Impact of Nitrogen Nutrition on Cannabis sativa: An Update on the Current Knowledge and Future Prospects
Abstract
1. Introduction
2. Physiological Effects of N Availability in Hemp
3. N Nutrition and Impact on the Plant Secondary Metabolism
4. Transcriptomic Datasets Identify Genes Involved in the Regulation of N Metabolism and Responsive to Abiotic Stresses in C. sativa
5. Breeding Strategies in Hemp: Nutrient Management and Synthesis of Secondary Metabolites
6. Manipulation of the Cannabinoid Biosynthetic Pathway and Their Relation with N
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Docimo, T.; Caruso, I.; Ponzoni, E.; Mattana, M.; Galasso, I. Molecular characterization of edestin gene family in Cannabis sativa L. Plant Physiol. Biochem. 2014, 84, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.; Struik, P.C.; Amaducci, S.; Stomph, T.; Yin, X. Hemp (Cannabis sativa L.) leaf photosynthesis in relation to nitrogen content and temperature: Implications for hemp as a bio-economically sustainable crop. GCB Bioenergy 2017, 9, 1573–1587. [Google Scholar] [CrossRef]
- Schluttenhofer, C.; Yuan, L. Challenges towards revitalizing hemp: A multifaceted crop. Trends Plants Sci. 2017, 22, 917–929. [Google Scholar] [CrossRef]
- Schultes, R.E.; Klein, W.M.; Plowman, T.; Lockwood, T.E. Cannabis: An example of taxonomic neglect. Bot. Mus. Leafl. Harv. Univ. 1974, 23, 337–367. [Google Scholar]
- Johnson, R. Hemp as an Agricultural Commodity; Congressional Research Service: Washington, DC, USA, 2014. [Google Scholar]
- Andre, C.M.; Hausman, J.F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Dingha, B.; Sandle, L.; Bhowmik, A.; Akotsen-Mensah, C.; Jackai, L.; Gibson, K.; Turco, R. Industrial hemp knowledge and interest among North Carolina organic farmers in the United States. Sustainability 2019, 11, 2691. [Google Scholar] [CrossRef]
- Van Bakel, H.; Stout, J.M.; Cote, A.G.; Tallon, C.M.; Sharpe, A.G.; Hughes, T.R.; Page, J.E. The draft genome and transcriptome of Cannabis sativa. Genome Biol. 2011, 12, R102. [Google Scholar] [CrossRef]
- Liu, J.; Qiao, Q.; Cheng, X.; Du, G.; Deng, G.; Zhao, M.; Liu, F. Transcriptome differences between fiber-type and seed-type Cannabis sativa variety exposed to salinity. Physiol. Mol. Biol. Plants 2016, 22, 429–443. [Google Scholar] [CrossRef]
- Masclaux-Daubresse, C.; Daniel-Vedele, F.; Dechorgnat, J.; Chardon, F.; Gaufichon, L.; Suzuki, A. Nitrogen uptake, assimilation and remobilization in plants: Challenges for sustainable and productive agriculture. Ann. Bot. 2010, 105, 1141–1157. [Google Scholar] [CrossRef]
- Curci, P.L.; Cigliano, R.A.; Zuluaga, D.L.; Janni, M.; Sanseverino, W.; Sonnante, G. Transcriptomic response of durum wheat to nitrogen starvation. Sci. Rep. 2017, 26, 1176. [Google Scholar] [CrossRef]
- Bernstein, N.; Gorelick, J.; Zerahia, R.; Koch, S. Impact of N, P, K, and humic acid supplementation on the chemical profile of medical cannabis (Cannabis sativa L.). Front. Plant Sci. 2019, 10, 736. [Google Scholar] [CrossRef] [PubMed]
- Struik, P.C.; Amaducci, S.; Bullard, M.J.; Stutterheim, N.C.; Venturi, G.; Cromack, H.T.H. Agronomy of fibre hemp (Cannabis sativa L.) in Europe. Ind. Crops Prod. 2000, 11, 107–118. [Google Scholar] [CrossRef]
- Tang, K.; Fracasso, A.; Struik, P.C.; Yin, X.; Amaducci, S. Water-and nitrogen-use efficiencies of hemp (Cannabis sativa L.) based on whole-canopy measurements and modeling. Front. Plant Sci. 2018, 9, 951. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.; Struik, P.C.; Yin, X.; Thouminot, C.; Bjelková, M.; Stramkale, V.; Amaducci, S. Comparing hemp (Cannabis sativa L.) cultivars for dual-purpose production under contrasting environments. Ind. Crops Prod. 2016, 87, 33–44. [Google Scholar] [CrossRef]
