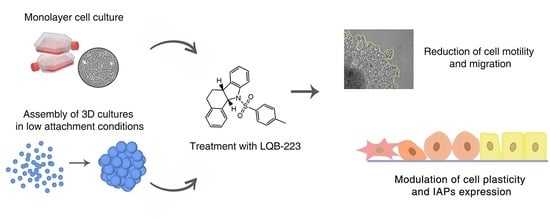
The LQB-223 Compound Modulates Antiapoptotic Proteins and Impairs Breast Cancer Cell Growth and Migration
Abstract
:1. Introduction
2. Results
2.1. Exposure to the LQB-223 Compound Modulates the Migratory Profile of Breast Cancer Cells
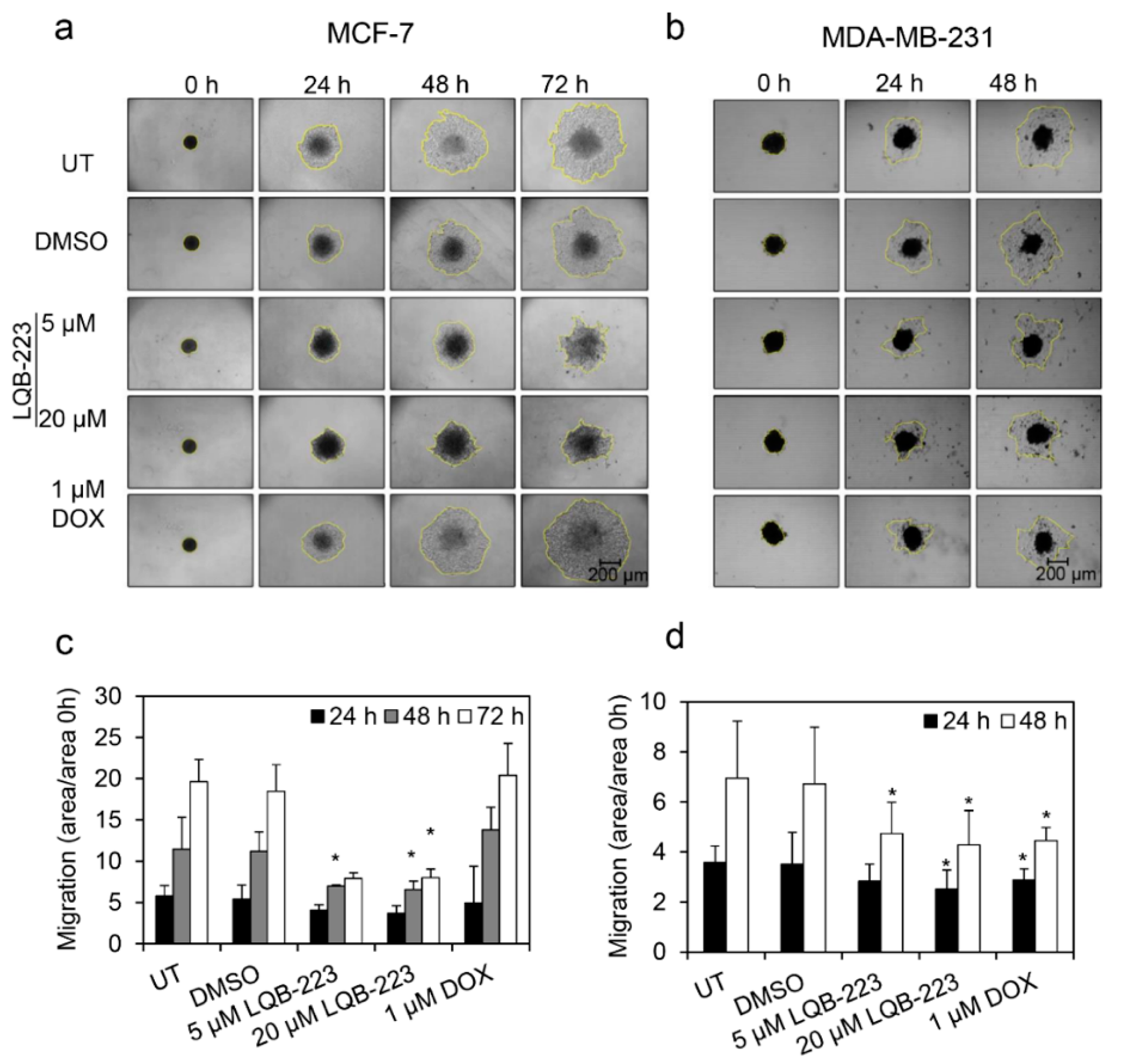
2.2. Cell Motility is Impaired in LQB-223-Treated Breast Cancer Cells
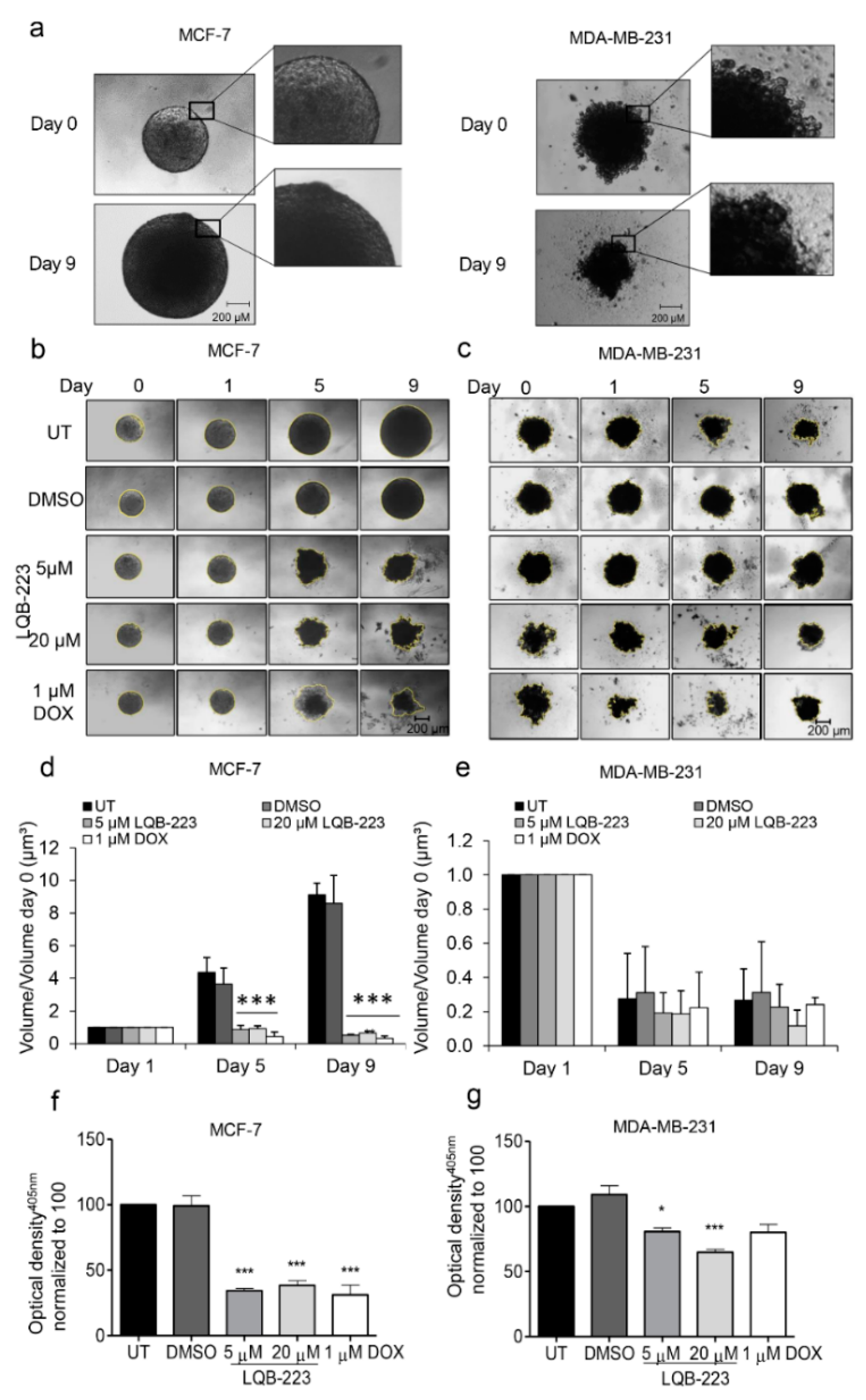
2.3. Treatment with LQB-223 Inhibits Cell Viability and Growth of 3D Cell Models of Breast Cancer
2.4. Cell Migration is Prevented by LQB-223 Treatment of Tridimensional Cultures
2.5. LQB-223 Treatment Leads to Changes in the mRNA Levels of KRT18, CDH1, and C-MYC
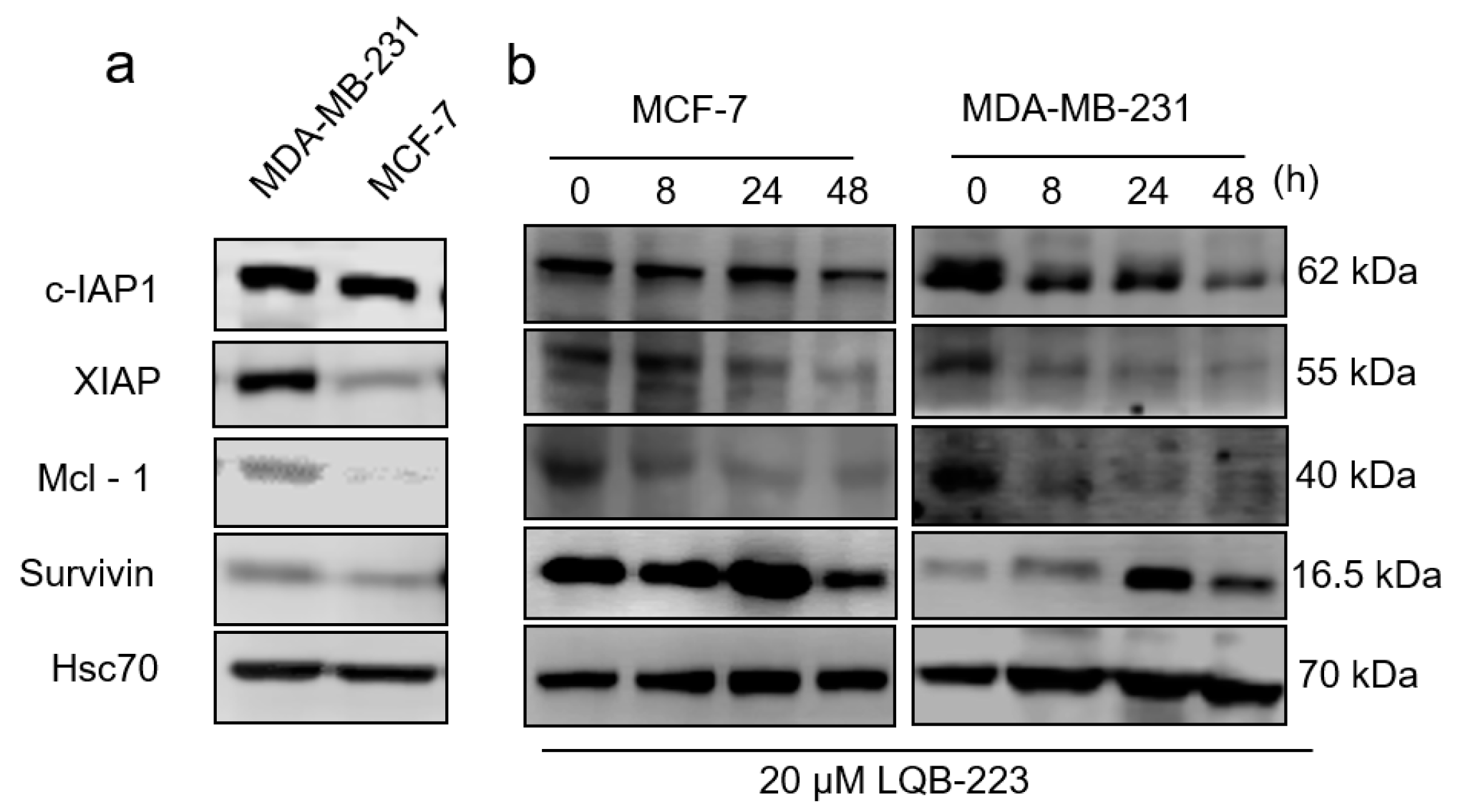
2.6. LQB-223 Treatment Modulates Protein Expression of Apoptotic Regulators
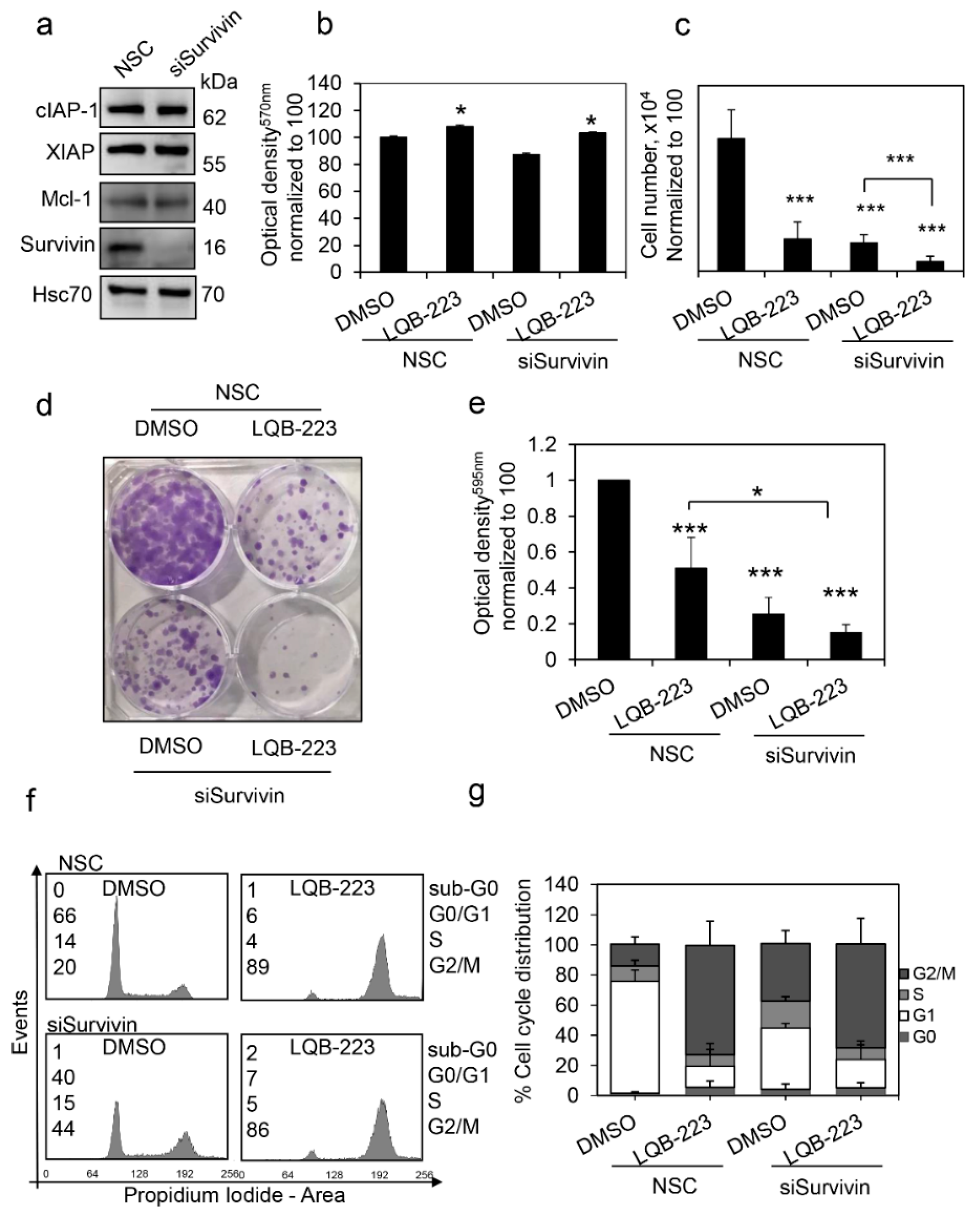
2.7. Survivin Inhibition Potentiates LQB-223-Induced Cytotoxicity in Breast Cancer Cells
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Drug Treatment
4.3. Wound Healing Assay
4.4. Crystal Violet Assay
4.5. Phagokinetic Track Motility Assay
4.6. Formation of 3D Cellular Structures
4.7. Growth Kinetics and Viability of Tridimensional Structures
4.8. Gelatin-Based Migration Assay in 3D Conformation
4.9. Western Blotting
4.10. Survivin Inhibition by Small Interfering RNA (siRNA)
4.11. Real-Time Quantitative PCR (qRT-PCR)
4.12. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Global Cancer Observatory. IARC: Lyon, France, 2018. Available online: https://gco.iarc.fr/ (accessed on 20 August 2019).
- Perou, C.M.; Sørlie, T.; Eisen, M.B.; van de Rijn, M.; Jeffrey, S.S.; Rees, C.A.; Pollack, J.R.; Ross, D.T.; Johnsen, H.; Akslen, L.A.; et al. Molecular portraits of human breast tumours. Nature 2000, 406, 747–752. [Google Scholar] [CrossRef]
- Curtis, C.; Shah, S.P.; Chin, S.F.; Turashvili, G.; Rueda, O.M.; Dunning, M.J.; Speed, D.; Lynch, A.G.; Samarajiwa, S.; Yuan, Y.; et al. The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 2012, 486, 346–352. [Google Scholar] [CrossRef]
