Telmisartan Lowers Elevated Blood Pressure in Psoriatic Mice without Attenuating Vascular Dysfunction and Inflammation
Abstract
1. Introduction
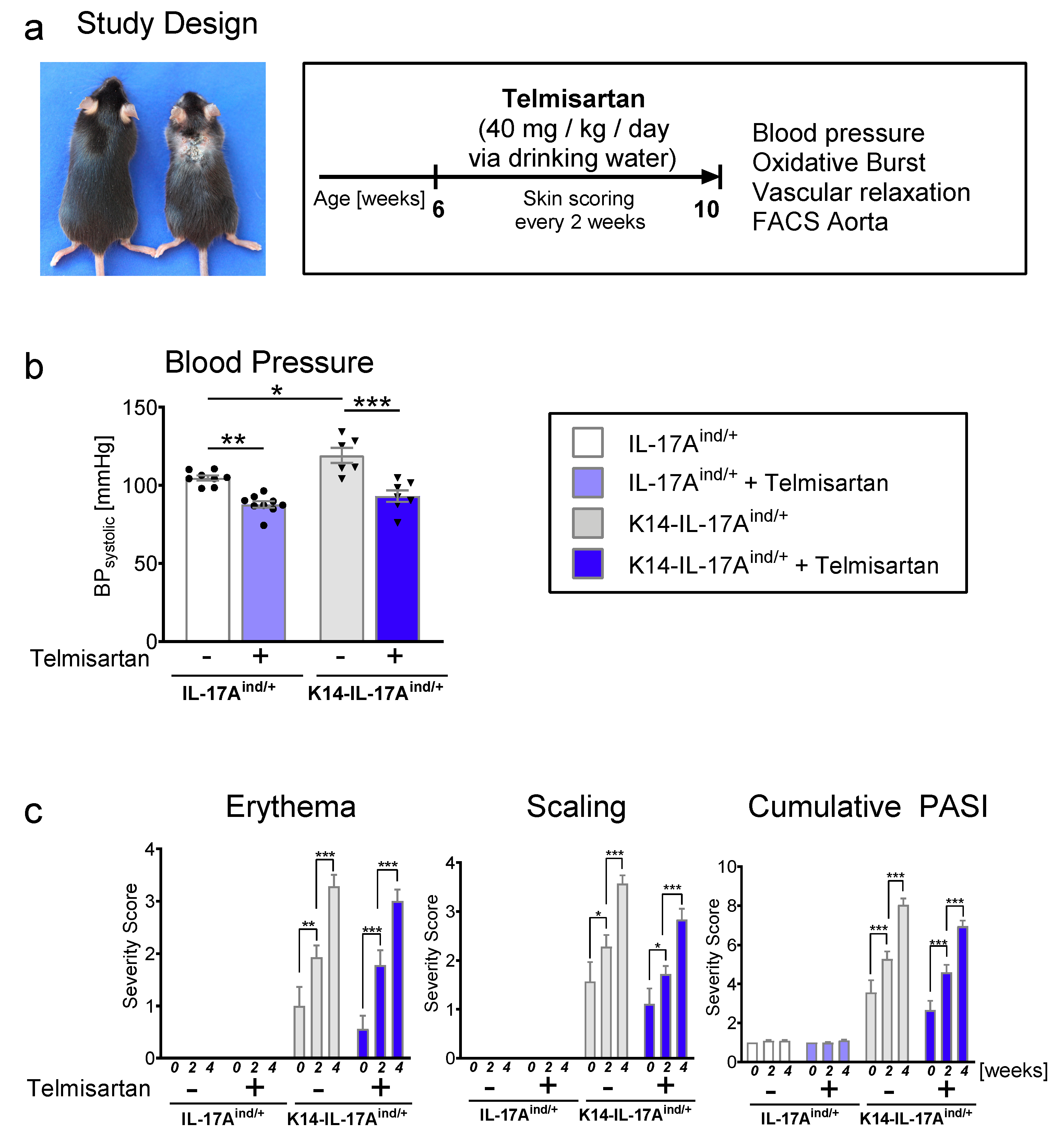
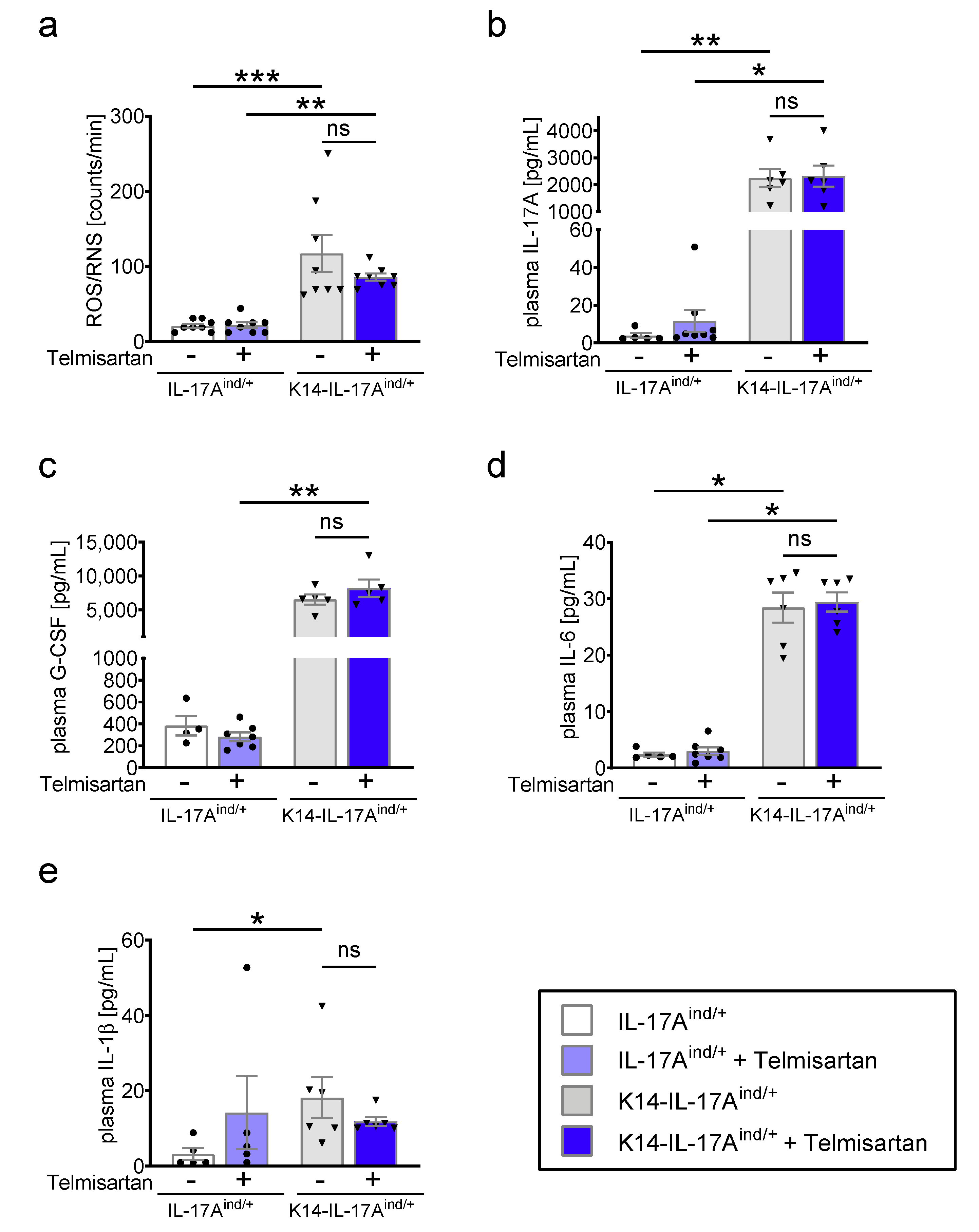
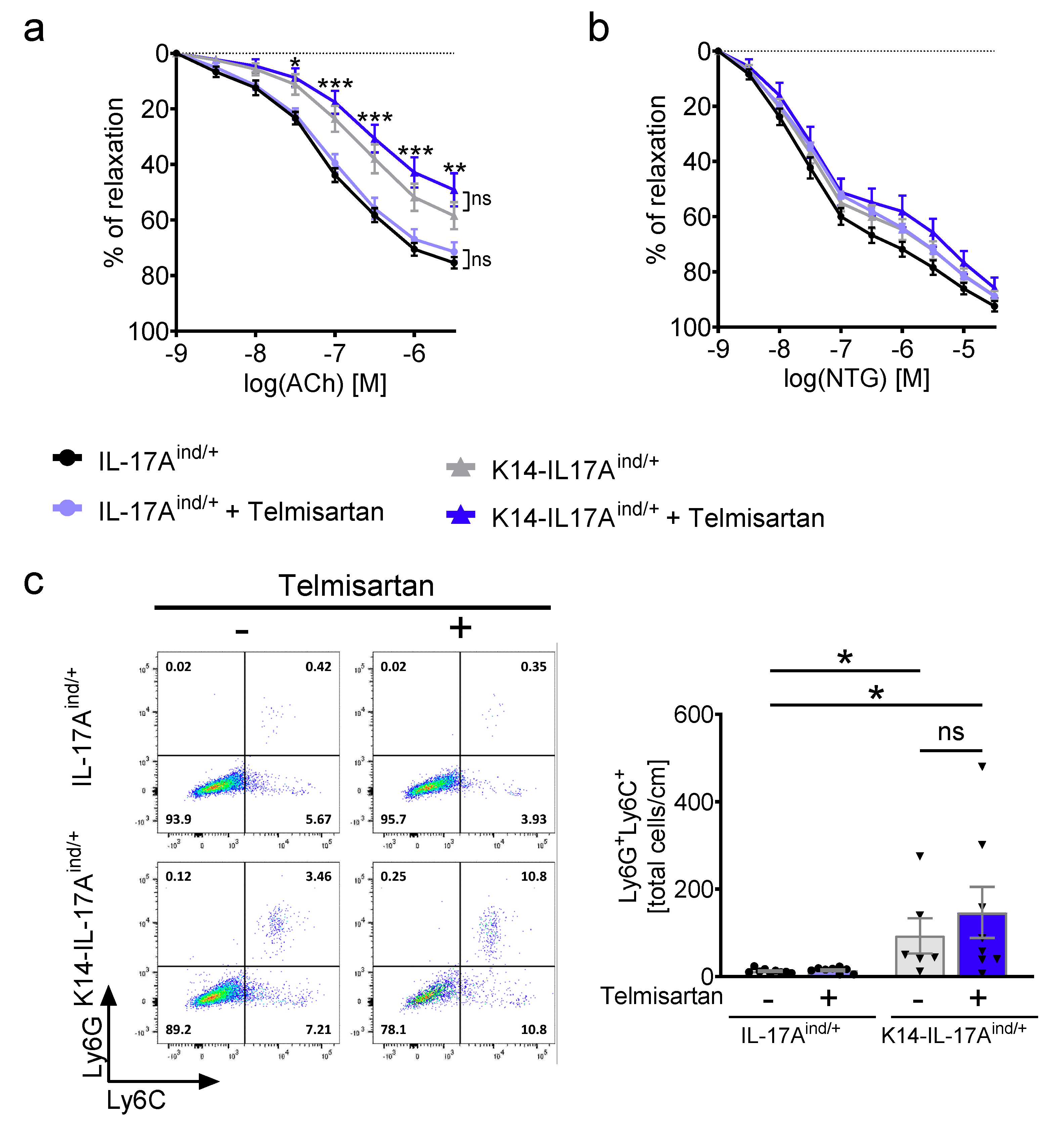
2. Results
3. Discussion
4. Materials and Methods
4.1. Mouse Models of Psoriasis-Like Skin Disease
4.2. Blood Pressure Measurements
4.3. Psoriasis Area and Severity Index (PASI)
4.4. Detection of ROS/RNS Formation with L-012-Enhanced Chemiluminescence
4.5. Vascular Tone Experiments
4.6. Cytokine Detection
4.7. Flow Cytometric Analysis
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Takeshita, J.; Grewal, S.; Langan, S.M.; Mehta, N.N.; Ogdie, A.; Van Voorhees, A.S.; Gelfand, J.M. Psoriasis and comorbid diseases: Implications for management. J. Am. Acad. Dermatol. 2017, 76, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Mehta, N.N.; Azfar, R.S.; Shin, D.B.; Neimann, A.L.; Troxel, A.B.; Gelfand, J.M. Patients with severe psoriasis are at increased risk of cardiovascular mortality: Cohort study using the General Practice Research Database. Eur. Heart J. 2010, 31, 1000–1006. [Google Scholar] [CrossRef] [PubMed]
- Gelfand, J.M.; Azfar, R.S.; Mehta, N.N. Psoriasis and cardiovascular risk: Strength in numbers. J. Investig. Dermatol. 2010, 130, 919–922. [Google Scholar] [CrossRef] [PubMed]
- Ahlehoff, O.; Skov, L.; Gislason, G.; Lindhardsen, J.; Kristensen, S.L.; Iversen, L.; Lasthein, S.; Gniadecki, R.; Dam, T.N.; Torp-Pedersen, C.; et al. Pharmacological undertreatment of coronary risk factors in patients with psoriasis: Observational study of the Danish nationwide registries. PLoS ONE 2012, 7, e36342. [Google Scholar] [CrossRef] [PubMed]
