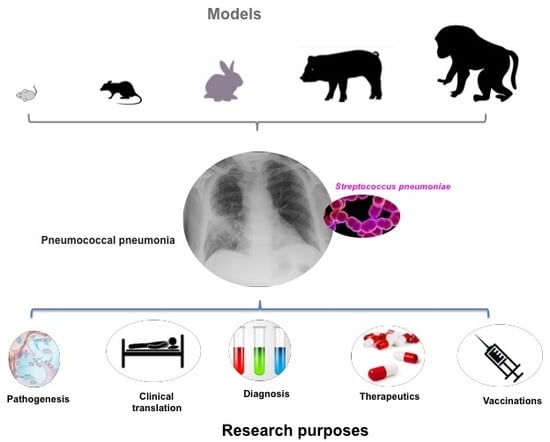
Animal Models of Pneumococcal pneumonia
Abstract
1. Introduction
2. Search Strategy and Selection Criteria
3. Mouse Models of Pneumococcal pneumonia
3.1. Mouse and Pneumococcal Strains
3.2. Routes of Infection
3.3. Particular Models of Pneumococcal Pneumonia in Mice
4. Rat Models
5. Rabbit Models
6. Swine Models
7. Non-Human Primates
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- GBD 2015 LRI Collaborators. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 1133–1161. [Google Scholar] [CrossRef]
- Musher, D.M.; Thorner, A.R. Community-acquired pneumonia. N. Engl. J. Med. 2014, 371, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Self, W.H.; Wunderink, R.G.; Fakhran, S.; Balk, R.; Bramley, A.M.; Reed, C.; Grijalva, C.G.; Anderson, E.J.; Courtney, D.M.; et al. CDC EPIC Study Team. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N. Engl. J. Med. 2015, 373, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Wunderink, R.G.; Waterer, G.W. Community-acquired pneumonia. N. Engl. J. Med. 2014, 370, 1863. [Google Scholar] [CrossRef] [PubMed]
- WHO. The Top 10 Causes of Death. 2016. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 24 May 2018).
- File, T.M., Jr. Clinical implications and treatment of multiresistant Streptococcus pneumoniae pneumonia. Clin. Microbiol. Infect. 2006, 12, 31–41. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Richter, S.S.; Heilmann, K.P.; Dohrn, C.L.; Riahi, F.; Beekmann, S.E.; Doern, G.V. Changing epidemiology of antimicrobial-resistant Streptococcus pneumoniae in the United States, 2004–2005. Clin. Infect. Dis. 2009, 48, e23–e33. [Google Scholar] [CrossRef] [PubMed]
- Yanagihara, K.; Izumikawa, K.; Higa, F.; Tateyama, M.; Tokimatsu, I.; Hiramatsu, K.; Fujita, J.; Kadota, J.; Kohno, S. Efficacy of azithromycin in the treatment of community-acquired pneumonia, including patients with macrolide-resistant Streptococcus pneumoniae infection. Intern. Med. 2009, 48, 527–535. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sohail, I.; Ghosh, S.; Mukundan, S.; Zelewski, S.; Khan, M.N. Role of Inflammatory Risk Factors in the Pathogenesis of Streptococcus pneumoniae. Front. Immunol. 2018, 9, 2275. [Google Scholar] [CrossRef] [PubMed]
- Weiser, J.N.; Ferreira, D.M.; Paton, J.C. Streptococcus pneumoniae: Transmission, colonization and invasion. Nat. Rev. Microbiol. 2018, 16, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Kadioglu, A.; Taylor, S.; Iannelli, F.; Pozzi, G.; Mitchell, T.J.; Andrew, P.W. Upper and lower respiratory tract infection by Streptococcus pneumoniae is affected by pneumolysin deficiency and differences in capsule type. Infect. Immun. 2002, 70, 2886–2890. [Google Scholar] [CrossRef] [PubMed]
- Briles, D.E.; Crain, M.J.; Gray, B.M.; Forman, C.; Yother, J. Strong association between capsular type and virulence for mice among human isolates of Streptococcus pneumoniae. Infect. Immun. 1992, 60, 111–116. [Google Scholar] [PubMed]
- Azoulay-Dupuis, E.; Rieux, V.; Muffat-Joly, M.; Bédos, J.P.; Vallée, E.; Rivier, C.; Isturiz, R.; Carbon, C.; Moine, P. Relationship between capsular type, penicillin susceptibility, and virulence of human Streptococcus pneumoniae isolates in mice. Antimicrob. Agents Chemother. 2000, 44, 1575–1577. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ogunniyi, A.D.; LeMessurier, K.S.; Graham, R.M.; Watt, J.M.; Briles, D.E.; Stroeher, U.H.; Paton, J.C. Contributions of pneumolysin, pneumococcal surface protein A (PspA), and PspC to pathogenicity of Streptococcus pneumoniae D39 in a mouse model. Infect. Immun. 2007, 75, 1843–1851. [Google Scholar] [CrossRef] [PubMed]
- Musher, D.M.; Phan, H.M.; Baughn, R.E. Protection against bacteremic pneumococcal infection by antibody to pneumolysin. J. Infect. Dis. 2001, 183, 827–830. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rubins, J.B.; Charboneau, D.; Paton, J.C.; Mitchell, T.J.; Andrew, P.W.; Janoff, E.N. Dual function of pneumolysin in the early pathogenesis of murine pneumococcal pneumonia. J. Clin. Invest. 1995, 95, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Andisi, V.F.; Hinojosa, C.A.; De Jong, A.; Kuipers, O.P.; Orihuela, C.J.; Bijlsma, J.J. Pneumococcal gene complex involved in resistance to extracellular oxidative stress. Infect. Immun. 2012, 80, 1037–1049. [Google Scholar] [CrossRef]
- Kalin, M. Pneumococcal serotypes and their clinical relevance. Thorax 1998, 53, 159–162. [Google Scholar] [CrossRef]
- Langley, J.M.; Kellner, J.D.; Solomon, N.; Robinson, J.L.; Le Saux, N.; McDonald, J.; Ulloa-Gutierrez, R.; Tan, B.; Allen, U.; Dobson, S.; et al. Empyema associated with community-acquired pneumonia: A Pediatric Investigator’s Collaborative Network on Infections in Canada (PICNIC) study. BMC Infect. Dis. 2008, 8, 129. [Google Scholar] [CrossRef]
- Hausdorff, W.P.; Bryant, J.; Paradiso, P.R.; Siber, G.R. Which pneumococcal serogroups cause the most invasive disease: Implications for conjugate vaccine formulation and use, part I. Clin. Infect. Dis. 2000, 30, 100–121. [Google Scholar] [CrossRef]
