Identification of Novel 1,3,5-Triphenylbenzene Derivative Compounds as Inhibitors of Hen Lysozyme Amyloid Fibril Formation
Abstract
1. Introduction
2. Results
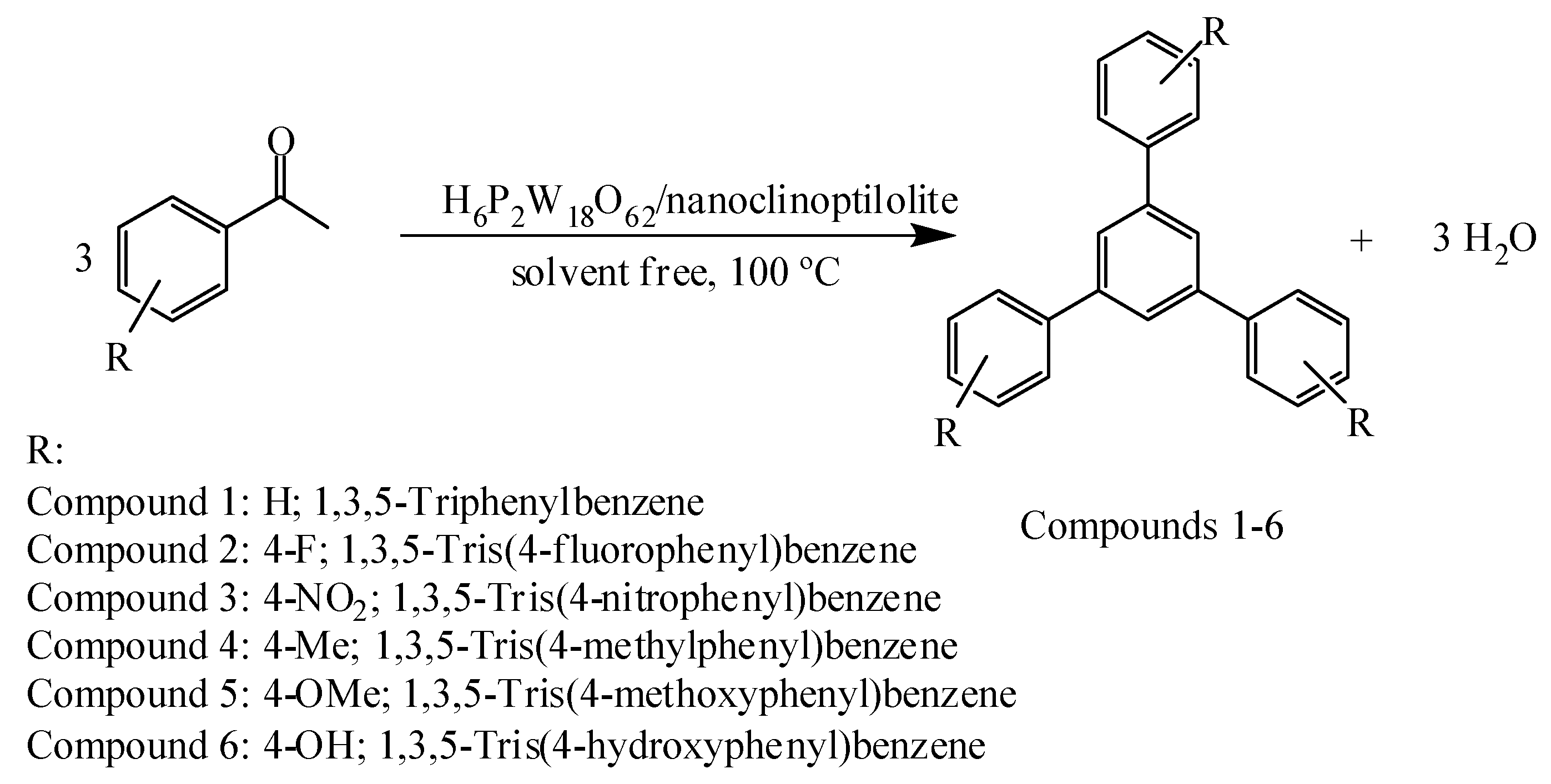
2.1. Chemistry of the Compounds
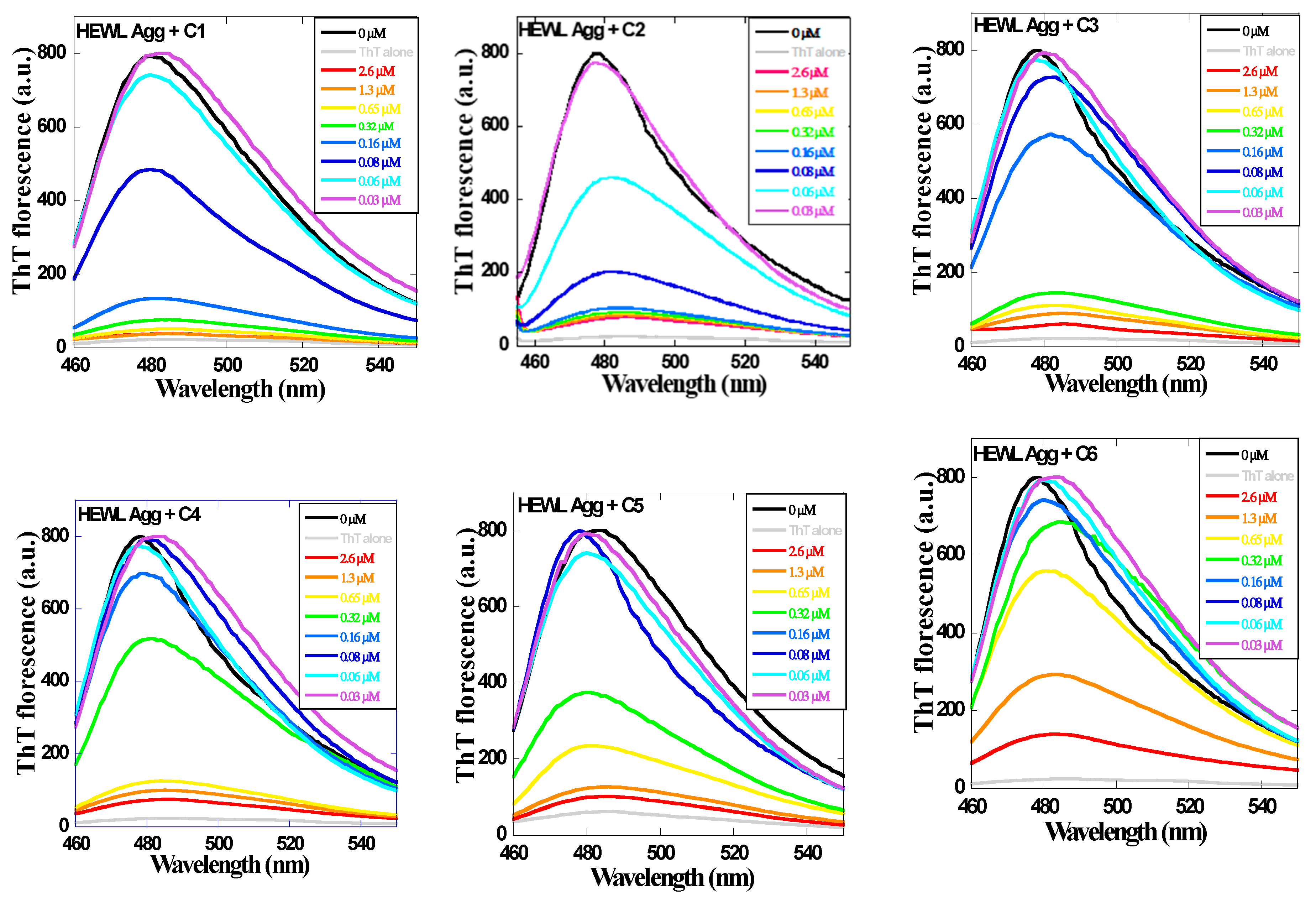
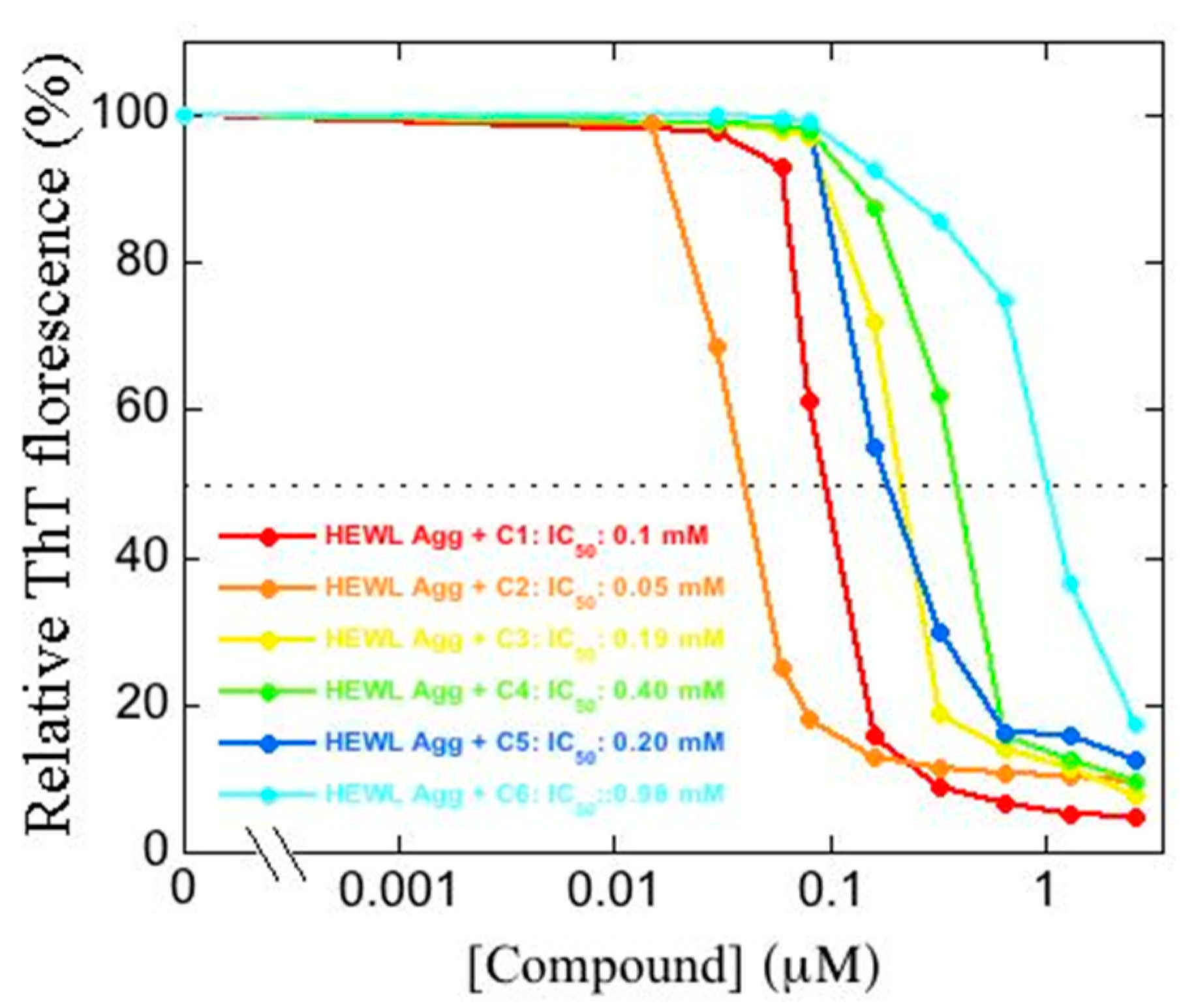
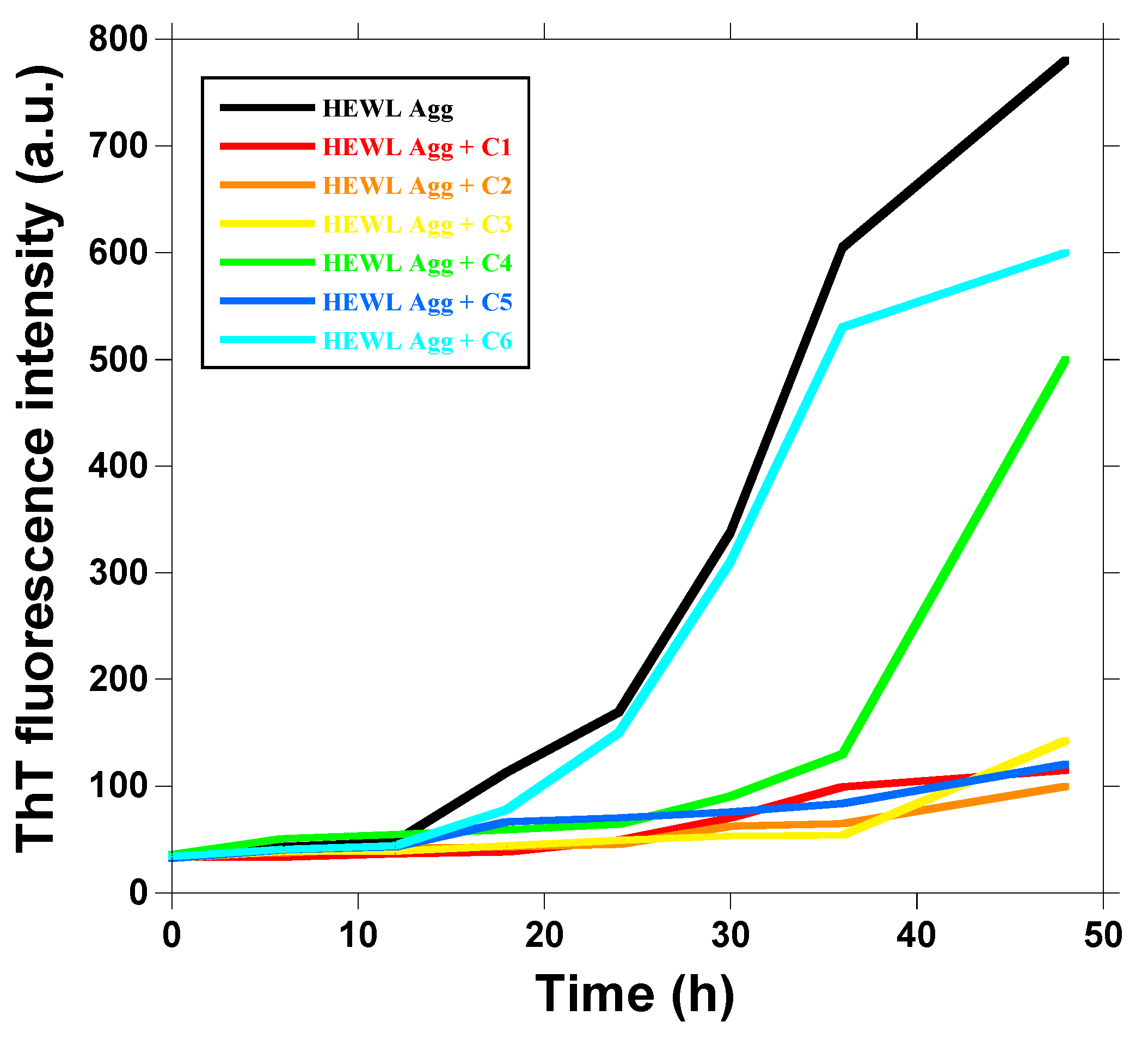
2.2. Effect of 1,3,5-Triarylbenzenes and Their Derivatives on Heat-Induced Fibrillation of HEWL Monitored with ThT Fluorescence
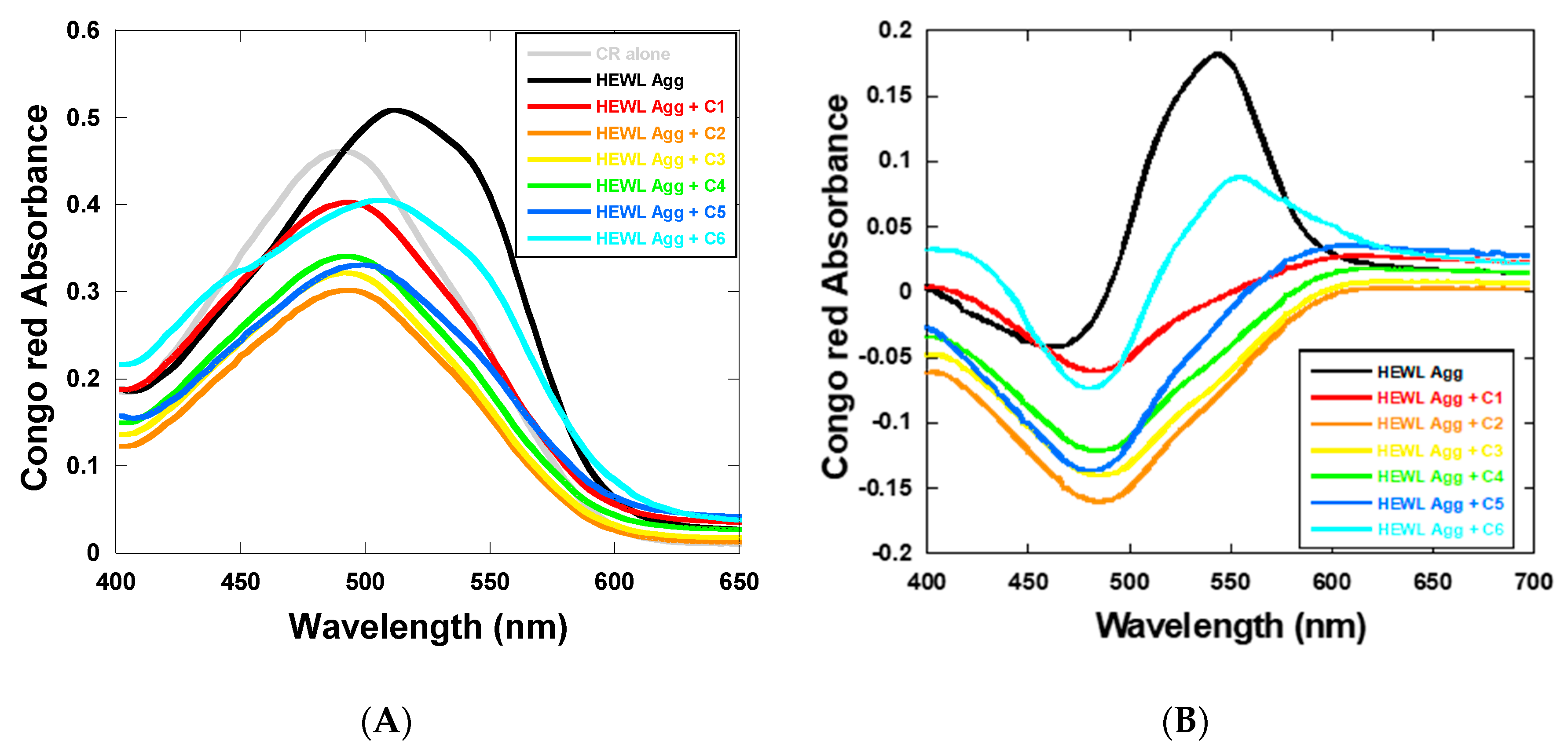
2.3. Effect of 1,3,5-Triarylbenzenes and Their Derivatives on Heat-Induced Fibrillation of HEWL Monitored with CR Assay
2.4. Effect of 1,3,5-Triarylbenzenes and Their Derivatives on Heat-Induced Fibrillation of HEWL Monitored with FTIR Spectroscopy
2.5. Effect of the Compounds on the Kinetic of HEWL Aggregation
2.6. Effect of the Compounds on HEWL Aggregation Morphology
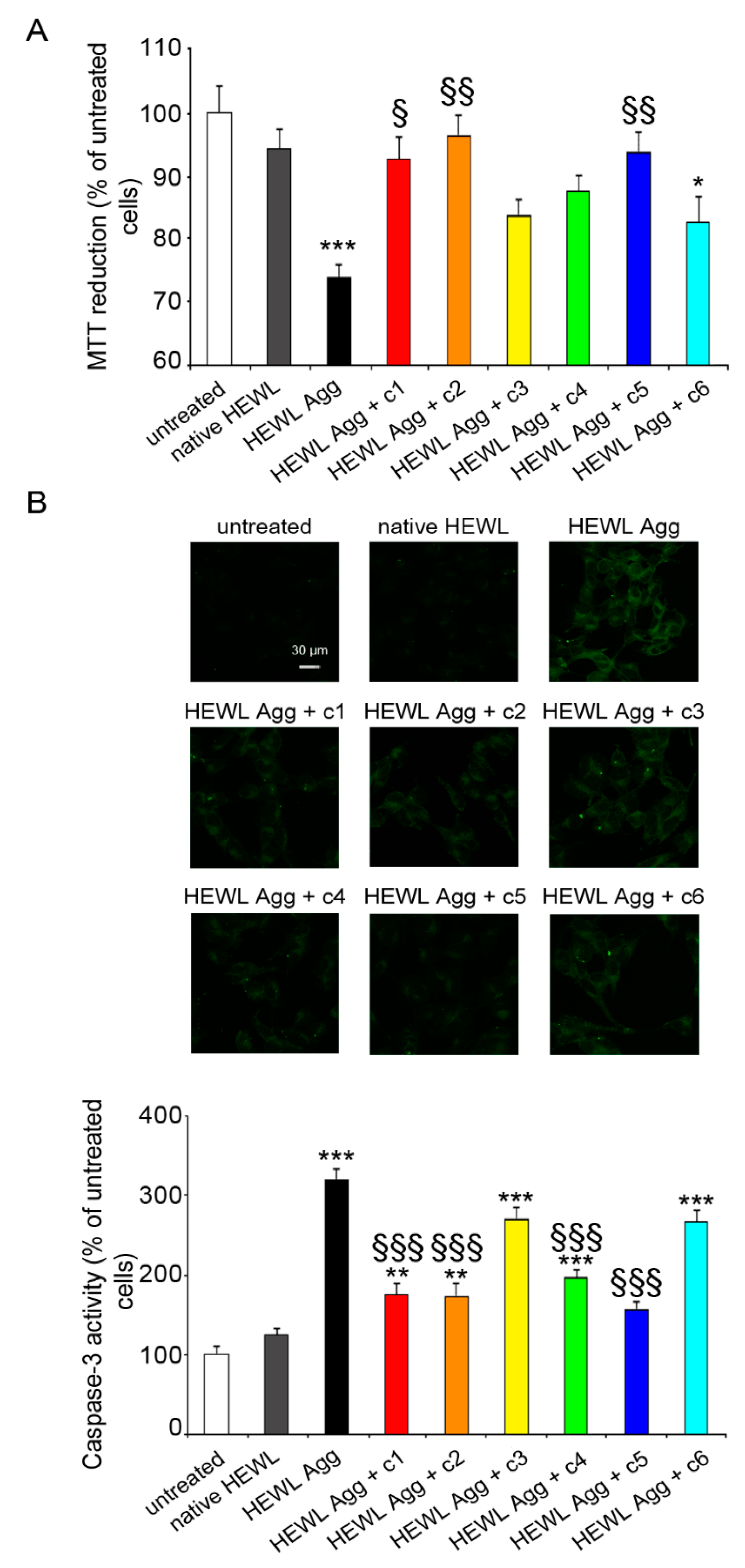
2.7. Effect of the Compounds on HEWL Amyloid Induced Cytotoxicity
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. General Chemistry
General Procedure for the Conversion of Acetophenone into 1,3,5-Triphenylbenzene
1,3,5-Triphenylbenzene (Compound 1)
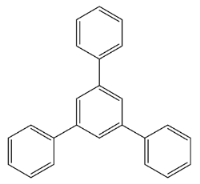
1,3,5-Tris(4-Fluorophenyl)Benzene (Compound 2)
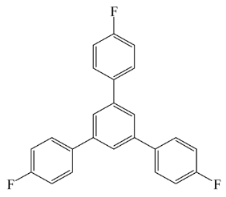
1,3,5-Tris(4-Nitrophenyl)Benzene (Compound 3)
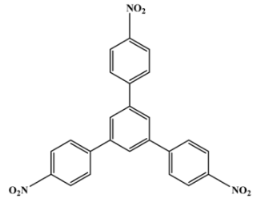
1,3,5-Tris(4-Methylphenyl)Benzene (Compound 4)

1,3,5-Tris(4-Methoxyphenyl)Benzene (Compound 5)
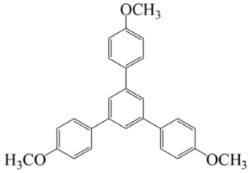
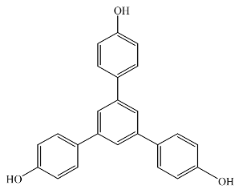
1,3,5-Tris(4-Hydroxyphenyl)Benzene (Compound 6)
4.3. Protein Aggregation
4.4. ThT Fluorescence Assay
4.5. CR Assay
4.6. FTIR Spectroscopy
4.7. Atomic Force Microscopy
4.8. Cell Cultures
4.9. 3-(4,5-Dimethylthiazol)-2,5-Diphenyltetrazolium Bromide (MTT) Reduction Test
4.10. Measurement of Caspase-3 Activity
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| HEWL | hen egg white lysozyme |
| ThT | thioflavin T |
| HPA/NCP | H6P2W18O62/nanoclinoptilolite |
| CR | Congo red |
| AFM | atomic force microscopy |
| FTIR | Fourier-transform infrared spectroscopy |
| MTT | 3-(4,5-dimethylthiazol)-2,5-diphenyltetrazolium bromide |
| NMR | nuclear magnetic resonance |
| TLC | thin layer chromatography |
