A Novel Hepatic Anti-Fibrotic Strategy Utilizing the Secretome Released from Etanercept-Synthesizing Adipose-Derived Stem Cells
Abstract
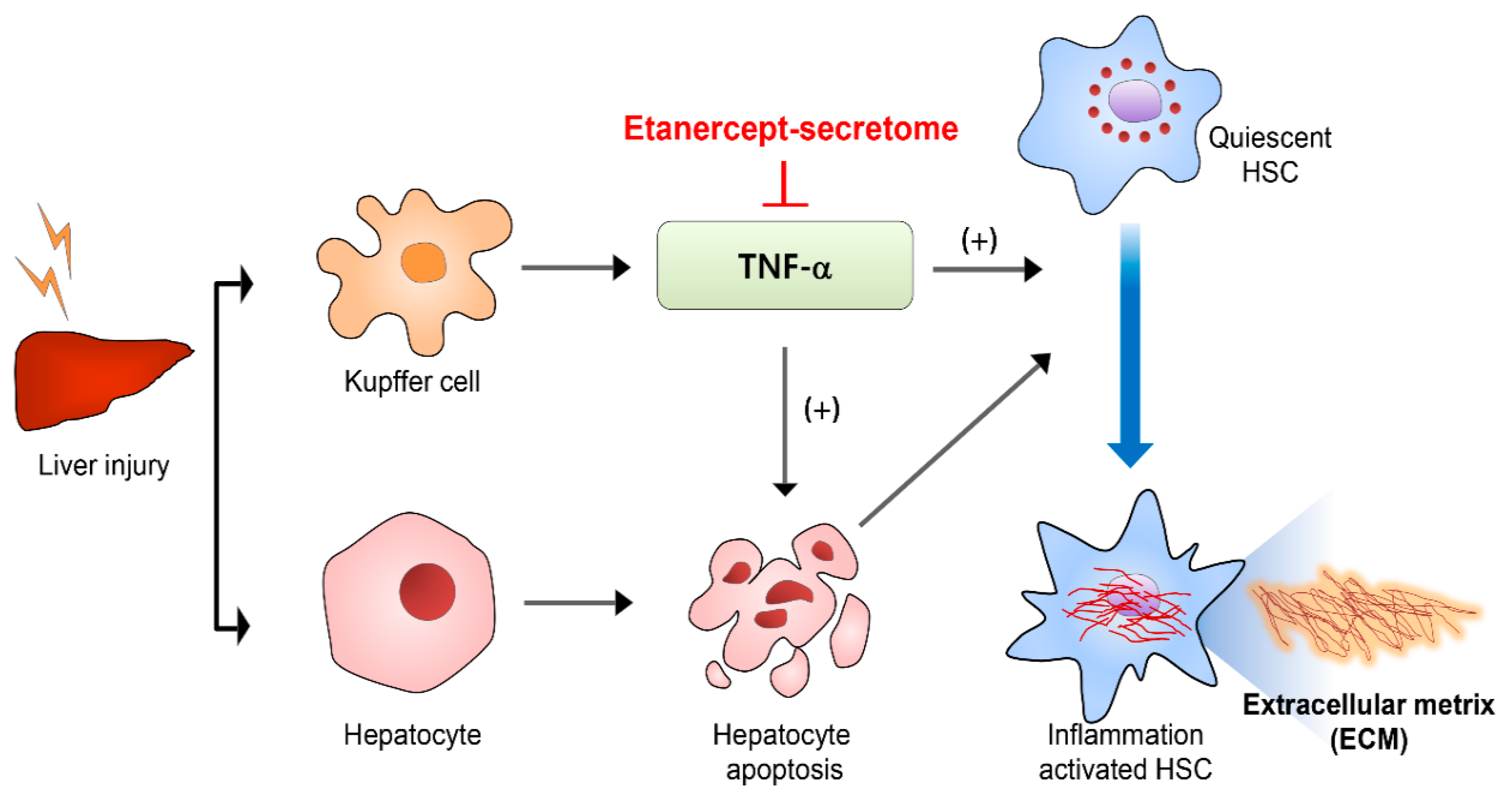
1. Introduction
2. Results
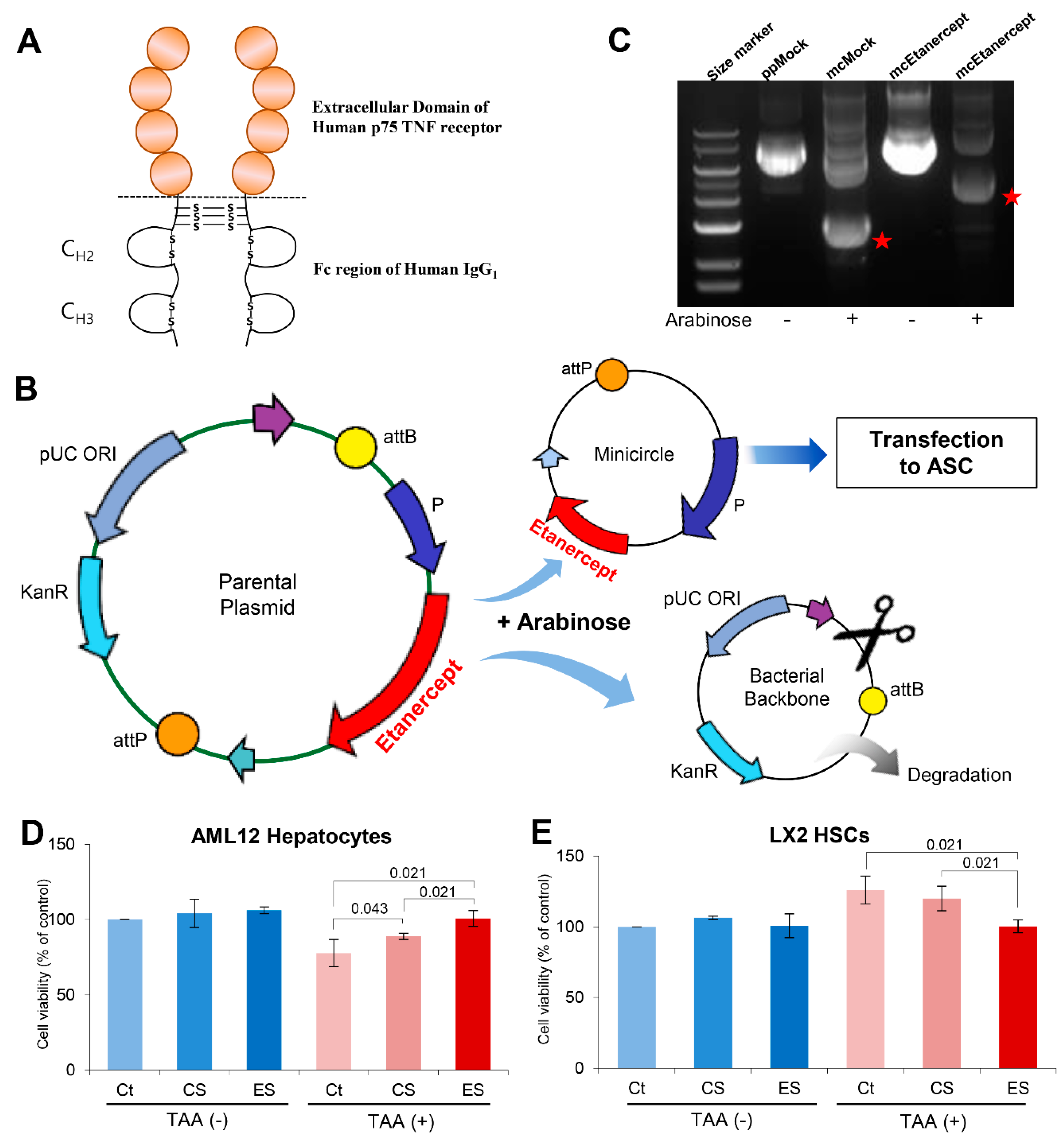
2.1. Attainment of Etanercept-Secretome from Etanercept-Synthesizing ASCs
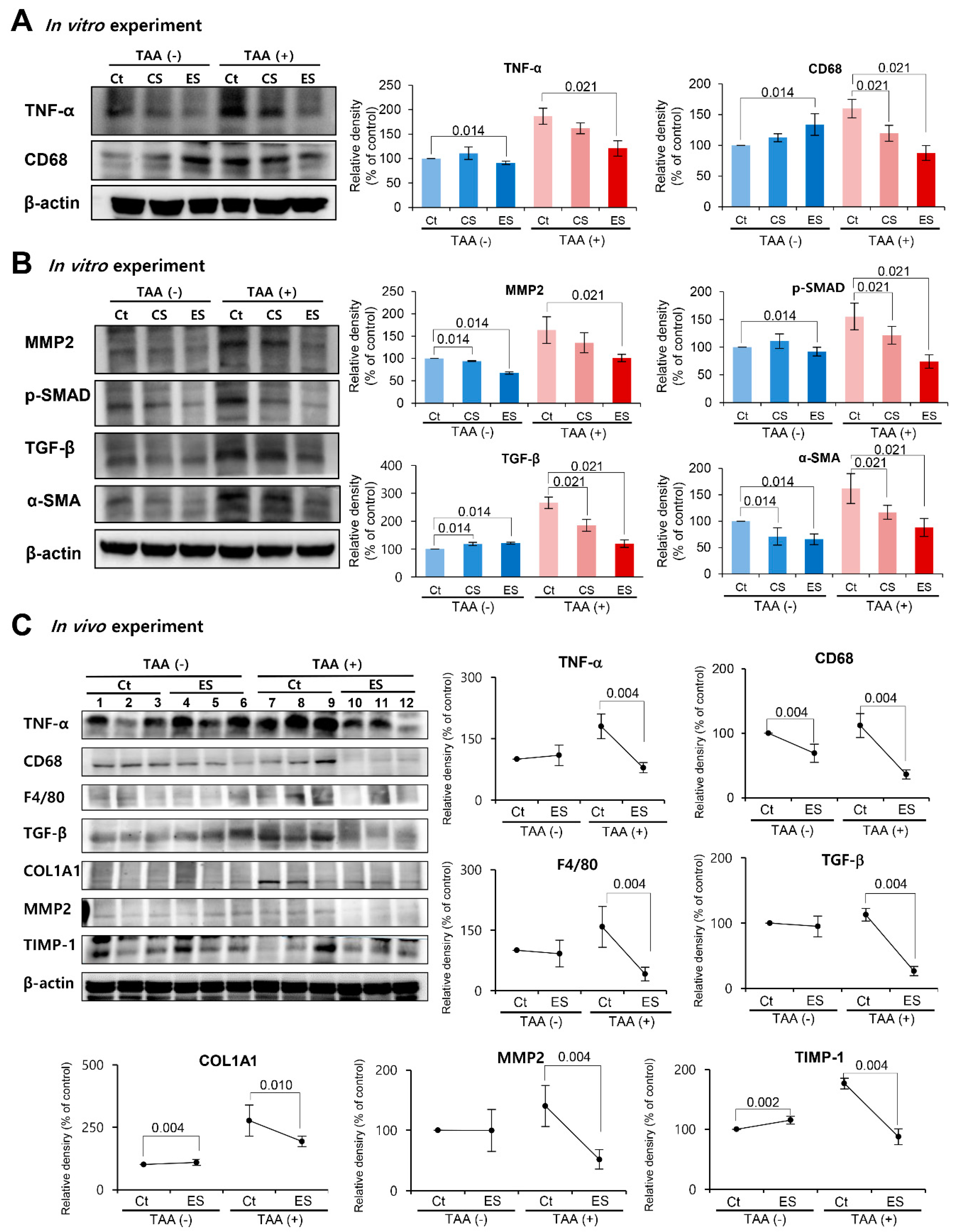
2.2. Effects of Etanercept-Secretome on the Protein Expression in HSCs in Vitro
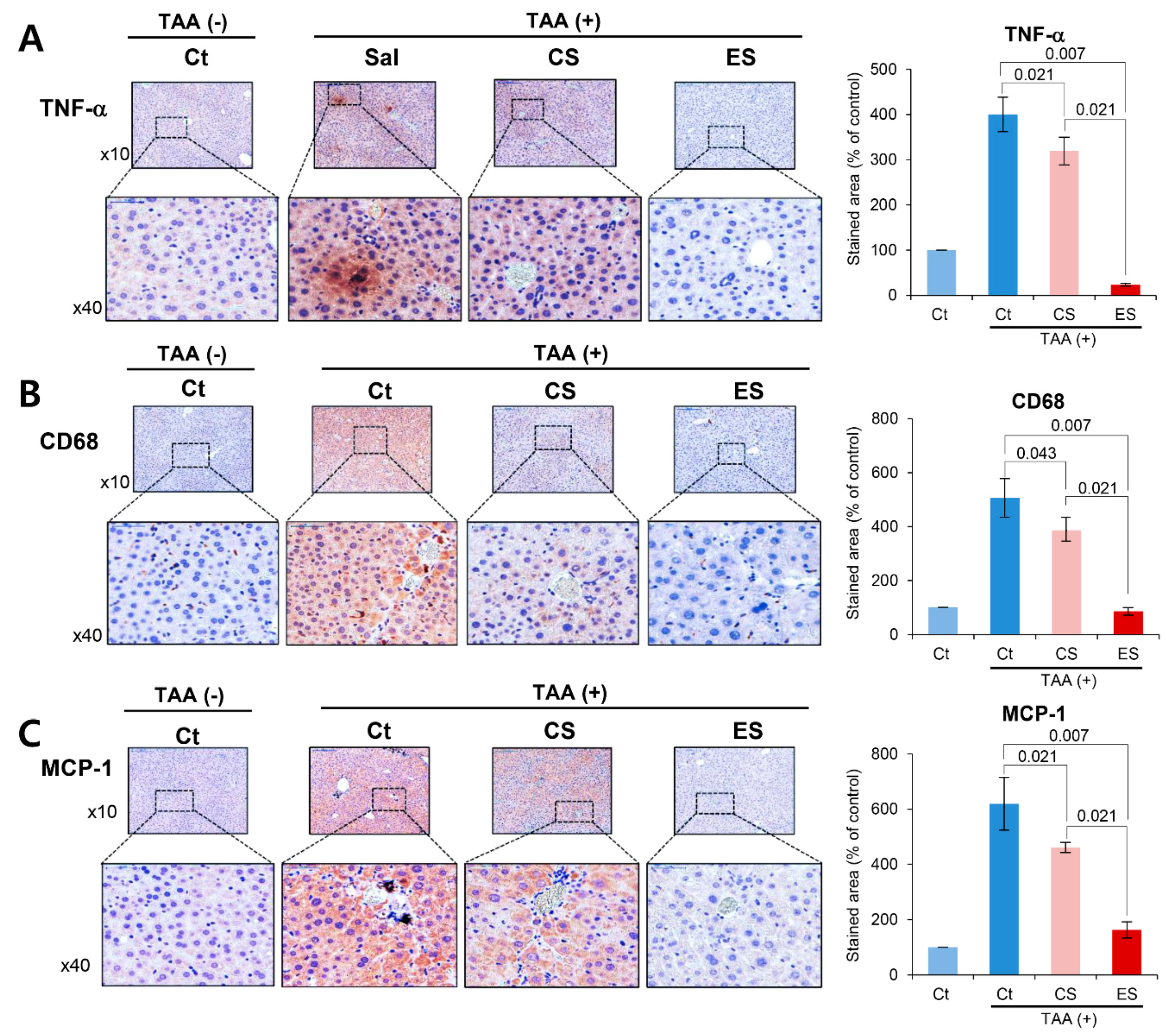
2.3. Effects of the Etanercept-Secretome in an In Vivo Liver Fibrosis Model
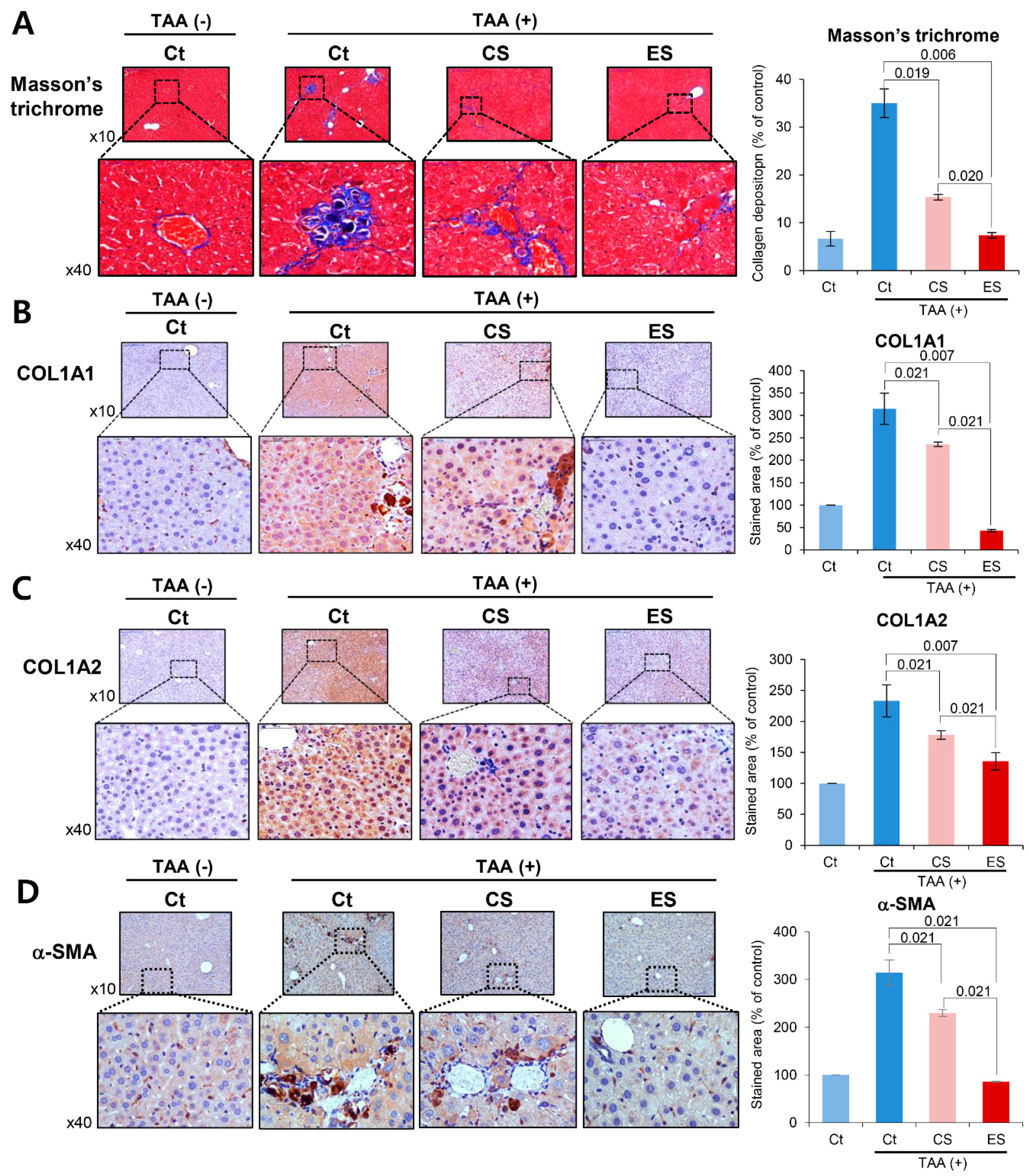
2.4. Histological Alternation of Liver Specimens after Injection of Etanercept-Secretome
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Generation of Etanercept-Synthesizing ASCs
4.3. Attainment of Secretome
4.4. Cell Proliferation Assay
4.5. Design of Animal Study
4.6. Western Blot Analysis
4.7. Serology Test and ELISA
4.8. Immunohistochemistry and Masson’s Trichrome Staining
4.9. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TNF-α | Tumor necrosis factor-α |
| ASCs | ASCs adipose-derived stem cells |
| HSCs | Hepatic stellate cells |
| BDL | Bile duct ligation |
References
- Schwabe, R.F.; Brenner, D.A. Mechanisms of Liver Injury. I. TNF-alpha-induced liver injury: Role of IKK, JNK, and ROS pathways. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G583–G589. [Google Scholar] [CrossRef] [PubMed]
- Gabele, E.; Froh, M.; Arteel, G.E.; Uesugi, T.; Hellerbrand, C.; Scholmerich, J.; Brenner, D.A.; Thurman, R.G.; Rippe, R.A. TNFalpha is required for cholestasis-induced liver fibrosis in the mouse. Biochem. Biophys. Res. Commun. 