Avian Expression Patterns and Genomic Mapping Implicate Leptin in Digestion and TNF in Immunity, Suggesting That Their Interacting Adipokine Role Has Been Acquired Only in Mammals
Abstract
1. Introduction
2. Results
2.1. Mapping Chicken TNF onto Chicken GGA16 Using Radiation-Hybrid (RH) Panel
2.2. Mapping the Chicken TNF and its Syntenic Genes Using Fluorescent in Situ Hybridization with Tyramide Signal Amplification (FISH-TSA)
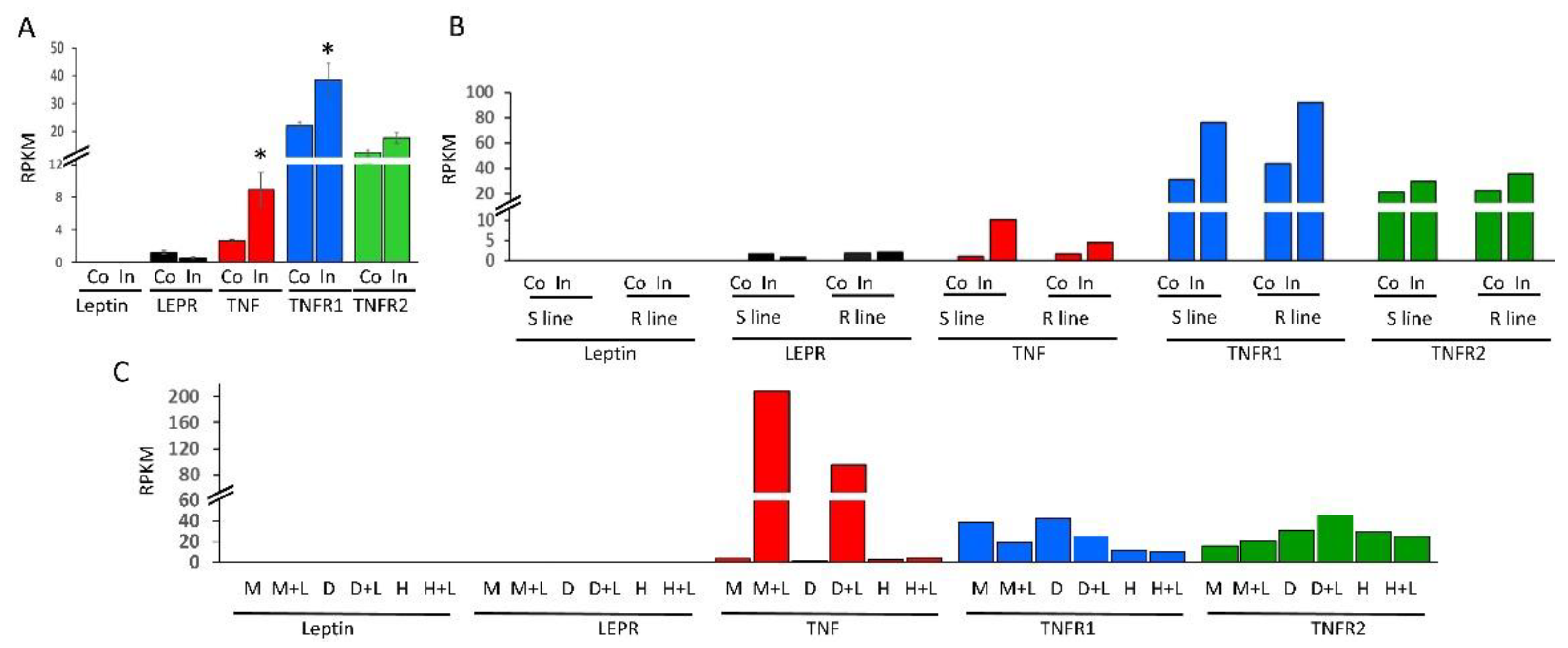
2.3. Expression Profiling of mRNAs of TNF, Leptin and Their Cognate Receptors in Immune Cells
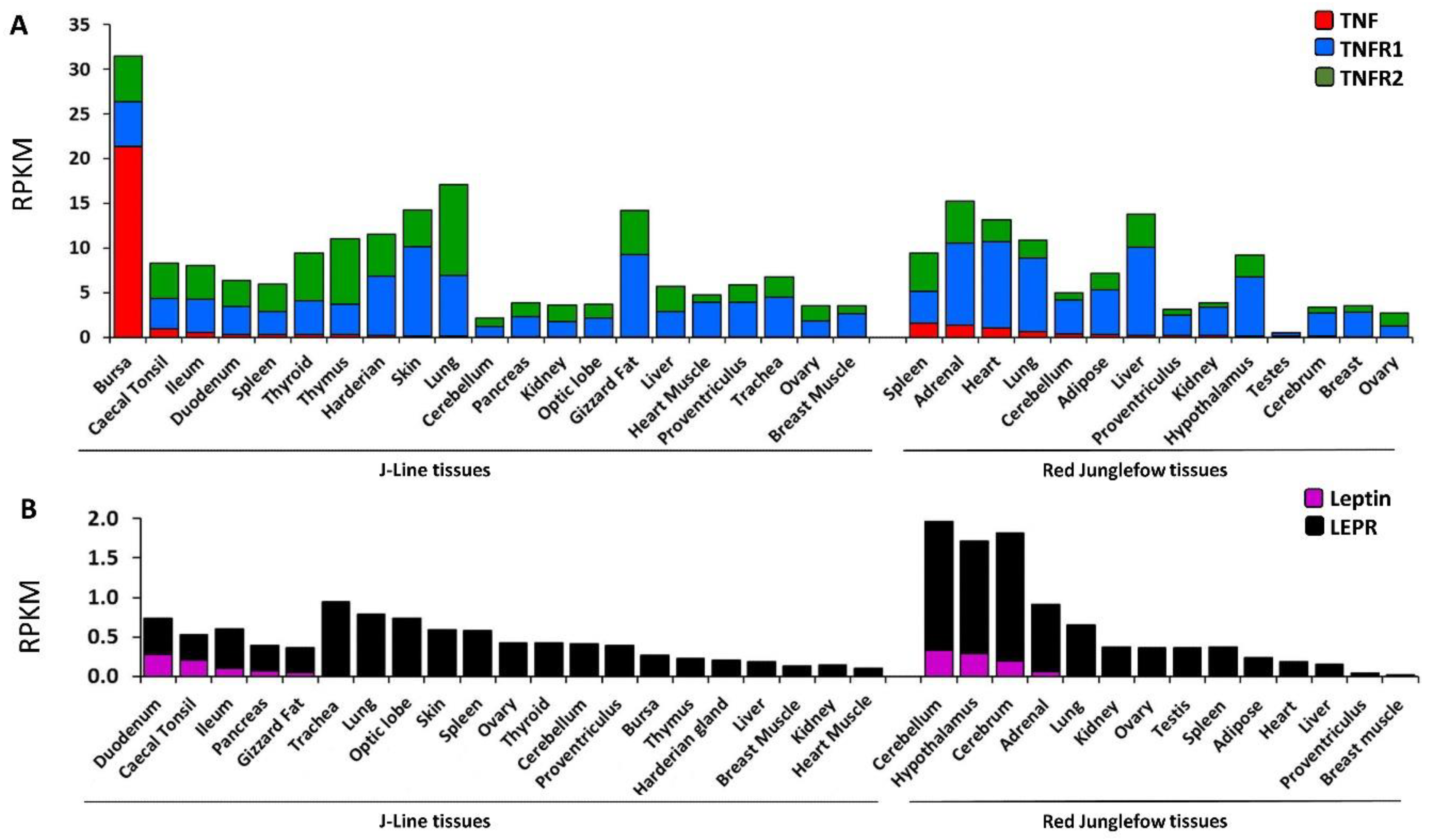
2.4. Expression Profiling of Leptin, TNF and Their Cognate Receptors in Variety of Chicken Tissues
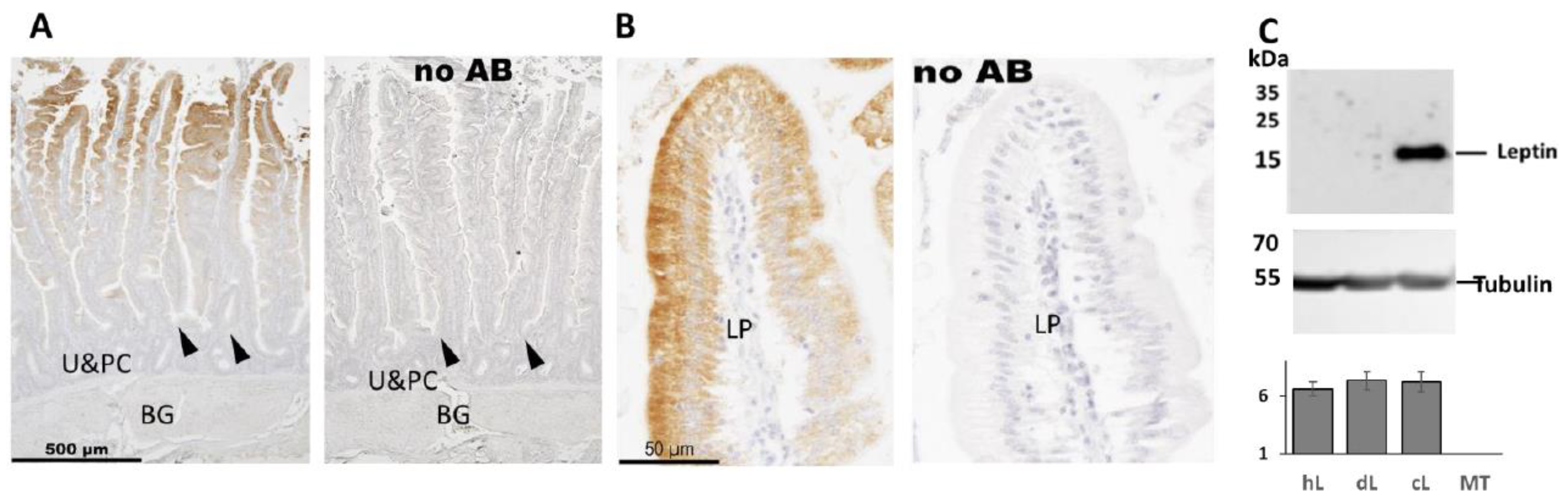
2.5. Immunohistochemistry Analysis of Leptin in Chicken Duodenum
3. Discussion
4. Materials and Methods
4.1. RH-Mapping
4.2. FISH-TSA Analysis
4.2.1. Chromosome Preparation and Cell Culture
4.2.2. cDNA Probe Preparation and Labelling
4.2.3. FISH-TSA
4.2.4. Microscopy and Processing of FISH-TSA Images
4.3. Bioinformatic Analysis
4.4. Animals and Tissue Sampling
4.5. Antibodies and Western Analysis
4.6. IHC Analysis
4.7. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J. The long road to leptin. J. Clin. Investig. 2016, 126, 4727–4734. [Google Scholar] [CrossRef] [PubMed]
- Uysal, K.T.; Wiesbrock, S.M.; Marino, M.W.; Hotamisligil, G.S. Protection from obesity-induced insulin resistance in mice lacking TNF-alpha function. Nature 1997, 389, 610–614. [Google Scholar] [CrossRef] [PubMed]
- Brenner, D.; Blaser, H.; Mak, T.W. Regulation of tumour necrosis factor signalling: Live or let die. Nat. Rev. Immunol. 2015, 15, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Carbone, F.; La Rocca, C.; Matarese, G. Immunological functions of leptin and adiponectin. Biochimie 2012, 94, 2082–2088. [Google Scholar] [CrossRef] [PubMed]
- Lord, G.M.; Matarese, G.; Howard, J.K.; Baker, R.J.; Bloom, S.R.; Lechler, R.I. Leptin modulates the T-cell immune response and reverses starvation-induced immunosuppression. Nature 1998, 394, 897–901. [Google Scholar] [CrossRef] [PubMed]