- Gu, G.; Dustin, D.; Fuqua, S.A. Targeted therapy for breast cancer and molecular mechanisms of resistance to treatment. Curr. Opin. Pharmacol. 2016, 31, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Shafei, A.; El-Bakly, W.; Sobhy, A.; Wagdy, O.; Reda, A.; Aboelenin, O.; Marzouk, A.; El Habak, K.; Mostafa, R.; Ali, M.A.; et al. A review on the efficacy and toxicity of different doxorubicin nanoparticles for targeted therapy in metastatic breast cancer. Biomed. Pharmacother. 2017, 95, 1209–1218. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Wang, Y.; Kiani, M.F.; Wang, B. Classification, treatment strategy, and associated drug resistance in breast cancer. Clin. Breast Cancer 2016, 16, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Levis, B.E.; Binkley, P.F.; Shapiro, C.L. Cardiotoxic effects of anthracycline-based therapy: What is the evidence and what are the potential harms? Lancet Oncol. 2017, 18, e445–e456. [Google Scholar] [CrossRef]
- Nebigil, C.G.; Désaubry, L. Updates in anthracycline-mediated cardiotoxicity. Front. Pharmacol. 2018, 9, 1262. [Google Scholar] [CrossRef] [PubMed]
- Buarque, C.D.; Salustiano, E.J.; Fraga, K.C.; Alves, B.R.; Costa, P.R. 11a-N-Tosyl-5-deoxi-pterocarpan (LQB-223), a promising prototype for targeting MDR leukemia cell lines. Eur. J. Med. Chem. 2014, 78, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Lemos, L.G.; Nestal de Moraes, G.; Delbue, D.; Vasconcelos Fda, C.; Bernardo, P.S.; Lam, E.W.; Buarque, C.D.; Costa, P.R.; Maia, R.C. 11a-N-Tosyl-5-deoxi-pterocarpan, LQB-223, a novel compound with potent antineoplastic activity toward breast cancer cells with different phenotypes. J. Cancer Res. Clin. Oncol. 2016, 142, 2119–2130. [Google Scholar] [CrossRef]
- Mendes, J.A.; Salustiano, E.J.; Pires, C.S.; Oliveira, T.; Barcellos, J.C.F.; Cifuentes, J.M.C.; Costa, P.R.R.; Rennó, M.N.; Buarque, C.D. 11a-N-tosyl-5-carbapterocarpans: Synthesis, antineoplastic evaluation and in silico prediction of ADMETox properties. Bioorg. Chem. 2018, 80, 585–590. [Google Scholar] [CrossRef]
- Silva, M.M.; Nascimento, E.O.; Júnior, E.F.S.; de Araújo Júnior, J.X.; Santana, C.C.; Grillo, L.A.M.; Figueiredo, I.M.; de Oliveira, R.S.; Costa, P.R.R.; Buarque, C.D.D.; et al. Interaction between bioactive compound 11a-N-tosyl-5-deoxi-pterocarpan (LQB-223) and Calf thymus DNA: Spectroscopic approach, electrophoresis and theoretical studies. Int. J. Biol. Macromol. 2017, 96, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Gordon, R.; Farmer, R.; Pattanayak, A.; Binkowski, A.; Huang, X.; Chavez, J.; Krishna, S.; Voll, E.; Pavese, J.; et al. Precision therapeutic targeting of human cancer cell motility. Nat. Commun. 2018, 9, 2454. [Google Scholar] [CrossRef] [PubMed]
