Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants
Abstract
1. Introduction
2. The Influence of Drought Stress on AMF Diversity and Growth
3. AMF and Host Plant Association During Drought Stress
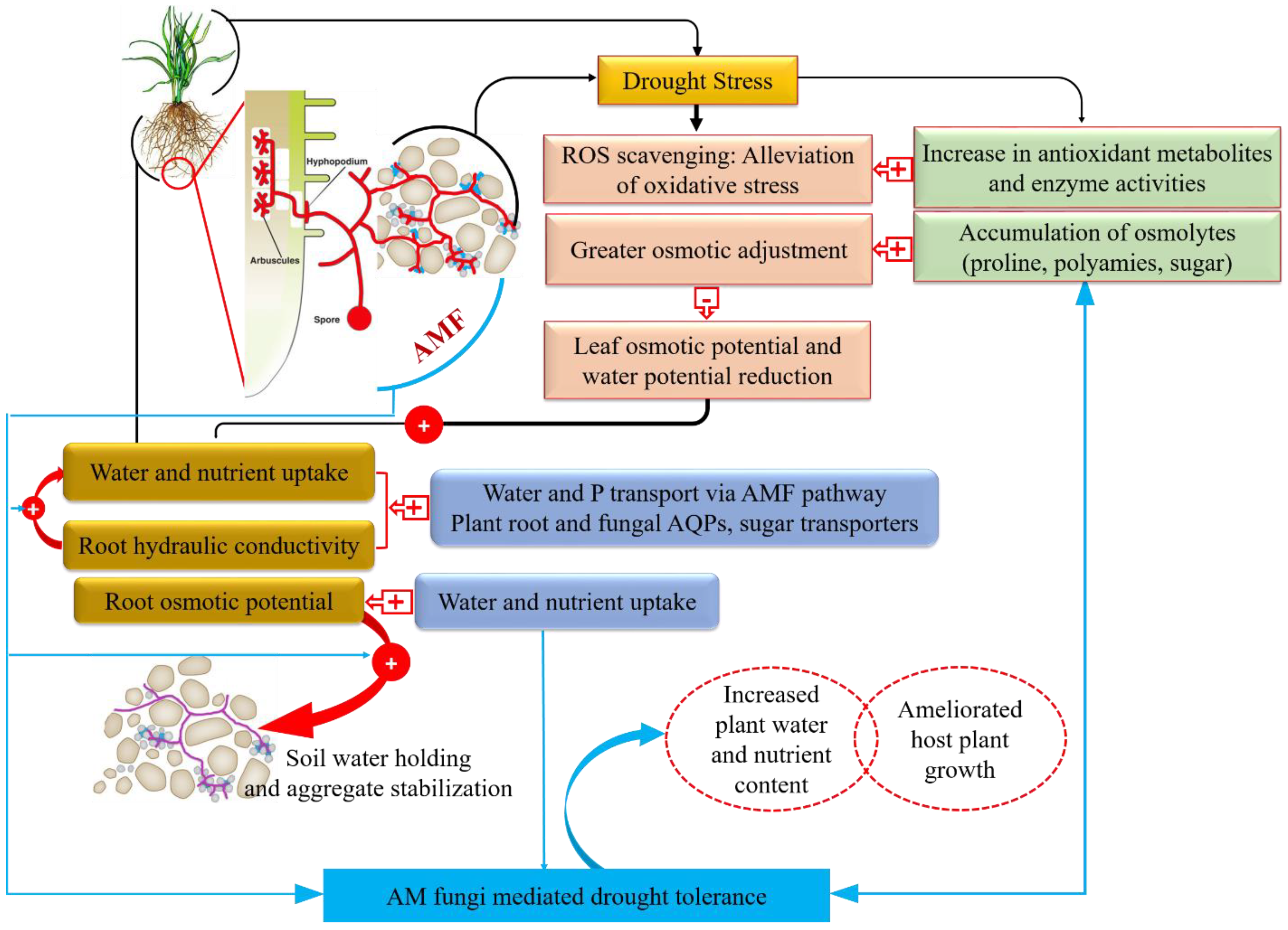
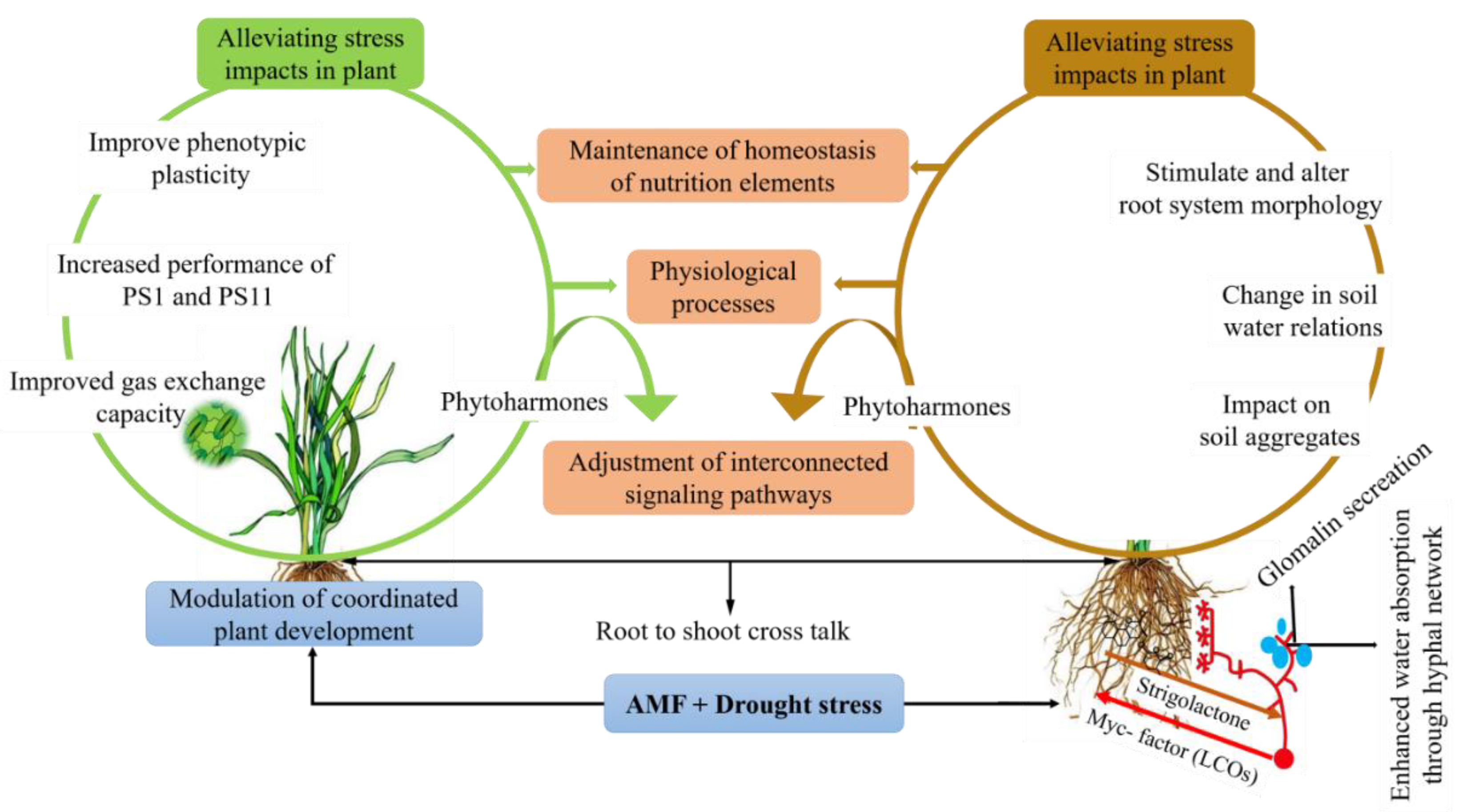
4. Mechanisms of AMF-Mediated Drought Stress Tolerance
4.1. AMF-Assisted Drought Stress Tolerance at Biochemical Level
4.2. AMF-Mediated Drought Stress Tolerance at Morphological Level
4.3. Fungal Water Absorption and Transport Against Drought Stress in AMF Association
5. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhang, F.; Zou, Y.N.; Wu, Q.S. Quantitative estimation of water uptake by mycorrhizal extraradical hyphae in citrus under drought stress. Sci. Hortic. 2018, 229, 132–136. [Google Scholar] [CrossRef]
- Parent, B.; Hachez, C.; Redondo, E.; Simonneau, T.; Chaumont, F.; Tardieu, F. Drought and abscisic acid effects on aquaporin content translate into changes in hydraulic conductivity and leaf growth rate: a trans-scale approach. Plant Physiol. 2009, 149, 2000–2012. [Google Scholar] [CrossRef] [PubMed]
- Candar-Cakir, B.; Arican, E.; Zhang, B. Small RNA and degradome deep sequencing reveals drought-and tissue-specific micrornas and their important roles in drought-sensitive and drought-tolerant tomato genotypes. Plant Biotechnol. J. 2016, 14, 1727–1746. [Google Scholar] [CrossRef] [PubMed]
- Batool, A.; Akram, N.A.; Cheng, Z.G.; Lv, G.C.; Ashraf, M.; Afzal, M.; Xiong, J.L.; Wang, J.Y.; Xiong, Y.C. Physiological and biochemical responses of two spring wheat genotypes to non-hydraulic root-to-shoot signalling of partial and full root-zone drought stress. Plant Physiol. Biochem. 2019, 139, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Selmar, D.; Kleinwächter, M. Influencing the product quality by deliberately applying drought stress during the cultivation of medicinal plants. Ind. Crops Prod. 2013, 42, 558–566. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Aroca, R.; Zamarreno, A.M.; Molina, S.; Andreo-Jimenez, B.; Porcel, R.; Garcia-Mina, J.M.; Ruyter-Spira, C.; Lopez-Raez, J.A. Arbuscular mycorrhizal symbiosis induces strigolactone biosynthesis under drought and improves drought tolerance in lettuce and tomato. Plant Cell Environ. 2016, 39, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Hao, Z.; Zhou, X.; Jiang, X.; Xu, L.; Wu, S.; Zhao, A.; Zhang, X.; Chen, B. Arbuscular mycorrhiza facilitates the accumulation of glycyrrhizin and liquiritin in Glycyrrhiza uralensis under drought stress. Mycorrhiza 2018, 28, 285–300. [Google Scholar] [CrossRef]
- Atkinson, N.J.; Urwin, P.E. The interaction of plant biotic and abiotic stresses: From genes to the field. J. Exp. Bot. 2012, 63, 3523–3544. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Aroca, R. Modulation of aquaporin genes by the arbuscular mycorrhizal symbiosis in relation to osmotic stress tolerance. In Symbioses and Stress; Springer: Dordrecht, The Netherlands, 2010; Volume 17, pp. 357–374. [Google Scholar]