- Loffreda, S.; Yang, S.Q.; Lin, H.Z.; Karp, C.L.; Brengman, M.L.; Wang, D.J.; Klein, A.S.; Bulkley, G.B.; Bao, C.; Noble, P.W.; et al. Leptin regulates proinflammatory immune responses. FASEB J. 1998, 12, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.Q. How much have we learnt about the TNF family of cytokines? Cytokine 2018, 101, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Grunfeld, C.; Zhao, C.; Fuller, J.; Pollack, A.; Moser, A.; Friedman, J.; Feingold, K.R. Endotoxin and cytokines induce expression of leptin, the ob gene product, in hamsters. J. Clin. Investig. 1996, 97, 2152–2157. [Google Scholar] [CrossRef]
- Park, H.K.; Ahima, R.S. Physiology of leptin: Energy homeostasis, neuroendocrine function and metabolism. Metabolism 2015, 64, 24–34. [Google Scholar] [CrossRef]
- Magor, K.E.; Miranzo Navarro, D.; Barber, M.R.; Petkau, K.; Fleming-Canepa, X.; Blyth, G.A.; Blaine, A.H. Defense genes missing from the flight division. Dev. Comp. Immunol. 2013, 41, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Dakovic, N.; Terezol, M.; Pitel, F.; Maillard, V.; Elis, S.; Leroux, S.; Lagarrigue, S.; Gondret, F.; Klopp, C.; Baeza, E.; et al. The loss of adipokine genes in the chicken genome and implications for insulin metabolism. Mol. Biol. Evol. 2014, 31, 2637–2646. [Google Scholar] [CrossRef] [PubMed]
- Bornelov, S.; Seroussi, E.; Yosefi, S.; Pendavis, K.; Burgess, S.C.; Grabherr, M.; Friedman-Einat, M.; Andersson, L. Correspondence on Lovell et al.: Identification of chicken genes previously assumed to be evolutionarily lost. Genome Biol. 2017, 18, 112. [Google Scholar] [CrossRef] [PubMed]
- Rohde, F.; Schusser, B.; Hron, T.; Farkasova, H.; Plachy, J.; Hartle, S.; Hejnar, J.; Elleder, D.; Kaspers, B. Characterization of chicken tumor necrosis factor-alpha, a long missed cytokine in birds. Front. Immunol. 2018, 9, 605. [Google Scholar] [CrossRef] [PubMed]
- Seroussi, E.; Cinnamon, Y.; Yosefi, S.; Genin, O.; Smith, J.G.; Rafati, N.; Bornelov, S.; Andersson, L.; Friedman-Einat, M. Identification of the Long-Sought Leptin in Chicken and Duck Expression Pattern of the Highly GC-Rich Avian leptin Fits an Autocrine/Paracrine Rather Than Endocrine Function. Endocrinology 2016, 157, 737–751. [Google Scholar] [CrossRef] [PubMed]
- Elleder, D.; Kaspers, B. After TNF-alpha, still playing hide-and-seek with chicken genes. Poult. Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Rozenboim, I.; Mahato, J.; Cohen, N.A.; Tirosh, O. Low protein and high-energy diet: A possible natural cause of fatty liver hemorrhagic syndrome in caged White Leghorn laying hens. Poult. Sci. 2016, 95, 612–621. [Google Scholar] [CrossRef]
