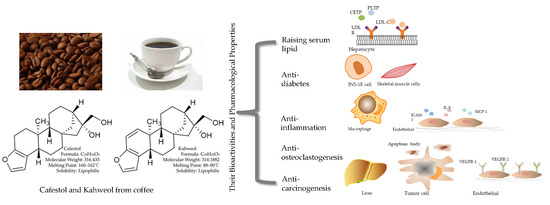
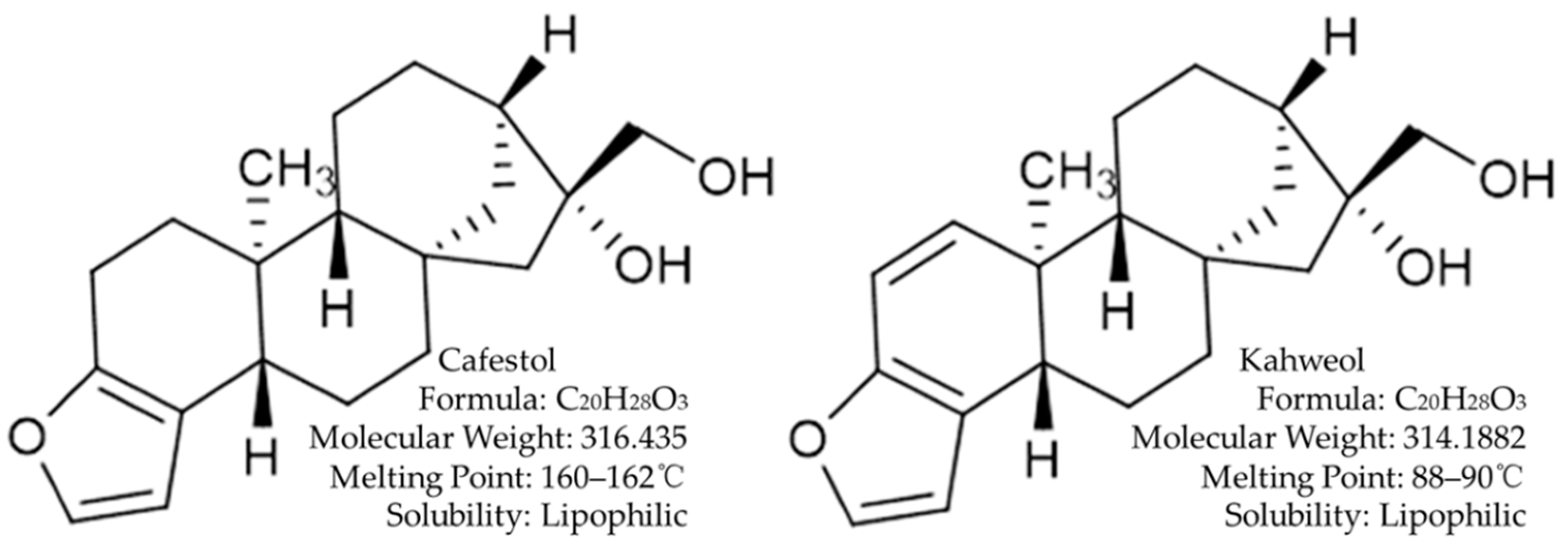
Cafestol and Kahweol: A Review on Their Bioactivities and Pharmacological Properties
Abstract
1. Introduction
2. Biological and Pharmacological Activities of Cafestol and Kahweol
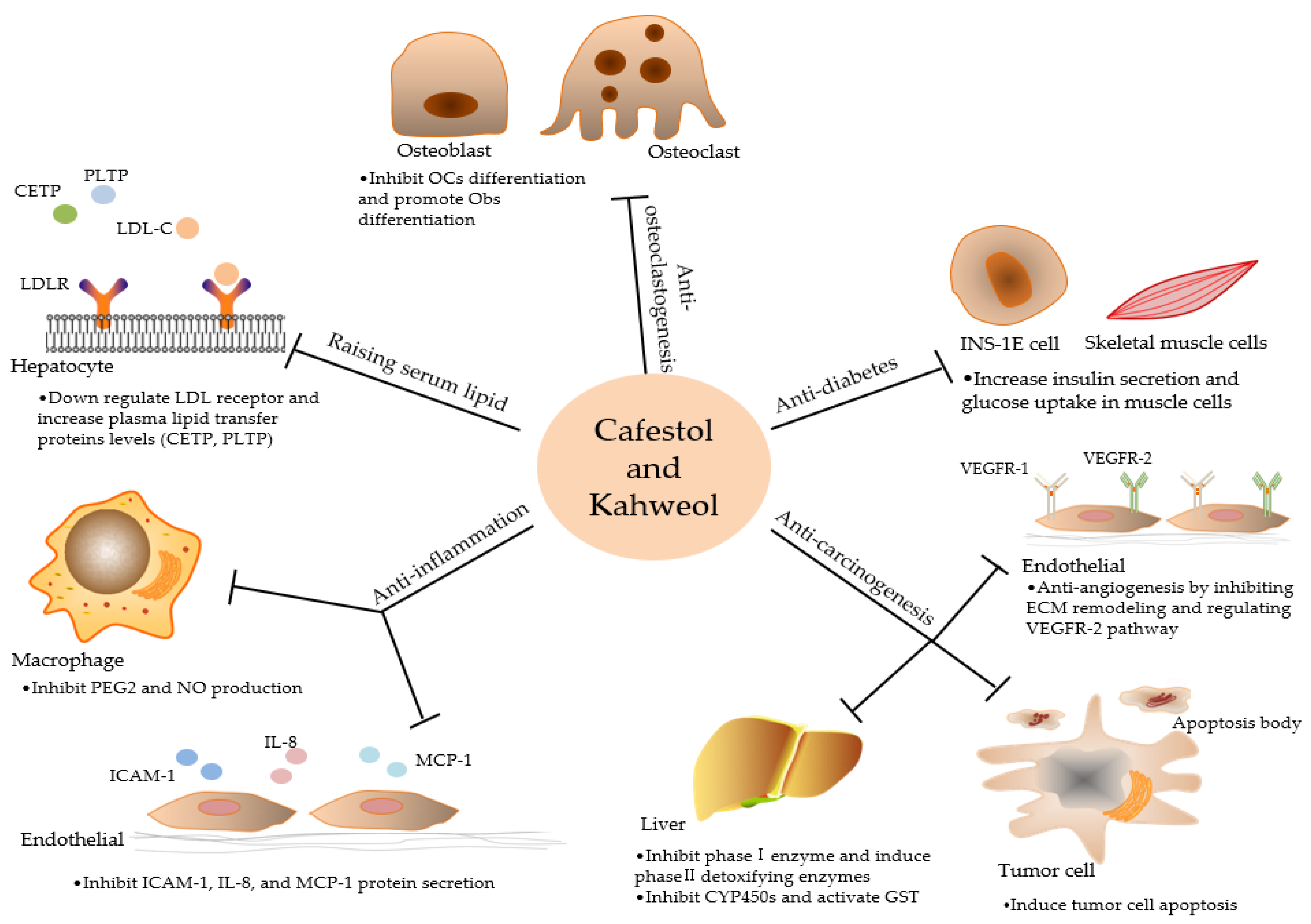
2.1. Raising Serum Lipid
2.2. Anti-inflammation
2.3. Anti- carcinogenesis
2.3.1. Inhibition for Early Mutagenic Event
2.3.2. Inducing Apoptosis
2.3.3. Anti-Angiogenesis
2.4. Potential Anti-Diabetic Capabilities
2.5. Anti-osteoclastogenesis
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| LDL | Low-density lipoprotein |
| GSH | Glutathione |
| FXR | Farnesoid X receptor |
| PXR | Pregnane X receptor |
| CETP | Cholesterolester transfer protein |
| PLTP | Phospholipid transfer protein |
| IL | Interleukin |
| TNF-α | Tumor Necrosis Factor-alpha |
| NO | Tumor Necrosis Factor-alpha |
| iNOS | Inducible nitric oxide synthase |
| PGE2 | Prostaglandin E2 |
| LPS | Lipopolysaccharide |
| COX-2 | Cyclooxygenase-2 |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| IKB | Inhibitor of nuclear factor kappa-B |
| IKK | Inhibitor of nuclear factor kappa-B kinase |
| NIK | NF-κB-inducible kinase |
| Akt | Protein kinase B |
| MAPK | Mitogen-activated protein kinase |
| AP-1 | Activator protein 1 |
| ERK | Extracellular regulated protein kinases |
| JNK | c-Jun N-terminal kinase |
| STAT | Signal transducers and activators of transcription |
| JAK | Janus kinase |
| MCP-1 | Monocyte chemotactic protein 1 |
| ICAM-1 | Intercellular cell adhesion molecule-1 |
| HUVECs | Human umbilical vein endothelial cells |
| HO-1 | Hemeoxygenase-1 |
| Sirt1 | Sirtuin 1 |
| ROS | Reactive oxygen species |
| Nrf2 | Nuclear factor erythroid-2-related factor 2 |
| CYP450s | Cytochrome P450 |
| Keap1 | Kelch-like ECH-associated protein-1 |
| ARE | Antioxidant response element |
| Bcl-2 | B-cell lymphoma-2 |
| mcl1 | Myeloid cell leukemia-1 |
| cFLIP | Cellular FLICE-inhibitory Protein |
| PI3K | Phosphatidylinositol 3′-kinase |
| Sp1 | Specificity protein 1 |
| ATF3 | Activating transcription factor 3 |
| MMP-2 | Matrix metalloproteinase-2 |
| GSK3β | Glycogen synthase kinase 3 beta |
| BTF3 | Basic transcription factor 3 |
| EC | Endothelial cell |
| ECM | Extracellular matrix |
| uPA | Urokinase type plasminogen activator |
| VEGF | Vascular endothelial growth factor |
| FAK | Focal adhesion kinase |
| CP | Cafestol palmitate |
| KP | Kahweol palmitate |
| T2D | Type-2-diabetes |
| PPAR | Peroxisome proliferators-activated receptor |
| C/EBPα | CCAAT/enhancer binding protein alpha |
| FABP 4 | Fatty acid-binding protein 4 |
| FASN | Fatty acid synthase |
| AMPK | Adenosine 5‘-monophosphate-activated protein kinase |
| PHIP | 2-Amino-1-methyl-6-phenylimidazo [4,5-bpyridine] |
| SULT | Sulfotransferase |
| OCs | Osteoclasts |
| OBs | Osteoblasts |
| NFATc1 | Nuclear factor of activated T cells cytoplasmic-1 |
| RANKL | Receptor activator of nuclear factor kappa-B ligand |
References
- Poole, R.; Kennedy, O.J.; Roderick, P.; Fallowfield, J.A.; Hayes, P.C.; Parkes, J. Coffee consumption and health: Umbrella review of meta-analyses of multiple health outcomes. BMJ (Clin. Res. Ed.) 2017, 359, j5024. [Google Scholar] [CrossRef]
- Ding, M.; Bhupathiraju, S.N.; Satija, A.; van Dam, R.M.; Hu, F.B. Long-term coffee consumption and risk of cardiovascular disease: A systematic review and a dose-response meta-analysis of prospective cohort studies. Circulation 2014, 129, 643–659. [Google Scholar] [CrossRef]
