Fusaricidin Produced by Paenibacillus polymyxa WLY78 Induces Systemic Resistance against Fusarium Wilt of Cucumber
Abstract
:1. Introduction
2. Results
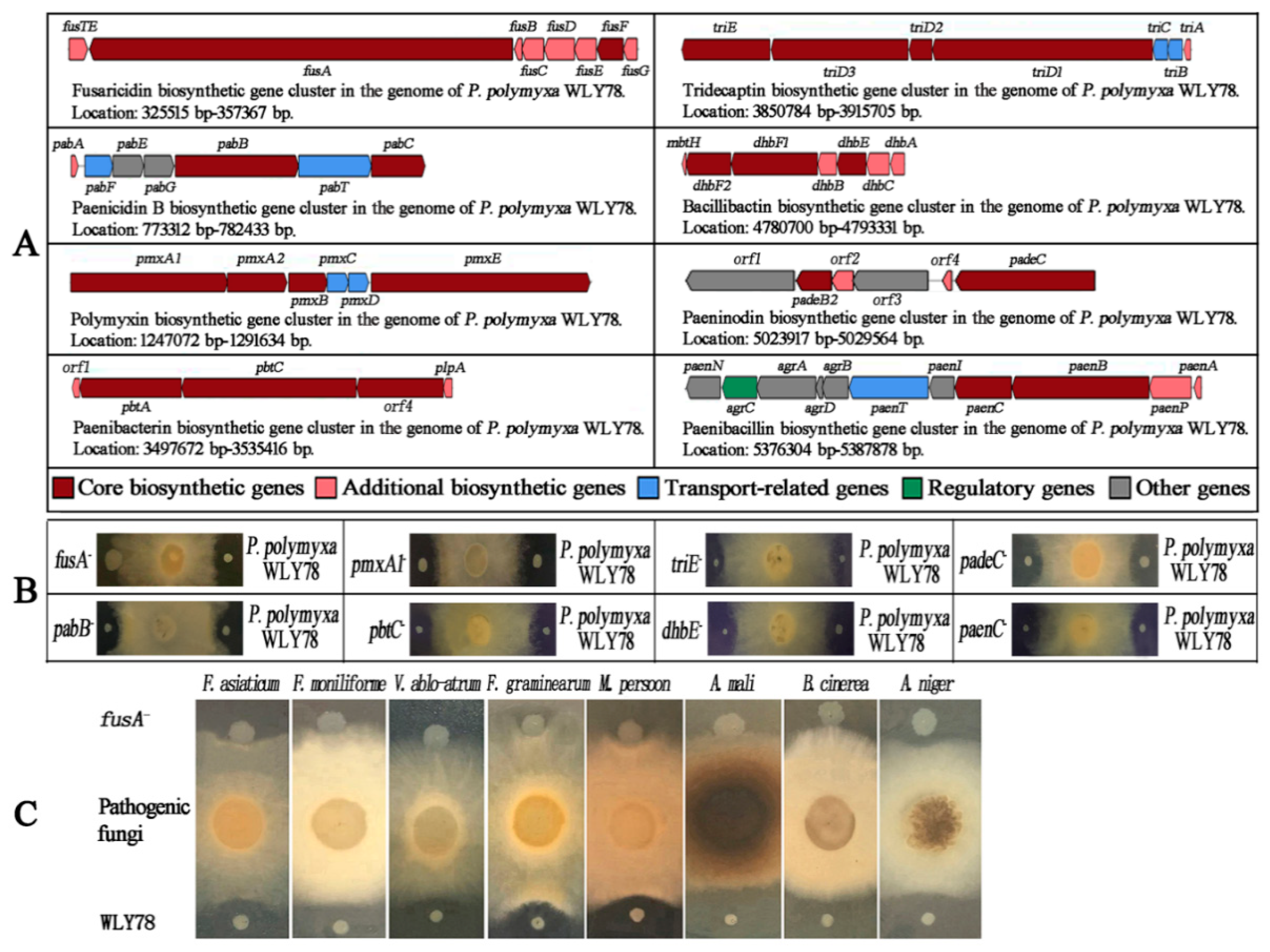
2.1. The fus Gene Cluster in P. polymyxa WLY78 Is Essential for Antifungal Activity
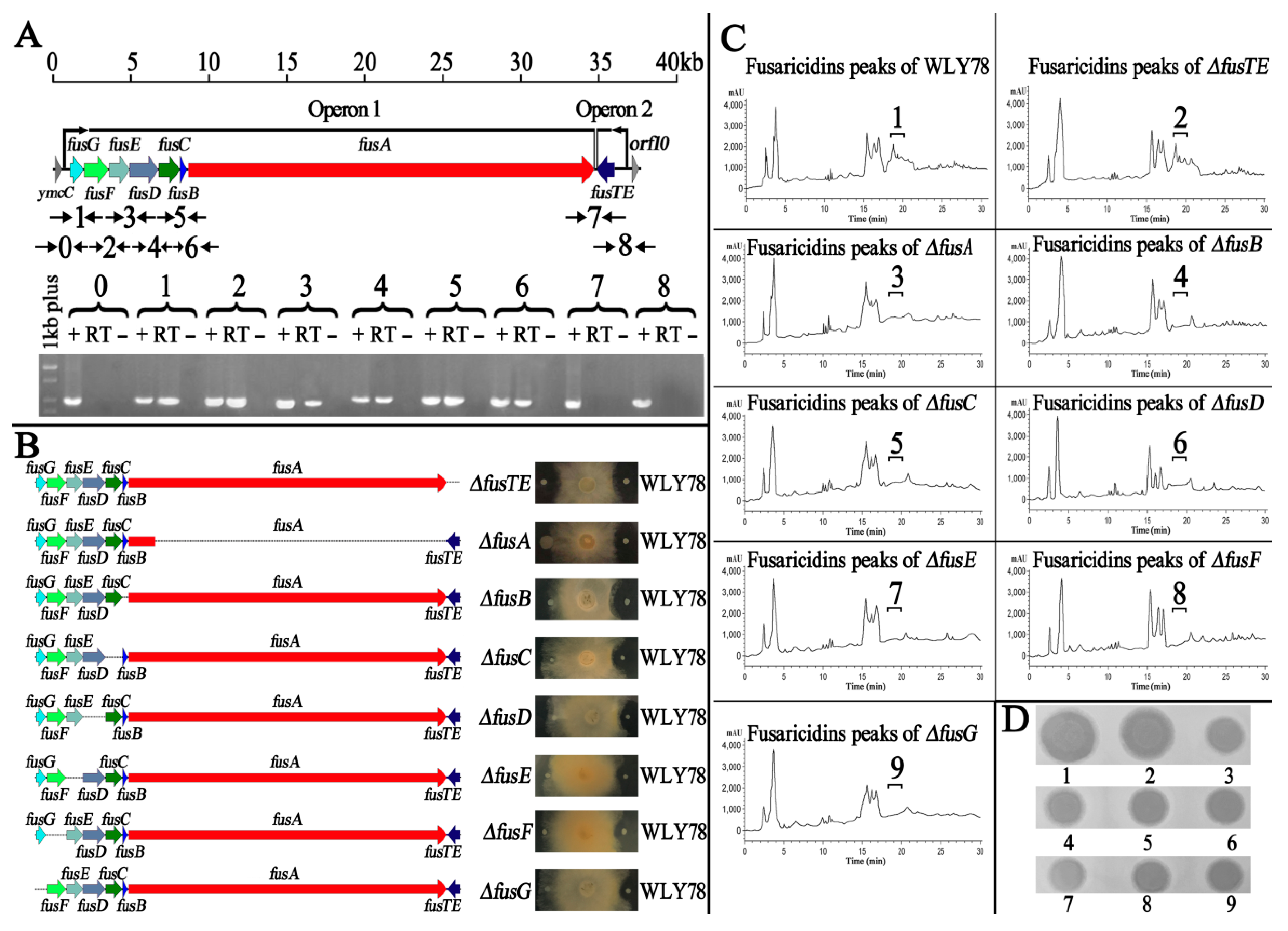
2.2. The Role of Each Gene within the fus Gene Cluster in Inhibition of F. oxysporum f. sp. cucumerium
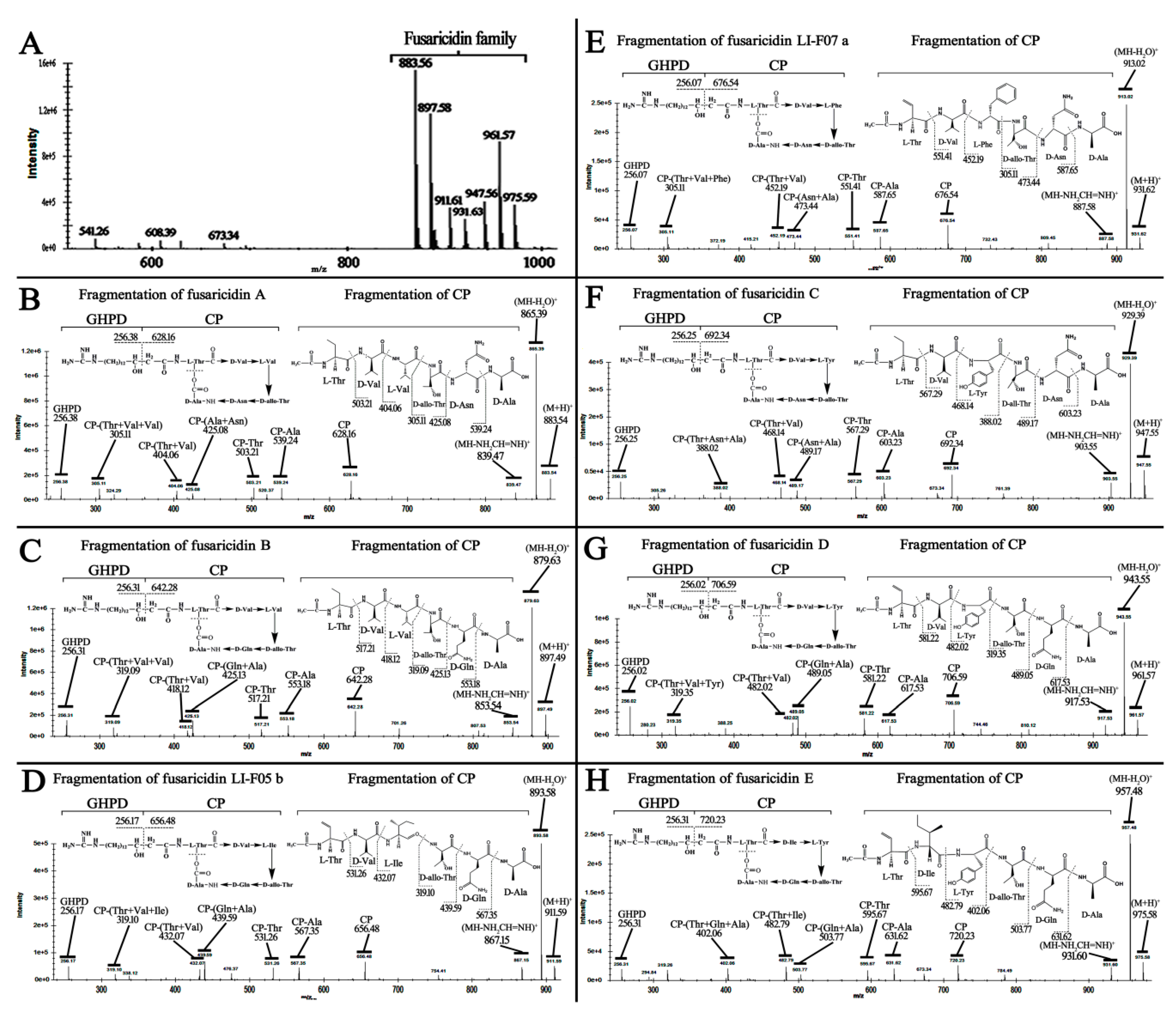
2.3. P. polymyxa WLY78 Produces Seven Forms of Fusaricidins and Fixes Nitrogen
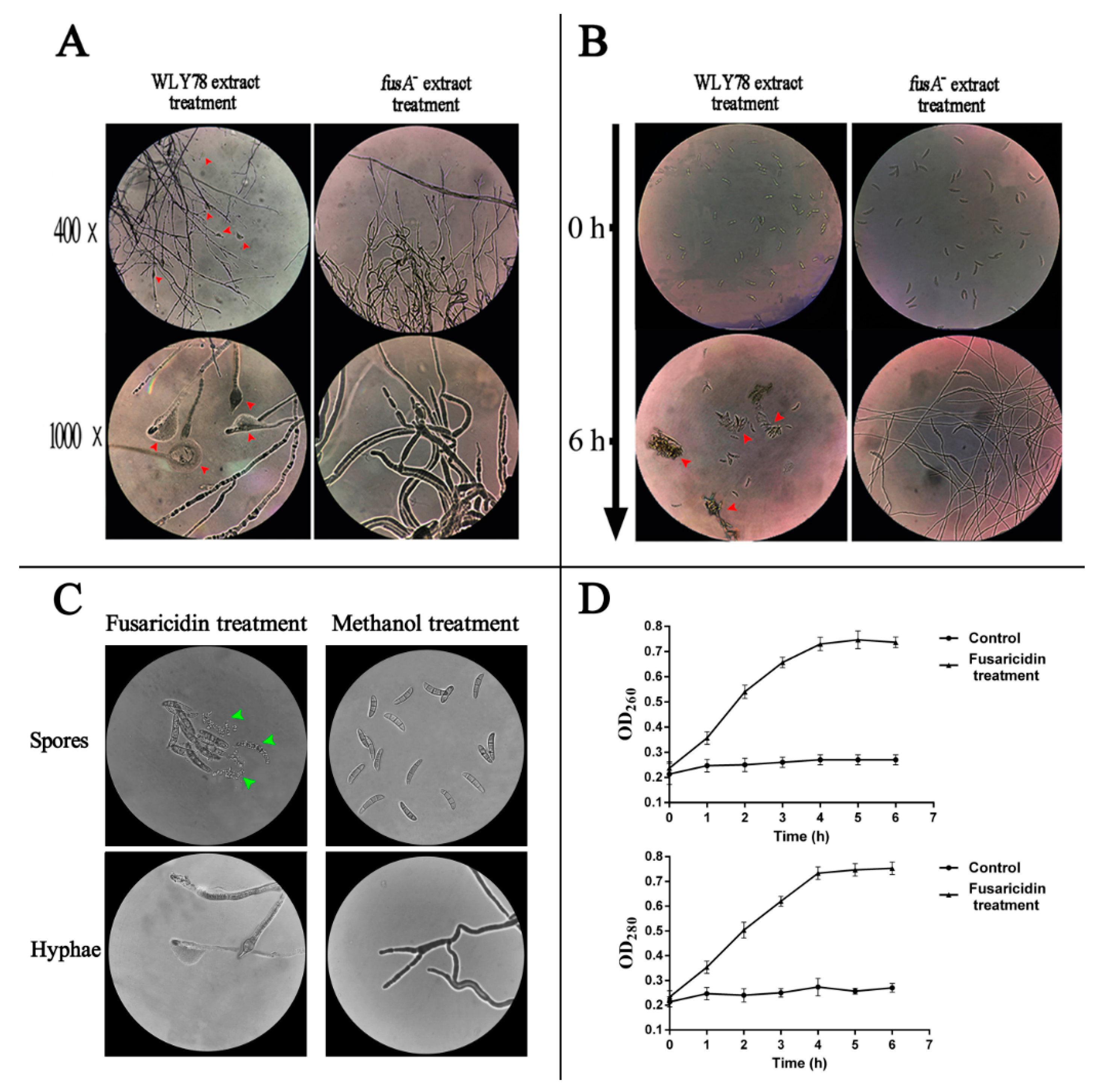
2.4. Fusaricidin Inhibits Spore Germination and Hypha Growth by Causing Cytoplasm Leakage
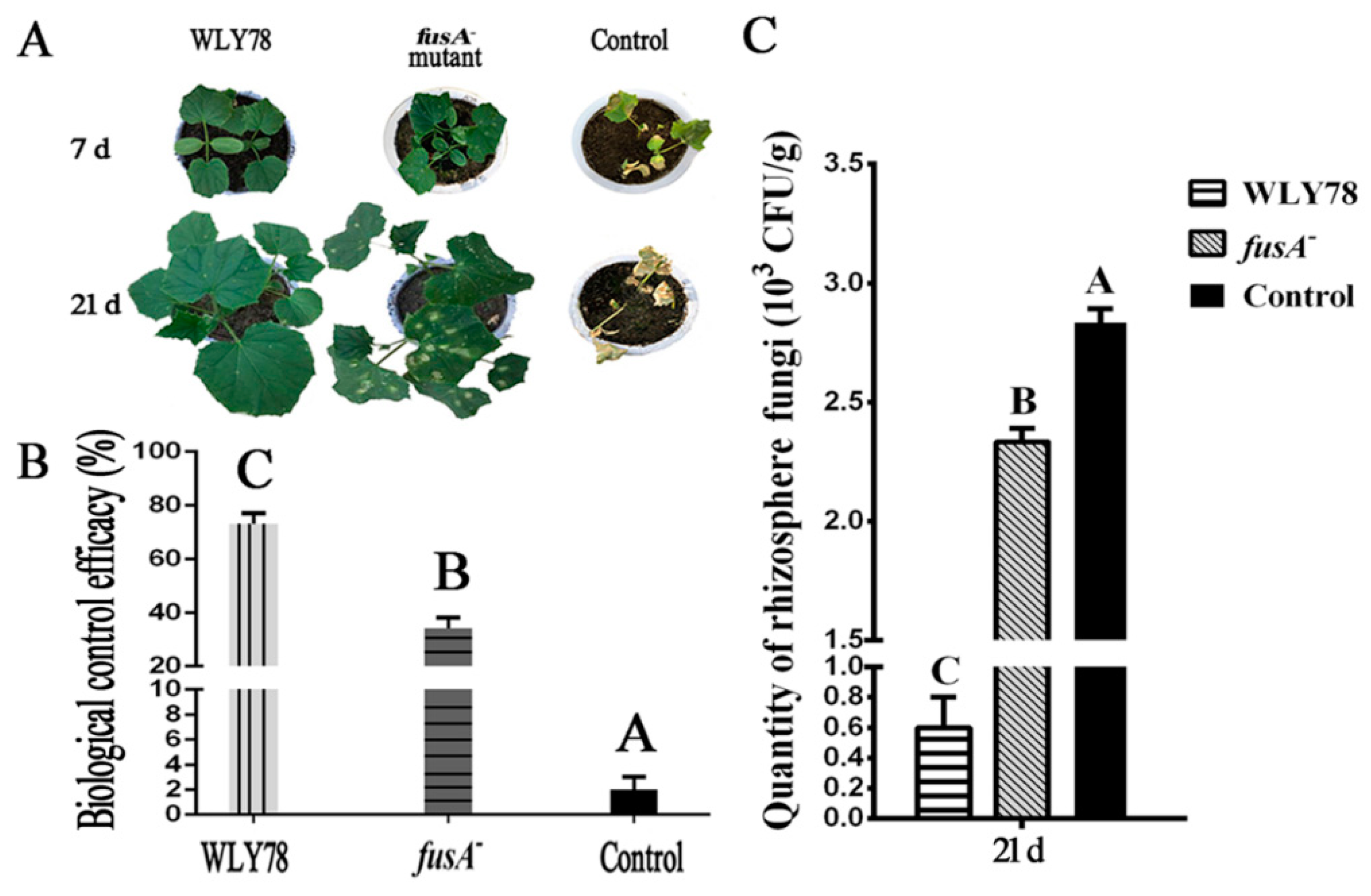
2.5. Fusaricidin Suppresses Fusarium Wilt of Cucumber In Vivo
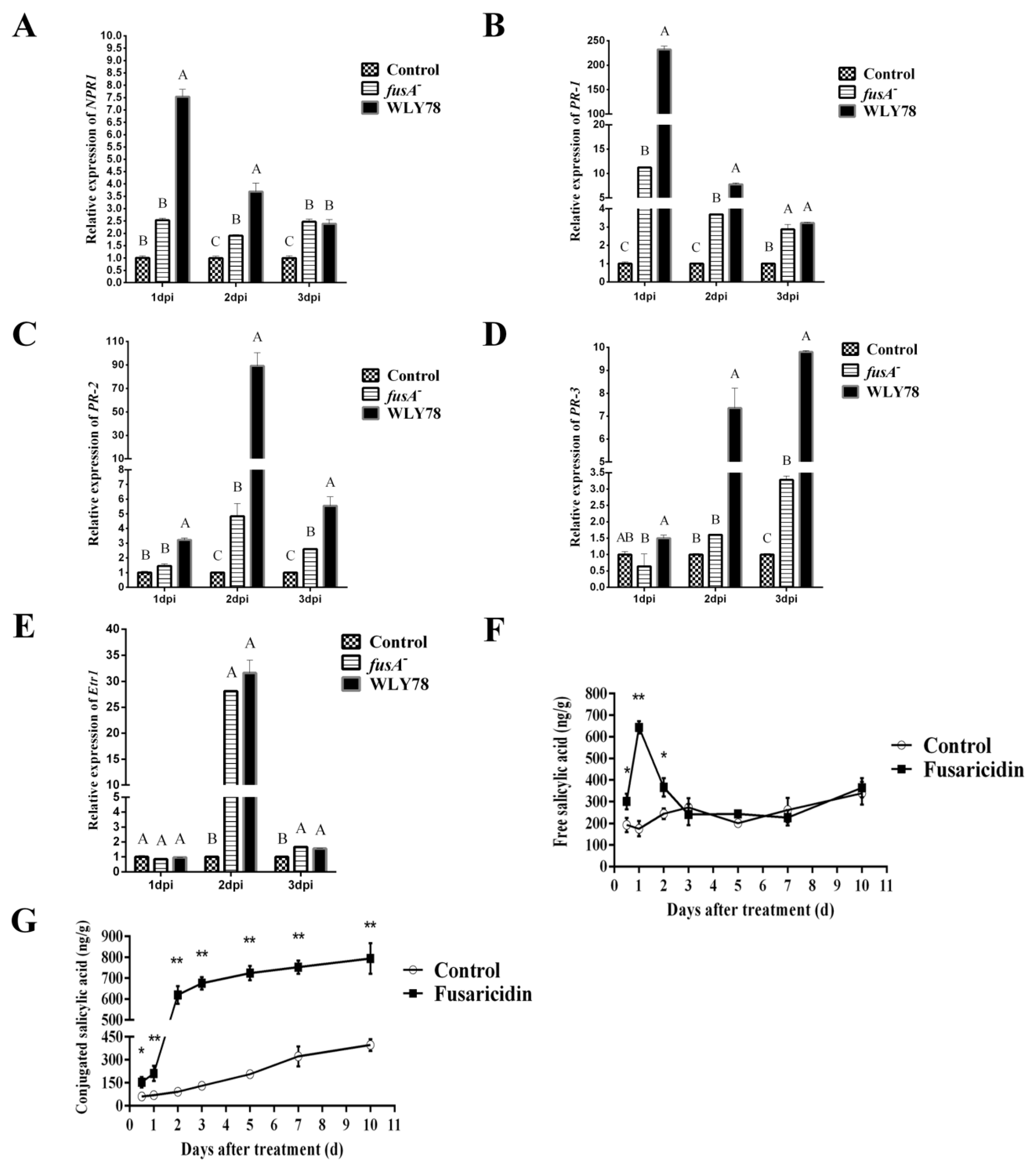
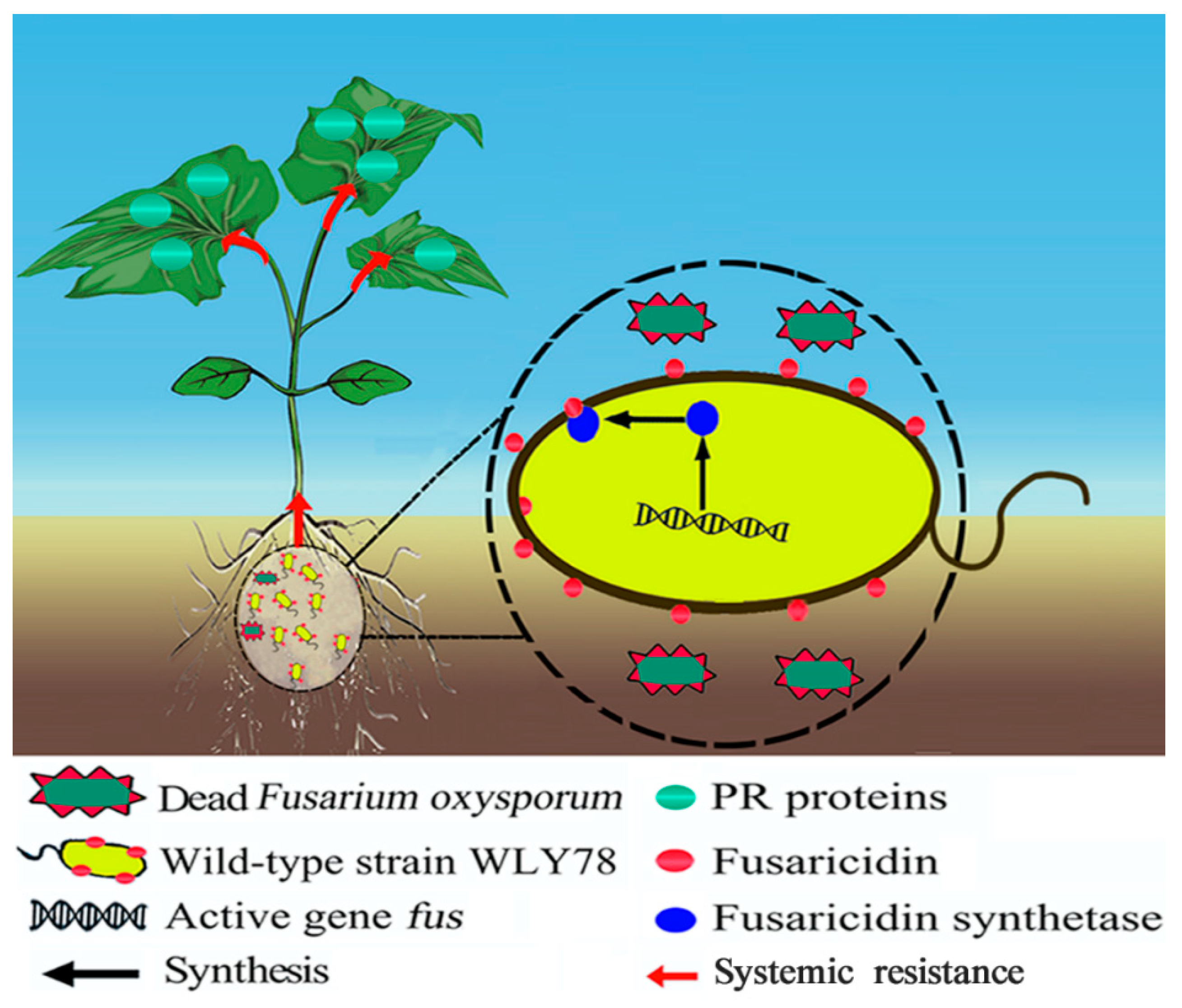
2.6. Fusaricidin Is Involved in Inducing Systemic Resistance
3. Discussion
4. Materials and Methods
4.1. Microorganisms, Plasmids, and Culture Conditions
4.2. Disruption of the Gene Cluster Involved in Synthesis of the Potential Antifungal Substances
4.3. Mutation of Each Gene within the fus Gene Cluster
4.4. Antifungal Activity Assay
4.5. RT-PCR Analysis
4.6. Extraction, Purification, and Antifungal Activity Assay of Fusaricidin
4.7. Identification of Fusaricidin
4.8. Acetylene Reduction Assays of Nitrogenase Activity
4.9. Inhibitory Mode of Fusaricidin against F. oxysporum f. sp. cucumerium
4.10. Plant Growth Conditions
4.11. Biocontrol Efficacy Assay
4.12. Detection of Expression Levels of Plant Resistance Gene
4.13. Detection of Salicylic Acid
4.14. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| fus | Fusaricidin biosynthesis gene |
| pab | Paenicidin B biosynthesis gene |
| pmx | Polymyxin biosynthesis gene |
| pbt | Paenibacterin biosynthesis gene |
| tri | Tridecaptin biosynthesis gene |
| dhb | Bacillibactin biosynthesis gene |
| pade | Paeninodin biosynthesis gene |
| paen | Paenibacillin biosynthesis gene |
| LC-MS | Liquid chromatography-mass spectrum |
| PR | Pathogenesis-related |
| ISR | Induced systemic resistance |
| SAR | Systemic acquired resistance |
| SA | Salicylic acid |
References
- Owen, J.H. Fusarium wilt of cucumber. Phytopathology 1955, 45, 435–439. [Google Scholar]