- Breslin, S.; O’Driscoll, L. Three-dimensional cell culture: The missing link in drug discovery. Drug Discov. Today 2013, 18, 240–249. [Google Scholar] [CrossRef] [PubMed]
- Hickman, J.A.; Graeser, R.; de Hoogt, R.; Vidic, S.; Brito, C.; Gutekunst, M.; van der Kuip, H. Three-dimensional models of cancer for pharmacology and cancer cell biology: Capturing tumor complexity in vitro/ex vivo. Biotechnol. J. 2014, 9, 1115–1128. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Liu, W.; Yu, W.; Lu, S.; Liu, M.; Kaplan, D.L.; Wang, X. Three-dimensional tissue culture model of human breast cancer for the evaluation of multidrug resistance. J. Tissue Eng. Regen. Med. 2018, 12, 1959–1971. [Google Scholar] [CrossRef] [PubMed]
- Froehlich, K.; Haeger, J.D.; Heger, J.; Pastuschek, J.; Photini, S.M.; Yan, Y.; Markert, U.R.; Lupp, A.; Pfarrer, C.; Mrowka, R.; et al. Generation of multicellular breast cancer tumor spheroids: Comparison of different protocols. J. Mammary Gland Biol. Neoplasia 2016, 21, 89–98. [Google Scholar] [CrossRef]
- Fulda, S. Regulation of cell migration, invasion and metastasis by IAP proteins and their antagonists. Oncogene 2014, 33, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Young, A.I.; Law, A.M.; Castillo, L.; Chong, S.; Cullen, H.D.; Koehler, M.; Lucas, M.C.; Herzog, S.; Brummer, T.; Lee, E.F.; et al. MCL-1 inhibition provides a new way to suppress breast cancer metastasis and increase sensitivity to dasatinib. Breast Cancer Res. 2016, 18, 125. [Google Scholar] [CrossRef]
- Dunnwald, L.K.; Rossing, M.A.; Li, C.I. Hormone receptor status, tumor characteristics, and prognosis: A prospective cohort of breast cancer patients. Breast Cancer Res. 2007, 9, R6. [Google Scholar] [CrossRef]
- Dai, X.; Xiang, L.; Li, T.; Bai, Z. Cancer hallmarks, biomarkers and breast cancer molecular subtypes. J. Cancer 2016, 7, 1281–1294. [Google Scholar] [CrossRef]