- Yalcin, B.; Nicod, J.; Bhomra, A.; Davidson, S.; Cleak, J.; Farinelli, L.; Østerås, M.; Whitley, A.; Yuan, W.; Gan, X.; et al. Commercially available outbred mice for genome-wide association studies. PLoS Genet. 2010, 6, e1001085. [Google Scholar] [CrossRef]
- Sandgren, A.; Sjostrom, K.; Olsson-Liljequist, B.; Christensson, B.; Samuelsson, A.; Kronvall, G.; Henriques Normark, B. Effect of clonal and serotype-specific properties on the invasive capacity of Streptococcus pneumoniae. J. Infect. Dis. 2004, 189, 785–796. [Google Scholar] [PubMed]
- Savulescu, C.; Krizova, P.; Lepoutre, A.; Mereckiene, J.; Vestrheim, D.F.; Ciruela, P.; Ordobas, M.; Guevara, M.; McDonald, E.; Morfeldt, E.; et al. Effect of high-valency pneumococcal conjugate vaccines on invasive pneumococcal disease in children in SpIDnet countries: An observational multicentre study. Lancet Respir. Med. 2017, 5, 648–656. [Google Scholar] [CrossRef]
- Thevaranjan, N.; Whelan, F.J.; Puchta, A.; Ashu, E.; Rossi, L.; Surette, M.G.; Bowdish, D.M. Streptococcus pneumoniae Colonization Disrupts the Microbial Community within the Upper Respiratory Tract of Aging Mice. Infect. Immun. 2016, 84, 906–916. [Google Scholar] [CrossRef] [PubMed]
- Azoulay-Dupuis, E.; Vallee, E.; Veber, B.; Bedos, J.P.; Bauchet, J.; Pocidalo, J.J. In vivo efficacy of a new fluoroquinolone, sparfloxacin, againstpenicillin-susceptible and -resistant and multiresistant strains of Streptococcus pneumoniae in a mouse model of pneumonia. Antimicrob. Agents Chemother. 1992, 36, 2698–2703. [Google Scholar] [CrossRef]
- Sauve, C.; Azoulay-Dupuis, E.; Moine, P.; Darras-Joly, C.; Rieux, V.; Carbon, C.; Bédos, J.P. Efficacies of cefotaxime and ceftriaxone in a mouse model of pneumonia induced by two penicillin- and cephalosporin-resistant strains of Streptococcus pneumoniae. Antimicrob. Agents Chemother. 1996, 40, 2829–2834. [Google Scholar] [CrossRef]
- Moine, P.; Vallée, E.; Azoulay-Dupuis, E.; Bourget, P.; Bédos, J.P.; Bauchet, J.; Pocidalo, J.J. In vivo efficacy of a broad-spectrum cephalosporin, ceftriaxone, against penicillin-susceptible and -resistant strains of Streptococcus pneumonia in a mouse pneumonia model. Antimicrob. Agents Chemother. 1994, 38, 1953–1958. [Google Scholar] [CrossRef]
- Sandgren, A.; Albiger, B.; Orihuela, C.J.; Tuomanen, E.; Normark, S.; Henriques-Normark, B. Virulence in mice of pneumococcal clonal types with known invasive disease potential in humans. J. Infect. Dis. 2005, 192, 791–800. [Google Scholar]
- Hinojosa, E.; Boyd, A.R.; Orihuela, C.J. Age-associated inflammation and toll-like receptor dysfunction prime the lungs for pneumococcal pneumonia. J. Infect. Dis. 2009, 200, 546–554. [Google Scholar] [CrossRef]
- Clement, C.G.; Tuvim, M.J.; Evans, C.M.; Tuvin, D.M.; Dickey, B.F.; Evans, S.E. Allergic lung inflammation alters neither susceptibility to Streptococcus pneumoniae infection nor inducibility of innate resistance in mice. Respir. Res. 2009, 10, 70. [Google Scholar] [CrossRef]
- Shivshankar, P.; Boyd, A.R.; Le Saux, C.J.; Yeh, I.T.; Orihuela, C.J. Cellular senescence increases expression of bacterial ligands in the lungs and is positively correlated with increased susceptibility to pneumococcal pneumonia. Aging Cell 2011, 10, 798–806. [Google Scholar] [CrossRef]
- Briles, D.E.; Hollingshead, S.K.; Paton, J.C.; Ades, E.W.; Novak, L.; van Ginkel, F.W.; Benjamin, W.H., Jr. Immunizations with pneumococcal surface protein A and pneumolysin are protective against pneumonia in a murine model of pulmonary infection with Streptococcus pneumoniae. J. Infect. Dis. 2003, 188, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Wanke-Jellinek, L.; Keegan, J.W.; Dolan, J.W.; Lederer, J.A. Characterization of lung infection-induced TCRγδ T cell phenotypes by CyTOF mass cytometry. J. Leukoc. Biol. 2016, 99, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Azoulay-Dupuis, E.; Mohler, J.; Bédos, J.P. Efficacy of BB-83698, a novel peptide deformylase inhibitor, in a mouse model of pneumococcal pneumonia. Antimicrob. Agents Chemother. 2004, 48, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Bédos, J.P.; Azoulay-Dupuis, E.; Moine, P.; Muffat-Joly, M.; Veber, B.; Pocidalo, J.J.; Vallée, E. Pharmacodynamic activities of ciprofloxacin and sparfloxacin in a murine pneumococcal pneumonia model: Relevance for drug efficacy. J. Pharm. Exp. Ther. 1998, 286, 29–35. [Google Scholar]
- Abgueguen, P.; Azoulay-Dupuis, E.; Noel, V.; Moine, P.; Rieux, V.; Fantin, B.; Bedos, J.P. Amoxicillin is effective against penicillin-resistant Streptococcus pneumonia strains in a mouse pneumonia model simulating human, pharmacokinetics. Antimicrob. Agents Chemother. 2007, 51, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Moine, P.; Mazoit, J.X.; Bédos, J.P.; Vallée, E.; Azoulay-Dupuis, E. Correlation between in vitro and in vivo activity of amoxicillin against Streptococcus pneumoniae in a murine pneumonia model. J. Pharm. Exp. Ther. 1997, 280, 310–315. [Google Scholar]
- Moine, P.; Mazoit, J.X. Streptococcus pneumoniae pneumonia in mice: Optimal amoxicillin dosing predicted from a pharmacokinetic-pharmacodynamic model. J. Pharmacol. Exp. Ther. 1999, 291, 1086–1092. [Google Scholar] [PubMed]