2009, 378, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Tarrats, N.; Moles, A.; Morales, A.; Garcia-Ruiz, C.; Fernandez-Checa, J.C.; Mari, M. Critical Role of Tumor Necrosis Factor Receptor 1, but not 2, in Hepatic Stellate Cell Proliferation, Extracellular Matrix Remodeling, and Liver Fibrogenesis. Hepatology 2011, 54, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Tomita, K.; Tamiya, G.; Ando, S.; Ohsumi, K.; Chiyo, T.; Mizutani, A.; Kitamura, N.; Toda, K.; Kaneko, T.; Horie, Y.; et al. Tumour necrosis factor alpha signalling through activation of Kupffer cells plays an essential role in liver fibrosis of non-alcoholic steatohepatitis in mice. Gut 2006, 55, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Hoshi, M.; Yasuda, I.; Saibara, T.; Moriwaki, H.; Kozawa, O. Tumor Necrosis Factor-alpha Promotes Cholestasis-Induced Liver Fibrosis in the Mouse through Tissue Inhibitor of Metalloproteinase-1 Production in Hepatic Stellate Cells. PLoS ONE 2013, 8, e65251. [Google Scholar] [CrossRef] [PubMed]
- Nagaki, M.; Naiki, T.; Brenner, D.A.; Osawa, Y.; Imose, M.; Hayashi, H.; Banno, Y.; Nakashima, S.; Moriwaki, H. Tumor necrosis factor alpha prevents tumor necrosis factor receptor-mediated mouse hepatocyte apoptosis, but not Fas-mediated apoptosis: Role of nuclear factor-kappa B. Hepatology 2000, 32, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Kirillova, I.; Peschon, J.J.; Fausto, N. Initiation of liver growth by tumor necrosis factor: Deficient liver regeneration in mice lacking type I tumor necrosis factor receptor. Proc. Natl. Acad. Sci. USA 1997, 94, 1441–1446. [Google Scholar] [CrossRef]
- HernandezMunoz, I.; DelaTorre, P.; SanchezAlcazar, J.A.; Garcia, I.; Santiago, E.; MunozYague, M.; SolisHerruzo, J.A. Tumor necrosis factor alpha inhibits collagen α1 (I) gene expression in rat hepatic stellate cells through a G protein. Gastroenterology 1997, 113, 625–640. [Google Scholar] [CrossRef]
