The Mechanisms Involved in Morphine Addiction: An Overview
Abstract
1. Introduction
2. Morphine and Its Receptors
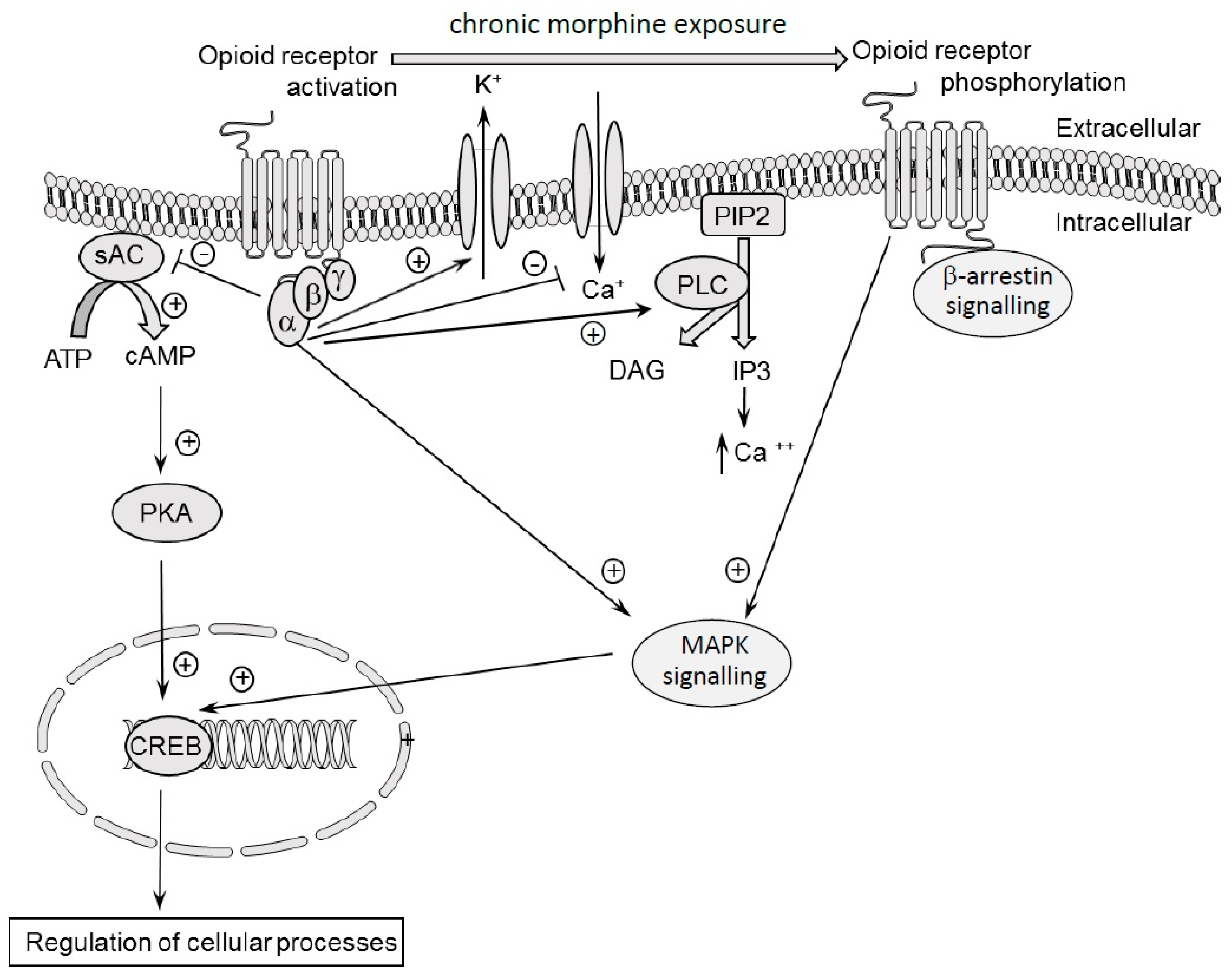
3. Molecular Effects of Acute and Chronic Dose of Morphine
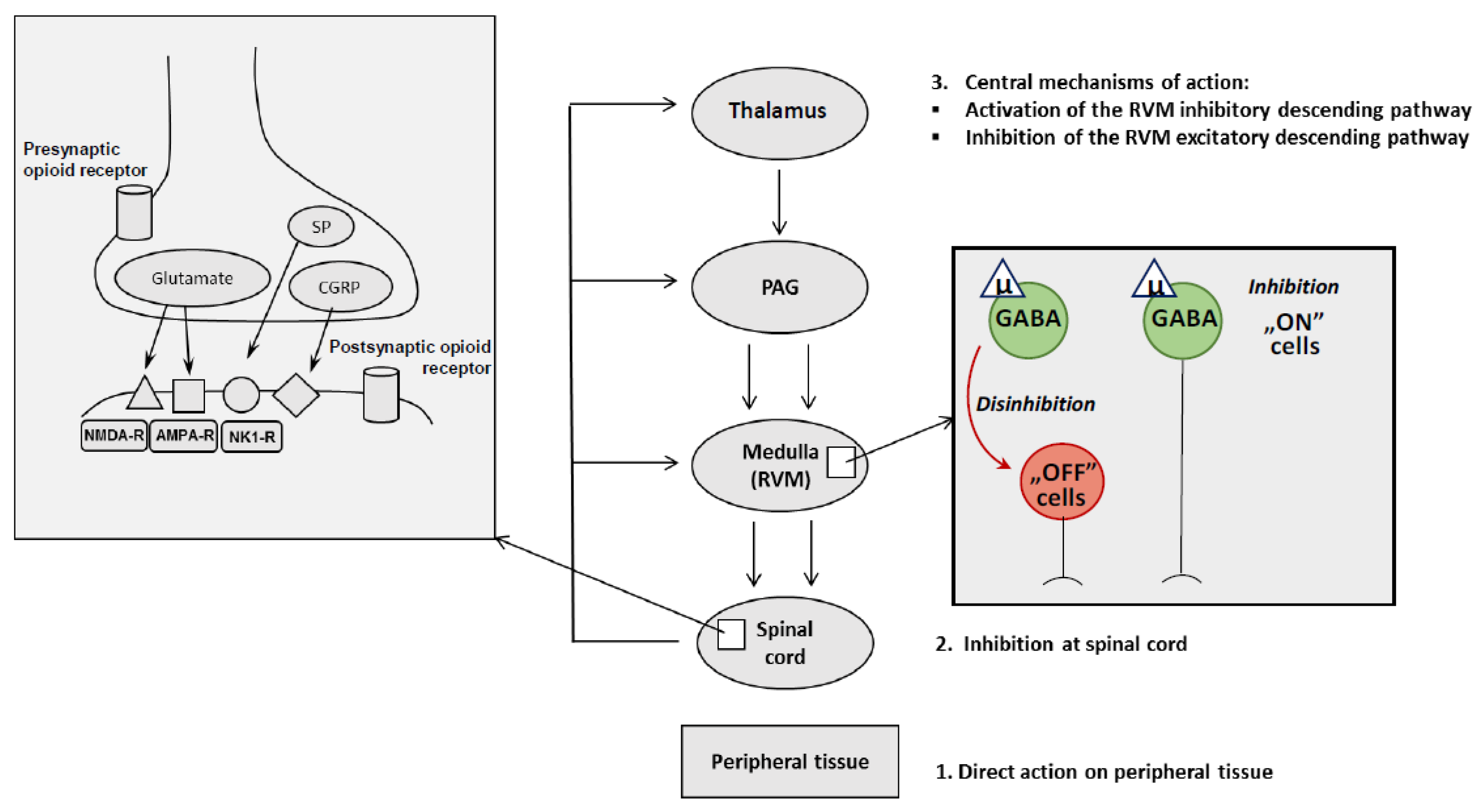
4. Opioid Analgesia
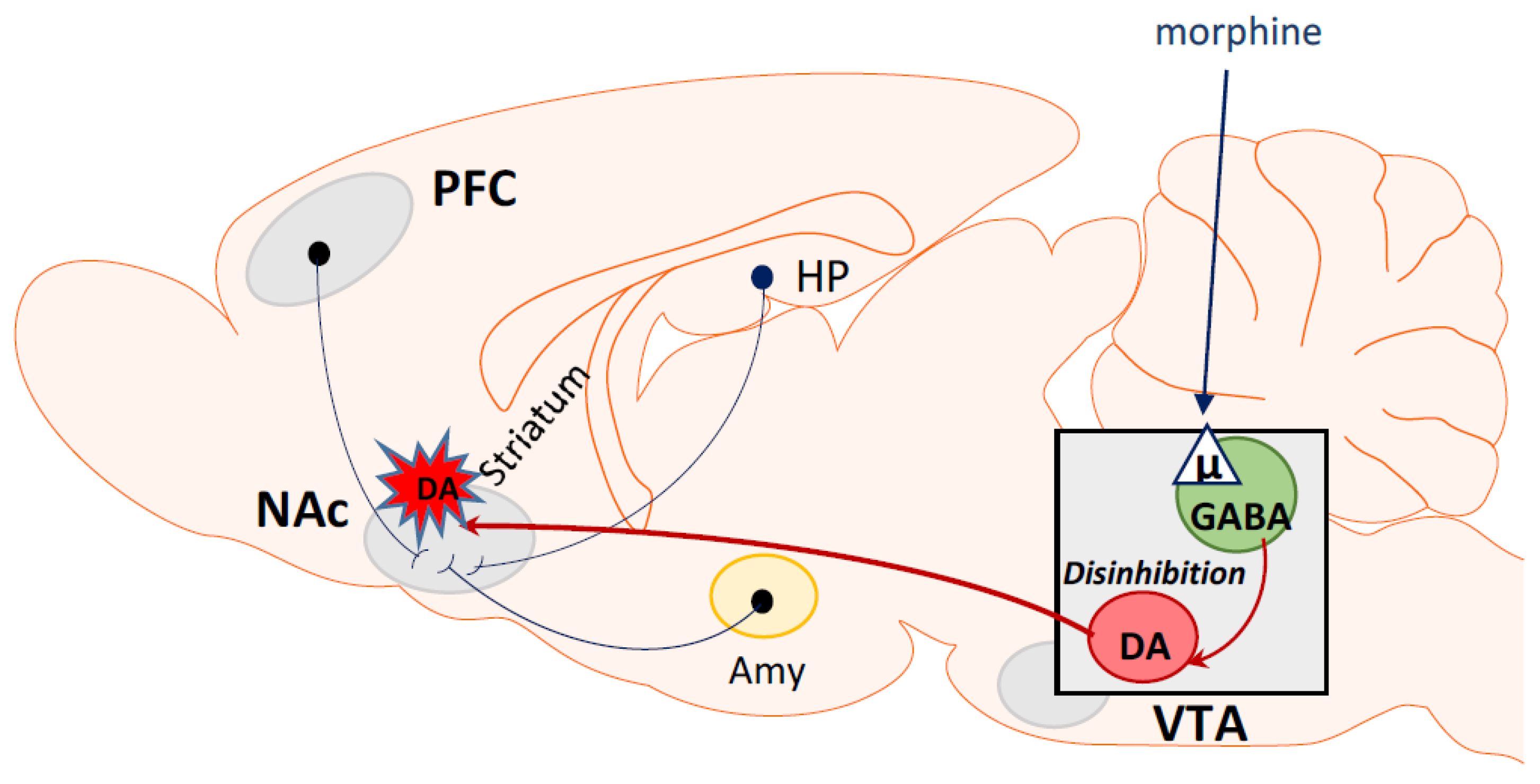
5. Opioid Rewarding Effects
6. Morphine as a Dependent Drug
7. Molecular Mechanisms of Morphine Tolerance and Dependence
8. Epigenetic Mechanisms of Morphine Tolerance and Dependence
9. Biased Opioid Ligands as a New Class of Opioid Analgesics
Author Contributions
Funding
Conflicts of Interest
References
- Nutt, D.J.; King, L.A.; Phillips, L.D. Drug harms in the UK: A multicriteria decision analysis. Lancet 2010, 376, 1558–1565. [Google Scholar] [CrossRef]
- Korsgaard, H.O.; Torgersen, S.; Wentzel-Larsen, T.; Ulberg, R. Substance abuse and personality disorder comorbidity in adolescent outpatients: Are girls more severely ill than boys? Child Adolesc. Psychiatry Ment. Health 2016, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Fridell, M.; Bäckström, M.; Hesse, M.; Krantz, P.; Perrin, S.; Nyhlén, A. Prediction of psychiatric comorbidity on premature death in a cohort of patients with substance use disorders: A 42-year follow-up. BMC Psychiatry 2019, 19, 150. [Google Scholar] [CrossRef] [PubMed]
- Spanagel, R.; Almeida, O.F.X.; Shippenberg, T.S. Long lasting changes in morphine-induced mesolimbic dopamine release after chronic morphine exposure. Synapse 1993, 14, 243–245. [Google Scholar] [CrossRef] [PubMed]
- Spanagel, R.; Shippenberg, T.S. Modulation of morphine induced sensitization by endogenous kappa opioid systems in the rat. Neurosci. Lett. 1993, 153, 232–236. [Google Scholar] [CrossRef]
- Tjon, G.H.K.; De Vries, T.J.; Ronken, E.; Hogenboom, F.; Wardeh, G.; Mulder, A.H.; Schoffelmeer, A.N. Repeated and chronic morphine administration causes differential long-lasting changes in dopaminergic neurotransmission in rat striatum without changing its δ- and κ-opioid receptor regulation. Eur. J. Pharmacol. 1994, 252, 205–212. [Google Scholar] [CrossRef]