- Desai, K.; Aiyappa, R.; Prabhu, J.S.; Nair, M.G.; Lawrence, P.V.; Korlimarla, A.; Ce, A.; Alexander, A.; Kaluve, R.S.; Manjunath, S.; et al. HR+HER2-breast cancers with growth factor receptor-mediated EMT have a poor prognosis and lapatinib downregulates EMT in MCF-7 cells. Tumour. Biol. 2017, 39, 1010428317695028. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.J.S.; Vani, M.G.; Hsieh, H.W.; Lin, C.C.; Wang, S.Y. Antcin-A Modulates Epithelial-to-Mesenchymal Transition and Inhibits Migratory and Invasive Potentials of Human Breast Cancer Cells via p53-Mediated miR-200c Activation. Planta Med. 2019, 85, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Cejalvo, J.M.; de Martínez Dueñas, E.; Galván, P.; García-Recio, S.; Burgués Gasión, O.; Paré, L.; Antolín, S.; Martinello, R.; Blancas, I.; Adamo, B.; et al. Intrinsic subtypes and gene expression profiles in primary and metastatic breast cancer. Cancer Res. 2017, 77, 2213–2221. [Google Scholar] [CrossRef] [PubMed]
- Sarisozen, C.; Abouzeid, A.H.; Torchilin, V.P. The effect of co-delivery of paclitaxel and curcumin by transferrin-targeted PEG-PE-based mixed micelles on resistant ovarian cancer in 3-D spheroids and in vivo tumors. Eur. J. Pharm. Biopharm. 2014, 88, 539–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.B.; Stein, R.; O’Hare, M.J. Three-dimensional in vitro tissue culture models of breast cancer—A review. Breast Cancer Res. Treat. 2004, 85, 281–291. [Google Scholar] [CrossRef]
- Wang, Y.; Mirza, S.; Wu, S.; Zeng, J.; Shi, W.; Band, H.; Band, V.; Duan, B. 3D hydrogel breast cancer models for studying the effects of hypoxia on epithelial to mesenchymal transition. Oncotarget 2018, 9, 32191–32203. [Google Scholar] [CrossRef]
- Friedrich, J.; Seidel, C.; Ebner, R.; Kunz-Schughart, L.A. Spheroid-based drug screen: Considerations and practical approach. Nat. Protoc. 2009, 4, 309–324. [Google Scholar] [CrossRef]
- Dubois, C.; Daumar, P.; Aubel, C.; Gauthier, J.; Vidalinc, B.; Mounetou, E.; Penault-Llorca, F.; Bamdad, M. The New Synthetic Serum-Free Medium OptiPASS Promotes High Proliferation and Drug Efficacy Prediction on Spheroids from MDA-MB-231 and SUM1315 Triple-Negative Breast Cancer Cell Lines. J. Clin. Med. 2019, 8, 397. [Google Scholar] [CrossRef]
- Wang, Y.; Shao, S.; Luo, M.; Huang, S.; Feng, L.; Yuan, N.; Wu, F.; Dang, C.; Zhao, X. Effects of rat bone marrow-derived mesenchymal stem cells on breast cancer cells with differing hormone receptor status. Oncol. Lett. 2017, 14, 7269–7275. [Google Scholar] [CrossRef]