- Houglum, K.; Buck, M.; Kim, D.J.; Chojkier, M. TNF-α inhibits liver collagen-α1 (I) gene expression through a tissue-specific regulatory region. Am. J. Physiol. Gastr. L 1998, 274, G840–G847. [Google Scholar] [CrossRef]
- Murdaca, G.; Spano, F.; Contatore, M.; Guastalla, A.; Magnani, O.; Puppo, F. Efficacy and safety of etanercept in chronic immune-mediated disease. Expert Opin. Drug Saf. 2014, 13, 649–661. [Google Scholar] [CrossRef]
- Park, N.; Rim, Y.A.; Jung, H.; Kim, J.; Yi, H.; Kim, Y.; Jang, Y.; Jung, S.M.; Lee, J.; Kwok, S.K.; et al. Etanercept-Synthesising Mesenchymal Stem Cells Efficiently Ameliorate Collagen-Induced Arthritis. Sci. Rep. 2017, 7, 39593. [Google Scholar] [CrossRef] [PubMed]
- Scott, L.J. Etanercept: A Review of Its Use in Autoimmune Inflammatory Diseases. Drugs 2014, 74, 1379–1410. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Kojika, E.; Hayashi, Y.; Kimura, M.; Nishikawa, K.; Yoshio, S.; Doi, H.; Kanto, T.; Kimura, K. Tumor necrosis factor-alpha-mediated hepatocyte apoptosis stimulates fibrosis in the steatotic liver in mice. Hepatol. Commun. 2018, 2, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Hamid, M.; Ahmed, R.R.; Moustafa, N.; Nady, R. The antifibrogenic effect of etanercept on development of liver cirrhosis induced by thioacetamide in rats. Ultrastruct. Pathol. 2017, 41, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.L.; Zheng, M.H.; Shi, K.Q.; Yang, T.; Chen, Y.P. A New Strategy for Treatment of Liver Fibrosis Letting MicroRNAs Do the Job. Biodrugs 2013, 27, 25–34. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, S. MicroRNAs in fibrosis: Opportunities and challenges. Arthritis Res. Ther. 2016, 18, 11. [Google Scholar] [CrossRef]