- Tjon, G.H.K.; Voorn, P.; Vanderschuren, L.J.M.J.; De Vries, T.J.; Michiels, N.H.L.M.; Jonker, A.J.; Klop, H.; Nestby, P.; Mulder, A.H.; Schoffelmeer, A.N. Delayed occurrence of enhanced striatal preprodynorphin gene expression in behaviorally sensitized rats: Differential long-term effects of intermittent and chronic morphine administration. Neuroscience 1997, 76, 167–176. [Google Scholar] [CrossRef]
- Koob, G.F.; Ahmed, S.H.; Boutrel, B.; Chen, S.A.; Kenny, P.J.; Markou, A.; O’Dell, L.E.; Parsons, L.H.; Sanna, P.P. Neurobiological mechanisms in the transition from drug use to drug dependence. Neurosci. Biobehav. Rev. 2004, 27, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J. Molecular mechanisms of drug addiction. Neuropharmacology 2004, 47, 24–32. [Google Scholar] [CrossRef]
- Robinson, T.E.; Kolb, B. Structural plasticity associated with exposure to drugs of abuse. Neuropharmacology 2004, 47, 33–46. [Google Scholar] [CrossRef]
- Motahari, A.A.; Sahraei, H.; Meftahi, G.H. Role of nitric oxide on dopamine release and morphine-dependency. Basic Clin. Neurosci. 2016, 7, 283–290. [Google Scholar] [PubMed]
- Goodman, A. Neurobiology of addiction. An integrative review. Biochem. Pharmacol. 2008, 75, 266–322. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, R.J. Endogenous opiates and behavior: 2014. Peptides 2016, 75, 18–70. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.E.; Berridge, K.C. The psychology and neurobiology of addiction: An incentive-sensitization view. Addiction 2000, 95, 91–117. [Google Scholar]
- WHO. Guidelines on Community Management of Opioid Overdose. 2014. Available online: https://www.who.int/substance_abuse/publications/drugs/en/ 15.06.2019 (accessed on 15 June 2019).
- WHO. Information Sheet on Opioid Overdose. Available online: https://www.who.int/substance_abuse/information-sheet/en/ 22.08.2019 (accessed on 22 August 2019).