- Bridgham, J.T.; Johnson, A.L. Expression and regulation of Fas antigen and tumor necrosis factor receptor type I in hen granulosa cells. Biol. Reprod. 2001, 65, 733–739. [Google Scholar] [CrossRef]
- Abdalla, S.A.; Horiuchi, H.; Furusawa, S.; Matsuda, H. Molecular study on chicken tumor necrosis factor receptor-II and tumor necrosis factor receptor-associated factor-5. Vet. Immunol. Immunopathol. 2004, 98, 31–41. [Google Scholar] [CrossRef]
- Horev, G.; Einat, P.; Aharoni, T.; Eshdat, Y.; Friedman-Einat, M. Molecular cloning and properties of the chicken leptin-receptor (CLEPR) gene. Mol. Cell. Endocrinol. 2000, 162, 95–106. [Google Scholar] [CrossRef]
- Ohkubo, T.; Tanaka, M.; Nakashima, K. Structure and tissue distribution of chicken leptin receptor (cOb-R) mRNA. Biochim. Biophys. Acta 2000, 1491, 303–308. [Google Scholar] [CrossRef]
- Dunn, I.C.; Boswell, T.; Friedman-Einat, M.; Eshdat, Y.; Burt, D.W.; Paton, I.R. Mapping of the leptin receptor gene (LEPR) to chicken chromosome 8. Anim. Genet. 2000, 31, 290. [Google Scholar] [CrossRef] [PubMed]
- Idriss, H.T.; Naismith, J.H. TNF alpha and the TNF receptor superfamily: Structure-function relationship(s). Microsc. Res. Tech. 2000, 50, 184–195. [Google Scholar] [CrossRef]
- Friedman-Einat, M.; Seroussi, E. Quack leptin. BMC Genom. 2014, 15, 551. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Friedman-Einat, M.; Cogburn, L.A.; Yosefi, S.; Hen, G.; Shinder, D.; Shirak, A.; Seroussi, E. Discovery and characterization of the first genuine avian leptin gene in the rock dove (Columba livia). Endocrinology 2014, 155, 3376–3384. [Google Scholar] [CrossRef]
- Prokop, J.W.; Schmidt, C.; Gasper, D.; Duff, R.J.; Milsted, A.; Ohkubo, T.; Ball, H.C.; Shawkey, M.D.; Mays, H.L., Jr.; Cogburn, L.A.; et al. Discovery of the elusive leptin in birds: Identification of several ‘missing links’ in the evolution of leptin and its receptor. PLoS ONE 2014, 9, e92751. [Google Scholar] [CrossRef]
- Huang, G.; Li, J.; Wang, H.; Lan, X.; Wang, Y. Discovery of a novel functional leptin protein (LEP) in zebra finches: Evidence for the existence of an authentic avian leptin gene predominantly expressed in the brain and pituitary. Endocrinology 2014, 155, 3385–3396. [Google Scholar] [CrossRef]
- Farkasova, H.; Hron, T.; Pačes, J.; Pajer, P.; Elleder, D. Identification of a GC-rich leptin gene in chicken. Agri Gene 2016, 1, 88–92. [Google Scholar] [CrossRef]
