Comparative Proteomic and Morpho-Physiological Analyses of Maize Wild-Type Vp16 and Mutant vp16 Germinating Seed Responses to PEG-Induced Drought Stress
Abstract
1. Introduction
2. Results
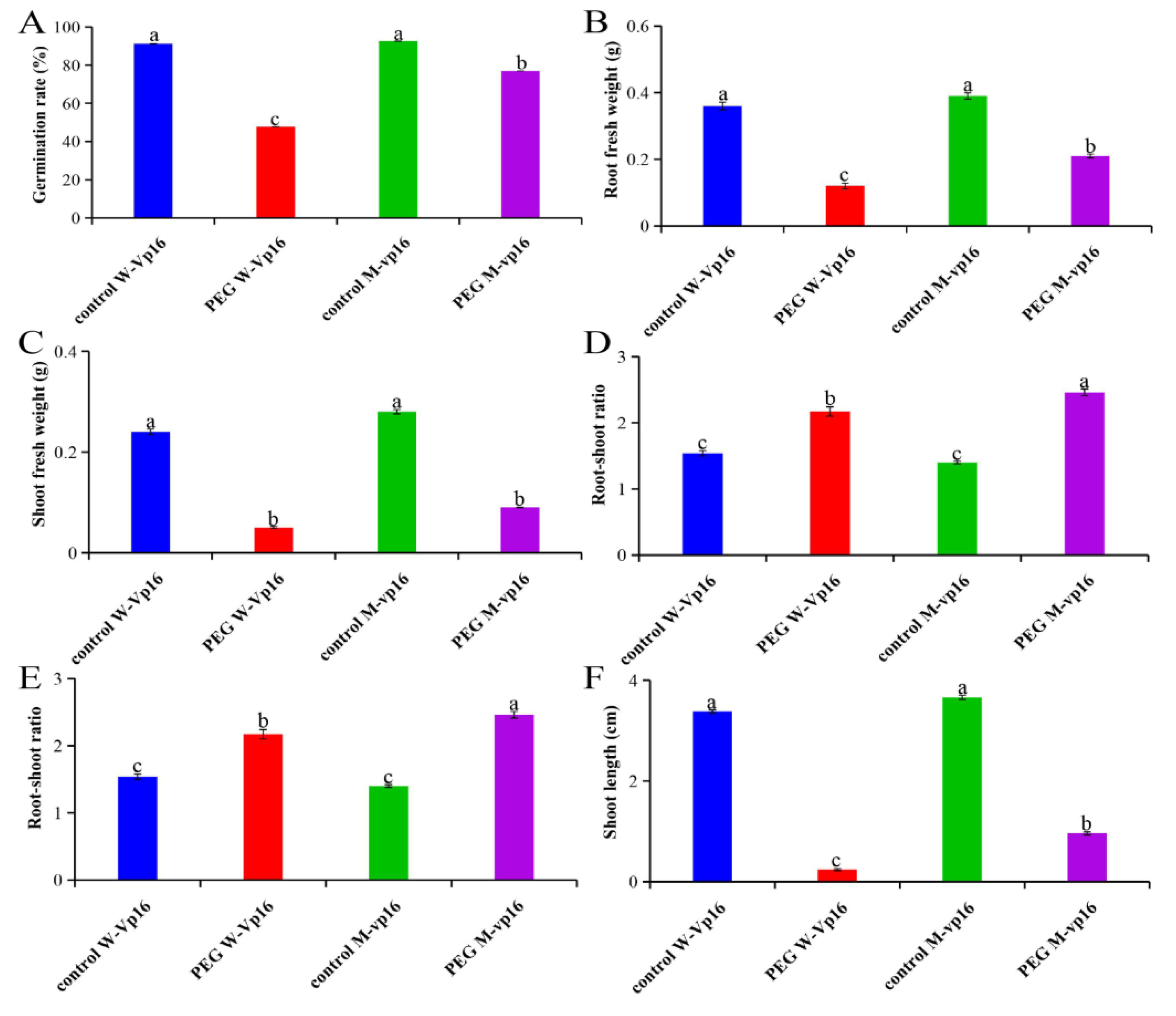
2.1. Morphological Responses of Wild-Type Vp16 and Mutant vp16 Maize Lines to Drought Stress
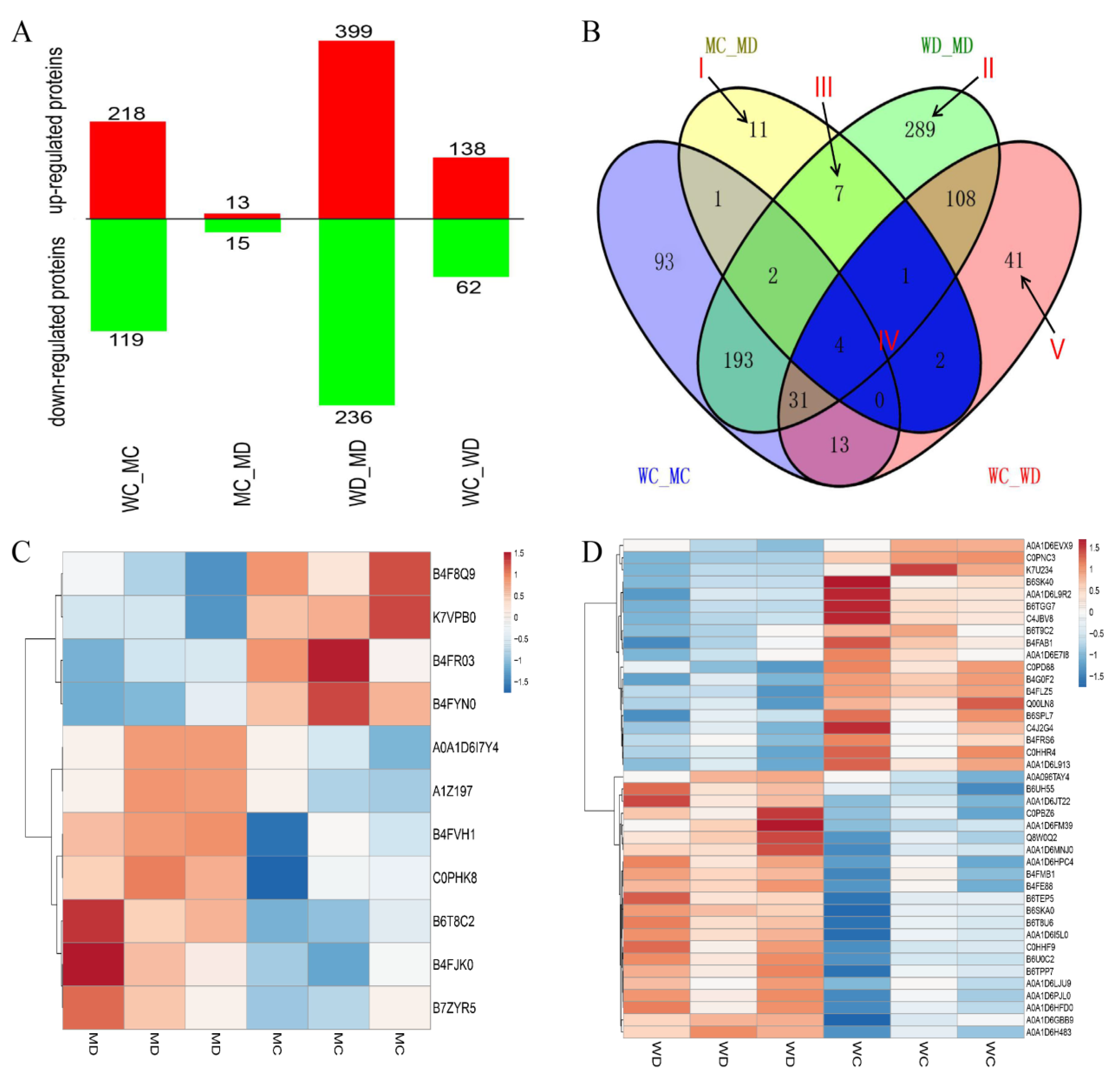
2.2. Summary Output Details of Maize Germinating Seed Proteins Identified by iTRAQ
2.3. Analysis of Differentially Abundant Proteins (DAPs) Identified in Different Comparisons
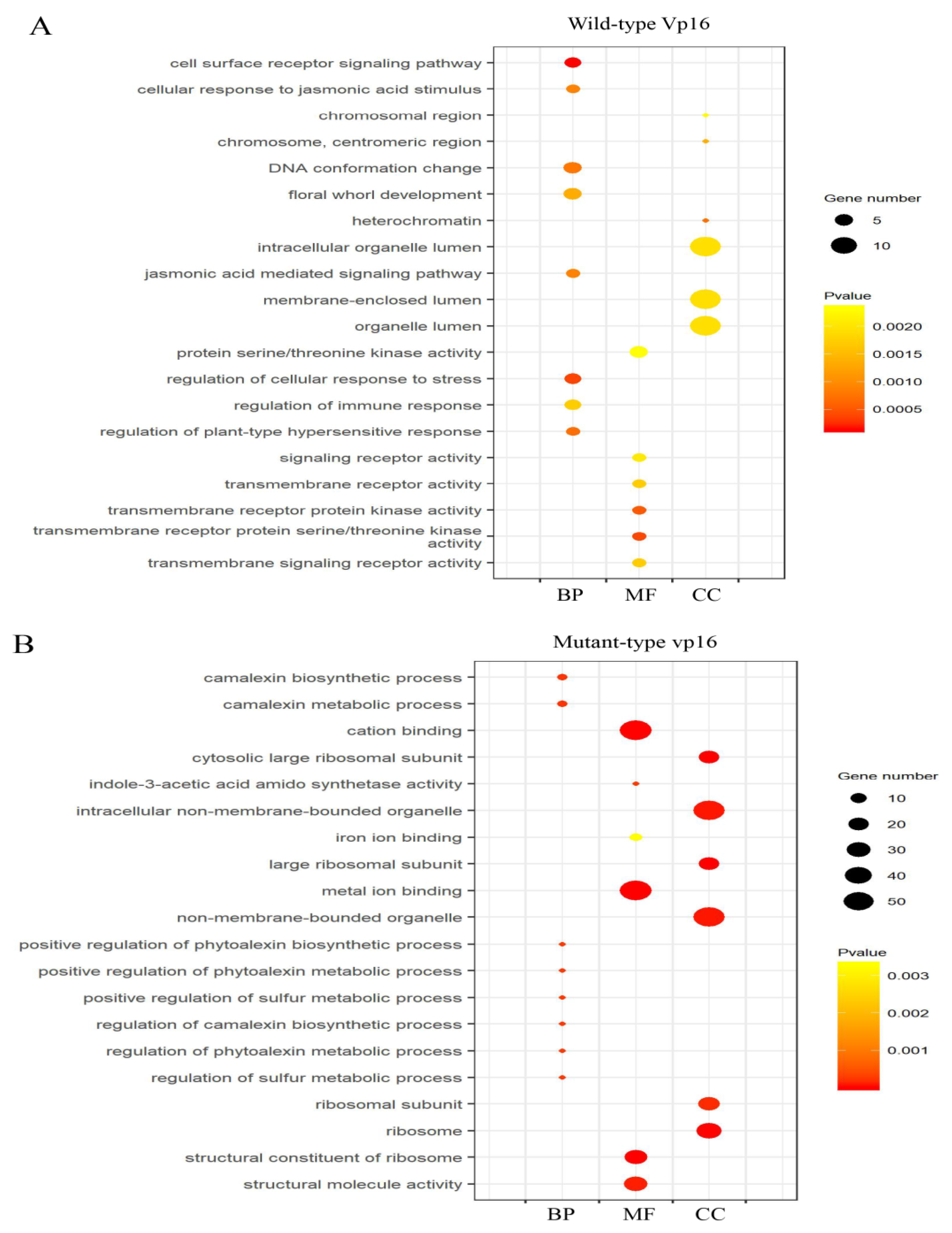
2.4. Gene Ontology (GO) Classification and Analysis of Drought-Responsive DAPs
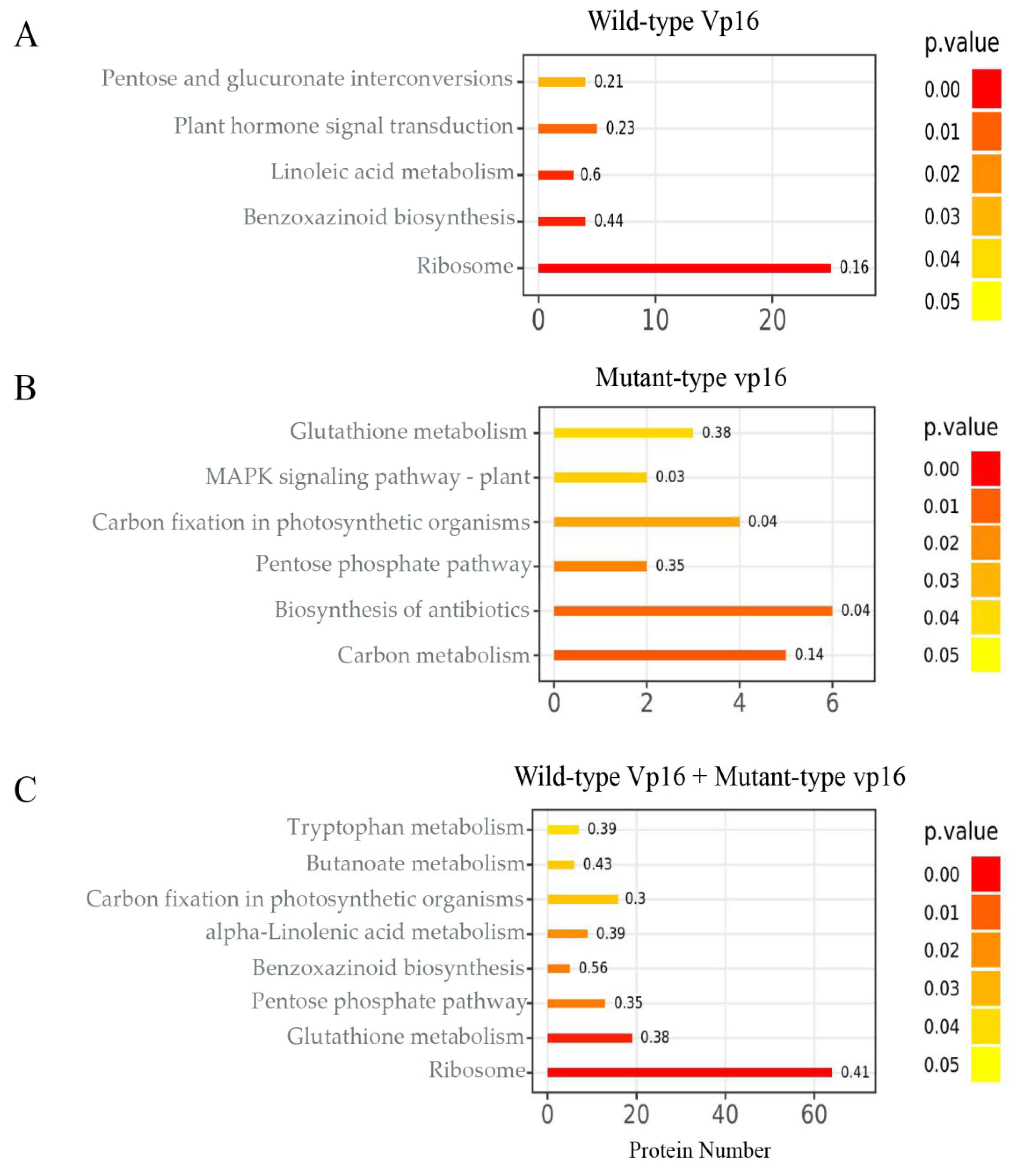
2.5. KEGG Pathway Enrichment Analysis of DAPs
2.6. Expression Levels of Genes Encoding DAPs in Response to Drought Stress
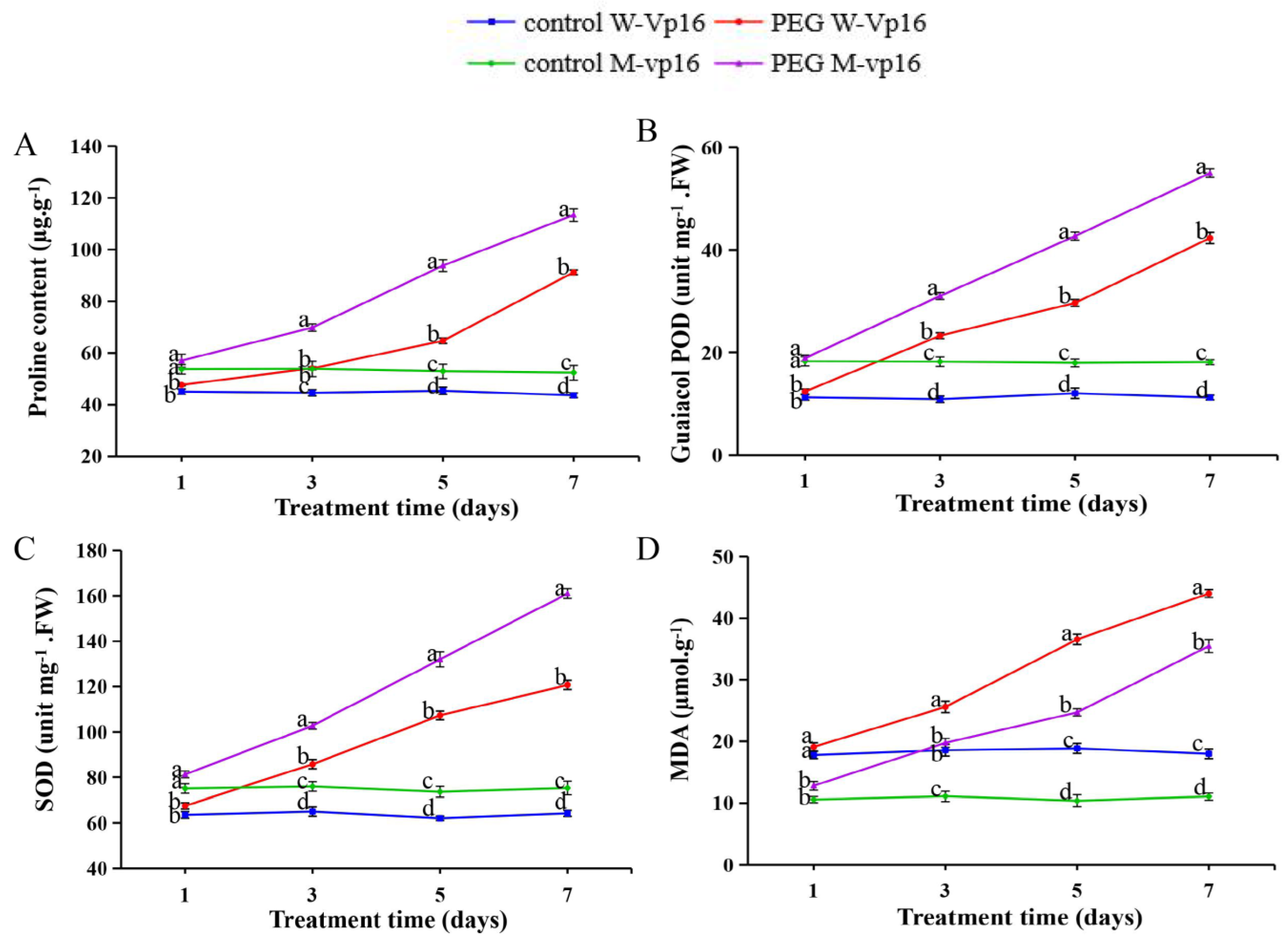
2.7. Physiological Responses of Wild-Type Vp16 and Mutant vp16 Maize Lines to Drought Stress
3. Discussion
3.1. Wild-Type Vp16 and Mutant vp16 Lines Showed Significant Variation in Their Morphological and Physiological Drought-Stress Responses
3.2. Carbohydrate/Energy Metabolism-Related Proteins under Drought