- Zhang, Z.; Jiang, W.; Jian, Q.; Song, W.; Zheng, Z.; Wang, D.; Liu, X. Residues and dissipation kinetics of triazole fungicides difenoconazole and propiconazole in wheat and soil in Chinese fields. Food Chem. 2015, 168, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Huo, Z.; Nan, Z.; Xu, Z.; Li, S.; Zhang, Q.; Qiu, M.; Yong, X.; Huang, Q.; Zhang, R.; Shen, Q.R. Optimization of survival and spore formation of Paenibacillus polymyxa SQR-21 during bioorganic fertilizer storage. Bioresour. Technol. 2012, 108, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Bent, E.; Breuil, C.; Enebak, S.; Chanway, P.C. Surface colonization of lodgepole pine (Pinus contora var latifolia) roots by Pseudomonas and Paenibacillus polymyxa under antibiotic conditions. Plant Soil 2002, 241, 187–196. [Google Scholar] [CrossRef]
- Raza, W.; Yang, X.M.; Wu, H.S.; Wang, Y.; Xu, Y.C.; Shen, Q.R. Isolation and characterisation of fusaricidin-type compound-producing strain of Paenibacillus polymyxa SQR-21 active against Fusarium oxysporum f. sp. nevium. Eur. J. Plant Pathol. 2009, 125, 471–483. [Google Scholar] [CrossRef]
- Haggag, W.M.; Timmusk, S. Colonization of peanut roots by biofilm-forming Paenibacillus polymyxa initiates biocontrol against crown rot disease. J. Appl. Microbiol. 2008, 104, 961–969. [Google Scholar] [CrossRef]
- Beatty, P.H.; Jensen, S.E. Paenibacillus polymyxa produces fusaricidin-type antifungal antibiotics active against Leptosphaeria maculans, the causative agent of blackleg disease of canola. Can. J. Microbiol. 2002, 48, 159–169. [Google Scholar] [CrossRef]
- Lamsal, K.; Kim, S.W.; Kim, Y.S.; Lee, Y.S. Biocontrol of late blight and plant growth promotion in tomato using rhizobacterial isolates. J. Microbiol. Biotechnol. 2013, 23, 897–904. [Google Scholar] [CrossRef]
- Bosmans, L.; Bruijn, I.D.; Gerards, S.; Moerkens, R.; van Looveren, L.; Wittemans, L.; van Calenberge, B.; Paeleman, A.; van Kerckhove, S.; Rozenski, J. Potential for biocontrol of hairy root disease by a Paenibacillus clade. Front. Microbiol. 2017, 8, 447. [Google Scholar] [CrossRef]
- Hao, Z.; Tuinen, D.V.; Wipf, D.; Fayolle, L.; Chataignier, O.; Li, X.; Chen, B.; Gianinazzi, S.; Gianinazzi-Pearson, V.; Adrian, M. Biocontrol of grapevine aerial and root pathogens by Paenibacillus sp. strain B2 and paenimyxin in vitro and in planta. Biol. Control 2017, 109, 42–50. [Google Scholar] [CrossRef]
