Surfactant Protein B Deficiency Induced High Surface Tension: Relationship between Alveolar Micromechanics, Alveolar Fluid Properties and Alveolar Epithelial Cell Injury
Abstract
1. Introduction
2. Results
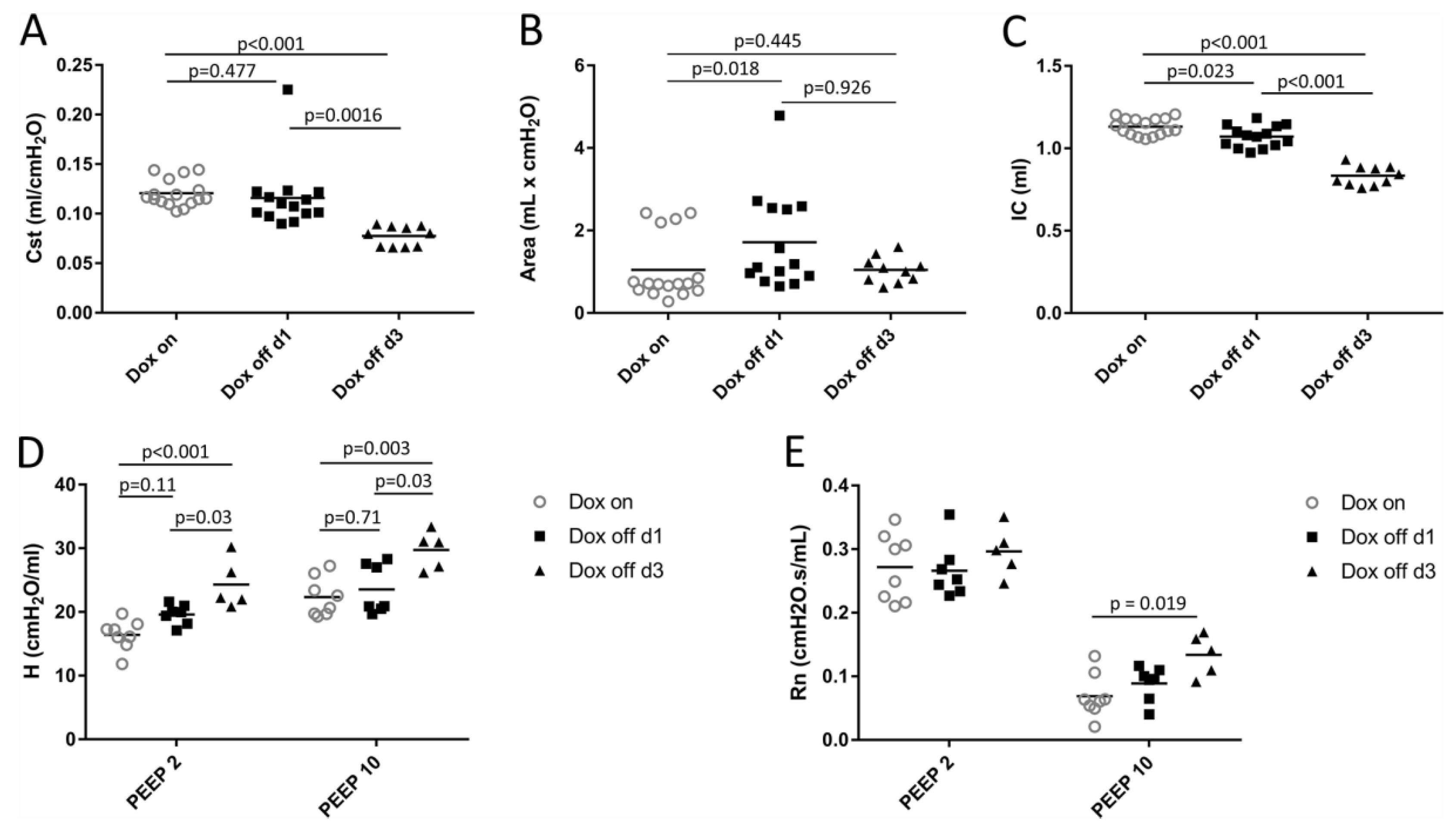
2.1. Lung Mechanics
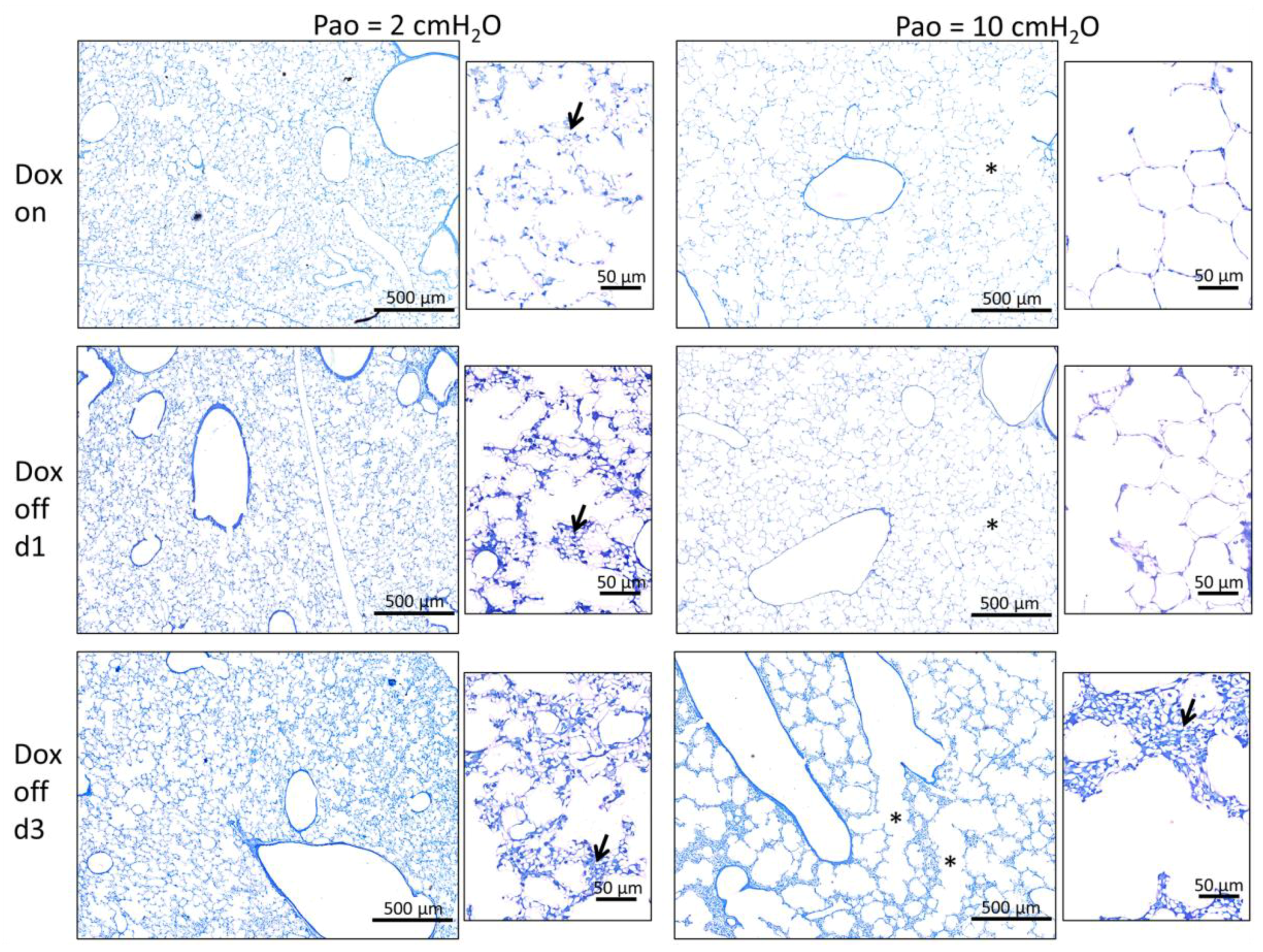
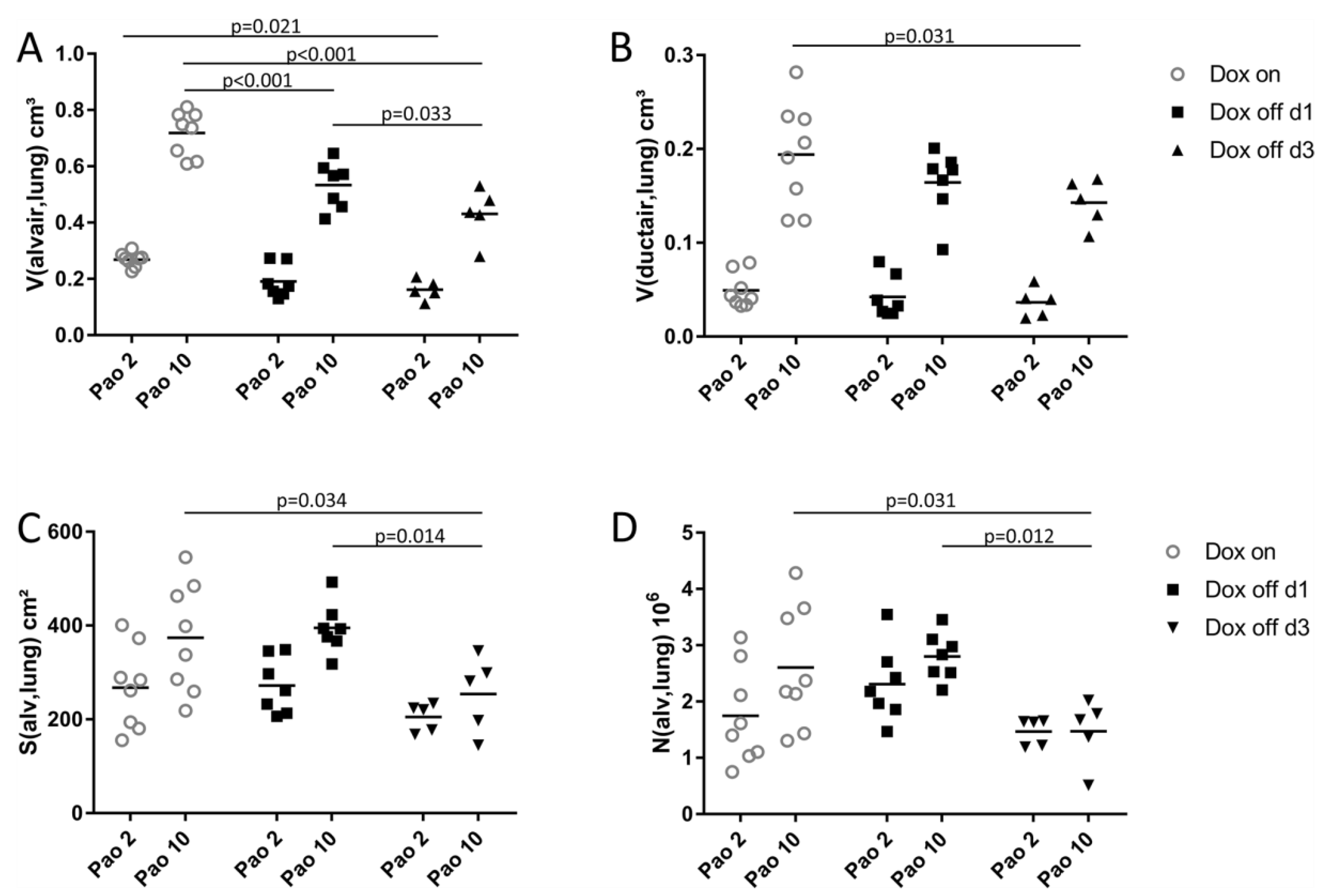
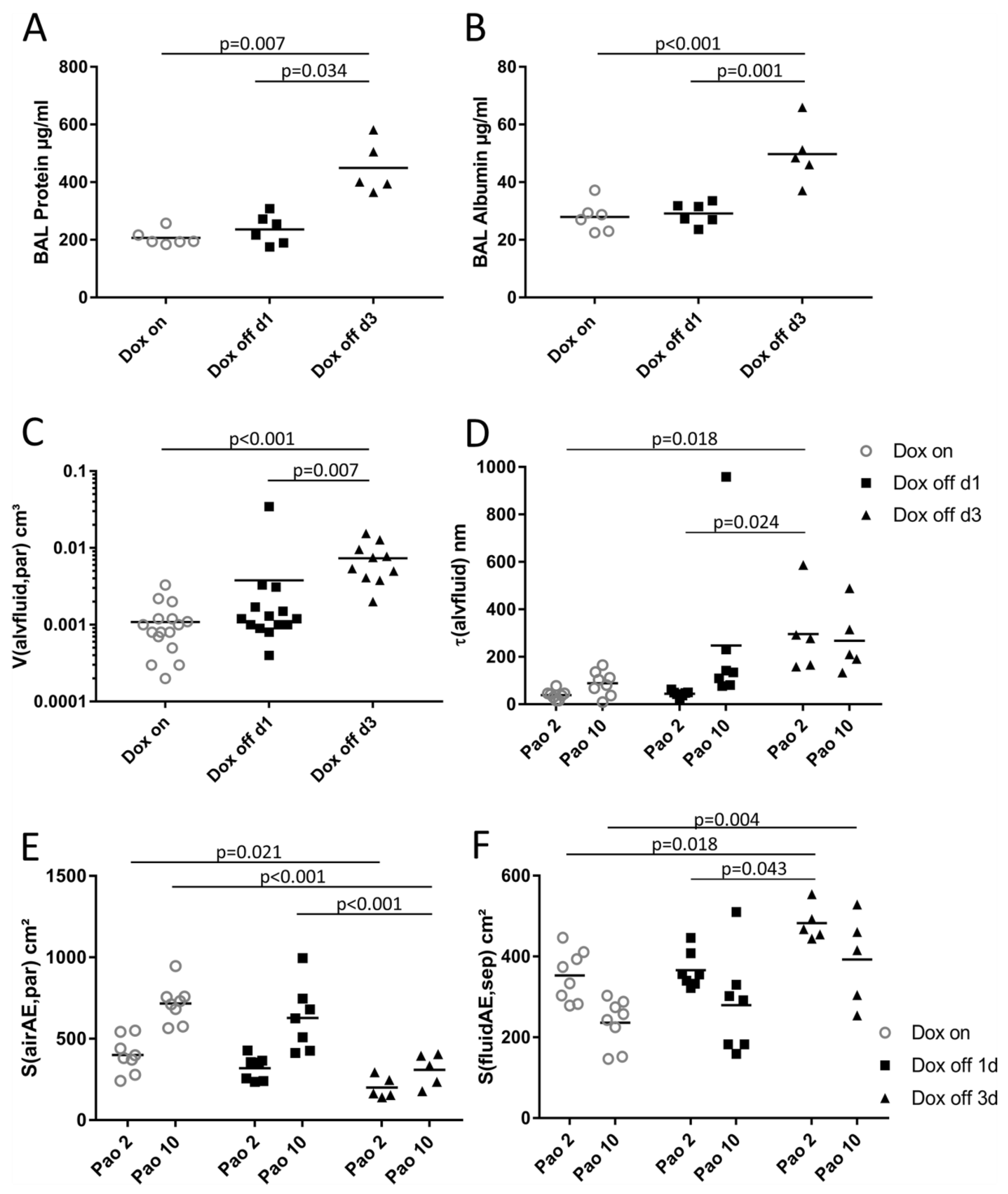
2.2. Progressive Disturbances of Acinar Micromechanics in Dox off Groups
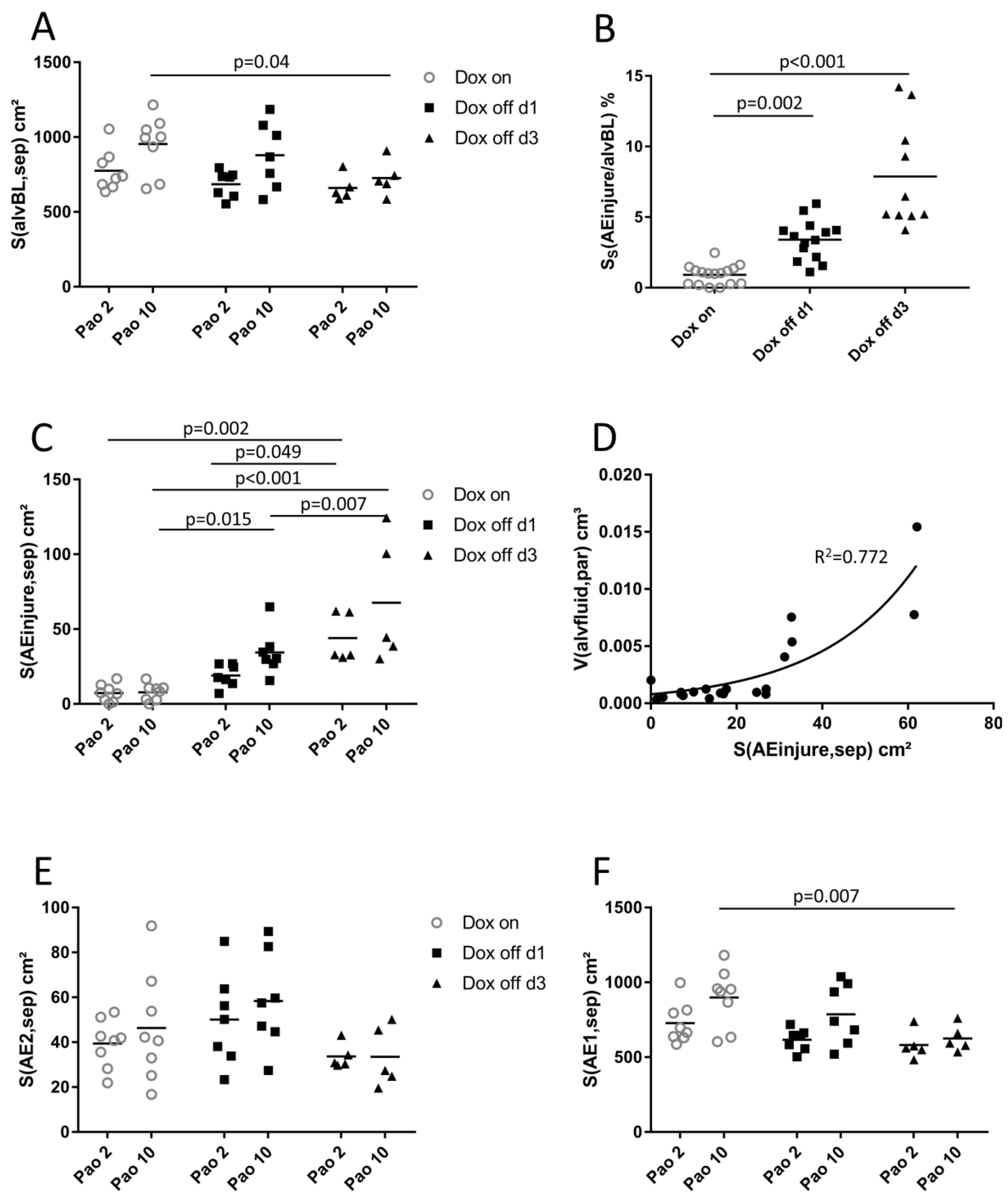
2.3. Composition of Inter-alveolar Septa and Intra-alveolar Fluid Properties
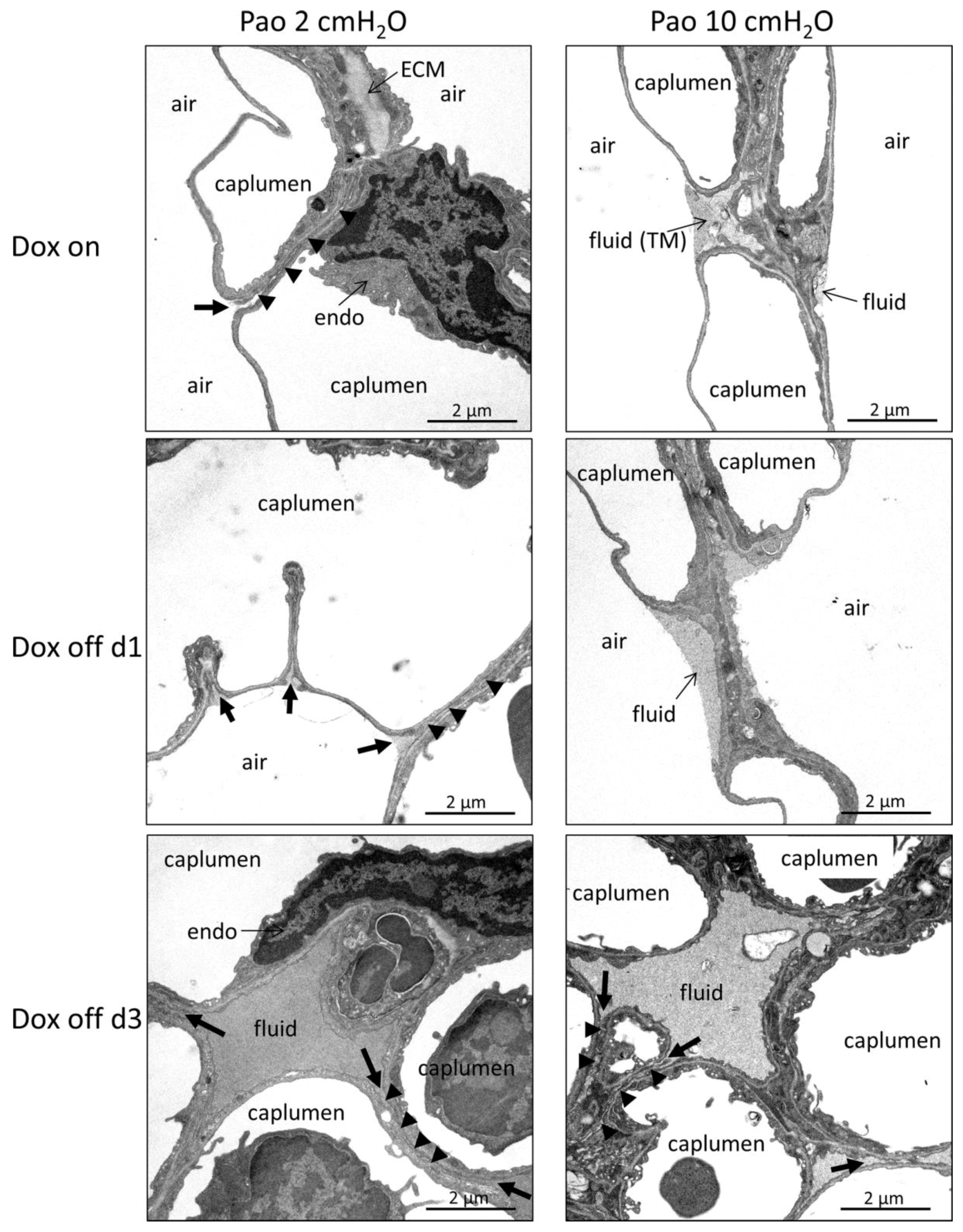
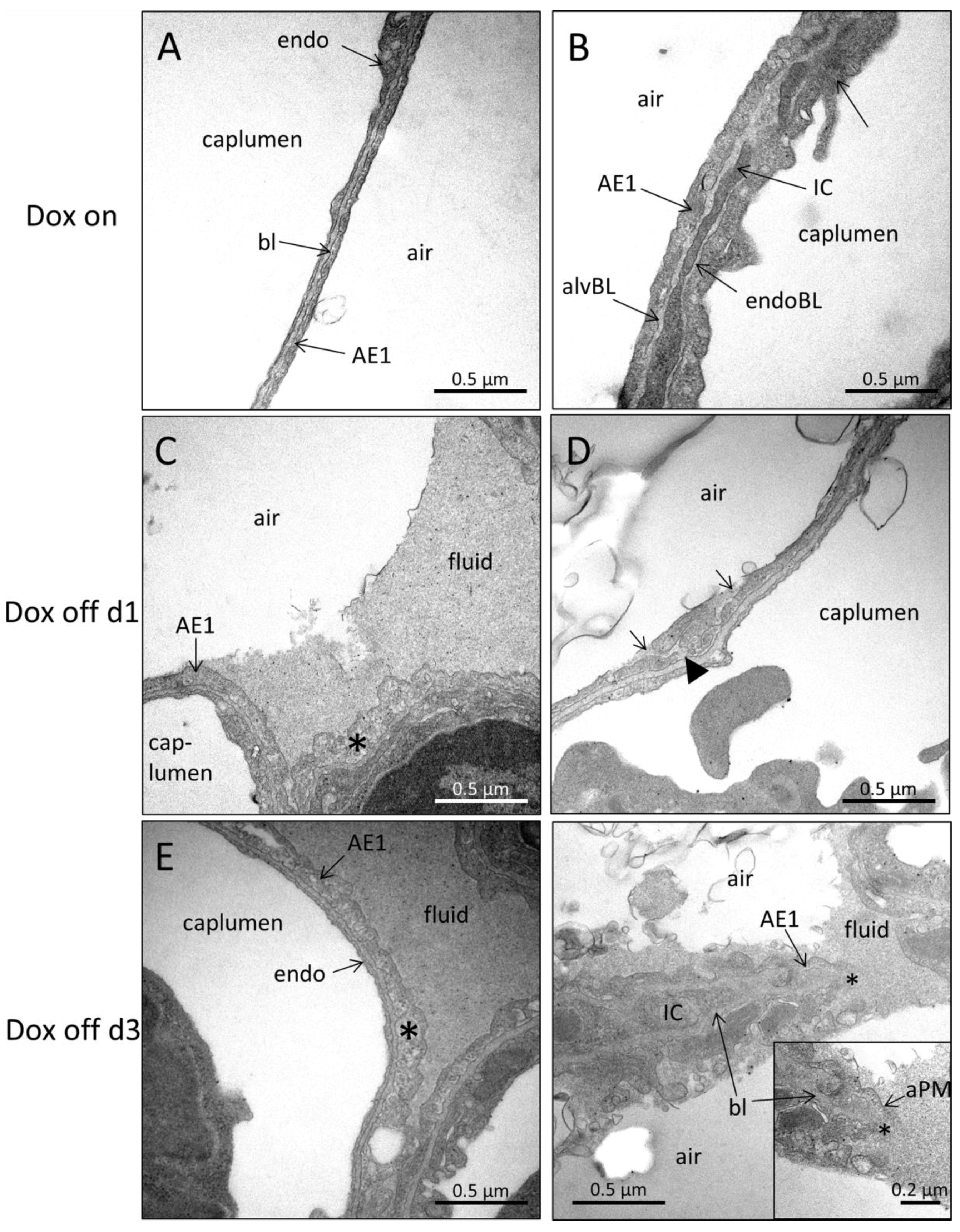
2.4. Ultrastructural Evaluation of Alveolar Epithelial Injury
2.5. Structure-function Relationships
3. Discussion
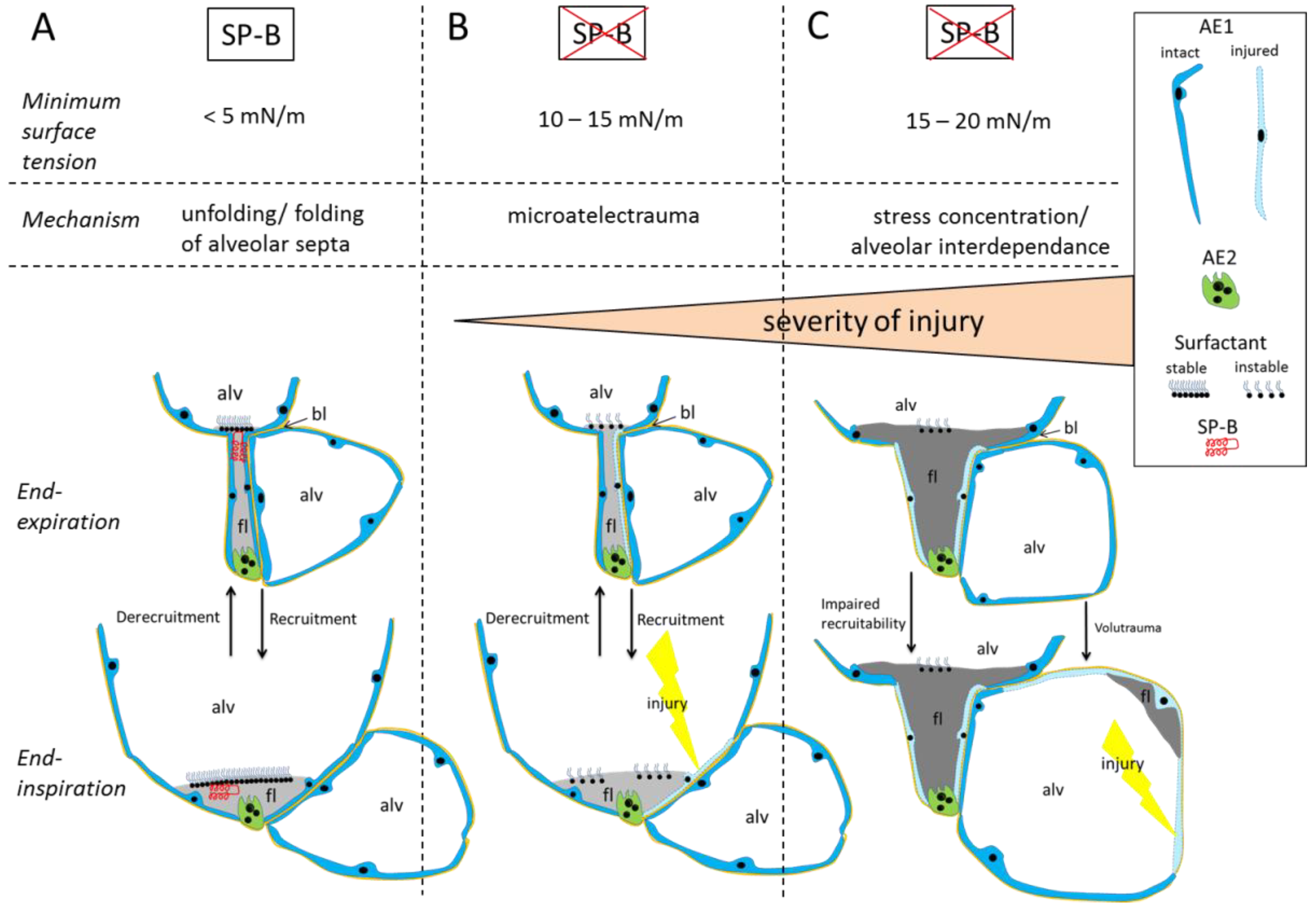
3.1. Alveolar Micromechanics
3.2. Alveolar Fluid Properties and the Relationship to Alveolar Micromechanics
3.3. Injury of Alveolar Epithelium in the Context of impaired Alveolar Micromechanics and Fluid Accumulation
3.4. Limitations
3.5. Summary
4. Materials and Methods
4.1. Animal Model and Study Groups
4.2. Experimental Protocol
4.3. Perfusion Fixation, Preparation and Sampling
4.4. Embedding for Light Microscopy
4.5. Embedding for Electron Microscopy
4.6. Design-based Stereology
4.7. Analyses of Broncho-alveolar Lavage Fluid
4.8. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lopez-Rodriguez, E.; Gay-Jordi, G.; Mucci, A.; Lachmann, N.; Serrano-Mollar, A. Lung surfactant metabolism: Early in life, early in disease and target in cell therapy. Cell Tissue Res. 2017, 367, 721–735. [Google Scholar] [CrossRef] [PubMed]
- Schürch, D.; Ospina, O.L.; Cruz, A.; Pérez-Gil, J. Combined and independent action of proteins SP-B and SP-C in the surface behavior and mechanical stability of pulmonary surfactant films. Biophys. J. 2010, 99, 3290–3299. [Google Scholar] [CrossRef] [PubMed]
- Ochs, M. The closer we look the more we see? Quantitative microscopic analysis of the pulmonary surfactant system. Cell Physiol. Biochem. 2010, 25, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Mead, J. Mechanical properties of lungs. Physiol. Rev. 1961, 41, 281–330. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, L.; Ochs, M. The micromechanics of lung alveoli: Structure and function of surfactant and tissue components. Histochem. Cell Biol. 2018, 150, 661–676. [Google Scholar] [CrossRef] [PubMed]
- Bachofen, H.; Schürch, S.; Urbinelli, M.; Weibel, E. Relations among alveolar surface tension, surface area, volume, and recoil pressure. J. Appl. Physiol. (1985) 1987, 62, 1878–1887. [Google Scholar] [CrossRef] [PubMed]
- Schürch, S.; Bachofen, H.; Possmayer, F. Surface activity in situ, in vivo, and in the captive bubble surfactometer. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 129, 195–207. [Google Scholar] [CrossRef]
- Almlén, A.; Stichtenoth, G.; Linderholm, B.; Haegerstrand-Björkman, M.; Robertson, B.; Johansson, J.; Curstedt, T. Surfactant proteins B and C are both necessary for alveolar stability at end expiration in premature rabbits with respiratory distress syndrome. J. Appl. Physiol. (1985) 2008, 104, 1101–1108. [Google Scholar] [CrossRef][Green Version]