3.3. Histone H2A Are the Main Histone Proteins Responsive to Drought
3.4. Protein Synthesis and Proteolysis-Related Proteins under Drought Stress
3.5. Stress Signal Transduction and Lipid-Metabolism-Related Proteins
3.6. Cellular Redox Homeostasis and Stress-Related Proteins under Drought
3.7. Most Significantly Enriched Metabolic Pathways in Response to Drought
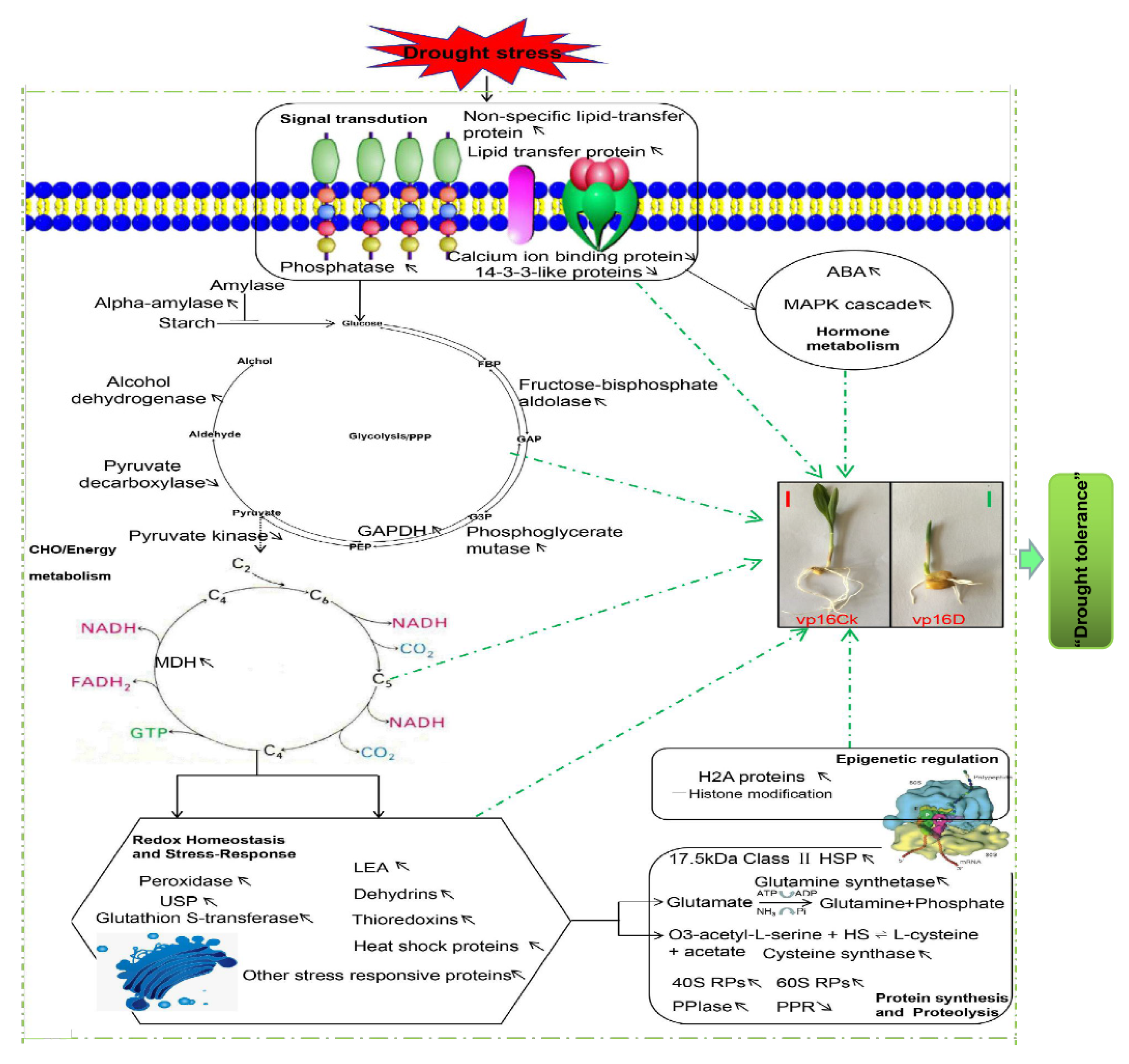
3.8. Proposed Hypothetical Model for Maize Germinating-Seed Drought Tolerance
4. Materials and Methods
4.1. Plant Materials and Drought-Stress Treatment
4.2. Growth Parameter Measurements and Physiological Assays
4.3. Protein Extraction
4.4. Protein Digestion and iTRAQ (Isobaric Tags for Relative and Absolute Quantification) Labeling
4.5. Strong Cation Exchange (SCX) and LC-MS/MS Analysis
4.6. Protein Identification and Quantification
4.7. Biological Function Classification, Pathway Enrichment and Hierarchal Clustering Analysis of DAPs
4.8. RNA Extraction, cDNA Synthesis and qRT-PCR Analysis
4.9. Statistical Analysis of Morpho-Physiological Data
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CDPKs | Calcium dependent protein kinases |