- Briles, D.E.; Nahm, M.; Schroer, K.; Davie, J.; Baker, P.; Kearney, J.; Barletta, R. Antiphosphocholine. Type 2 streptococcus pneumoniae. J. Exp. Med. 1981, 153, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Tessier, P.R.; Kim, M.K.; Zhou, W.; Xuan, D.; Li, C.; Ye, M.; Nightingale, C.H.; Nicolau, D.P. Pharmacodynamic assessment of clarithromycin in a murine model of pneumococcal pneumonia. Antimicrob. Agents Chemother. 2002, 46, 1425–1434. [Google Scholar] [CrossRef] [PubMed]
- Bou Ghanem, E.N.; Clark, S.; Du, X.; Wu, D.; Camili, A.; Leong, J.M.; Meydani, S.N. The α-tocopherol form of vitamin E reverses age-associated susceptibility to streptococcus pneumoniae lung infection by modulating pulmonary neutrophil recruitment. J. Immunol. 2015, 194, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.J.; Rooney, K.; Choi, A.M.K.; Stout-Delgado, H.W. NLRP3 inflammasome activation in aged macrophages is diminished during Streptococcus pneumoniae infection. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L372–L387. [Google Scholar] [CrossRef] [PubMed]
- Janesch, P.; Stulik, L.; Rouha, H.; Varga, C.; Steinhäuser, J.; Zerbs, M.; Nagy, E. Age-related changes in the levels and kinetics of pulmonary cytokine and chemokine responses to Streptococcuspneumoniae in mouse pneumonia models. Cytokine 2018, 111, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.E.; José, R.J.; Brown, J.S.; Chambers, R.C. Enhanced inflammation in aged mice following infection with Streptococcus pneumoniae is associated with decreased IL-10 and augmented chemokine production. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 308, L539–L549. [Google Scholar] [CrossRef] [PubMed]
- Rubins, J.B.; Charboneau, D. Effect of anesthetics on pathogenesis of experimentally induced murine pneumococcal pneumonia. Comp. Med. 2000, 50, 292–295. [Google Scholar] [PubMed]
- Majhi, A.; Kundu, K.; Adhikary, R.; Banerjee, M.; Mahanti, S.; Basu, A.; Bishayi, B. Combination therapy with ampicillin azithromycin in an experimental pneumococcal pneumonia is bactericidal effective in down regulating inflammation in mice. J. Inflamm. (Lond.) 2014, 11, 5. [Google Scholar] [CrossRef]
- Brumshagen, C.; Maus, R.; Bischof, A.; Ueberberg, B.; Bohling, J.; Osterholzer, J.J.; Ogunniyi, A.D.; Paton, J.C.; Welte, T.; Maus, U.A. FMS-like tyrosine kinase 3 ligand treatment of mice aggravates acute lung injury in response to Streptococcus pneumoniae: Role of pneumolysin. Infect. Immun. 2012, 80, 4281–4290. [Google Scholar] [PubMed]
- Henken, S.; Bohling, J.; Martens-Lobenhoffer, J.; Paton, J.C.; Ogunniyi, A.D.; Briles, D.E.; Salisbury, V.C.; Wedekind, D.; Bode-Böger, S.M.; Welsh, T.; et al. Efficacy profiles of daptomycin for treatment of invasive and non invasive pulmonary infections with Streptococcus pneumoniae. Antimicrob. Agents Chemother. 2010, 54, 707–717. [Google Scholar]
- Dommaschk, A.; Ding, N.; Tort Tarres, M.; Bittersohl, L.F.; Maus, R.; Stolper, J.; Jonigk, D.; Braubach, P.; Lippmann, T.; Welte, T.; et al. Nasopharyngeal colonization with Streptococcus pneumoniae triggers dendritic cell dependent antibody responses against invasive disease in mice. Eur. J. Immunol. 2017, 47, 540–551. [Google Scholar] [CrossRef]
- Herbold, W.; Maus, R.; Hahn, I.; Ding, N.; Srivastava, M.; Christman, J.W.; Mack, M.; Reutershan, J.; Briles, D.E.; Paton, J.C.; et al. Importance of CXC chemokine receptor 2 in alveolar neutrophil and exudate macrophage recruitment in response to pneumococcal lung infection. Infect. Immun. 2010, 78, 2620–2630. [Google Scholar] [CrossRef]
- Mracsko, E.; Stegemann-Koniszewski, S.; Na, S.Y.; Dalpke, A.; Bruder, D.; Lasitschka, F.; Veltkamp, R. A Mouse Model of Post-Stroke Pneumonia Induced by Intra-Tracheal Inoculation with Streptococcus pneumoniae. Cereb. Dis. 2017, 43, 99–109. [Google Scholar] [CrossRef]
- Kim, M.K.; Zhou, W.; Tessier, P.R.; Xuan, D.; Ye, M.; Nightingale, C.H.; Nicolau, D.P. Bactericidal effect and pharmacodynamics of cethromycin (ABT-773) in a murine pneumococcal pneumonia model. Antimicrob. Agents Chemother. 2002, 46, 3185–3192. [Google Scholar] [CrossRef] [PubMed]
- Dandekar, P.K.; Williams, P.; Tessier, P.R.; Farrell, D.J.; Nightingale, C.H.; Nicolau, D.P. Assessment of the efficacy of telithromycin simulating human exposures against S. pneumoniae with ribosomal mutations in a murine pneumonia model. Int. J. Antimicrob. Agents. 2005, 25, 530–534. [Google Scholar] [CrossRef] [PubMed]
- Azoulay-Dupuis, E.; Vallée, E.; Bedos, J.P.; Muffat-Joly, M.; Pocidalo, J.J. Prophylactic and therapeutic activities of azithromycin in a mouse model of pneumococcal pneumonia. Antimicrob. Agents Chemother. 1991, 35, 1024–1028. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, Y.; Ouellet, N.; Deslauriers, A.M.; Simard, M.; Olivier, M.; Bergeron, M.G. Cytokine kinetics and other host factors in response to pneumococcal pulmonary infection in mice. Infect. Immun. 1998, 66, 912–922. [Google Scholar] [PubMed]
- Calame, W.; Douwes-Idema, A.E.; van den Barselaar, M.T.; Mattie, H. Contribution ofalveolar phagocytes to antibiotic efficacy in an experimental lung infection with Streptococcus pneumoniae. J. Infect. 2001, 42, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Feldmesser, M.; Casadevall, A. Effect of serum IgG1 to Cryptococcus neoformans glucuronoxylomannan on murine pulmonary infection. J. Immunol. 1997, 158, 790–799. [Google Scholar]
