Physiological and Metabolomic Responses of Kale to Combined Chilling and UV-A Treatment
Abstract
1. Introduction
2. Results
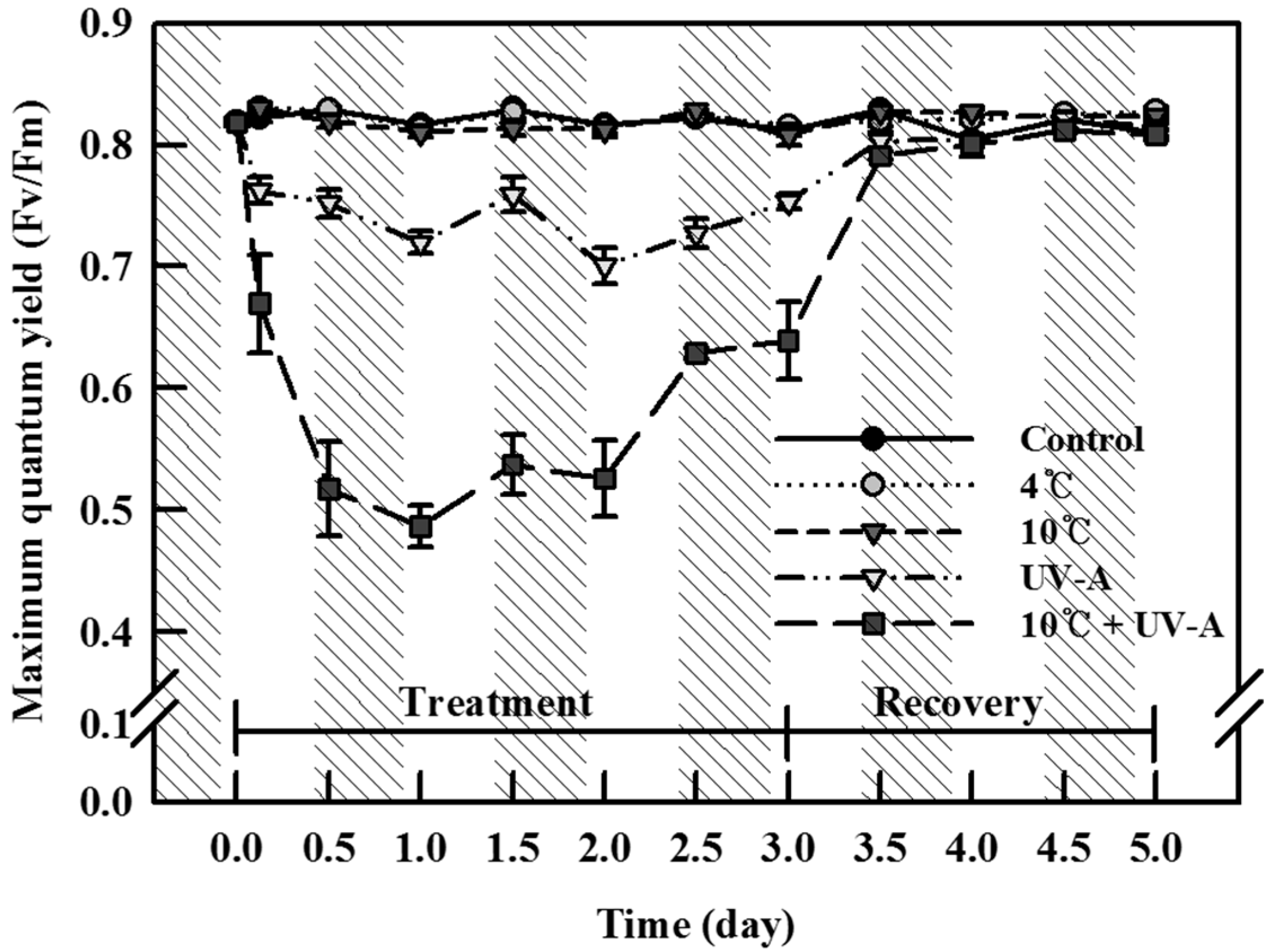
2.1. Chlorophyll Fluorescence
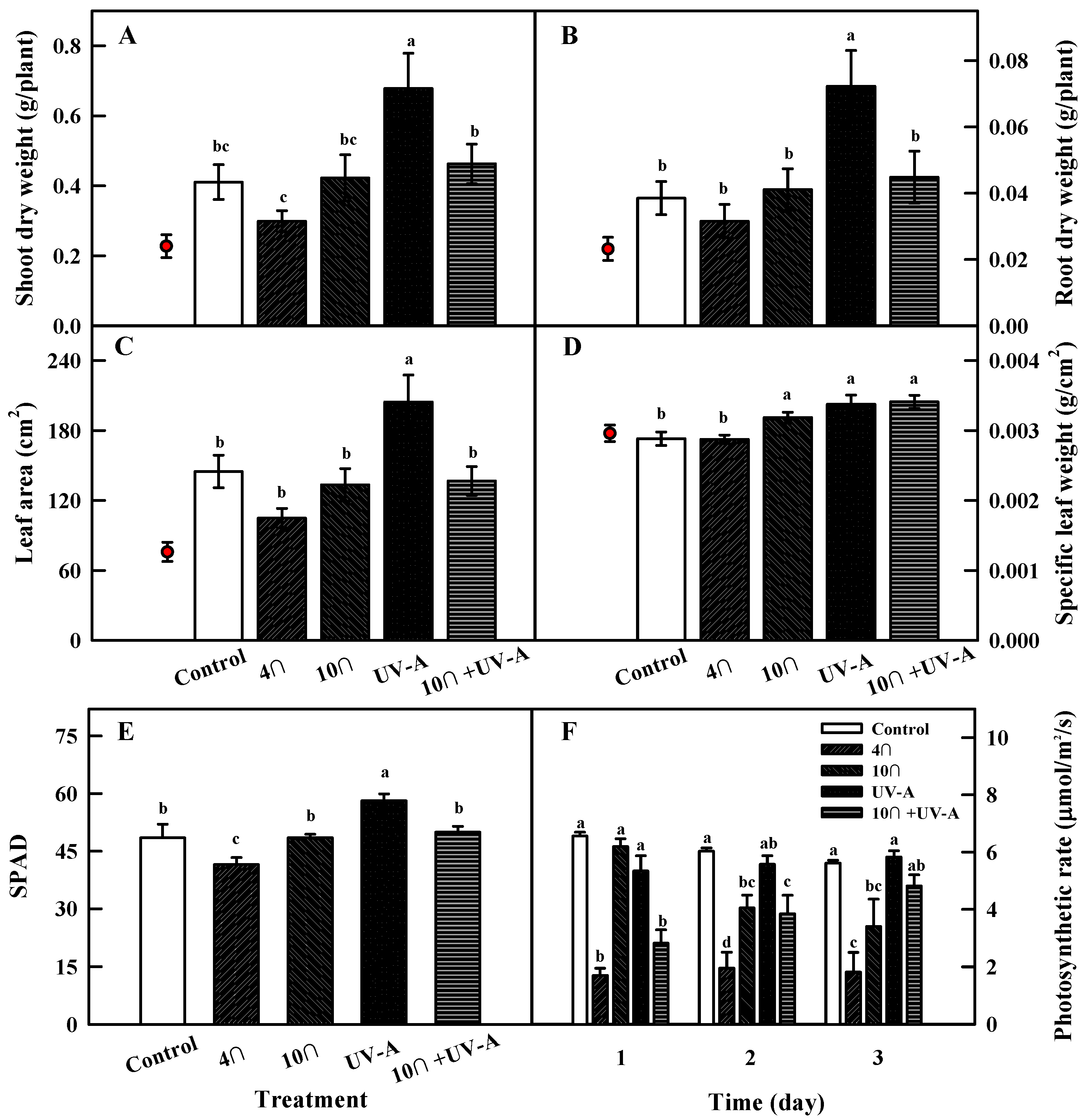
2.2. Growth Characteristics
2.3. Photosynthetic Rate
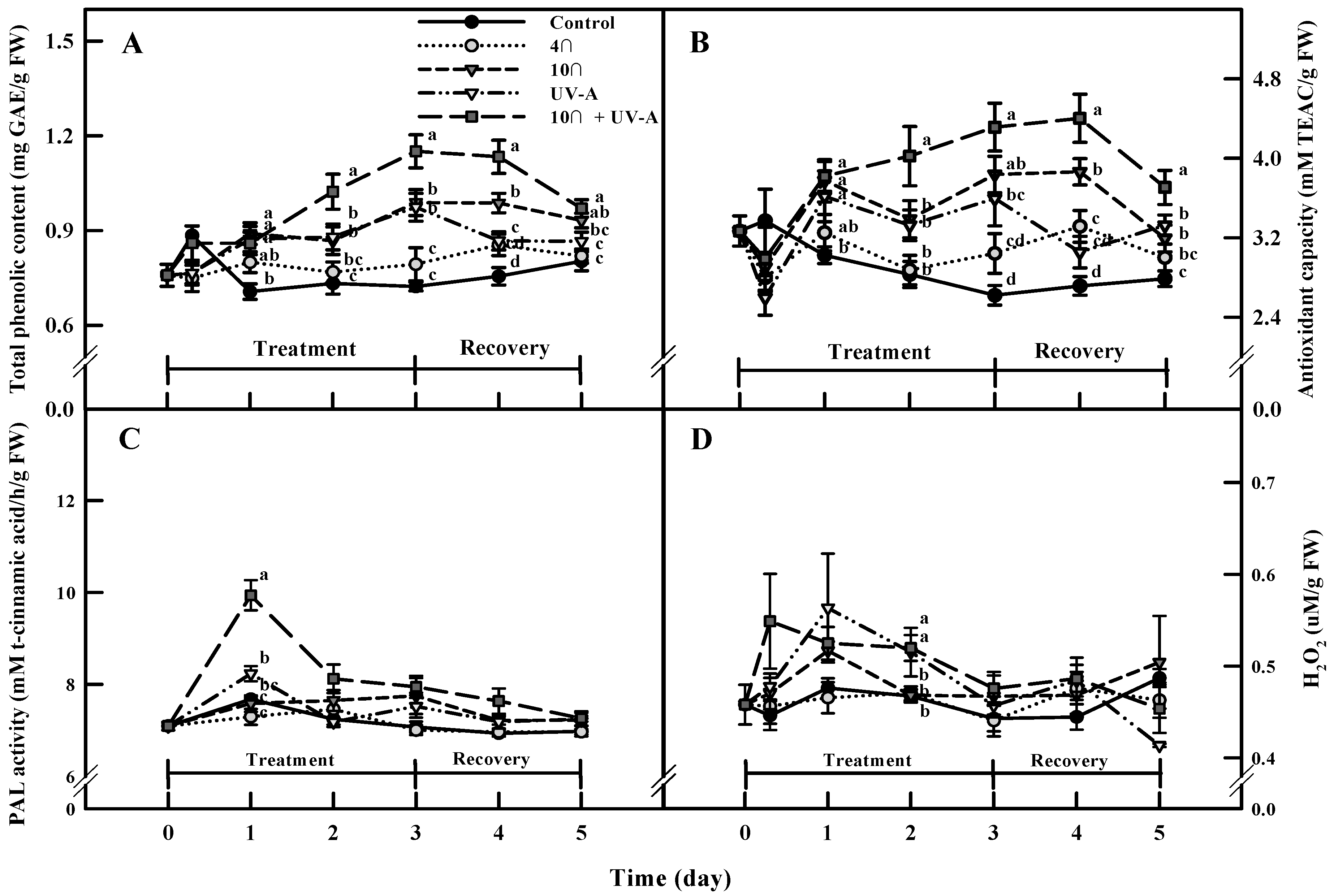
2.4. Total Phenolic Content and Antioxidant Capacity
2.5. Phenylalanine Ammonia-Lyase (PAL) Activity and ROS (H2O2) Levels
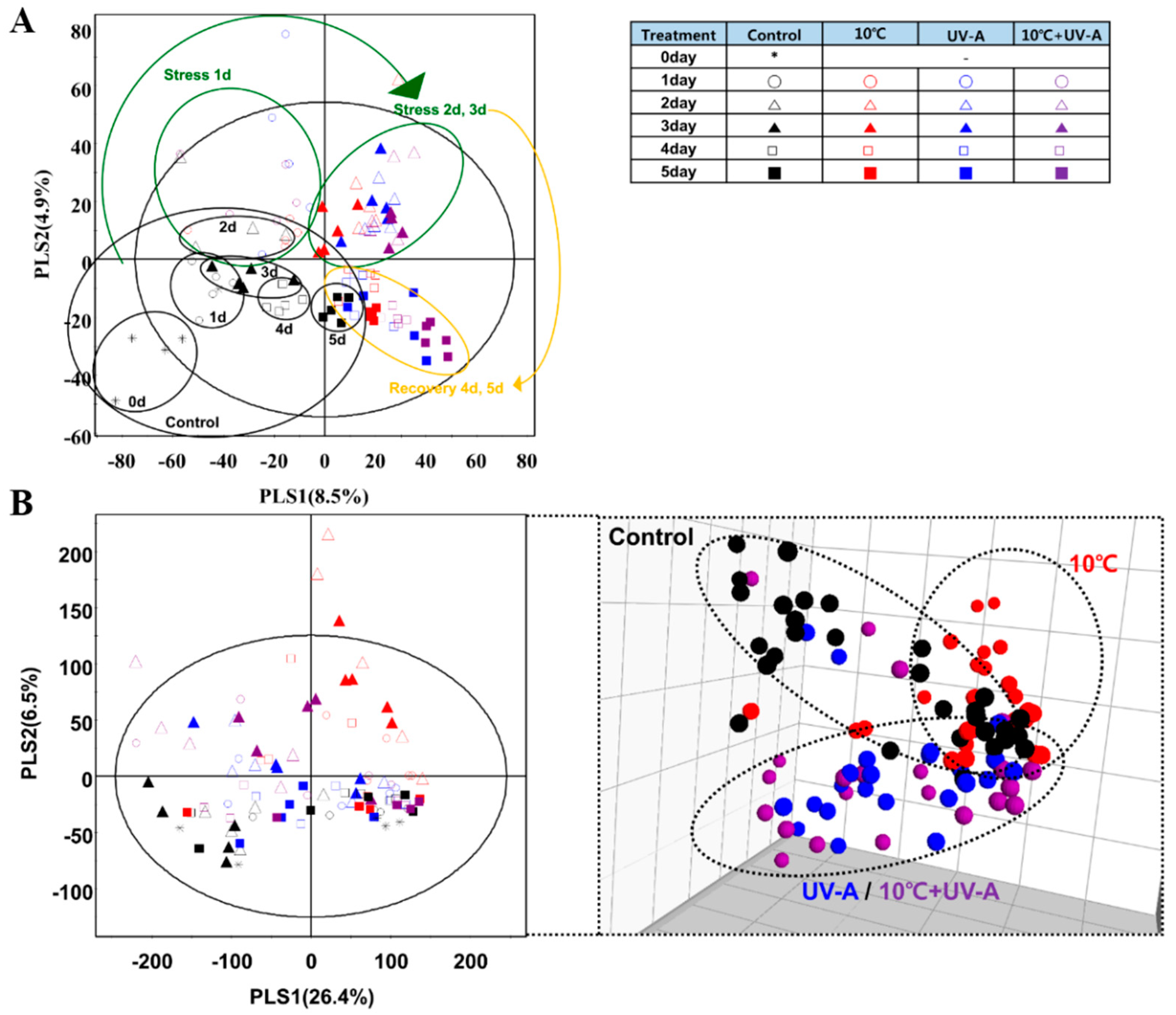
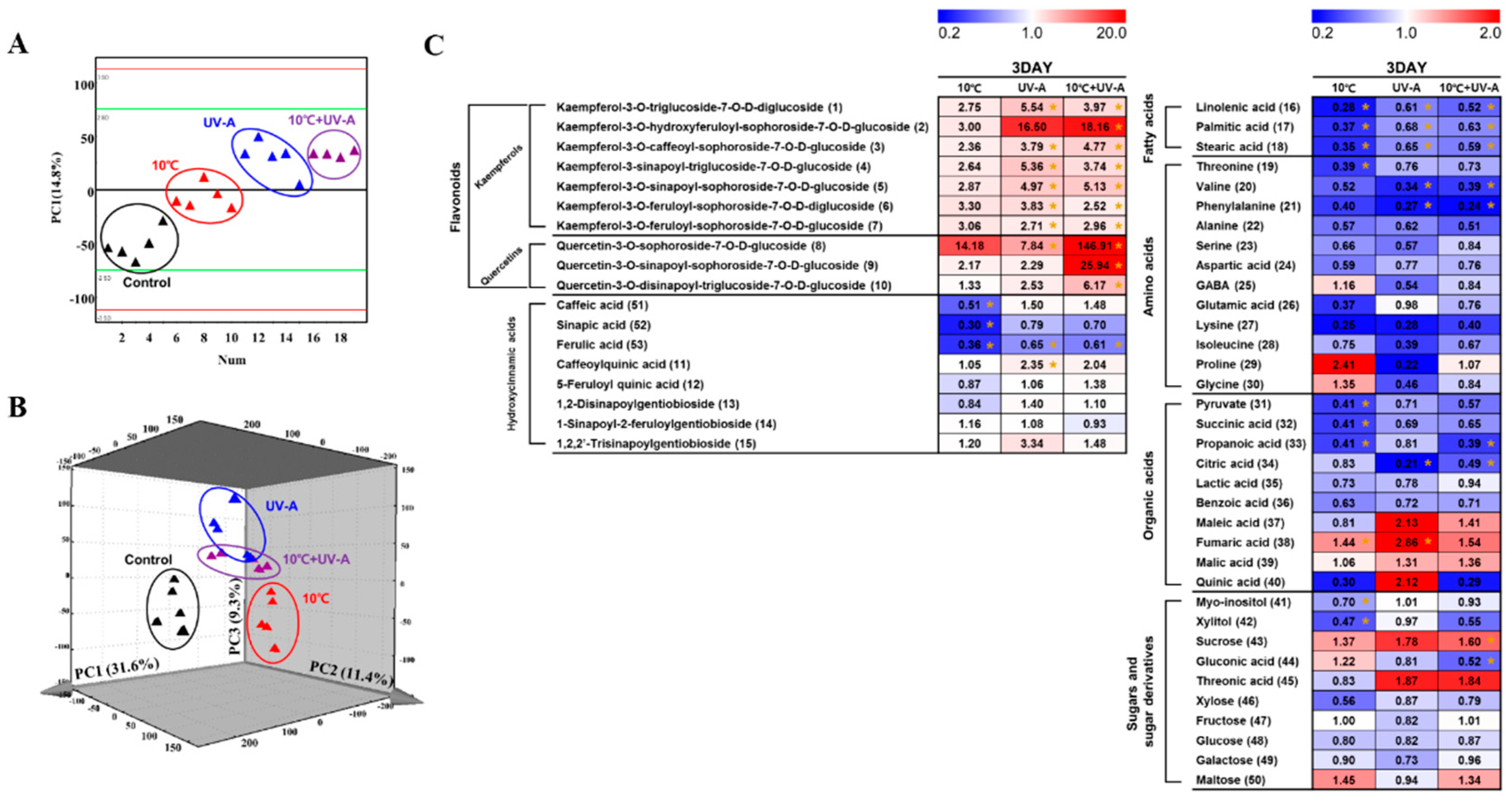
2.6. Nontargeted Metabolite Profiling
2.7. Metabolic Variations at 3 Days of Treatment
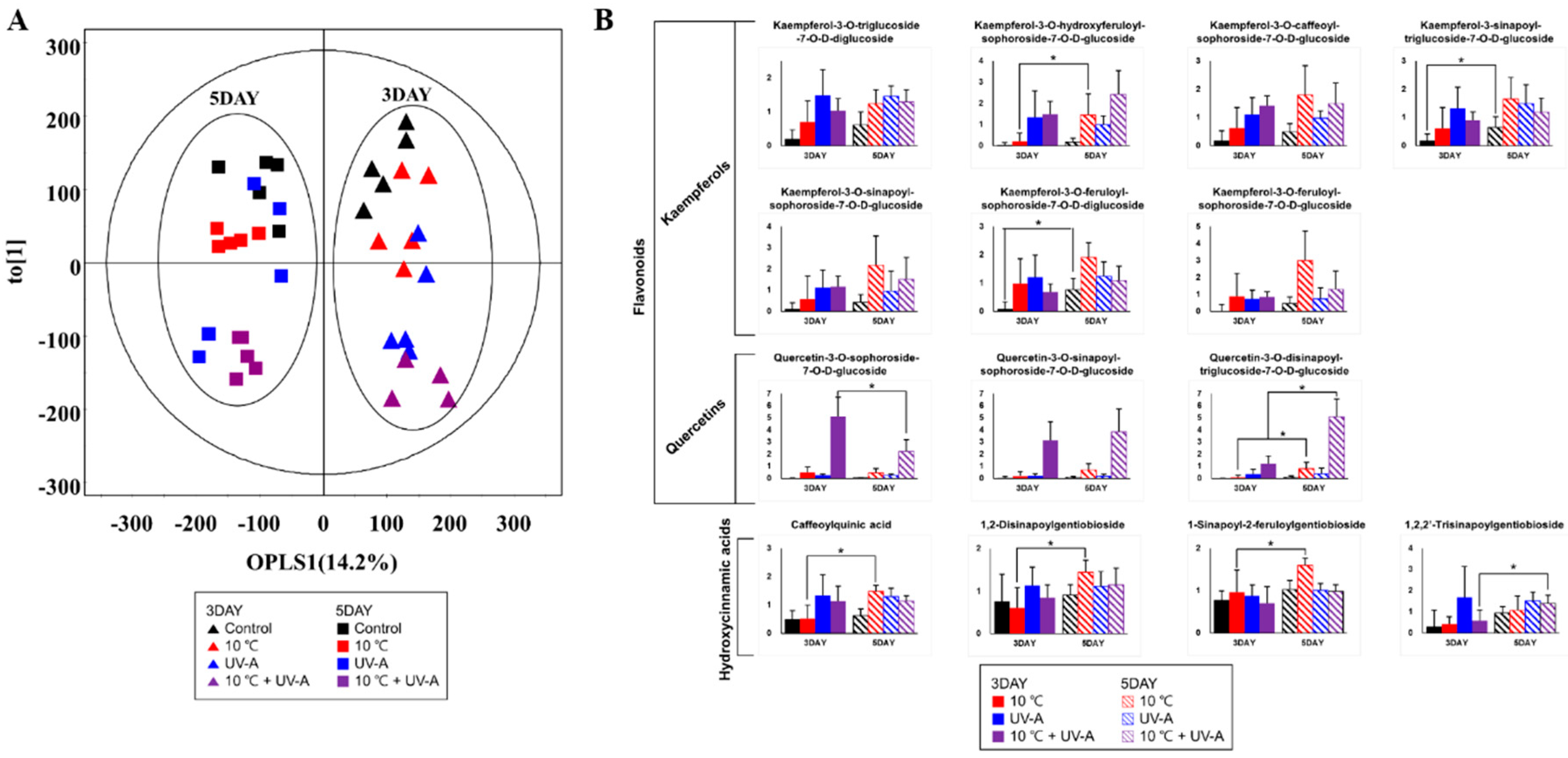
2.8. Changes in Secondary Metabolite Levels after 2 Days of Recovery
3. Discussion
3.1. Effects of Single and combined Stress Treatments on Plant Biomass
3.2. Effects of Single and Combined Stress Treatments on the Biosynthesis of Bioactive Compounds
3.3. Effects of Single and Combination Stress Treatments on Metabolite Levels
4. Materials and Methods
4.1. Plant Materials and Cultivation Conditions
4.2. Environmental Stress Treatments
4.3. Chlorophyll Fluorescence Measurements
4.4. Growth Characteristics
4.5. Chlorophyll Content and Photosynthetic Rate Measurements
4.6. Total Phenolic Content and Antioxidant Capacity
4.7. ROS (H2O2) Measurements
4.8. PAL Activity Measurements
4.9. Metabolite Extraction
4.10. UPLC–Q–TOF–MS and UHPLC–LTQ–IT–MS/MS Analysis
4.11. GC–TOF–MS Analysis
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ROS | Reactive oxygen species |
| ASC | Ascorbate |
| GSH | Glutathione |
| SLW | Specific leaf weight |
| UPLC-Q-TOF-MS | Ultra-performance liquid chromatography-quadrupole-time of flight mass spectrometry |