- Duarte, G.V.; Oliveira, M.F.; Follador, I.; Silva, T.S.; Carvalho, E.M.F. Diagnosis and underdiagnosis of comorbidities in psoriasis patients-need for a multidisciplinary approach. An. Bras. Dermatol. 2016, 91, 743–747. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, S.S.; Levy, D.; Vasan, R.S.; Wang, T.J. The Framingham Heart Study and the epidemiology of cardiovascular disease: A historical perspective. Lancet 2014, 383, 999–1008. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.W.; Harskamp, C.T.; Armstrong, E.J. The association between psoriasis and hypertension: A systematic review and meta-analysis of observational studies. J. Hypertens. 2013, 31, 433–442, discussion 442–433. [Google Scholar] [CrossRef]
- Takeshita, J.; Grewal, S.; Langan, S.M.; Mehta, N.N.; Ogdie, A.; Van Voorhees, A.S.; Gelfand, J.M. Psoriasis and comorbid diseases: Epidemiology. J. Am. Acad. Dermatol. 2017, 76, 377–390. [Google Scholar] [CrossRef]
- Takeshita, J.; Wang, S.; Shin, D.B.; Mehta, N.N.; Kimmel, S.E.; Margolis, D.J.; Troxel, A.B.; Gelfand, J.M. Effect of psoriasis severity on hypertension control: A population-based study in the United Kingdom. JAMA Dermatol. 2015, 151, 161–169. [Google Scholar] [CrossRef]
- Karbach, S.; Croxford, A.L.; Oelze, M.; Schüler, R.; Minwegen, D.; Wegner, J.; Koukes, L.; Yogev, N.; Nikolaev, A.; Reißig, S.; et al. Interleukin 17 drives vascular inflammation, endothelial dysfunction, and arterial hypertension in psoriasis-like skin disease. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 2658–2668. [Google Scholar] [CrossRef] [PubMed]