- Dolan, J.M.; Weinberg, J.B.; O’Brien, E.; Abashian, A.; Procario, M.C.; Aronoff, D.M.; Crofford, L.J.; Peters-Golden, M.; Ward, L.; Mancuso, P. Increased lethality and defective pulmonary clearance of Streptococcus pneumoniae in microsomal prostaglandin E synthase-1-knockout mice. Am. J. Physiol Lung Cell Mol. Physiol. 2016, 310, L1111–L1120. [Google Scholar] [CrossRef]
- Mancuso, P.; OBrien, E.; Prano, J.; Goel, D.; Aronoff, D.M. No Impairment in hostdefense against Streptococcus pneumoniae in obese CPEfat/fat mice. PLoS ONE 2014, 9, e106420. [Google Scholar] [CrossRef]
- Yamamoto, K.; Ahyi, A.N.; Pepper-Cunningham, Z.A.; Ferrari, J.D.; Wilson, A.A.; Jones, M.R.; Quinton, L.J.; Mizgerd, J.P. Roles of lung epithelium in neutrophil recruitment during pneumococcal pneumonia. Am. J. Respir. Cell Mol. Biol. 2014, 50, 253–262. [Google Scholar] [CrossRef]
- Suzuki, H.; Hogg, J.C.; van Eeden, S.F. Sequestration and homing of bone marrow-derived lineage negative progenitor cells in the lung during pneumococcal pneumonia. Respir. Res. 2008, 9, 25. [Google Scholar] [CrossRef]
- Winter, C.; Taut, K.; Länger, F.; Mack, M.; Briles, D.E.; Paton, J.C.; Maus, R.; Srivastava, M.; Welte, T.; Maus, U.A. FMS-like tyrosine kinase 3 ligand aggravates the lung inflammatory response to Streptococcus pneumoniae infection in mice: Role of dendritic cells. J. Immunol. 2007, 179, 3099–3108. [Google Scholar] [CrossRef] [PubMed]
- Burns, T.; Abadi, M.; Pirofski, L.A. Modulation of the lung inflammatory response to serotype 8 pneumococcal infection by a human immunoglobulin m monoclonal antibody to serotype 8 capsular polysaccharide. Infect. Immun. 2005, 73, 4530–4538. [Google Scholar] [CrossRef] [PubMed]
- Southam, D.S.; Dolovich, M.; O’Byrne, P.M.; Inman, M.D. Distribution of intranasal instillations in mice: Effects of volume, time, body position, and anesthesia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 282, L833–L839. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, B.; Guo, Y.; Li, W.; Tian, Y.; Sonnenberg, G.F.; Weiser, J.N.; Ni, X.; Shen, H. Cross-protective mucosal immunity mediated by memory Th17 cells against Streptococcus pneumoniae lung infection. Mucosal Immunol. 2017, 10, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.M.; Khandavilli, S.; Camberlein, E.; Hyams, C.; Baxendale, H.E.; Brown, J.S. Protective contributions against invasive Streptococcus pneumoniae pneumonia of antibody and Th17-cell responses to nasopharyngeal colonization. PLoS ONE 2011, 6, e25558. [Google Scholar] [CrossRef] [PubMed]
- Matthias, K.A.; Roche, A.M.; Standish, A.J.; Shchepetov, M.; Weiser, J.N. Neutrophil-toxin interactions promote antigen delivery and mucosal clearance of Streptococcus pneumoniae. J. Immunol. 2008, 180, 6246–6254. [Google Scholar] [CrossRef] [PubMed]
- Clement, C.G.; Evans, S.E.; Evans, C.M.; Hawke, D.; Kobayashi, R.; Reynolds, P.R.; Moghaddam, S.J.; Scott, B.L.; Melicoff, E.; Adachi, R.; et al. Stimulation of lung innate immunity protects against lethal pneumococcal pneumonia in mice. Am. J. Respir. Crit. Care Med. 2008, 177, 1322–1330. [Google Scholar] [CrossRef]
- Tuvim, M.J.; Clement, C.G.; Huang, E.S.; Cote, G.J.; Evans, S.E.; Lei, X.; Deftos, L.J.; Gagel, R.F.; Dickey, B.F. Deletion of the gene encoding calcitonin and calcitonin gene-related peptide α does not affect the outcome of severe infection in mice. Am. J. Respir. Cell Mol. Biol. 2013, 49, 151–155. [Google Scholar] [CrossRef]
- Ershler, W.B.; Hebert, J.C.; Blow, A.J.; Granter, S.R.; Lynch, J. Effect of thymosin alpha one on specific antibody response and susceptibility to infection in young and aged mice. Int. J. Immunopharmacol. 1985, 7, 465–471. [Google Scholar] [CrossRef]
- Iizawa, Y.; Kitamoto, N.; Hiroe, K.; Nakao, M. Streptococcus pneumoniae in the nasal cavity of mice causes lower respiratory tract infection after airway bstruction. J. Med. Microbiol. 1996, 44, 490–495. [Google Scholar] [CrossRef]
- Hoover, J.L.; Lewandowski, T.F.; Mininger, C.L.; Singley, C.M.; Sucoloski, S.; Rittenhouse, S.A. Robust Pneumonia Model in Immunocompetent Rodents to Evaluate Antibacterial Efficacy against S. pneumoniae, H. influenzae, K. pneumoniae, P. aeruginosa or A. Baumannii. J. Vis. Exp. 2017, 119, e55068. [Google Scholar]
- Mold, C.; Rodic-Polic, B.; Du Clos, T.W. Protection from Streptococcus pneumoniae infection by C-reactive protein and natural antibody requires complement but not Fc gamma receptors. J. Immunol. 2002, 168, 6375–6381. [Google Scholar] [CrossRef] [PubMed]
- Tokairin, Y.; Shibata, Y.; Sata, M.; Abe, S.; Takabatake, N.; Igarashi, A.; Ishikawa, T.; Inoue, S.; Kubota, I. Enhanced immediate inflammatory response to Streptococcus pneumoniae in the lungs of mice with pulmonary emphysema. Respirology 2008, 13, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Siggins, R.W.; Melvan, J.N.; Welsh, D.A.; Bagby, G.J.; Nelson, S.; Zhang, P. Alcohol suppresses the granulopoietic response to pulmonary Streptococcus pneumonia infection with enhancement of STAT3 signaling. J. Immunol. 2011, 186, 4306–4313. [Google Scholar] [CrossRef]