- Matute-Bello, G.; Downey, G.; Moore, B.B.; Groshong, S.D.; Matthay, M.A.; Slutsky, A.S.; Kuebler, W.M.; Group, A.L.I.i.A.S. An official American Thoracic Society workshop report: Features and measurements of experimental acute lung injury in animals. Am. J. Respir. Cell Mol. Biol. 2011, 44, 725–738. [Google Scholar] [CrossRef]
- Cabrera-Benítez, N.E.; Parotto, M.; Post, M.; Han, B.; Spieth, P.M.; Cheng, W.E.; Valladares, F.; Villar, J.; Liu, M.; Sato, M.; et al. Mechanical stress induces lung fibrosis by epithelial-mesenchymal transition. Crit. Care Med. 2012, 40, 510–517. [Google Scholar] [CrossRef]
- Wilson, T.A.; Bachofen, H. A model for mechanical structure of the alveolar duct. J. Appl. Physiol. (1985) 1982, 52, 1064–1070. [Google Scholar] [CrossRef] [PubMed]
- Schiller, H.J.; McCann, U.G.; Carney, D.E.; Gatto, L.A.; Steinberg, J.M.; Nieman, G.F. Altered alveolar mechanics in the acutely injured lung. Crit. Care Med. 2001, 29, 1049–1055. [Google Scholar] [CrossRef] [PubMed]
- Tabuchi, A.; Nickles, H.T.; Kim, M.; Semple, J.W.; Koch, E.; Brochard, L.; Slutsky, A.S.; Pries, A.R.; Kuebler, W.M. Acute Lung Injury Causes Asynchronous Alveolar Ventilation That Can Be Corrected by Individual Sighs. Am J. Respir. Crit. Care Med. 2016, 193, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Greene, K.; Wright, J.; Steinberg, K.; Ruzinski, J.; Caldwell, E.; Wong, W.; Hull, W.; Whitsett, J.; Akino, T.; Kuroki, Y.; et al. Serial changes in surfactant-associated proteins in lung and serum before and after onset of ARDS. Am. J. Respir Crit. Care Med. 1999, 160, 1843–1850. [Google Scholar] [CrossRef] [PubMed]
- Gregory, T.J.; Longmore, W.J.; Moxley, M.A.; Whitsett, J.A.; Reed, C.R.; Fowler, A.A.; Hudson, L.D.; Maunder, R.J.; Crim, C.; Hyers, T.M. Surfactant chemical composition and biophysical activity in acute respiratory distress syndrome. J. Clin. Investig. 1991, 88, 1976–1981. [Google Scholar] [CrossRef]
- Schmidt, R.; Markart, P.; Ruppert, C.; Wygrecka, M.; Kuchenbuch, T.; Walmrath, D.; Seeger, W.; Guenther, A. Time-dependent changes in pulmonary surfactant function and composition in acute respiratory distress syndrome due to pneumonia or aspiration. Respir. Res. 2007, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Albert, R.K. The role of ventilation-induced surfactant dysfunction and atelectasis in causing acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 2012, 185, 702–708. [Google Scholar] [CrossRef]
- Nieman, G.F.; Bredenberg, C.E. High surface tension pulmonary edema induced by detergent aerosol. J. Appl. Physiol. 1985, 58, 129–136. [Google Scholar] [CrossRef]
- Lutz, D.; Gazdhar, A.; Lopez-Rodriguez, E.; Ruppert, C.; Mahavadi, P.; Gunther, A.; Klepetko, W.; Bates, J.H.; Smith, B.; Geiser, T.; et al. Alveolar Derecruitment and Collapse Induration as Crucial Mechanisms in Lung Injury and Fibrosis. Am. J. Respir. Cell Mol. Biol. 2015, 52, 232–243. [Google Scholar] [CrossRef]
- Nesslein, L.L.; Melton, K.R.; Ikegami, M.; Na, C.L.; Wert, S.E.; Rice, W.R.; Whitsett, J.A.; Weaver, T.E. Partial SP-B deficiency perturbs lung function and causes air space abnormalities. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 288, L1154–L1161. [Google Scholar] [CrossRef]
