Machine Learning to Understand the Immune-Inflammatory Pathways in Fibromyalgia
Abstract
1. Introduction
2. Results
2.1. Socio-Demographic and Clinical Data in Patients and Controls
2.2. Associations Between Cytokines and Diagnosis
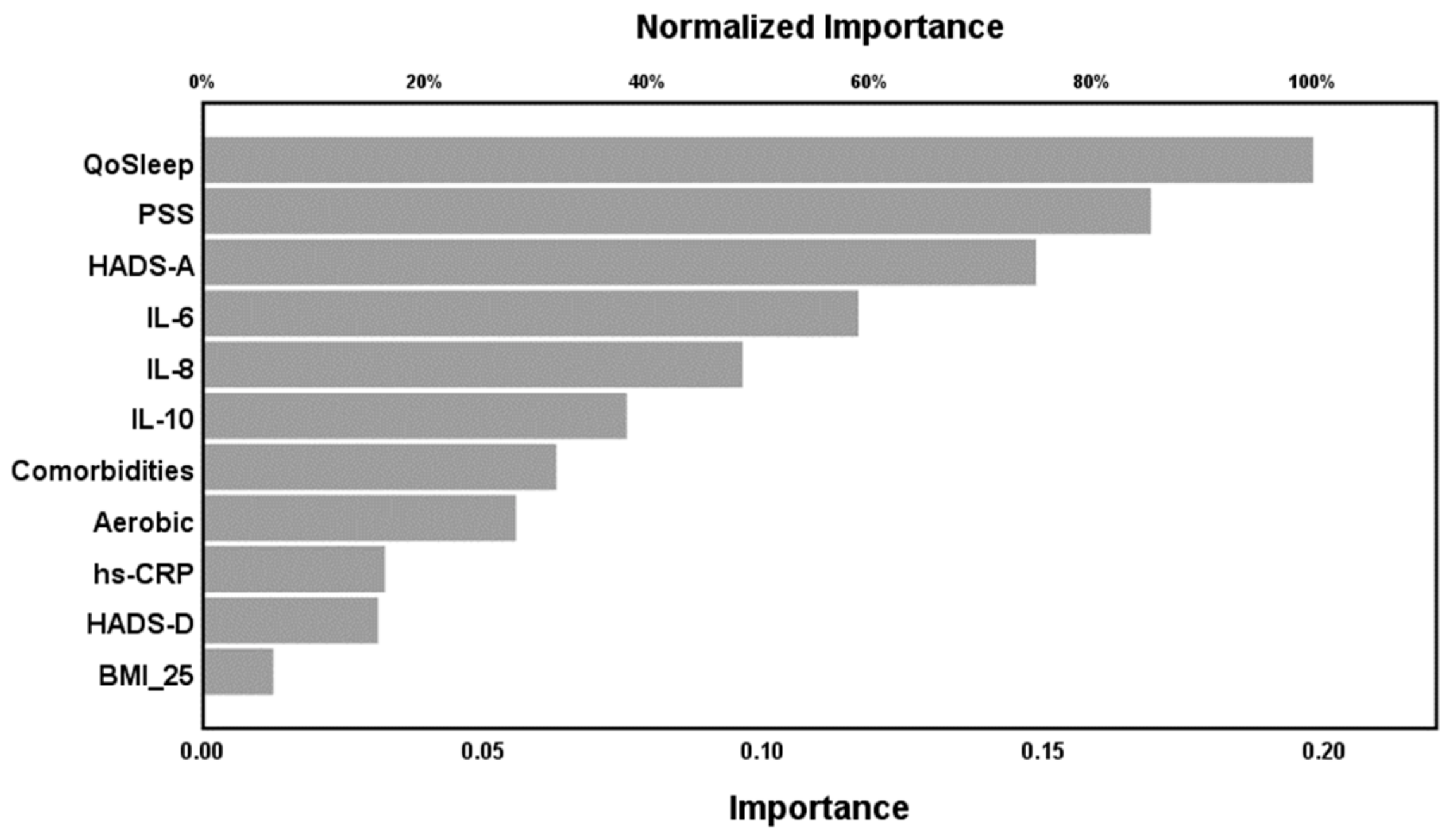
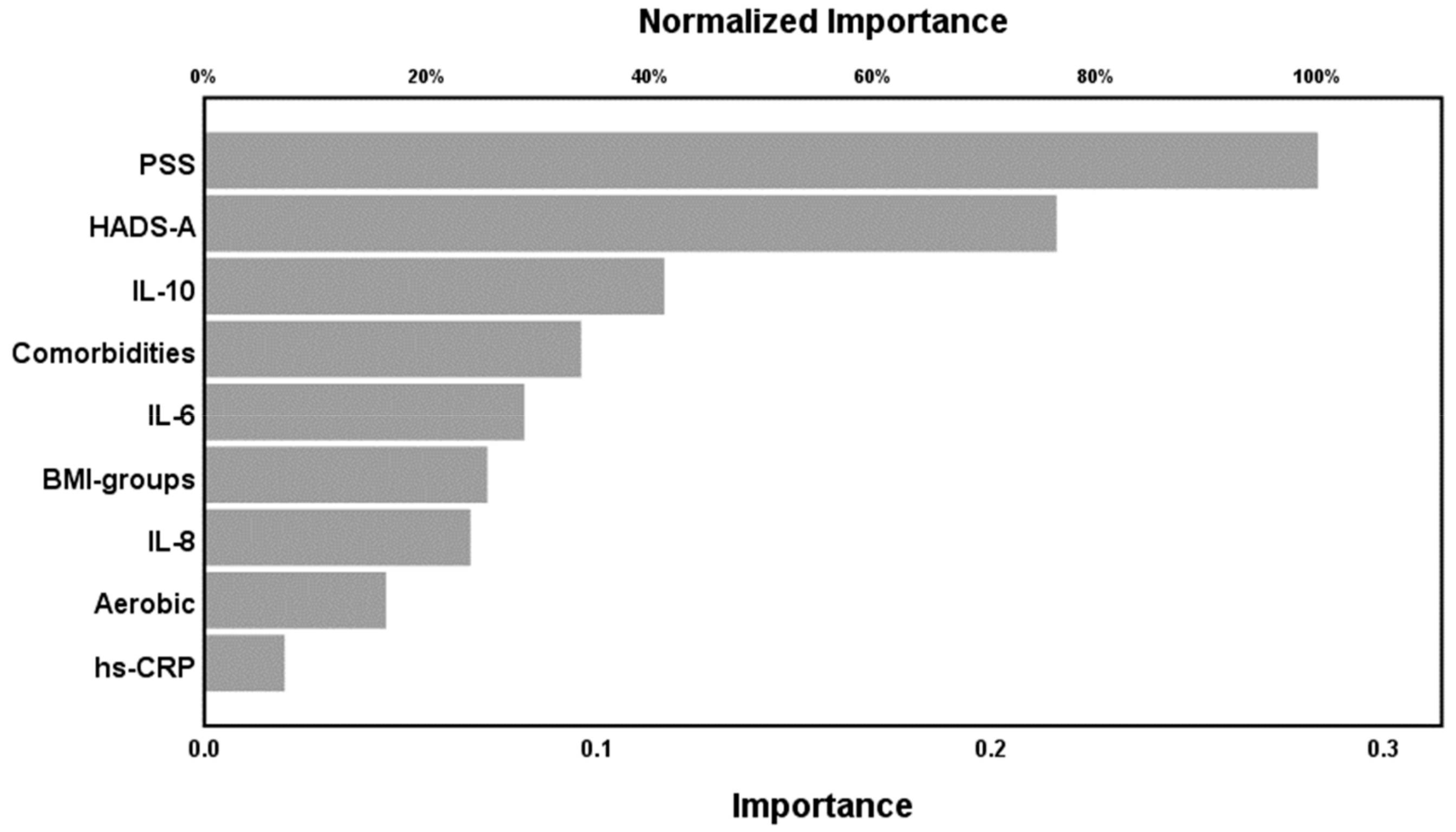
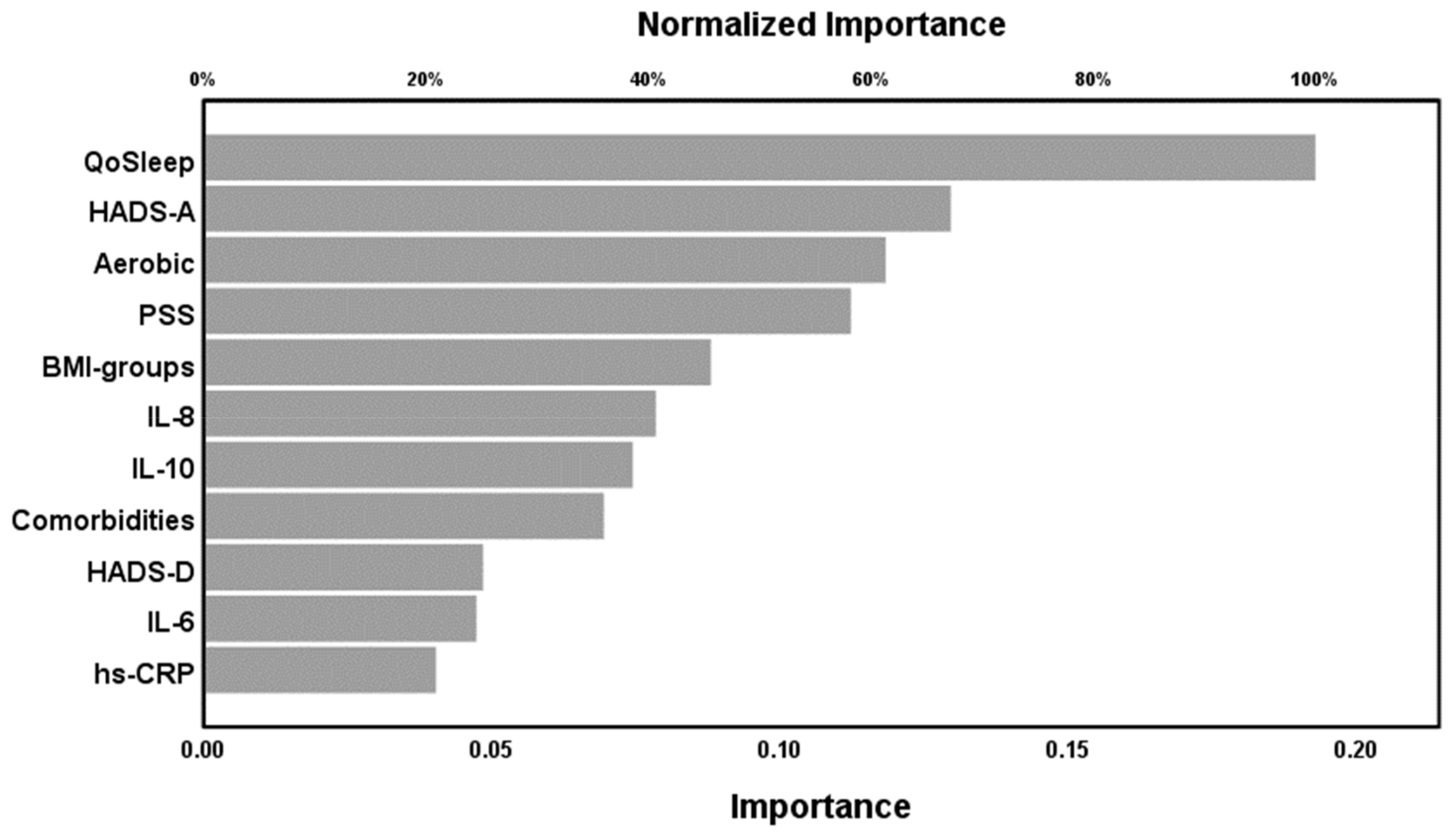
2.3. Prediction of FM
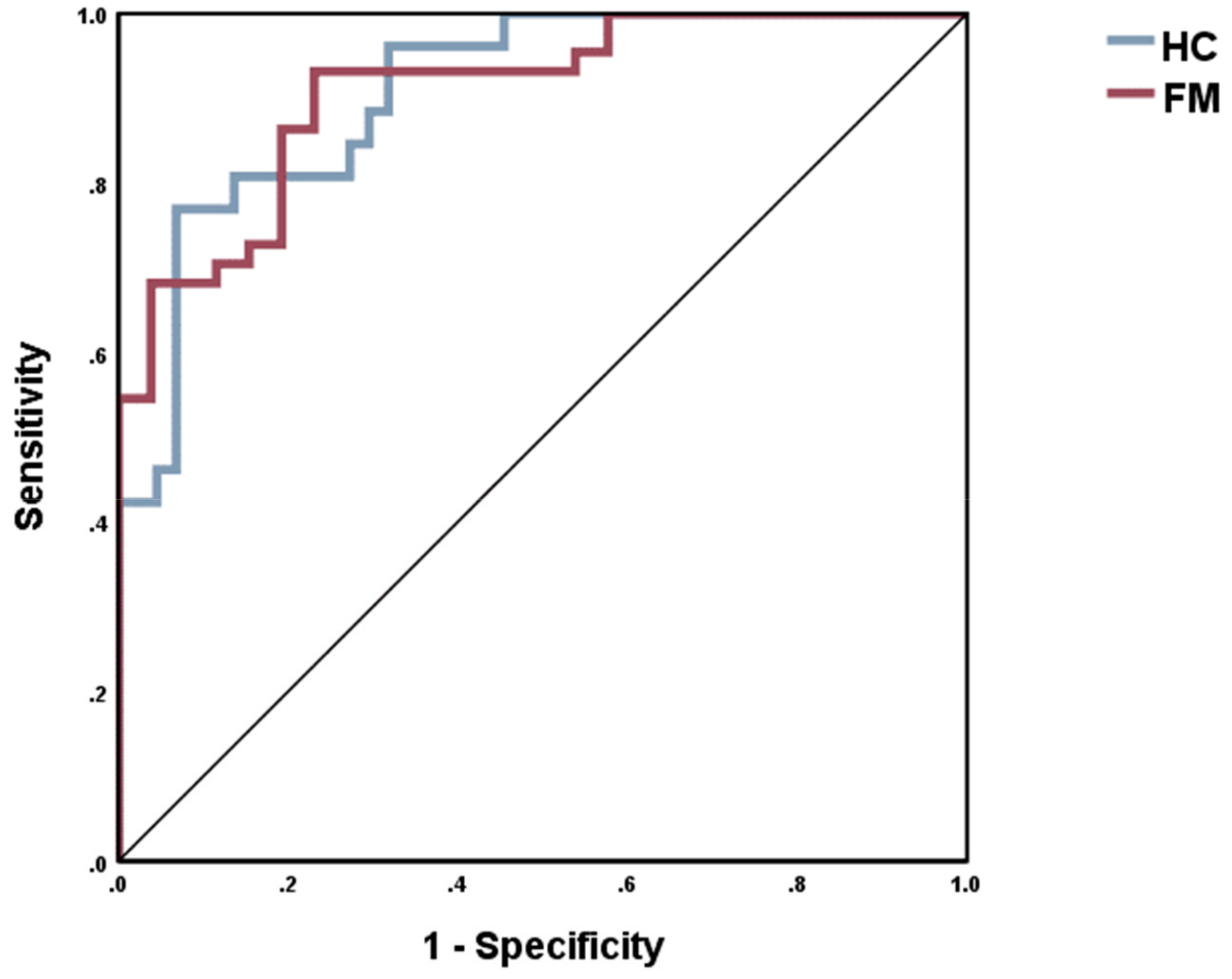
2.4. Results of NN
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Clinical Measurements
4.3. Blood Assays
4.4. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| FM | Fibromyalgia |
| BMI | Body Mass Index |
| IL | Interleukin |
| hs-CRP | High Sensitivity C-Reactive Protein |
| WPI | Widespread Pain Index |
| SSS | Symptom Severity Scale |
| HADS | Hospital Anxiety and Depression Scale |
| PSS-10 | Perceived Stress Scale |
| IRS | Immune-inflammatory response system |
| ADs | Antidepressants |
| NN | Neural Networks |
| ACR | American College of Rheumatology |
| FS | Fibromyalgia Symptoms Scale |
| FIQ-R | Fibromyalgia Impact Questionnaire |
| SDs | Standard deviations |
| GLM | Multivariate general linear model |
| FDR | False discovery rate |
| CI | Confidence Intervals |
| ROC | Receiving operating curve |
| LIF | Leukemia inhibitor factor |
References
- Häuser, W.; Ablin, J.; Fitzcharles, M.-A.; Littlejohn, G.; Luciano, J.V.; Usui, C.; Walitt, B.; Wolfe, F.; Wolfe, F.; Walitt, B.; et al. Fibromyalgia. Nat. Rev. Dis. Prim. 2015, 1, 15022. [Google Scholar] [CrossRef] [PubMed]
- Sluka, K.; Clauw, D. Neurobiology of fibromyalgia and chronic widespread pain. Neuroscience 2016, 338, 114–129. [Google Scholar] [CrossRef] [PubMed]
- Ranzolin, A.; Duarte, A.L.B.P.; Bredemeier, M.; da Costa Neto, C.A.; Ascoli, B.M.; Wollenhaupt-Aguiar, B.; Kapczinski, F.; Xavier, R.M. Evaluation of cytokines, oxidative stress markers and brain-derived neurotrophic factor in patients with fibromyalgia—A controlled cross-sectional study. Cytokine 2016, 84, 25–28. [Google Scholar] [CrossRef] [PubMed]
- Bazzichi, L.; Rossi, A.; Massimetti, G.; Giannaccini, G.; Giuliano, T.; De Feo, F.; Ciapparelli, A.; Dell’Osso, L.; Bombardieri, S. Cytokine patterns in fibromyalgia and their correlation with clinical manifestations. Clin. Exp. Rheumatol. 2007, 25, 225–230. [Google Scholar] [PubMed]
- Bäckryd, E.; Lind, A.-L.; Thulin, M.; Larsson, A.; Gerdle, B.; Gordh, T. High levels of cerebrospinal fluid chemokines point to the presence of neuroinflammation in peripheral neuropathic pain. Pain 2017, 158, 2487–2495. [Google Scholar] [CrossRef]
- Malhotra, D.; Saxena, A.K.; Dar, S.A.; Kumar, V.; Nasare, N.; Tripathi, A.K.; Banerjee, B.D. Evaluation of cytokine levels in fibromyalgia syndrome patients and its relationship to the severity of chronic pain. J. Musculoskelet. Pain 2012, 20, 164–169. [Google Scholar] [CrossRef]
- Xiao, Y.; Russell, I.J.; Liu, Y.-G. A brain-derived neurotrophic factor polymorphism Val66Met identifies fibromyalgia syndrome subgroup with higher body mass index and C-reactive protein. Rheumatol. Int. 2012, 32, 2479–2485. [Google Scholar] [CrossRef]