- Wang, A.; Wang, S.; Zhu, C.; Huang, H.; Wu, L.; Wan, X.; Yang, X.; Zhang, H.; Miao, R.; He, L.; et al. Coffee and cancer risk: A meta-analysis of prospective observational studies. Sci. Rep. 2016, 6, 33711. [Google Scholar] [CrossRef]
- Shang, F.; Li, X.; Jiang, X. Coffee consumption and risk of the metabolic syndrome: A meta-analysis. Diabetes Metab. 2016, 42, 80–87. [Google Scholar] [CrossRef]
- Bravi, F.; Tavani, A.; Bosetti, C.; Boffetta, P.; La Vecchia, C. Coffee and the risk of hepatocellular carcinoma and chronic liver disease: A systematic review and meta-analysis of prospective studies. Eur. J. Cancer Prev. 2017, 26, 368–377. [Google Scholar] [CrossRef]
- Ludwig, I.A.; Clifford, M.N.; Lean, M.E.; Ashihara, H.; Crozier, A. Coffee: Biochemistry and potential impact on health. Food Funct. 2014, 5, 1695–1717. [Google Scholar] [CrossRef]
- Godos, J.; Pluchinotta, F.R.; Marventano, S.; Buscemi, S.; Li Volti, G.; Galvano, F.; Grosso, G. Coffee components and cardiovascular risk: Beneficial and detrimental effects. Int. J. Food Sci. Nutr. 2014, 65, 925–936. [Google Scholar] [CrossRef]
- Van Cruchten, S.T.; de Haan, L.H.; Mulder, P.P.; Kunne, C.; Boekschoten, M.V.; Katan, M.B.; Aarts, J.M.; Witkamp, R.F. The role of epoxidation and electrophile-responsive element-regulated gene transcription in the potentially beneficial and harmful effects of the coffee components cafestol and kahweol. J. Nutr. Biochem. 2010, 21, 757–763. [Google Scholar] [CrossRef]
- De Roos, B.; Caslake, M.J.; Stalenhoef, A.F.; Bedford, D.; Demacker, P.N.; Katan, M.B.; Packard, C.J. The coffee diterpene cafestol increases plasma triacylglycerol by increasing the production rate of large VLDL apolipoprotein B in healthy normolipidemic subjects. Am. J. Clin. Nutr. 2001, 73, 45–52. [Google Scholar] [CrossRef][Green Version]
- Halvorsen, B.; Ranheim, T.; Nenseter, M.S.; Huggett, A.C.; Drevon, C.A. Effect of a coffee lipid (cafestol) on cholesterol metabolism in human skin fibroblasts. J. Lipid Res. 1998, 39, 901–912. [Google Scholar]
- Urgert, R.; Kosmeijer-Schuil, T.G.; Katan, M.B. Intake levels, sites of action and excretion routes of the cholesterol-elevating diterpenes from coffee beans in humans. Biochem. Soc. Trans. 1996, 24, 800–806. [Google Scholar] [CrossRef]
- De Roos, B.; Meyboom, S.; Kosmeijer-Schuil, T.G.; Katan, M.B. Absorption and urinary excretion of the coffee diterpenes cafestol and kahweol in healthy ileostomy volunteers. J. Intern. Med. 1998, 244, 451–460. [Google Scholar] [CrossRef]
- Cruchten, S.T.; de Waart, D.R.; Kunne, C.; Hooiveld, G.J.; Boekschoten, M.V.; Katan, M.B.; Elferink, R.P.; Witkamp, R.F. Absorption, distribution, and biliary excretion of cafestol, a potent cholesterol-elevating compound in unfiltered coffees, in mice. Drug Metab. Dispos. 2010, 38, 635–640. [Google Scholar] [CrossRef]
- De Roos, B.; Van Tol, A.; Urgert, R.; Scheek, L.M.; Van Gent, T.; Buytenhek, R.; Princen, H.M.; Katan, M.B. Consumption of French-press coffee raises cholesteryl ester transfer protein activity levels before LDL cholesterol in normolipidaemic subjects. J. Intern. Med. 2000, 248, 211–216. [Google Scholar] [CrossRef]
- Thelle, D.S. Coffee, tea and coronary heart disease. Curr. Opin. Lipidol. 1995, 6, 25–27. [Google Scholar] [CrossRef]
- Urgert, R.; Weusten-van der Wouw, M.P.; Hovenier, R.; Meyboom, S.; Beynen, A.C.; Katan, M.B. Diterpenes from coffee beans decrease serum levels of lipoprotein(a) in humans: Results from four randomised controlled trials. Eur. J. Clin. Nutr. 1997, 51, 431–436. [Google Scholar] [CrossRef]