- Ikegami, M.; Whitsett, J.A.; Martis, P.C.; Weaver, T.E. Reversibility of lung inflammation caused by SP-B deficiency. Am. J. Physiol. Lung Cell Mol. Physiol. 2005, 289, L962–L970. [Google Scholar] [CrossRef] [PubMed]
- Bilek, A.M.; Dee, K.C.; Gaver, D.P. Mechanisms of surface-tension-induced epithelial cell damage in a model of pulmonary airway reopening. J. Appl. Physiol. (1985) 2003, 94, 770–783. [Google Scholar] [CrossRef] [PubMed]
- Hobi, N.; Ravasio, A.; Haller, T. Interfacial stress affects rat alveolar type II cell signaling and gene expression. Am J. Physiol. Lung Cell Mol. Physiol. 2012, 303, L117–L129. [Google Scholar] [CrossRef] [PubMed]
- Ravasio, A.; Hobi, N.; Bertocchi, C.; Jesacher, A.; Dietl, P.; Haller, T. Interfacial sensing by alveolar type II cells: A new concept in lung physiology? Am. J. Physiol. Cell Physiol. 2011, 300, C1456–C1465. [Google Scholar] [CrossRef] [PubMed]
- Mead, J.; Takishima, T.; Leith, D. Stress distribution in lungs: A model of pulmonary elasticity. J. Appl. Physiol. 1970, 28, 596–608. [Google Scholar] [CrossRef]
- Knudsen, L.; Lopez-Rodriguez, E.; Berndt, L.; Steffen, L.; Ruppert, C.; Bates, J.H.T.; Ochs, M.; Smith, B.J. Alveolar Micromechanics in Bleomycin-induced Lung Injury. Am. J. Respir. Cell Mol. Biol. 2018, 59, 757–769. [Google Scholar] [CrossRef]
- Slutsky, A.S.; Ranieri, V.M. Ventilator-induced lung injury. N. Engl. J. Med. 2013, 369, 2126–2136. [Google Scholar] [CrossRef]
- Glindmeyer, H.W.; Smith, B.J.; Gaver, D.P. In situ enhancement of pulmonary surfactant function using temporary flow reversal. J. Appl. Physiol. 2012, 112, 149–158. [Google Scholar] [CrossRef]
- Ramsingh, R.; Grygorczyk, A.; Solecki, A.; Cherkaoui, L.S.; Berthiaume, Y.; Grygorczyk, R. Cell deformation at the air-liquid interface induces Ca2+-dependent ATP release from lung epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2011, 300, L587–L595. [Google Scholar] [CrossRef][Green Version]
- Wu, Y.; Kharge, A.B.; Perlman, C.E. Lung ventilation injures areas with discrete alveolar flooding, in a surface tension-dependent fashion. J. Appl. Physiol. (1985) 2014, 117, 788–796. [Google Scholar] [CrossRef]
- Hantos, Z.; Daroczy, B.; Suki, B.; Nagy, S.; Fredberg, J.J. Input impedance and peripheral inhomogeneity of dog lungs. J. Appl. Physiol. 1992, 72, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Allen, G.B.; Pavone, L.A.; DiRocco, J.D.; Bates, J.H.; Nieman, G.F. Pulmonary impedance and alveolar instability during injurious ventilation in rats. J. Appl. Physiol. (1985) 2005, 99, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Massa, C.B.; Allen, G.B.; Bates, J.H. Modeling the dynamics of recruitment and derecruitment in mice with acute lung injury. J. Appl. Physiol. 2008, 105, 1813–1821. [Google Scholar] [CrossRef] [PubMed]
- Paré, P.D.; Mitzner, W. Airway-parenchymal interdependence. Compr. Physiol. 2012, 2, 1921–1935. [Google Scholar] [CrossRef] [PubMed]
- Gil, J.; Bachofen, H.; Gehr, P.; Weibel, E. Alveolar volume-surface area relation in air- and saline-filled lungs fixed by vascular perfusion. J. Appl. Physiol. (1985) 1979, 47, 990–1001. [Google Scholar] [CrossRef] [PubMed]