- Maes, M.; Libbrecht, I.; Van Hunsel, F.; Lin, A.H.; De Clerck, L.; Stevens, W.; Kenis, G.; de Jongh, R.; Bosmans, E.; Neels, H. The immune-inflammatory pathophysiology of fibromyalgia: Increased serum soluble gp130, the common signal transducer protein of various neurotrophic cytokines. Psychoneuroendocrinology 1999, 24, 371–383. [Google Scholar] [CrossRef]
- Maes, M.; Nowak, G.; Caso, J.R.; Leza, J.C.; Song, C.; Kubera, M.; Klein, H.; Galecki, P.; Noto, C.; Glaab, E.; et al. Toward Omics-Based, Systems Biomedicine, and Path and Drug Discovery Methodologies for Depression-Inflammation Research. Mol. Neurobiol. 2016, 2927–2935. [Google Scholar] [CrossRef]
- Üçeyler, N.; Häuser, W.; Sommer, C. Systematic review with meta-analysis: Cytokines in fibromyalgia syndrome. BMC Musculoskelet. Disord. 2011, 12, 245. [Google Scholar] [CrossRef]
- Rodriguez-Pinto, I.; Agmon-Levin, N.; Howard, A.; Shoenfeld, Y. Fibromyalgia and cytokines. Immunol. Lett. 2014, 161, 200–203. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Glaser, R. Methodological issues in behavioral immunology research with humans. Brain. Behav. Immun. 1988, 2, 67–78. [Google Scholar] [CrossRef]
- Krueger, J.M.; Rector, D.M.; Churchill, L. Sleep and Cytokines. Sleep Med. Clin. 2007, 2, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Kheirandish-Gozal, L.; Gozal, D. Obstructive Sleep Apnea and Inflammation: Proof of Concept Based on Two Illustrative Cytokines. Int. J. Mol. Sci. 2019, 20, 459. [Google Scholar] [CrossRef] [PubMed]
- Bystrom, J.; Clanchy, F.I.; Taher, T.E.; Mangat, P.; Jawad, A.S.; Williams, R.O.; Mageed, R.A. TNFα in the regulation of Treg and Th17 cells in rheumatoid arthritis and other autoimmune inflammatory diseases. Cytokine 2018, 101, 4–13. [Google Scholar] [CrossRef] [PubMed]
- Cataldi, C.; Mari, N.L.; Lozovoy, M.A.B.; Martins, L.M.M.; Reiche, E.M.V.; Maes, M.; Dichi, I.; Simão, A.N.C. Proinflammatory and anti-inflammatory cytokine profiles in psoriasis: Use as laboratory biomarkers and disease predictors. Inflamm. Res. 2019, 68, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Vogelzangs, N.; de Jonge, P.; Smit, J.H.; Bahn, S.; Penninx, B.W. Cytokine production capacity in depression and anxiety. Transl. Psychiatry 2016, 6, e825. [Google Scholar] [CrossRef]
- Köhler, C.A.; Freitas, T.H.; Maes, M.; de Andrade, N.Q.; Liu, C.S.; Fernandes, B.S.; Stubbs, B.; Solmi, M.; Veronese, N.; Herrmann, N.; et al. Peripheral cytokine and chemokine alterations in depression: A meta-analysis of 82 studies. Acta Psychiatr. Scand. 2017, 135, 373–387. [Google Scholar] [CrossRef]
- Szałach, Ł.P.; Lisowska, K.A.; Cubała, W.J. The Influence of Antidepressants on the Immune System. Arch. Immunol. Ther. Exp. (Warsz) 2019, 67, 143–151. [Google Scholar] [CrossRef]
- Maes, M. The immunoregulatory effects of antidepressants. Hum. Psychopharmacol. 2001, 16, 95–103. [Google Scholar] [CrossRef]