- Urgert, R.; Katan, M.B. The cholesterol-raising factor from coffee beans. Annu. Rev. Nutr. 1997, 17, 305–324. [Google Scholar] [CrossRef]
- Rustan, A.C.; Halvorsen, B.; Huggett, A.C.; Ranheim, T.; Drevon, C.A. Effect of coffee lipids (cafestol and kahweol) on regulation of cholesterol metabolism in HepG2 cells. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 2140–2149. [Google Scholar] [CrossRef]
- Post, S.M.; de Roos, B.; Vermeulen, M.; Afman, L.; Jong, M.C.; Dahlmans, V.E.; Havekes, L.M.; Stellaard, F.; Katan, M.B.; Princen, H.M. Cafestol increases serum cholesterol levels in apolipoprotein E*3-Leiden transgenic mice by suppression of bile acid synthesis. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1551–1556. [Google Scholar] [CrossRef]
- Ricketts, M.L.; Boekschoten, M.V.; Kreeft, A.J.; Hooiveld, G.J.; Moen, C.J.; Muller, M.; Frants, R.R.; Kasanmoentalib, S.; Post, S.M.; Princen, H.M.; et al. The cholesterol-raising factor from coffee beans, cafestol, as an agonist ligand for the farnesoid and pregnane X receptors. Mol. Endocrinol. 2007, 21, 1603–1616. [Google Scholar] [CrossRef]
- Post, S.M.; de Wit, E.C.; Princen, H.M. Cafestol, the cholesterol-raising factor in boiled coffee, suppresses bile acid synthesis by downregulation of cholesterol 7 alpha-hydroxylase and sterol 27-hydroxylase in rat hepatocytes. Arterioscler. Thromb. Vasc. Biol. 1997, 17, 3064–3070. [Google Scholar] [CrossRef]
- Grundy, S.M. Cholesterol-raising effects of coffee: Clues to regulation of cholesterol metabolism. J. Intern. Med. 1995, 238, 475–477. [Google Scholar] [CrossRef]
- van Tol, A.; Urgert, R.; de Jong-Caesar, R.; van Gent, T.; Scheek, L.M.; de Roos, B.; Katan, M.B. The cholesterol-raising diterpenes from coffee beans increase serum lipid transfer protein activity levels in humans. Atherosclerosis 1997, 132, 251–254. [Google Scholar] [CrossRef]
- Urgert, R.; Katan, M.B. The cholesterol-raising factor from coffee beans. J. R. Soc. Med. 1996, 89, 618–623. [Google Scholar] [CrossRef]
- Kim, J.Y.; Jung, K.S.; Jeong, H.G. Suppressive effects of the kahweol and cafestol on cyclooxygenase-2 expression in macrophages. FEBS Lett. 2004, 569, 321–326. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kim, D.H.; Jeong, H.G. Inhibitory effect of the coffee diterpene kahweol on carrageenan-induced inflammation in rats. BioFactors (Oxf. Engl.) 2006, 26, 17–28. [Google Scholar] [CrossRef]
- Shen, C.P.; Luo, J.G.; Yang, M.H.; Kong, L.Y. Cafestol-Type Diterpenoids from the Twigs of Tricalysia fruticosa with Potential Anti-inflammatory Activity. J. Nat. Prod. 2015, 78, 1322–1329. [Google Scholar] [CrossRef]
- Chen, F.; Demers, L.M.; Shi, X. Upstream signal transduction of NF-kappaB activation. Curr. Drug Targets 2002, 1, 137–149. [Google Scholar] [CrossRef]
- Kim, J.Y.; Jung, K.S.; Lee, K.J.; Na, H.K.; Chun, H.K.; Kho, Y.H.; Jeong, H.G. The coffee diterpene kahweol suppress the inducible nitric oxide synthase expression in macrophages. Cancer Lett. 2004, 213, 147–154. [Google Scholar] [CrossRef]
- De Lucia, M.; Panzella, L.; Melck, D.; Giudicianni, I.; Motta, A.; Napolitano, A.; d’Ischia, M. Differential reactivity of purified bioactive coffee furans, cafestol and kahweol, with acidic nitrite: Product characterization and factors controlling nitrosation versus ring-opening pathways. Chem. Res. Toxicol. 2009, 22, 1922–1928. [Google Scholar] [CrossRef]
