C2H2-Type Zinc Finger Proteins (DkZF1/2) Synergistically Control Persimmon Fruit Deastringency
Abstract
1. Introduction
2. Results
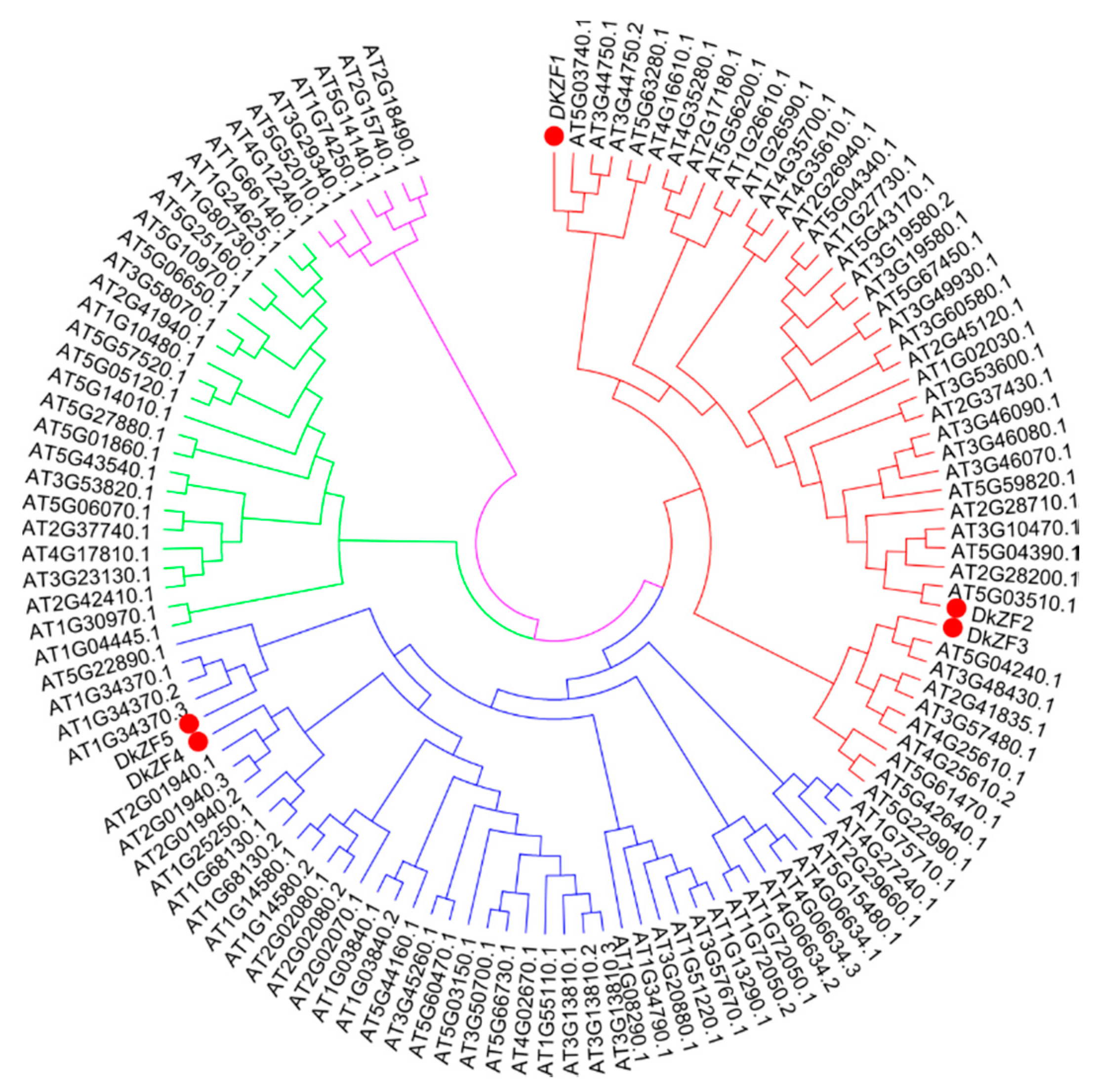
2.1. Phylogeny and Sequence Analyses of DkZFs
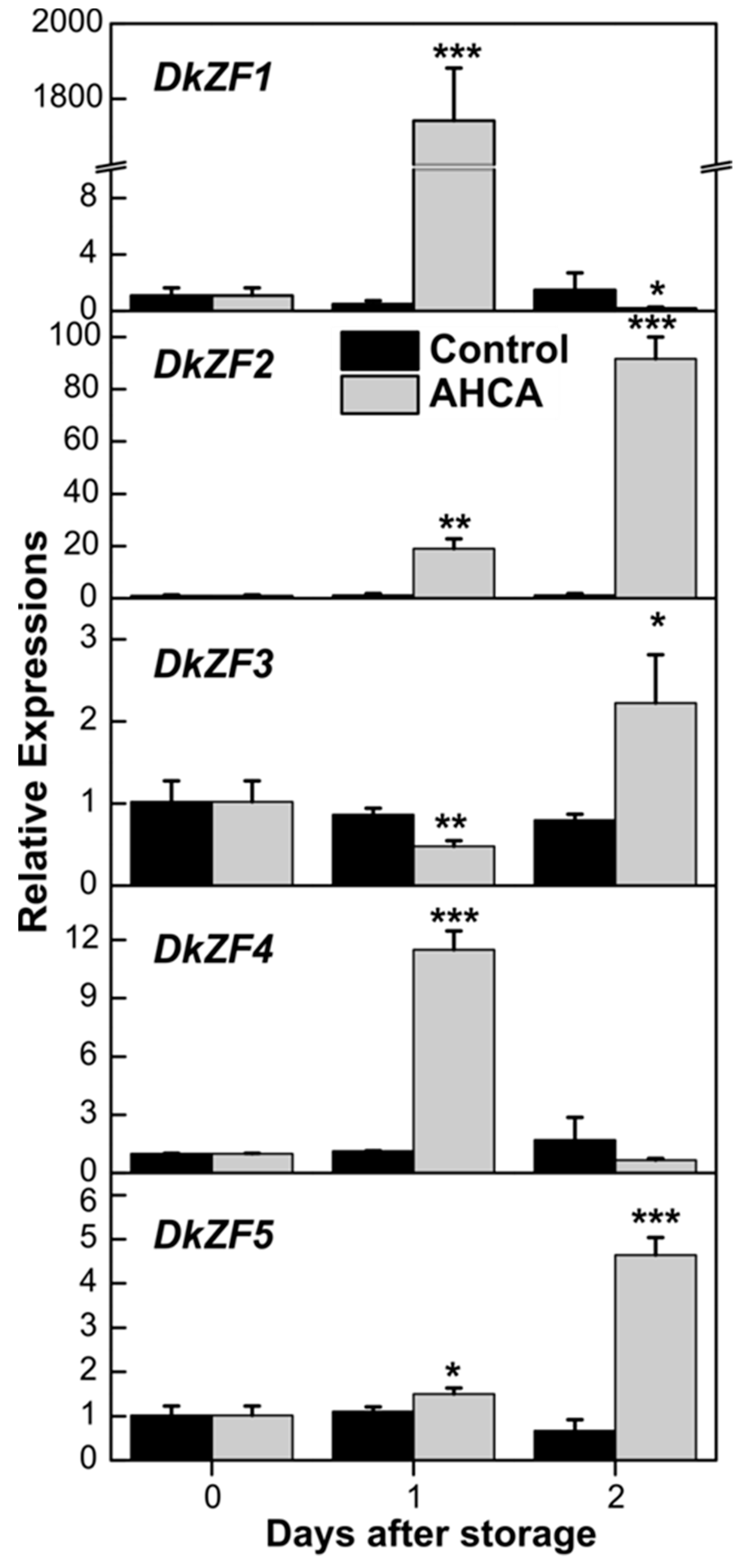
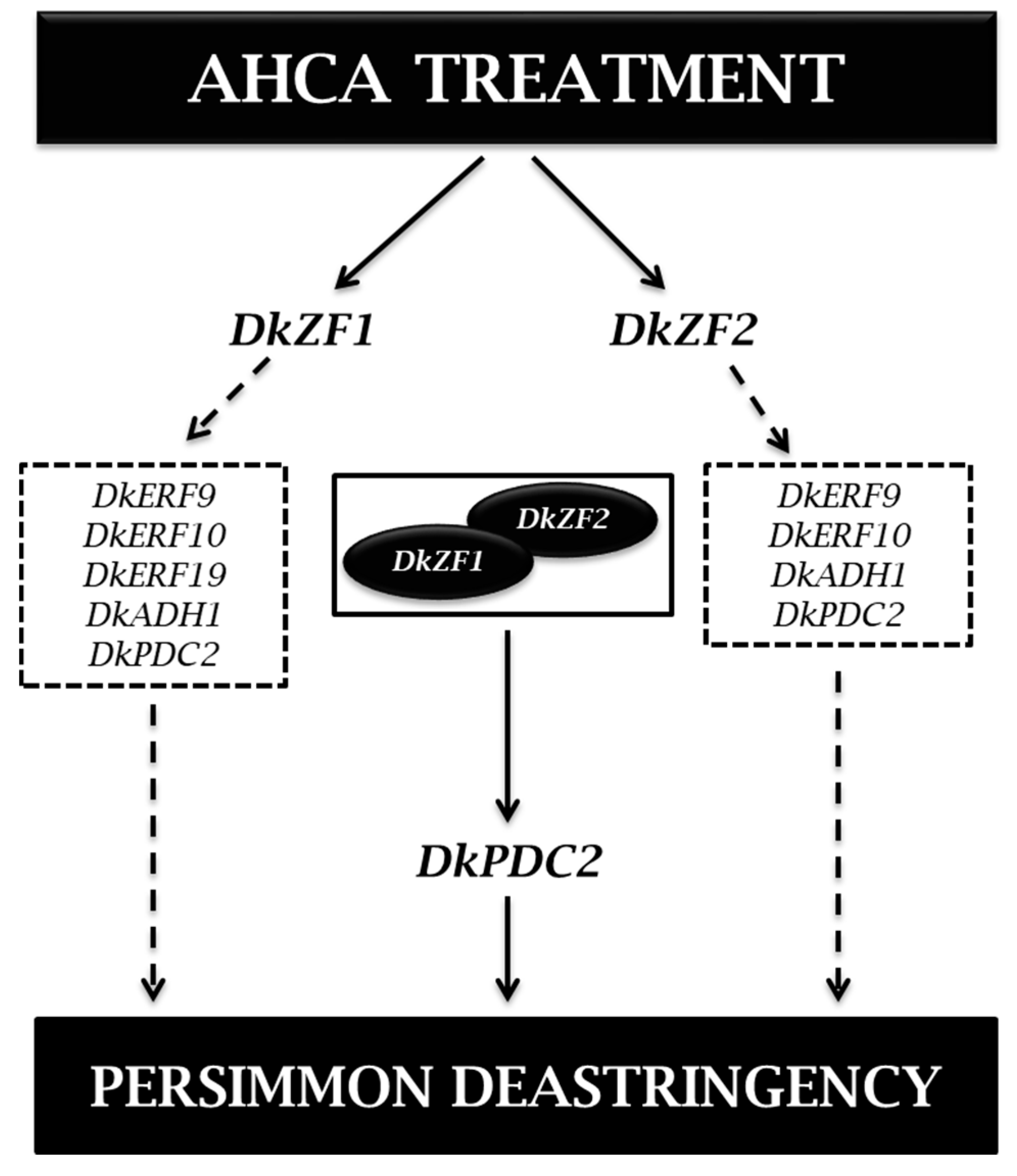
2.2. Expression of DkZFs in Response to AHCA Treatment
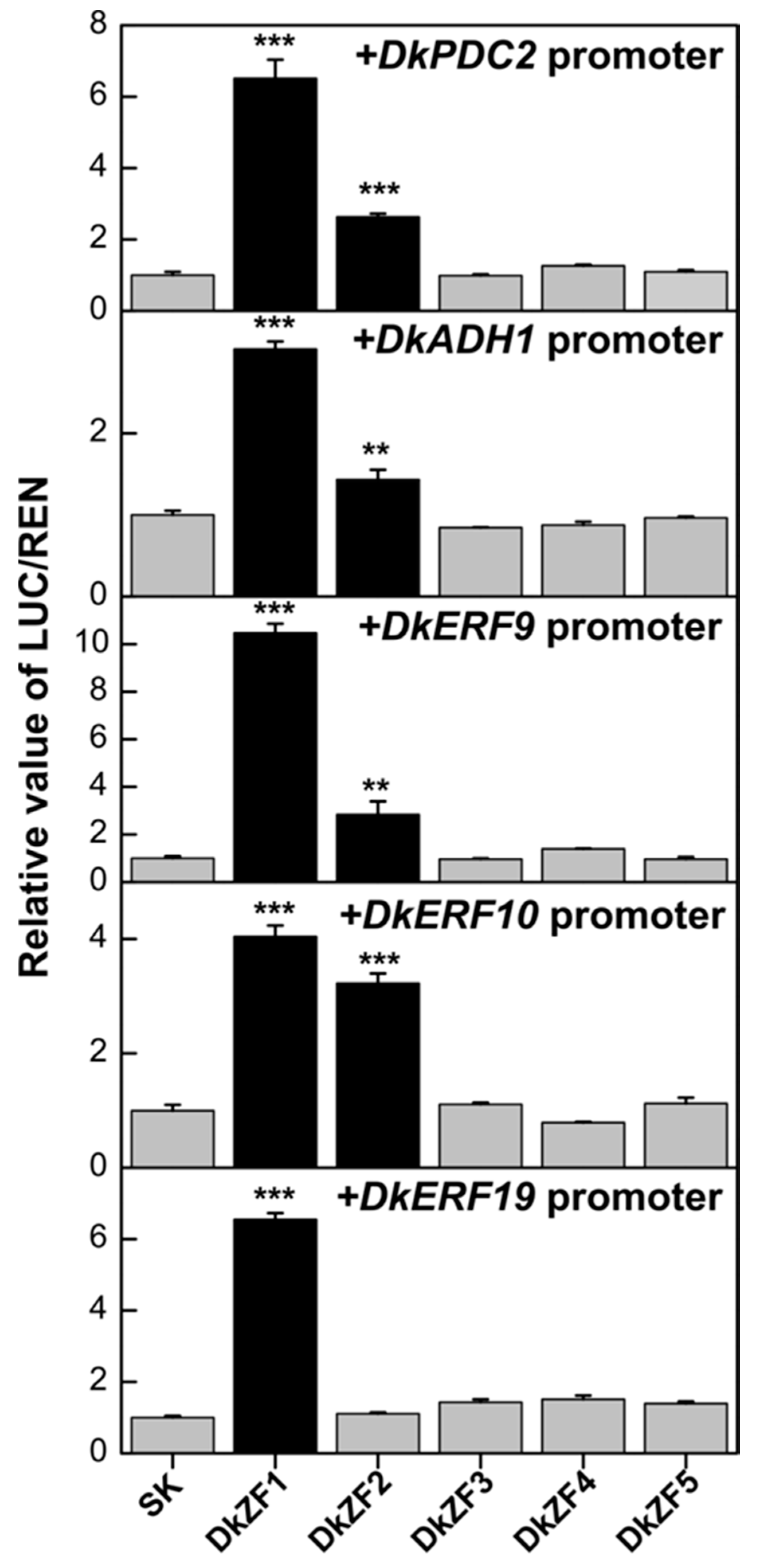
2.3. Transcriptional Effects of DkZFs on Promoters of Deastringency-Related Genes
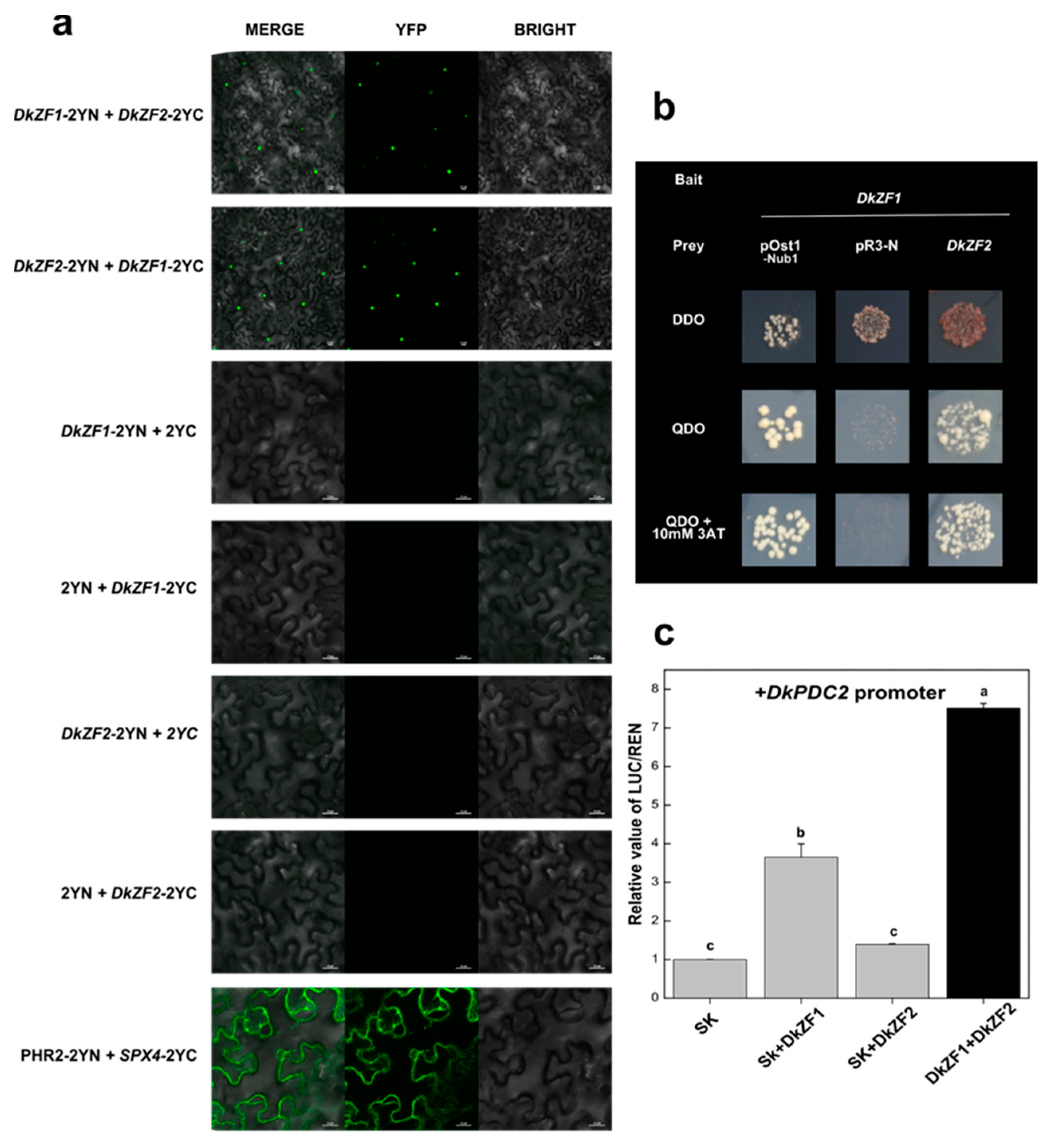
2.4. Synergistic Regulations of DkZF2 and DkZF1 on DkPDC2 Promoter
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Treatments
4.2. Gene Isolation and Sequence Analysis
4.3. RNA Extraction and cDNA Synthesis
4.4. Oligonucleotide Primers and Real-Time PCR
4.5. Dual-Luciferase Assay
4.6. Bimolecular Fluorescence Complementation (BiFC) Assays
4.7. Yeast Two-Hybrid Assays
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CTAB | Cetyltrimethyl Ammonium Bromide |
| MEGA | Molecular Evolutionary Genetics Analysis |
| RNA-seq | Ribose nucleic acid sequencing |
| Y2H | Yeast two hybrid |
References
- Banti, V.; Giuntoli, B.; Gonzali, S.; Loreti, E.; Magneschi, L.; Novi, G.; Paparelli, E.; Parlanti, S.; Pucciariello, C.; Santaniello, A. Low oxygen response mechanisms in green organisms. Int. J. Mol. Sci. 2013, 14, 4734–4761. [Google Scholar] [CrossRef] [PubMed]
- Ismond, K.P.; Dolferus, R.; De Pauw, M.; Dennis, E.S.; Good, A.G. Enhanced low oxygen survival in Arabidopsis through increased metabolic flux in the fermentative pathway. Plant Physiol. 2003, 132, 1292–1302. [Google Scholar] [CrossRef] [PubMed]
- Rocha, M.; Licausi, F.; Araújo, W.L.; Nunes Nesi, A.; Sodek, L.; Fernie, A.R.; Van Dongen, J.T. Glycolysis and the tricarboxylic acid cycle are linked by alanine aminotransferase during hypoxia induced by waterlogging of Lotus japonicus. Plant Physiol. 2010, 152, 1501–1513. [Google Scholar] [CrossRef] [PubMed]