| DMSO | dimethyl sulfoxide |
| mp | melting point |
| DMEM | Dulbecco’s modified Eagle’s medium |
| PBS | phosphate buffered saline |
References
- Chiti, F.; Dobson, C.M. Protein Misfolding, Amyloid Formation, and Human Disease: A Summary of Progress Over the Last Decade. Annu. Rev. Biochem. 2017, 86, 27–68. [Google Scholar] [CrossRef] [PubMed]
- Sipe, J.D.; Benson, M.D.; Buxbaum, J.N.; Ikeda, S.-I.; Merlini, G.; Saraiva, M.J.M.; Westermark, P. Amyloid fibril proteins and amyloidosis: Chemical identification and clinical classification International Society of Amyloidosis 2016 Nomenclature Guidelines. Amyloid 2016, 23, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Sgarbossa, A. Protein Aggregation: Emerging Strategies against Amyloidogenesis. Int. J. Mol. Sci. 2012, 13, 17121–17137. [Google Scholar] [CrossRef] [PubMed]
- Dobson, C.M. Protein folding and misfolding. Nature 2003, 426, 884–890. [Google Scholar] [CrossRef] [PubMed]
- Kayed, R.; McIntire, T.M.; Milton, S.C.; Glabe, C.G.; Head, E.; Thompson, J.L.; Cotman, C.W. Common Structure of Soluble Amyloid Oligomers Implies Common Mechanism of Pathogenesis. Science 2003, 300, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Baglioni, S.; Casamenti, F.; Bucciantini, M.; Luheshi, L.M.; Taddei, N.; Chiti, F.; Dobson, C.M.; Stefani, M. Prefibrillar Amyloid Aggregates Could Be Generic Toxins in Higher Organisms. J. Neurosci. 2006, 26, 8160–8167. [Google Scholar] [CrossRef]
- Benilova, I.; Karran, E.; De Strooper, B. The toxic Aβ oligomer and Alzheimer’s disease: An emperor in need of clothes. Nat. Neurosci. 2012, 15, 349–357. [Google Scholar] [CrossRef]
- Lee, C.-C.; Nayak, A.; Sethuraman, A.; Belfort, G.; McRae, G.J. A Three-Stage Kinetic Model of Amyloid Fibrillation. Biophys. J. 2007, 92, 3448–3458. [Google Scholar] [CrossRef]
- Knowles, T.P.J.; Waudby, C.A.; Devlin, G.L.; Cohen, S.I.A.; Aguzzi, A.; Vendruscolo, M.; Terentjev, E.M.; Welland, M.E.; Dobson, C.M.; Waudby, C.; et al. An Analytical Solution to the Kinetics of Breakable Filament Assembly. Science 2009, 326, 1533–1537. [Google Scholar] [CrossRef]
- Arosio, P.; Vendruscolo, M.; Dobson, C.M.; Knowles, T.P. Chemical kinetics for drug discovery to combat protein aggregation diseases. Trends Pharmacol. Sci. 2014, 35, 127–135. [Google Scholar] [CrossRef]