- Assmus, B.; Honold, J.; Schachinger, V.; Britten, M.B.; Fischer-Rasokat, U.; Lehmann, R.; Teupe, C.; Pistorius, K.; Martin, H.; Abolmaali, N.D.; et al. Transcoronary transplantation of progenitor cells after myocardial infarction. New Engl. J. Med. 2006, 355, 1222–1232. [Google Scholar] [CrossRef]
- Lee, T.J.; Bhang, S.H.; Yang, H.S.; La, W.G.; Yoon, H.H.; Shin, J.Y.; Seong, J.Y.; Shin, H.; Kim, B.S. Enhancement of long-term angiogenic efficacy of adipose stem cells by delivery of FGF2. Microvasc. Res. 2012, 84, 1–8. [Google Scholar] [CrossRef]
- Suzuki, K.; Murtuza, B.; Beauchamp, J.R.; Smolenski, R.T.; Varela-Carver, A.; Fukushima, S.; Coppen, S.R.; Partridge, T.A.; Yacoub, M.H. Dynamics and mediators of acute graft attrition after myoblast transplantation to the heart. Faseb. J. 2004, 18, 1153–1155. [Google Scholar] [CrossRef]
- Toma, C.; Pittenger, M.F.; Cahill, K.S.; Byrne, B.J.; Kessler, P.D. Human mesenchymal stem cells differentiate to a cardiomyocyte phenotype in the adult murine heart. Circulation 2002, 105, 93–98. [Google Scholar] [CrossRef]
- Lee, S.C.; Jeong, H.J.; Lee, S.K.; Kim, S.J. Lipopolysaccharide preconditioning of adipose-derived stem cells improves liver-regenerating activity of the secretome. Stem Cell Res. Ther. 2015, 6, 75. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, F.G.; Carvalho, M.M.; Panchalingam, K.M.; Rodrigues, A.J.; Mendes-Pinheiro, B.; Anjo, S.; Manadas, B.; Behie, L.A.; Sousa, N.; Salgado, A.J. Impact of the Secretome of Human Mesenchymal Stem Cells on Brain Structure and Animal Behavior in a Rat Model of Parkinson’s Disease. Stem Cells Transl. Med. 2017, 6, 634–646. [Google Scholar] [CrossRef] [PubMed]