- Pfeilschifter, J.; Köditz, R.; Pfohl, M.; Schatz, H. Changes in proinflammatory cytokine activity after menopause. Endocr. Rev. 2002, 23, 90–119. [Google Scholar] [CrossRef] [PubMed]
- Divani, A.A.; Luo, X.; Datta, Y.H.; Flaherty, J.D.; Panoskaltsis-Mortari, A. Effect of Oral and Vaginal Hormonal Contraceptives on Inflammatory Blood Biomarkers. Mediat. Inflamm. 2015, 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Ortega, E.; García, J.J.; Bote, M.E.; Martín-Cordero, L.; Escalante, Y.; Saavedra, J.M.; Northoff, H.; Giraldo, E.; Ortega, E. Exercise in fibromyalgia and related inflammatory disorders: Known effects and unknown chances. Rheumatol. Int. 2007, 27, 441–446. [Google Scholar]
- Bote, M.E.; Garcia, J.J.; Hinchado, M.D.; Ortega, E. An exploratory study of the effect of regular aquatic exercise on the function of neutrophils from women with fibromyalgia: Role of IL-8 and noradrenaline. Brain Behav. Immun. 2014, 39, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Nilsonne, G.; Lekander, M.; Åkerstedt, T.; Axelsson, J.; Ingre, M. Diurnal variation of circulating interleukin-6 in humans: A meta-analysis. PLoS ONE 2016, 11, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Grudet, C.; Malm, J.; Westrin, A.; Brundin, L. Suicidal patients are deficient in vitamin D, associated with a pro-inflammatory status in the blood. Psychoneuroendocrinology 2014, 50C, 210–219. [Google Scholar] [CrossRef]
- Pérez-Aranda, A.; Andrés-Rodríguez, L.; Feliu-Soler, A.; Núñez, C.; Stephan-Otto, C.; Pastor-Mira, M.A.; López-Roig, S.; Peñacoba, C.; Calandre, E.P.; Slim, M.; et al. Clustering a large Spanish sample of patients with fibromyalgia using the Fibromyalgia Impact Questionnaire–Revised. Pain 2019, 160, 908–921. [Google Scholar] [CrossRef]
- Bäckryd, E.; Tanum, L.; Lind, A.-L.; Larsson, A.; Gordh, T. Evidence of both systemic inflammation and neuroinflammation in fibromyalgia patients, as assessed by a multiplex protein panel applied to the cerebrospinal fluid and to plasma. J. Pain Res. 2017, 10, 515–525. [Google Scholar] [CrossRef]
- Tsilioni, I.; Russell, I.J.; Stewart, J.M.; Gleason, R.M.; Theoharides, C.; Theoharides, T.C.; Theoharides, C. Neuropeptides CRH, SP, HK-1 and inflammatory cytokines IL-6, TNF are increased in serum of patients with Fibromyalgia Syndrome implicating mast cells. J. Pharmacol. Exp. Ther. 2016, 356, 664–672. [Google Scholar] [CrossRef]
- Mendieta, D.; De la Cruz-Aguilera, D.L.; Barrera-Villalpando, M.I.; Becerril-Villanueva, E.; Arreola, R.; Hernández-Ferreira, E.; Pérez-Tapia, S.M.; Pérez-Sánchez, G.; Garcés-Alvarez, M.E.; Aguirre-Cruz, L.; et al. IL-8 and IL-6 primarily mediate the inflammatory response in fibromyalgia patients. J. Neuroimmunol. 2016, 290, 22–25. [Google Scholar] [CrossRef]