- Dolferus, R.; Klok, E.J.; Delessert, C.; Wilson, S.; Ismond, K.P.; Good, A.G.; Peacock, W.J.; Dennis, E.S. Enhancing the anaerobic response. Ann. Bot. 2003, 91, 111–117. [Google Scholar] [CrossRef]
- Loreti, E.; Poggi, A.; Novi, G.; Alpi, A.; Perata, P. A genome wide analysis of the effects of sucrose on gene expression in Arabidopsis seedlings under anoxia. Plant Physiol. 2005, 137, 1130–1138. [Google Scholar] [CrossRef]
- Min, T.; Yin, X.R.; Shi, Y.N.; Luo, Z.R.; Yao, Y.C.; Grierson, D.; Ferguson, I.B.; Chen, K.S. Ethylene-responsive transcription factors interact with promoters of ADH and PDC involved in persimmon (Diospyros kaki) fruit de-astringency. J. Exp. Bot. 2012, 63, 6393–6405. [Google Scholar] [CrossRef]
- Ali, S.; Khan, A.S.; Malik, A.U.; Shahid, M. Effect of controlled atmosphere storage on pericarp browning, bioactive compounds and antioxidant enzymes of litchi fruits. Food Chem. 2016, 206, 18–29. [Google Scholar] [CrossRef]
- Bekele, E.A.; Ampofo, A.J.; Alis, R.; Hertog, M.L.; Nicolai, B.M.; Geeraerd, A.H. Dynamics of metabolic adaptation during initiation of controlled atmosphere storage of ‘Jonagold’apple: Effects of storage gas concentrations and conditioning. Postharvest Biol. Technol. 2016, 117, 9–20. [Google Scholar] [CrossRef]
- Matityahu, I.; Marciano, P.; Holland, D.; Ben-Arie, R.; Amir, R. Differential effects of regular and controlled atmosphere storage on the quality of three cultivars of pomegranate (Punica granatum L.). Postharvest Biol. Technol. 2016, 115, 132–141. [Google Scholar] [CrossRef]