- Shen, T.; Lee, J.; Lee, E.; Kim, S.H.; Kim, T.W.; Cho, J.Y. Cafestol, a coffee-specific diterpene, is a novel extracellular signal-regulated kinase inhibitor with AP-1-targeted inhibition of prostaglandin E2 production in lipopolysaccharide-activated macrophages. Biol. Pharm. Bull. 2010, 33, 128–132. [Google Scholar] [CrossRef]
- Shen, T.; Park, Y.C.; Kim, S.H.; Lee, J.; Cho, J.Y. Nuclear factor-kappaB/signal transducers and activators of transcription-1-mediated inflammatory responses in lipopolysaccharide-activated macrophages are a major inhibitory target of kahweol, a coffee diterpene. Biol. Pharm. Bull. 2010, 33, 1159–1164. [Google Scholar] [CrossRef]
- Villarino, A.V.; Kanno, Y.; O’Shea, J.J. Mechanisms and consequences of Jak-STAT signaling in the immune system. Nat. Immunol. 2017, 18, 374–384. [Google Scholar] [CrossRef]
- Cheng, J.J.; Wung, B.S.; Chao, Y.J.; Wang, D.L. Cyclic strain-induced reactive oxygen species involved in ICAM-1 gene induction in endothelial cells. Hypertension 1998, 31, 125–130. [Google Scholar] [CrossRef]
- Wung, B.S.; Cheng, J.J.; Hsieh, H.J.; Shyy, Y.J.; Wang, D.L. Cyclic strain-induced monocyte chemotactic protein-1 gene expression in endothelial cells involves reactive oxygen species activation of activator protein 1. Circ. Res. 1997, 81, 1–7. [Google Scholar] [CrossRef]
- Hao, W.R.; Sung, L.C.; Chen, C.C.; Chen, P.Y. Cafestol Inhibits Cyclic-Strain-Induced Interleukin-8, Intercellular Adhesion Molecule-1, and Monocyte Chemoattractant Protein-1 Production in Vascular Endothelial Cells. Oxidative Med. Cell. Longev. 2018, 2018, 7861518. [Google Scholar] [CrossRef]
- Moolgavkar, S.H. The multistage theory of carcinogenesis and the age distribution of cancer in man. J. Natl. Cancer Inst. 1978, 61, 49–52. [Google Scholar] [CrossRef]
- Turesky, R.J.; Lang, N.P.; Butler, M.A.; Teitel, C.H.; Kadlubar, F.F. Metabolic activation of carcinogenic heterocyclic aromatic amines by human liver and colon. Carcinogenesis 1991, 12, 1839–1845. [Google Scholar] [CrossRef]
- Mori, Y.; Koide, A.; Kobayashi, Y.; Morimura, K.; Kaneko, M.; Fukushima, S. Effect of ethanol treatment on metabolic activation and detoxification of esophagus carcinogenic N-nitrosamines in rat liver. Mutagenesis 2002, 17, 251–256. [Google Scholar] [CrossRef]
- Huber, W.W.; Rossmanith, W.; Grusch, M.; Haslinger, E.; Prustomersky, S.; Peter-Vorosmarty, B.; Parzefall, W.; Scharf, G.; Schulte-Hermann, R. Effects of coffee and its chemopreventive components kahweol and cafestol on cytochrome P450 and sulfotransferase in rat liver. Food Chem. Toxicol. 2008, 46, 1230–1238. [Google Scholar] [CrossRef]
- Lee, K.J.; Choi, J.H.; Jeong, H.G. Hepatoprotective and antioxidant effects of the coffee diterpenes kahweol and cafestol on carbon tetrachloride-induced liver damage in mice. Food Chem. Toxicol. 2007, 45, 2118–2125. [Google Scholar] [CrossRef]
- Cavin, C.; Holzhaeuser, D.; Scharf, G.; Constable, A.; Huber, W.W.; Schilter, B. Cafestol and kahweol, two coffee specific diterpenes with anticarcinogenic activity. Food Chem. Toxicol. 2002, 40, 1155–1163. [Google Scholar] [CrossRef]
- Cavin, C.; Holzhauser, D.; Constable, A.; Huggett, A.C.; Schilter, B. The coffee-specific diterpenes cafestol and kahweol protect against aflatoxin B1-induced genotoxicity through a dual mechanism. Carcinogenesis 1998, 19, 1369–1375. [Google Scholar] [CrossRef]
- Higgins, L.G.; Cavin, C.; Itoh, K.; Yamamoto, M.; Hayes, J.D. Induction of cancer chemopreventive enzymes by coffee is mediated by transcription factor Nrf2. Evidence that the coffee-specific diterpenes cafestol and kahweol confer protection against acrolein. Toxicol. Appl. Pharmacol. 2008, 226, 328–337. [Google Scholar] [CrossRef]