- Gaver, D.P.; Halpern, D.; Jensen, O.E.; Grotberg, J.B. The steady motion of a semi-infinite bubble through a flexible-walled channel. J. Fluid Mech. 1996, 319, 25–65. [Google Scholar] [CrossRef]
- Kay, S.S.; Bilek, A.M.; Dee, K.C.; Gaver, D.P. Pressure gradient, not exposure duration, determines the extent of epithelial cell damage in a model of pulmonary airway reopening. J. Appl. Physiol. 2004, 97, 269–276. [Google Scholar] [CrossRef]
- Nieman, G.F.; Gatto, L.A.; Habashi, N.M. Impact of mechanical ventilation on the pathophysiology of progressive acute lung injury. J. Appl. Physiol. (1985) 2015, 119, 1245–1261. [Google Scholar] [CrossRef]
- Naire, S.; Jensen, O.E. Epithelial cell deformation during surfactant-mediated airway reopening: A theoretical model. J. Appl. Physiol. (1985) 2005, 99, 458–471. [Google Scholar] [CrossRef]
- Matute-Bello, G.; Frevert, C.W.; Martin, T.R. Animal models of acute lung injury. Am. J. Physiol. Lung Cell Mol. Physiol. 2008, 295, L379–L399. [Google Scholar] [CrossRef]
- Roan, E.; Waters, C.M. What do we know about mechanical strain in lung alveoli? Am. J. Physiol. Lung Cell Mol. Physiol. 2011, 301, L625–L635. [Google Scholar] [CrossRef] [PubMed]
- Tschumperlin, D.J.; Margulies, S.S. Alveolar epithelial surface area-volume relationship in isolated rat lungs. J. Appl. Physiol. (1985) 1999, 86, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Bachofen, H.; Schürch, S. Alveolar surface forces and lung architecture. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 129, 183–193. [Google Scholar] [CrossRef]
- Smith, B.J.; Grant, K.A.; Bates, J.H. Linking the development of ventilator-induced injury to mechanical function in the lung. Ann. Biomed. Eng. 2013, 41, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Beike, L.; Wrede, C.; Hegermann, J.; Lopez-Rodriguez, E.; Kloth, C.; Gauldie, J.; Kolb, M.; Maus, U.A.; Ochs, M.; Knudsen, L. Surfactant dysfunction and alveolar collapse are linked with fibrotic septal wall remodeling in the TGF-β1-induced mouse model of pulmonary fibrosis. Lab. Investig. 2019, 99, 830–852. [Google Scholar] [CrossRef]
- Smith, B.J.; Bartolak-Suki, E.; Suki, B.; Roy, G.S.; Hamlington, K.L.; Charlebois, C.M.; Bates, J.H.T. Linking Ventilator Injury-Induced Leak across the Blood-Gas Barrier to Derangements in Murine Lung Function. Front. Physiol. 2017, 8, 466. [Google Scholar] [CrossRef] [PubMed]
- Allen, G.B.; Leclair, T.; Cloutier, M.; Thompson-Figueroa, J.; Bates, J.H. The response to recruitment worsens with progression of lung injury and fibrin accumulation in a mouse model of acid aspiration. Am. J. Physiol. Lung Cell Mol. Physiol. 2007, 292, L1580–L1589. [Google Scholar] [CrossRef]
- Weibel, E. What makes a good lung? Swiss Med. Wkly. 2009, 139, 375–386. [Google Scholar]
- Ghadiali, S.; Huang, Y. Role of airway recruitment and derecruitment in lung injury. Crit. Rev. Biomed. Eng. 2011, 39, 297–317. [Google Scholar] [CrossRef]