| GC-TOF-MS | Gas chromatography time-of-flight mass spectrometry |
| PCA | Principal component analysis |
| PLS-DA | Partial least square-discriminant analysis |
| VIP | Variable importance in projection |
| PPFD | Photosynthetic photon flux density |
| PSI | Photosystem I |
| PSII | Photosystem II |
| PAL | Phenylalanine ammonia-lyase |
| UHPLC-LTQ-IT-MS/MS | Ultra-high-performance liquid chromatography linear trap quadrupole tandem mass Spectrometry |
| QC | Quality control |
| MSTFA | N-methyl-N-trimethylsilyl trifluoroacetamide |
| ANOVA | Analysis of variance |
References
- Miller, G.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467. [Google Scholar] [CrossRef] [PubMed]
- Gechev, T.S.; Hille, J. Hydrogen peroxide as a signal controlling plant programmed cell death. J. Cell Biol. 2005, 168, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Rivero, R.M.; Ruiz, J.M.; García, P.C.; López-Lefebre, L.R.; Sánchez, E.; Romero, L. Resistance to cold and heat stress: Accumulation of phenolic compounds in tomato and watermelon plants. Plant Sci. 2001, 160, 315–321. [Google Scholar] [CrossRef]
- Mazorra, L.M.; Nunez, M.; Hechavarria, M.; Coll, F.; Sánchez-Blanco, M.J. Influence of brassinosteroids on antioxidant enzymes activity in tomato under different temperatures. Biol. Plant 2002, 45, 593–596. [Google Scholar] [CrossRef]
- Jansen, M.A.; Hectors, K.; O’Brien, N.M.; Guisez, Y.; Potters, G. Plant stress and human health: Do human consumers benefit from UV-B acclimated crops? Plant Sci. 2008, 175, 449–458. [Google Scholar] [CrossRef]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 1–12. [Google Scholar] [CrossRef]