- Tao, K.S.; Wang, W.; Wang, L.; Cao, D.Y.; Li, Y.Q.; Wu, S.X.; Dou, K.F. The multifaceted mechanisms for coffee’s anti-tumorigenic effect on liver. Med Hypotheses 2008, 71, 730–736. [Google Scholar] [CrossRef]
- Huber, W.W.; Teitel, C.H.; Coles, B.F.; King, R.S.; Wiese, F.W.; Kaderlik, K.R.; Casciano, D.A.; Shaddock, J.G.; Mulder, G.J.; Ilett, K.F.; et al. Potential chemoprotective effects of the coffee components kahweol and cafestol palmitates via modification of hepatic N-acetyltransferase and glutathione S-transferase activities. Environ. Mol. Mutagen. 2004, 44, 265–276. [Google Scholar] [CrossRef]
- Huber, W.W.; Scharf, G.; Nagel, G.; Prustomersky, S.; Schulte-Hermann, R.; Kaina, B. Coffee and its chemopreventive components Kahweol and Cafestol increase the activity of O6-methylguanine-DNA methyltransferase in rat liver--comparison with phase II xenobiotic metabolism. Mutat. Res. 2003, 522, 57–68. [Google Scholar] [CrossRef]
- Huber, W.W.; Scharf, G.; Rossmanith, W.; Prustomersky, S.; Grasl-Kraupp, B.; Peter, B.; Turesky, R.J.; Schulte-Hermann, R. The coffee components kahweol and cafestol induce gamma-glutamylcysteine synthetase, the rate limiting enzyme of chemoprotective glutathione synthesis, in several organs of the rat. Arch. Toxicol. 2002, 75, 685–694. [Google Scholar] [CrossRef]
- Huber, W.W.; Prustomersky, S.; Delbanco, E.; Uhl, M.; Scharf, G.; Turesky, R.J.; Thier, R.; Schulte-Hermann, R. Enhancement of the chemoprotective enzymes glucuronosyl transferase and glutathione transferase in specific organs of the rat by the coffee components kahweol and cafestol. Arch. Toxicol. 2002, 76, 209–217. [Google Scholar] [CrossRef]
- Lam, L.K.; Sparnins, V.L.; Wattenberg, L.W. Effects of derivatives of kahweol and cafestol on the activity of glutathione S-transferase in mice. J. Med. Chem. 1987, 30, 1399–1403. [Google Scholar] [CrossRef]
- Muriel, P.; Arauz, J. Coffee and liver diseases. Fitoterapia 2010, 81, 297–305. [Google Scholar] [CrossRef]
- Lima, C.S.; Spindola, D.G.; Bechara, A.; Garcia, D.M.; Palmeira-Dos-Santos, C.; Peixoto-da-Silva, J.; Erustes, A.G.; Michelin, L.F.G.; Pereira, G.J.S.; Smaili, S.S.; et al. Cafestol, a diterpene molecule found in coffee, induces leukemia cell death. Biomed. Pharmacother. 2017, 92, 1045–1054. [Google Scholar] [CrossRef]
- Kotowski, U.; Heiduschka, G.; Seemann, R.; Eckl-Dorna, J.; Schmid, R.; Kranebitter, V.; Stanisz, I.; Brunner, M.; Lill, C.; Thurnher, D. Effect of the coffee ingredient cafestol on head and neck squamous cell carcinoma cell lines. Strahlenther. Und Onkol. 2015, 191, 511–517. [Google Scholar] [CrossRef]
- Woo, S.M.; Min, K.J.; Seo, B.R.; Nam, J.O.; Choi, K.S.; Yoo, Y.H.; Kwon, T.K. Cafestol overcomes ABT-737 resistance in Mcl-1-overexpressed renal carcinoma Caki cells through downregulation of Mcl-1 expression and upregulation of Bim expression. Cell Death Dis. 2014, 5, e1514. [Google Scholar] [CrossRef]
- Lee, K.A.; Chae, J.I.; Shim, J.H. Natural diterpenes from coffee, cafestol and kahweol induce apoptosis through regulation of specificity protein 1 expression in human malignant pleural mesothelioma. J. Biomed. Sci. 2012, 19, 60. [Google Scholar] [CrossRef]
- Choi, M.J.; Park, E.J.; Oh, J.H.; Min, K.J.; Yang, E.S.; Kim, Y.H.; Lee, T.J.; Kim, S.H.; Choi, Y.H.; Park, J.W.; et al. Cafestol, a coffee-specific diterpene, induces apoptosis in renal carcinoma Caki cells through down-regulation of anti-apoptotic proteins and Akt phosphorylation. Chem. Biol. Interact. 2011, 190, 102–108. [Google Scholar] [CrossRef]