- Chiavolini, D.; Pozzi, G.; Ricci, S. Animal Models of Streptococcus pneumoniae Disease. Clin. Microbiol. Rev. 2008, 10, 666–685. [Google Scholar] [CrossRef]
- Feldman, C.; Munro, N.C.; Jeffery, P.K.; Mitchell, T.J.; Andrew, P.W.; Boulnois, G.J.; Guerreiro, D.; Rohde, J.A.L.; Todd, H.C.; Cole, P.J.; et al. Pneumolysin induces the salient histologic features of pneumococcal infection in the rat lung in vivo. Am. J. Respir. Cell Mol. Biol. 1991, 5, 416–423. [Google Scholar] [CrossRef]
- Smith, G.M.; Abbott, K.H. Development of experimental respiratory infections in neutropenic rats with either penicillin-resistant Streptococcus pneumoniae or beta-lactamase-producing Haemophilus influenzae. Antimicrob. Agents Chemother. 1994, 38, 608–610. [Google Scholar] [CrossRef][Green Version]
- Saladino, R.A.; Stack, A.M.; Fleisher, G.R.; Thompson, C.M.; Briles, D.E.; Kobzik l Siber, G.R. Development of a model of low-inoculum Streptococcus pneumoniae intrapulmonary infection in infant rats. Infect. Immun. 1997, 65, 4701–4704. [Google Scholar]
- Bakker-Woudenberg, I.A.J.M.; De Jong-Hoenderop, J.Y.T.; Michel, M.F. Efficacy of antimicrobial chemotherapy in experimental rat pneumonia: Effects of impaired phagocytosis. Infect. Immun. 1979, 25, 366–375. [Google Scholar]
- Alcantara, R.B.; Preheim, L.C.; Gentry-Nielsen, M.J. Pneumolysin- induced complement depletion during experimental pneumococcal bacteremia. Infect. Immun. 2001, 69, 3569–3575. [Google Scholar] [CrossRef]
- Aslami, H.; Pulskens, W.P.; Kuipers, M.T.; Bos, A.P.; Van Kuilenburg, A.B.P.; Wanders, R.J.A.; Roelofsen, J.; Roelofs, J.J.T.H.; Kerindongo, R.P.; Beurskens, C.J.P.; et al. Hydrogen Sulfide Donor NaHS Reduces Organ Injury in a Rat Model of Pneumococcal Pneumosepsis, Associated with Improved Bio-Energetic Status. PLoS ONE 2013, 8, e63497. [Google Scholar] [CrossRef] [PubMed]
- Peterson, G.E.; Silva, S.S.; Amantea, S.L.; Miorelli, P.; Sanches, P.; Kulczynski, J.; Roesch, E.; Fraga, J.C. Accuracy of Complement Activation Product Levels to Detect Infected Pleural Effusion in Rats. Pediatric Pulmonol. 2017, 52, 757–762. [Google Scholar] [CrossRef] [PubMed]
- van den Boogaard, F.E.; Hofstra, J.J.; Van ’t Veer, C.; Levi, M.M.; Roelofs, J.J.T.H.; Van der Poll, T.; Schultz, M.J. Feasibility and Safety of Local Treatment with Recombinant Human Tissue Factor Pathway Inhibitor in a Rat Model of Streptococcus pneumoniae Pneumonia. PLoS ONE 2015, 10, e0127261. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Candiani, G.; Abbondi, M.; Borgonovi, M.; Williams, R. Experimental lobar pneumonia due to penicillin-susceptible and penicillin-resistant Streptococcus pneumoniae in immunocompetent and neutropenic rats: Efficacy of penicillin and teicoplanin treatment. J. Antimicrob. Chemother. 1997, 39, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Kuikka, A.; Syrjanen, J.; Renkonen, O.V.; Valtonen, V.V. Pneumococcal bacteraemia during a recent decade. J. Infect. 1992, 24, 157–168. [Google Scholar] [CrossRef]
- Davis, C.C.; Mellencamp, M.A.; Preheim, L.C. A model of pneumococcal pneumonia in chronically intoxicated rats. J. Infect. Dis. 1991, 163, 799–805. [Google Scholar] [CrossRef]
- Mellencamp, M.A.; Preheim, L.C. Pneumococcal pneumonia in a rat model of cirrhosis: Effects of cirrhosis on pulmonary defense mechanisms against Streptococcus pneumoniae. J. Infect. Dis. 1991, 163, 102–108. [Google Scholar] [CrossRef]
- Hoover, J.L.; Singley, C.M.; Elefante, P.; DeMarsh, P.; Zalacain, M.; Rittenhouse, S. Reducing antibacterial development risk for GSK1322322 by exploring potential human dose regimens in nonclinical efficacy studies using immunocompetent rats. Antimicrob. Agents Chemother. 2017, 61, e00959-17. [Google Scholar] [CrossRef]
- Smith, G.M.; Slocombe, B.; Abbott, K.H.; Mizen, L.W. Activity of amoxicillin-clavulanate against penicillin-resistant Streptococcus pneumoniae in an experimental respiratory infection model in rats. Antimicrob. Agents Chemother. 1998, 42, 813–817. [Google Scholar] [CrossRef]
- Leung, L.S.; Szal, E.G.J.; Drachman, R.H. Increased susceptibility of splenectomized rats to infection with Diplococcus pneumoniae. J. Infect. Dis. 1972, 126, 507–513. [Google Scholar] [CrossRef]
- Alcantara, R.B.; Preheim, L.C.; Gentry, M.J. Role of pneumolysin’s complement-activating activity during pneumococcal bacteremia in cirrhotic rats. Infect. Immun. 1999, 67, 2862–2866. [Google Scholar] [PubMed]
- Gullstrand, P.; Alwmark, A.; Schalen, C. Effect of steroids on the outcome of penicillin treatment in pneumococcal sepsis in splenectomized rats. Surgery 1982, 91, 222–225. [Google Scholar] [PubMed]
- Offenbart, K.; Christensen, P.; Gullstrand, P.; Prellner, K.; Seger, R.A. Treatment of pneumococcal postsplenectomy sepsis in the rat with human gamma-globulin. J. Surg. Res. 1986, 40, 198–201. [Google Scholar] [CrossRef]