- Yeung, K.T.; Yang, J. Epithelial-mesenchymal transition in tumor metastasis. Mol. Oncol. 2017, 11, 28–39. [Google Scholar] [CrossRef]
- Ko, J.H.; Yang, M.H.; Baek, S.H.; Nam, D.; Jung, S.H.; Ahn, K.S. Theacrine attenuates epithelial mesenchymal transition in human breast cancer MDA-MB-231 cells. Phytother. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Vuoriluoto, K.; Haugen, H.; Kiviluoto, S.; Mpindi, J.P.; Nevo, J.; Gjerdrum, C.; Tiron, C.; Lorens, J.B.; Ivaska, J. Vimentin regulates EMT induction by Slug and oncogenic H-Ras and migration by governing Axl expression in breast cancer. Oncogene 2011, 30, 1436–1448. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Li, H.; Ren, G. Epithelial-mesenchymal transition and drug resistance in breast cancer (Review). Int. J. Oncol. 2015, 47, 840–848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jackstadt, R.; Hermeking, H. MicroRNAs as regulators and mediators of c-MYC function. Biochim. Biophys. Acta 2015, 1849, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Orlandini, L.F.; Reis, F.J.C.D.; da Silveira, W.A.; Tiezzi, M.G.; de Andrade, J.M.; Ribeiro-Silva, A.; Deaton, R.; Bosland, M.; Tiezzi, D.G. Identification of a Subtype of Poorly Differentiated Invasive Ductal Carcinoma of the Breast Based on Vimentin and E-cadherin Expression. Rev. Bras. Ginecol. Obstet. 2018, 40, 779–786. [Google Scholar] [CrossRef] [Green Version]
- Qian, X.L.; Zhang, J.; Li, P.Z.; Lang, R.G.; Li, W.D.; Sun, H.; Liu, F.F.; Guo, X.J.; Gu, F.; Fu, L. Dasatinib inhibits c-src phosphorylation and prevents the proliferation of Triple-Negative Breast Cancer (TNBC) cells which overexpress Syndecan-Binding Protein (SDCBP). PLoS ONE 2017, 12, e0171169. [Google Scholar] [CrossRef] [PubMed]
- Lou, L.; Yu, Z.; Wang, Y.; Wang, S.; Zhao, Y. c-Src inhibitor selectively inhibits triple-negative breast cancer overexpressed Vimentin in vitro and in vivo. Cancer Sci. 2018, 109, 1648–1659. [Google Scholar] [CrossRef]
- Ocana, A.; Gil-Martin, M.; Antolín, S.; Atienza, M.; Montaño, Á.; Ribelles, N.; Urruticoechea, A.; Falcón, A.; Pernas, S.; Orlando, J.; et al. Efficacy and safety of dasatinib with trastuzumab and paclitaxel in first line HER2-positive metastatic breast cancer: Results from the phase II GEICAM/2010-04 study. Breast Cancer Res. Treat. 2019, 174, 693–701. [Google Scholar] [CrossRef]