- Fodero-Tavoletti, M.T.; Villemagne, V.L.; Rowe, C.C.; Masters, C.L.; Barnham, K.J.; Cappai, R. Amyloid-β: The seeds of darkness. Int. J. Biochem. Cell Biol. 2011, 43, 1247–1251. [Google Scholar] [CrossRef] [PubMed]
- Lannfelt, L.; Blennow, K.; Zetterberg, H.; Batsman, S.; Ames, D.; Harrison, J.; Maters, C.L.; Targum, S.; Bush, A.I.; Murdoch, R.; et al. Safety, efficacy, and biomarker findings of PBT2 in targeting Aβ as a modifying therapy for Alzheimer’s disease: A phase IIa, double-blind, randomized, placebo-controlled trial. Lancet Neurol. 2008, 7, 779–786. [Google Scholar] [CrossRef]
- Soto, C.; Saborio, G.P.; Permanne, B. Inhibiting the conversion of soluble amyloid-beta peptide into abnormally folded amyloidogenic intermediates: Relevance for Alzheimer’s disease therapy. Acta. Neurol. Scand. Suppl. 2000, 176, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, R.; Lam, K.S.; Jin, L.W.; Duan, Y. Alzheimer’s Disease Drug Candidates Stabilize A-b Protein Native Structure by Interacting with the Hydrophobic Core. Biophys. J. 2011, 100, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
- Ankarcrona, M.; Winblad, B.; Monteiro, C.; Fearns, C.; Powers, E.T.; Johansson, J.; Westermark, G.T.; Presto, J.; Ericzon, B.-G.; Kelly, J.W. Current and future treatment of amyloid diseases. J. Intern. Med. 2016, 280, 177–202. [Google Scholar] [CrossRef] [PubMed]
- Blennow, K.; de Leon, M.J.; Zetterberg, H. Alzheimer’s disease. Lancet 2006, 368, 387–403. [Google Scholar] [CrossRef]
- Permanne, B.; Adessi, C.; Saborio, G.P.; Fraga, S.; Frossard, M.J.; van Dorpe, J.; Dewachter, I.; Banks, W.A.; van Leuven, F.; Soto, C. Reduction of amyloid load and cerebral damage in a transgenic mouse model of Alzheimer’s disease by treatment with a beta-sheet breaker peptide. FASEB J. 2002, 16, 860–862. [Google Scholar] [CrossRef]
- Chiti, F.; Stefani, M.; Taddei, N.; Ramponi, G.; Dobson, C.M. Rationalization of the effects of mutations on peptide and protein aggregation rates. Nature 2003, 424, 805–808. [Google Scholar] [CrossRef]
- Belli, M.; Ramazzotti, M.; Chiti, F. Prediction of amyloid aggregation in vivo. EMBO Rep. 2011, 12, 657–663. [Google Scholar] [CrossRef]