- Linger, P.; Mussig, J.; Fischer, H.; Kobert, J. Industrial hemp (Cannabis sativa L.) growing on heavy metal contaminated soil: Fibre quality and phytoremediation potential. Ind. Crops Prod. 2002, 16, 33–42. [Google Scholar] [CrossRef]
- Luyckx, M.; Berni, R.; Cai, G.; Lutts, S.; Guerriero, G. Impact of heavy metals on non-food herbaceous crops and prophylactic role of Si. In Plant Metallomics and Functional Omics; Sablok, G., Ed.; Springer: Cham, Switzerland, 2019; pp. 303–321. [Google Scholar]
- Bouloc, P.; van der Werf, H.M.G. The role of hemp in sustainable development. In Hemp: Industrial Production and Uses; Bouloc, P., Allegret, S., Arnaud, L., Eds.; CABI: Wallingford, UK, 2013; pp. 278–289. [Google Scholar]
- Venturi, G.; Amaducci, M.T. Canapa (Cannabis sativa L.). In Le Colture da Fibra; Venturi, G., Amaducci, M.T., Eds.; Collana PRisCA; Edagricole: Bologna, Italy, 1999; pp. 33–55. [Google Scholar]
- Amaducci, S.; Gusovius, H.J. Hemp-cultivation, extraction and processing. In Industrial Applications of Natural Fibres: Structure, Properties and Technical Applications; Müssig, J., Ed.; Wiley: West Sussex, UK, 2010; pp. 109–134. [Google Scholar]
- Cosentino, S.L.; Riggi, E.; Testa, G.; Scordia, D.; Copani, V. Evaluation of European developed fibre hemp genotypes (Cannabis sativa L.) in semi-arid Mediterranean environment. Ind. Crops Prod. 2013, 50, 312–324. [Google Scholar] [CrossRef]
- Van Oosten, M.; Costa, A.; Punzo, P.; Landi, S.; Ruggiero, A.; Batelli, G.; Grillo, S. Genetics of drought stress tolerance in crop plants. In Drought Stress Tolerance in Plants; Hossain, M.A., Wani, S.H., Bhattachajee, S., Burrit, D., Eds.; Springer: Cham, Switzerland, 2016; Volume 2, pp. 39–70. ISBN 9783319324210. [Google Scholar] [CrossRef]
- Landi, S.; Esposito, S. Nitrate uptake affects cell wall synthesis and modelling. Front. Plant Sci. 2017, 8, 1376. [Google Scholar] [CrossRef]
- Gao, C.; Cheng, C.; Zhao, L.; Yu, Y.; Tang, Q.; Xin, P.; Liu, T.; Yan, Z.; Guo, Y.; Zang, G. Genome-wide expression profiles of hemp (Cannabis sativa L.) in response to drought stress. Int. J. Genom. 2018, 2018, 3057272. [Google Scholar] [CrossRef]
- Guerriero, G.; Behr, M.; Hausman, J.M.; Legay, S. Textile hemp vs. salinity: Insights from a targeted gene expression analysis. Genes 2017, 8, 242. [Google Scholar] [CrossRef]
- Aerts, R.; Chapin, F.S. The mineral nutrition of wild plants revisited: A re-evaluation of processes and patterns. In Advances in Ecological Research; Academic Press: Cambridge, MA, USA, 2000; Volume 30, pp. 1–67. [Google Scholar]
- Carrera, A.L.; Bertiller, M.B.; Sain, C.L.; Mazzarini, M.J. Relationship between plant nitrogen conservation strategies and the dynamics of soil nitrogen in the arid Patagonian Monte, Argentina. Plant Soil 2003, 255, 595–604. [Google Scholar] [CrossRef]
- Li, L.J.; Zeng, D.H.; Mao, R.; Yu, Z.Y. Nitrogen and phosphorus resorption of Artemisia scoparia, Chenopodium acuminatum, Cannabis sativa and Phragmites communis under nitrogen and phosphorus additionsin a semiarid grassland, China. Plant Soil Environ. 2012, 58, 446–451. [Google Scholar] [CrossRef]
- Aubin, M.; Seguin, P.; Vanasse, A.; Tremblay, G.F.; Mustafa, A.; Charron, J.F. Industrial hemp response to nitrogen, phosphorus, and potassium fertilization. Crop Forage Turfgrass Manag. 2015, 1. [Google Scholar] [CrossRef]
- Malceva, M.; Vikmane, M.; Stramkale, V. Changes of photosynthesis-related parameters and productivity of Cannabis sativa under different nitrogen supply. Environ. Exp. Biol. 2011, 9, 61–69. [Google Scholar]