- Bornelov, S.; Seroussi, E.; Yosefi, S.; Benjamini, S.; Miyara, S.; Ruzal, M.; Grabherr, M.; Rafati, N.; Molin, A.M.; Pendavis, K.; et al. Comparative omics and feeding manipulations in chicken indicate a shift of the endocrine role of visceral fat towards reproduction. BMC Genom. 2018, 19, 295. [Google Scholar] [CrossRef]
- Nedwin, G.E.; Naylor, S.L.; Sakaguchi, A.Y.; Smith, D.; Jarrett-Nedwin, J.; Pennica, D.; Goeddel, D.V.; Gray, P.W. Human lymphotoxin and tumor necrosis factor genes: Structure, homology and chromosomal localization. Nucleic Acids Res. 1985, 13, 6361–6373. [Google Scholar] [CrossRef]
- Inoko, H.; Trowsdale, J. Linkage of TNF genes to the HLA-B locus. Nucleic Acids Res. 1987, 15, 8957–8962. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kaufman, J.; Milne, S.; Gobel, T.W.; Walker, B.A.; Jacob, J.P.; Auffray, C.; Zoorob, R.; Beck, S. The chicken B locus is a minimal essential major histocompatibility complex. Nature 1999, 401, 923–925. [Google Scholar] [CrossRef] [PubMed]
- Solinhac, R.; Leroux, S.; Galkina, S.; Chazara, O.; Feve, K.; Vignoles, F.; Morisson, M.; Derjusheva, S.; Bed’hom, B.; Vignal, A.; et al. Integrative mapping analysis of chicken microchromosome 16 organization. BMC Genom. 2010, 11, 616. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, J. Generalists and specialists: A new view of how MHC Class I molecules fight infectious pathogens. Trends Immunol. 2018, 39, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Warren, W.C.; Hillier, L.W.; Tomlinson, C.; Minx, P.; Kremitzki, M.; Graves, T.; Markovic, C.; Bouk, N.; Pruitt, K.D.; Thibaud-Nissen, F.; et al. A New Chicken Genome Assembly Provides Insight into Avian Genome Structure. G3 Genes Genomes Genet. 2017, 7, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Kuprash, D.V.; Alimzhanov, M.B.; Tumanov, A.V.; Grivennikov, S.I.; Shakhov, A.N.; Drutskaya, L.N.; Marino, M.W.; Turetskaya, R.L.; Anderson, A.O.; Rajewsky, K.; et al. Redundancy in tumor necrosis factor (TNF) and lymphotoxin (LT) signaling in vivo: Mice with inactivation of the entire TNF/LT locus versus single-knockout mice. Mol. Cell. Biol. 2002, 22, 8626–8634. [Google Scholar] [CrossRef]
- Morisson, M.; Lemiere, A.; Bosc, S.; Galan, M.; Plisson-Petit, F.; Pinton, P.; Delcros, C.; Feve, K.; Pitel, F.; Fillon, V.; et al. ChickRH6: A chicken whole-genome radiation hybrid panel. Genet. Sel. Evol. 2002, 34, 521–533. [Google Scholar] [CrossRef]