- Taira, S.; Ikeda, K.; Ohkawa, K. Comparison of insolubility of tannins induced by acetaldehyde vapor in fruits of three types of astringent persimmon. J. Jpn. Soc. Hort. Sci. Food Tech. 2001, 48, 684–687. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Apple peels as a value-added food ingredient. J. Agric. Food Chem. 2003, 51, 1676–1683. [Google Scholar] [CrossRef] [PubMed]
- Galvis Sánchez, A.C.; Gil, I.A.; Gil, M.I. Comparative study of six pear cultivars in terms of their phenolic and vitamin C contents and antioxidant capacity. J. Sci. Food Agric. 2003, 83, 995–1003. [Google Scholar] [CrossRef]
- Remorini, D.; Tavarini, S.; Degl, I.E.; Loreti, F.; Massai, R.; Guidi, L. Effect of rootstocks and harvesting time on the nutritional quality of peel and flesh of peach fruits. Food Chem. 2008, 110, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Moon, J.Y.; Kim, H.; Lee, D.-S.; Cho, M.; Choi, H.K.; Kim, Y.S.; Mosaddik, A.; Cho, S.K. Antioxidant and antiproliferative activities of mango (Mangifera indica L.) flesh and peel. Food Chem. 2010, 121, 429–436. [Google Scholar] [CrossRef]
- Li, Y.; Guo, C.; Yang, J.; Wei, J.; Xu, J.; Cheng, S. Evaluation of antioxidant properties of pomegranate peel extract in comparison with pomegranate pulp extract. Food Chem. 2006, 96, 254–260. [Google Scholar] [CrossRef]
- Kim, J.G.; Beppu, K.; Kataoka, I. Varietal differences in phenolic content and astringency in skin and flesh of hardy kiwifruit resources in Japan. Sci. Hortic. 2009, 120, 551–554. [Google Scholar] [CrossRef]
- Silva, B.M.; Andrade, P.B.; Ferreres, F.; Domingues, A.L.; Seabra, R.M.; Ferreira, M.A. Phenolic profile of quince fruit (Cydonia oblonga Miller) (pulp and peel). J. Agric. Food Chem. 2002, 50, 4615–4618. [Google Scholar] [CrossRef]
- Akagi, T.; Ikegami, A.; Suzuki, Y.; Yoshida, J.; Yamada, M.; Sato, A.; Yonemori, K. Expression balances of structural genes in shikimate and flavonoid biosynthesis cause a difference in proanthocyanidin accumulation in persimmon (Diospyros kaki Thunb.) fruit. Planta 2009, 230, 899–915. [Google Scholar] [CrossRef]