- Ogden, M.; Hoefgen, R.; Roessner, U.; Persson, S.; Khan, G.A. Feeding the walls: How does nutrient availability regulate cell wall composition? Int. J. Mol. Sci. 2018, 9, 2691. [Google Scholar] [CrossRef]
- Berni, R.; Guerriero, G.; Cai, G. One for all and all for one! Increased plant heavy metal tolerance by growth promoting microbes: A metabolomic standpoint. In Plant Metallomics and Functional Omics; Sablok, G., Ed.; Springer Nature Switzerland AG: Cham, Switzerland, 2019. [Google Scholar]
- Lyu, D.; Backer, R.; Robinson, W.G.; Smith, D.L. Plant growth-promoting rhizobacteria for Cannabis production: Yield, cannabinoid profile and disease resistance. Front. Microbiol. 2019, 10, 1761. [Google Scholar] [CrossRef]
- Backer, R.; Rokem, J.S.; Ilangumaran, G.; Lamont, J.; Praslickova, D.; Ricci, E.; Subramanian, S.; Smith, D.L. Plant growth-promoting rhizobacteria: Context, mechanisms of action, and roadmap to commercialization of biostimulants for sustainable agriculture. Front. Plant Sci. 2018, 9, 1473. [Google Scholar] [CrossRef]
- Conant, R.; Walsh, R.; Walsh, M.; Bell, C.; Wallenstein, M. Effects of a microbial biostimulant, Mammoth P™, on Cannabis sativa bud yield. J. Hortic. For. 2017, 4. ISSN 2376-0354. [Google Scholar] [CrossRef]
- Losak, T.; Richter, R. Split nitrogen doses and their efficiency in poppy (Papaver somniferum L.) nutrition. Plant Soil Environ. UZPI 2005, 50, 484–488. [Google Scholar] [CrossRef]
- Mur, L.A.J.; Simpson, C.; Kumari, A.; Gupta, A.K.; Gupta, K.J. Moving nitrogen to the centre of plant defence against pathogens. Ann. Bot. 2017, 119, 703–709. [Google Scholar] [CrossRef]
- Flores-Sanchez, I.J.; Verpoorte, R. Secondary metabolism in Cannabis. Phytochem. Rev. 2008, 7, 615–639. [Google Scholar] [CrossRef]
- Fritz, C.; Palacios-Rojas, N.; Feil, R.; Stitt, M. Regulation of secondary metabolism by the carbon-nitrogen status in tobacco: Nitrate inhibits large sectors of phenylpropanoid metabolism. Plant J. 2006, 46, 533–548. [Google Scholar] [CrossRef]
- Razal, R.A.; Ellis, S.; Singh, S.; Lewis, N.G.; Towers, G.H.N. Nitrogen recycling in phenylpropanoid metabolism. Phytochemistry 1996, 41, 31–35. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Landi, S.; Nurcato, R.; de Lillo, A.; Lentini, M.; Grillo, S.; Esposito, S. Glucose-6-phosphate dehydrogenase plays a central role in the response of tomato (Solanum lycopersicum) plants to short and long-term drought. Plant Physiol. Biochem. 2016, 105, 79–89. [Google Scholar] [CrossRef]
- Landi, S.; Hausman, J.F.; Guerriero, G.; Esposito, S. Poaceae vs. abiotic stress: Focus on drought and salt stress, recent insight and perspectives. Front. Plant Sci. 2017, 8, 1214. [Google Scholar] [CrossRef]
- Bandurska, H.; Niedziela, J.; Pietrowska-Borek, M.; Nuc, K.; Chadzinikolau, T.; Radzikowska, D. Regulation of proline biosynthesis and resistance to drought stress in two barley (Hordeum vulgare L.) genotypes of different origin. Plant Physiol. Biochem. 2017, 118, 427–437. [Google Scholar] [CrossRef]
- Miyake, K.; Ito, T.; Senda, M.; Ishikawa, R.; Harada, T.; Niizeki, M.; Akada, S. Isolation of a subfamily of genes for R2R3-MYB transcription factors showing up-regulated expression under nitrogen nutrient-limited conditions. Plant Mol. Biol. 2003, 53, 237–245. [Google Scholar] [CrossRef]
- Todd, C.D.; Zeng, P.; Huete, A.M.; Hoyos, M.E.; Polacco, J.C. Transcripts of MYB-like genes respond to phosphorous and nitrogen deprivation in Arabidopsis. Planta 2004, 219, 1003–1009. [Google Scholar] [CrossRef]
- Dai, X.; Wang, Y.; Zhang, W.H. OsWRKY74, a WRKY transcription factor, modulates tolerance to phosphate starvation in rice. J. Exp. Bot. 2016, 67, 947–960. [Google Scholar] [CrossRef]