- Lee, B.; Farag, A.M.; Park, B.H.; Kloepper, W.J.; Lee, H.S.; Ryu, C.-M. Induced resistance by a long-chain bacterial volatile: Elicitation of plant systemic defense by a C13 volatile produced by Paenibacillus polymyxa. PLoS ONE 2012, 7, e48744. [Google Scholar] [CrossRef] [PubMed]
- Kajimura, Y.; Kaneda, M. Fusaricidin A, a new depsipeptide antibiotic produced by Bacillus polymyxa KT-8 taxonomy, fermentation, isolation, structure elucidation and biological activity. J. Antibiot. 1996, 49, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, S.; Senthilkumar, S.; Gnanamanickam, S.; Inayathullah, M.; Jayakumar, R. Isolation and partial characterization of antifungal protein from Bacillus polymyxa strain VLB16. Process Biochem. 2005, 40, 3236–3243. [Google Scholar] [CrossRef]
- Cho, K.M.; Hong, S.Y.; Lee, S.M.; Kim, Y.H.; Kahng, G.G.; Kim, H.; Dae, Y.H. A cel44C-man26A gene of endophytic Paenibacillus polymyxa GS01 has multi-glycosyl hydrolases in two catalytic domains. Appl. Microbiol. Biotechnol. 2006, 73, 618–630. [Google Scholar] [CrossRef]
- Cochrane, S.A.; Vederas, J.C. Lipopeptides from Bacillus and Paenibacillus spp.: A gold mine of antibiotic candidates. Med. Res. Rev. 2016, 36, 4–31. [Google Scholar] [CrossRef]
- Sieber, S.A.; Marahiel, M.A. Molecular mechanisms underlying nonribosomal peptide synthesis: Approaches to new antibiotics. Chem. Rev. 2005, 105, 715–738. [Google Scholar] [CrossRef]
- Kuroda, J.; Fukai, T.; Nomura, T. Collision-induced dissociation of ring-opened cyclic depsipeptides with a guanidino group by electrospray ionization/ion trap mass spectrometry. J. Mass Spectrom. 2001, 36, 30. [Google Scholar] [CrossRef]
- Nomura, T.; Kuroda, J.; Fukai, T.; Konishi, M.; Uno, J.; Kurusu, K. LI-F antibiotics, a family of antifungal cyclic depsipeptides produced by Bacillus polymyxa L-1129. Heterocycles 2000, 53, 1533. [Google Scholar] [CrossRef]
- Kajimura, Y.; Kaneda, M. Fusaricidins B, C and D, new depsipeptide antibiotics produced by Bacillus polymyxa KT-8: Isolation, structure elucidation and biological activity. J. Antibiot. 1997, 50, 220–228. [Google Scholar] [CrossRef]