- Saia, S.; Amato, G.; Frenda, A.S.; Giambalvo, D.; Ruisi, P. Influence of arbuscular mycorrhizae on biomass production and nitrogen fixation of berseem clover plants subjected to water stress. PLoS ONE 2014, 9, e90738. [Google Scholar] [CrossRef]
- Latef, A.A.H.A.; Hashem, A.; Rasool, S.; Abd_Allah, E.F.; Alqarawi, A.A.; Egamberdieva, D.; Jan, S.; Anjum, N.A.; Ahmad, P. Arbuscular mycorrhizal symbiosis and abiotic stress in plants: A review. J. Plant Biol. 2016, 59, 407–426. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008. [Google Scholar]
- Auge, R.M.; Toler, H.D.; Saxton, A.M. Arbuscular mycorrhizal symbiosis alters stomatal conductance of host plants more under drought than under amply watered conditions: A meta-analysis. Mycorrhiza 2015, 25, 13–24. [Google Scholar] [CrossRef]
- Wu, Q.S.; Xia, R.X.; Zou, Y.N. Reactive oxygen metabolism in mycorrhizal and non-mycorrhizal citrus (Poncirus trifoliata) seedlings subjected to water stress. J. Plant Physiol. 2006, 163, 1101–1110. [Google Scholar] [CrossRef]
- Ruiz-Sanchez, M.; Armada, E.; Munoz, Y.; Garcia de Salamone, I.E.; Aroca, R.; Ruiz-Lozano, J.M.; Azcon, R. Azospirillum and arbuscular mycorrhizal colonization enhance rice growth and physiological traits under well-watered and drought conditions. J. Plant Physiol. 2011, 168, 1031–1037. [Google Scholar] [CrossRef]
- Sanchez-Blanco, M.J.; Ferrandez, T.; Morales, M.A.; Morte, A.; Alarcon, J.J. Variations in water status, gas exchange, and growth in Rosmarinus officinalis plants infected with Glomus deserticola under drought conditions. J. Plant Physiol. 2004, 161, 675–682. [Google Scholar] [CrossRef]
- Chitarra, W.; Pagliarani, C.; Maserti, B.; Lumini, E.; Siciliano, I.; Cascone, P.; Schubert, A.; Gambino, G.; Balestrini, R.; Guerrieri, E. Insights on the impact of arbuscular mycorrhizal symbiosis on tomato tolerance to water stress. Plant Physiol. 2016, 171, 1009–1023. [Google Scholar] [CrossRef]
- Fernández-Lizarazo, J.C.; Moreno-Fonseca, L.P. Mechanisms for tolerance to water-deficit stress in plants inoculated with arbuscular mycorrhizal fungi. A review. Agron. Colomb. 2016, 34, 179–189. [Google Scholar] [CrossRef]
- Xu, L.; Li, T.; Wu, Z.; Feng, H.; Yu, M.; Zhang, X.; Chen, B. Arbuscular mycorrhiza enhances drought tolerance of tomato plants by regulating the 14-3-3 genes in the ABA signaling pathway. Appl. Soil Ecol. 2018, 125, 213–221. [Google Scholar] [CrossRef]
- Bahadur, A.; Jin, Z.; Jiang, S.; Chai, Y.; Zhang, Q.; Pan, J.; Liu, Y.; Feng, H. Arbuscular mycorrhizal spores distribution across different ecosystems of Qinghai Tibetan Plateau. Pak. J. Bot. 2019, 51, 1481–1492. [Google Scholar] [CrossRef]
- Opik, M.; Zobel, M.; Cantero, J.J.; Davison, J.; Facelli, J.M.; Hiiesalu, I.; Jairus, T.; Kalwij, J.M.; Koorem, K.; Leal, M.E.; et al. Global sampling of plant roots expands the described molecular diversity of arbuscular mycorrhizal fungi. Mycorrhiza 2013, 23, 411–430. [Google Scholar] [CrossRef]