- Black, A.R.; Black, J.D.; Azizkhan-Clifford, J. Sp1 and kruppel-like factor family of transcription factors in cell growth regulation and cancer. J. Cell. Physiol. 2001, 188, 143–160. [Google Scholar] [CrossRef]
- Um, H.J.; Oh, J.H.; Kim, Y.N.; Choi, Y.H.; Kim, S.H.; Park, J.W.; Kwon, T.K. The coffee diterpene kahweol sensitizes TRAIL-induced apoptosis in renal carcinoma Caki cells through down-regulation of Bcl-2 and c-FLIP. Chem. -Biol. Interact. 2010, 186, 36–42. [Google Scholar] [CrossRef]
- Cardenas, C.; Quesada, A.R.; Medina, M.A. Insights on the antitumor effects of kahweol on human breast cancer: Decreased survival and increased production of reactive oxygen species and cytotoxicity. Biochem. Biophys. Res. Commun. 2014, 447, 452–458. [Google Scholar] [CrossRef]
- Choi, D.W.; Lim, M.S.; Lee, J.W.; Chun, W.; Lee, S.H.; Nam, Y.H.; Park, J.M.; Choi, D.H.; Kang, C.D.; Lee, S.J.; et al. The Cytotoxicity of Kahweol in HT-29 Human Colorectal Cancer Cells Is Mediated by Apoptosis and Suppression of Heat Shock Protein 70 Expression. Biomol. Ther. 2015, 23, 128–133. [Google Scholar] [CrossRef]
- Oh, S.H.; Hwang, Y.P.; Choi, J.H.; Jin, S.W.; Lee, G.H.; Han, E.H.; Chung, Y.H.; Chung, Y.C.; Jeong, H.G. Kahweol inhibits proliferation and induces apoptosis by suppressing fatty acid synthase in HER2-overexpressing cancer cells. Food Chem. Toxicol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Chae, J.I.; Jeon, Y.J.; Shim, J.H. Anti-proliferative properties of kahweol in oral squamous cancer through the regulation specificity protein 1. Phytother. Res. 2014, 28, 1879–1886. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.H.; Lee, J.T.; Yang, E.S.; Chang, J.S.; Lee, D.S.; Kim, S.H.; Choi, Y.H.; Park, J.W.; Kwon, T.K. The coffee diterpene kahweol induces apoptosis in human leukemia U937 cells through down-regulation of Akt phosphorylation and activation of JNK. Apoptosis 2009, 14, 1378–1386. [Google Scholar] [CrossRef] [PubMed]
- Park, G.H.; Song, H.M.; Jeong, J.B. The coffee diterpene kahweol suppresses the cell proliferation by inducing cyclin D1 proteasomal degradation via ERK1/2, JNK and GKS3beta-dependent threonine-286 phosphorylation in human colorectal cancer cells. Food Chem. Toxicol. 2016, 95, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.G.; Hwang, Y.P.; Jeong, H.G. Kahweol blocks STAT3 phosphorylation and induces apoptosis in human lung adenocarcinoma A549 cells. Toxicol. Lett. 2009, 187, 28–34. [Google Scholar] [CrossRef]
- Yan, C.; Lu, D.; Hai, T.; Boyd, D.D. Activating transcription factor 3, a stress sensor, activates p53 by blocking its ubiquitination. EMBO J. 2005, 24, 2425–2435. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Wolfgang, C.D.; Hai, T. Activating transcription factor 3, a stress-inducible gene, suppresses Ras-stimulated tumorigenesis. J. Biol. Chem. 2006, 281, 10473–10481. [Google Scholar] [CrossRef]
- Chen, H.H.; Wang, D.L. Nitric oxide inhibits matrix metalloproteinase-2 expression via the induction of activating transcription factor 3 in endothelial cells. Mol. Pharmacol. 2004, 65, 1130–1140. [Google Scholar] [CrossRef]
- Park, G.H.; Song, H.M.; Jeong, J.B. Kahweol from Coffee Induces Apoptosis by Upregulating Activating Transcription Factor 3 in Human Colorectal Cancer Cells. Biomol. Ther. 2017, 25, 337–343. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Bang, W.; Cho, J.H.; Lee, R.H.; Kim, S.H.; Kim, M.S.; Park, S.M.; Shin, J.C.; Chung, H.J.; Oh, K.B.; et al. Kahweol induces apoptosis by suppressing BTF3 expression through the ERK signaling pathway in non-small cell lung cancer cells. Int. J. Oncol. 2016, 49, 2294–2302. [Google Scholar] [CrossRef]