| DAPs | Differentially abundant proteins |
| GADPH | Glyceraldehyde-3-phosphate dehydrogenase |
| GO | Gene ontology |
| GSTs | Glutathione-S-transferases |
| HSPs | Heat shock proteins |
| iTRAQ | Isobaric tags for relative and absolute quantification |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| LC-MS/MS | Liquid chromatography-tandem mass spectrometry |
| MAPK | Mitogen-activated protein kinases |
| MDA | Malondialdehyde |
| MDH | Malate dehydrogenase |
| nsLTPs | Non-specific lipid transfer proteins |
| PEG | Polyethylene-glycol |
| POD | Guaiacol peroxidase |
| PPIase | Peptidylprolyl isomerase |
| qRT-PCR | Quantitative real-time polymerase chain reaction |
| RPs | Ribosomal proteins |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
References
- Al-Whaibi, M.H. Plant heat-shock proteins: A mini review. J. King Saud Univ. Sci. 2010, 23, 139–150. [Google Scholar] [CrossRef]
- Farooq, M.; Wahid, A.; Kobayashi, N.; Fujita, D.; Basra, S.M.A. Plant drought stress: Effects, mechanisms and management. Agron. Sustain. Dev. 2009, 29, 185–212. [Google Scholar] [CrossRef]
