Benzo[b]tellurophenes as a Potential Histone H3 Lysine 9 Demethylase (KDM4) Inhibitor
Abstract
:1. Introduction
2. Results and Discussion
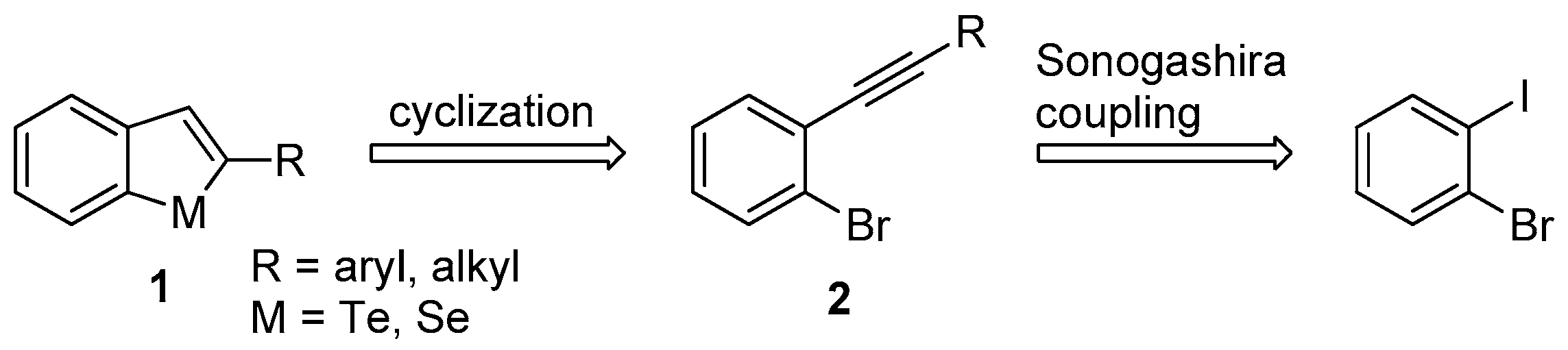
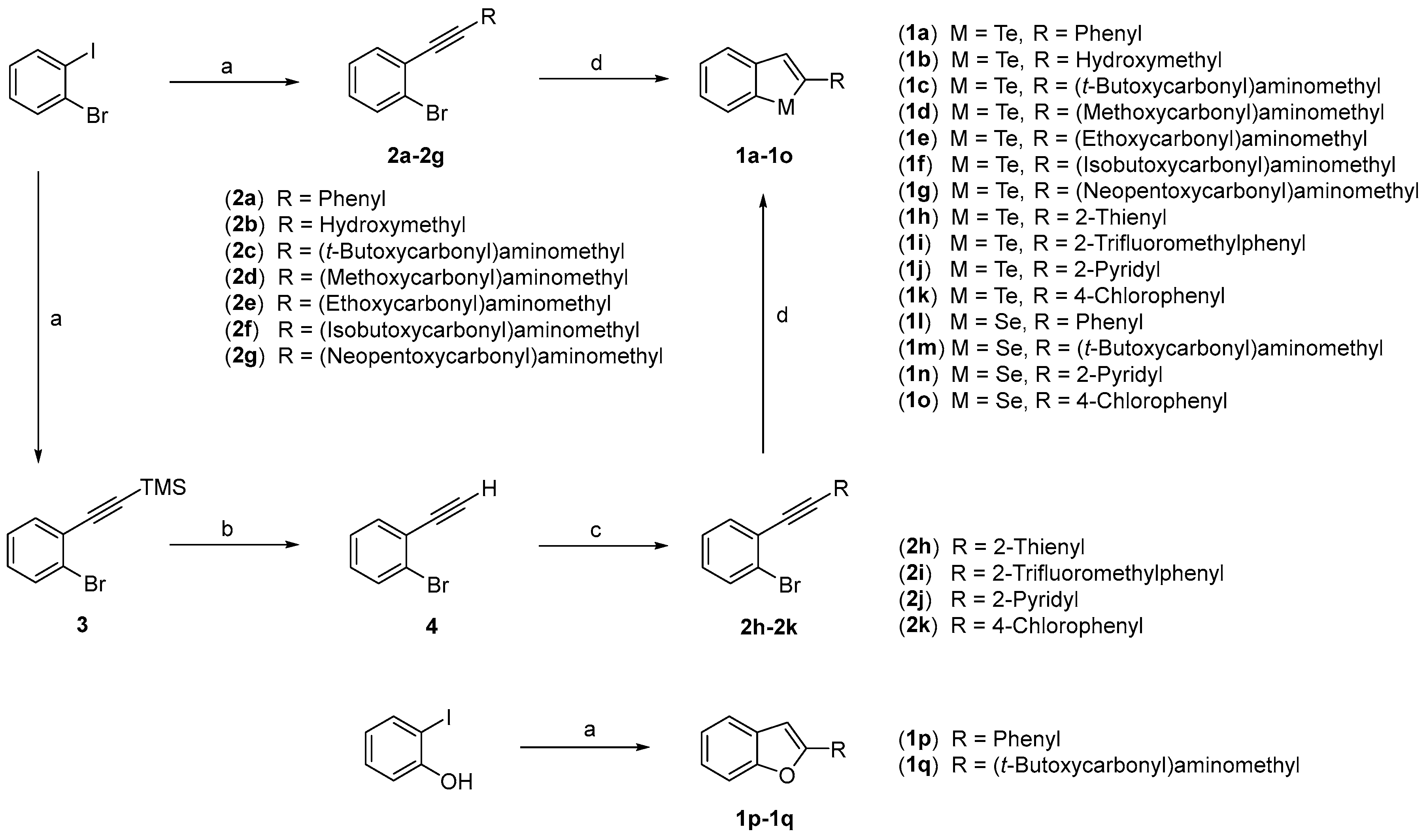
2.1. Chemistry
2.2. Biological Evaluation
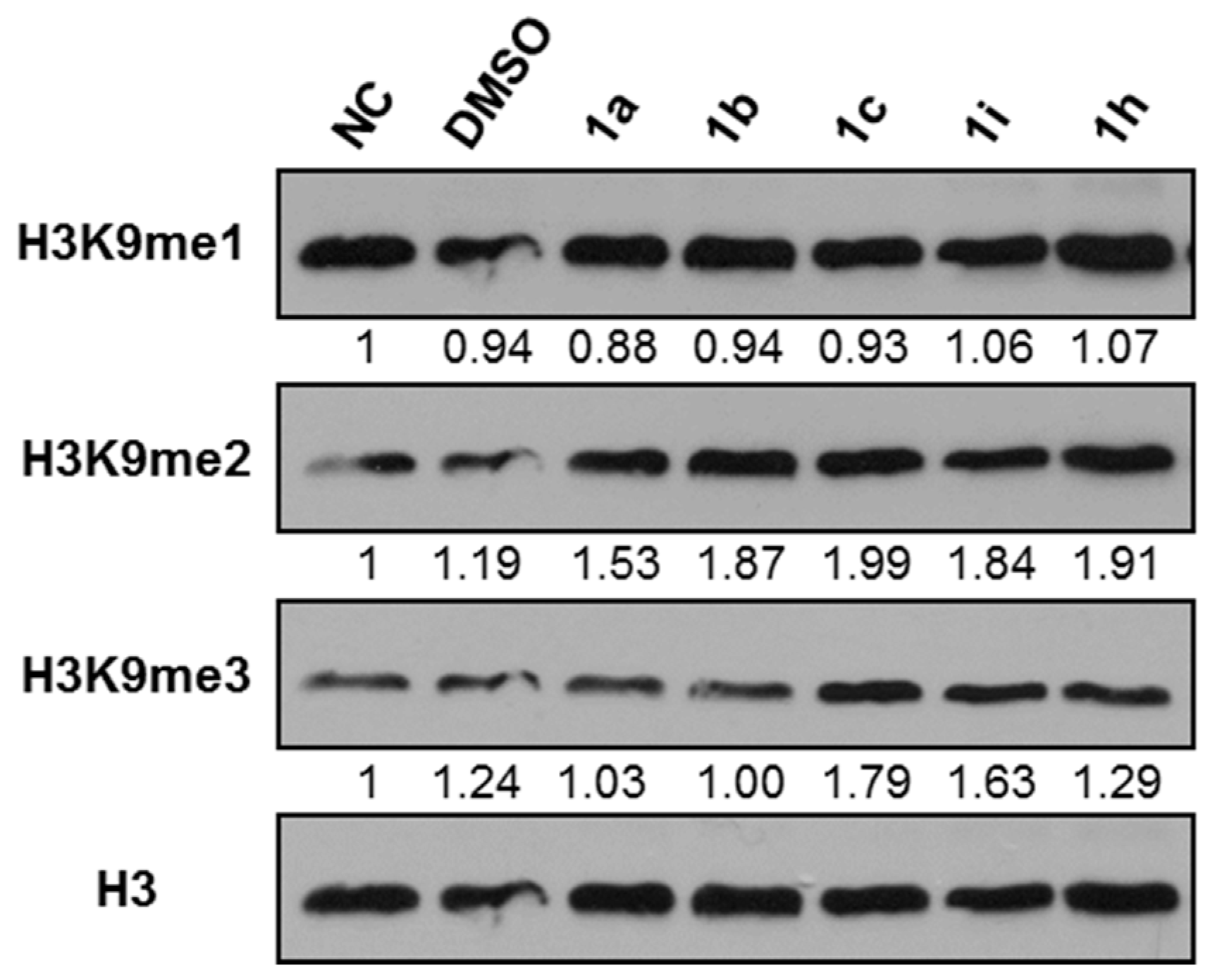
2.2.1. Effects of Compounds on Histone Methylation in cervical cancer HeLa Cells
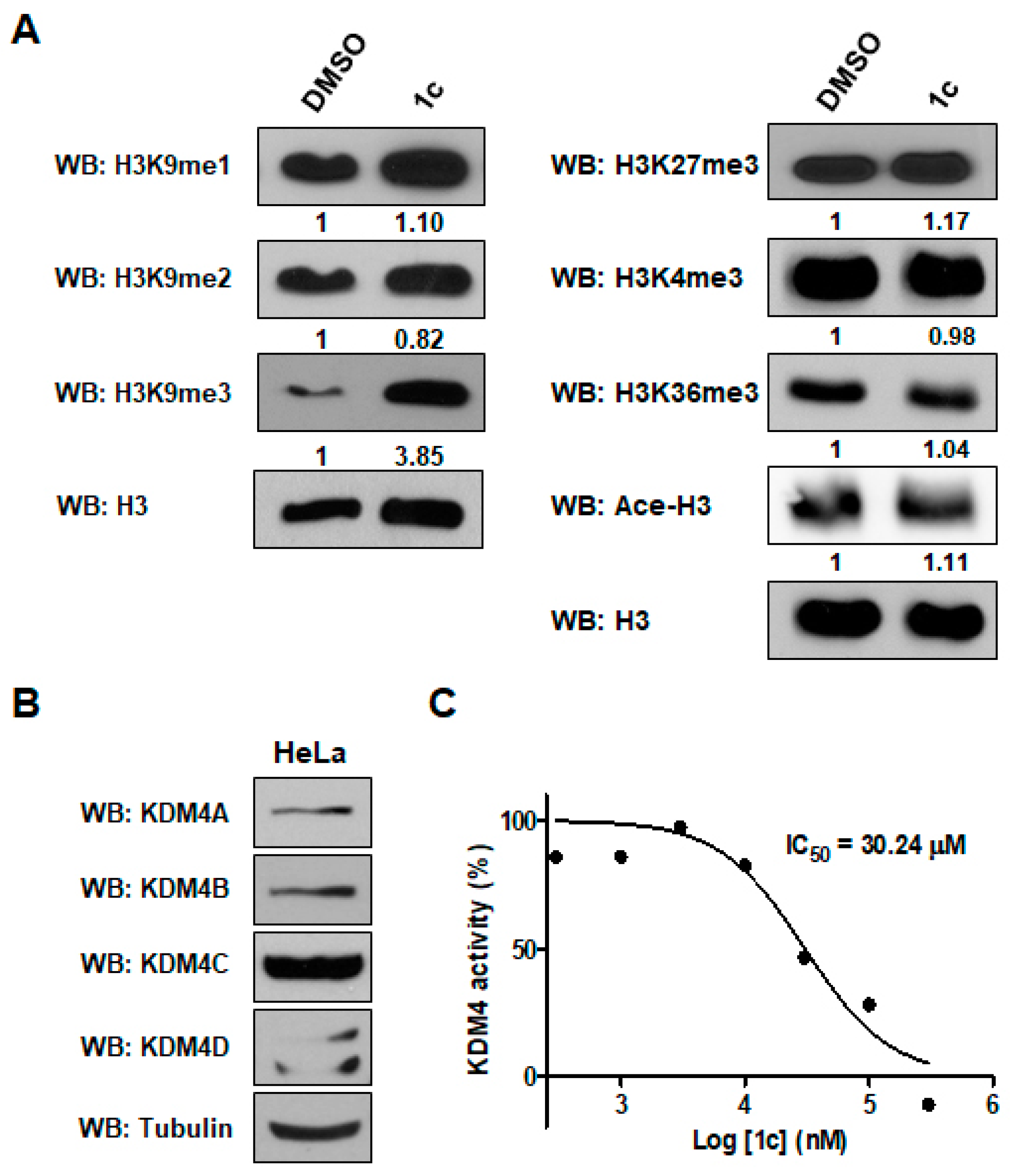
2.2.2. KDM4 Inhibitory Assays
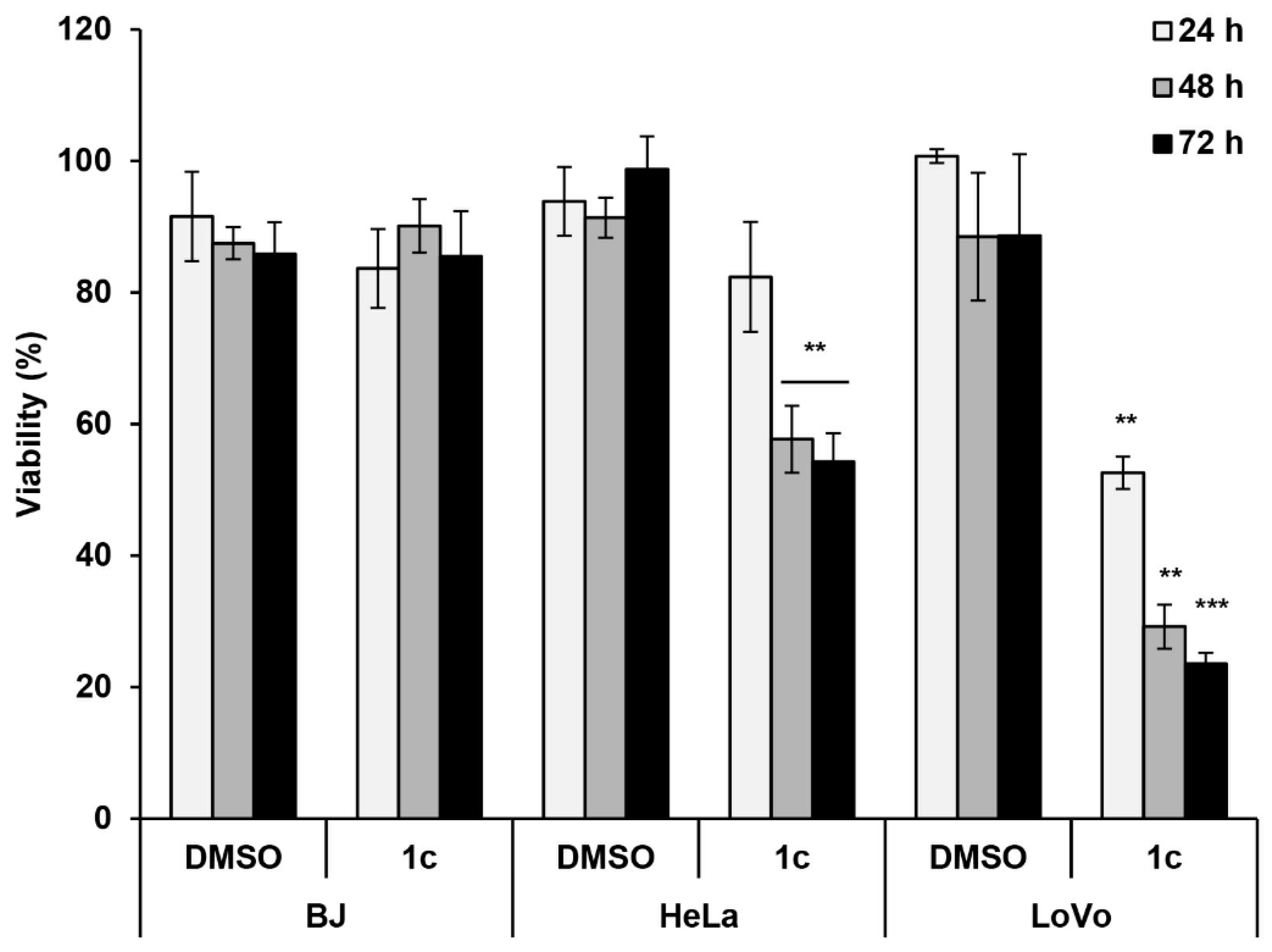
2.2.3. Effects of 1c on Cytotoxicity of Human Cervical HeLa and Colon LoVo Cancer Cells
3. Materials and Methods
3.1. Chemistry
3.1.1. General Procedure for the Preparation of Compounds 2a, 2b, and 3
3.1.2. General Procedure for the Preparation of Compound 2c–2g
3.1.3. Procedure for the Preparation of Compound 4
3.1.4. General Procedure for the Preparation of Compound 2h–2k
3.1.5. General Procedure for the Preparation of Benzo[b]tellurophenes and Benzo[b]selenophenes (1a−1o)
3.1.6. Procedure for the Preparation of Benzo[b]furans (1p, 1q)
3.2. Biology
3.2.1. Acid Extraction of Histones
3.2.2. Western Blot Analysis
3.2.3. Histone Demethylase Assay
3.2.4. MTT Assay
3.2.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Radadiya, A.; Shah, A. Bioactive benzofuran derivatives: An insight on lead developments, radioligands and advances of the last decade. Eur. J. Med. Chem. 2015, 97, 356–376. [Google Scholar] [CrossRef] [PubMed]