- Bi, H.; Luang, S.; Li, Y.; Bazanova, N.; Morran, S.; Song, Z.; Perera, M.A.; Hrmova, M.; Borisjuk, N.; Lopato, S. Identification and characterization of wheat drought-responsive MYB transcription factors involved in the regulation of cuticle biosynthesis. J. Exp. Bot. 2016, 67, 5363–5380. [Google Scholar] [CrossRef] [PubMed]
- Casaretto, J.A.; El-kereamy, A.; Zeng, B.; Stiegelmeyer, S.M.; Chen, X.; Bi, Y.M.; Rothstein, S.J. Expression of OsMYB55 in maize activates stress-responsive genes and enhances heat and drought tolerance. BMC Genom. 2016, 17, 312. [Google Scholar] [CrossRef] [PubMed]
- He, G.H.; Xu, J.Y.; Wang, Y.X.; Liu, J.M.; Li, P.S.; Chen, M.; Ma, Y.Z.; Xu, Z.S. Drought-responsive WRKY transcription factor genes TaWRKY1 and TaWRKY33 from wheat confer drought and/or heat resistance in Arabidopsis. BMC Plant Biol. 2016, 16, 116. [Google Scholar] [CrossRef] [PubMed]
- Saud, S.; Fahad, S.; Yajun, C.; Ihsan, M.Z.; Hammad, H.M.; Nasim, W.; Amanullah, J.; Arif, M.; Alharby, H. Effects of nitrogen supply on water stress and recovery mechanisms in Kentucky bluegrass plants. Front. Plant Sci. 2017, 8, 983. [Google Scholar] [CrossRef]
- Yang, S.Y.; Hao, D.L.; Song, Z.Z.; Yang, G.Z.; Wang, L.; Su, Y.H. RNA-Seq analysis of differentially expressed genes in rice under varied nitrogen supplies. Gene 2015, 25, 305–317. [Google Scholar] [CrossRef]
- Wang, X.; Wang, L.; Shangguan, Z. Leaf gas exchange and fluorescence of two winter wheat varieties in response to drought stress and nitrogen supply. PLoS ONE 2016, 11, e0165733. [Google Scholar] [CrossRef]
- Cao, X.; Zong, C.; Zhu, C.; Zhang, J.; Zhu, L.; Wu, L.; Jin, Q. Variability of leaf photosynthetic characteristics in rice and its relationship with resistance to water stress under different nitrogen nutrition regimes. Physiol. Plant. 2018. [Google Scholar] [CrossRef]
- Kanno, Y.; Hanada, A.; Chiba, Y.; Ichikawa, T.; Nakazawa, M.; Matsui, M.; Koshiba, T.; Kamiya, Y.; Seo, M. Identification of an abscisic acid transporter by functional screening using the receptor complex as a sensor. Proc. Natl. Acad. Sci. USA 2012, 109, 9653–9658. [Google Scholar] [CrossRef]
- Hsu, P.K.; Tsay, Y.F. Two phloem nitrate transporters, NRT1.11 and NRT1.12, are important for redistributing xylem-borne nitrate to enhance plant growth. Plant Physiol. 2013, 163, 844–856. [Google Scholar] [CrossRef]
- Guo, F.Q.; Young, J.; Crawford, N.M. The nitrate transporter AtNRT1.1 (CHL1) functions in stomatal opening and contributes to drought susceptibility in Arabidopsis. Plant Cell 2003, 15, 107–117. [Google Scholar] [CrossRef]
- Álvarez-Aragón, R.; Rodríguez-Navarro, A. Nitrate-dependent shoot sodium accumulation and osmotic functions of sodium in Arabidopsis under saline conditions. Plant J. 2017, 91, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Remans, T.; Nacry, P.; Pervent, M.; Girin, T.; Tillard, P.; Lepetit, M.; Gojon, A. A central role for the nitrate transporter NRT2.1 in the integrated morphological and physiological responses of the root system to nitrogen limitation in Arabidopsis. Plant Physiol. 2006, 140, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Chen, D.; Min, D.; Li, W.; Xu, Z.; Zhou, Y.; Li, L.; Chen, M.; Ma, Y. AtTGA4, a bZIP transcription factor, confers drought resistance by enhancing nitrate transport and assimilation in Arabidopsis thaliana. Biochem. Biophys. Res. Commun. 2015, 13, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Fu, Y.L.; Pike, S.M.; Bao, J.; Tian, W.; Zhang, Y.; Chen, C.Z.; Zhang, Y.; Li, H.M.; Huang, J.; et al. The Arabidopsis nitrate transporter NRT1.8 functions in nitrate removal from the xylem sap and mediates cadmium tolerance. Plant Cell 2010, 22, 1633–1646. [Google Scholar] [CrossRef]