- Aslam, M.; Maqbool, M.A.; Cengiz, R. Drought stress in maize (Zea mays L.): Effects, resistance mechanisms, global achievements and biological strategies for improvement. In SpringerBriefs in Agriculture; Springer: Cham, Switzerland, 2015; ISBN 978-3-319-25440-1. [Google Scholar] [CrossRef]
- Miao, Z.; Han, Z.; Zhang, T.; Chen, S.; Ma, C. A systems approach to spatio-temporal understanding of the drought stress response in maize. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fahad, S.; Bajwa, A.A.; Nazir, U.; Anjum, S.A.; Farooq, A.; Zohaib, A.; Sadia, S.; Nasim, W.; Adkins, S.; Saud, S. Crop production under drought and heat Stress: Plant responses and management options. Front. Plant Sci. 2017, 8, 1147. [Google Scholar] [CrossRef]
- Feller, U.; Vaseva, I.I. Extreme climatic events: Impacts of drought and high temperature on physiological processes in agronomically important plants. Front. Environ. Sci. 2014, 2, 1–39. [Google Scholar] [CrossRef]
- F.A.O (Food and Agricultural Organization of the United Nations). High Level Expert Forum—How to Feed the World in 2050. 2009. Available online: www.fao.org/ (accessed on 23 June 2019).