- Ouledali, S.; Ennajeh, M.; Zrig, A.; Gianinazzi, S.; Khemira, H. Estimating the contribution of arbuscular mycorrhizal fungi to drought tolerance of potted olive trees (Olea europaea). Acta Physiol. Plant 2018, 40, 81. [Google Scholar] [CrossRef]
- Basu, S.; Rabara, R.C.; Negi, S. AMF: The future prospect for sustainable agriculture. Physiol. Mol. Plant Pathol. 2018, 102, 36–45. [Google Scholar] [CrossRef]
- Meddich, A.; Jaiti, F.; Bourzik, W.; Asli, A.E.; Hafidi, M. Use of mycorrhizal fungi as a strategy for improving the drought tolerance in date palm (Phoenix dactylifera). Sci. Hortic. 2015, 192, 468–474. [Google Scholar] [CrossRef]
- Sykorova, Z.; Ineichen, K.; Wiemken, A.; Redecker, D. The cultivation bias: different communities of arbuscular mycorrhizal fungi detected in roots from the field, from bait plants transplanted to the field, and from a greenhouse trap experiment. Mycorrhiza 2007, 18, 1–14. [Google Scholar] [CrossRef]
- Stahl, P.D.; Christensen, M. Population variation in the mycorrhizal fungus Glomus mosseae: Breadth of environmental tolerance. Mycol. Res. 1991, 95, 300–307. [Google Scholar] [CrossRef]
- Sylvia, D.M.; Williams, S.E. Vesicualr arbuscular mycorrhizae and environmental stress. In Mycorrhiza in Sustainable Agriculture; Bethlenfalvay, G.T., Linderman, R.D., Eds.; ASA Special Publication: Madison, WI, USA, 1992; pp. 101–124. [Google Scholar]
- ÖPik, M.; Moora, M.; Liira, J.; Zobel, M. Composition of root-colonizing arbuscular mycorrhizal fungal communities in different ecosystems around the globe. J. Ecol. 2006, 94, 778–790. [Google Scholar] [CrossRef]
- Mohammad, M.J.; Hamad, S.R.; Malkawi, H.I. Population of arbuscular mycorrhizal fungi in semi-arid environment of Jordan as influenced by biotic and abiotic factors. J. Arid Environ. 2003, 53, 409–417. [Google Scholar] [CrossRef]
- Panwar, J.; Tarafdar, J.C. Distribution of three endangered medicinal plant species and their colonization with arbuscular mycorrhizal fungi. J. Arid Environ. 2006, 65, 337–350. [Google Scholar] [CrossRef]
- Tian, H.; Gai, J.P.; Zhang, J.L.; Christie, P.; Li, X.L. Arbuscular mycorrhizal fungi associated with wild forage plants in typical steppe of eastern Inner Mongolia. Eur. J. Soil Biol. 2009, 45, 321–327. [Google Scholar] [CrossRef]
- Verma, N.; Tarafdar, J.C.; Srivastava, K.K.; Panwar, J. Arbuscular mycorrhizal (AM) diversity in Prosopis cineraria (L.) Druce under arid agroecosystems. Agric. Sci. China 2008, 7, 754–761. [Google Scholar] [CrossRef]
- Bray, E.A. Plant responses to water deficit. Trends Plant. Sci. 1997, 2, 48–54. [Google Scholar] [CrossRef]
- Manoharan, P.T.; Shanmugaiah, V.; Balasubramanian, N.; Gomathinayagam, S.; Sharma, M.P.; Muthuchelian, K. Influence of AM fungi on the growth and physiological status of Erythrina variegata Linn. grown under different water stress conditions. Eur. J. Soil Biol. 2010, 46, 51–156. [Google Scholar] [CrossRef]