- Keri, R.S.; Chand, K.; Budagumpi, S.; Balappa Somappa, S.; Patil, S.A.; Nagaraja, B.M. An overview of benzo[b]thiophene-based medicinal chemistry. Eur. J. Med. Chem. 2017, 138, 1002–1033. [Google Scholar] [CrossRef] [PubMed]
- Majid, M.H.; Vahideh, Z. Recent Advances in the Synthesis of Biologically Active Compounds Containing Benzo[b]Furans as a Framework. Curr. Org. Synth. 2016, 13, 780–833. [Google Scholar]
- Khanam, H.; Shamsuzzaman. Bioactive Benzofuran derivatives: A review. Eur. J. Med. Chem. 2015, 97, 483–504. [Google Scholar] [CrossRef]
- Tan, H.W.; Mo, H.-Y.; Lau, A.T.Y.; Xu, Y.-M. Selenium Species: Current Status and Potentials in Cancer Prevention and Therapy. Int. J. Mol. Sci. 2018, 20, 75. [Google Scholar] [CrossRef]
- Müller, A.; Cadenas, E.; Graf, P.; Sies, H. A novel biologically active seleno-organic compound—1: Glutathione peroxidase-like activity in vitro and antioxidant capacity of PZ 51 (Ebselen). Biochem. Pharmacol. 1984, 33, 3235–3239. [Google Scholar] [CrossRef]
- Choi, Y.-S.; Kim, Y.-J.; Lee, J.; Lee, J.; Jeong, J.-H. Synthesis and Evaluation of Selenoflavones That Have Potential Neuroprotective Effects. Heterocycles 2014, 89, 2794–2805. [Google Scholar]