- Yu, W.; Yin, C.; Zhou, C.; Ye, B. Prediction of the mechanism of action of fusaricidin on Bacillus subtilis. PLoS ONE 2012, 7, e50003. [Google Scholar] [CrossRef]
- Ma, Z.; Ongena, M.; Höfte, M. The cyclic lipopeptide orfamide induces systemic resistance in rice to Cochliobolus miyabeanus but not to Magnaporthe oryzae. Plant Cell Rep. 2017, 36, 1731–1746. [Google Scholar] [CrossRef] [PubMed]
- Hua, J.; Meyerowitz, E.M. Ethylene responses are negatively regulated by a receptor gene family in Arabidopsis thaliana. Cell 1998, 94, 261–271. [Google Scholar] [CrossRef]
- Zeriouh, H.; Romero, D.; Garcia-Gutierrez, L.; Cazorla, F.M.; de Vicente, A.; Perez-Garcia, A. The iturin-like lipopeptides are essential components in the biological control arsenal of Bacillus subtilis against bacterial diseases of cucurbits. Mol. Plant-Microbe Interact. 2011, 24, 1540–1552. [Google Scholar] [CrossRef] [PubMed]
- Farace, G.; Fernandez, O.; Jacquens, L.; Coutte, F.; Krier, F.; Jacques, P.; Clément, C.; Barka, E.A.; Jacquard, C.; Dorey, S. Cyclic lipopeptides from Bacillus subtilis activate distinct patterns of defence responses in grapevine. Mol. Plant Pathol. 2015, 16, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Jourdan, E.; Henry, G.; Duby, F.; Dommes, J.; Barthélemy, J.P.; Thonart, P.; Ongena, M. Insights into the defense-related events occurring in plant cells following perception of surfactin-type lipopeptide from Bacillus subtilis. Mol. Plant-Microbe Interact. 2009, 22, 456–468. [Google Scholar] [CrossRef]
- Pajerowska-Mukhtar, K.M.; Emerine, D.K.; Mukhtar, M.S. Tell me more: Roles of NPRs in plant immunity. Trends Plant Sci. 2013, 18, 402–411. [Google Scholar] [CrossRef]
- Pieterse, C.M.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.; Bakker, P.A. Induced systemic resistance by beneficial microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Feys, B.J.; Parker, J.E. Interplay of signaling pathways in plant disease resistance. Trends Genet. 2000, 16, 449–455. [Google Scholar] [CrossRef]
- Tornero, P.; Gadea, J.; Conejero, V.; Vera, P. Two PR-1 genes from tomato are differentially regulated and reveal a novel mode of expression for a pathogenesis-related gene during the hypersensitive response and development. Mol. Plant-Microbe Interact. 1997, 10, 624–634. [Google Scholar] [CrossRef]