- Lesschaeve, I.; Noble, A.C. Polyphenols: Factors influencing their sensory properties and their effects on food and beverage preferences. Am. J. Clin. Nutr. 2005, 81, 330S–335S. [Google Scholar] [CrossRef]
- Min, T.; Fang, F.; Ge, H.; Shi, Y.N.; Luo, Z.R.; Yao, Y.C.; Grierson, D.; Yin, X.R.; Chen, K.S. Two novel anoxia-induced ethylene response factors that interact with promoters of deastringency-related genes from persimmon. PLoS ONE 2014, 9, e97043. [Google Scholar] [CrossRef]
- Min, T.; Wang, M.M.; Wang, H.; Liu, X.; Fang, F.; Grierson, D.; Yin, X.R.; Chen, K.S. Isolation and expression of NAC genes during persimmon fruit postharvest astringency removal. Int. J. Mol. Sci. 2015, 16, 1894–1906. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.G.; Gong, Z.Y.; Wang, M.M.; Li, X.; Grierson, D.; Yin, X.R.; Chen, K.S. A transcription factor network responsive to high CO2/hypoxia is involved in deastringency in persimmon fruit. J. Exp. Bot. 2018, 69, 2061–2070. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.G.; Gong, Z.Y.; Huang, J.; Grierson, D.; Chen, K.S.; Yin, X.R. High-CO2/hypoxia-responsive transcription factors DkERF24 and DkWRKY1 interact and activate DkPDC2 promoter. Plant Physiol. 2019, 180, 621–633. [Google Scholar] [CrossRef] [PubMed]
- Jamil, W.; Wu, W.; Ahmad, M.; Zhu, Q.G.; Liu, X.F.; Jin, R.; Yin, X.R. High-CO2/hypoxia-modulated NAC transcription factors involved in de-astringency of persimmon fruit. Sci. Hortic. 2019, 252, 201–207. [Google Scholar] [CrossRef]
- Jin, R.; Zhu, Q.G.; Shen, X.Y.; Wang, M.M.; Jamil, W.; Grierson, D.; Yin, X.R.; Chen, K.S. DkNAC7, a novel high CO2/hypoxia-induced NAC transcription factor, regulates persimmon fruit de-astringency. PLoS ONE 2018, 13, e0194326. [Google Scholar] [CrossRef]