- Iinuma, H.; Okinaga, K. Prevention of pneumococcal bacteremia by immunization with type 6 pneumococcal capsular polysaccharide vaccine in splenectomized rats. J. Infect. Dis. 1989, 160, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Mapara, M.; Thomas, B.S.; Bhat, K.M. Rabbit as an animal model for experimental research. Dent. Res. J. 2012, 9, 111–118. [Google Scholar]
- Ganzinger, U.; Haslberger, A. Pharmacokinetics of cephalosporins in normal and septicemic rabbits. Antimicrob. Agents Chemother. 1985, 28, 473–477. [Google Scholar] [CrossRef]
- Spence, S. The Dutch-Belted rabbit: An alternative breed for developmental toxicity testing. Birth Defects Res. B Dev. Reprod. Toxicol. 2003, 68, 439–448. [Google Scholar] [CrossRef]
- Tuomanen, E.; Rich, R.; Zak, O. Induction of pulmonary inflammation by components of the pneumococcal cell surface. Am. Rev. Respir. Dis. 1987, 135, 869–874. [Google Scholar] [CrossRef]
- Charles, P.E.; Piroth, L.; Desbiolles, N.; Lequeu, C.; Martin, L.; Portier, H.; Chavanet, P. New model of ventilator-associated pneumonia in immunocompetent rabbits. Crit. Care Med. 2002, 30, 2278–2283. [Google Scholar] [CrossRef]
- Piroth, L.; Martin, L.; Coulon, A.; Lequeu, C.; Duong, M.; Buisson, M.; Portier, H.; Chavanet, P. Development of a new experimental model of penicillin-resistant Streptococcus pneumoniae pneumonia and amoxicillin treatment by reproducing human pharmacokinetics. Antimicrob. Agents Chemother. 1999, 43, 2484–2492. [Google Scholar] [CrossRef]
- Yershov, A.L.; Jordan, B.S.; Guymon, C.H.; Dubick, M.A. Relationship between the inoculum dose of Streptococcus pneumoniae and pneumonia onset in a rabbit model. Eur. Respir. J. 2005, 25, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Cabellos, C.; MacIntyre, D.E.; Forrest, M.; Burroughs, M.; Prasad, S.; Tuomanen, E. Differing roles for platelet-activating factor during inflammation of lung and subarachnoid space. The special case of Streptococcus pneumoniae. J. Clin. Investig. 1992, 90, 612–618. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Idanpaan-Heikkila, I.P.; Simon, M.; Zopf, D.; Vullo, T.; Cahill, P.; Sokol, K.; Tuomanen, E. Oligosaccharides interfere with the establishment and progression of experimental pneumococcal pneumonia. J. Infect. Dis. 1997, 176, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Croisier, D.; Chavanet, P.; Lequeu, C.; Ahanou, A.; Nierlich, A.; Neuwirth, C.; Piroth, L.; Duong, M.; Buisson, M.; Portier, H. Efficacy and pharmacodynamics of simulated human-like treatment with levafloxacin on experimental pneumonia induced with penicillin-resistant pneumococci with various susceptibilities to fluoroquinolones. J. Antimicrob. Chemother. 2002, 50, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Piroth, L.; Desbiolles, N.; Mateo-Ponce, V.; Martin, L.; Lequeu, C.; Charles, P.E.; Portier, H.; Chavanet, P. HMR 3647 human-like treatment of experimental pneumonia due to penicillin-resistant and erythromycin-resistant Streptococcus pneumoniae. J. Antimicrob. Chemother. 2001, 47, 33–42. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Croisier, D.; Etienne, M.; Bergoin, E.; Charles, P.E.; Lequeu, C.; Piroth, L.; Portier, H.; Chavanet, P. Mutant selection window in levofloxacin and moxifloxacin treatments of experimental pneumococcal pneumonia in a rabbit model of human therapy. Antimicrob. Agents Chemother. 2004, 48, 1699–1707. [Google Scholar] [CrossRef] [PubMed]
- Croisier-Bertin, D.; Piroth, L.; Charles, P.E.; Larribeau, A.; Biek, D.; Ge, Y.; Chavanet, P. Ceftaroline versus Ceftriaxone in a Highly Penicillin-Resistant Pneumococcal Pneumonia Rabbit Model Using Simulated Human Dosing. Antimicrob. Agents Chemother. 2011, 7, 3557–3563. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Guckian, J.C.; Morrey, B.F.; Kirby, H.B. Role of lysosomes and cathepsin inhibitor in plasma during pneumococcal infection. J. Infect. Dis. 1970, 122, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Guckian, J.C. Role of metabolism in pathogenesis of bacteremia due to Diplococcus pneumoniae in rabbits. J. Infect. Dis. 1973, 127, 1–8. [Google Scholar] [CrossRef]
- Guckian, J.C. Coagulopathy in experimental sepsis with Streptococcus pneumoniae in rabbits: Effect of drug therapy and splenectomy. J. Infect. Dis. 1976, 134, 150–157. [Google Scholar] [CrossRef]
- Coln, D.; Horton, J.; Ogden, M.; Williams, S. Clearance of pneumococcal organisms after repair of injured spleens. J. Pediatr. Surg. 1983, 18, 280–281. [Google Scholar] [CrossRef]
- Horton, J.; Ogden, M.E.; Williams, S.; Coln, D. The importance of splenic blood flow in clearing pneumococcal organisms. Ann. Surg. 1982, 195, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Meurens, F.; Summerfield, A.; Nauwynck, H.; Saif, L.; Gerdts, V. The pig: A model for human infectious diseases. Trends Microbiol. 2012, 20, 1. [Google Scholar] [CrossRef] [PubMed]
- Summerfield, A. Special issue on porcine immunology: An introduction from the guest editor. Dev. Comp. Immunol. 2009, 33, 265–266. [Google Scholar] [CrossRef] [PubMed]
- Swindle, M.M.; Makin, A.; Herron, A.J.; Clubb FJJr Frazier, K.S. Swine as models in biomedical research and toxicology testing. Vet. Pathol. 2012, 49, 344–356. [Google Scholar] [CrossRef]