- Fan, Q.J.; Liu, J.H. Colonization with arbuscular mycorrhizal fungus affects growth, drought tolerance and expression of stress-responsive genes in Poncirus trifoliata. Acta Physiol. Plant 2011, 33, 1533–1542. [Google Scholar] [CrossRef]
- Zou, Y.N.; Wang, P.; Liu, C.Y.; Ni, Q.D.; Zhang, D.J.; Wu, Q.S. Mycorrhizal trifoliate orange has greater root adaptation of morphology and phytohormones in response to drought stress. Sci. Rep. 2017, 7, 41134. [Google Scholar] [CrossRef] [PubMed]
- Oldroyd, G.E.D. Speak, friend, and enter: Signalling systems that promote beneficial symbiotic associations in plants. Nat. Rev. Microbiol. 2013, 11, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Verma, J.P. Does plant-microbe interaction confer stress tolerance in plants: A review? Microbiol. Res. 2018, 207, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.H.; Zou, Y.N.; Rahman, M.M.; Ni, Q.D.; Wu, Q.S. Mycorrhizas alter sucrose and proline metabolism in trifoliate orange exposed to drought stress. Sci. Rep. 2017, 7, 42389. [Google Scholar] [CrossRef] [PubMed]
- Barros, V.; Frosi, G.; Santos, M.; Ramos, D.G.; Falcao, H.M.; Santos, M.G. Arbuscular mycorrhizal fungi improve photosynthetic energy use efficiency and decrease foliar construction cost under recurrent water deficit in woody evergreen species. Plant Physiol. Biochem. 2018, 127, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Auge, R.M. Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 2001, 11, 3–42. [Google Scholar] [CrossRef]
- Wu, Q.S.; Zou, Y.N. Arbuscular mycorrhizal fungi and tolerance of drought stress in plants. In Arbuscular Mycorrhizas and Stress Tolerance of Plants; Wu, Q.S., Ed.; Springer: Singapore, 2017; pp. 25–41. [Google Scholar]
- Pavithra, D.; Yapa, N. Arbuscular mycorrhizal fungi inoculation enhances drought stress tolerance of plants. Groundw. Sustain. Develop. 2018, 7, 490–494. [Google Scholar] [CrossRef]
- Kapoor, R.; Evelin, H.; Mathur, P.; Giri, B. Arbuscular mycorrhiza: Approaches for abiotic stress tolerance in crop plants for sustainable agriculture. In Plant Acclimation to Environmental Stress; Tuteja, N., Gill, S.S., Eds.; Springer: New York, NY, USA, 2013; pp. 359–401. [Google Scholar]
- Gong, M.; Tang, M.; Chen, H.; Zhang, Q.; Feng, X. Effects of two Glomus species on the growth and physiological performance of Sophora davidii seedlings under water stress. New For. 2012, 44, 399–408. [Google Scholar] [CrossRef]
- Pagano, M.C. Drought stress and mycorrhizal plant. In Use of Microbes for the Alleviation of Soil Stresses; Miransari, M., Ed.; Springer: New York, NY, USA, 2014; Volume 1, pp. 97–110. [Google Scholar]