- Jung, H.; Kim, B.; Moon, B.I.; Oh, E.S. Cytokeratin 18 is necessary for initiation of TGF-β1-induced epithelial-mesenchymal transition in breast epithelial cells. Mol. Cell Biochem. 2016, 423, 21–28. [Google Scholar] [CrossRef]
- Turksen, K.; Troy, T.C. Barriers built on claudins. J. Cell Sci. 2004, 117, 2435–2447. [Google Scholar] [CrossRef] [Green Version]
- Dias, K.; Dvorkin-Gheva, A.; Hallett, R.M.; Wu, Y.; Hassell, J.; Pond, G.R.; Levine, M.; Whelan, T.; Bane, A.L. Claudin-Low Breast Cancer; Clinical & Pathological Characteristics. PLoS ONE 2017, 12, e0168669. [Google Scholar] [CrossRef]
- Yen, T.Y.; Bowen, S.; Yen, R.; Piryatinska, A.; Macher, B.A.; Timpe, L.C. Glycoproteins in claudin-low breast cancer cell lines have a unique expression profile. J. Proteome Res. 2017, 16, 1391–1400. [Google Scholar] [CrossRef] [PubMed]
- Prat, A.; Karginova, O.; Parker, J.S.; Fan, C.; He, X.; Bixby, L.; Harrell, J.C.; Roman, E.; Adamo, B.; Troester, M.; et al. Characterization of cell lines derived from breast cancers and normal mammary tissues for the study of the intrinsic molecular subtypes. Breast Cancer Res. Treat. 2013, 142, 237–255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sawyer, D.B. Anthracyclines and heart failure. N. Engl. J. Med. 2013, 368, 1154–1156. [Google Scholar] [CrossRef] [PubMed]
- Vejpongsa, P.; Yeh, E.T. Topoisomerase 2β: A promising molecular target for primary prevention of anthracycline-induced cardiotoxicity. Clin. Pharmacol. Ther. 2014, 95, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Mahjoub, M.A.; Bakhshinejad, B.; Sadeghizadeh, M.; Babashah, S. Combination treatment with dendrosomal nanocurcumin and doxorubicin improves anticancer effects on breast cancer cells through modulating CXCR4/NF-κB/Smo regulatory network. Mol. Biol. Rep. 2017, 44, 341–351. [Google Scholar] [CrossRef]
- Zhong, Z.F.; Tan, W.; Tian, K.; Yu, H.; Qiang, W.A.; Wang, Y.T. Combined effects of furanodiene and doxorubicin on the migration and invasion of MDA-MB-231 breast cancer cells in vitro. Oncol. Rep. 2017, 37, 2016–2024. [Google Scholar] [CrossRef]
- Jin, X.; Wei, Y.; Liu, Y.; Lu, X.; Ding, F.; Wang, J.; Yang, S. Resveratrol promotes sensitization to Doxorubicin by inhibiting epithelial-mesenchymal transition and modulating SIRT1/β-catenin signaling pathway in breast cancer. Cancer Med. 2019, 8, 1246–1257. [Google Scholar] [CrossRef]