- Smirnoff, N. The role of active oxygen in the response of plants to water deficit and desiccation. New Phytol. 1993, 125, 27–58. [Google Scholar] [CrossRef]
- Zhang, J.; Kirkham, M.B. Enzymatic responses of the ascorbate-glutathione cycle to drought in sorghum and sunflower plants. Plant Sci. 1996, 113, 139–147. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Agati, G.; Azzarello, E.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012, 196, 67–76. [Google Scholar] [CrossRef]
- Lee, J.H.; Oh, M.M. Short-term low temperature increases phenolic antioxidant levels in kale. Hortic. Environ. Biotechnol. 2015, 56, 588–596. [Google Scholar] [CrossRef]
- Ochoa-Velasco, C.E.; Avila-Sosa, R.; Navarro-Cruz, A.R.; López-Malo, A.; Palou, E. Biotic and abiotic factors to increase bioactive compounds in fruits and vegetables. Food Bioconvers. 2017, 2017, 317–349. [Google Scholar] [CrossRef]
- Albert, A.; Sareedenchai, V.; Heller, W.; Seidlitz, H.K.; Zidorn, C. Temperature is the key to altitudinal variation of phenolics in Arnica montana L. cv. ARBO. Oecologia 2009, 160, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef] [PubMed]
- León-Chan, R.G.; López-Meyer, M.; Osuna-Enciso, T.; Sañudo-Barajas, J.A.; Heredia, J.B.; León-Félix, J. Low temperature and ultraviolet-B radiation affect chlorophyll content and induce the accumulation of UV-B-absorbing and antioxidant compounds in bell pepper (Capsicum annuum) plants. Environ. Exp. Bot. 2017, 139, 143–151. [Google Scholar] [CrossRef]
- Elstner, E.F.; Oswald, W. Mechanisms of oxygen activation during plant stress. Proc. R. Soc. Edinb. 1994, 102, 131–154. [Google Scholar] [CrossRef]