- Jansing, N.L.; McClendon, J.; Henson, P.M.; Tuder, R.M.; Hyde, D.M.; Zemans, R.L. Unbiased Quantitation of Alveolar Type II to Alveolar Type I Cell Transdifferentiation during Repair after Lung Injury in Mice. Am. J. Respir. Cell Mol. Biol. 2017, 57, 519–526. [Google Scholar] [CrossRef]
- Hamlington, K.L.; Smith, B.J.; Dunn, C.M.; Charlebois, C.M.; Roy, G.S.; Bates, J.H.T. Linking lung function to structural damage of alveolar epithelium in ventilator-induced lung injury. Respir. Physiol. Neurobiol. 2018, 255, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Fredberg, J.J.; Kamm, R.D. Stress transmission in the lung: Pathways from organ to molecule. Annu. Rev. Physiol. 2006, 68, 507–541. [Google Scholar] [CrossRef] [PubMed]
- Perlman, C.E.; Lederer, D.J.; Bhattacharya, J. Micromechanics of alveolar edema. Am. J. Respir. Cell Mol. Biol. 2011, 44, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Schirrmann, K.; Mertens, M.; Kertzscher, U.; Kuebler, W.M.; Affeld, K. Theoretical modeling of the interaction between alveoli during inflation and deflation in normal and diseased lungs. J. Biomech. 2010, 43, 1202–1207. [Google Scholar] [CrossRef] [PubMed]
- Loring, S.H.; Topulos, G.P.; Hubmayr, R.D. Transpulmonary Pressure: The Importance of Precise Definitions and Limiting Assumptions. Am. J. Respir. Crit. Care Med. 2016, 194, 1452–1457. [Google Scholar] [CrossRef]
- Albert, R.K.; Smith, B.; Perlman, C.E.; Schwartz, D.A. Is Progression of Pulmonary Fibrosis due to Ventilation-induced Lung Injury? Am. J. Respir. Crit. Care Med. 2019, 200, 140–151. [Google Scholar] [CrossRef]
- Hamlington, K.L.; Bates, J.H.T.; Roy, G.S.; Julianelle, A.J.; Charlebois, C.; Suki, B.; Smith, B.J. Alveolar leak develops by a rich-get-richer process in ventilator-induced lung injury. PLoS ONE 2018, 13, e0193934. [Google Scholar] [CrossRef]
- Scherle, W. A simple method for volumetry of organs in quantitative stereology. Mikroskopie 1970, 26, 57–60. [Google Scholar]
- Tschanz, S.; Schneider, J.P.; Knudsen, L. Design-based stereology: Planning, volumetry and sampling are crucial steps for a successful study. Ann. Anat. 2014, 196, 3–11. [Google Scholar] [CrossRef]
- Ochs, M.; Mühlfeld, C. Quantitative microscopy of the lung: A problem-based approach. Part 1: Basic principles of lung stereology. Am. J. Physiol. Lung Cell Mol. Physiol. 2013, 305, L15–L22. [Google Scholar] [CrossRef]
- Muhlfeld, C.; Knudsen, L.; Ochs, M. Stereology and morphometry of lung tissue. Methods in Mol. Biol. (Clifton, N.J.) 2013, 931, 367–390. [Google Scholar] [CrossRef]
- Hsia, C.; Hyde, D.; Ochs, M.; Weibel, E. An official research policy statement of the American Thoracic Society/European Respiratory Society: Standards for quantitative assessment of lung structure. Am. J. Respir. Crit. Care Med. 2010, 181, 394–418. [Google Scholar] [CrossRef] [PubMed]
- Weibel, E.; Hsia, C.; Ochs, M. How much is there really? Why stereology is essential in lung morphometry. J. Appl. Physiol. 2007, 102, 459–467. [Google Scholar] [CrossRef]
- Gundersen, H.; Jensen, E. The efficiency of systematic sampling in stereology and its prediction. J. Microsc. 1987, 147, 229–263. [Google Scholar] [CrossRef] [PubMed]
- Ochs, M. A brief update on lung stereology. J. Microsc. 2006, 222, 188–200. [Google Scholar] [CrossRef] [PubMed]