- Hao, T.; Chen, S. Colonization of wheat, maize and cucumber by Paenibacillus polymyxa WLY78. PLoS ONE 2017, 12, e0169980. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, L.; Liu, Z.; Liu, Z.; Zhao, D.; Liu, X.; Zhang, B.; Xie, J.; Hong, Y.; Li, P. A minimal nitrogen fixation gene cluster from Paenibacillus sp. WLY78 enables expression of active nitrogenase in Escherichia coli. PLoS Genet. 2013, 9, e1003865. [Google Scholar] [CrossRef]
- Xie, J.; Shi, H.; Du, Z.; Wang, T.; Liu, X.; Chen, S. Comparative genomic and functional analysis reveal conservation of plant growth promoting traits in Paenibacillus polymyxa and its closely related species. Sci. Rep. 2016, 9, 21329. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Ravnskov, S.; Larsen, J. Biocontrol of Pythium damping-off in cucumber by arbuscular mycorrhiza-associated bacteria from the genus Paenibacillus. Fungicides 2007, 52, 863–875. [Google Scholar] [CrossRef]
- Wang, L.; Xie, Y.; Cui, Y.; Xu, J.; He, W.; Chen, H.; Guo, J. Conjunctively screening of biocontrol agents (BCAs) against Fusarium root rot and Fusarium head blight caused by Fusarium graminearum. Microbiol. Res. 2015, 177, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Lu, Z.; Bi, H.; Lu, F.; Zhang, C.; Bie, X. Isolation and characterization of peptide antibiotics LI-F04 and polymyxin B6 produced by Paenibacillus polymyxa strain JSa-9. Peptides 2011, 32, 1917–1923. [Google Scholar] [CrossRef] [PubMed]
- Lohans, C.T.; Huang, Z.; van Belkum, M.J.; Giroud, M.; Sit, C.S.; Steels, E.M.; Zheng, J.; Whittal, R.M.; McMullen, L.M.; Vederas, J.C. Structural Characterization of the Highly Cyclized Lantibiotic Paenicidin A via a Partial Desulfurization/Reduction Strategy. J. Am. Chem. Soc. 2012, 134, 19540–19543. [Google Scholar] [CrossRef]
- Deris, Z.Z.; Akter, J.; Sivanesan, S.; Roberts, K.D.; Thompson, P.E.; Nation, R.L.; Li, J.; Velkov, T. A secondary mode of action of polymyxins against gram-negative bacteria involves the inhibition of NADH-quinone oxidoreductase activity. J. Antibiot. 2014, 67, 147. [Google Scholar] [CrossRef]
- Cochranea, A.S.; Findlay, B.; Bakhtiary, A.; Acedo, Z.J.; Rodriguez-Lopez, M.E.; Mercier, P.; John, V.C. Antimicrobial lipopeptide tridecaptin A1 selectively binds to Gram-negative lipid II. Proc. Natl. Acad. Sci. USA 2016, 113, 11561. [Google Scholar] [CrossRef]