- An, S.Y.; Han, J.; Lim, H.J.; Park, S.Y.; Kim, J.H.; Do, B.R.; Kim, J.H. Valproic acid promotes differentiation of hepatocyte-like cells from whole human umbilical cord-derived mesenchymal stem cells. Tissue Cell 2014, 46, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Elsharkawy, A.M.; Oakley, F.; Mann, D.A. The role and regulation of hepatic stellate cell apoptosis in reversal of liver fibrosis. Apoptosis 2005, 10, 927–939. [Google Scholar] [CrossRef]
- Hu, J.; Xu, Y.; Hao, J.; Wang, S.; Li, C.; Meng, S. MiR-122 in hepatic function and liver diseases. Protein Cell 2012, 3, 364–371. [Google Scholar] [CrossRef]
- Li, J.; Ghazwani, M.; Zhang, Y.; Lu, J.; Li, J.; Fan, J.; Gandhi, C.R.; Li, S. miR-122 regulates collagen production via targeting hepatic stellate cells and suppressing P4HA1 expression. J. Hepatol. 2013, 58, 522–528. [Google Scholar] [CrossRef]
- Swioklo, S.; Constantinescu, A.; Connon, C.J. Alginate-Encapsulation for the Improved Hypothermic Preservation of Human Adipose-Derived Stem Cells. Stem Cells Transl. Med. 2016, 5, 339–349. [Google Scholar] [CrossRef]
- Szabo, G.; Bala, S. MicroRNAs in liver disease. Nat. Rev. Gastroenterol. Hepatol. 2013, 10, 542–552. [Google Scholar] [CrossRef]
- Zeng, C.; Wang, Y.L.; Xie, C.; Sang, Y.; Li, T.J.; Zhang, M.; Wang, R.; Zhang, Q.; Zheng, L.; Zhuang, S.M. Identification of a novel TGF-beta-miR-122-fibronectin 1/serum response factor signaling cascade and its implication in hepatic fibrogenesis. Oncotarget 2015, 6, 12224–12233. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef]
- Hessien, M.H.; El-Sharkawi, I.M.; El-Barbary, A.A.; El-Beltagy, D.M.; Snyder, N. Non-invasive index of liver fibrosis induced by alcohol, thioacetamide and schistosomal infection in mice. BMC Gastroenterol. 2010, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.S.; Wanibuchi, H.; Morimura, K.; Wongpoomchai, R.; Chusiri, Y.; Gonzalez, F.J.; Fukushima, S. Role of CYP2E1 in thioacetamide-induced mouse hepatotoxicity. Toxicol. Appl. Pharm. 