- Braendgaard, H.; Gundersen, H.J. The impact of recent stereological advances on quantitative studies of the nervous system. J. Neurosci. Methods 1986, 18, 39–78. [Google Scholar] [CrossRef]
- Ochs, M.; Nyengaard, L.R.; Lung, A.; Knudsen, L.; Voigt, M.; Wahlers, T.; Richter, J.; Gundersen, H.J.G. The number of alveoli in the human lung. Am. J. Respir. Crit. Care Med. 2004, 169, 120–124. [Google Scholar] [CrossRef]
- Gundersen, H.; Bagger, P.; Bendtsen, T.; Evans, S.; Korbo, L.; Marcussen, N.; Møller, A.; Nielsen, K.; Nyengaard, J.; Pakkenberg, B. The new stereological tools: Disector, fractionator, nucleator and point sampled intercepts and their use in pathological research and diagnosis. APMIS 1988, 96, 857–881. [Google Scholar] [CrossRef]
- Hyde, D.; Tyler, N.; Putney, L.; Singh, P.; Gundersen, H. Total number and mean size of alveoli in mammalian lung estimated using fractionator sampling and unbiased estimates of the Euler characteristic of alveolar openings. Anat. Rec. A Discov. Mol. Cell Evol. Biol. 2004, 277, 216–226. [Google Scholar] [CrossRef]
- Tschanz, S.A.; Burri, P.H.; Weibel, E.R. A simple tool for stereological assessment of digital images: The STEPanizer. J. Microsc. 2011, 243, 47–59. [Google Scholar] [CrossRef]
- Fehrenbach, H.; Schepelmann, D.; Albes, J.; Bando, T.; Fischer, F.; Fehrenbach, A.; Stolte, N.; Wahlers, T.; Richter, J. Pulmonary ischemia/reperfusion injury: A quantitative study of structure and function in isolated heart-lungs of the rat. Anat. Rec. 1999, 255, 84–89. [Google Scholar] [CrossRef]
- Steffen, L.; Ruppert, C.; Hoymann, H.G.; Funke, M.; Ebener, S.; Kloth, C.; Mühlfeld, C.; Ochs, M.; Knudsen, L.; Lopez-Rodriguez, E. Surfactant replacement therapy reduces acute lung injury and collapse induration-related lung remodeling in the bleomycin model. Am. J. Physiol. Lung Cell Mol. Physiol. 2017, 313, L313–L327. [Google Scholar] [CrossRef] [PubMed]
| Group | Dox on | Dox off d1 | Dox off d3 | 2wayANOVA | ||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Pao 2 | Pao 10 | group | Pao |
| V(lung) cm3 | 0.52 (0.05) | 1.18 (0.10) | 0.42 (0.08) | 0.95 (0.10) | 0.40 (0.05) | 0.80 (0.14) | < 0.01 | < 0.01 |
| V(alvair,lung) cm3 | 0.27 (0.02) | 0.72 (0.07) | 0.19 (0.05) | 0.53 (0.08) | 0.16 (0.03) | 0.43 (0.08) | < 0.001 | < 0.001 |
| V(ductair,lung) cm3 | 0.05 (0.02) | 0.19 (0.05) | 0.04 (0.02) | 0.16 (0.03) | 0.04 (0.01) | 0.14 (0.02) | n.s. | < 0.01 |
| V(sep,lung) cm3 | 0.13 (0.02) | 0.12 (0.02) | 0.12 (0.01) | 0.13 (0.03) | 0.13 (0.01) | 0.12 (0.02) | n.s. | n.s. |
| S(alv,lung) cm2 | 268 (83.0) | 374 (110) | 272 (55.0) | 395 (50.0) | 205 (26.7) | 254 (72.6) | 0.008 | 0.001 |
| τ (sep) µm | 10.3 (2.91) | 6.77 (1.04) | 9.37 (1.24) | 6.53 (1.70) | 13.2 (1.19) | 10.2 (2.91) | < 0.001 | < 0.001 |
| N(alv,lung) 106 | 1.75 (0.81) | 2.61 (1.01) | 2.31 (0.63) | 2.80 (0.39) | 1.47 (0.21) | 1.47 (0.52) | 0.005 | n.s. |
| Dox on | Dox off d1 | Dox off d3 | 2wayANOVA | |||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Group | Pao |
| V(AE1,sep) mm3 | 15.2 (2.01) | 15.4 (3.19) | 11.6 (2.00) | 16.5 (3.14) | 14.4 (0.97) | 13.0 (1.86) | n.s. | n.s. |