- Xu, S.; Jiang, Y.; Cui, W.; Jin, Q.; Zhang, Y.; Bu, D.; Fu, J.; Wang, R.; Zhou, F.; Shen, W. Hydrogen enhances adaptation of rice seedlings to cold stress via the reestablishment of redox homeostasis mediated by miRNA expression. Plant Soil 2017, 414, 15–67. [Google Scholar] [CrossRef]
- Allen, D.J.; Ort, D.R. Impacts of chilling temperatures on photosynthesis in warm-climate plants. Trends Plant Sci. 2001, 6, 36–42. [Google Scholar] [CrossRef]
- Verdaguer, D.; Jansen, M.A.; Llorens, L.; Morales, L.O.; Neugart, S. UV-A radiation effects on higher plants: Exploring the known unknown. Plant Sci. 2017, 255, 72–81. [Google Scholar] [CrossRef]
- Lee, J.H.; Son, K.H.; Oh, M.M. Short-term ultraviolet (UV)-A light emitting diode (LED) radiation improves biomass and bioactive compounds of kale. Front. Plant Sci. 2019, 10, 1042. [Google Scholar] [CrossRef]
- Tezuka, T.; Yamaguchi, F.; Ando, Y. Physiological activation in radish plants by UV-A radiation. J. Photochem. Photobiol. B Biol. 1994, 24, 33–40. [Google Scholar] [CrossRef]
- Zhang, Z.; Jia, Y.; Gao, H.; Zhang, L.; Li, H.; Meng, Q. Characterization of PSI recovery after chilling-induced photoinhibition in cucumber (Cucumis sativus L.) leaves. Planta 2011, 234, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Coto-Montes, A.; Poeggeler, B. Circadian rhythms, oxidative stress, and antioxidative defense mechanisms. Chronobiol. Int. 2003, 20, 921–962. [Google Scholar] [CrossRef] [PubMed]
- Pastori, G.; Foyer, C.H.; Mullineaux, P. Low temperature-induced changes in the distribution of H2O2 and antioxidants between the bundle sheath and mesophyll cells of maize leaves. J. Exp. Bot. 2000, 51, 107–113. [Google Scholar] [CrossRef]