- Nepolean, T.; Kaul, J.; Mukri, G. Genomics-enabled next-generation breeding approaches for developing system-specific drought tolerant hybrids in maize. Front. Plant Sci. 2018, 9, 361. [Google Scholar] [CrossRef]
- Thirunavukkarasu, N.; Sharma, R.; Singh, N.; Shiriga, K.; Mohan, S.; Mittal, S.; Mittal, S.; Mallikarjuna, M.G.; Rao, A.R.; Dash, P.K.; et al. Genomewide expression and functional interactions of genes under drought stress in maize. Int. J. Genom. 2017, 2568706. [Google Scholar] [CrossRef]
- Shiferaw, B.; Prasanna, B.M.; Hellin, J.; Bänziger, M. Crops that feed the world 6. Past successes and future challenges to the role played by maize in global food security. Food Secur. 2011, 3, 307–327. [Google Scholar] [CrossRef]
- Gong, F.; Yang, L.; Tai, F.; Hu, X.; Wang, W. “Omics” of maize stress response for sustainable food production: Opportunities and challenges. OMICS 2014, 18, 714–732. [Google Scholar] [CrossRef]
- Yin, X.G.; Jørgen, E.O.; Wang, M.; Kersebaum, K.-C.; Chen, H.; Baby, S.; Ozturk, I.; Chen, F. Adapting maize production to drought in the northeast farming region of China. Eur. J. Agron. 2016, 77, 47–58. [Google Scholar] [CrossRef]
- Kaya, M.D.; Okçu, G.; Atak, M.; Çıkılı, Y.; Kolsarıcı, Ö. Seed treatments to overcome salt and drought stress during germination in sunflower (Helianthus annuus L.). Eur. J. Agron. 2006, 24, 291–295. [Google Scholar] [CrossRef]
- Hussain, H.A.; Hussain, S.; Khaliq, A.; Ashraf, U.; Anjum, S.A.; Men, S.N.; Wang, L.C. Chilling and drought stresses in crop plants: Implications, cross talk, and potential management opportunities. Front. Plant Sci. 2018, 9, 393. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Ning, F.; Zhang, Q.; Wu, X.; Wang, W. Enhancing omics research of crop responses to drought under field conditions. Front. Plant Sci. 2017, 8, 174. [Google Scholar] [CrossRef] [PubMed]
- Khodarahmpour, Z. Evaluation of maize (Zea mays L.) hybrids, seed germination and seedling characters in water stress conditions. Afr. J. Agric. Res. 2017, 7, 6049–6053. [Google Scholar] [CrossRef]
- Álvarez-Iglesias, L.; de la Roza-Delgado, B.; Reigosa, M.J.; Revilla, P.; Pedrol, N. A simple, fast and accurate screening method to estimate maize (Zea mays L) tolerance to drought at early stages. Maydica 2017, 62, 1–12. [Google Scholar]
- Agrawal, L.; Gupta, S.; Mishra, S.K.; Pandey, G.; Kumar, S.; Chauhan, P.S.; Chakrabarty, D.; Nautiyal, C.S. Elucidation of complex nature of PEG induced drought-stress response in rice root using comparative proteomics approach. Front. Plant Sci. 2016, 7, 1466. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Yang, H.; Wang, W.; Wu, J.; Hu, X. Quantitative proteomic analyses identify aba-related proteins and signal pathways in maize leaves under drought conditions. Front. Plant Sci. 2016, 7, 1827. [Google Scholar] [CrossRef]
- Osmolovskaya, N.; Shumilina, J.; Kim, A.; Didio, A.; Grishina, T.; Bilova, T.; Keltsieva, O.A.; Zhukov, V.; Tikhonovich, I.; Tarakhovskaya, E.; et al. Methodology of drought stress research: Experimental setup and physiological characterization. Int. J. Mol. Sci. 2018, 19, 4089. [Google Scholar] [CrossRef]
- Wang, X.L.; Cai, X.F.; Xu, C.X.; Wang, Q.H.; Dai, S.J. Drought-responsive mechanisms in plant leaves revealed by proteomics. Int. J. Mol. Sci. 2016, 17, 1706. [Google Scholar] [CrossRef]
- Wahid, A.; Gelani, S.; Ashraf, M.; Foolad, M.R. Heat tolerance in plants: An overview. Environ. Exp. Bot. 2007, 61, 199–223. [Google Scholar] [CrossRef]
- Shan, X.; Li, Y.; Jiang, Y.; Jiang, Z.; Hao, W.; Yuan, Y. Transcriptome profile analysis of maize seedlings in response to high-salinity, drought and cold stresses by deep sequencing. Plant Mol. Biol. Rep. 2013, 31, 1485–1491. [Google Scholar] [CrossRef]
- Min, H.; Chen, C.; Wei, S.; Shang, X.; Sun, M.; Xia, R.; Liu, X.G.; Hao, D.Y.; Chen, H.B.; Xie, Q. Identification of drought tolerant mechanisms in maize seedlings based on transcriptome analysis of recombination inbred lines. Front. Plant Sci. 2016, 7, 1080. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhang, H.; Wang, T.; Chen, S.; Dai, S. Proteomics-based investigation of salt-responsive mechanisms in plant roots. J. Proteom. 2013, 82, 230–253. [Google Scholar] [CrossRef]
- Kosova, K.; Vitamvas, P.; Prasil, I.T.; Renaut, J. Plant proteome changes under abiotic stress- contribution of proteomics studies to understanding plant stress response. J. Proteom. 2011, 74, 1301–1322. [Google Scholar] [CrossRef] [PubMed]
- Kamel, C.; Sonia, A.R.; Claudette, J.; Dominique, J. Proteomic analysis of seed dormancy in Arabidopsis. Physiol. Plant 2006, 142, 1493–1510. [Google Scholar] [CrossRef]
- Zhang, H.; Lian, C.; Shen, Z. Proteomic identification of small, copper-responsive proteins in germinating embryos of Oryza sativa. Ann. Bot. 2009, 103, 923–930. [Google Scholar] [CrossRef] [PubMed]
- ProtTech. Two Different Methods in Protein Identification by Mass Spectrometry. ProtTech. Inc.. 2010. Available online: http://www.prottech.com/ (accessed on 10 February 2019).