- Karlsson, M.C.; Gonzalez, S.F.; Welin, J.; Fuxe, J. Epithelial-mesenchymal transition in cancer metastasis through the lymphatic system. Mol. Oncol. 2017, 11, 781–791. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Q.; Zhang, X.; Cai, H.; Zhang, P.; Kong, D.; Ge, X.; Du, M.; Liang, R.; Dong, W. Anticancer effects of plant derived Anacardic acid on human breast cancer MDA-MB-231 cells. Am. J. Transl. Res. 2018, 10, 2424–2434. [Google Scholar]
- Evans, M.K.; Brown, M.C.; Geradts, J.; Bao, X.; Robinson, T.J.; Jolly, M.K.; Vermeulen, P.B.; Palmer, G.M.; Gromeier, M.; Levine, H.; et al. XIAP Regulation by MNK Links MAPK and NFκB Signaling to Determine an Aggressive Breast Cancer Phenotype. Cancer Res. 2018, 78, 1726–1738. [Google Scholar] [CrossRef] [PubMed]
- Sumalatha, K.R.; Abiramasundari, G.; Chetan, G.K.; Divya, T.; Sudhandiran, G.; Sreepriya, M. XIAP inhibitor and antiestrogen embelin abrogates metastasis and augments apoptosis in estrogen receptor positive human breast adenocarcinoma cell line MCF-7. Mol. Biol. Rep. 2014, 41, 935–946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, F.; Ambrosini, G.; Chu, E.Y.; Plescia, J.; Tognin, S.; Marchisio, P.C.; Altieri, D.C. Control of apoptosis and mitotic spindle checkpoint by survivin. Nature 1998, 396, 580–584. [Google Scholar] [CrossRef] [PubMed]
- Altieri, D.C. Survivin, versatile modulation of cell division and apoptosis in cancer. Oncogene 2003, 22, 8581–8589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, T.; Uchida, H. Inhibition of Survivin by Adenovirus Vector Enhanced Paclitaxel-induced Apoptosis in Breast Cancer Cells. Anticancer Res. 2018, 38, 4281–4288. [Google Scholar] [CrossRef]
- Wang, X.; Simpson, E.R.; Brown, K.A. p53: Protection against Tumor Growth beyond Effects on Cell Cycle and Apoptosis. Cancer Res. 2015, 75, 5001–5007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitagawa, M.; Lee, S.H. The chromosomal passenger complex (CPC) as a key orchestrator of orderly mitotic exit and cytokinesis. Front. Cell Dev. Biol. 2015, 3, 14. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, B.; Mani, A.M.; Wu, Z.; Fan, Y.; Li, W.; Wu, Z.H. Survivin inhibitors mitigate chemotherapeutic resistance in breast cancer cells by suppressing genotoxic nuclear factor-κB activation. J. Pharmacol. Exp. Ther. 2018, 366, 184–193. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]