- Huang, E.; Yousef, E.A. Paenibacterin, a novel broad-spectrum lipopeptide antibiotic, neutralises endotoxins and promotes survival in a murine model of Pseudomonas aeruginosa-induced sepsis. Int. J. Antimicrob. Agents 2014, 44, 74–77. [Google Scholar] [CrossRef]
- Hollensteiner, J.; Wemheuer, F.; Harting, R.; Kolarzyk, A.M.; Valerio, S.M.D.; Poehlein, A.; Brzuszkiewicz, E.B.; Nesemann, K.; Braus-Stromeyer, S.A.; Braus, G.H.; et al. Bacillus thuringiensis and Bacillus weihenstephanensis inhibit the growth of phytopathogenic Verticillium species. Front. Microbiol. 2017, 7, 1–19. [Google Scholar] [CrossRef]
- Zhu, S.; Hegemann, J.D.; Fage, C.D.; Zimmermann, M.; Xie, X.; Linne, U.; Marahiel, M.A. Insights into the unique phosphorylation of the lasso peptide paeninodin. J. Biol. Chem. 2016, 291, 13662–13678. [Google Scholar] [CrossRef] [PubMed]
- Debois, M.; Ongena, M.; Cawoy, H.; de Pauw, E. MALDI-FTICR MS imaging as a powerful tool to identify Paenibacillus antibiotics involved in the inhibition of plant pathogens. J. Am. Soc. Mass Spectrom. 2013, 24, 1202–1213. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.K.; Park, S.Y.; Kim, R.; Lee, C.H.; Kim, J.F.; Park, S.H. Identification and functional analysis of the fusaricidin biosynthetic gene of Paenibacillus polymyxa E681. Biochem. Biophys. Res. Commun. 2008, 365, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Beatty, P.K.; Shah, S.; Jensen, S.E. Use of PCR-targeted mutagenesis to disrupt production of fusaricidin-type antifungal antibiotics in Paenibacillus polymyxa. Appl. Environ. Microbiol. 2007, 73, 3480–3489. [Google Scholar] [CrossRef]
- Li, J.; Jensen, S.E. Nonribosomal biosynthesis of fusaricidins by Paenibacillus polymyxa PKB1 involves direct activation of a D-amino acid. Chem. Biol. 2008, 15, 118–127. [Google Scholar] [CrossRef]
- Li, S.Q.; Zhang, R.F.; Wang, Y.; Zhang, N.; Shao, J.H.; Qiu, M.H.; Shen, B.; Yin, X.H.; Shen, Q.R. Promoter analysis and transcription regulation of fus gene cluster responsible for fusaricidin synthesis of Paenibacillus polymyxa SQR-21. Appl. Microbiol. Biotechnol. 2013, 97, 9479–9489. [Google Scholar] [CrossRef]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389. [Google Scholar] [CrossRef]