- Deakin, J.E.; Papenfuss, A.T.; Belov, K.; Cross, J.G.; Coggill, P.; Palmer, S.; Sims, S.; Speed, T.P.; Beck, S.; Graves, J.A. Evolution and comparative analysis of the MHC Class III inflammatory region. BMC Genom. 2006, 7, 281. [Google Scholar] [CrossRef]
- Pasparakis, M.; Alexopoulou, L.; Episkopou, V.; Kollias, G. Immune and inflammatory responses in TNF alpha-deficient mice: A critical requirement for TNF alpha in the formation of primary B cell follicles, follicular dendritic cell networks and germinal centers, and in the maturation of the humoral immune response. J. Exp. Med. 1996, 184, 1397–1411. [Google Scholar] [CrossRef]
- Flier, J.S. Lowered leptin slims immune response. Nat. Med. 1998, 4, 1124–1125. [Google Scholar] [CrossRef]
- Naylor, C.; Petri, W.A., Jr. Leptin Regulation of Immune Responses. Trends Mol. Med. 2016, 22, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Francisco, V.; Pino, J.; Campos-Cabaleiro, V.; Ruiz-Fernandez, C.; Mera, A.; Gonzalez-Gay, M.A.; Gomez, R.; Gualillo, O. Obesity, Fat Mass and Immune System: Role for Leptin. Front. Physiol. 2018, 9, 640. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Chang, S.; Xie, Q.; Black-Pyrkosz, A.; Zhang, H. Comparative transcriptomics of genetically divergent lines of chickens in response to Marek’s disease virus challenge at cytolytic phase. PLoS ONE 2017, 12, e0178923. [Google Scholar] [CrossRef] [PubMed]
- Bacon, L.D.; Hunt, H.D.; Cheng, H.H. A review of the development of chicken lines to resolve genes determining resistance to diseases. Poult. Sci. 2000, 79, 1082–1093. [Google Scholar] [CrossRef] [PubMed]
- Bush, S.J.; Freem, L.; MacCallum, A.J.; O’Dell, J.; Wu, C.; Afrasiabi, C.; Psifidi, A.; Stevens, M.P.; Smith, J.; Summers, K.M.; et al. Combination of novel and public RNA-seq datasets to generate an mRNA expression atlas for the domestic chicken. BMC Genom. 2018, 19, 594. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Hsueh, W.; Gonzalez-Crussi, F. Cellular localization of tumor necrosis factor (TNF)-alpha transcripts in normal bowel and in necrotizing enterocolitis. TNF gene expression by Paneth cells, intestinal eosinophils, and macrophages. Am. J. Pathol. 1993, 142, 1858–1865. [Google Scholar] [PubMed]
- Santaolalla, R.; Abreu, M.T. Innate immunity in the small intestine. Curr. Opin. Gastroenterol. 2012, 28, 124–129. [Google Scholar] [CrossRef]