| Gene | Protein | Forward 5’–3’ | Reverse 5’–3’ |
|---|---|---|---|
| CDH1 | E-cadherin | GAATGACAACAAGCCCGAAT | GACCTCCATCACAGAGGTTCC |
| CLDN3 | claudin-3 | CTGCTCTGCTGCTCGTGTCC | TTAGACGTAGTCCTTGCGGTCGTAG |
| VIM | vimentin | GACAATGCGTCTCTGGCACGTCTT | TCCTCCGCCTCCTGCAGGTTCTT |
| KRT18 | cytokeratin-18 | GCGAGAAGGAGACCATGCA | GGTGTTCCCGGATTTTGATCT |
| C-MYC | c-Myc | AATGAAAAGGCCCCCAAGGTAGTTATCC | GTCGTTTCCGCAACAAGTCCTCTTC |
| ACTB | β-actin | GGCGGCACCACCATGTACCCT | AGGGGCCGGACTCGTCATACT |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lemos, L.G.T.; Longo, G.M.d.C.; Mendonça, B.d.S.; Robaina, M.C.; Brum, M.C.M.; Cirilo, C.d.A.; Gimba, E.R.P.; Costa, P.R.R.; Buarque, C.D.; Nestal de Moraes, G.; et al. The LQB-223 Compound Modulates Antiapoptotic Proteins and Impairs Breast Cancer Cell Growth and Migration. Int. J. Mol. Sci. 2019, 20, 5063. https://doi.org/10.3390/ijms20205063
Lemos LGT, Longo GMdC, Mendonça BdS, Robaina MC, Brum MCM, Cirilo CdA, Gimba ERP, Costa PRR, Buarque CD, Nestal de Moraes G, et al. The LQB-223 Compound Modulates Antiapoptotic Proteins and Impairs Breast Cancer Cell Growth and Migration. International Journal of Molecular Sciences. 2019; 20(20):5063. https://doi.org/10.3390/ijms20205063
Chicago/Turabian StyleLemos, Lauana Greicy Tonon, Gabriel Mello da Cunha Longo, Bruna dos Santos Mendonça, Marcela Cristina Robaina, Mariana Concentino Menezes Brum, Caíque de Assis Cirilo, Etel Rodrigues Pereira Gimba, Paulo Roberto Ribeiro Costa, Camilla Djenne Buarque, Gabriela Nestal de Moraes, and et al. 2019. "The LQB-223 Compound Modulates Antiapoptotic Proteins and Impairs Breast Cancer Cell Growth and Migration" International Journal of Molecular Sciences 20, no. 20: 5063. https://doi.org/10.3390/ijms20205063
APA StyleLemos, L. G. T., Longo, G. M. d. C., Mendonça, B. d. S., Robaina, M. C., Brum, M. C. M., Cirilo, C. d. A., Gimba, E. R. P., Costa, P. R. R., Buarque, C. D., Nestal de Moraes, G., & Maia, R. C. (2019). The LQB-223 Compound Modulates Antiapoptotic Proteins and Impairs Breast Cancer Cell Growth and Migration. International Journal of Molecular Sciences, 20(20), 5063. https://doi.org/10.3390/ijms20205063