- Takatsuji, H. Zinc-finger proteins: The classical zinc finger emerges in contemporary plant science. Plant Mol. Biol. 1999, 39, 1073–1078. [Google Scholar] [CrossRef]
- Jenkins, T.H.; Li, J.; Scutt, C.P.; Gilmartin, P.M. Analysis of members of the Silene latifolia Cys 2/His 2 zinc-finger transcription factor family during dioecious flower development and in a novel stamen-defective mutant ssf1. Planta 2005, 220, 559–571. [Google Scholar] [CrossRef]
- Schumann, U.; Prestele, J.; OGeen, H.; Brueggeman, R.; Wanner, G.; Gietl, C. Requirement of the C3HC4 zinc RING finger of the Arabidopsis PEX10 for photorespiration and leaf peroxisome contact with chloroplasts. Proc. Natl. Acad. Sci. USA 2007, 104, 1069–1074. [Google Scholar] [CrossRef]
- Moore, M.; Ullman, C. Recent developments in the engineering of zinc finger proteins. Brief. Func. Genom. 2003, 1, 342–355. [Google Scholar] [CrossRef][Green Version]
- Ciftci, Y.S.; Morsy, M.R.; Song, L.; Coutu, A.; Krizek, B.A.; Lewis, M.W.; Warren, D.; Cushman, J.; Connolly, E.L.; Mittler, R. The EAR-motif of the Cys2/His2-type zinc finger protein ZAT7 plays a key role in the defense response of Arabidopsis to salinity stress. J. Biol. Chem. 2007, 282, 9260–9268. [Google Scholar] [CrossRef]
- Kim, S.H.; Hong, J.K.; Lee, S.C.; Sohn, K.H.; Jung, H.W.; Hwang, B.K. CAZFP1, Cys 2/His 2-type zinc-finger transcription factor gene functions as a pathogen-induced early-defense gene in Capsicum annuum. Plant Mol. Biol. 2004, 55, 883–904. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, H.; Maruyama, K.; Sakuma, Y.; Meshi, T.; Iwabuchi, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Arabidopsis Cys2/His2-type zinc-finger proteins function as transcription repressors under drought, cold, and high-salinity stress conditions. Plant Physiol. 2004, 136, 2734–2746. [Google Scholar] [CrossRef] [PubMed]