- Weidner, S.; Kordala, E.; Brosowska-Arendt, W.; Karamac, M.; Kosinska, A.; Amarowicz, R. Phenolic compounds and properties of antioxidants in grapevine roots [Vitis vinifera L.] under low-temperature stress followed by recovery. Acta Soc. Bot. Pol. 2009, 78, 279–286. [Google Scholar] [CrossRef]
- Kotilainen, T.; Tegelberg, R.; Julkunen-tiitto, R.; Lindfors, A.; Aphalo, P.J. Metabolite specific effects of solar UV-A and UV-B on alder and birch leaf phenolics. Glob. Chang. Biol. 2008, 14, 1294–1304. [Google Scholar] [CrossRef]
- Lee, M.J.; Son, J.E.; Oh, M.M. Growth and phenolic compounds of Lactuca sativa L. grown in a closed-type plant production system with UV-A,-B, or-C lamp. J. Sci. Food Agric. 2014, 94, 197–204. [Google Scholar] [CrossRef]
- Brazaitytė, A.; Viršilė, A.; Jankauskienė, J.; Sakalauskienė, S.; Samuolienė, G.; Sirtautas, R.; Novičkovas, A.; Dabašinskas, L.; Miliauskienė, J.; Vaštakaitė, V.; et al. Effect of supplemental UV-A irradiation in solid-state lighting on the growth and phytochemical content of microgreens. Int. Agrophys. 2015, 29, 13–22. [Google Scholar] [CrossRef]
- Ebrahim, M.K.H. Stress tolerance responses of two cotton cultivars exposed to ultraviolet-A (366 nm) radiation: Photosynthetic performance and chemical constituents. Acta Agron. Hung. 2004, 52, 113–124. [Google Scholar] [CrossRef]
- Sivadasan, U.; Randriamanana, T.R.; Julkunen-Tiitto, R.; Nybakken, L. The vegetative buds of Salix myrsinifolia are responsive to elevated UV-B and temperature. Plant Physiol. Biochem. 2015, 93, 66–73. [Google Scholar] [CrossRef]
- Bilger, W.; Rolland, M.; Nybakken, L. UV screening in higher plants induced by low temperature in the absence of UV-B radiation. Photochem. Photobiol. Sci. 2007, 6, 190–195. [Google Scholar] [CrossRef]
