2017, 4, 93. [Google Scholar] [CrossRef]
- Barile, L.; Gherghiceanu, M.; Popescu, L.M.; Moccetti, T.; Vassalli, G. Ultrastructural evidence of exosome secretion by progenitor cells in adult mouse myocardium and adult human cardiospheres. J. Biomed. Biotechnol. 2012, 20, 2012. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Vassalli, G. Exosomes: Therapy delivery tools and biomarkers of diseases. Pharmacol. Ther. 2017, 174, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Thery, C. Exosomes: Secreted vesicles and intercellular communications. F1000 Biol. Rep. 2011, 3, 15. [Google Scholar] [CrossRef] [PubMed]
- Cervio, E.; Barile, L.; Moccetti, T.; Vassalli, G. Exosomes for Intramyocardial Intercellular Communication. Stem Cells Int. 2015, 2015, 482171. [Google Scholar] [CrossRef] [PubMed]
- Gallet, R.; Dawkins, J.; Valle, J.; Simsolo, E.; de Couto, G.; Middleton, R.; Tseliou, E.; Luthringer, D.; Kreke, M.; Smith, R.R.; et al. Exosomes secreted by cardiosphere-derived cells reduce scarring, attenuate adverse remodelling, and improve function in acute and chronic porcine myocardial infarction. Eur. Heart J. 2016, 38, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Wang, H.; Zhang, X.; Zhao, S.; Zhou, Z.; Mu, X.; Zhao, C.; Teng, W. The Role of SDF-1/CXCR4/CXCR7 in Neuronal Regeneration after Cerebral Ischemia. Front. Neurosci. 2017, 11, 590. [Google Scholar] [CrossRef] [PubMed]
- Uematsu, M.; Yoshizaki, T.; Shimizu, T.; Obata, J.E.; Nakamura, T.; Fujioka, D.; Watanabe, K.; Watanabe, Y.; Kugiyama, K. Sustained myocardial production of stromal cell-derived factor-1alpha was associated with left ventricular adverse remodeling in patients with myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2015, 309, H1764–H1771. [Google Scholar] [CrossRef] [PubMed]
- Ceradini, D.J.; Kulkarni, A.R.; Callaghan, M.J.; Tepper, O.M.; Bastidas, N.; Kleinman, M.E.; Capla, J.M.; Galiano, R.D.; Levine, J.P.; Gurtner, G.C. Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat. Med. 2004, 10, 858–864. [Google Scholar] [CrossRef] [PubMed]
- Peled, A.; Petit, I.; Kollet, O.; Magid, M.; Ponomaryov, T.; Byk, T.; Nagler, A.; Ben-Hur, H.; Many, A.; Shultz, L.; et al. Dependence of human stem cell engraftment and repopulation of NOD/SCID mice on CXCR4. Science 1999, 283, 845–848. [Google Scholar] [CrossRef]
- Elmadbouh, I.; Haider, H.; Jiang, S.; Idris, N.M.; Lu, G.; Ashraf, M. Ex vivo delivered stromal cell-derived factor-1alpha promotes stem cell homing and induces angiomyogenesis in the infarcted myocardium. J. Mol. Cell. Cardiol. 2007, 42, 792–803. [Google Scholar] [CrossRef]
- Hu, X.; Dai, S.; Wu, W.J.; Tan, W.; Zhu, X.; Mu, J.; Guo, Y.; Bolli, R.; Rokosh, G. Stromal cell derived factor-1 alpha confers protection against myocardial ischemia/reperfusion injury: Role of the cardiac stromal cell derived factor-1 alpha CXCR4 axis. Circulation 2007, 116, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wang, T.; Zhang, D.; Zhao, T.; Dai, B.; Ashraf, A.; Wang, X.; Xu, M.; Millard, R.W.; Fan, G.C.; et al. Mesenchymal stem cells overexpressing CXCR4 attenuate remodeling of postmyocardial infarction by releasing matrix metalloproteinase-9. Stem Cells Dev. 2012, 21, 778–789. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Z.; Li, Y.L.; Huang, W.; Cai, W.F.; Liang, J.; Paul, C.; Jiang, L.; Wu, Z.C.; Xu, M.; Zhu, P.; et al. Paracrine effect of CXCR4-overexpressing mesenchymal stem cells on ischemic heart injury. Cell Biochem. Funct. 2017, 35, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Fan, G.C.; Zhou, X.; Zhao, T.; Pasha, Z.; Xu, M.; Zhu, Y.; Ashraf, M.; Wang, Y. Over-expression of CXCR4 on mesenchymal stem cells augments myoangiogenesis in the infarcted myocardium. J. Mol. Cell. Cardiol. 2008, 44, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.; Ma, R.; Cai, W.; Huang, W.; Paul, C.; Liang, J.; Wang, Y.; Zhao, T.; Kim, H.W.; Xu, M.; et al. Exosomes Secreted from CXCR4 Overexpressing Mesenchymal Stem Cells Promote Cardioprotection via Akt Signaling Pathway following Myocardial Infarction. Stem Cells Int. 2015, 2015, 659890. [Google Scholar] [CrossRef] [PubMed]