2008, 228, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Kodama, T.; Takehara, T.; Hikita, H.; Shimizu, S.; Shigekawa, M.; Tsunematsu, H.; Li, W.; Miyagi, T.; Hosui, A.; Tatsumi, T.; et al. Increases in p53 expression induce CTGF synthesis by mouse and human hepatocytes and result in liver fibrosis in mice. J. Clin. Invest. 2011, 121, 3343–3356. [Google Scholar] [CrossRef] [PubMed]
- Palacios, R.S.; Roderfeld, M.; Hemmann, S.; Rath, T.; Atanasova, S.; Tschuschner, A.; Gressner, O.A.; Weiskirchen, R.; Graf, J.; Roeb, E. Activation of hepatic stellate cells is associated with cytokine expression in thioacetamide-induced hepatic fibrosis in mice. Lab. Invest. 2008, 88, 1192–1203. [Google Scholar] [CrossRef] [PubMed]
- Wallace, M.C.; Hamesch, K.; Lunova, M.; Kim, Y.; Weiskirchen, R.; Strnad, P.; Friedman, S.L. Standard Operating Procedures in Experimental Liver Research: Thioacetamide model in mice and rats. Lab. Anim. 2015, 49, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.B.; Chuang, H.R.; Yang, L.C.; Lin, W.C. A Standardized Aqueous Extract of Anoectochilus formosanus Ameliorated Thioacetamide-Induced Liver Fibrosis in Mice: The Role of Kupffer Cells. Biosci. Biotech. Bioch. 2010, 74, 781–787. [Google Scholar] [CrossRef]
- Huang, B.; Cheng, X.X.; Wang, H.F.; Huang, W.J.; Hu, Z.L.G.; Wang, D.; Zhang, K.; Zhang, H.; Xue, Z.Y.; Da, Y.R.; et al. Mesenchymal stem cells and their secreted molecules predominantly ameliorate fulminant hepatic failure and chronic liver fibrosis in mice respectively. J. Transl. Med. 2016, 14. [Google Scholar] [CrossRef]
- Kim, Y.H.; Cho, K.A.; Park, M.; Kim, H.S.; Park, J.W.; Woo, S.Y.; Ryu, K.H. Conditioned Medium from Tonsil-Derived Mesenchymal Stem Cells Relieves CCl4-Induced Liver Fibrosis in Mice. Tissue Eng. Regen. Med. 2019, 16, 51–58. [Google Scholar] [CrossRef]