- Andersson, C.-M.; Hallberg, A.; Brattsand, R.; Cotgreave, I.A.; Engman, L.; Persson, J. Glutathione peroxidase-like activity of diaryl tellurides. Bioorganic Med. Chem. Lett. 1993, 3, 2553–2558. [Google Scholar] [CrossRef]
- Sredni, B.; Albeck, M.; Kazimirsky, G.; Shalit, F. The immunomodulator AS101 administered orally as a chemoprotective and radioprotective agent. Int. J. Immunopharmacol. 1992, 14, 613–619. [Google Scholar] [CrossRef]
- Sredni, B. Immunomodulating tellurium compounds as anti-cancer agents. Semin. Cancer Biol. 2012, 22, 60–69. [Google Scholar] [CrossRef]
- Caracelli, I.; Vega-Teijido, M.; Zukerman-Schpector, J.; Cezari, M.H.S.; Lopes, J.G.S.; Juliano, L.; Santos, P.S.; Comasseto, J.V.; Cunha, R.L.O.R.; Tiekink, E.R.T. A tellurium-based cathepsin B inhibitor: Molecular structure, modelling, molecular docking and biological evaluation. J. Mol. Struct. 2012, 1013, 11–18. [Google Scholar] [CrossRef]
- Angeli, A.; Tanini, D.; Capperucci, A.; Supuran, C.T. First evaluation of organotellurium derivatives as carbonic anhydrase I, II, IV, VII and IX inhibitors. Bioorganic Chem. 2018, 76, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.P.; Poon, J.-F.; Engman, L. Catalytic Antioxidants: Regenerable Tellurium Analogues of Vitamin E. Org. Lett. 2013, 15, 6274–6277. [Google Scholar] [CrossRef] [PubMed]