- Orfei, P.; Bigetti, E.; Patrizio, A.; Pinto, G. The use of alfentanil for short duration surgery in pediatric anesthesia. Minerva Anestesiol. 2000, 66, 123–129. [Google Scholar] [PubMed]
- White, L.D.; Hodge, A.; Vlok, R.; Hurtado, G.; Eastern, K.; Melhuish, T.M. Efficacy and adverse effects of buprenorphine in acute pain management: Systematic review and meta-analysis of randomised controlled trials. Br. J. Anaesth. 2018, 120, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Saadabadi, A. Buprenorphine; StatPearls Publishing: Treasure Island, FL, USA, 2018. [Google Scholar]
- Harricharan, S.; Farah, K. Buprenorphine Formulations for the Treatment of Opioid Use Disorders: A Review of Comparative Clinical Effectiveness. Cost-Effectiveness and Guidelines; CADTH Rapid Response Reports; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2017. [Google Scholar]
- Szabova, A.; Sadhasivam, S.; Wang, Y.; Nick, T.G.; Goldschneider, K. Comparison of postoperative analgesia with epidural butorphanol/bupivacaine versus fentanyl/bupivacaine following pediatric urological procedures. J. Opioid Manag. 2010, 6, 401–407. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Yokoyama, T.; Nagao, Y.; Nakagawa, T.; Magaribuchi, T. Treatment of epidural morphine induced pruritus with butorphanol. Masui 2009, 58, 178–182. [Google Scholar]
- Chung, K.F.; Pavord, I.D. Prevalence, pathogenesis, and causes of chronic cough. Lancet 2008, 371, 1364–1374. [Google Scholar] [CrossRef]
- Peechakara, B.V.; Gupta, M. Codeine; StatPearls Publishing: Treasure Island, FL, USA, 2018. [Google Scholar]
- Palmer, K.R.; Corbett, C.L.; Holdsworth, C.D. Double-blind cross-over study comparing loperamide; codeine and diphenoxylate in the treatment of chronic diarrhea. Gastroenterology 1980, 79, 1272–1275. [Google Scholar] [CrossRef]
- Koskela, H.; Naaranlahti, T. Drug therapy for cough. Duodecim 2016, 132, 455–460. [Google Scholar] [PubMed]
- Leung, F.W.; Rao, S.S. Approach to fecal incontinence and constipation in older hospitalized patients. Hosp. Pract. (1995) 2011, 39, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Schiller, L.R. Treatment of Fecal Incontinence. Curr. Treat. Options Gastroenterol. 2003, 6, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Leppert, W. Dihydrocodeine as an opioid analgesic for the treatment of moderate to severe chronic pain. Curr. Drug Metab. 2010, 11, 494–506. [Google Scholar] [CrossRef] [PubMed]
- Schug, S.A.; Ting, S. Fentanyl Formulations in the Management of Pain: An Update. Drugs 2017, 77, 747–763. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, S. Hydrocodone Bitartrate ER (Hysingla® ER): A Review in Chronic Pain. Clin. Drug Investig. 2016, 36, 969–980. [Google Scholar] [CrossRef]
- Davis, M.P.; McPherson, M.L.; Mehta, Z.; Behm, B.; Fernandez, C. What Parenteral Opioids to Use in Face of Shortages of Morphine, Hydromorphone, and Fentanyl. Am. J. Hosp. Palliat. Med. 2018, 35, 1118–1122. [Google Scholar] [CrossRef]
- Schildmann, E.K.; Rémi, C.; Bausewein, C. Levodropropizine in the management of cough associated with cancer or nonmalignant chronic disease—A systematic review. J. Pain Palliat. Care Pharmacother. 2011, 25, 209–218. [Google Scholar] [CrossRef]
- Gudin, J.; Fudin, J.; Nalamachu, S. Levorphanol use: Past, present and future. Postgrad. Med. 2015, 128, 46–53. [Google Scholar] [CrossRef]
- Lasoff, D.R.; Koh, C.H.; Corbett, B.; Minns, A.B.; Cantrell, F.L. Loperamide Trends in Abuse and Misuse Over 13 Years: 2002–2015. Pharmacotherapy 2017, 37, 249–253. [Google Scholar] [CrossRef]
- Singer, J.; Jank, A.; Amara, S.; Stepan, P.D.; Kaisers, U.; Hoehne, C. Efficacy and Effects of Parenteral Pethidine or Meptazinol and Regional Analgesia for Pain Relief during Delivery. A Comparative Observational Study. Geburtshilfe Frauenheilkd. 2016, 76, 964–971. [Google Scholar] [CrossRef]
- Johnson, K.; Gerada, C.; Greenough, A. Treatment of neonatal abstinence syndrome. Arch. Dis. Child. Fetal Neonatal Ed. 2003, 88, F2–F5. [Google Scholar] [CrossRef] [PubMed]
- Anderson, I.B.; Kearney, T.E. Medicine Cabinet Use of methadone. West. J. Med. 2000, 172, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Siu, A.; Robinson, C.A. Neonatal Abstinence Syndrome: Essentials for the Practitioner. J. Pediatr. Pharmacol. Ther. 2014, 19, 147–155. [Google Scholar]
- Murphy, P.B.; Barrett, M.J. Morphine; StatPearls Publishing: Treasure Island, FL, USA, 2018. [Google Scholar]
- Bolser, D.C. Pharmacologic Management of Cough. Otolaryngol. Clin. N. Am. 2010, 43, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Logash, M.; Pokotylo, P.; Zboina, B.; Stępień, R.B. Nalbuphine: Some aspects of the research and applications. Med. Stud. 2017, 33, 146–154. [Google Scholar] [CrossRef]
- Zeng, Z.; Lu, J.; Shu, C.; Chen, Y.; Guo, T.; Wu, Q.; Yao, S.; Yina, P. A Comparision of Nalbuphine with Morphine for Analgesic Effects and Safety: Meta-Analysis of Randomized Controlled Trials. Sci. Rep. 2015, 5, 10927. [Google Scholar] [CrossRef] [PubMed]
- Kubica-Cielińska, A.; Zielińska, M. The use of nalbuphine in paediatric anaesthesia. Anaesthesiol. Intensive Ther. 2015, 47, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Laux, G. Update Psychopharmacotherapy. Med. Monatsschr. Pharm. 2017, 40, 4–14. [Google Scholar]
- Behar, E.; Bagnulo, R.; Coffin, P.O. Acceptability and Feasibility of Naloxone Prescribing in Primary Care Settings: A Systematic Review. Prev. Med. 2018, 114, 79–87. [Google Scholar] [CrossRef]
- Jordan, M.R.; Morrisonponce, D. Naloxone; StatPearls Publishing: Treasure Island, FL, USA, 2018. [Google Scholar]
- Liu, M.; Wittbrodt, E. Low-dose oral naloxone reverses opioid-induced constipation and analgesia. J. Pain Symptom. Manag. 2002, 23, 48–53. [Google Scholar] [CrossRef]
- Sokol, R.; LaVertu, A.E.; Morrill, D.; Albanese, C.; Schuman-Olivier, Z. Group-based treatment of opioid use disorder with buprenorphine: A systematic review. J. Subst. Abuse Treat. 2018, 84, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Gastfriend, D.R. A pharmaceutical industry perspective on the economics of treatments for alcohol and opioid use disorders. Ann. N. Y. Acad. Sci. 2014, 1327, 112–130. [Google Scholar] [CrossRef] [PubMed]
- Gkegkes, I.D.; Minis, E.E.; Iavazzo, C. Oxycodone/naloxone in postoperative pain management of surgical patients. J. Opioid Manag. 2018, 14, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Wang, L.; Gao, Y.; Zhou, H.; Wang, J. Analgesic Effect of Preoperative Pentazocine for Laparoscopic Cholecystectomy. Cureus 2016, 8, e948. [Google Scholar] [CrossRef] [PubMed]
- Yasaei, R.; Saadabadi, A. Meperidine; StatPearls Publishing: Treasure Island, FL, USA, 2018. [Google Scholar]
- Leong, W.L.; Sng, B.L.; Sia, A.T. A comparison between remifentanil and meperidine for labor analgesia: A systematic review. Anesth. Analg. 2011, 113, 818–825. [Google Scholar] [CrossRef] [PubMed]
- Bushuven, S.; Kreuer, S.; Kranke, P. Remifentanil Up2date—Part 1. Anasthesiol. Intensivmed. Notfallmed. Schmerzther. 2017, 52, 543–553. [Google Scholar]
- Mandel, J.E. Considerations for the use of short-acting opioids in general anesthesia. J. Clin. Anesth. 2014, 26, S1–S7. [Google Scholar] [CrossRef]
- Zajączkowska, R.; Przewłocka, B.; Kocot-Kępska, M.; Mika, J.; Leppert, W.; Wordliczek, J. Tapentadol—A representative of a new class of MOR-NRI analgesics. Pharmacol. Rep. 2018, 70, 812–820. [Google Scholar] [CrossRef]