- Kojima, Y. Mesenchymal Stem Cells Cultured in Hypoxic Conditions Had a Greater Therapeutic Effect on Mice with Liver Cirrhosis through Prostaglandin E2 and miR210 Production Compared to Those Cultured under Normal Oxygen Conditions. Hepatology 2018, 68, 640a. [Google Scholar]
- Mehrabani, D.; Khajehahmadi, Z.; Tajik, P.; Tamadon, A.; Rahmanifar, F.; Ashraf, M.; Tanideh, N.; Zare, S. Regenerative Effect of Bone Marrow-derived Mesenchymal Stem Cells in Thioacetamide-induced Liver Fibrosis of Rats. Arch. Razi. Inst. 2019, 74, 279–286. [Google Scholar]
- Truong, N.H.; Nguyen, N.H.; Le, T.V.; Vu, N.B.; Huynh, N.; Nguyen, T.V.; Le, H.M.; Phan, N.K.; Pham, P.V. Comparison of the Treatment Efficiency of Bone Marrow-Derived Mesenchymal Stem Cell Transplantation via Tail and Portal Veins in CCl4-Induced Mouse Liver Fibrosis. Stem Cells Int. 2016, 2016, 5720413. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Tsuchiya, A.; Seino, S.; Kawata, Y.; Kojima, Y.; Ikarashi, S.; Starkey Lewis, P.J.; Lu, W.Y.; Kikuta, J.; Kawai, H.; et al. Mesenchymal Stem Cells and Induced Bone Marrow-Derived Macrophages Synergistically Improve Liver Fibrosis in Mice. Stem Cells Transl. Med. 2019, 8, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Canbay, A.; Feldstein, A.E.; Higuchi, H.; Werneburg, N.; Grambihler, A.; Bronk, S.F.; Gores, G.J. Kupffer cell engulfment of apoptotic bodies stimulates death ligand and cytokine expression. Hepatology 2003, 38, 1188–1198. [Google Scholar] [CrossRef] [PubMed]