- Britt, A. Repair of damaged bases. In The Arabidopsis Book; Somerville, C.R., Meyerowitz, E.M., Eds.; American Society Plant Biologists: Rockville, MD, USA, 2002. [Google Scholar] [CrossRef]
- Ramakrishna, A.; Ravishankar, G.A. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal. Behav. 2011, 6, 1720–1731. [Google Scholar] [CrossRef] [PubMed]
- Steindal, A.L.H.; Rødven, R.; Hansen, E.; Mølmann, J. Effects of photoperiod, growth temperature and cold acclimatisation on glucosinolates, sugars and fatty acids in kale. Food Chem. 2015, 174, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Neugart, S.; Krumbein, A.; Zrenner, R. Influence of light and temperature on gene expression leading to accumulation of specific flavonol glycosides and hydroxycinnamic acid derivatives in kale (Brassica oleracea var. sabellica). Front. Plant Sci. 2016, 7, 326. [Google Scholar] [CrossRef] [PubMed]
- Zietz, M.; Weckmüller, A.; Schmidt, S.; Rohn, S.; Schreiner, M.; Krumbein, A.; Kroh, L.W. Genotypic and climatic influence on the antioxidant activity of flavonoids in kale (Brassica oleracea var. sabellica). J. Agric. Food Chem. 2010, 58, 2123–2130. [Google Scholar] [CrossRef] [PubMed]
- Solecka, D. Role of phenylpropanoid compounds in plant responses to different stress factors. Acta Physiol. Plant. 1997, 19, 257–268. [Google Scholar] [CrossRef]
- Chalker-Scott, L. The role of phenolic compounds in plant stress responses. In Low Temperature Stress Physiology in Crop; Paul, H.L., Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1989. [Google Scholar]