- Wolfert, C.; Merbach, M.; Stammler, G.; Emrich, O.; Meid, A.D.; Burhenne, J.; Blank, A.; Mikus, G. Management of chronic pain using extended release tilidine: Quality of life and implication of comedication on tilidine metabolism. Schmerz 2017, 31, 516–523. [Google Scholar] [CrossRef]
- Bravo, L.; Mico, J.A.; Berrocoso, E. Discovery and development of tramadol for the treatment of pain. Expert Opin. Drug Discov. 2017, 12, 1281–1291. [Google Scholar] [CrossRef] [PubMed]
- Gray, K.; Adhikary, S.D.; Janicki, P. Pharmacogenomics of analgesics in anesthesia practice: A current update of literature. J. Anaesthesiol. Clin. Pharmacol. 2018, 34, 155–160. [Google Scholar] [PubMed]
- Khan, A.H.; Rasaily, D. Tramadol Use in Premature Ejaculation: Daily versus Sporadic Treatment. Indian J. Psychol. Med. 2013, 35, 256–259. [Google Scholar] [CrossRef] [PubMed]
- Waldhoer, M.; Bartlett, S.E.; Whistler, J.L. Opioid receptors. Annu. Rev. Biochem. 2004, 73, 953–990. [Google Scholar] [CrossRef]
- Dhawan, B.N.; Cesselin, F.; Raghubir, R.; Reisine, T.; Bradley, P.B.; Portoghese, P.S.; Hamon, M. International Union of Pharmacology. XII. Classification of opioid receptors. Pharmacol. Rev. 1996, 48, 567–592. [Google Scholar]
- Pert, C.B.; Snyder, S.H. Opiate receptor: Demonstration in nervous tissue. Science 1973, 179, 1011–1014. [Google Scholar] [CrossRef] [PubMed]
- Gray, A.C.; Coupar, I.M.; White, P.J. Comparison of opioid receptor distributions in the rat central nervous system. Life Sci. 2006, 79, 674–685. [Google Scholar] [CrossRef] [PubMed]
- Moskowitz, A.S.; Goodman, R.R. Autoradiographic distribution of mu1 and mu2 opioid binding in the mouse central nervous system. Brain Res. 1985, 360, 117–129. [Google Scholar] [CrossRef]
- Wittert, G.; Hope, P.; Pyle, D. Tissue distribution of opioid receptors gene expression in the rat. Biochem. Biophys. Res. Commun. 1996, 218, 877–881. [Google Scholar] [CrossRef]
- Gray, A.C.; Coupar, I.M.; White, P.J. Comparison of opioid receptor distributions in the rat ileum. Life Sci. 2006, 78, 1610–1616. [Google Scholar] [CrossRef]
- Hughes, J.; Kosterlitz, H.W.; Smith, T.W. The distribution of methionine-enkephalin and leucine-enkephalin in the brain and peripheral tissues. Br. J. Pharmacol. 1977, 61, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.D.; Galligan, J.J. Function of opioids in the enteric nervous system. Neurogastroenterol. Motil. 2004, 16, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Zebraski, S.E.; Kochenash, S.M.; Raffa, R.B. Lung opioid receptors: Pharmacology and possible target for nebulized morphine in dyspnea. Life Sci. 2000, 66, 2221–2231. [Google Scholar] [CrossRef]
- Pattinson, K.T.S. Opioids and the control of respiration. Br. J. Anaesth. 2008, 100, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Klee, W.A.; Nirenberg, M. Opiate-dependent modulation of adenylate cyclase. Proc. Natl. Acad. Sci. USA 1977, 74, 3365–3369. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, S.; Chang, A.; Liu, N.J.; Gintzler, A.R. Chronic opioid treatment augments caveolin-1 scaffolding: Relevance to stimulatory μ-opioid receptor adenylyl cyclase signaling. J. Neurochem. 2016, 139, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Fleming, L.M.; Ponjee, G.; Childers, S.R. Inhibition of protein phosphorylation by opioid-inhibited adenylyl cyclase in rat brain membranes. J. Pharmacol. Exp. Ther. 1992, 260, 1416–1424. [Google Scholar] [PubMed]
- Zhang, Z.; Pan, Z.Z. Synaptic mechanism for functional synergism between delta- and mu-opioid receptors. J. Neurosci. 2010, 30, 4735–4745. [Google Scholar] [CrossRef] [PubMed]
- Law, P.Y.; Wong, Y.H.; Loh, H.H. Molecular mechanisms and regulation of opioid receptor signaling. Annu. Rev. Pharmacol. Toxicol. 2000, 40, 389–430. [Google Scholar] [CrossRef]
- North, R.A.; Williams, J.T.; Surprenant, A.; Christie, M.J. Mu and delta receptors belong to a family of receptors that are coupled to potassium channels. Proc. Natl. Acad. Sci. USA 1987, 84, 5487–5491. [Google Scholar] [CrossRef]
- Moises, H.C.; Rusin, K.I.; Macdonald, R.L. Mu- and kappa-opioid receptors selectively reduce the same transient components of high-threshold calcium current in rat dorsal root ganglion sensory neurons. J. Neurosci. 1994, 14, 5903–5916. [Google Scholar] [CrossRef] [PubMed]
- Zamponi, G.W.; Snutch, T.P. Modulation of voltage-dependent calcium channels by G proteins. Curr. Opin. Neurobiol. 1998, 8, 351–356. [Google Scholar] [CrossRef]
- Zamponi, G.W.; Snutch, T.P. Modulating modulation: Crosstalk between regulatory pathways of presynaptic calcium channels. Mol. Interv. 2002, 2, 476–478. [Google Scholar] [CrossRef] [PubMed]
- Torrecilla, M.; Marker, C.L.; Cintora, S.C.; Stoffel, M.; Williams, J.T.; Wickman, K. G-protein-gated potassium channels containing Kir3.2 and Kir3.3 subunits mediate the acute inhibitory effects of opioids on locus ceruleus neurons. J. Neurosci. 2002, 22, 4328–4334. [Google Scholar] [CrossRef] [PubMed]
- Torrecilla, M.; Quillinan, N.; Williams, J.T.; Wickman, K. Pre- and postsynaptic regulation of locus coeruleus neurons after chronic morphine treatment: A study of GIRK-knockout mice. Eur. J. Neurosci. 2008, 28, 618–624. [Google Scholar] [CrossRef]
- Allouche, S.; Noble, F.; Marie, N. Opioid receptor desensitization: Mechanisms and its link to tolerance. Front. Pharmacol. 2014, 5, 280. [Google Scholar] [CrossRef] [PubMed]
- Fields, H. State-dependent opioid control of pain. Nat. Rev. Neurosci. 2004, 5, 565–575. [Google Scholar] [CrossRef]
- Raehal, K.M.; Bohn, L.M. β-arrestins: Regulatory role and therapeutic potential in opioid and cannabinoid receptor-mediated analgesia. Handb. Exp. Pharmacol. 2014, 219, 427–443. [Google Scholar]
- Gomes, I.; Gupta, A.; Filipovska, J.; Szeto, H.H.; Pintar, J.E.; Devi, L.A. A role for heterodimerization of μ and δ opiate receptors in enhancing morphine analgesia. Proc. Natl. Acad. Sci. USA 2004, 101, 5135–5139. [Google Scholar] [CrossRef]
- Di Chiara, G. Role of dopamine in the behavioural actions of nicotine related to addiction. Eur. J. Pharmacol. 2000, 393, 295–314. [Google Scholar] [CrossRef]
- Anderson, S.M.; Pierce, R.C. Cocaine-induced alterations in dopamine receptor signaling: Implications for reinforcement and reinstatement. Pharmacol. Ther. 2005, 106, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Briand, L.A.; Flagel, S.B.; Garcia-Fuster, M.J.; Watson, S.J.; Akil, H.; Sarter, M.; Robinson, T.E. Persistent alterations in cognitive function and prefrontal dopamine D2 receptors following extended, but not limited, access to self-administered cocaine. Neuropsychopharmacology 2008, 33, 2969–2980. [Google Scholar] [CrossRef] [PubMed]
- Listos, J.; Baranowska-Bosiacka, I.; Wąsik, A.; Talarek, S.; Tarnowski, M.; Listos, P.; Łupina, M.; Antkiewicz-Michaluk, L.; Gutowska, I.; Tkacz, M.; et al. The adenosinergic system is involved in sensitization to morphine withdrawal signs in rats-neurochemical and molecular basis in dopaminergic system. Psychopharmacology 2016, 233, 2383–2397. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A.; Rompre, P.P. Brain dopamine and reward. Annu. Rev. Psychol. 1989, 40, 191–225. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.W.; North, R.A. Opioids excite dopamine neurons by hyperpolarization of local interneurons. J. Neurosci. 1992, 12, 4. [Google Scholar] [CrossRef]