- Schüler, R.; Brand, A.; Klebow, S.; Wild, J.; Veras, F.P.; Ullmann, E.; Roohani, S.; Kolbinger, F.; Kossmann, S.; Wohn, C.; et al. Antagonization of IL-17A Attenuates Skin Inflammation and Vascular Dysfunction in Mouse Models of Psoriasis. J. Investig. Dermatol. 2019, 139, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Roubille, C.; Richer, V.; Starnino, T.; McCourt, C.; McFarlane, A.; Fleming, P.; Siu, S.; Kraft, J.; Lynde, C.; Pope, J.; et al. Evidence-based Recommendations for the Management of Comorbidities in Rheumatoid Arthritis, Psoriasis, and Psoriatic Arthritis: Expert Opinion of the Canadian Dermatology-Rheumatology Comorbidity Initiative. J. Rheumatol. 2015, 42, 1767–1780. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, P.; Schulz, E.; Oelze, M.; Müller, J.; Schuhmacher, S.; Alhamdani, M.S.; Debrezion, J.; Hortmann, M.; Reifenberg, K.; Fleming, I.; et al. AT1-receptor blockade by telmisartan upregulates GTP-cyclohydrolase I and protects eNOS in diabetic rats. Free Radic. Biol. Med. 2008, 45, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Croxford, A.L.; Karbach, S.; Kurschus, F.C.; Wörtge, S.; Nikolaev, A.; Yogev, N.; Klebow, S.; Schüler, R.; Reissig, S.; Piotrowski, C.; et al. IL-6 regulates neutrophil microabscess formation in IL-17A-driven psoriasiform lesions. J. Investig. Dermatol. 2014, 134, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Syrbe, U.; Moebes, A.; Scholze, J.; Swidsinski, A.; Dorffel, Y. Effects of the angiotensin II type 1 receptor antagonist telmisartan on monocyte adhesion and activation in patients with essential hypertension. Hypertens. Res. Off. J. Jpn. Soc. Hypertens. 2007, 30, 521–528. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Song, K.H.; Bae, S.J.; Chang, J.; Park, J.H.; Jo, I.; Cho, K.W.; Cho, D.H. Telmisartan mitigates hyperglycemia-induced vascular inflammation by increasing GSK3beta-Ser (9) phosphorylation in endothelial cells and mouse aortas. Biochem. Biophy. Res. Commun. 2017, 491, 903–911. [Google Scholar] [CrossRef]
- Dey, A.K.; Joshi, A.A.; Chaturvedi, A.; Lerman, J.B.; Aberra, T.M.; Rodante, J.A.; Teague, H.L.; Harrington, C.L.; Rivers, J.P.; Chung JHKabbany, M.T. Association Between Skin and Aortic Vascular Inflammation in Patients With Psoriasis: A Case-Cohort Study Using Positron Emission Tomography/Computed Tomography. JAMA Cardiol. 2017, 2, 1013–1018. [Google Scholar] [CrossRef]
- Shaaban, D.; Al-Mutairi, N. The effect of tumor necrosis factor inhibitor therapy on the incidence of myocardial infarction in patients with psoriasis: A retrospective study. J. Dermatol. Treat. 2018, 29, 3–7. [Google Scholar] [CrossRef]
- Ahlehoff, O.; Skov, L.; Gislason, G.; Lindhardsen, J.; Kristensen, S.L.; Iversen, L.; Lasthein, S.; Gniadecki, R.; Dam, T.N.; Torp-Pedersen CHansen, P.R. Cardiovascular disease event rates in patients with severe psoriasis treated with systemic anti-inflammatory drugs: A Danish real-world cohort study. J. Intern. Med. 2013, 273, 197–204. [Google Scholar] [CrossRef]
- Haak, S.; Croxford, A.L.; Kreymborg, K.; Heppner, F.L.; Pouly, S.; Becher BWaisman, A. IL-17A and IL-17F do not contribute vitally to autoimmune neuro-inflammation in mice. J. Clin. Investig. 