- Lee, B.R.; Muneer, S.; Avice, J.C.; Jung, W.J.; Kim, T.H. Mycorrhizal colonisation and P-supplement effects on N uptake and N assimilation in perennial ryegrass under well-watered and drought-stressed conditions. Mycorrhiza 2012, 22, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Gholamhoseini, M.; Ghalavand, A.; Dolatabadian, A.; Jamshidi, E.; Khodaei-Joghan, A. Effects of arbuscular mycorrhizal inoculation on growth, yield, nutrient uptake and irrigation water productivity of sunflowers grown under drought stress. Agr. Water Manag. 2013, 117, 106–114. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Gómez, M.; Azcón, R. Influence of different Glomus species on the time course of physiological plant responses of lettuce to progressive drought stress periods. Plant Sci. 1995, 110, 37–44. [Google Scholar] [CrossRef]
- Doubková, P.; Vlasáková, E.; Sudová, R. Arbuscular mycorrhizal symbiosis alleviates drought stress imposed on Knautia arvensis plants in serpentine soil. Plant Soil 2013, 370, 149–161. [Google Scholar] [CrossRef]
- Bahmani, M.; Naghdi, R.; Kartoolinejad, D. Milkweed seedlings tolerance against water stress: Comparison of inoculations with Rhizophagus irregularis and Pseudomonas putida. Environ. Technol. 2018, 10, 111–121. [Google Scholar] [CrossRef]
- Yooyongwech, S.; Samphumphuang, T.; Tisarum, R.; Theerawitaya, C.; Chaum, S. Arbuscular mycorrhizal fungi (AMF) improved water deficit tolerance in two different sweet potato genotypes involves osmotic adjustments via soluble sugar and free proline. Sci. Hortic. 2016, 198, 107–117. [Google Scholar] [CrossRef]
- Amiri, R.; Nikbakht, A.; Etemadi, N. Alleviation of drought stress on rose geranium [Pelargonium graveolens (L.) Herit.] in terms of antioxidant activity and secondary metabolites by mycorrhizal inoculation. Sci. Hortic. 2015, 197, 373–380. [Google Scholar] [CrossRef]
- Huang, Y.M.; Zou, Y.N.; Wu, Q.S. Alleviation of drought stress by mycorrhizas is related to increased root H2O2 efflux in trifoliate orange. Sci. Rep. 2017, 7, 42335. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Y.; Srivastava, A.K.; Wu, Q.S. Mycorrhizal fungi regulate root responses and leaf physiological activities in Trifoliate orange. Not. Bot. Horti Agrobot. Cluj Napoca 2017, 45, 17–21. [Google Scholar] [CrossRef][Green Version]
- Zhao, R.; Guo, W.; Bi, N.; Guo, J.; Wang, L.; Zhao, J.; Zhang, J. Arbuscular mycorrhizal fungi affect the growth, nutrient uptake and water status of maize (Zea mays L.) grown in two types of coal mine spoils under drought stress. Appl. Soil Ecol. 2015, 88, 41–49. [Google Scholar] [CrossRef]
- Quiroga, G.; Erice, G.; Aroca, R.; Chaumont, F.; Ruiz-Lozano, J.M. Enhanced drought stress tolerance by the arbuscular mycorrhizal symbiosis in a drought-sensitive maize cultivar is related to a broader and differential regulation of host plant aquaporins than in a drought-tolerant cultivar. Front. Plant Sci. 2017, 8, 1056. [Google Scholar] [CrossRef]
- Wu, Q.S.; Srivastava, A.K.; Zou, Y.N. AMF-induced tolerance to drought stress in citrus: A review. Sci. Hortic. 2013, 164, 77–87. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M. Arbuscular mycorrhizal symbiosis and alleviation of osmotic stress. New perspectives for molecular studies. Mycorrhiza 2003, 13, 309–317. [Google Scholar] [CrossRef]