- Sekirnik, R.; Rose, N.R.; Thalhammer, A.; Seden, P.T.; Mecinovic, J.; Schofield, C.J. Inhibition of the histone lysine demethylase JMJD2A by ejection of structural Zn(II). Chem. Commun. 2009, 42, 6376–6378. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Lan, F.; Matson, C.; Mulligan, P.; Whetstine, J.R.; Cole, P.A.; Casero, R.A.; Shi, Y. Histone Demethylation Mediated by the Nuclear Amine Oxidase Homolog LSD1. Cell 2004, 119, 941–953. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Shilatifard, A. Histone Demethylation by Hydroxylation: Chemistry in Action. ACS Chem. Biol. 2006, 1, 75–81. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, Z.; Yung, S.; Lu, Q. Epigenetic dynamics in immunity and autoimmunity. Int. J. Biochem. Cell Biol. 2015, 67, 65–74. [Google Scholar] [CrossRef]
- Young, L.C.; Hendzel, M.J. The oncogenic potential of Jumonji D2 (JMJD2/KDM4) histone demethylase overexpression. Biochem. Cell Biol. 2013, 91, 369–377. [Google Scholar] [CrossRef]
- Berry, W.L.; Shin, S.; Lightfoot, S.A.; Janknecht, R. Oncogenic features of the JMJD2A histone demethylase in breast cancer. Int. J. Oncol. 2012, 41, 1701–1706. [Google Scholar]
- Ishimura, A.; Terashima, M.; Kimura, H.; Akagi, K.; Suzuki, Y.; Sugano, S.; Suzuki, T. Jmjd2c histone demethylase enhances the expression of Mdm2 oncogene. Biochem. Biophys. Res. Commun. 2009, 389, 366–371. [Google Scholar] [CrossRef]