- Wallace, D.J.; Linker-Israeli, M.; Hallegua, D.; Silverman, S.; Silver, D.; Weisman, M.H.; Linker-Israeli, M.; Hallegua, D.; Silverman, S.; Silver, D.; et al. Cytokines play an aetiopathogenetic role in fibromyalgia: A hypothesis and pilot study. Rheumatology (Oxford) 2001, 40, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Togo, F.; Natelson, B.H.; Adler, G.K.; Ottenweller, J.E.; Goldenberg, D.L.; Struzik, Z.R.; Yamamoto, Y. Plasma cytokine fluctuations over time in healthy controls and patients with fibromyalgia. Exp. Biol. Med. (Maywood) 2009, 234, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Behm, F.G.; Gavin, I.M.; Karpenko, O.; Lindgren, V.; Gaitonde, S.; Gashkoff, P.A.; Gillis, B.S. Unique immunologic patterns in fibromyalgia. BMC Clin. Pathol. 2012, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Haynes, W.L.; Michalek, J.E.; Russell, I.J. Elevated serum high-sensitivity C-reactive protein levels in fibromyalgia syndrome patients correlate with body mass index, interleukin-6, interleukin-8, erythrocyte sedimentation rate. Rheumatol. Int. 2013, 33, 1259–1264. [Google Scholar] [CrossRef] [PubMed]
- Ernberg, M.; Christidis, N.; Ghafouri, B.; Bileviciute-Ljungar, I.; Lofgren, M.; Bjersing, J.; Palstam, A.; Larsson, A.; Mannerkorpi, K.; Gerdle, B.; et al. Plasma Cytokine Levels in Fibromyalgia and Their Response to 15 Weeks of Progressive Resistance Exercise or Relaxation Therapy. Mediators Inflamm. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ortega, E.; Bote, M.E.; Giraldo, E.; García, J.J. Aquatic exercise improves the monocyte pro- and anti-inflammatory cytokine production balance in fibromyalgia patients. Scand. J. Med. Sci. Sport. 2012, 22, 104–112. [Google Scholar] [CrossRef]
- Elmas, O.; Yildiz, S.; Bilgin, S.; Demirci, S.; Comlekci, S.; Koyuncuoglu, H.R.; Akkus, S.; Colak, O.H.; Koklukaya, E.; Arslan, E.; et al. Physiological parameters as a tool in the diagnosis of fibromyalgia syndrome in females: A preliminary study. Life Sci. 2016, 145, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Akturk, S.; Buyukavci, R.; Aktürk, S.; Büyükavcı, R.; Akturk, S.; Buyukavci, R. Evaluation of blood neutrophil-lymphocyte ratio and platelet distribution width as inflammatory markers in patients with fibromyalgia. Clin. Rheumatol. 2017, 36, 1885–1889. [Google Scholar] [CrossRef]
- Gota, C.E.; Kaouk, S.; Wilke, W.S. Fibromyalgia and obesity: The association between body mass index and disability, depression, history of abuse, medications, and comorbidities. J. Clin. Rheumatol. 2015, 21, 289–295. [Google Scholar] [CrossRef]
- Feinberg, T.; Sambamoorthi, U.; Lilly, C.; Innes, K.K. Potential Mediators between Fibromyalgia and C-Reactive protein: Results from a Large U.S. Community Survey. BMC Musculoskelet. Disord. 2017, 18, 294. [Google Scholar] [CrossRef]
- Moraes, J.B.; Maes, M.; Barbosa, D.S.; Ferrari, T.Z.; Uehara, M.K.S.; Carvalho, A.F.; Nunes, S.O.V. Elevated C-reactive Protein Levels in Women with Bipolar Disorder may be Explained by a History of Childhood Trauma, Especially Sexual Abuse, Body Mass Index and Age. CNS Neurol. Disord. Drug Targets 2017, 16, 514–521. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Congio, A.; Moraes, J.B.; Bonifacio, K.L.; Barbosa, D.S.; Vargas, H.O.; Morris, G.; Puri, B.K.; Michelin, A.P.; Nunes, S.O.V. Early Life Trauma Predicts Affective Phenomenology and the Effects are Partly Mediated by Staging Coupled with Lowered Lipid-Associated Antioxidant Defences. Biomol. Concepts 2018, 9, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Devos, N.; Wauters, A.; Demedts, P.; Maurits, V.; Neels, H.; Bosmans, E.; Altamura, C.; Lin, A.; Song, C.; et al. Inflammatory markers in younger vs elderly normal volunteers and in patients with Alzheimer’s disease. J. Psychiatr. Res. 1999, 33, 397–405. [Google Scholar] [CrossRef]
- Maes, M.; Carvalho, A.F. The Compensatory Immune-Regulatory Reflex System (CIRS) in Depression and Bipolar Disorder. Mol. Neurobiol. 2018, 55, 8885–8903. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Anderson, G.; Kubera, M.; Berk, M. Targeting classical IL-6 signalling or IL-6 trans -signalling in depression? Expert Opin. Ther. Targets 2014, 18, 495–512. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.Q.; Liu, Z.; Liu, Z.H.; Chen, S.P.; Li, M.; Shahveranov, A.; Ye, D.W.; Tian, Y.K. Interleukin-6: An emerging regulator of pathological pain. J. Neuroinflammation 2016, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rabe, B.; Chalaris, A.; May, U.; Waetzig, G.H.; Seegert, D.; Williams, A.S.; Jones, S.A.; Rose-John, S.; Scheller, J. Transgenic blockade of interleukin 6 transsignaling abrogates inflammation. Blood 2008, 111, 1021–1028. [Google Scholar] [CrossRef] [PubMed]
- Uçeyler, N.; Valenza, R.; Stock, M.; Schedel, R.; Sprotte, G.; Sommer, C. Reduced levels of antiinflammatory cytokines in patients with chronic widespread pain. Arthritis Rheum. 2006, 54, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Brewer, K.; Friesland, A.; Whitfield, B.; Nolan, T. Neuroprotective Effects of Ceftriaxone Following Excitotoxic Spinal Cord Injury. Acad. Emerg. Med. 1999, 14, S174. [Google Scholar] [CrossRef]
- Carlson, N.R. Fisiología de la Conducta; Pearson Educación: Madrid, Spain, 2014. [Google Scholar]
- Abraham, K.E.; Mcmillen, D.; Brewer, K.L. The effects of endogenous interleukin-10 on gray matter damage and the development of pain behaviors following excitotoxic spinal cord injury in the mouse. Neuroscience 2004, 124, 945–952. [Google Scholar] [CrossRef]
- Jostock, T.; Müllberg, J.; Ozbek, S.; Atreya, R.; Blinn, G.; Voltz, N.; Fischer, M.; Neurath, M.F.; Rose-John, S. Soluble gp130 is the natural inhibitor of soluble interleukin-6 receptor transsignaling responses. Eur. J. Biochem. 2001, 268, 160–167. [Google Scholar] [CrossRef]
- Metcalfe, S.M.; Strom, T.B.; Williams, A.; Fahmy, T.M. Multiple Sclerosis and the LIF/IL-6 Axis: Use of Nanotechnology to Harness the Tolerogenic and Reparative Properties of LIF. Nanobiomedicine 2015, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- McKay Hart, A.; Wiberg, M.; Terenghi, G. Exogenous leukaemia inhibitory factor enhances nerve regeneration after late secondary repair using a bioartificial nerve conduit. Br. J. Plast. Surg. 2003, 56, 444–450. [Google Scholar] [CrossRef]