- Smith, S.E.; Facelli, E.; Pope, S.; Smith, F.A. Plant performance in stressful environments: interpreting new and established knowledge of the roles of arbuscular mycorrhizas. Plant Soil 2010, 326, 3–20. [Google Scholar] [CrossRef]
- Demidchik, V. Mechanisms of oxidative stress in plants: From classical chemistry to cell biology. Environ. Exp. Bot. 2015, 109, 212–228. [Google Scholar] [CrossRef]
- Fobert, P.R.; Despres, C. Redox control of systemic acquired resistance. Curr. Opin. Plant Biol. 2005, 8, 378–382. [Google Scholar] [CrossRef]
- Rhoads, D.M.; Umbach, A.L.; Subbaiah, C.C.; Siedow, J.N. Mitochondrial reactive oxygen species. Contribution to oxidative stress and interorganellar signaling. Plant Physiol. 2006, 141, 357–366. [Google Scholar] [CrossRef]
- Abbaspour, H.; Saeidi-Sar, S.; Afshari, H.; Abdel-Wahhab, M.A. Tolerance of Mycorrhiza infected pistachio (Pistacia vera L.) seedling to drought stress under glasshouse conditions. J. Plant Physiol. 2012, 169, 704–709. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 1360–1385. [Google Scholar] [CrossRef]
- Zou, Y.N.; Huang, Y.M.; Wu, Q.S.; He, X.H. Mycorrhiza-induced lower oxidative burst is related with higher antioxidant enzyme activities, net H2O2 effluxes, and Ca2+ influxes in trifoliate orange roots under drought stress. Mycorrhiza 2015, 25, 143–152. [Google Scholar] [CrossRef]
- Latef, A.A.H.A.; Chaoxing, H. Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Sci. Hortic. 2011, 127, 228–233. [Google Scholar] [CrossRef]
- Chang, M.; Liao, L.; Lin, J.; Liu, Z.; Hsu, Y.; Lee, T. Modulation of antioxidant defense system and NADPH oxidase in Pluchea indica leaves by water deficit stress. Bot. Stud. 2012, 53, 45–54. [Google Scholar]
- Essahibi, A.; Benhiba, L.; Babram, M.A.; Ghoulam, C.; Qaddoury, A. Influence of arbuscular mycorrhizal fungi on the functional mechanisms associated with drought tolerance in carob (Ceratonia siliqua L.). Trees 2017, 32, 87–97. [Google Scholar] [CrossRef]
- Barzana, G.; Aroca, R.; Ruiz-Lozano, J.M. Localized and non-localized effects of arbuscular mycorrhizal symbiosis on accumulation of osmolytes and aquaporins and on antioxidant systems in maize plants subjected to total or partial root drying. Plant Cell Environ. 2015, 38, 1613–1627. [Google Scholar] [CrossRef]
- Marulanda, A.; Porcel, R.; Barea, J.M.; Azcon, R. Drought tolerance and antioxidant activities in lavender plants colonized by native drought-tolerant or drought-sensitive Glomus Species. Microb. Ecol. 2007, 54, 543–552. [Google Scholar] [CrossRef]
- Miransari, M.; Abrishamchi, A.; Khoshbakht, K.; Niknam, V. Plant hormones as signals in arbuscular mycorrhizal symbiosis. Crit. Rev. Biotechnol. 2014, 34, 123–133. [Google Scholar] [CrossRef]
- Miransari, M. Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant Biol. 2010, 12, 563–569. [Google Scholar] [CrossRef]