- McLaughlin, R.F.; Tyler, W.S.; Canada, R.O. A study of the subgross pulmonary anatomy of various mammals. Am. J. Anat. 1961, 108, 149–165. [Google Scholar] [CrossRef]
- Marquette, C.H.; Wermert, D.; Wallet, F.; Copin, M.C.; Tonnel, A.B. Characterization of an animal model of ventilator-acquired pneumonia. Chest 1999, 115, 200–209. [Google Scholar] [CrossRef]
- Luna, C.M.; Sibila, O.; Agusti, C.; Torres, A. Animal models of ventilator-associated pneumonia. Eur. Respir. J. 2009, 33, 182–188. [Google Scholar] [CrossRef]
- Marquette, C.H.; Mensier, E.; Copin, M.C.; Desmidt, A.; Freitag, L.; Witt, C.; Petyt, L.; Ramon, P. Experimental models of tracheobronchial stenoses: A useful tool for evaluating airway stents. Ann. Thorac. Surg. 1995, 60, 651–656. [Google Scholar] [CrossRef]
- Wermert, D.; Marquette, C.H.; Copin, M.C.; Wallet, F.; Fraticelli, A.; Ramon, P.; Tonnel, A.B. Influence of pulmonary bacteriology and histology on the yield of diagnostic procedures in ventilator-acquired pneumonia. Am. J. Respir. Crit. Care Med. 1998, 158, 139–147. [Google Scholar] [CrossRef]
- Luna, C.M.; Bruno, D.A.; García-Morato, J.; Mann, K.C.; Risso Patrón, J.; Sagardía, J.; Absi, R.; García Bottino, M.; Marchetti, D.; Famiglietti, A.; et al. Effect of linezolid compared with glycopeptides in methicillin-resistant Staphylococcus aureus severe pneumonia in piglets. Chest 2009, 135, 1564–1571. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, I.; Wallet, F.; Nicolas-Robin, A.; Ferrari, F.; Marquette, C.H.; Rouby, J.J. Lung deposition and efficiency of nebulized amikacin during Escherichia coli pneumonia inventilated piglets. Am. J. Respir. Crit. Care Med 2002, 166, 1375–1381. [Google Scholar] [CrossRef] [PubMed]
- Sibila, O.; Agusti, C.; Torres, A.; Baquero, S.; Gando, S.; Patrón, J.R.; Morato, J.G.; Goffredo, D.H.; Bassi, N.; Luna, C.M. Experimental Pseudomonas aeruginosa pneumonia: Evaluation of the associated inflammatory response. Eur. Respir. J. 2007, 30, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- De Greeff, A.; Van Selm, S.; Buys, H.; Harders-Westerveen, J.F.; Tunjungputri, R.N.; De Mast, Q.; Van der Ven, A.J.; Stockhofe-Zurwieden, N.; De Jonge, M.I.; Smith, H.E. Pneumococcal colonization and invasive disease studied in a porcine model. BMC Microbiol. 2016, 16, 102. [Google Scholar] [CrossRef] [PubMed]
- Shak, J.R.; Cremers, A.J.; Gritzfeld, J.F.; De Jonge, M.I.; Hermans, P.W.; Vidal, J.E.; Klugman, K.P.; Gordon, S.P. Impact of experimental human pneumococcal carriage on nasopharyngeal bacterial densities in healthy adults. PLoS ONE 2014, 9, e98829. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tunjungputri, R.N.; De Jonge, M.I.; De Greeff, A.; van Selm, S.; Buys, H.; Harders-Westerveen, J.F.; Stockhofe-Zurwieden, N.; Urbanus, R.T.; De Groot, P.G.; Smith, H.E.; et al. Invasive pneumococcal disease leads to activation and hyperreactivity of platelets. Thrombosis Res. 2016, 144, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Philipp, M.T.; Doyle, L.A.; Martin, D.S.; Plauché, G.B.; Phillippi-Falkenstein, K.M.; Bohm, R.P., Jr. A rhesus macaque model of Streptococcus pneumoniae carriage. J. Med. Primatol. 2012, 41, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Proctor, M.; Manning, P.J. Production of immunoglobulin A protease by Streptococcus pneumoniae from animals. Infect. Immun. 1990, 58, 2733–2737. [Google Scholar]
- McNeely, T.B.; Staub, J.M.; Rusk, C.M.; Blum, M.J.; Donnelly, J.J. Antibody responses to capsular polysaccharide backbone and O-acetate side groups of Streptococcus pneumoniae type 9V in humans and rhesus macaques. Infect. Immun. 1998, 66, 3705–3710. [Google Scholar] [PubMed]
- Nelson, S.; Happel, K.I.; Zhang, P.; Myers, L.; Dufour, J.P.; Bagby, G.J. Effect of bacterial pneumonia on lung simian immunodeficiency virus (SIV) replication in alcohol consuming SIV-infected rhesus macaques. Alcohol Clin. Exp. Res. 2013, 37, 969–977. [Google Scholar] [CrossRef]
- Berendt, R.F.; Long, G.G.; Walker, J.S. Influenza alone and in sequence with pneumonia due to Streptococcus pneumoniae in the squirrel monkey. J. Infect. Dis. 1975, 132, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Philipp, M.T.; Purcell, J.E.; Martin, D.S.; Buck, W.R.; Plauché, G.B.; Ribka, E.P.; DeNoel, P.; Hermand, P.; Leiva, L.E.; Bagby, G.J.; et al. Experimental infection of rhesus macaques with Streptococcus pneumoniae: A possible model for vaccine assessment. J. Med. Primatol. 2006, 35, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Kraft, B.D.; Piantadosi, C.A.; Benjamin, A.M.; Lucas, J.E.; Zaas, A.K.; Betancourt-Quiroz, M.; Woods, C.W.; Chang, A.L.; Roggli, V.L.; Marshall, C.D.; et al. Development of a novel preclinical model of pneumococcal pneumonia in nonhuman primates. Am. J. Respir. Cell Mol. Biol. 2014, 50, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Reyes, L.F.; Restrepo, M.I.; Hinojosa, C.A.; Soni, N.J.; Shenoy, A.T.; Gilley, R.P.; Gonzalez-Juarbe, N.; Noda, J.R.; Winter, V.T.; de la Garza, M.A.; et al. A Non-Human Primate Model of Severe Pneumococcal Pneumonia. PLoS ONE 2016, 11, e0166092. [Google Scholar] [CrossRef] [PubMed]