- Luo, W.; Chang, R.; Zhong, J.; Pandey, A.; Semenza, G.L. Histone demethylase JMJD2C is a coactivator for hypoxia-inducible factor 1 that is required for breast cancer progression. Proc. Natl. Acad. Sci. USA 2012, 109, E3367–E3376. [Google Scholar] [CrossRef] [PubMed]
- Westaway, S.M.; Preston, A.G.; Barker, M.D.; Brown, F.; Brown, J.A.; Campbell, M.; Chung, C.W.; Drewes, G.; Eagle, R.; Garton, N.; et al. Cell Penetrant Inhibitors of the KDM4 and KDM5 Families of Histone Lysine Demethylases. 2. Pyrido[3,4-d]pyrimidin-4(3H)-one Derivatives. J. Med. Chem. 2016, 59, 1370–1387. [Google Scholar] [CrossRef] [PubMed]
- Westaway, S.M.; Preston, A.G.; Barker, M.D.; Brown, F.; Brown, J.A.; Campbell, M.; Chung, C.W.; Diallo, H.; Douault, C.; Drewes, G.; et al. Cell Penetrant Inhibitors of the KDM4 and KDM5 Families of Histone Lysine Demethylases. 1. 3-Amino-4-pyridine Carboxylate Derivatives. J. Med. Chem. 2016, 59, 1357–1369. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.K.; Bonaldi, T.; Cuomo, A.; Del Rosario, J.R.; Hosfield, D.J.; Kanouni, T.; Kao, S.C.; Lai, C.; Lobo, N.A.; Matuszkiewicz, J.; et al. Design of KDM4 Inhibitors with Antiproliferative Effects in Cancer Models. ACS Med. Chem. Lett. 2017, 8, 869–874. [Google Scholar] [CrossRef]
- Carter, D.M.; Specker, E.; Przygodda, J.; Neuenschwander, M.; von Kries, J.P.; Heinemann, U.; Nazare, M.; Gohlke, U. Identification of a Novel Benzimidazole Pyrazolone Scaffold That Inhibits KDM4 Lysine Demethylases and Reduces Proliferation of Prostate Cancer Cells. Slas Discov. Adv. Life Sci. R D 2017, 22, 801–812. [Google Scholar] [CrossRef]