- Bado, A.; Levasseur, S.; Attoub, S.; Kermorgant, S.; Laigneau, J.P.; Bortoluzzi, M.N.; Moizo, L.; Lehy, T.; Guerre-Millo, M.; Le Marchand-Brustel, Y.; et al. The stomach is a source of leptin. Nature 1998, 394, 790–793. [Google Scholar] [CrossRef]
- Peters, J.H.; Ritter, R.C.; Simasko, S.M. Leptin and CCK selectively activate vagal afferent neurons innervating the stomach and duodenum. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 290, R1544–R1549. [Google Scholar] [CrossRef]
- Yarandi, S.S.; Hebbar, G.; Sauer, C.G.; Cole, C.R.; Ziegler, T.R. Diverse roles of leptin in the gastrointestinal tract: Modulation of motility, absorption, growth, and inflammation. Nutrition 2011, 27, 269–275. [Google Scholar] [CrossRef]
- Sambrook, J.G.; Figueroa, F.; Beck, S. A genome-wide survey of Major Histocompatibility Complex (MHC) genes and their paralogues in zebrafish. BMC Genom. 2005, 6, 152. [Google Scholar] [CrossRef] [PubMed]
- O’Hare, T.H.; Delany, M.E. Genetic variation exists for telomeric array organization within and among the genomes of normal, immortalized, and transformed chicken systems. Chromosome Res. 2009, 17, 947–964. [Google Scholar] [CrossRef] [PubMed]
- Seroussi, E.; Pitel, F.; Leroux, S.; Morisson, M.; Bornelov, S.; Miyara, S.; Yosefi, S.; Cogburn, L.A.; Burt, D.W.; Andersson, L.; et al. Mapping of leptin and its syntenic genes to chicken chromosome 1p. BMC Genet. 2017, 18, 77. [Google Scholar]
- Wajant, H.; Siegmund, D. TNFR1 and TNFR2 in the control of the life and death balance of macrophages. Front. Cell Dev. Biol. 2019, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Wajant, H.; Pfizenmaier, K.; Scheurich, P. Tumor necrosis factor signaling. Cell Death Differ. 2003, 10, 45–65. [Google Scholar] [CrossRef] [PubMed]
- Cammisotto, P.; Bendayan, M. A review on gastric leptin: The exocrine secretion of a gastric hormone. Anat. Cell Biol. 2012, 45, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Inagaki-Ohara, K. Gastric leptin and tumorigenesis: Beyond obesity. Int. J. Mol. Sci. 2019, 20, 2622. [Google Scholar] [CrossRef]
- Darcel, N.P.; Liou, A.P.; Tome, D.; Raybould, H.E. Activation of vagal afferents in the rat duodenum by protein digests requires PepT1. J. Nutr. 2005, 135, 1491–1495. [Google Scholar] [CrossRef][Green Version]