- Armstrong, A.H.; Chen, J.; McKoy, A.F.; Hecht, M.H. Mutations That Replace Aromatic Side Chains Promote Aggregation of the Alzheimer’s Aβ Peptide. Biochemistry 2011, 50, 4058–4067. [Google Scholar] [CrossRef][Green Version]
- Reinke, A.A.; Gestwicki, J.E. Structure?activity Relationships of Amyloid Beta-aggregation Inhibitors Based on Curcumin: Influence of Linker Length and Flexibility. Chem. Boil. Drug Des. 2007, 70, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Gazit, E. A possible role for pi-stacking in the self-assembly of amyloid fibrils. FASEB J. 2002, 16, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Tracz, S.M.; Abedini, A.; Driscoll, M.; Raleigh, D.P. Role of Aromatic Interactions in Amyloid Formation by Peptides Derived from Human Amylin†. Biochemistry 2004, 43, 15901–15908. [Google Scholar] [CrossRef] [PubMed]
- Milardi, D.; Sciacca, M.F.M.; Pappalardo, M.; Grasso, D.M.; la Rosa, C. The role of aromatic side-chains in amyloid growth and membrane interaction of the islet amyloid polypeptide fragment LANFLVH. Eur. Biophys. J. 2011, 40, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Marek, P.; Abedini, A.; Song, B.; Kanungo, M.; Johnson, M.E.; Gupta, R.; Zaman, W.; Wong, S.S.; Raleigh, D.P. Aromatic Interactions Are Not Required for Amyloid Fibril Formation by Islet Amyloid Polypeptide but Do Influence the Rate of Fibril Formation and Fibril Morphology. Biochemistry 2007, 46, 3255–3261. [Google Scholar] [CrossRef]
- Fauchère, J.L.; Charton, M.; Kier, L.B.; Verloop, A.; Pliska, V. Amino acid side chain parameters for correlation studies in biology and pharmacology. Int. J. Pept. Protein Res. 1988, 32, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Bemporad, F.; Taddei, N.; Stefani, M.; Chiti, F. Assessing the role of aromatic residues in the amyloid aggregation of human muscle acylphosphatase. Protein Sci. 2006, 15, 862–870. [Google Scholar] [CrossRef]
- Marshall, K.E.; Morris, K.L.; Charlton, D.; O’Reilly, N.; Lewis, L.; Walden, H.; Serpell, L.C. Hydrophobic, Aromatic, and Electrostatic Interactions Play a Central Role in Amyloid Fibril Formation and Stability. Biochemistry 2011, 50, 2061–2071. [Google Scholar] [CrossRef]
- Azriel, R.; Gazit, E. Analysis of the Minimal Amyloid-forming Fragment of the Islet Amyloid Polypeptide. J. Boil. Chem. 2001, 276, 34156–34161. [Google Scholar] [CrossRef]