| V(AE2,sep) mm3 | 4.92 (1.36) | 7.73 (3.01) | 6.24 (1.66) | 6.78 (2.20) | 5.96 (2.58) | 4.67 (2.00) | n.s. | n.s. |
| V(endo,sep) mm3 | 20.8 (1.58) | 20.7 (2.94) | 18.7 3.42 | 21.3 (4.97) | 20.2 (1.68) | 20.1 (2.89) | n.s. | n.s. |
| V(othercells,sep) mm3 | 8.04 (1.95) | 9.02 (2.07) | 8.01 (1.38) | 12.6 (5.49) | 9.05 (0.8) | 9.12 (1.4) | n.s. | n.s. |
| V(ECM,sep) mm3 | 3.78 (0.95) | 4.15 (1.65) | 3.60 (0.73) | 6.42 (2.99) | 3.52 (0.79) | 4.69 (2.30) | n.s. | 0.024 |
| V(caplumen,sep) mm3 | 73.7 (16.3) | 63.7 (15.2) | 75.3 (8.89) | 57.5 (12.5) | 73.04 (9.34) | 63.05 (10.8) | n.s. | 0.009 |
| S(endoBL,sep) cm2 | 775 (138) | 915 (188) | 700 (81.0) | 804 (175) | 689 (76.3) | 739 (83.5) | n.s. | 0.049 |
| Dox on | Dox off d1 | Dox off d3 | 2wayANOVA | |||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Group | Pao |
| V(alvfluid,par) mm3 | 0.79 (0.32) | 1.39 (0.95) | 0.92 (0.26) | 6.64 (11.4) | 8.03 (3.95) | 6.63 (3.95) | 0.032 | n.s. |
| S(airAE,par) cm2 | 401 (103) | 717 (112) | 320 (76.2) | 628 (190) | 200 (60.0) | 310 (89.2) | < 0.001 | < 0.001 |
| S(fluidAE,par) cm2 | 353 (58.7) | 236 (55.3) | 366 (29.4) | 280 (113) | 483 (39.3) | 393 (101) | < 0.001 | < 0.001 |
| τ(alvfluid) nm | 39.0 (18.6) | 89.0 (48.0) | 44.2 (16.6) | 248 (294) | 297 (155) | 268 (125) | 0.006 | n.s. |
| Dox on | Dox off d1 | Dox off d3 | 2wayANOVA | |||||
|---|---|---|---|---|---|---|---|---|
| Parameter | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Pao 2 | Pao 10 | Group | Pao |
| S(alvBL,sep) cm2 | 775 (129) | 954 (181) | 686 (82.4) | 879 (206) | 660 (76.3) | 727 (105) | 0.034 | 0.006 |
| SS(AEinjure/alvBL)% | 0.97 (0.76) | 0.86 (0.55) | 2.78 (0.99) | 4.03 (1.39) | 6.83 (2.54) | 8.94 (4.12) | < 0.001 | n.s. |
| SS(AE1/alvBL)% | 93.3 (1.27) | 94.0 (2.15) | 90.1 (2.10) | 89.4 (2.30) | 88.0 (3.44) | 86.4 (3.45) | < 0.001 | n.s. |
| SS(AE2/alvBL)% | 5.16 (1.31) | 5.09 (2.37) | 7.15 (2.21) | 6.59 (1.70) | 5.21 (1.18) | 4.67 (1.73) | 0.033 | n.s. |
| S(AEinjure,sep) cm2 | 7.31 (5.48) | 7.87 (5.13) | 19.0 (4.07) | 34.4 (14.1) | 44.1 (14.4) | 67.7 (37.6) | < 0.001 | 0.025 |
| S(AE1,sep) cm2 | 728 (126) | 900 (184) | 617 (62.6) | 786 (188) | 582 (84.9) | 626 (78.2) | 0.004 | 0.011 |
| S(AE2,sep) cm2 | 39.5 (9.99) | 46.4 (22.7) | 50.1 (20.1) | 58.4 (20.1) | 33.7 (4.97) | 33.5 (12.0) | 0.03 | n.s. |
| Definition | Abbreviation | Test System | Magnification |
|---|---|---|---|
| Volume of parenchyma | V(par,lung) | Point counting (P) | 5 × |
| Volume of non-parenchyma (conducting airways, larger vessels, connective tissue) | V(nonpar,lung) | Point counting (P) | 5 × |
| Volume of alveolar airspaces within lung parenchyma | V(alvair,lung) | Point counting(P) | 20 × |
| Volume of ductal airspaces within lung parenchyma | V(ductair,lung) | Point counting (P) | 20 × |
| Volume of interalveolar septa within lung parenchyma | V (sep,lung) | Point counting (P) | 20 × |
| Surface area of alveoli within lung parenchyma | S (alv,lung) | Intersection counting (I) | 20 × |
| Arithmetic mean septal wall thickness | τ(sep) | Volume-to-surface ratio | 20 × |
| Number alveoli within lung parenchyma | N(alv,lung) | Physical disector | 10 × |
| Volume of alveolar epithelial type 1 cells within septal walls | V(AE1,sep) | Point counting (P) | 11.000 × |