- Pitel, F.; Abasht, B.; Morisson, M.; Crooijmans, R.P.; Vignoles, F.; Leroux, S.; Feve, K.; Bardes, S.; Milan, D.; Lagarrigue, S.; et al. A high-resolution radiation hybrid map of chicken chromosome 5 and comparison with human chromosomes. BMC Genom. 2004, 5, 66. [Google Scholar] [CrossRef]
- de Givry, S.; Bouchez, M.; Chabrier, P.; Milan, D.; Schiex, T. CARHTA GENE: Multipopulation integrated genetic and radiation hybrid mapping. Bioinformatics 2005, 21, 1703–1704. [Google Scholar] [CrossRef]
- Voorrips, R.E. MapChart: Software for the graphical presentation of linkage maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Plachy, J. The chicken—A laboratory animal of the class Aves. Folia Biol.-Prague 2000, 46, 17–23. [Google Scholar]
- Federspiel, M.J.; Hughes, S.H. Retroviral gene delivery. Method Cell Biol. 1997, 52, 179. [Google Scholar]
- Courtet, M.; Flajnik, M.; Du Pasquier, L. Major histocompatibility complex and immunoglobulin loci visualized by in situ hybridization on Xenopus chromosomes. Dev. Comp. Immunol. 2001, 25, 149–157. [Google Scholar] [CrossRef]
- Himly, M.; Foster, D.N.; Bottoli, I.; Iacovoni, J.S.; Vogt, P.K. The DF-1 chicken fibroblast cell line: Transformation induced by diverse oncogenes and cell death resulting from infection by avian leukosis viruses. Virology 1998, 248, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Krylov, V.; Tlapakova, T.; Macha, J. Localization of the single copy gene Mdh2 on Xenopus tropicalis chromosomes by FISH-TSA. Cytogenet. Genome Res. 2007, 116, 110–112. [Google Scholar] [CrossRef]
- Knytl, M.; Tlapakova, T.; Vankova, T.; Krylov, V. Silurana Chromosomal Evolution: A new piece to the puzzle. Cytogenet. Genome Res. 2018, 156, 223–228. [Google Scholar] [CrossRef]
- Knytl, M.; Smolik, O.; Kubickova, S.; Tlapakova, T.; Evans, B.J.; Krylov, V. Chromosome divergence during evolution of the tetraploid clawed frogs, Xenopus mellotropicalis and Xenopus epitropicalis as revealed by Zoo-FISH. PLoS ONE 2017, 12, e0177087. [Google Scholar] [CrossRef]
- Carabajal Paladino, L.Z.; Nguyen, P.; Sichova, J.; Marec, F. Mapping of single-copy genes by TSA-FISH in the codling moth, Cydia pomonella. BMC Genet. 2014, 15 (Suppl. 2), S15. [Google Scholar] [CrossRef]
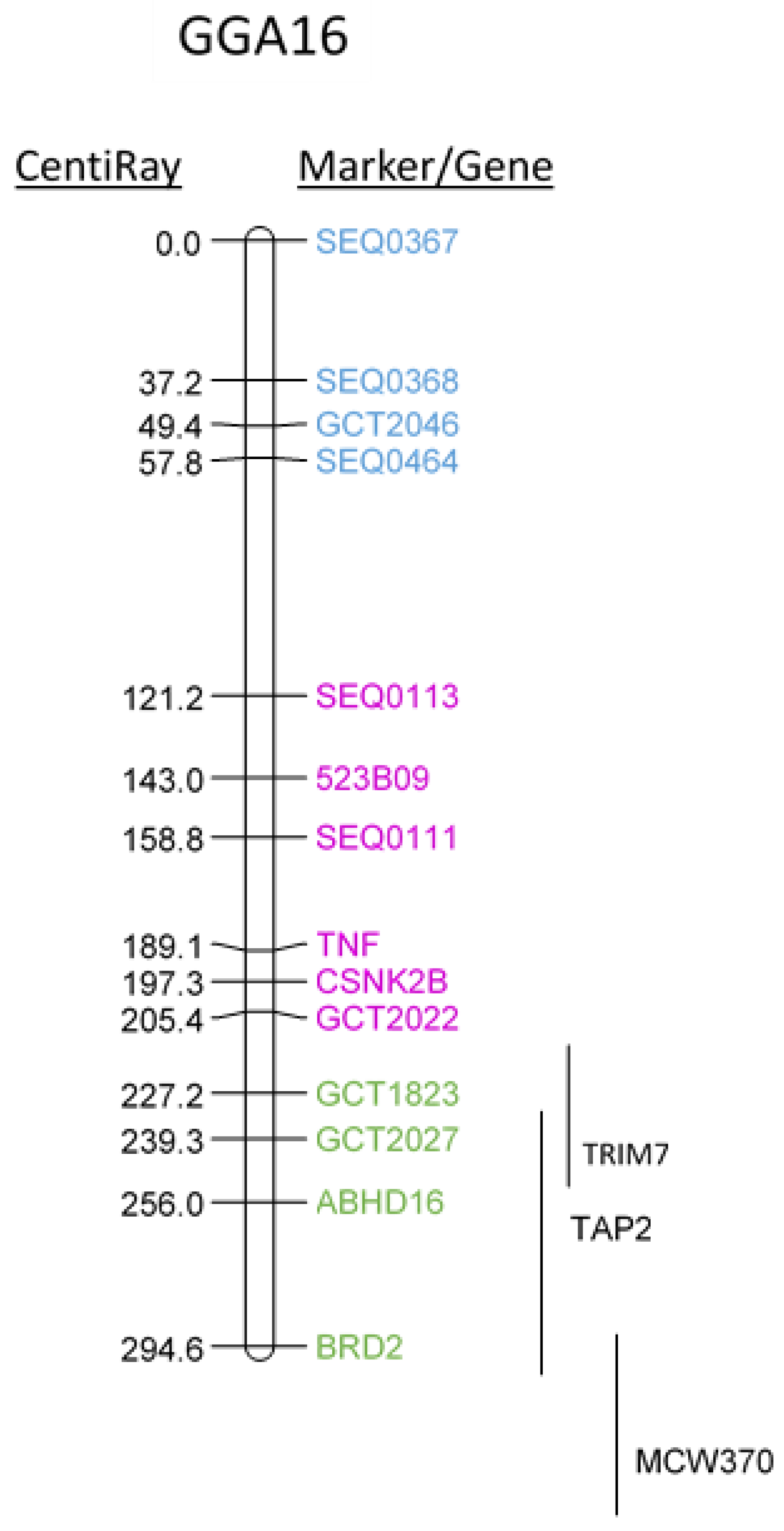
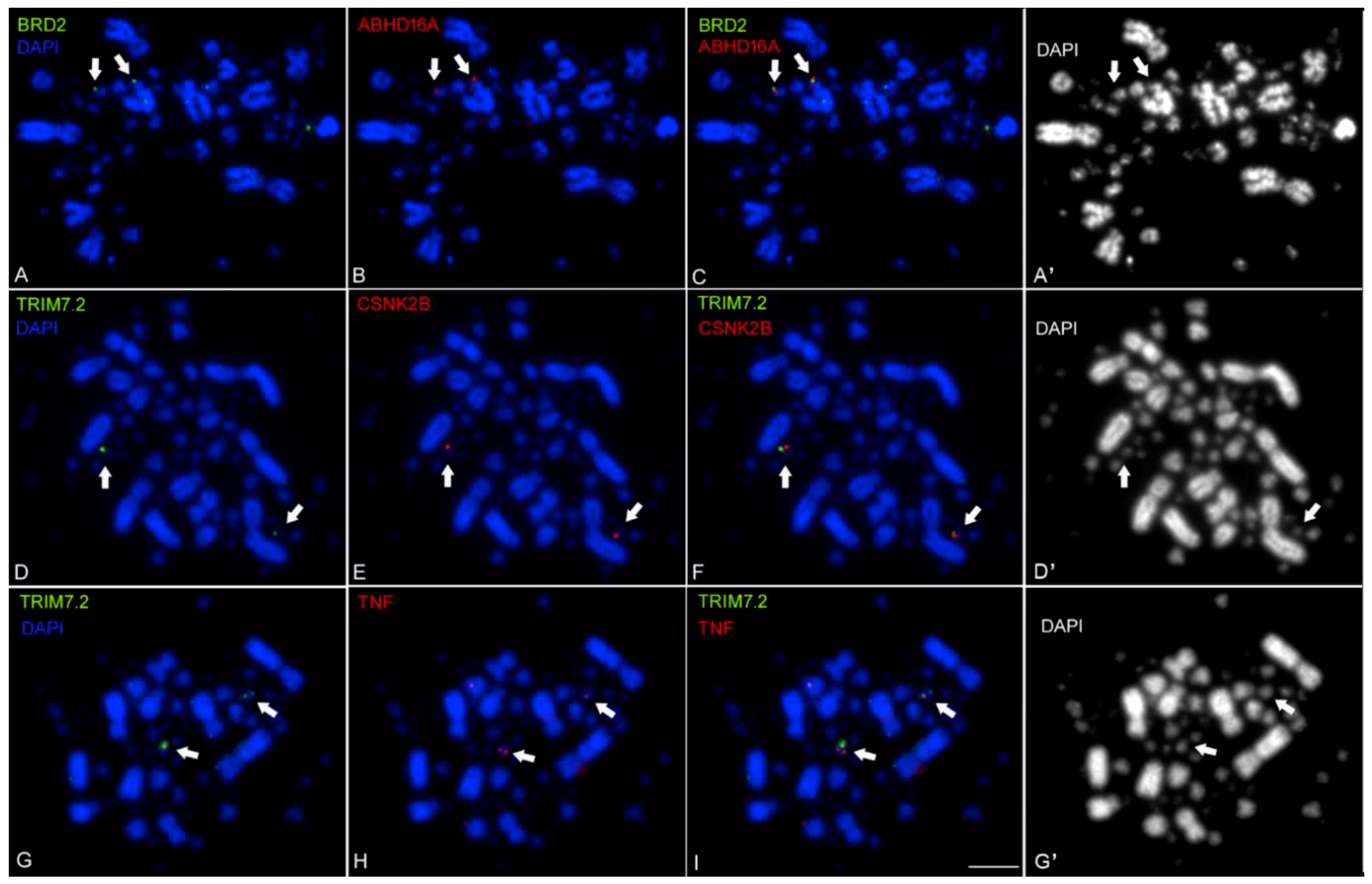
| Gene Name | Position RH-Mapping (CentiRay) | Linkage by FISH | Position GGA16 | Position HSA6 | |
|---|---|---|---|---|---|