- Sepehrizadeh, Z.; Bahrololoumi Shapourabadi, M.; Ahmadi, S.; Hashemi Bozchlou, S.; Zarrindast, M.R.; Sahebgharani, M. Decreased AMPA GluR2, but not GluR3, mRNA expression in rat amygdala and dorsal hippocampus following morphine-induced behavioural sensitization. Clin. Exp. Pharmacol. Physiol. 2008, 35, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Farahmandfar, M.; Karimian, S.M.; Zarrindast, M.R.; Kadivar, M.; Afrouzi, H.; Naghdi, N. Morphine sensitization increases the extracellular level of glutamate in CA1 of rat hippocampus via μ-opioid receptor. Neurosci. Lett. 2011, 494, 130–134. [Google Scholar] [CrossRef]
- Reith, M.E.; Li, M.Y.; Yan, Q.S. Extracellular dopamine, norepinephrine, and serotonin in the ventral tegmental area and nucleus accumbens of freely moving rats during intracerebral dialysis following systemic administration. Psychopharmacology 1997, 134, 309–317. [Google Scholar] [CrossRef]
- Carr, D.B.; Sesack, S.R. GABA-containing neurons in the rat ventral tegmental area project to the prefrontal cortex. Synapse 2000, 38, 114–123. [Google Scholar] [CrossRef]
- Frankowska, M.; Wydra, K.; Faron-Górecka, A.; Zaniewska, M.; Kuśmider, M.; Dziedzicka-Wasylewska, M.; Filip, M. Alterations in gamma-aminobutyric acid(B) receptor binding in the rat brain after reinstatement of cocaine-seeking behavior. Pharmacol. Rep. 2008, 60, 834–843. [Google Scholar]
- Zaniewska, M.; Filip, M.; Przegalinski, E. The Involvement of Norepinephrine in Behaviors Related to Psychostimulant Addiction. Curr. Neuropharmacol. 2015, 13, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Sahraei, H.; Zarei, F.; Eidi, A.; Oryan, S.; Shams, J.; Khoshbaten, A.; Zarrindast, M.R. The role of nitric oxide within the nucleus accumbens on the acquisition and expression of morphine-induced place preference in morphine sensitized rats. Eur. J. Pharmacol. 2007, 556, 99–106. [Google Scholar] [CrossRef]
- Alijanpour, S.; Tirgar, F.; Zarrindast, M.R. Role of dorsal hippocampal orexin-1 receptors in memory restoration induced by morphine sensitization phenomenon. Neuroscience 2016, 312, 215–226. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.E.; Berridge, K.C. The incentive sensitization theory of addiction: Some current issues. Philos. Trans. R. Soc. B 2008, 363, 3137–3146. [Google Scholar] [CrossRef]
- Listos, J.; Baranowska-Bosiacka, I.; Talarek, S.; Listos, P.; Orzelska, J.; Fidecka, S.; Gutowska, I.; Kolasa, A.; Rybicka, M.; Chlubek, D. The effect of perinatal lead exposure on dopamine receptor D2 expression in morphine dependent rats. Toxicology 2013, 310, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.M.; Castonguay, A.; Ghogha, A.; Vayssiere, P.; Pradhan, A.A.; Xue, L.; Mehrabani, S.; Wu, J.; Levitt, P.; Olmstead, M.C.; et al. Neuroimmune regulation of GABAergic neurons within the ventral tegmental area during withdrawal from chronic morphine. Neuropsychopharmacology 2016, 41, 949–959. [Google Scholar] [CrossRef]
- Harris, A.C.; Gewirtz, J.C. Acute opioid dependence: Characterizing the early adaptations underlying drug withdrawal. Psychopharmacology 2005, 178, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.J.; Cahill, C.M. Neurobiology of opioid dependence in creating addiction vulnerability. F1000Research 2016, 5. [Google Scholar] [CrossRef]
- Schulteis, G.; Markou, A.; Gold, L.H.; Stinus, L.; Koob, G.F. Relative sensitivity to naloxone of multiple indices of opiate withdrawal: A quantitative dose-response analysis. J. Pharmacol. Exp. Ther. 1994, 271, 1391–1398. [Google Scholar] [PubMed]
- Zhang, Z.; Schulteis, G. Withdrawal from acute morphine dependence is accompanied by increased anxiety-like behavior in the elevated plus maze. Pharmacol. Biochem. Behav. 2008, 89, 392–403. [Google Scholar] [CrossRef]
- Done, C.; Silverstone, P.; Sharp, T. Effect of naloxone-precipitated morphine withdrawal on noradrenaline release in rat hippocampus in vivo. Eur. J. Pharmacol. 1992, 215, 333–336. [Google Scholar] [CrossRef]
- Diaz, S.L.; Kemmling, A.; Balerio, G.N. Baclofen reestablishes striatal and cortical dopamine concentrations during naloxone-precipitated withdrawal. Neurochem. Int. 2003, 42, 293–298. [Google Scholar] [CrossRef]
- Hooshmandi, M.; Hosseinmardi, N.; Janahmadi, M.; Khakpai, F.; Rohampour, K.; Doostmohammadi, J. Antagonism of orexin type-1 receptors (OX1Rs) attenuates naloxone-precipitated morphine withdrawal syndrome in rat dorsal hippocampus. Pharmacol. Biochem. Behav. 2017, 158, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Stinus, L.; Le Moal, M.; Bloom, F.E. Opponent process theory of motivation: Neurobiological evidence from studies of opiate dependence. Neurosci. Biobehav. Rev. 1989, 13, 135–140. [Google Scholar] [CrossRef]
- Acquas, E.; Di Chiara, G. Depression of mesolimbic dopamine transmission and sensitization to morphine during opiate abstinence. J. Neurochem. 1992, 58, 1620–1625. [Google Scholar] [CrossRef] [PubMed]
- Diana, M.; Pistis, M.; Muntoni, A.; Gessa, G. Profound decrease of mesolimbic dopaminergic neuronal activity in morphine withdrawn rats. J. Pharmacol. Exp. Ther. 1995, 272, 781–785. [Google Scholar] [PubMed]
- Elman, I.; Borsook, D.; Volkow, N.D. Pain and suicidality: Insights from reward and addiction neuroscience. Prog. Neurobiol. 2013, 109, 1–27. [Google Scholar] [CrossRef]
- Fox, M.E.; Rodeberg, N.T.; Wightman, R.M. Reciprocal catecholamine changes during opiate exposure and withdrawal. Neuropsychopharmacology 2017, 42, 671–681. [Google Scholar] [CrossRef]
- Sepulveda, M.J.; Hernandez, L.; Rada, P.; Tucci, S.; Contreras, E. Effect of precipitated withdrawal on extracellular glutamate and aspartate in the nucleus accumbens of chronically morphine-treated rats: An in vivo microdialysis study. Pharmacol. Biochem. Behav. 1998, 60, 255–262. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, X.; Zhang, Y.M.; Liu, H.; Jiang, Q.; Pang, G.; Tao, X.; Dong, L.; Stackman, R.W., Jr. Activation of serotonin 5-HT(2C) receptor suppresses behavioral sensitization and naloxone-precipitated withdrawal symptoms in morphine-dependent mice. Neuropharmacology 2016, 101, 246–254. [Google Scholar] [CrossRef]
- Zhou, Y.; Bendor, J.; Hofmann, L.; Randesi, M.; Ho, A.; Kreek, M.J. Mu opioid receptor and orexin/hypocretin mRNA levels in the lateral hypothalamus and striatum are enhanced by morphine withdrawal. J. Endocrinol. 2006, 191, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Matinfar, M.; Esfahani, M.M.; Aslany, N.; Davoodi, S.H.; Parsaei, P.; Zarei, G.; Reisi, P. Effect of repeated morphine withdrawal on spatial learning, memory and serum cortisol level in mice. Adv. Biomed. Res. 2013, 2, 80. [Google Scholar] [PubMed]
- Meye, F.J.; van Zessen, R.; Smidt, M.P.; Adan, R.A.; Ramakers, G.M. Morphine withdrawal enhances constitutive μ-opioid receptor activity in the ventral tegmental area. J. Neurosci. 2012, 32, 16120–16128. [Google Scholar] [CrossRef] [PubMed]