- Sashida, H.; Sadamori, K.; Tsuchiya, T. A Convenient One-Pot Preparation of Benzo[b]-tellurophenes, -selenophenes, and -thiophenes from o-Bromoethynylbenzenes. Synth. Commun. 1998, 28, 713–727. [Google Scholar] [CrossRef]
- Okuma, K.; Yahata, S.; Nagahora, N.; Shioji, K. Reaction of Acetophenone Hydrazones with TeCl4: Novel Formation of 2,5-Diaryltellurophenes and Benzotellurophenes. Chem. Lett. 2017, 46, 405–407. [Google Scholar] [CrossRef]
- Shechter, D.; Dormann, H.L.; Allis, C.D.; Hake, S.B. Extraction, purification and analysis of histones. Nat. Protoc. 2007, 2, 1445–1457. [Google Scholar] [CrossRef]
| Cpds | Structure | Activity * | Cpds | Structure | Activity * |
|---|---|---|---|---|---|
| 1a | 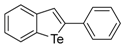 | 1.03 | 1j | 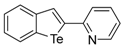 | 1.06 |
| 1b |  | 1.00 | 1k | 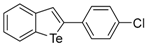 | 0.98 |
| 1c | 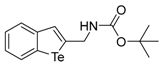 | 1.79 | 1l | 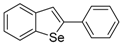 | 0.97 |
| 1d | 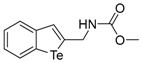 | 1.06 | 1m | 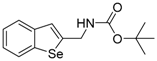 | 0.90 |
| 1e | 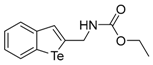 | 1.03 | 1n |  | 1.04 |