- Profit, A.A.; Felsen, V.; Chinwong, J.; Mojica, E.-R.E.; Desamero, R.Z.B. Evidence of π-stacking interactions in the self-assembly of hIAPP(22–29). Proteins Struct. Funct. Bioinform. 2013, 81, 690–703. [Google Scholar] [CrossRef]
- Ahmad, E.; Ahmad, A.; Singh, S.; Arshad, M.; Khan, A.H.; Khan, R.H. A mechanistic approach for islet amyloid polypeptide aggregation to develop anti-amyloidogenic agents for type-2 diabetes. Biochemistry 2011, 93, 793–805. [Google Scholar] [CrossRef] [PubMed]
- Ardah, M.T.; Paleologou, K.E.; Lv, G.; Khair, S.B.A.; Kazim, A.S.; Minhas, S.T.; Al-Tel, T.H.; Al-Hayani, A.A.; Haque, M.E.; Eliezer, D.; et al. Structure activity relationship of phenolic acid inhibitors of α-synuclein fibril formation and toxicity. Front. Aging Neurosci. 2014, 6, 197. [Google Scholar] [CrossRef] [PubMed]
- Ramshini, H.; Mannini, B.; Khodayari, K.; Ebrahim-Habibi, A.; Moghaddasi, A.S.; Tayebee, R.; Chiti, F. Bis(indolyl)phenylmethane derivatives are effective small molecules for inhibition of amyloid fibril formation by hen lysozyme. Eur. J. Med. Chem. 2016, 124, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Lakey-Beitia, J.; Berrocal, R.; Rao, K.S.; Durant, A.A. Polyphenols as therapeutic molecules in Alzheimer’s disease through modulating Amyloid pathways. Mol. Neurobiol. 2015, 51, 466–479. [Google Scholar] [CrossRef] [PubMed]
- Begum, A.N.; Jones, M.R.; Lim, G.P.; Morihara, T.; Kim, P.; Heath, D.D.; Rock, C.L.; Pruitt, M.A.; Yang, F.; Hudspeth, B.; et al. Curcumin structure-function, bioavailability, and efficacy in models of neuroinflammation and Alzheimer’s disease. J. Pharmacol. Exp. Ther. 2008, 326, 196–208. [Google Scholar] [CrossRef]
- Levy-Sakin, M.; Shreberk, M.; Daniel, Y.; Gazit, E. Targeting insulin amyloid assembly by small aromatic molrcules: Toward rational design aggregation inhibitors. Islets 2009, 1, 210–215. [Google Scholar] [CrossRef]
- Krebs, M.R.; Wilkins, D.K.; Chung, E.W.; Pitkeathly, M.C.; Chamberlain, A.K.; Zurdo, J.; Robinson, C.V.; Dobson, C.M. Formation and seeding of amyloid fibrils from wild-type hen lysozyme and a peptide fragment from the beta-domain. J. Mol. Biol. 2000, 300, 541–549. [Google Scholar] [CrossRef]