- Taochy, C.; Gaillard, I.; Ipotesi, E.; Oomen, R.; Leonhardt, N.; Zimmermann, S.; Peltier, J.B.; Szponarski, W.; Simonneau, T.; Sentenac, H.; et al. The Arabidopsis root stele transporter NPF2.3 contributes to nitrate translocation to shoots under salt stress. Plant J. 2015, 83, 466–479. [Google Scholar] [CrossRef]
- Gollhofer, J.; Timofeev, R.; Lan, P.; Schmidt, W.; Buckhout, T.J. Vacuolar-iron-transporter1-like proteins mediate iron homeostasis in Arabidopsis. PLoS ONE 2014, 9, e110468. [Google Scholar] [CrossRef]
- Garcia, M.J.; Lucena, C.; Romera, F.J.; Alcantara, E.; Perez-Vicente, R. Ethylene and nitric oxide involvement in the up-regulation of key genes related to iron acquisition and homeostasis in Arabidopsis. J. Exp. Bot. 2010, 61, 3885–3899. [Google Scholar] [CrossRef]
- Antolín-Llovera, M.; Petutsching, E.K.; Ried, M.K.; Lipka, V.; Nürnberger, T.; Robatzek, S.; Parniske, M. Knowing your friends and foes–plant receptorlike kinases as initiators of symbiosis or defence. New Phytol. 2014, 204, 791–802. [Google Scholar] [CrossRef]
- Dufayard, J.F.; Bettembourg, M.; Fischer, I.; Droc, G.; Guiderdoni, E.; Périn, C.; Chantret, N.; Diévart, A. New insights on leucine-rich repeats receptor-like kinase orthologous relationships in angiosperms. Front. Plant Sci. 2017, 8, 381. [Google Scholar] [CrossRef]
- Winter, D.; Vinegar, B.; Nahal, H.; Ammar, R.; Wilson, G.V.; Provart, N.J. An “Electronic Fluorescent Pictograph” browser for exploring and analyzing large-scale biological data sets. PLoS ONE 2007, 2, e718. [Google Scholar] [CrossRef]
- Xie, W.; Wang, G.; Yuan, M.; Yao, W.; Lyu, K.; Zhao, H.; Yang, M.; Li, P.; Zhang, X.; Wang, Q.; et al. Breeding signatures of rice improvement revealed by a genomic variation map from a large germplasm collection. Proc. Natl. Acad. Sci. USA 2015, 112, 5411–5419. [Google Scholar] [CrossRef] [PubMed]
- Jallouli, S.; Ayadi, S.; Landi, S.; Capasso, G.; Santini, G.; Chamekh, Z.; Zouri, I.; Azaiez, F.E.B.; Trifa, Y.; Esposito, S. Physiological and molecular osmotic stress responses in three durum wheat (Triticum turgidum ssp Durum) genotype. Agronomy 2019, 19, 550. [Google Scholar] [CrossRef]
- Lehmann, T.; Skrok, A.; Dabert, M. Stress-induced changes in glutamate dehydrogenase activity imply its role in adaptation to C and N metabolism in lupine embryos. Physiol. Plant. 2010, 138, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Miflin, B.J.; Habash, D.Z. The role of glutamine synthetase and glutamate dehydrogenase in nitrogen assimilation and possibilities for improvement in the nitrogen utilization of crops. J. Exp. Bot. 2002, 53, 979–987. [Google Scholar] [CrossRef]
- Ranalli, P. Current status and future scenarios of hemp breeding. Euphytica 2004, 140, 121–131. [Google Scholar] [CrossRef]
- Ranalli, P.; Venturi, G. Hemp as a raw material for industrial applications. Euphytica 2004, 140, 1–6. [Google Scholar] [CrossRef]
- Salentijn, E.M.J.; Zhang, Q.; Amaducci, S.; Yang, M.; Trindade, L.M. New developments in fiber hemp (Cannabis sativa L.) breeding. Ind. Crops Prod. 2015, 68, 32–41. [Google Scholar] [CrossRef]
- Elshire, R.J.; Glaubitz, J.C.; Sun, Q.; Poland, J.A.; Kawamoto, K.; Buckler, E.S. A robust, simple genotyping-by-sequencing (GBS) approach for high diversityspecies. PLoS ONE 2011, 6, e19379. [Google Scholar] [CrossRef]
- Comai, L. The advantages and disadvantages of being polyploid. Nat. Rev. Genet. 2005, 6, 836–846. [Google Scholar] [CrossRef]
- Sattler, M.C.; Carvalho, C.R.; Clarindo, W.R. The polyploidy and its key role in plant breeding. Planta 2016, 243, 281–296. [Google Scholar] [CrossRef]