- Sampath, S.C.; Sampath, S.C.; Ho, A.T.V.; Corbel, S.Y.; Millstone, J.D.; Lamb, J.; Walker, J.; Kinzel, B.; Schmedt, C.; Blau, H.M. Induction of muscle stem cell quiescence by the secreted niche factor Oncostatin M. Nat. Commun. 2018, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Clauw, D.J.; Fitzcharles, M.-A.; Goldenberg, D.L.; Häuser, W.; Katz, R.S.; Mease, P.; Russell, A.S.; Russell, I.J.; Winfield, J.B. Fibromyalgia Criteria and Severity Scales for Clinical and Epidemiological Studies: A Modification of the ACR Preliminary Diagnostic Criteria for Fibromyalgia. J. Rheumatol. 2011, 38, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Smythe, H.A.; Yunus, M.B.; Bennett, R.M.; Bombardier, C.; Goldenberg, D.L.; Tugwell, P.; Campbell, S.M.; Abeles, M.; Clark, P.; et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum. 1990, 33, 160–172. [Google Scholar] [CrossRef]
- Feliu-Soler, A.; Borràs, X.; Peñarrubia-María, M.T.; Rozadilla-Sacanell, A.; D’Amico, F.; Moss-Morris, R.; Howard, M.A.; Fayed, N.; Soriano-Mas, C.; Puebla-Guedea, M.; et al. Cost-utility and biological underpinnings of Mindfulness-Based Stress Reduction (MBSR) versus a psychoeducational programme (FibroQoL) for fibromyalgia: A 12-month randomised controlled trial (EUDAIMON study). BMC Complement. Altern. Med. 2016, 16, 81. [Google Scholar] [CrossRef]
- Luciano, J.V.; Aguado, J.; Serrano-Blanco, A.; Calandre, E.P.; Rodriguez-Lopez, C.M. Dimensionality, reliability and validity of the Revised Fibromyalgia Impact Questionnaire (FIQR) in two Spanish samples. Arthritis Care Res. (Hoboken). 2013, 65, 1682–1689. [Google Scholar] [CrossRef]
- Bennett, R.M.; Friend, R.; Jones, K.D.; Ward, R.; Han, B.K.; Ross, R.L. The Revised Fibromyalgia Impact Questionnaire (FIQR): Validation and psychometric properties. Arthritis Res. Ther. 2009, 11, 415. [Google Scholar] [CrossRef]
- Carrillo-de-la-Peña, M.T.; Triñanes, Y.; Conzález-Villar, A.; Romero-Yuste, S.; Gómez-Perretta, C.; Arias, M.; Wolfe, F. Convergence between the 1990 and 2010 ACR diagnostic criteria and validation of the Spanish version of the Fibromyalgia Survey Questionnaire (FSQ). Rheumatol. Int. 2014, 35, 141–151. [Google Scholar] [CrossRef]
- Luciano, J.V.; Barrada, J.R.; Aguado, J.; Osma, J.; García-Campayo, J. Bifactor analysis and construct validity of the HADS: A cross-sectional and longitudinal study in fibromyalgia patients. Psychol. Assess. 2014, 26, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr. Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- SPSS I. IBM SPSS Neural Networks 22. Available online: http://www.sussex.ac.uk/its/pdfs/SPSS_Neural_Network_22.pdf (accessed on 20 March 2016).
| Variables | FM (n = 66) | HC (n = 35) | p |
|---|---|---|---|
| Sociodemographic & biometric variables | |||
| Age (years) | 53.36 (8.05) | 54.43 (7.84) | 0.55 |
| Employed (n, %) | 26 (38.40) | 26 (74.30%) | <0.001 * |
| With primary studies or more (n, %) | 62 (94.00%) | 35 (100.00%) | <0.001 * |
| Living with someone (n, % Yes) | 61 (90.10%) | 30 (85.70%) | 0.23 |
| BMI (kg/m2) | 28.79 (5.27) | 24.03 (3.67) | <0.001 * |
| Duration of illness (years) | 12.98 (9.48) | na | na |