- Min, K.J.; Um, H.J.; Kim, J.I.; Kwon, T.K. The coffee diterpene kahweol enhances sensitivity to sorafenib in human renal carcinoma Caki cells through down-regulation of Mcl-1 and c-FLIP expression. Oncotarget 2017, 8, 83195–83206. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J. Tumor angiogenesis: Therapeutic implications. New Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [PubMed]
- Okubo, Y.; Motohashi, O.; Nakayama, N.; Nishimura, K.; Kasajima, R.; Miyagi, Y.; Shiozawa, M.; Yoshioka, E.; Suzuki, M.; Washimi, K.; et al. The clinicopathological significance of angiogenesis in hindgut neuroendocrine tumors obtained via an endoscopic procedure. Diagn. Pathol. 2016, 11, 128. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, C.; Quesada, A.R.; Medina, M.A. Anti-angiogenic and anti-inflammatory properties of kahweol, a coffee diterpene. PLoS ONE 2011, 6, e23407. [Google Scholar] [CrossRef]
- Stetler-Stevenson, W.G. Matrix metalloproteinases in angiogenesis: A moving target for therapeutic intervention. J. Clin. Investig. 1999, 103, 1237–1241. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Gerber, H.P.; LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 2003, 9, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Wu, H.J.; Guan, J.L. Nuclear FAK and its kinase activity regulate VEGFR2 transcription in angiogenesis of adult mice. Sci. Rep. 2018, 8, 2550. [Google Scholar] [CrossRef]
- Wang, S.; Yoon, Y.C.; Sung, M.J.; Hur, H.J.; Park, J.H. Antiangiogenic properties of cafestol, a coffee diterpene, in human umbilical vein endothelial cells. Biochem. Biophys. Res. Commun. 2012, 421, 567–571. [Google Scholar] [CrossRef]
- Moeenfard, M.; Cortez, A.; Machado, V.; Costa, R.; Luis, C.; Coelho, P.; Soares, R.; Alves, A.; Borges, N.; Santos, A. Anti-Angiogenic Properties of Cafestol and Kahweol Palmitate Diterpene Esters. J. Cell. Biochem. 2016, 117, 2748–2756. [Google Scholar] [CrossRef]
- Albertin, G.; Guidolin, D.; Sorato, E.; Oselladore, B.; Tortorella, C.; Ribatti, D. Urotensin-II-stimulated expression of pro-angiogenic factors in human vascular endothelial cells. Regul. Pept. 2011, 172, 16–22. [Google Scholar] [CrossRef]
- Lee, C.Y.; Tsai, Y.T.; Loh, S.H.; Liu, J.C.; Chen, T.H.; Chao, H.H.; Cheng, T.H.; Chen, J.J. Urotensin II induces interleukin 8 expression in human umbilical vein endothelial cells. PLoS ONE 2014, 9, e90278. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.T.; Sung, L.C.; Haw, W.R.; Chen, C.C.; Huang, S.F.; Liu, J.C.; Cheng, T.H.; Chen, P.Y.; Loh, S.H.; Tsai, C.S. Cafestol, a coffee diterpene, inhibits urotensin II-induced interleukin-8 expression in human umbilical vein endothelial cells. Eur. J. Pharmacol. 2018, 820, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Mellbye, F.B.; Jeppesen, P.B.; Hermansen, K.; Gregersen, S. Cafestol, a Bioactive Substance in Coffee, Stimulates Insulin Secretion and Increases Glucose Uptake in Muscle Cells: Studies in Vitro. J. Nat. Prod. 2015, 78, 2447–2451. [Google Scholar] [CrossRef] [PubMed]