- Vogel, J.T.; Zarka, D.G.; Van Buskirk, H.A.; Fowler, S.G.; Thomashow, M.F. Roles of the CBF2 and ZAT12 transcription factors in configuring the low temperature transcriptome of Arabidopsis. Plant J. 2005, 41, 195–211. [Google Scholar] [CrossRef] [PubMed]
- Yin, M.; Wang, Y.; Zhang, L.; Li, J.; Quan, W.; Yang, L.; Wang, Q.; Chan, Z. The Arabidopsis Cys2/His2 zinc finger transcription factor ZAT18 is a positive regulator of plant tolerance to drought stress. J. Exp. Bot. 2017, 68, 2991–3005. [Google Scholar] [PubMed]
- Zhang, S.; Zhang, D.; Fan, S.; Du, L.; Shen, Y.; Xing, L.; Li, Y.; Ma, J.; Han, M. Effect of exogenous GA3 and its inhibitor paclobutrazol on floral formation, endogenous hormones, and flowering-associated genes in ‘Fuji’ apple (Malus Domestica Borkh.). Plant Physiol. Biochem. 2016, 107. [Google Scholar] [CrossRef]
- Pandey, D.M.; Kim, S.R. Identification and expression analysis of hypoxia stress inducible CCCH-type zinc finger protein genes in rice. J. Plant Biol. 2012, 55, 489–497. [Google Scholar] [CrossRef]
- Huang, J.; Sun, S.J.; Xu, D.Q.; Yang, X.; Bao, Y.-M.; Wang, Z.F.; Tang, H.J.; Zhang, H. Increased tolerance of rice to cold, drought and oxidative stresses mediated by the overexpression of a gene that encodes the zinc finger protein ZFP245. Biochem. Biophys. Res. Commun. 2009, 389, 556–561. [Google Scholar] [CrossRef]
- Zhang, H.; Ni, L.; Liu, Y.; Wang, Y.; Zhang, A.; Tan, M.; Jiang, M. The C2H2-type Zinc Finger Protein ZFP182 is Involved in Abscisic Acid-Induced Antioxidant Defense in Rice F. J. Integr. Plant Biol. 2012, 54, 500–510. [Google Scholar] [CrossRef]
- Shi, H.; Liu, G.; Wei, Y.; Chan, Z. The zinc-finger transcription factor ZAT6 is essential for hydrogen peroxide induction of anthocyanin synthesis in Arabidopsis. Plant Mol. Biol. 2018, 97, 165–176. [Google Scholar] [CrossRef]