| In menopause (n, % Yes) | 33 (55.00%) | 22 (62.90%) | 0.72 |
| Number of comorbidities | 0.97 (1.24) | 0.26 (0.51) | <0.001 * |
| Antidepressants intake (n, % Yes) | 25 (37.90%) | 1 (2.90%) | <0.001 * |
| Clinical variables | |||
| FIQ-R (0–100) | 59.51 (21.06) | ||
| WPI (0–19) | 13.5 (3.70) | 1.6 (1.80) | <0.001 * |
| SSS (0–12) | 8.4 (2.60) | 2.1 (1.80) | <0.001 * |
| FS (0–31) | 22.00 (5.50) | 3.6 (3.20) | <0.001 * |
| HADS_A (0–21) | 10.55 (4.41) | 6.17 (3.20) | <0.001 * |
| HADS_D (0–21) | 7.77 (5.06) | 3.14 (3.33) | <0.001 * |
| HADS Total (0–42) | 18.32 (8.17) | 9.20 (6.07) | <0.001 * |
| PSS-10 (0–40) | 22.00 (9.47) | 12.57 (6.49) | <0.001 * |
| Health behaviours | |||
| Sleep quality (0–10) | 3.79 (2.07) | 6.43 (1.61) | <0.001 * |
| Healthy diet (0–10) | 6.84 (2.01) | 6.52 (2.25) | 0.24 |
| Smoke (n, % Yes) | 12.00 (19.70%) | 5.00 (14.30%) | 0.36 |
| Practice of aerobic exercise (n, % Yes) | 37.00 (60.70%) | 32.00 (91.40%) | 0.001 * |
| Immune biomarkers | |||
| IL-6 (pg/mL)* | 5.93 (9.27) | 7.56 (5.04) | 0.007 * |
| IL-10 (pg/mL)* | 10.29 (10.78) | 17.94 (15.02) | 0.04 * |
| CXCL8 (pg/mL)* | 11.68 (8.90) | 14.39 (10.29) | 0.04 * |
| hs-CRP (mg/L)* | 2.69 (2.96) | 1.33 (1.76) | 0.005 * |
| Tests | Dependent Variables | Explanatory Variables | F | df | p |
|---|---|---|---|---|---|
| Multivariate | 4 cytokines + 1 composite | Diagnosis | 3.50 | 4/96 | 0.010 * |
| Between-subject effects | IL-6 | Diagnosis | 7.54 | 1/99 | 0.007 * |
| CXCL-8 | Diagnosis | 4.59 | 1/99 | 0.035 * | |
| IL-10 | Diagnosis | 5.00 | 1/99 | 0.028 * | |
| hs-CRP | Diagnosis | 8.44 | 1/99 | 0.005 * | |
| zIL6 + zIL10 + zCXCL-8 | Diagnosis | 8.74 | 1/99 | 0.004 * | |
| Multivariate | 4 cytokines + 1 composite | Diagnosis | 2.20 | 4/92 | 0.075 |
| Age | 2.81 | 4/92 | 0.030 * | ||
| QoSleep | 1.81 | 4/92 | 0.134 | ||
| BMI groups | 1.81 | 8/186 | 0.077 | ||
| Between-subject effects | IL-6 | Diagnosis | 5.45 | 1/95 | 0.022 * |
| CXCL-8 | Diagnosis | 4.40 | 1/95 | 0.039 * | |
| IL-10 | Diagnosis | 7.11 | 1/95 | 0.009 * | |
| hs-CRP | Diagnosis | 0.70 | 1/95 | 0.406 | |
| zIL6 + zIL10 + zCXCL-8 | Diagnosis | 8.78 | 1/95 | 0.004 * | |
| hs-CRP | BMI groups | 6.46 | 1/95 | 0.002 * | |
| IL-10 | QoSleep | 4.40 | 1/95 | 0.039 * | |
| IL-6 | Age | 8.51 | 1/95 | 0.004 * |
| Variables | HC | FMS |
|---|---|---|
| zIL-6 | 0.370 (0.196) | −0.218 (0.124) |
| zCXCL-8 | 0.338 (0.205) | −0.215 (0.130) |
| zIL-10 | 0.446 (0.204) | −0.253 (0.129) |
| zhs-CRP | −0.069 (0.190) | 0.135 (0.120) |
| zIL6 + zIL10 + zCXCL-8 | 0.472 (0.198) | −0.281 (0.125) |
| No | Explanatory Variables | B | SE | Wald | df | p | OR | 95% CI |
|---|---|---|---|---|---|---|---|---|
| #1 | IL-10 | −0.710 | 0.337 | 4.447 | 1 | 0.035 | 0.492 | 0.254–0.951 |
| Comorbidities | 1.302 | 0.653 | 3.976 | 1 | 0.046 | 3.676 | 1.022–13.218 | |
| QoSleep | −1.020 | 0.236 | 18.720 | 1 | <0.001 | 0.360 | 0.227–0.572 | |
| Aerobic exercise | −2.738 | 0.956 | 8.201 | 1 | 0.004 | 0.065 | 0.010–0.421 | |