- Galili, G. The aspartate-family pathway of plants: Linking production of essential amino acids with energy and stress regulation. Plant Signal. Behav. 2011, 6, 192–195. [Google Scholar] [CrossRef]
- Hildebrandt, T.M.; Nesi, A.N.; Araújo, W.L.; Braun, H.P. Amino acid catabolism in plants. Mol. Plant 2015, 8, 1563–1579. [Google Scholar] [CrossRef]
- Zhang, X.; Ding, X.; Ji, Y.; Wang, S.; Chen, Y.; Luo, J.; Shen, Y.; Peng, L. Measurement of metabolite variations and analysis of related gene expression in Chinese liquorice (Glycyrrhiza uralensis) plants under UV-B irradiation. Sci. Rep. 2018, 8, 6144. [Google Scholar] [CrossRef]
- Dixon, R.A.; Paiva, N.L. Stress-induced phenylpropanoid metabolism. Plant Cell 1995, 7, 1085. [Google Scholar] [CrossRef]
- Amarowicz, R.; Weidner, S.; Wójtowicz, I.; Karmac, M.; Kosinska, A.; Rybarczyk, A. Influence of low-temperature stress on changes in the composition of grapevine leaf phenolic compounds and their antioxidant properties. Funct. Plant Sci. Biot. 2010, 4, 90–96. [Google Scholar]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Calif. Agric. Exp. Stat. Circ. 1950, 347, 1–37. [Google Scholar]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence-a practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Gillespie, K.M. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protoc. 2007, 2, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Miller, N.J.; Rice-Evans, C.A. Spectrophotometric determination of antioxidant activity. Redox Rep. 1996, 2, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Boo, H.O.; Heo, B.G.; Gorinstein, S.; Chon, S.U. Positive effects of temperature and growth conditions on enzymatic and antioxidant