- Morozova-Roche, L.A.; Zurdo, J.; Spencer, A.; Noppe, W.; Receveur, V.; Archer, D.B.; Joniau, M.; Dobson, C.M. Amyloid Fibril Formation and Seeding by Wild-Type Human Lysozyme and Its Disease-Related Mutational Variants. J. Struct. Boil. 2000, 130, 339–351. [Google Scholar] [CrossRef]
- Goda, S.; Takano, K.; Yamagata, Y.; Nagata, R.; Akutsu, H.; Maki, S.; Namba, K.; Yutani, K. Amyloid protofilament formation of hen egg lysozyme in highly concentrated ethanol solution. Protein Sci. 2000, 9, e369–e375. [Google Scholar] [CrossRef]
- Cao, A.; Hu, D.; Lai, L. Formation of amyloid fibrils from fully reduced hen egg white lysozyme. Protein Sci. 2004, 13, 319–324. [Google Scholar] [CrossRef]
- Swaminathan, R.; Ravi, V.K.; Kumar, S.; Kumar, M.V.; Chandra, N. Lysozyme: A model protein for amyloid research. Adv. Protein Chem. Struct. Biol. 2011, 84, 63–111. [Google Scholar] [PubMed]
- Xu, M.; Shashilov, V.A.; Ermolenkov, V.V.; Fredriksen, L.; Zagorevski, D.; Lednev, I.K. The first step of hen egg white lysozyme fibrillation, irreversible partial unfolding, is a two-state transition. Protein Sci. 2007, 16, 815–832. [Google Scholar] [CrossRef] [PubMed]
- Tayebee, R.; Jarrahi, M.; Maleki, B.; Mokhtari, Z.B.; Baghbanian, S.M.; Razi, M.K. A new method for the preparation of 1,3,5-triarylbenzenes catalyzed by nanoclinoptilolite/HDTMA. RSC Adv. 2015, 5, 10869–10877. [Google Scholar] [CrossRef]
- Tayebee, R.; Jarrahi, M. ChemInform Abstract: H6P2W18O62/Nanoclinoptilolite as an Efficient Nanohybrid Catalyst in the Cyclotrimerization of Aryl Methyl Ketones under Solvent-Free Conditions. RSC Adv. 2015, 46, 21206–21214. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Thornberry, N.A. The caspase family of cysteine proteases. Br. Med. Bull. 1997, 53, 478–490. [Google Scholar] [CrossRef]
- Re, F.; Airoldi, C.; Zona, C.; Masserini, M.; la Ferla, B.; Quattrocchi, N.; Nicotra, F. Beta amyloid aggregation inhibitors: Small molecules as candidate drugs for therapy of Alzheimer’s disease. Curr. Med. Chem. 2010, 17, 2990–3006. [Google Scholar] [CrossRef]
- Wang, X.-Q.; Xia, C.-L.; Chen, S.-B.; Tan, J.-H.; Ou, T.-M.; Huang, S.-L.; Li, D.; Gu, L.-Q.; Huang, Z.-S. Design, synthesis, and biological evaluation of 2-arylethenylquinoline derivatives as multifunctional agents for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2015, 89, 349–361. [Google Scholar] [CrossRef]