- Botker, H.E.; Hausenloy, D.; Andreadou, I.; Antonucci, S.; Boengler, K.; Davidson, S.M.; Deshwal, S.; Devaux, Y.; Di Lisa, F.; Di Sante, M.; et al. Practical guidelines for rigor and reproducibility in preclinical and clinical studies on cardioprotection. Basic Res. Cardiol. 2018, 113, 39. [Google Scholar] [CrossRef] [PubMed]
- Arslan, F.; Lai, R.C.; Smeets, M.B.; Akeroyd, L.; Choo, A.; Aguor, E.N.; Timmers, L.; van Rijen, H.V.; Doevendans, P.A.; Pasterkamp, G.; et al. Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 2013, 10, 301–312. [Google Scholar] [CrossRef]
- Andriolo, G.; Provasi, E.; Lo Cicero, V.; Brambilla, A.; Soncin, S.; Torre, T.; Milano, G.; Biemmi, V.; Vassalli, G.; Turchetto, L.; et al. Exosomes From Human Cardiac Progenitor Cells for Therapeutic Applications: Development of a GMP-Grade Manufacturing Method. Front. Physiol. 2018, 9, 1169. [Google Scholar] [CrossRef]
- Vandergriff, A.C.; de Andrade, J.B.; Tang, J.; Hensley, M.T.; Piedrahita, J.A.; Caranasos, T.G.; Cheng, K. Intravenous Cardiac Stem Cell-Derived Exosomes Ameliorate Cardiac Dysfunction in Doxorubicin Induced Dilated Cardiomyopathy. Stem Cells Int. 2015, 2015, 960926. [Google Scholar] [CrossRef]
- Marbán, E. A mechanistic roadmap for the clinical application of cardiac cell therapies. Nat. Biomed. Eng. 2018, 2, 353–361. [Google Scholar] [CrossRef]
- Chen, K.H.; Chen, C.H.; Wallace, C.G.; Yuen, C.M.; Kao, G.S.; Chen, Y.L.; Shao, P.L.; Chen, Y.L.; Chai, H.T.; Lin, K.C.; et al. Intravenous administration of xenogenic adipose-derived mesenchymal stem cells (ADMSC) and ADMSC-derived exosomes markedly reduced brain infarct volume and preserved neurological function in rat after acute ischemic stroke. Oncotarget 2016, 7, 74537–74556. [Google Scholar] [CrossRef] [PubMed]
- Sluijter, J.P.G.; Davidson, S.M.; Boulanger, C.M.; Buzas, E.I.; de Kleijn, D.P.V.; Engel, F.B.; Giricz, Z.; Hausenloy, D.J.; Kishore, R.; Lecour, S.; et al. Extracellular vesicles in diagnostics and therapy of the ischaemic heart: Position Paper from the Working Group on Cellular Biology of the Heart of the European Society of Cardiology. Cardiovasc. Res. 2018, 114, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.C.; Andriantsitohaina, R. Extracellular Vesicles in Metabolic Syndrome. Circ. Res. 2017, 120, 1674–1686. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Nishikawa, M.; Shinotsuka, H.; Matsui, Y.; Ohara, S.; Imai, T.; Takakura, Y. Visualization and in vivo tracking of the exosomes of murine melanoma B16-BL6 cells in mice after intravenous injection. J. Biotechnol. 2013, 165, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Csak, T.; Momen-Heravi, F.; Lippai, D.; Kodys, K.; Catalano, D.; Satishchandran, A.; Ambros, V.; Szabo, G. Biodistribution and function of extracellular miRNA-155 in mice. Sci. Rep. 2015, 5, 10721. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.T.; Hartner, W.C.; Kale, A.; Torchilin, V.P. Gene delivery into ischemic myocardium by double-targeted lipoplexes with anti-myosin antibody and TAT peptide. Gene Ther. 2009, 16, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Abe, T.; Sakaue-Sawano, A.; Kiyonari, H.; Shioi, G.; Inoue, K.; Horiuchi, T.; Nakao, K.; Miyawaki, A.; Aizawa, S.; Fujimori, T. Visualization of cell cycle in mouse embryos with Fucci2 reporter directed by Rosa26 promoter. Development 2013, 140, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, Y.; Zhao, Z.; Meng, Q.; Yu, Y.; Sun, J.; Yang, Z.; Chen, Y.; Li, J.; Ma, T.; et al. Engineered Exosomes With Ischemic Myocardium-Targeting Peptide for Targeted Therapy in Myocardial Infarction. J. Am. Heart Assoc. 2018, 7, e008737. [Google Scholar] [CrossRef]