- Wu, X.; Wang, W. Increasing confidence of proteomics data regarding the identification of stress-responsive proteins in crop plants. Front. Plant Sci. 2016, 7, 702. [Google Scholar] [CrossRef]
- Kamal, A.H.M.; Cho, K.; Choi, J.S.; Jin, Y.; Park, C.S.; Lee, J.S.; Woo, S.H. Patterns of protein expression in water-stressed wheat chloroplasts. Biol. Plant. 2013, 57, 305–312. [Google Scholar] [CrossRef]
- Cheng, L.; Gao, X.; Li, S.; Shi, M.; Javeed, H.; Jing, X.; Yang, G.; He, G. Proteomic analysis of soybean [Glycine max (L.) Meer.] seeds during imbibition at chilling temperature. Mol. Breed. 2010, 26, 1–17. [Google Scholar] [CrossRef]
- Zheng, J.; Fu, J.; Gou, M.; Huai, J.; Liu, Y.; Jian, M.; Huang, Q.; Guo, X.; Dong, Z.; Wang, H.; et al. Genome-wide transcriptome analysis of two maize inbred lines under drought stress. Plant Mol. Biol. 2010, 72, 407–421. [Google Scholar] [CrossRef]
- Anjum, S.A.; Xie, X.Y.; Wang, L.C.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar]
- Khodarahmpour, Z.; Motamedi, M. Evaluation of drought and salinity stress effects on germination and early growth of two cultivars of maize (Zea mays L.). Afr. J. Biotechnol. 2011, 10, 14868–14872. [Google Scholar] [CrossRef]
- Westgate, M.E.; Boyer, J.S. Osmotic adjustment and the inhibition of leaf, root, stem and silk growth at low water potentials in maize. Planta 1985, 164, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Moussa, H.R.; Abdel-Aziz, S.M. Comparative response of drought tolerant and drought sensitive maize genotypes to water stress. Aust. J. Crop Sci. 2008, 1, 31–36. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 217037. [Google Scholar] [CrossRef]
- Fang, Y.; Xiong, L. General mechanisms of drought response and their application in drought resistance improvement in plants. Cell. Mol. Life Sci. 2015, 72, 673–689. [Google Scholar] [CrossRef]
- Wang, T.; Chen, X.; Zhu, F.; Li, F.; Li, L.; Yang, Q.; Chi, X.; Yu, S. Characterization of peanut germin-like proteins, AhGLPs in plant development and defense. PLoS ONE 2013, 8, e61722. [Google Scholar] [CrossRef]
- Caruso, G.; Cavaliere, C.; Foglia, P.; Gubbiotti, R.; Samperi, R.; Laganà, A. Analysis of drought responsive proteins in wheat (Triticum durum) by 2D-PAGE and MALDI-TOF mass spectrometry. Plant Sci. 2009, 6, 570–576. [Google Scholar] [CrossRef]
- Salekdeh, G.H.; Siopongco, J.; Wade, L.J.; Ghareyazie, B.; Bennett, J. Proteomic analysis of rice leaves during drought stress and recovery. Proteomics 2002, 2, 1131–1145. [Google Scholar] [CrossRef]
- Urban, M.O.; Vasek, J.; Klima, M.; Krtkova, J.; Kosova, K.; Prasil, I.T.; Vitamvas, P. Proteomic and physiological approach reveals drought- induced changes in rapeseeds: Water-saver and water- spender strategy. J. Proteom. 2016, 152, 188–205. [Google Scholar] [CrossRef] [PubMed]
- Vitamvas, P.; Urban, M.O.; Škodáček, K.; Kosova, K.; Pitelkova, I.; Vitamvas, J.; Renaut, J.; Prášil, I.T. Quantitative analysis of proteome extracted from barely crowns grown under different drought condition. Front. Plant Sci. 2015, 6, 479. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lei, L.; Lai, J.; Zhao, H.; Song, W. Effects of drought stress and water recovery on physiological responses and gene expression in maize seedlings. BMC Plant Biol. 2018, 18, 68. [Google Scholar] [CrossRef] [PubMed]
- Zenda, T.; Liu, S.; Wang, X.; Liu, G.; Jin, H.; Dong, A.; Yang, Y.; Duan, H. Key maize drought-responsive genes and pathways revealed by comparative transcriptome and physiological analyses of contrasting inbred lines. Int. J. Mol. Sci. 2019, 20, 1268. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Sasaki, T.; Ueda, M.; Sako, K.; Seki, M. Chromatin changes in response to drought, salinity, heat, and cold stresses in plants. Front. Plant Sci. 2015, 6, 114. [Google Scholar] [CrossRef] [PubMed]
- Mozgova, I.; Mikulski, P.; Pecinka, A.; Farrona, S. Epigenetic mechanisms of abiotic stress response and memory in plants. Epigenet. Plants Agron. Import. Fundam. Appl. 2019, 1–64. [Google Scholar] [CrossRef]
- Yuan, L.; Liu, X.; Luo, M.; Yang, S.; Wu, K. Involvement of histone modifications in plant abiotic stress responses. J. Integr. Plant Biol. 2013, 55, 892–901. [Google Scholar] [CrossRef]
- Luo, F.; Deng, X.; Liu, Y.; Yan, Y. Identification of phosphorylation proteins in response to water deficit during wheat flag leaf and grain development. Bot. Stud. 2018, 59, 28–35. [Google Scholar] [CrossRef]
- Kim, Y.E.; Hipp, M.S.; Bracher, A.; Hayer-Hartl, M.; Ulrich Hartl, F. Molecular chaperone functions in protein folding and proteostasis. Annu. Rev. Biochem. 2013, 82, 323–355. [Google Scholar] [CrossRef]