- Lopez-Raez, J.A. How drought and salinity affect arbuscular mycorrhizal symbiosis and strigolactone biosynthesis? Planta 2016, 243, 1375–1385. [Google Scholar] [CrossRef]
- Pandey, A.; Sharma, M.; Pandey, G.K. Emerging roles of strigolactones in plant responses to stress and development. Front. Plant Sci. 2016, 7, 434. [Google Scholar] [CrossRef]
- Chabaud, M.; Genre, A.; Sieberer, B.J.; Faccio, A.; Fournier, J.; Novero, M.; Barker, D.G.; Bonfante, P. Arbuscular mycorrhizal hyphopodia and germinated spore exudates trigger Ca2+ spiking in the legume and nonlegume root epidermis. New Phytol. 2011, 189, 347–355. [Google Scholar] [CrossRef]
- Lopez-Obando, M.; Ligerot, Y.; Bonhomme, S.; Boyer, F.D.; Rameau, C. Strigolactone biosynthesis and signaling in plant development. Development 2015, 142, 3615–3619. [Google Scholar] [CrossRef]
- Kretzschmar, T.; Kohlen, W.; Sasse, J.; Borghi, L.; Schlegel, M.; Bachelier, J.B.; Reinhardt, D.; Bours, R.; Bouwmeester, H.J.; Martinoia, E. A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 2012, 483, 341–344. [Google Scholar] [CrossRef]
- Mori, N.; Nishiuma, K.; Sugiyama, T.; Hayashi, H.; Akiyama, K. Carlactone-type strigolactones and their synthetic analogues as inducers of hyphal branching in arbuscular mycorrhizal fungi. Phytochemistry 2016, 130, 90–98. [Google Scholar] [CrossRef]
- Li, T.; Sun, Y.; Ruan, Y.; Xu, L.; Hu, Y.; Hao, Z.; Zhang, X.; Li, H.; Wang, Y.; Yang, L.; et al. Potential role of D-myo-inositol-3-phosphate synthase and 14-3-3 genes in the crosstalk between Zea mays and Rhizophagus intraradices under drought stress. Mycorrhiza 2016, 26, 879–893. [Google Scholar] [CrossRef]
- Calvo-Polanco, M.; Sánchez-Romera, B.; Aroca, R. Arbuscular mycorrhizal fungi and the tolerance of plants to drought and salinity. In Symbiotic Endophytes; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; Volume 37, pp. 271–288. [Google Scholar]
- Hong, J.H.; Seah, S.W.; Xu, J. The root of ABA action in environmental stress response. Plant Cell Rep. 2013, 32, 971–983. [Google Scholar] [CrossRef]
- Martin-Rodriguez, J.A.; Huertas, R.; Ho-Plagaro, T.; Ocampo, J.A.; Tureckova, V.; Tarkowska, D.; Ludwig-Muller, J.; Garcia-Garrido, J.M. Gibberellin-abscisic acid balances during arbuscular mycorrhiza formation in tomato. Front. Plant Sci. 2016, 7, 1273. [Google Scholar] [CrossRef]
- Herrera-Medina, M.J.; Steinkellner, S.; Vierheilig, H.; Bote, J.A.O.; Garcia Garrido, J.M. Abscisic acid determines arbuscule development and functionality in the tomato arbuscular mycorrhiza. New Phytol. 2007, 175, 554–564. [Google Scholar] [CrossRef]
- Pozo, M.J.; Lopez-Raez, J.A.; Azcon-Aguilar, C.; Garcia-Garrido, J.M. Phytohormones as integrators of environmental signals in the regulation of mycorrhizal symbioses. New Phytol. 2015, 205, 1431–1436. [Google Scholar] [CrossRef]
- Esch, H.; Hundeshagen, B.; Sehneider-Poetsch, H.; Bothe, H. Demonstration of abscisic acid in spores and hyphae of the arbuscular-mycorrhizal fungus Glomus and in the NE-fixing cyanobacterium Anabaena variabilis. Plant Sci. 1994, 99, 9–16. [Google Scholar] [CrossRef]