| 1f | 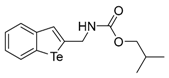 | 1.30 | 1o | 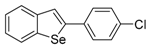 | 0.86 |
| 1g |  | 0.88 | 1p | 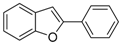 | 0.96 |
| 1h | 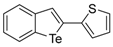 | 1.29 | 1q | 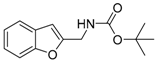 | 1.13 |
| 1i | 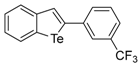 | 1.63 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.-J.; Lee, D.H.; Choi, Y.-S.; Jeong, J.-H.; Kwon, S.H. Benzo[b]tellurophenes as a Potential Histone H3 Lysine 9 Demethylase (KDM4) Inhibitor. Int. J. Mol. Sci. 2019, 20, 5908. https://doi.org/10.3390/ijms20235908
Kim Y-J, Lee DH, Choi Y-S, Jeong J-H, Kwon SH. Benzo[b]tellurophenes as a Potential Histone H3 Lysine 9 Demethylase (KDM4) Inhibitor. International Journal of Molecular Sciences. 2019; 20(23):5908. https://doi.org/10.3390/ijms20235908
Chicago/Turabian StyleKim, Yoon-Jung, Dong Hoon Lee, Yong-Sung Choi, Jin-Hyun Jeong, and So Hee Kwon. 2019. "Benzo[b]tellurophenes as a Potential Histone H3 Lysine 9 Demethylase (KDM4) Inhibitor" International Journal of Molecular Sciences 20, no. 23: 5908. https://doi.org/10.3390/ijms20235908
APA StyleKim, Y.-J., Lee, D. H., Choi, Y.-S., Jeong, J.-H., & Kwon, S. H. (2019). Benzo[b]tellurophenes as a Potential Histone H3 Lysine 9 Demethylase (KDM4) Inhibitor. International Journal of Molecular Sciences, 20(23), 5908. https://doi.org/10.3390/ijms20235908