- Bagheri, M.; Mansouri, H. Effect of induced polyploidy on some biochemical parameters in Cannabis sativa L. Appl. Biochem. Biotechnol. 2015, 175, 2366–2375. [Google Scholar] [CrossRef]
- Laverty, K.U.; Stout, J.M.; Sullivan, M.J.; Hardik, S.; Navdeep, G.; Holbrook, L.; Deikus, G.; Sebra, R.; Hughes, T.R.; Page, J.E.; et al. A physical and genetic map of Cannabis sativa identifies extensive rearrangements at the THC/CBD acid synthase loci. Genome Res. 2019, 29, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Parsons, J.L.; Martin, S.L.; James, T.; Golenia, G.; Boudko, E.A.; Hepworth, S.R. Polyploidization for the genetic improvement of Cannabis sativa. Front. Plant Sci. 2019, 10, 476. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.A.; Wingen, L.U.; Griffiths, M.; Pound, M.P.; Gaju, O.; Foulkes, M.J.; le Gouis, J.; Griffiths, S.; Bennett, M.J.; Wells, D.M.; et al. Phenotyping pipeline reveals major seedling root growth QTL in hexaploid wheat. J. Exp. Bot. 2015, 66, 2283–2292. [Google Scholar] [CrossRef] [PubMed]
- Bretagnolle, F.; Thompson, J.D. Gametes with the somatic chromosome number: Mecchanisms of their formation and role in the evolution of autopolyploid plants. New Phytol. 1995, 129, 1–22. [Google Scholar] [CrossRef]
- Salentijn, E.M.J.; Petit, J.; Trindade, L.M. The complex interactions between flowering behavior and fiber quality in hemp. Front. Plant Sci. 2019, 10, 614. [Google Scholar] [CrossRef]
- Ruggiero, A.; Punzo, P.; Landi, S.; Costa, A.; van Ooosten, M.; Grillo, S. Improving plant water use efficiency through molecular genetics. Horticulturae 2017, 3, 31. [Google Scholar] [CrossRef]
- Zeng, M.; Guo, H.Y.; Guo, R.; Yang, M.; Mao, K.M. A study on phytoremediation of Cannabis sativa L. in heavy metals polluted soil. Chin. J. Soil Sci. 2013, 44, 472–476. [Google Scholar]
- Van der Werf, H.M.G.; van Geel, W.C.A.; van Gils, L.J.C.; Haverkort, A.J. Nitrogen fertilization and row width affect self-thinning and productivity of fibre hemp (Cannabis sativa L.). Field Crops Res. 1995, 42, 27–37. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Lupidi, G.; Nabissi, M.; Petrelli, R.; Kamte, S.L.N.; Cappellacci, L.; Fiorini, D.; Sut, S.; Dall’Acqua, S.; et al. The crop-residue of fiber hemp cv. Futura 75: From a waste product to a source of botanical insecticides. Environ. Sci. Pollut. Res. 2018, 25, 10515–10525. [Google Scholar] [CrossRef]
- Kowalska, M.; Ziomek, M.; Żbikowska, A. Stability of cosmetic emulsion containing different amount of hemp oil. Int. J. Cosmet. Sci. 2015, 37, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Mikulcová, V.; Kašpárková, V.; Humpolíček, P.; Buňková, L. Formulation, characterization and properties of hemp seed oil and its emulsions. Molecules 2017, 22, 700. [Google Scholar] [CrossRef] [PubMed]
- Corroon, J.; Phillips, J.A. A cross-sectional study of cannabidiol users. Cannabis Cannabinoid Res. 2018, 3, 152–161. [Google Scholar] [CrossRef]
- Sirikantaramas, S.; Morimoto, S.; Shoyama, Y.; Ishikawa, Y.; Wada, Y.; Shoyama, Y.; Taura, F. The gene controlling marijuana psychoactivity: Molecular cloning and heterologous expression of Δ1-tetrahydrocannabinolic acid synthase from Cannabis sativa L. J. Biol. Chem. 2004, 279, 39767–39774. [Google Scholar] [CrossRef] [PubMed]
- Taura, F.; Sirikantaramas, S.; Shoyama, Y.; Yoshikai, K.; Shoyama, Y.; Morimoto, S. Cannabidiolic-acid synthase, the chemotype-determining enzyme in the fiber-type Cannabis sativa. FEBS Lett. 2007, 581, 2929–2934. [Google Scholar] [CrossRef]
- Rodziewicz, P.; Loroch, S.; Marczak, Ł.; Sickmann, A.; Kayser, O. Cannabinoid synthases and osmoprotective metabolites accumulate in the exudates of Cannabis sativa L. glandular trichomes. Plant Sci. 2019, 284, 108–116. [Google Scholar] [CrossRef]