- Bilecki, W.; Zapart, G.; Ligeza, A.; Wawrzczak-Bargiela, A.; Urbański, M.J.; Przewłocki, R. Regulation of the extracellular signal-regulated kinases following acute and chronic opioid treatment. Cell. Mol. Life Sci. 2005, 62, 2369–2375. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.L.; He, J.H.; Ding, H.L.; Zeng, Y.M. Activation of the spinal ERK signaling pathway contributes naloxone-precipitated withdrawal in morphine-dependent rats. Pain 2005, 118, 336–349. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, K.; Kawasaki, S.; Kobori, T.; Fujita-Hamabe, W.; Mizoguchi, H.; Yamada, K.; Nabeshima, T.; Tokuyama, S. Involvement of matrix metalloproteinase-9 in the development of morphine tolerance. Eur. J. Pharmacol. 2012, 683, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Gustin, H.B.; Akil, H. Opioid analgesics. In Goodman and Gilman’s The Pharmacological Basis of Therapeutics; Hardman, J.G., Limbird, L.E., Eds.; McGraw-Hill: New York, NY, USA, 2001; pp. 569–619. [Google Scholar]
- Mitra, S.; Sinatra, R.S. Perioperative management of acute pain in the opioid-dependent patient. Anesthesiology 2004, 101, 212–227. [Google Scholar] [CrossRef]
- Cecchi, M.; Capriles, N.; Watson, S.J.; Akil, H. Differential responses to morphine-induced analgesia in the tail-flick test. Behav. Brain Res. 2008, 194, 146–151. [Google Scholar] [CrossRef]
- Célérier, E.; Yazdi, M.T.; Castañé, A.; Ghozland, S.; Nyberg, F.; Maldonado, R. Effects of nandrolone on acute morphine responses, tolerance and dependence in mice. Eur. J. Pharmacol. 2003, 465, 69–81. [Google Scholar] [CrossRef]
- Joharchi, K.; Jorjani, M. The role of nitric oxide in diabetes-induced changes of morphine tolerance in rats. Eur. J. Pharmacol. 2007, 570, 66–71. [Google Scholar] [CrossRef]
- Zarrindast, M.R.; Dinkoub, Z.; Homayoun, H.; Bakhtiarian, A.; Khavandgar, S. Dopamine receptor mechanism(s) and morphine tolerance in mice. J. Psychopharmacol. 2002, 16, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, E.; Bagcivan, I.; Gursoy, S. Role of D1/D2 dopamine receptors antagonist perphenazine in morphine analgesia and tolerance in rats. Bosn. J. Basic Med. Sci. 2013, 13, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.P.; Jain, N.K.; Kulkarni, S.K. Fluoxetine suppresses morphine tolerance and dependence: Modulation of NO-cGMP/DA/serotoninergic pathways. Methods Find. Exp. Clin. Pharmacol. 2003, 25, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Gawel, K.; Gibula-Bruzda, E.; Dziedzic, M.; Jenda-Wojtanowska, M.; Marszalek-Grabska, M.; Silberring, J.; Kotlinska, J.H. Cholinergic activation affects the acute and chronic antinociceptive effects of morphine. Physiol. Behav. 2017, 169, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, H.; Ghaemi-Jandabi, M.; Azizi, H.; Semnanian, S. The role of orexin type-1 receptors in the development of morphine tolerance in locus coeruleus neurons: An electrophysiological perspective. Brain Res. 2016, 1646, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Wilson-Poe, A.R.; Lau, B.K.; Vaughan, C.W. Repeated morphine treatment alters cannabinoid modulation of GABAergic synaptic transmission within the rat periaqueductal grey. Br. J. Pharmacol. 2015, 172, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.S.; Hou, Y.; Yan, T.L.; Guo, Y.Y.; Han, W.; Guan, F.L.; Chen, T.; Li, T. Dopamine D3 receptor-regulated NR2B subunits of N-methyl-d-aspartate receptors in the nucleus accumbens involves in morphine-induced locomotor activity. CNS Neurosci. Ther. 2014, 20, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Guegan, T.; Cebrià, J.P.; Maldonado, R.; Martin, M. Morphine-induced locomotor sensitization produces structural plasticity in the mesocorticolimbic system dependent on CB1-R activity. Addict. Biol. 2016, 21, 1113–1126. [Google Scholar] [CrossRef] [PubMed]
- Shippenberg, T.S.; Heidbreder, C. Sensitization to the conditioned rewarding effects of cocaine: Pharmacological and temporal characteristics. J. Pharmacol. Exp. Ther. 1995, 273, 808–815. [Google Scholar]
- Manzanedo, C.; Aguilar, M.A.; Rodríguez-Arias, M.; Miñarro, J. Sensitization to the rewarding effects of morphine depends on dopamine. J. Neuroreport. 2005, 16, 201–205. [Google Scholar] [CrossRef]
- Rothwell, P.E.; Gewirtz, J.C.; Thomas, M.J. Episodic withdrawal promotes psychomotor sensitization to morphine. Neuropsychopharmacology 2010, 35, 2579–2589. [Google Scholar] [CrossRef] [PubMed]
- Vanderschuren, L.J.; Kalivas, P.W. Alterations in dopaminergic and glutamatergic transmission in the induction and expression of behavioral sensitization: A critical review of preclinical studies. Psychopharmacology 2000, 151, 99–120. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.E. LTP may trigger addiction. Mol. Interv. 2003, 3, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Vanderschuren, L.J.; Pierce, R.C. Sensitization processes in drug addiction. In Behavioral Neuroscience of Drug Addiction 2010; Springer: Berlin/Heidelberg, Germany, 2010; pp. 179–195. [Google Scholar]
- Le Moal, M.; Simon, H. Mesocorticolimbic dopaminergic network: Functional and regulatory roles. Physiol. Rev. 1991, 71, 155–234. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.E.; Berridge, K.C. The neural basis of drug craving: An incentive-sensitization theory of addiction. Brain Res. Rev. 1993, 18, 247–291. [Google Scholar] [CrossRef]
- Kalivas, P.W.; Duffy, P. Sensitization to repeated morphine injection in the rat: Possible involvement of A10 dopamine neurons. J. Pharmacol. Exp. Ther. 1987, 241, 204–212. [Google Scholar]
- Reisi, Z.; Bani-Ardalan, M.; Zarepour, L.; Haghparast, A. Involvement of D1/D2 dopamine receptors within the nucleus accumbens and ventral tegmental area in the development of sensitization to antinociceptive effect of morphine. Pharmacol. Biochem. Behav. 2014, 118, 16–21. [Google Scholar] [CrossRef]
- Jeziorski, M.; White, F.J. Dopamine receptor antagonists prevent expression, but not development, of morphine sensitization. Eur. J. Pharmacol. 1995, 275, 235–244. [Google Scholar] [CrossRef]
- Borgkvist, A.; Valjent, E.; Santini, E.; Hervé, D.; Girault, J.A.; Fisone, G. Delayed, context- and dopamine D1receptor-dependent activation of ERK in morphine-sensitized mice. Neuropharmacology 2008, 55, 230–237. [Google Scholar] [CrossRef]
- Jeziorski, M.; White, F.J.; Wolf, M.E. MK-801 prevents the development of behavioral sensitization during repeated morphine administration. Synapse 1994, 16, 137–147. [Google Scholar] [CrossRef]
- Carlezon, W.A., Jr.; Rasmussen, K.; Nestler, E.J. AMPA antagonist LY293558 blocks the development, without blocking the expression, of behavioral sensitization to morphine. Synapse 1999, 31, 256–262. [Google Scholar] [CrossRef]
- Stafford, K.; Gomes, A.B.; Shen, J.; Yoburn, B.C. Mu-Opioid receptor downregulation contributes to opioid tolerance in vivo. Biochem. Behav. 2001, 69, 233–237. [Google Scholar] [CrossRef]
- Dang, V.C.; Christie, M.J. Mechanisms of rapid opioid receptor desensitization, resensitization and tolerance in brain neurons. Br. J. Pharmacol. 2012, 165, 1704–1716. [Google Scholar] [CrossRef] [PubMed]
- Anselmi, L.; Jaramillo, I.; Palacios, M.; Huynh, J.; Sternini, C. Ligand-induced μ opioid receptor internalization in enteric neurons following chronic treatment with the opiate fentanyl. J. Neurosci. Res. 2013, 9, 854–860. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J. Reflections on: “A general role for adaptations in G-Proteins and the cyclic AMP system in mediating the chronic actions of morphine and cocaine on neuronal function”. Brain Res. 2016, 1645, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Bohn, L.M.; Gainetdinov, R.R.; Lin, F.T.; Lefkowitz, R.J.; Caron, M.G. Mu-opioid receptor desensitization by beta-arrestin-2 determines morphine tolerance but not dependence. Nature 2000, 408, 720–723. [Google Scholar] [CrossRef] [PubMed]