- Brogden, K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar] [CrossRef]
- Vater, J.; Niu, B.; Dietel, K.; Borriss, R. Characterization of novel fusaricidins produced by Paenibacillus polymyxa-M1 using MALDI-TOF mass spectrometry. J. Am. Soc. Mass Spectrom. 2015, 26, 1548–1558. [Google Scholar] [CrossRef]
- Reimann, M.; Sandjo, L.P.; Antelo, L.; Thines, E.; Siepe, I.; Opatz, T. A new member of the fusaricidin family-structure elucidation and synthesis of fusaricidin E. Beilstein J. Org. Chem. 2017, 13, 1430–1438. [Google Scholar] [CrossRef]
- van de Mortel, J.E.; de Vos, R.C.; Dekkers, E.; Pineda, A.; Guillod, L.; Bouwmeester, K.; van Loon, J.J.A.; Dicke, M.; Raaijmakers, J.M. Metabolic and Transcriptomic Changes Induced in Arabidopsis by the Rhizobacterium Pseudomonas fluorescens SS101. Plant Physiol. 2012, 160, 2173–2188. [Google Scholar] [CrossRef] [PubMed]
- Tjamos, S.E.; Flemetakis, E.; Paplomatas, E.J.; Katinakis, P. Induction of resistance to Verticillium dahliae in Arabidopsis thaliana by the biocontrol agent K-165 and pathogenesis-related proteins gene expression. Mol. Plant-Microbe Interact. 2005, 18, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Hennig, J.; Malamy, J.; Grynkiewicz, G.; Indulski, J.; Klessig, D.F. Interconversion of the salicylic acid signal and its glucoside in tobacco. Plant J. 2002, 4, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Paulus, H.; Gray, E. The biosynthesis of polymyxin B by growing cultures of Bacillus polymyxa. J. Biol. Chem. 1964, 239, 865–871. [Google Scholar] [PubMed]
- Wang, T.; Zhao, X.; Shi, H.; Sun, L.; Li, Y.; Li, Q.; Zhang, H.; Chen, S.; Li, J. Positive and negative regulation of transferred nif genes mediated by indigenous GlnR in Gram-positive Paenibacillus polymyxa. PLoS Genet. 2018, 14, e1007629. [Google Scholar] [CrossRef]
- Li, Y.; Gu, Y.; Li, J.; Xu, M.; Wei, Q.; Wang, Y. Biocontrol agent Bacillus amyloliquefaciens LJ02 induces systemic resistance against cucurbits powdery mildew. Front. Microbiol. 2015, 6, 883. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Al-Tuwaijri, M. Studies on Fusarium wilt disease of cucumber. J. Appl. Pham. Sci. 2015, 5, 110–119. [Google Scholar] [CrossRef]