- Ciftci, Y.S.; Mittler, R. The zinc finger network of plants. Cell. Mol. Life Sci. 2008, 65, 1150–1160. [Google Scholar] [CrossRef]
- Davletova, S.; Schlauch, K.; Coutu, J.; Mittler, R. The zinc-finger protein Zat12 plays a central role in reactive oxygen and abiotic stress signaling in Arabidopsis. Plant Physiol. 2005, 139, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Le, C.T.T.; Brumbarova, T.; Ivanov, R.; Stoof, C.; Weber, E.; Mohrbacher, J.; Fink-Straube, C.; Bauer, P. Zinc finger of Arabidopsis thaliana12 (ZAT12) interacts with FER-like iron deficiency-induced transcription factor (FIT) linking iron deficiency and oxidative stress responses. Plant Physiol. 2016, 170, 540–557. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.; Shulaev, V.; Mittler, R. Reactive oxygen signaling and abiotic stress. Physiol. Plant 2008, 133, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Salvador, A.; Arnal, L.; Besada, C.; Larrea, V.; Quiles, A.; Pérez, M.I. Physiological and structural changes during ripening and deastringency treatment of persimmon fruit cv.‘Rojo Brillante’. Postharvest Biol. Technol. 2007, 46, 181–188. [Google Scholar] [CrossRef]
- Espley, R.V.; Hellens, R.P.; Putterill, J.; Stevenson, D.E.; Kutty, A.S.; Allan, A.C. Red colouration in apple fruit is due to the activity of the MYB transcription factor, MdMYB10. Plant J. 2007, 49, 414–427. [Google Scholar] [CrossRef]
- Liu, X.; Feng, C.; Zhang, M.; Yin, X.; Xu, C.; Chen, K. The MrWD40-1 gene of Chinese bayberry (Myrica rubra) interacts with MYB and bHLH to enhance anthocyanin accumulation. Plant Mol. Biol. Rep. 2013, 31, 1474–1484. [Google Scholar] [CrossRef]