- Canbay, A.; Taimr, P.; Torok, N.; Higuchi, H.; Friedman, S.; Gores, G.J. Apoptotic body engulfment by a human stellate cell line is profibrogenic. Lab. Invest. 2003, 83, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Canbay, A.; Friedman, S.; Gores, G.J. Apoptosis: The nexus of liver injury and fibrosis. Hepatology 2004, 39, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Elpek, G.O. Cellular and molecular mechanisms in the pathogenesis of liver fibrosis: An update. World J. Gastroenterol. 2014, 20, 7260–7276. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, J.H.; Kim, O.-H.; Lee, S.C.; Kim, K.-H.; Park, J.H.; Lee, J.I.; Lee, K.H.; Hong, H.-E.; Seo, H.; Choi, H.J.; et al. A Novel Hepatic Anti-Fibrotic Strategy Utilizing the Secretome Released from Etanercept-Synthesizing Adipose-Derived Stem Cells. Int. J. Mol. Sci. 2019, 20, 6302. https://doi.org/10.3390/ijms20246302
Han JH, Kim O-H, Lee SC, Kim K-H, Park JH, Lee JI, Lee KH, Hong H-E, Seo H, Choi HJ, et al. A Novel Hepatic Anti-Fibrotic Strategy Utilizing the Secretome Released from Etanercept-Synthesizing Adipose-Derived Stem Cells. International Journal of Molecular Sciences. 2019; 20(24):6302. https://doi.org/10.3390/ijms20246302
Chicago/Turabian StyleHan, Jae Hyun, Ok-Hee Kim, Sang Chul Lee, Kee-Hwan Kim, Jung Hyun Park, Jae Im Lee, Kyung Hee Lee, Ha-Eun Hong, Haeyeon Seo, Ho Joong Choi, and et al. 2019. "A Novel Hepatic Anti-Fibrotic Strategy Utilizing the Secretome Released from Etanercept-Synthesizing Adipose-Derived Stem Cells" International Journal of Molecular Sciences 20, no. 24: 6302. https://doi.org/10.3390/ijms20246302
APA StyleHan, J. H., Kim, O.-H., Lee, S. C., Kim, K.-H., Park, J. H., Lee, J. I., Lee, K. H., Hong, H.-E., Seo, H., Choi, H. J., Ju, J. H., & Kim, S.-J. (2019). A Novel Hepatic Anti-Fibrotic Strategy Utilizing the Secretome Released from Etanercept-Synthesizing Adipose-Derived Stem Cells. International Journal of Molecular Sciences, 20(24), 6302. https://doi.org/10.3390/ijms20246302