- Gagne, S.J.; Stout, J.M.; Liu, E.; Boubakir, Z.; Clark, S.M.; Page, J.E. Identification of olivetolic acid cyclase from Cannabis sativa reveals a unique catalytic route to plant polyketides. Proc. Natl. Acad. Sci. USA 2012, 109, 12811–12816. [Google Scholar] [CrossRef]
- Stout, J.M.; Boubakir, Z.; Ambrose, S.J.; Purves, R.W.; Page, J.E. The hexanoyl-CoA precursor for cannabinoid biosynthesis is formed by an acyl-activating enzyme in Cannabis sativa trichomes. Plant J. 2012, 71, 353–365. [Google Scholar] [CrossRef]
- Fellermeier, M.; Zenk, M.H. Prenylation of olivetolate by a hemp transferase yields cannabigerolic acid, the precursor of tetrahydrocannabinol. FEBS Lett. 1998, 427, 283–285. [Google Scholar] [CrossRef]
- Luo, X.; Reiter, M.A.; d’Espaux, L.; Wong, J.; Denby, C.M.; Lechner, A.; Zhang, Y.; Grzybowski, A.T.; Harth, S.; Lee, H.; et al. Complete biosynthesis of cannabinoids and their unnatural analogues in yeast. Nature 2019, 567, 123–126. [Google Scholar] [CrossRef]
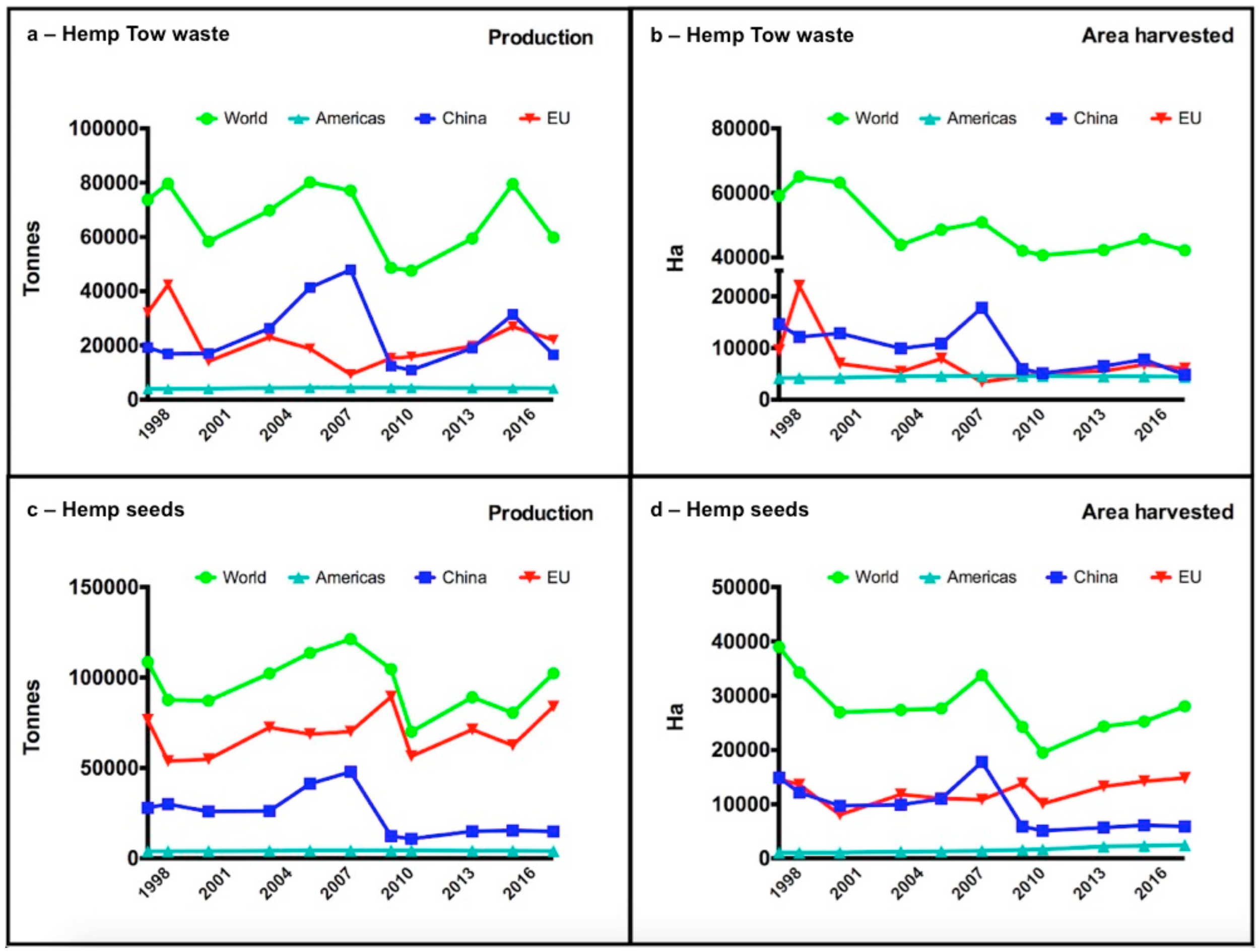
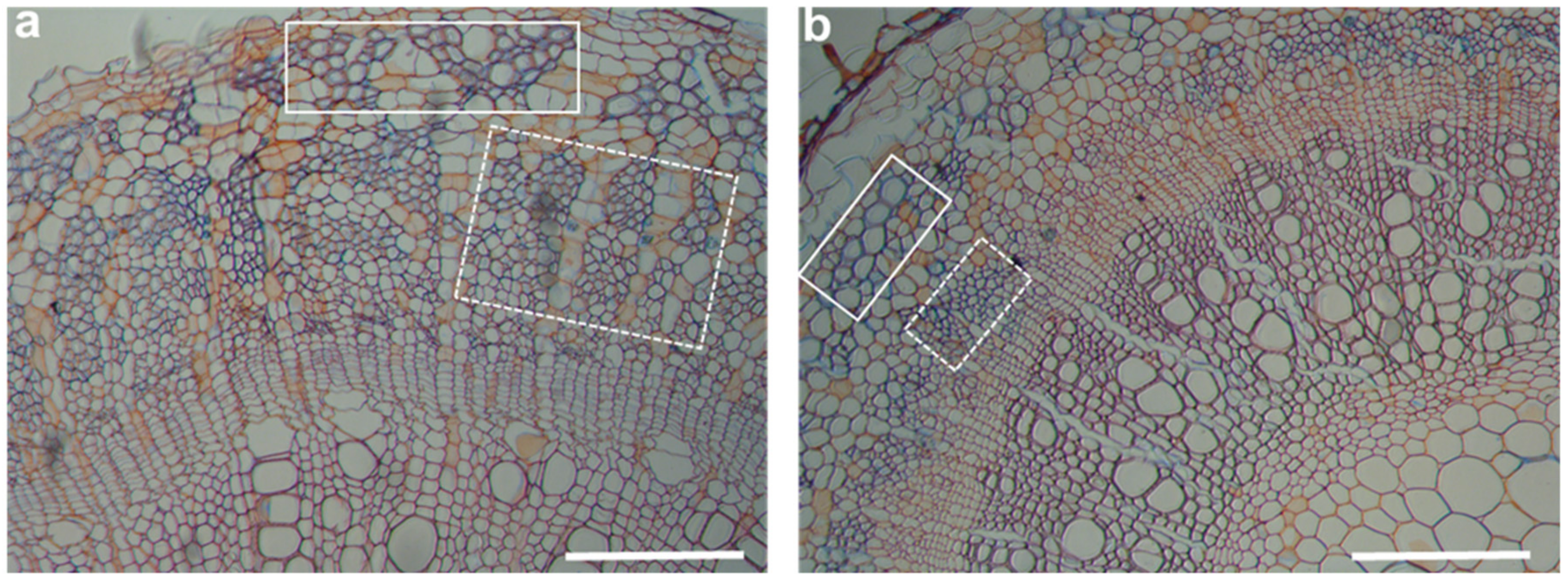
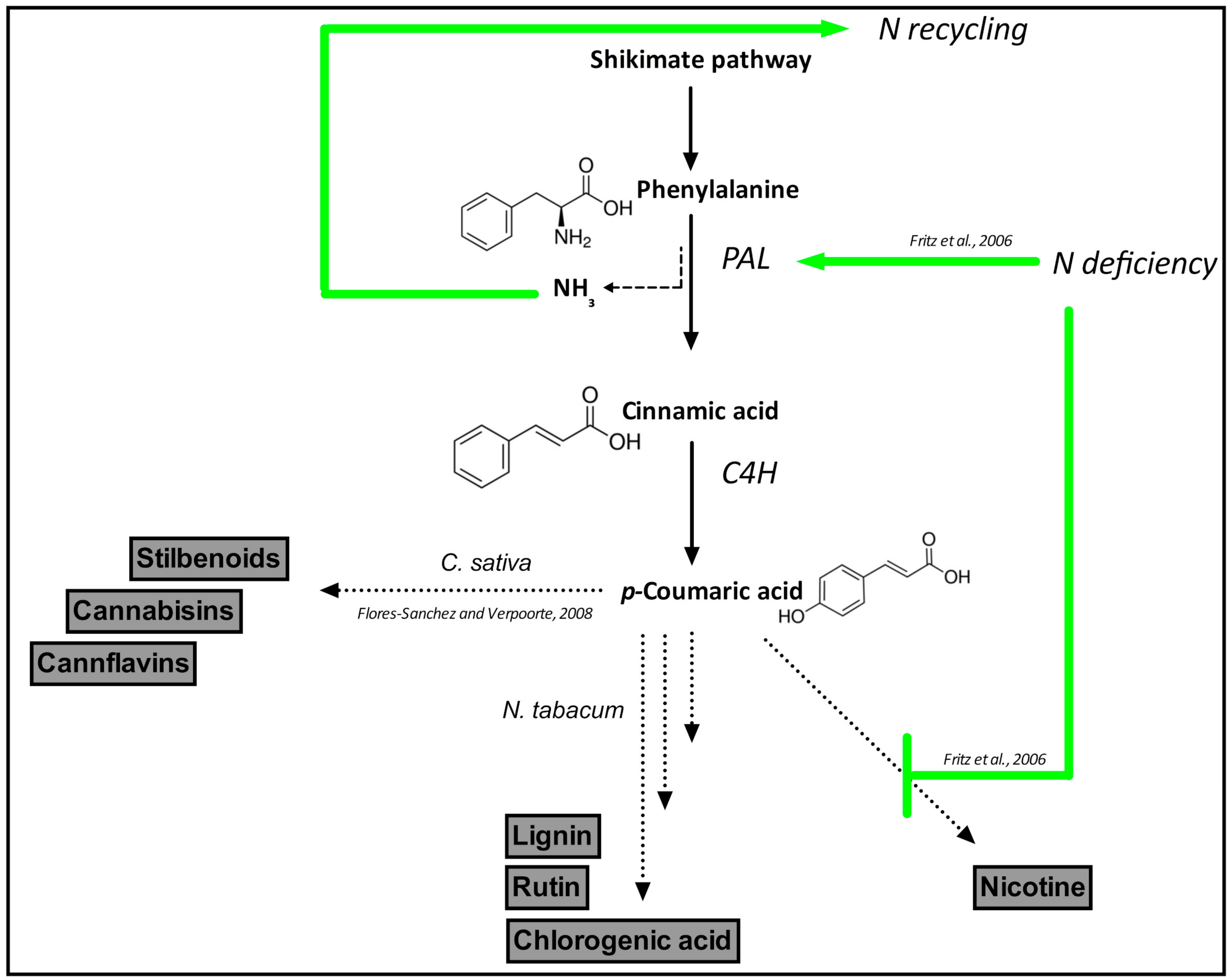
| Plant | Control | N Supply | P Supply | N + P Addition | ||||
|---|---|---|---|---|---|---|---|---|
| NRE (%) | PRE (%) | NRE (%) | PRE (%) | NRE (%) | PRE (%) | NRE (%) | PRE (%) | |
| C. sativa | 62 | 42 | 35 | 35 | 52 | 38 | 25 | 41 |
| P. communis | 42 | 10 | 45 | 20 | 58 | 10 | 60 | 15 |
| A. scoparia | 42 | 50 | 20 | 62 | 55 | 40 | 22 | 50 |
| C. acuminatum | 48 | 45 | 30 | 60 | 40 | 42 | 18 | 58 |
| Locus | Behavior vs. Abiotic Stress | Annotation | GO or KEGG Categories Related to N Metabolism |
|---|---|---|---|
| gi_351617093_gb_JP471394.1 | UP upon Drought | MYBR domain class transcription factor | GO:0010243 |
| gi_351606916_gb_JP461241.1 | UP upon Drought | WRKY transcription factor 33-like | GO:0010243 |
| gi_351617961_gb_JP472262.1 | UP upon Drought | Vinorine synthase-like | GO:0006807 |
| gi_351624360_gb_JP478661.1 | UP upon Drought | Triacylglycerol lipase | GO:0006807 |
| gi_351627864_gb_JP480747.1 | UP upon Drought | Vinorine synthase-like | GO:0034641 |
| gi_351612890_gb_JP467191.1 | UP upon Drought | Transcription factor LHY | GO:0010243 |
| gi_351603990_gb_JP458344.1 | UP upon Drought | Cysteine-rich receptor-like protein kinase | GO:0071705 |
| gi_351624708_gb_JP479009.1 | UP upon Drought | Delta-1-pyrroline-5-carboxylate synthetase | GO:0034641 |
| gi_351618788_gb_JP473089.1 | UP upon Drought | Pleiotropic drug resistance protein | GO:0006995 |
| gi_351599092_gb_JP453596.1 | UP upon Drought | ACD1-like | GO:0044270 |
| gi_351596616_gb_JP451172.1 | UP upon Drought | Probable peptide/nitrate transporter | GO:0006807 |
| gi_351625252_gb_JP479553.1 | UP upon Drought | Glucose-methanol-choline oxidoreductase | GO:0006807 |
| gi_351629105_gb_JP481988.1 | UP upon Drought | Hypothetical protein | GO:0034641 |
| gi_351628557_gb_JP481440.1 | UP upon Drought | Deoxytaxol N-benzoyltransferase | GO:0006807 |
| gi_351622676_gb_JP476977.1 | UP upon Drought | Shikimate O-hydroxycinnamoyltransferase | GO:0034641 |
| gi_351597997_gb_JP452531.1 | UP upon Drought | Aldehyde dehydrogenase | GO:0006807 |
| gi_351624288_gb_JP478589.1 | UP upon Drought | Hypothetical protein | GO:0051171 |
| gi_351615767_gb_JP470068.1 | UP upon Drought | MYB domain protein 20 | GO:0071705 |
| gi_351623654_gb_JP477955.1 | DOWN upon Drought | 21 kDa protein | GO:0071705 |
| gi_351629055_gb_JP481938.1 | DOWN upon Drought | Urea-proton symporter | GO:0006995 |
| gi_351624448_gb_JP478749.1 | DOWN upon Drought | Allantoinase | GO:0006995 |
| gi_351624658_gb_JP478959.1 | DOWN upon Drought | Salutaridinol 7-O-acetyltransferase | GO:0006807 |