- Zhu, Q.G.; Wang, M.M.; Gong, Z.Y.; Fang, F.; Sun, N.J.; Li, X.; Grierson, D.; Yin, X.R.; Chen, K.S. Involvement of DkTGA1 transcription factor in anaerobic response leading to persimmon fruit postharvest de-astringency. PLoS ONE 2016, 11, e0155916. [Google Scholar] [CrossRef]
- Wang, M.M.; Zhu, Q.G.; Deng, C.L.; Luo, Z.R.; Sun, N.J.; Grierson, D.; Yin, X.R.; Chen, K.S. Hypoxia-responsive ERF s involved in postdeastringency softening of persimmon fruit. Plant Biotechnol. J. 2017, 15, 1409–1419. [Google Scholar] [CrossRef]
- Yin, X.R.; Shi, Y.N.; Min, T.; Luo, Z.R.; Yao, Y.C.; Xu, Q.; Ferguson, I.; Chen, K.S. Expression of ethylene response genes during persimmon fruit astringency removal. Planta 2012, 235, 895–906. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamil, W.; Wu, W.; Gong, H.; Huang, J.-W.; Ahmad, M.; Zhu, Q.-G.; Jin, R.; Liu, X.-F.; Yin, X.-R. C2H2-Type Zinc Finger Proteins (DkZF1/2) Synergistically Control Persimmon Fruit Deastringency. Int. J. Mol. Sci. 2019, 20, 5611. https://doi.org/10.3390/ijms20225611
Jamil W, Wu W, Gong H, Huang J-W, Ahmad M, Zhu Q-G, Jin R, Liu X-F, Yin X-R. C2H2-Type Zinc Finger Proteins (DkZF1/2) Synergistically Control Persimmon Fruit Deastringency. International Journal of Molecular Sciences. 2019; 20(22):5611. https://doi.org/10.3390/ijms20225611
Chicago/Turabian StyleJamil, Wajeeha, Wei Wu, Hui Gong, Jing-Wen Huang, Mudassar Ahmad, Qing-Gang Zhu, Rong Jin, Xiao-Fen Liu, and Xue-Ren Yin. 2019. "C2H2-Type Zinc Finger Proteins (DkZF1/2) Synergistically Control Persimmon Fruit Deastringency" International Journal of Molecular Sciences 20, no. 22: 5611. https://doi.org/10.3390/ijms20225611
APA StyleJamil, W., Wu, W., Gong, H., Huang, J.-W., Ahmad, M., Zhu, Q.-G., Jin, R., Liu, X.-F., & Yin, X.-R. (2019). C2H2-Type Zinc Finger Proteins (DkZF1/2) Synergistically Control Persimmon Fruit Deastringency. International Journal of Molecular Sciences, 20(22), 5611. https://doi.org/10.3390/ijms20225611