| #2 | IL-10 | −0.742 | 0.325 | 5.211 | 1 | 0.022 | 0.476 | 0.252–0.900 |
| HADS-A | 0.179 | 0.080 | 5.014 | 1 | 0.025 | 1.196 | 1.023–1.399 | |
| QoSleep | −0.816 | 0.203 | 16.157 | 1 | <0.001 | 0.442 | 0.297–0.658 |
| Models | NN#1 | NN#2 | NN#3 | |
|---|---|---|---|---|
| Input Layer | Number of units | 11 + bias | 9 | 11 + bias |
| Rescaling method | Standardized | Standardized | Adjusted normalized | |
| Hidden layers | Number of hidden layers | 2 | 1 | 2 |
| Number of units in hidden layer 1 | 4 | 4 | 3 | |
| Number of units in hidden layer 2 | 3 | - | 2 | |
| Activation Function | Hyperbolic tangent | Hyperbolic tangent | Hyperbolic tangent | |
| Output layer | Dependent variables | WPI score | SSS score | FM vs. controls (group membership) |
| Number of units | 1 | 1 | 2 | |
| Rescaling method | Standardized | Standardized | ||
| Activation function | Identity | Identity | Identity | |
| Error function | Sum of squares (SS) | Sum of squares (SS) | SS | |
| Training | SS error | 10.742 | 10.096 | 6.971 |
| Relative error or % incorrect | 0.413 | 0.439 | 18.7% | |
| Prediction (sensitivity - specificity) | 82.1%–80.0% | |||
| SS error | 4.959 | 3.857 | 1.612 | |
| Testing | Relative error or % incorrect | 0.421 | 0.378 | 9.1% |
| Prediction (sensitivity - specificity) | 93.3%–83.3% | |||
| AUC ROC | 0.908 | |||
| Holdout | Relative error or % incorrect | 0.512 | 0.485 | 12.9% |
| Prediction (sensitivity-specificity) | 86.4%–88.9% | |||
| Correlation with predicted values | R = 0.598 | R = 0.625 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrés-Rodríguez, L.; Borràs, X.; Feliu-Soler, A.; Pérez-Aranda, A.; Rozadilla-Sacanell, A.; Arranz, B.; Montero-Marin, J.; García-Campayo, J.; Angarita-Osorio, N.; Maes, M.; et al. Machine Learning to Understand the Immune-Inflammatory Pathways in Fibromyalgia. Int. J. Mol. Sci. 2019, 20, 4231. https://doi.org/10.3390/ijms20174231
Andrés-Rodríguez L, Borràs X, Feliu-Soler A, Pérez-Aranda A, Rozadilla-Sacanell A, Arranz B, Montero-Marin J, García-Campayo J, Angarita-Osorio N, Maes M, et al. Machine Learning to Understand the Immune-Inflammatory Pathways in Fibromyalgia. International Journal of Molecular Sciences. 2019; 20(17):4231. https://doi.org/10.3390/ijms20174231
Chicago/Turabian StyleAndrés-Rodríguez, Laura, Xavier Borràs, Albert Feliu-Soler, Adrián Pérez-Aranda, Antoni Rozadilla-Sacanell, Belén Arranz, Jesús Montero-Marin, Javier García-Campayo, Natalia Angarita-Osorio, Michael Maes, and et al. 2019. "Machine Learning to Understand the Immune-Inflammatory Pathways in Fibromyalgia" International Journal of Molecular Sciences 20, no. 17: 4231. https://doi.org/10.3390/ijms20174231
APA StyleAndrés-Rodríguez, L., Borràs, X., Feliu-Soler, A., Pérez-Aranda, A., Rozadilla-Sacanell, A., Arranz, B., Montero-Marin, J., García-Campayo, J., Angarita-Osorio, N., Maes, M., & Luciano, J. V. (2019). Machine Learning to Understand the Immune-Inflammatory Pathways in Fibromyalgia. International Journal of Molecular Sciences, 20(17), 4231. https://doi.org/10.3390/ijms20174231