- Mellbye, F.B.; Jeppesen, P.B.; Shokouh, P.; Laustsen, C.; Hermansen, K.; Gregersen, S. Cafestol, a Bioactive Substance in Coffee, Has Antidiabetic Properties in KKAy Mice. J. Nat. Prod. 2017, 80, 2353–2359. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Kim, N.J.; Song, J.K.; Chun, K.H. Kahweol inhibits lipid accumulation and induces Glucose-uptake through activation of AMP-activated protein kinase (AMPK). BMB Rep. 2017, 50, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Lee, S.G.; Kang, Y.J.; Kwon, T.K.; Nam, J.O. Kahweol inhibits adipogenesis of 3T3-L1 adipocytes through downregulation of PPARgamma. Nat. Prod. Res. 2018, 32, 1216–1219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.B.; Zhou, G.; Li, C. AMPK: An emerging drug target for diabetes and the metabolic syndrome. Cell Metab. 2009, 9, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, K.; Neutzsky-Wulff, A.V.; Bonewald, L.F.; Karsdal, M.A. Local communication on and within bone controls bone remodeling. Bone 2009, 44, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, Z.; Duan, N.; Zhu, G.; Schwarz, E.M.; Xie, C. Osteoblast-osteoclast interactions. Connect. Tissue Res. 2018, 59, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Fumimoto, R.; Sakai, E.; Yamaguchi, Y.; Sakamoto, H.; Fukuma, Y.; Nishishita, K.; Okamoto, K.; Tsukuba, T. The coffee diterpene kahweol prevents osteoclastogenesis via impairment of NFATc1 expression and blocking of Erk phosphorylation. J. Pharmacol. Sci. 2012, 118, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Fukuma, Y.; Sakai, E.; Nishishita, K.; Okamoto, K.; Tsukuba, T. Cafestol has a weaker inhibitory effect on osteoclastogenesis than kahweol and promotes osteoblast differentiation. BioFactors (Oxf. Engl.) 2015, 41, 222–231. [Google Scholar] [CrossRef] [PubMed]
| Activities | Mechanism | Comparison of efficiency |
|---|---|---|
| Raising serum lipid | •Down regulate LDL receptor and increase plasma lipid transfer proteins levels (CETP, PLTP) | Cafestol is far stronger than kahweol |
| Anti-inflammation | •Inhibit the expression of iNOS and COX-2 and the secretion of pro-inflammatory cytokines | Kahweol might be more effective in antioxidant activity |
| •Inhibit phase I enzyme and induce phase II detoxifying enzymes: Nrf2/ARE | ||
| Anti-carcinogenesis | •Induce apoptosis by regulating Bcl-2 family proteins and cyclins •Anti-angiogenesis by inhibiting ECM remodeling and regulating VEGFR-2 pathway | Kahweol exhibited stronger anti-angiogenic properties than cafestol in some studies |
| Anti-diabetes | •Increase insulin secretion and glucose uptake in muscle cells •Inhibit adipogenesis | Not mentioned |
| Anti-osteoclastogenesis | •Inhibit differentiation and bone resorbing activity of OCs •Promote OBs differentiation | Kahweol stronger in inhibiting osteoclastogenesis than cafestol |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, Y.; Wang, C.; Xu, J.; Wang, S. Cafestol and Kahweol: A Review on Their Bioactivities and Pharmacological Properties. Int. J. Mol. Sci. 2019, 20, 4238. https://doi.org/10.3390/ijms20174238
Ren Y, Wang C, Xu J, Wang S. Cafestol and Kahweol: A Review on Their Bioactivities and Pharmacological Properties. International Journal of Molecular Sciences. 2019; 20(17):4238. https://doi.org/10.3390/ijms20174238
Chicago/Turabian StyleRen, Yaqi, Chunlan Wang, Jiakun Xu, and Shuaiyu Wang. 2019. "Cafestol and Kahweol: A Review on Their Bioactivities and Pharmacological Properties" International Journal of Molecular Sciences 20, no. 17: 4238. https://doi.org/10.3390/ijms20174238
APA StyleRen, Y., Wang, C., Xu, J., & Wang, S. (2019). Cafestol and Kahweol: A Review on Their Bioactivities and Pharmacological Properties. International Journal of Molecular Sciences, 20(17), 4238. https://doi.org/10.3390/ijms20174238