- Fotovat, R.; Alikhani, M.; Valizadeh, M.; Mirzaei, M.; Salekdeh, G.H. A proteomics approach to Discover drought tolerance proteins in wheat pollen grain at meiosis stage. Protein Pept. Lett. 2017, 24, 26–36. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, Y.; Zhang, J.; Shi, W.; Qian, C.; Peng, X. Identification of aluminum-responsive proteins in rice roots by a proteomic approach: Cysteine synthase as a key player in Al response. Proteomics 2007, 7, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Singh, S.; Singh, H.; Chawla, M.; Dutta, T.; Kaur, H.; Bender, K.; Snedden, W.A.; Kapoor, S.; Pareek, A. Characterization of peptidyl-prolyl cis-trans isomerase- and calmodulin-binding activity of a cytosolic Arabidopsis thaliana cyclophilin AtCyp19-3. PLoS ONE 2015, 10, e0136692. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.G.; Lee, J.S.; Kim, J.T.; Kwon, Y.S.; Bae, D.W.; Bae, H.H.; Son, B.Y.; Baek, S.B.; Kwon, Y.U.; Woo, M.O.; et al. Physiological and proteomic analysis of the response to drought stress in an inbred Korean maize line. Plant Omics 2015, 8, 159–168. [Google Scholar] [CrossRef]
- Sharma, A.D.; Singh, P. Comparative studies on drought-induced changes in peptidyl prolyl cis-trans isomerase activity in drought-tolerant and susceptible cultivars of Sorghum bicolor. Curr. Sci. 2003, 84, 911–918. [Google Scholar] [CrossRef]
- Zhu, J.K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Cramer, G.R.; Urano, K.; Delrot, S.; Pezzotti, M.; Shinozaki, K. Effects of abiotic stress on plants: A systems biology perspective. Biogeochemistry 2011, 11, 163. [Google Scholar] [CrossRef] [PubMed]
- Jonak, C.; Kiegerl, S.; Ligterink, W.; Barker, P.J.; Huskisson, N.S.; Hirt, H. Stress signaling in plants: A mitogen-activated protein kinase pathway is activated by cold and drought. Proc. Natl. Acad. Sci. USA 1996, 93, 11274–11279. [Google Scholar] [CrossRef]
- Edstam, M.M.; Viitanen, L.; Salminen, T.A.; Edqvist, J. Evolutionary history of the non-specific lipid transfer proteins. Mol. Plant 2011, 4, 947–964. [Google Scholar] [CrossRef]
- Molina, A.; Segura, A.; García-Olmedo, F. Lipid transfer proteins (nsLTPs) from barley and maize leaves are potent inhibitors of bacterial and fungal plant pathogens. FEBS Lett. 1993, 316, 119–122. [Google Scholar] [CrossRef]
- Zenda, T.; Liu, S.; Wang, X.; Jin, H.; Liu, G.; Duan, H. Comparative proteomic and physiological analyses of two divergent maize inbred lines provide more insights into drought-stress tolerance mechanisms. Int. J. Mol. Sci. 2018, 19, 3225. [Google Scholar] [CrossRef]
- Pitzschke, A.; Forzani, C.; Hirt, H. Reactive oxygen species signaling in plants. Antioxid. Redox Signal. 2006, 8, 1757–1764. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Harb, A. Identification of candidate genes for drought stress tolerance. Drought Stress Toler. Plants 2016, 2, 385–414. [Google Scholar] [CrossRef]
- Schloß, P.; Walter, C.; Mäder, M. Basic peroxidases in isolated vacuoles of nicotiana tabacum L. Planta 1987, 170, 225–229. [Google Scholar] [CrossRef]
- Faghani, E.; Gharechahi, J.; Komatsu, S.; Mirzaei, M.; Khavarinejad, R.A.; Najafi, F.; Farsad, L.K.; Salekdeh, G.H. Comparative physiology and proteomic analysis of two wheat genotypes contrasting in drought tolerance. J. Proteom. 2015, 114, 1–15. [Google Scholar] [CrossRef]
- Benešová, M.; Holá, D.; Fischer, L.; Jedelský, P.L.; Hnilička, F.; Wilhelmová, N.; Rothová, O.; Kočová, M.; Procházková, D.; Honnerová, J.; et al. The physiology and proteomics of drought tolerance in maize: Early stomatal closure as a cause of lower tolerance to short-term dehydration? PLoS ONE 2012, 7, e38017. [Google Scholar] [CrossRef]
- Xu, C.P.; Huang, B. Comparative analysis of drought responsive proteins in kentucky bluegrass cultivars contrasting in drought tolerance. Crop Sci. 2010, 50, 2543–2552. [Google Scholar] [CrossRef]
- Zang, X.; Komatsu, S. A proteomics approach for identifying osmotic-stress-related proteins in rice. Phytochemistry 2007, 68, 426–437. [Google Scholar] [CrossRef]
- Ji, W.; Zhu, Y.M.; Li, Y.; Yang, L.A.; Zhao, X.W.; Cai, H.; Bai, X. Over-expression of a glutathione S-transferase gene, GsGST, from wild soybean (Glycine soja) enhances drought and salt tolerance in transgenic tobacco. Biotechnol. Lett. 2010, 32, 1173–1179. [Google Scholar] [CrossRef]
- Waters, E.R.; Lee, G.J.; Vierling, E. Evolution, structure and function of the small heat shock proteins in plants. J. Exp. Bot. 1996, 47, 325–338. [Google Scholar] [CrossRef]
- Meyer, Y.; Siala, W.; Bashandy, T.; Riondet, C.; Vignols, F.; Reichheld, J.P. Glutaredoxins and thioredoxins in plants. Biochem. Biophys. Acta 2008, 1783, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Campbell, S.A.; Close, T.J. Dehydrins: Genes, proteins, and associations with phenotypic traits. New Phytol. 1997, 137, 61–74. [Google Scholar] [CrossRef]
- Hanin, M.; Brini, F.; Ebel, C.; Toda, Y.; Takeda, S.; Masmoudi, K. Plant dehydrins and stress tolerance versatile proteins for complex mechanisms. Plant Signal. Behav. 2011, 6, 1503–1509. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Zhao, Y.; Wang, Y.; Shi, Z.; Zhang, P.; Zhang, Y.; Song, W.; Zhao, J. Comparative proteomics of contrasting maize genotypes provides insights into salt-stress tolerance mechanisms. J. Proteome Res. 2018, 17, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Bai, X.; Jiang, C.; Li, Z. Phosphoproteomic analysis of two contrasting maize inbred lines provides insights into the mechanism of salt-stress tolerance. Int. J. Mol. Sci. 2019, 20, 1886. [Google Scholar] [CrossRef]