status in lettuce plants. Plant Sci. 2011, 181, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Jung, E.S.; Singh, D.; Kim, S.; Lee, Y.W.; Kim, J.G.; Lee, C.H. Spatial (cap & stipe) metabolomic variations affect functional components between brown and white beech mushrooms. Food Res. Int. 2017, 102, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.S.; Lee, S.; Lim, S.H.; Ha, S.H.; Liu, K.H.; Lee, C.H. Metabolite profiling of the short-term responses of rice leaves (Oryza sativa cv. Ilmi) cultivated under different LED lights and its correlations with antioxidant activities. Plant Sci. 2013, 210, 61–69. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-H.; Kwon, M.C.; Jung, E.S.; Lee, C.H.; Oh, M.-M. Physiological and Metabolomic Responses of Kale to Combined Chilling and UV-A Treatment. Int. J. Mol. Sci. 2019, 20, 4950. https://doi.org/10.3390/ijms20194950
Lee J-H, Kwon MC, Jung ES, Lee CH, Oh M-M. Physiological and Metabolomic Responses of Kale to Combined Chilling and UV-A Treatment. International Journal of Molecular Sciences. 2019; 20(19):4950. https://doi.org/10.3390/ijms20194950
Chicago/Turabian StyleLee, Jin-Hui, Min Cheol Kwon, Eun Sung Jung, Choong Hwan Lee, and Myung-Min Oh. 2019. "Physiological and Metabolomic Responses of Kale to Combined Chilling and UV-A Treatment" International Journal of Molecular Sciences 20, no. 19: 4950. https://doi.org/10.3390/ijms20194950
APA StyleLee, J.-H., Kwon, M. C., Jung, E. S., Lee, C. H., & Oh, M.-M. (2019). Physiological and Metabolomic Responses of Kale to Combined Chilling and UV-A Treatment. International Journal of Molecular Sciences, 20(19), 4950. https://doi.org/10.3390/ijms20194950