- Sang, Z.; Qiang, X.; Li, Y.; Yuan, W.; Liu, Q.; Shi, Y.; Ang, W.; Luo, Y.; Tan, Z.; Deng, Y. Design, synthesis and evaluation of scutellarein-O-alkylamines as multifunctional agents for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2015, 94, 348–366. [Google Scholar] [CrossRef]
- Sacchettini, J.C.; Kelly, J.W. Therapeutic strategies for human amyloid diseases. Nat. Rev. Drug Discov. 2002, 1, 267–275. [Google Scholar] [CrossRef]
- Tartaglia, G.G.; Cavalli, A.; Pellarin, R.; Caflisch, A. The role of aromaticity, exposed surface, and dipole moment in determining protein aggregation rates. Protein Sci. 2004, 13, 1939–1941. [Google Scholar] [CrossRef] [PubMed]
- Makin, O.S.; Atkins, E.; Sikorski, P.; Johansson, J.; Serpell, L.C. Molecular basis for amyloid fibril formation and stability. Proc. Natl. Acad. Sci. USA 2005, 102, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Profit, A.A.; Vedad, J.; Saleh, M.; Desamero, R.Z. Aromaticity and amyloid formation: Effect of π-electron distribution and aryl substituent geometry on the self-assembly of peptides derived from hIAPP(22–29). Arch. Biochem. Biophys. 2015, 567, 46–58. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Porat, Y.; Stepensky, A.; Ding, F.-X.; Naider, F.; Gazit, E. Completely different amyloidogenic potential of nearly identical peptide fragments. Biopolymers 2003, 69, 161–164. [Google Scholar] [CrossRef]
- Hirata, K.; Sugimoto, T.; Konnob, T. Moriia, Amyloid-forming propensity of the hydrophobic non-natural amino acid on the fibril-forming core peptide of human tau. Bioorg. Med. Chem. Lett. 2007, 17, 2971–2974. [Google Scholar] [CrossRef]
- Ramshini, H.; Mohammad-Zadeh, M.; Ebrahim-Habibi, A. Inhibition of amyloid fibril formation and cytotoxicity by a chemical analog of Curcumin as a stable inhibitor. Int. J. Boil. Macromol. 2015, 78, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Ladiwala, A.R.; Dordick, J.S.; Tessier, P.M. Aromatic small molecules remodel toxic soluble oligomers of amyloid beta through three independent pathways. J. Biol. Chem. 2011, 286, 3209–3218. [Google Scholar] [CrossRef] [PubMed]