2009, 119, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Hafner, M.; Wenk, J.; Nenci, A.; Pasparakis, M.; Scharffetter-Kochanek, K.; Smyth, N.; Peters, T.; Kess, D.; Holtkötter, O.; Shephard, P.; et al. Keratin 14 Cre transgenic mice authenticate keratin 14 as an oocyte-expressed protein. Genesis 2004, 38, 176–181. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wild, J.; Schüler, R.; Knopp, T.; Molitor, M.; Kossmann, S.; Münzel, T.; Daiber, A.; Waisman, A.; Wenzel, P.; Karbach, S.H. Telmisartan Lowers Elevated Blood Pressure in Psoriatic Mice without Attenuating Vascular Dysfunction and Inflammation. Int. J. Mol. Sci. 2019, 20, 4261. https://doi.org/10.3390/ijms20174261
Wild J, Schüler R, Knopp T, Molitor M, Kossmann S, Münzel T, Daiber A, Waisman A, Wenzel P, Karbach SH. Telmisartan Lowers Elevated Blood Pressure in Psoriatic Mice without Attenuating Vascular Dysfunction and Inflammation. International Journal of Molecular Sciences. 2019; 20(17):4261. https://doi.org/10.3390/ijms20174261
Chicago/Turabian StyleWild, Johannes, Rebecca Schüler, Tanja Knopp, Michael Molitor, Sabine Kossmann, Thomas Münzel, Andreas Daiber, Ari Waisman, Philip Wenzel, and Susanne Helena Karbach. 2019. "Telmisartan Lowers Elevated Blood Pressure in Psoriatic Mice without Attenuating Vascular Dysfunction and Inflammation" International Journal of Molecular Sciences 20, no. 17: 4261. https://doi.org/10.3390/ijms20174261
APA StyleWild, J., Schüler, R., Knopp, T., Molitor, M., Kossmann, S., Münzel, T., Daiber, A., Waisman, A., Wenzel, P., & Karbach, S. H. (2019). Telmisartan Lowers Elevated Blood Pressure in Psoriatic Mice without Attenuating Vascular Dysfunction and Inflammation. International Journal of Molecular Sciences, 20(17), 4261. https://doi.org/10.3390/ijms20174261