| TRIM 7.2 | Tripartite motif containing 7 | 200–240 | CSNK2B, TNF | 2,482,253..2,492,415 | |
| BRD2 | Bromodomain containing 2 | 294.6 | ABHD16A | 2,574,363..2,583,216 | 32968594..32981505 |
| TAP2 | Transporter 2, ATP-binding cassette | 220–300 | 2,601,162..2,604,724 | 32821833..32838823 | |
| C4A | Complement 4 | 2,610,038..2,624,392 | 31982057..32002680 | ||
| TNF | Tumor Necrosis Factor | 189.1 | Unknown | 31575565..31578336 | |
| CSNK2B | Casein kinase II subunit beta | 197.3 | Unknown | 31665880..31670070 | |
| ABHD16A | Abhydrolase domain containing 16A | 256 | Unknown | 31686949..31703360 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seroussi, E.; Knytl, M.; Pitel, F.; Elleder, D.; Krylov, V.; Leroux, S.; Morisson, M.; Yosefi, S.; Miyara, S.; Ganesan, S.; et al. Avian Expression Patterns and Genomic Mapping Implicate Leptin in Digestion and TNF in Immunity, Suggesting That Their Interacting Adipokine Role Has Been Acquired Only in Mammals. Int. J. Mol. Sci. 2019, 20, 4489. https://doi.org/10.3390/ijms20184489
Seroussi E, Knytl M, Pitel F, Elleder D, Krylov V, Leroux S, Morisson M, Yosefi S, Miyara S, Ganesan S, et al. Avian Expression Patterns and Genomic Mapping Implicate Leptin in Digestion and TNF in Immunity, Suggesting That Their Interacting Adipokine Role Has Been Acquired Only in Mammals. International Journal of Molecular Sciences. 2019; 20(18):4489. https://doi.org/10.3390/ijms20184489
Chicago/Turabian StyleSeroussi, Eyal, Martin Knytl, Frédérique Pitel, Daniel Elleder, Vladimir Krylov, Sophie Leroux, Mireille Morisson, Sara Yosefi, Shoval Miyara, Saibaba Ganesan, and et al. 2019. "Avian Expression Patterns and Genomic Mapping Implicate Leptin in Digestion and TNF in Immunity, Suggesting That Their Interacting Adipokine Role Has Been Acquired Only in Mammals" International Journal of Molecular Sciences 20, no. 18: 4489. https://doi.org/10.3390/ijms20184489
APA StyleSeroussi, E., Knytl, M., Pitel, F., Elleder, D., Krylov, V., Leroux, S., Morisson, M., Yosefi, S., Miyara, S., Ganesan, S., Ruzal, M., Andersson, L., & Friedman-Einat, M. (2019). Avian Expression Patterns and Genomic Mapping Implicate Leptin in Digestion and TNF in Immunity, Suggesting That Their Interacting Adipokine Role Has Been Acquired Only in Mammals. International Journal of Molecular Sciences, 20(18), 4489. https://doi.org/10.3390/ijms20184489