- Jones, E.E.; Alford, P.L.; Reingold, A.L.; Russell, H.; Keeling, M.E.; Broome, C.V. Predisposition to invasive pneumococcal illness following parainfluenza type 3 virus infection in chimpanzees. J. Am. Vet. Med. Assoc. 1984, 185, 1351–1353. [Google Scholar]
- Szentiks, C.A.; Köndgen, S.; Silinski, S.; Speck, S.; Leendertz, F.H. Lethal pneumonia in a captive juvenile chimpanzee (Pan troglodytes) due to human-transmitted human respiratory syncytial virus (HRSV) and infection with Streptococcus pneumoniae. J. Med. Primatol. 2009, 38, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Reyes, L.F.; Restrepo, M.I.; Hinojosa, C.A.; Soni, N.J.; Anzueto, A.; Babu, B.L.; Gonzalez-Juarbe, N.; Rodriguez, A.H.; Jimenez, A.; Chalmers, J.D.; et al. Severe Pneumococcal Pneumonia Causes Acute Cardiac Toxicity and Subsequent Cardiac Remodeling. Am. J. Respir. Crit. Care Med. 2017, 196, 609–620. [Google Scholar] [CrossRef]
- Dalli, J.; Kraft, B.D.; Colas, R.A.; Shinohara, M.; Fredenburgh, L.E.; Hess, D.R.; Chiang, N.; Welty-Wolf, K.; Choi, A.M.; Piantadosi, C.A.; et al. The Regulation of Proresolving Lipid Mediator Profiles in Baboon Pneumonia by Inhaled Carbon Monoxide. Am. J. Respir. Cell Mol. Biol. 2015, 53, 314–325. [Google Scholar] [CrossRef]
- Shinohara, M.; Kibi, M.; Riley, I.R.; Chiang, N.; Dalli, J.; Kraft, B.D.; Piantadosi, C.A.; Choi, A.M.; Serhan, C.N. Cell-cell interactions and bronchoconstrictor eicosanoid reduction with inhaled carbon monoxide and resolvin D1. Am. J. Physiol Lung Cell Mol. Physiol. 2014, 307, L746–L757. [Google Scholar] [CrossRef]
- Fredenburgh, L.E.; Kraft, B.D.; Hess, D.R.; Harris, R.S.; Wolf, M.A.; Suliman, H.B.; Roggli, V.L.; Davies, J.D.; Winkler, T.; Stenzler, A.; et al. Effects of inhaled CO administration on acute lung injury in baboons with pneumococcal pneumonia. Am. J. Physiol Lung Cell Mol. Physiol. 2015, 309, L834–L846. [Google Scholar] [CrossRef]
| Clinical presentation |
|
| Complications |
|
| Physical exam |
|
| Laboratory findings |
|
| Imaging (CXR, CT, lung US) |
|
| Purpose of the Model | Specific Aim | References |
| TREATMENT | Antibiotic efficacy testing | [13,25,26,27,34,35,36,37,38,40,46,48,52,53,54] |
| PREVENTION | Vaccine efficacy testing | [23,32] |
| PATHOGENESIS | Bacterial capsule as virulence factor | [12,18,19,20] |
| Virulence factors other than capsule | [11,16,17] | |
| Host defence mechanisms against pneumococcal infection | [29,30,31,33,39,41,42,43,44,49,50,55,56,57,58,60,61,65,66,73] | |
| Role of airway obstruction in pathogenesis | [71] | |
| Mechanisms of tissue damage in pneumococcal pneumonia | [29,47] |
| Type of Model | ||||
|---|---|---|---|---|
| Purpose of Model | Rat | Rabbit | Pig | NHP |
| TREATMENT [References] | Efficacy of new antibiotics [72,76,83,85,89,90] | Evaluate pharmacokinetic and efficacy of different drugs [101,105,106,107,108] | Development of directed therapies against pneumococcus [125,126,127] | Test new drugs for ARDS [139,140,141] |
| Test new drugs for sepsis [82,84,93,94] | Test new drugs in pneumococcal sepsis [113] | Therapies against inflammatory complications [125,126,127] | ||
| PREVENTION [References] | Test vaccination efficacy and safety [95] | Development of vaccines [114,125,126,127] | Test vaccination efficacy and safety [136,137,138] | |
| PATHOGENESIS [References] | Impaired phagocytosis [76,77,78,79,80] | Measure inoculum size [99,100,101,102] | Evaluate VAP [114,119,120,121,122,123,124] | Pneumococcal colonization [128] |
| Effect of pneumolysin [77,78,79,80,81] | Effect of pneumoccoccal cell surface components [99,103] | Assess pneumococcal invasive disease [125,126,127] | Assess pneumococcal components [129,130,131] | |
| Measure inoculum size [82,83] | Role of the platelet-activating factor on lung inflammation [99,103] | Evaluate oropharyngeal colonization [125,126,127] | Measure inoculum size [134,135] | |
| Measure inflammatory levels in the lungs [82,84] | Role of host oligosaccharides [104] | Evaluate pneumococcal transmission [125,126,127] | Assess severity of illness [132,133] | |
| Assess virulence of different serotypes [78,79,83] | Assess different pneumococcal serotypes [18,101,105,106] | Evaluate platelet activation and hyperreactivity reactions [125,126,127] | ||
| Evaluate pneumonia in immunocompromised host [76,85,86,87,88] | Pneumococcal sepsis [108,109,110,111,112] | |||
| Pneumococcal sepsis [76,88,91,92] | ||||
| Evaluate pneumococcal complications [83] | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borsa, N.; Di Pasquale, M.; Restrepo, M.I. Animal Models of Pneumococcal pneumonia. Int. J. Mol. Sci. 2019, 20, 4220. https://doi.org/10.3390/ijms20174220
Borsa N, Di Pasquale M, Restrepo MI. Animal Models of Pneumococcal pneumonia. International Journal of Molecular Sciences. 2019; 20(17):4220. https://doi.org/10.3390/ijms20174220
Chicago/Turabian StyleBorsa, Noemi, Marta Di Pasquale, and Marcos I. Restrepo. 2019. "Animal Models of Pneumococcal pneumonia" International Journal of Molecular Sciences 20, no. 17: 4220. https://doi.org/10.3390/ijms20174220
APA StyleBorsa, N., Di Pasquale, M., & Restrepo, M. I. (2019). Animal Models of Pneumococcal pneumonia. International Journal of Molecular Sciences, 20(17), 4220. https://doi.org/10.3390/ijms20174220