- Tayebee, R.; Savoji, K.; Razi, M.K.; Maleki, B. Environmentally friendly cyclotrimerization of substituted acetophenones catalyzed by a new nano-composite of γ-Al 2 O 3 nanoparticles decorated with H 5 PW 10 V 2 O 40. RSC Adv. 2016, 60, 55319–55326. [Google Scholar] [CrossRef]
- Romanellia, G.P.; Ruiza, D.M.; Bideberripea, H.P.; Autinob, J.C.; Baronetti, G.T.; Thomasa, H.J. Silicagel-supported H6P2W18O62. 24H2O: A reusable catalyst to prepare diphenylmethyl (DPM) ethers. Arkivoc 2007, 2007, 1–8. [Google Scholar]
- Sophianopoulos, A.J.; Rhodes, C.; Holcomb, D.N.; van Holde, K.E. Physical studies of lysozyme. I. Characterization. J. Biol. Chem. 1962, 237, 1107–1112. [Google Scholar]
- Nilsson, M.R. Techniques to study amyloid fibril formation in vitro. Methods 2004, 34, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Cascella, R.; Capitini, C.; Fani, G.; Dobson, C.M.; Cecchi, C.; Chiti, F. Quantification of the Relative Contributions of Loss-of-function and Gain-of-function Mechanisms in TAR DNA-binding Protein 43 (TDP-43) Proteinopathies. J. Boil. Chem. 2016, 291, 19437–19448. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramshini, H.; Tayebee, R.; Bigi, A.; Bemporad, F.; Cecchi, C.; Chiti, F. Identification of Novel 1,3,5-Triphenylbenzene Derivative Compounds as Inhibitors of Hen Lysozyme Amyloid Fibril Formation. Int. J. Mol. Sci. 2019, 20, 5558. https://doi.org/10.3390/ijms20225558
Ramshini H, Tayebee R, Bigi A, Bemporad F, Cecchi C, Chiti F. Identification of Novel 1,3,5-Triphenylbenzene Derivative Compounds as Inhibitors of Hen Lysozyme Amyloid Fibril Formation. International Journal of Molecular Sciences. 2019; 20(22):5558. https://doi.org/10.3390/ijms20225558
Chicago/Turabian StyleRamshini, Hassan, Reza Tayebee, Alessandra Bigi, Francesco Bemporad, Cristina Cecchi, and Fabrizio Chiti. 2019. "Identification of Novel 1,3,5-Triphenylbenzene Derivative Compounds as Inhibitors of Hen Lysozyme Amyloid Fibril Formation" International Journal of Molecular Sciences 20, no. 22: 5558. https://doi.org/10.3390/ijms20225558
APA StyleRamshini, H., Tayebee, R., Bigi, A., Bemporad, F., Cecchi, C., & Chiti, F. (2019). Identification of Novel 1,3,5-Triphenylbenzene Derivative Compounds as Inhibitors of Hen Lysozyme Amyloid Fibril Formation. International Journal of Molecular Sciences, 20(22), 5558. https://doi.org/10.3390/ijms20225558