| Volume of alveolar epithelial type 2 cells within septal walls | V(AE2,sep) | Point counting (P) | 11.000 × |
| Volume of endothelial cell within septal walls | V(endo,sep) | Point counting (P) | 11.000 × |
| Volume of other cells (e.g., macrophages, fibroblasts) | V(othercells,sep) | Point counting (P) | 11.000 × |
| Volume of extracellular matrix within septal walls | V(ECM,sep) | Point counting (P) | 11.000 × |
| Volume of capillary lumen within septal walls | V(cap,sep) | Point counting (P) | 11.000 × |
| Volume of alveolar fluid within parenchyma | V(alvfluid,par) | Point counting (P) | 11.000 × |
| Surface area of the alveolar epithelial basal lamina within septal walls | S(alvBL,sep) | Intersection counting (I) | 11.000 × |
| Surface area of the basal lamina covered by AE1 cells | S(AE1,sep) | Intersection counting (I) | 11.000 × |
| Surface area of the basal lamina covered by AE2 cells | S(AE2,sep) | Intersection counting (I) | 11.000 × |
| Surface area of the basal lamina covered by injured alveolar epithelial cells | S(AEinjure,sep) | Intersection counting (I) | 11.000 × |
| Surface area of the endothelial basal lamina within septal walls | S(endoBL,sep) | Intersection (I) counting | 11.000 × |
| Surface area of the alveolar epithelium covered by air within parenchyma | S(airAE,par) | Intersection (I) counting | 11.000 × |
| Surface area of alveolar epithelium covered by alveolar fluid within parenchyma | S(fluidAE,par) | Intersection (I) counting | 11.000 × |
| Arithmetic mean thickness of alveolar fluid | τ(alvfluid) | Volume-to-surface ratio | 11.000 × |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rühl, N.; Lopez-Rodriguez, E.; Albert, K.; Smith, B.J.; Weaver, T.E.; Ochs, M.; Knudsen, L. Surfactant Protein B Deficiency Induced High Surface Tension: Relationship between Alveolar Micromechanics, Alveolar Fluid Properties and Alveolar Epithelial Cell Injury. Int. J. Mol. Sci. 2019, 20, 4243. https://doi.org/10.3390/ijms20174243
Rühl N, Lopez-Rodriguez E, Albert K, Smith BJ, Weaver TE, Ochs M, Knudsen L. Surfactant Protein B Deficiency Induced High Surface Tension: Relationship between Alveolar Micromechanics, Alveolar Fluid Properties and Alveolar Epithelial Cell Injury. International Journal of Molecular Sciences. 2019; 20(17):4243. https://doi.org/10.3390/ijms20174243
Chicago/Turabian StyleRühl, Nina, Elena Lopez-Rodriguez, Karolin Albert, Bradford J Smith, Timothy E Weaver, Matthias Ochs, and Lars Knudsen. 2019. "Surfactant Protein B Deficiency Induced High Surface Tension: Relationship between Alveolar Micromechanics, Alveolar Fluid Properties and Alveolar Epithelial Cell Injury" International Journal of Molecular Sciences 20, no. 17: 4243. https://doi.org/10.3390/ijms20174243
APA StyleRühl, N., Lopez-Rodriguez, E., Albert, K., Smith, B. J., Weaver, T. E., Ochs, M., & Knudsen, L. (2019). Surfactant Protein B Deficiency Induced High Surface Tension: Relationship between Alveolar Micromechanics, Alveolar Fluid Properties and Alveolar Epithelial Cell Injury. International Journal of Molecular Sciences, 20(17), 4243. https://doi.org/10.3390/ijms20174243