- Szalai, G.; Kellős, T.; Galiba, G.; Kocsy, G. Glutathione as an antioxidant and regulatory molecule in plants under abiotic stress conditions. J. Plant Growth Regul. 2009, 28, 66–80. [Google Scholar] [CrossRef]
- Zeng, W.; Peng, Y.; Zhao, X.; Wu, B.; Chen, F.; Ren, B.; Zhuang, Z.; Gao, Q.; Ding, Y. Comparative proteomics analysis of the seedling root response of drought-sensitive and drought-tolerant maize varieties to drought stress. Int. J. Mol. Sci. 2019, 20, 2793. [Google Scholar] [CrossRef]
- Yang, W.F.; Tian, Y.H.; Wang, T.T.; Wang, R.N.; Tao, Y.S. Isolating and confirming the MuDR-inserted flanking sequences of maize. Cytol. Genet. 2017, 51, 142–148. [Google Scholar] [CrossRef]
- Bates, T.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Han, L.B.; Song, G.L.; Zhang, X. Preliminary observation of physiological responses of three turfgrass species to traffic stress. HortTechnology 2008, 18, 139–143. [Google Scholar] [CrossRef]
- Ma, Q.L.; Kang, J.M.; Long, R.C.; Cui, Y.J.; Zhang, T.J.; Xiong, J.B.; Yang, Q.C.; Sun, Y. Proteomic analysis of salt and osmotic-drought stress in alfalfa seedlings. J. Integr. Agric. 2016, 15, 2266–2278. [Google Scholar] [CrossRef]
- Dhindsa, R.S.; Plumb-Dhindsa, P.; Thorpe, T.A. Leaf senescence: Correlated with increased leaves of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J. Exp. Bot. 1981, 32, 93–101. [Google Scholar] [CrossRef]
- Wisniewski, J.R.; Zougman, A.; Nagaraj, N.; Mann, M. Universal sample preparation method for proteome analysis. Nat. Methods 2009, 6, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Shi, S. Physiological and proteomic responses of contrasting alfalfa (Medicago sativa L.) varieties to PEG-Induced osmotic stress. Front. Plant Sci. 2018, 9, 242. [Google Scholar] [CrossRef] [PubMed]
- Swägger, H. Tricine-SDS-PAGE. Nat. Protoc. 2006, 1, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Method 2016, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
| No | Accession 1 | Gene Name/ID 2 | Annotation 3 | Cover. (%) 4 | Pept. 5 | Fold Change 6 | p-Value 7 | KEGG Pathways 8 |
|---|---|---|---|---|---|---|---|---|
| 1 | B6T8C2 | Zm00001d006547 | Histone H2A | 36.08 | 2 | 1.81 | 7.33 × 10−3 | |
| 2 | B4FJK0 | 100216750 | Histone H2A | 33.75 | 2 | 1.73 | 4.53 × 10−2 | |
| 3 | A0A1D6I7Y4 | 100282031 | Peptidylprolyl isomerase | 35.29 | 2 | 1.32 | 2.11 × 10−2 | |
| 4 | A1Z197 | Zm00001d024755 | Uncharacterized protein | 12.71 | 3 | 1.25 | 5.05 × 10−4 | Plant–pathogen interaction |
| 5 | B4FVH1 | Zm00001d009640 | Malate dehydrogenase | 69.41 | 5 | 1.23 | 2.66 × 10−2 | Glyoxylate and dicarboxylate metabolism |
| 6 | B7ZYR5 | Zm00001d044434 | Uncharacterized protein | 9.49 | 3 | 1.22 | 2.88 × 10−2 | |
| 7 | C0PHK8 | Zm00001d018529 | Uncharacterized protein | 15.73 | 6 | 1.20 | 4.66 × 10−2 | |
| 8 | B4FR03 | 100272828 | Nucleic acid-binding OB-fold-like protein | 48.63 | 5 | 0.83 | 2.97 × 10−2 | Homologous recombination |
| 9 | B4FYN0 | 100285351 | Mitochondrial import inner membrane translocase subunit TIM13 | 28.74 | 2 | 0.83 | 6.11 × 10−3 | |
| 10 | B4F8Q9 | 100191413 | Calcium ion binding | 3.14 | 2 | 0.80 | 2.69 × 10−2 | |
| 11 | K7VPB0 | 103636586 | Pentatricopeptide repeat-containing protein mitochondrial | 7.06 | 3 | 0.73 | 6.22 × 10−3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, S.; Zenda, T.; Dong, A.; Yang, Y.; Liu, X.; Wang, Y.; Li, J.; Tao, Y.; Duan, H. Comparative Proteomic and Morpho-Physiological Analyses of Maize Wild-Type Vp16 and Mutant vp16 Germinating Seed Responses to PEG-Induced Drought Stress. Int. J. Mol. Sci. 2019, 20, 5586. https://doi.org/10.3390/ijms20225586
Liu S, Zenda T, Dong A, Yang Y, Liu X, Wang Y, Li J, Tao Y, Duan H. Comparative Proteomic and Morpho-Physiological Analyses of Maize Wild-Type Vp16 and Mutant vp16 Germinating Seed Responses to PEG-Induced Drought Stress. International Journal of Molecular Sciences. 2019; 20(22):5586. https://doi.org/10.3390/ijms20225586
Chicago/Turabian StyleLiu, Songtao, Tinashe Zenda, Anyi Dong, Yatong Yang, Xinyue Liu, Yafei Wang, Jiao Li, Yongsheng Tao, and Huijun Duan. 2019. "Comparative Proteomic and Morpho-Physiological Analyses of Maize Wild-Type Vp16 and Mutant vp16 Germinating Seed Responses to PEG-Induced Drought Stress" International Journal of Molecular Sciences 20, no. 22: 5586. https://doi.org/10.3390/ijms20225586
APA StyleLiu, S., Zenda, T., Dong, A., Yang, Y., Liu, X., Wang, Y., Li, J., Tao, Y., & Duan, H. (2019). Comparative Proteomic and Morpho-Physiological Analyses of Maize Wild-Type Vp16 and Mutant vp16 Germinating Seed Responses to PEG-Induced Drought Stress. International Journal of Molecular Sciences, 20(22), 5586. https://doi.org/10.3390/ijms20225586