- Bohn, L.M.; Lefkowitz, R.J.; Gainetdinov, R.R.; Peppel, K.; Caron, M.G.; Lin, F.T. Enhanced morphine analgesia in mice lacking beta-arrestin 2. Science 1999, 286, 2495–2498. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.L.; Zhang, J.S.; Zhang, X.Q.; Yue, W.; Ma, L. Differential regulation of beta-arrestin 1 and beta-arrestin 2 gene expression in rat brain by morphine. Neuroscience 2003, 117, 383–389. [Google Scholar] [CrossRef]
- Al-Hasani, R.; Bruchas, M.R. Molecular mechanisms of opioid receptor-dependent signaling and behavior. Anesthesiology 2011, 115, 1363–1381. [Google Scholar] [CrossRef]
- Narita, M.; Ioka, M.; Suzuki, M.; Narita, M.; Suzuki, T. Effect of repeated administration of morphine on the activity of extracellular signal regulated kinase in the mouse brain. Neurosci. Lett. 2002, 324, 97–100. [Google Scholar] [CrossRef]
- Macey, T.A.; Bobeck, E.N.; Hegarty, D.M.; Aicher, S.A.; Ingram, S.L.; Morgan, M.M. Extracellular signal-regulated kinase 1/2 activation counteracts morphine tolerance in the periaqueductal gray of the rat. J. Pharmacol. Exp. Ther. 2009, 331, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Ferrer-Alcón, M.; García-Fuster, M.J.; La Harpe, R.; García-Sevilla, J.A. Long-term regulation of signaling components of adenylyl cyclase and mitogen-activated protein kinase in the pre-frontal cortex of human opiate addicts. J. Neurochem. 2004, 90, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Muller, D.L.; Unterwald, E.M. In vivo regulation of extracellular signal-regulated protein kinase (ERK) and protein kinase B (Akt) phosphorylation by acute and chronic morphine. J. Pharmacol. Exp. Ther. 2004, 310, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Asensio, V.J.; Miralles, A.; García-Sevilla, J.A. Stimulation of mitogen-activated protein kinase kinases (MEK1/2) by mu-, delta- and kappa-opioid receptor agonists in the rat brain: Regulation by chronic morphine and opioid withdrawal. Eur. J. Pharmacol. 2006, 539, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, E.; Lehmann, D.; Vivoli, E.; Norcini, M.; Ghelardini, C. Involvement of PLC-beta3 in the effect of morphine on memory retrieval in passive avoidance task. J. Psychopharmacol. 2010, 24, 891–896. [Google Scholar] [CrossRef] [PubMed]
- Nabemoto, M.; Mashimo, M.; Someya, A.; Nakamura, H.; Hirabayashi, T.; Fujino, H.; Kaneko, M.; Okuma, Y.; Saito, T.; Yamaguchi, N.; et al. Release of arachidonic acid by 2-arachidonoyl glycerol and HU210 in PC12 cells; roles of Src, phospholipase C and cytosolic phospholipase A(2)alpha. Eur. J. Pharmacol. 2008, 590, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Smith, F.L.; Lohmann, A.B.; Dewey, W.L. Involvement of phospholipid signal transduction pathways in morphine tolerance in mice. Br. J. Pharmacol. 1999, 128, 220–226. [Google Scholar] [CrossRef]
- Williams, J.T.; Ingram, S.L.; Henderson, G.; Chavkin, C.; von Zastrow, M.; Schulz, S.; Koch, T.; Evans, C.J.; Christie, M.J. Regulation of µ-opioid receptors: Desensitization, phosphorylation, internalization, and tolerance. Pharmacol. Rev. 2013, 65, 223–254. [Google Scholar] [CrossRef]
- Skinner, M.K. Environmental epigenetic transgenerational inheritance and somatic epigenetic mitotic stability. Epigenetics 2011, 6, 838–842. [Google Scholar]
- Novikova, S.I.; He, F.; Bai, J.; Cutrufello, N.J.; Lidow, M.S.; Undieh, A.S. Maternal cocaine administration in mice alters DNA methylation and gene expression in hippocampal neurons of neonatal and prepubertal offspring. PLoS ONE 2008, 3, e1919. [Google Scholar] [CrossRef]
- Listos, J.; Talarek, S.; Gryzińska, M.; Listos, P.; Kędzierska, E.; Orzelska-Górka, J.; Łupina, M.; Kotlińska, J.H. Mephedrone exposure in adolescent rats alters the rewarding effect of morphine in adults. Eur. J. Pharmacol. 2017, 810, 63–69. [Google Scholar]
- Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002, 16, 6–21. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.K.; Song, K.Y.; Kim, C.S.; Choi, H.S.; Guo, X.H.; Law, P.Y.; Wei, L.N.; Loh, H.H. Evidence of endogenous mu opioid receptor regulation by epigenetic control of the promoters. Mol. Cell. Biol. 2007, 27, 4720–4736. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.K.; Kim, C.S.; Kim, D.K.; Law, P.Y.; Wei, L.N.; Loh, H.H. Up-regulation of the mu-opioid receptor gene is mediated through chromatin remodeling and transcriptional factors in differentiated neuronal cells. Mol. Pharmacol. 2010, 78, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Mashayekhi, F.J.; Rasti, M.; Rahvar, M.; Mokarram, P.; Namavar, M.R.; Owji, A.A. Expression levels of the BDNF gene and histone modifications around its promoters in the ventral tegmental area and locus ceruleus of rats during forced abstinence from morphine. Neurochem. Res. 2012, 37, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Maze, I.; Dietz, D.M.; Scobie, K.N.; Kennedy, P.J.; Damez-Werno, D.; Neve, R.L.; Zachariou, V.; Shen, L.; Nestler, E.J. Morphine epigenomically regulates behavior through alterations in histone H3 lysine 9 dimethylation in the nucleus accumbens. J. Neurosci. 2012, 32, 17454–17464. [Google Scholar] [CrossRef] [PubMed]
- Ciccarelli, A.; Calza, A.; Santoru, F.; Grasso, F.; Concas, A.; Sassoè-Pognetto, M.; Giustetto, M. Morphine withdrawal produces ERK-dependent and ERK-independent epigenetic marks in neurons of the nucleus accumbens and lateral septum. Neuropharmacology 2013, 70, 168–179. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, J.; Li, M.; Sui, N. Distinctive Roles of 5-aza-2′-deoxycytidine in Anterior Agranular Insular and Basolateral Amygdala in Reconsolidation of Aversive Memory Associated with Morphine in Rats. Front. Behav. Neurosci. 2016, 10, 50. [Google Scholar] [CrossRef]
- Zhang, J.J.; Han, J.; Sui, N. Okadaic acid blocks the effects of 5-aza-2-deoxycytidine on consolidation, acquisition and retrieval of morphine-induced place preference in rats. Neuropharmacology 2014, 86, 282–293. [Google Scholar] [CrossRef]
- Barrow, T.M.; Byun, H.M.; Li, X.; Smart, C.; Wang, Y.X.; Zhang, Y.; Baccarelli, A.A.; Guo, L. The effect of morphine upon DNA methylation in ten regions of the rat brain. Epigenetics 2017, 12, 1038–1047. [Google Scholar] [CrossRef]
- Chen, X.T.; Pitis, P.; Liu, G.; Yuan, C.; Gotchev, D.; Cowan, C.L.; Rominger, D.H.; Koblish, M.; Dewire, S.M.; Crombie, A.L.; et al. Structure-activity relationships and discovery of a G protein biased µ opioid receptor ligand, [(3-methoxythiophen-2-yl)methyl]({2-[(9R)-9-(pyridin-2-yl)-6-oxaspiro-[4.5]de can-9-yl]ethyl})amine (TRV130), for the treatment of acute severe pain. J. Med. Chem. 2013, 56, 8019–8031. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.X.; Cheng, T.; Li, W.H.; Liu, G.X.; Zhu, W.L.; Tang, Y. Computational insights into the G-protein-biased activation and inactivation mechanisms of the μ opioid receptor. Acta Pharmacol. Sin. 2018, 39, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Madariaga-Mazón, A.; Marmolejo-Valencia, A.F.; Li, Y.; Toll, L.; Houghten, R.A.; Martinez-Mayorga, K. Mu-Opioid receptor biased ligands: A safer and painless discovery of analgesics? Drug Discov. Today 2017, 22, 1719–1729. [Google Scholar] [CrossRef] [PubMed]