- Muhlstedt, S.; Ghadge, S.K.; Duchene, J.; Qadri, F.; Jarve, A.; Vilianovich, L.; Popova, E.; Pohlmann, A.; Niendorf, T.; Boye, P.; et al. Cardiomyocyte-derived CXCL12 is not involved in cardiogenesis but plays a crucial role in myocardial infarction. J. Mol. Med. 2016, 94, 1005–1014. [Google Scholar] [CrossRef]
- Bobis-Wozowicz, S.; Miekus, K.; Wybieralska, E.; Jarocha, D.; Zawisz, A.; Madeja, Z.; Majka, M. Genetically modified adipose tissue-derived mesenchymal stem cells overexpressing CXCR4 display increased motility, invasiveness, and homing to bone marrow of NOD/SCID mice. Exp. Hematol. 2011, 39, 686–696.e4. [Google Scholar] [CrossRef]
- Muhlethaler-Mottet, A.; Liberman, J.; Ascencao, K.; Flahaut, M.; Balmas Bourloud, K.; Yan, P.; Jauquier, N.; Gross, N.; Joseph, J.M. The CXCR4/CXCR7/CXCL12 Axis Is Involved in a Secondary but Complex Control of Neuroblastoma Metastatic Cell Homing. PLoS ONE 2015, 10, e0125616. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.A.; Maarouf, O.; Cheema, F.H.; Liu, C.; Zhang, Q.Y.; Kraus, C.; Zeeshan Afzal, M.; Firdous, M.; Klinakis, A.; Efstratiadis, A.; et al. SDF-1 activates papillary label-retaining cells during kidney repair from injury. Am. J. Physiol. Renal Physiol. 2012, 302, F1362–F1373. [Google Scholar] [CrossRef] [PubMed]
- Zomer, A.; Maynard, C.; Verweij, F.J.; Kamermans, A.; Schafer, R.; Beerling, E.; Schiffelers, R.M.; de Wit, E.; Berenguer, J.; Ellenbroek, S.I.J.; et al. In Vivo imaging reveals extracellular vesicle-mediated phenocopying of metastatic behavior. Cell 2015, 161, 1046–1057. [Google Scholar] [CrossRef] [PubMed]
- Claycomb, W.C.; Lanson, N.A., Jr.; Stallworth, B.S.; Egeland, D.B.; Delcarpio, J.B.; Bahinski, A.; Izzo, N.J., Jr. HL-1 cells: A cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proc. Natl. Acad. Sci. USA 1998, 95, 2979–2984. [Google Scholar] [CrossRef] [PubMed]
- Altomare, C.; Cervio, E.; Alessandra, C.; Milano, G.; Torre, T.; Demertzis, S.; Barile, L.; Vassalli, G. Induced Pluripotent Stem (IPS) Cells to Assess the Cardioprotective and Proangiogenic Activities of Exosomes Secreted by Human Cardiac Progenitor Cells. Biophys. J. 2016, 110, 595a–596a. [Google Scholar] [CrossRef]
- Alessandra Ciullo, E.C.; Foglieni, C.; Paganetti, P.; Camici, G.; Moccetti, T.; Vassalli, G.; Barile, L. New molecular approaches to quantitatively assess exosome uptake by specific target cells. J. Extracell. Vesicles 2016, 5, PW1.09. [Google Scholar]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciullo, A.; Biemmi, V.; Milano, G.; Bolis, S.; Cervio, E.; Fertig, E.T.; Gherghiceanu, M.; Moccetti, T.; Camici, G.G.; Vassalli, G.; et al. Exosomal Expression of CXCR4 Targets Cardioprotective Vesicles to Myocardial Infarction and Improves Outcome after Systemic Administration. Int. J. Mol. Sci. 2019, 20, 468. https://doi.org/10.3390/ijms20030468
Ciullo A, Biemmi V, Milano G, Bolis S, Cervio E, Fertig ET, Gherghiceanu M, Moccetti T, Camici GG, Vassalli G, et al. Exosomal Expression of CXCR4 Targets Cardioprotective Vesicles to Myocardial Infarction and Improves Outcome after Systemic Administration. International Journal of Molecular Sciences. 2019; 20(3):468. https://doi.org/10.3390/ijms20030468
Chicago/Turabian StyleCiullo, Alessandra, Vanessa Biemmi, Giuseppina Milano, Sara Bolis, Elisabetta Cervio, Emanuel Tudor Fertig, Mihaela Gherghiceanu, Tiziano Moccetti, Giovanni G. Camici, Giuseppe Vassalli, and et al. 2019. "Exosomal Expression of CXCR4 Targets Cardioprotective Vesicles to Myocardial Infarction and Improves Outcome after Systemic Administration" International Journal of Molecular Sciences 20, no. 3: 468. https://doi.org/10.3390/ijms20030468
APA StyleCiullo, A., Biemmi, V., Milano, G., Bolis, S., Cervio, E., Fertig, E. T., Gherghiceanu, M., Moccetti, T., Camici, G. G., Vassalli, G., & Barile, L. (2019). Exosomal Expression of CXCR4 Targets Cardioprotective Vesicles to Myocardial Infarction and Improves Outcome after Systemic Administration. International Journal of Molecular Sciences, 20(3), 468. https://doi.org/10.3390/ijms20030468