| gi_351597746_gb_JP452287.1 | DOWN upon Drought | Anthranilate N-benzoyltransferase | GO:0006807 |
| gi_351605867_gb_JP460208.1 | DOWN upon Drought | Nitrate transporter 1.2 | GO:0006807 |
| gi_351602034_gb_JP456469.1 | DOWN upon Drought | Nitrate transporter 1.2 | GO:0006807 |
| gi_351619289_gb_JP473590.1 | DOWN upon Drought | Non-symbiotic hemoglobin 2 | GO:0009399 |
| gi_351591331_gb_JP449779.1 | DOWN upon Drought | Protein PHR1-LIKE 1 | GO:0034641 |
| gi_351625347_gb_JP479648.1 | DOWN upon Drought | Pleiotropic drug resistance protein 2 | GO:0006995 |
| gi_351590806_gb_JP449264.1 | DOWN upon Drought | Vacuolar iron transporter | GO:0009399 |
| gi_351598838_gb_JP453346.1 | DOWN upon Drought | Pleiotropic drug resistance protein | GO:0006995 |
| gi_351620563_gb_JP474864.1 | DOWN upon Drought | Leucine-rich repeat family protein | GO:0006995 |
| gi_351597483_gb_JP452028.1 | DOWN upon Drought | LRR receptor-like serine/threonine kinase | GO:0071705 |
| gi_351596259_gb_JP450816.1 | DOWN upon Drought | Glutamine synthetase leaf isozyme | GO:0009399/Ko00910 |
| gi_351624862_gb_JP479163.1 | DOWN upon Drought | LRR receptor-like serine/threonine kinase | GO:0071705 |
| gi_351624507_gb_JP478808.1 | DOWN upon Drought | L-3-cyanoalanine synthase 1 | GO:0051410 |
| gi_351623596_gb_JP477897.1 | DOWN upon Drought | Tropinone reductase | GO:0044271 |
| gi_351605608_gb_JP459952.1 | DOWN upon Drought | Major facilitator superfamily protein | GO:0006807 |
| gi_351606133_gb_JP460466.1 | DOWN upon Drought | BAHD acyltransferase | GO:0006807 |
| gi_351623568_gb_JP477869.1 | DOWN upon Drought | Tocopherol O-methyltransferase | GO:0034641 |
| gi_351601188_gb_JP455639.1 | DOWN upon Drought | LRR receptor-like serine/threonine kinase | GO:0071705 |
| gi_351598272_gb_JP452798.1 | DOWN upon Drought | Aminomethyltransferase, mitochondrial | ko00910 |
| gi_351612051_gb_JP466352.1 | DOWN upon Drought | Carbonic anhydrase 2 | ko00910 |
| gi_351615730_gb_JP470031.1 | DOWN upon Drought | Carbonic anhydrase, chloroplastic | ko00910 |
| gi_351617853_gb_JP472154.1 | DOWN upon Drought | Carbonic anhydrase, chloroplastic | ko00910 |
| gi_351621906_gb_JP476207.1 | DOWN upon Drought | Bifunctional monodehydroascorbate reductase | ko00910 |
| PK00197.1 | UP upon Salinity | Glutamate dehydrogenase | ko00910 |
| PK06425.1 | UP upon Salinity | Glutamate synthase | ko00910 |
| PK21222.1 | DOWN upon Salinity | Carbonic anhydrase 1 | ko00910 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Landi, S.; Berni, R.; Capasso, G.; Hausman, J.-F.; Guerriero, G.; Esposito, S. Impact of Nitrogen Nutrition on Cannabis sativa: An Update on the Current Knowledge and Future Prospects. Int. J. Mol. Sci. 2019, 20, 5803. https://doi.org/10.3390/ijms20225803
Landi S, Berni R, Capasso G, Hausman J-F, Guerriero G, Esposito S. Impact of Nitrogen Nutrition on Cannabis sativa: An Update on the Current Knowledge and Future Prospects. International Journal of Molecular Sciences. 2019; 20(22):5803. https://doi.org/10.3390/ijms20225803
Chicago/Turabian StyleLandi, Simone, Roberto Berni, Giorgia Capasso, Jean-Francois Hausman, Gea Guerriero, and Sergio Esposito. 2019. "Impact of Nitrogen Nutrition on Cannabis sativa: An Update on the Current Knowledge and Future Prospects" International Journal of Molecular Sciences 20, no. 22: 5803. https://doi.org/10.3390/ijms20225803
APA StyleLandi, S., Berni, R., Capasso, G., Hausman, J.-F., Guerriero, G., & Esposito, S. (2019). Impact of Nitrogen Nutrition on Cannabis sativa: An Update on the Current Knowledge and Future Prospects. International Journal of Molecular Sciences, 20(22), 5803. https://doi.org/10.3390/ijms20225803