- Manglik, A.; Lin, H.; Aryal, D.K.; McCorvy, J.D.; Dengler, D.; Corder, G.; Levit, A.; Kling, R.C.; Bernat, V.; Hübner, H.; et al. Structure-based discovery of opioid analgesics with reduced side effects. Nature 2016, 537, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Negus, S.S.; Freeman, K.B. Abuse Potential of Biased Mu Opioid Receptor Agonists. Trends Pharmacol. Sci. 2018, 39, 916–919. [Google Scholar] [CrossRef]
- Ranjan, R.; Pandey, S.; Shukla, A.K. Biased Opioid Receptor Ligands: Gain without Pain. Trends Endocrinol. Metab. 2017, 28, 247–249. [Google Scholar] [CrossRef] [PubMed]
| Drug Names | Structural Formula | Indications |
|---|---|---|
| Alfentanil | 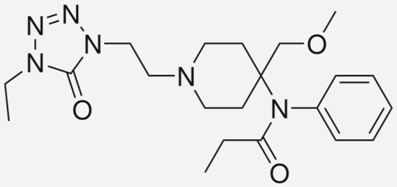 | - anaesthesia in surgery [17] |
| Buprenorphine |  | - relieve moderate-to-severe pain [18] - substitute treatment for opioid addiction [19] - Neonatal Abstinence Syndrome [20] |
| Butorphanol | 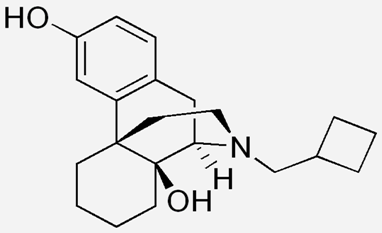 | - treat moderate-to-severe pain [21] - relieve acute morphine-induced pruritus [22] |
| Codeine | 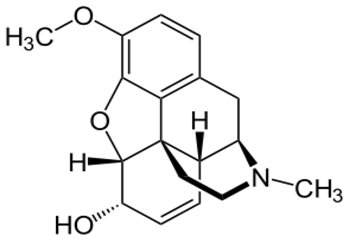 | - treatment of chronic cough [23] - relief of moderate-to-severe pain [24] - treat persistent diarrhoea [25] |
| Dextromethorphan | 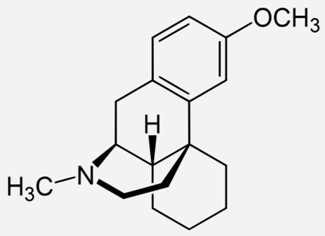 | - temporary relief of coughs without phlegm [26] |
| Diphenoxylate | 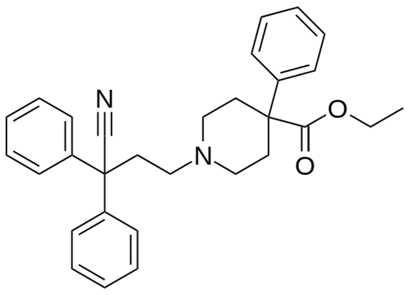 | - acute and chronic diarrhoea of various origins [27] - reduction in the amount of faecal fluid after ileostomy and colostomy [28] |
| Dihydrocodeine | 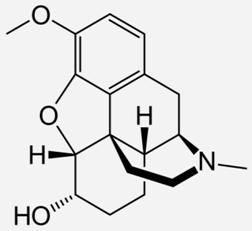 | - treat moderate-to-severe pain [29] - treat dry cough [29] - treat diarrhoea [29] |
| Fentanyl |  | - treatment of severe, chronic pain [30] - used for surgical anaesthesia [30] |
| Hydrocodone | 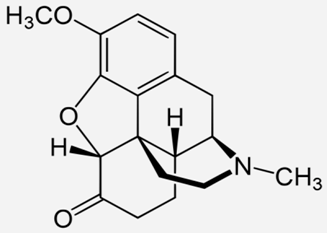 | - the management of pain severe enough to require daily, around-the-clock use [31] |
| Hydromorphone | 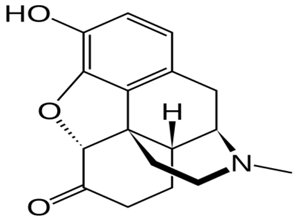 | - relieve moderate-to-severe pain [32] |
| Laevodropropizine | 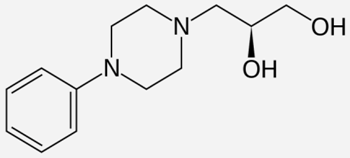 | - treat dry cough [33] |
| Levorphanol |  | - use in moderate-to-severe pain [34] |
| Loperamide | 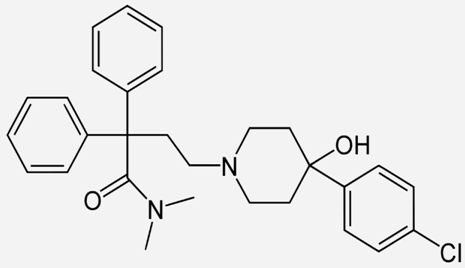 | - stop diarrhoea [35] |
| Meptazinol | 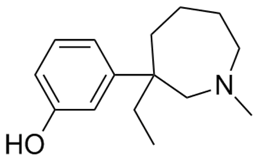 | - relieve moderate-to-severe pain (among others, obstetrics) [36] |
| Methadone | 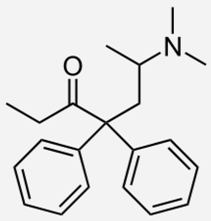 | - treatment of opiate dependence [37] - the treatment chronic, severe pain [38] - treatment of neonatal abstinence syndrome [39] |
| Morphine | 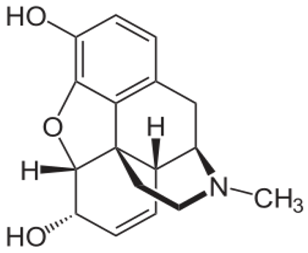 | - moderate-to-severe pain relief [40] - used for procedural sedation [40] - sporadically used as an antitussive drug [41] - treatment of Neonatal Abstinence Syndrome [39] |
| Nalbuphine | 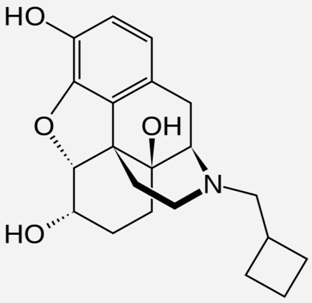 | - itching treatment [42] - recommended for weak-to-moderately severe pain [42] - sedation [43] - anaesthesia [44] |
| Nalmefene | 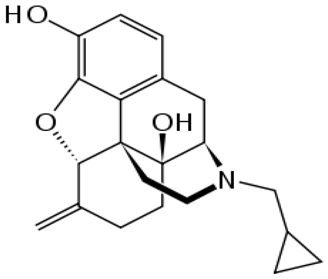 | - reduction of alcohol consumption [45] |
| Naloxone | 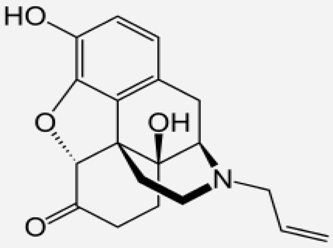 | - Treatment of poisoning, overdose of opioid substances [46] - reversal of undesirable effects from opioid used during anaesthesia [47] - counteracting the occurrence of opioid-induced constipation [48] - substitute treatment of opioid dependence [49] |
| Naltrexone | 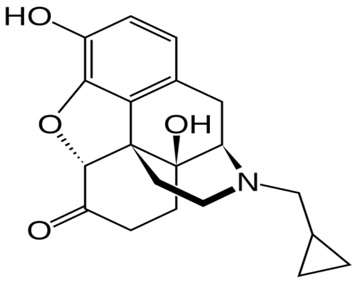 | - treatment of alcoholism [50] |
| Oxycodone | 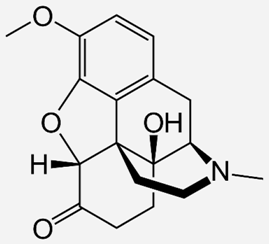 | - treatment of opioid-induced constipation [48] - pain treatment [51] |
| Pentazocine |  | - treatment of moderate-to-severe pain [52] - a preanaesthetic or preoperative medication [52] |
| Pethidine | 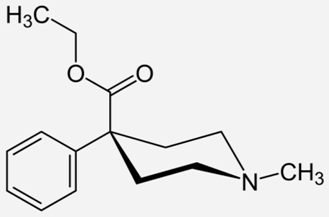 | - the treatment of moderate-to-severe pain [53] - used as an adjunct to preoperative medications to reduce shivering [53] - relief of childbirth pain [54] |
| Remifentanil | 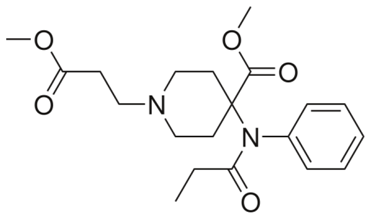 | - relief of childbirth pain [54] - treatment of moderate-to-severe pain [55] - providing anaesthesia and sedation [55] |
| Sulfentanil | 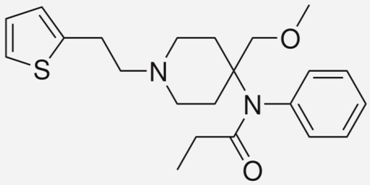 | - management of moderate-to-severe pain [56] - anaesthesia [56] |
| Tapentadol | 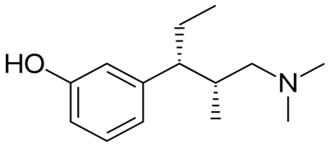 | - to help relieve moderate-to-severe pain [57] |
| Tilidine |  | - treatment of pain [58] |
| Tramadol | 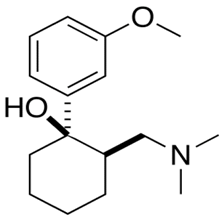 | - treat moderate-to-severe chronic pain [59] - relief of childbirth pain [59] - anaesthesia [60] - premature ejaculation [61] |
| Experimental Procedures for Phases of Addiction |
|---|
| Morphine dependence is obtained commonly by chronic administration of increasing doses (from 10 to 50–100 mg/kg) of morphine, twice a day for 5–9 consecutive days. |
| Morphine withdrawal is obtained in morphine dependent animals either by discontinuation of chronic morphine administration or via administration of an opioid receptor antagonist, such as naloxone, at a range of doses from 1 to 6 mg/kg. The severity of morphine withdrawal symptoms is analyzed on the basis of the number of withdrawal episodes, such as jumpings, paw tremors, teeth chattering, wet dog shakes and diarrhoea. |
| Morphine tolerance is obtained by repeated administration of the same dose of morphine (10 mg/kg) for several (3–7) consecutive days. Commonly, it is analyzed by comparison of the reaction of animals on nociceptive stimulus, recorded on the first and last day of morphine administration. Morphine tolerance commonly is measured in behavioural tests, such as the tail immersion test or the hot plate test. |
| Morphine-induced behavioral sensitization is related closely to the environment in which the addictive substance is taken and reflects morphine-seeking behavior in studied animals. It is obtained by administration of a challenge dose of morphine (at range of 1–10 mg/kg) in morphine dependent animals after several days (7–10) of a morphine-free period. It is measured as an increase in locomotor activity of animals, rarely as the enhanced rewarding effect. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Listos, J.; Łupina, M.; Talarek, S.; Mazur, A.; Orzelska-Górka, J.; Kotlińska, J. The Mechanisms Involved in Morphine Addiction: An Overview. Int. J. Mol. Sci. 2019, 20, 4302. https://doi.org/10.3390/ijms20174302
Listos J, Łupina M, Talarek S, Mazur A, Orzelska-Górka J, Kotlińska J. The Mechanisms Involved in Morphine Addiction: An Overview. International Journal of Molecular Sciences. 2019; 20(17):4302. https://doi.org/10.3390/ijms20174302
Chicago/Turabian StyleListos, Joanna, Małgorzata Łupina, Sylwia Talarek, Antonina Mazur, Jolanta Orzelska-Górka, and Jolanta Kotlińska. 2019. "The Mechanisms Involved in Morphine Addiction: An Overview" International Journal of Molecular Sciences 20, no. 17: 4302. https://doi.org/10.3390/ijms20174302
APA StyleListos, J., Łupina, M., Talarek, S., Mazur, A., Orzelska-Górka, J., & Kotlińska, J. (2019). The Mechanisms Involved in Morphine Addiction: An Overview. International Journal of Molecular Sciences, 20(17), 4302. https://doi.org/10.3390/ijms20174302