- Faheem, M.; Raza, W.; Zhong, W.; Nan, Z.; Shen, Q.R.; Xu, Y.C. Evaluation of the biocontrol potential of Streptomyces goshikiensis YCXU against Fusarium oxysporum f. sp. niveum. Biol. Control 2015, 81, 101–110. [Google Scholar] [CrossRef]
- Kenneth, J.; Livak, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔcT method. Methods 2001, 25, 402–408. [Google Scholar]
- Cameron, R.K.; Paiva, N.L.; Lamb, C.J.; Dixon, R.A. Accumulation of salicylic acid and PR-1 gene transcripts in relation to the systemic acquired resistance (SAR) response induced by Pseudomonas syringae pv. tomato in Arabidopsis. Physiol. Mol. Plant Pathol. 1999, 55, 121–130. [Google Scholar] [CrossRef]
| Gene | Length (bp) | Accession NO. of GenBank | Predicted Product |
|---|---|---|---|
| fusTE | 1035 | AYC81014.1 | Alpa/beta hydrolase-thioesterase |
| fusG | 771 | AYC81021.1 | Enoyl-acyl carrier protein reductase |
| fusF | 1461 | AYC81020.1 | Acyl-CoA ligase |
| fusE | 1224 | AYC81019.1 | Aldehyde dehydrogenase |
| fusD | 1701 | AYC81018.1 | Acetolactate synthase large subunit |
| fusC | 1242 | AYC81017.1 | 3-Oxoacyl-acyl carrier protein synthase |
| fusB | 408 | AYC81016.1 | (3R)-Hydroxymyristoyl-acyl carrier protein |
| fusA | 23730 | AYC81015.1 | Non-ribosomal polypeptide synthetase |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Chen, S. Fusaricidin Produced by Paenibacillus polymyxa WLY78 Induces Systemic Resistance against Fusarium Wilt of Cucumber. Int. J. Mol. Sci. 2019, 20, 5240. https://doi.org/10.3390/ijms20205240
Li Y, Chen S. Fusaricidin Produced by Paenibacillus polymyxa WLY78 Induces Systemic Resistance against Fusarium Wilt of Cucumber. International Journal of Molecular Sciences. 2019; 20(20):5240. https://doi.org/10.3390/ijms20205240
Chicago/Turabian StyleLi, Yunlong, and Sanfeng Chen. 2019. "Fusaricidin Produced by Paenibacillus polymyxa WLY78 Induces Systemic Resistance against Fusarium Wilt of Cucumber" International Journal of Molecular Sciences 20, no. 20: 5240. https://doi.org/10.3390/ijms20205240
APA StyleLi, Y., & Chen, S. (2019). Fusaricidin Produced by Paenibacillus polymyxa WLY78 Induces Systemic Resistance against Fusarium Wilt of Cucumber. International Journal of Molecular Sciences, 20(20), 5240. https://doi.org/10.3390/ijms20205240