- Danneberg, G.; Latus, C.; Zimmer, W.; Hundeshagen, B.; Schneider-Poetsch, H.; Bothe, H. Influence of vesicular-arbuscular mycorrhiza on phytohormone balances in maize (Zea mays L.). J. Plant Physiol. 1993, 141, 33–39. [Google Scholar] [CrossRef]
- Bheemareddy, V.S.; Lakshman, H.C. Effect of AM fungus Glomus fasciculatumon metabolite accumulation in four varieties of Triticum aestivum L. under short-term water stress. Vegetos 2011, 24, 41–49. [Google Scholar]
- Yooyongwech, S.; Phaukinsang, N.; Cha-um, S.; Supaibulwatana, K. Arbuscular mycorrhiza improved growth performance in Macadamia tetraphylla L. grown under water deficit stress involves soluble sugar and proline accumulation. Plant Growth Regul. 2013, 69, 285–293. [Google Scholar] [CrossRef]
- Zhang, B.; Chang, S.X.; Anyia, A.O. Mycorrhizal inoculation and nitrogen fertilization affect the physiology and growth of spring wheat under two contrasting water regimes. Plant Soil 2015, 398, 47–57. [Google Scholar] [CrossRef]
- Goicoechea, N.; Szalai, G.; Antolín, M.C.; Sánchez-Díaz, M.; Paldi, E. Influence of arbuscular mycorrhizae and Rhizobium on free polyamines and proline levels in water-stressed alfalfa. J. Plant Physiol. 1998, 153, 706–711. [Google Scholar] [CrossRef]
- Chaves, M.; Maroco, J.; Pereira, J. Understanding plant responses to drought- from genes to the whole plant. Funct. Plant Biol. 2003, 30, 239–264. [Google Scholar] [CrossRef]
- Bakr, J.; Pék, Z.; Helyes, L.; Posta, K. Mycorrhizal inoculation alleviates water deficit impact on field-grown processing tomato. Pol. J. Environ. Stud. 2018, 27, 1949–1958. [Google Scholar] [CrossRef]
- Porcel, R.; Azcón, R.; Ruiz-Lozano, J.M. Evaluation of the role of genes encoding for Δ1-pyrroline-5-carboxylate synthetase (P5CS) during drought stress in arbuscular mycorrhizal Glycine max and Lactuca sativa plants. Physiol. Mol. Plant Pathol. 2004, 65, 211–221. [Google Scholar] [CrossRef]
- Zhang, H.H.; Tang, M.; Chen, H.; Zheng, C.L.; Niu, Z.C. Effect of inoculation with AM fungi on lead uptake, translocation and stress alleviation of Zea mays L. seedlings planting in soil with increasing lead concentrations. Eur. J. Soil Biol. 2010, 46, 306–311. [Google Scholar] [CrossRef]
- Finlay, R.; Söderström, B. Mycorrhiza and carbon flow to the soil. In Mycorrhizal Functioning: An Integrative Plant-Fungal Process; Allen, M., Ed.; Chapman and Hall: New York, NY, USA, 1992; pp. 134–160. [Google Scholar]
- Koyro, H.W.; Ahmad, P.; Geissler, N. Abiotic Stress Responses in Plants: An Overview; Springer: New York, NY, USA, 2012; pp. 1–28. [Google Scholar]
- De Carvalho, K.; de Campos, M.K.; Domingues, D.S.; Pereira, L.F.; Vieira, L.G. The accumulation of endogenous proline induces changes in gene expression of several antioxidant enzymes in leaves of transgenic Swingle citrumelo. Mol. Biol. Rep. 2013, 40, 3269–3279. [Google Scholar] [CrossRef]
- Asrar, A.A.; Abdel-Fattah, G.M.; Elhindi, K.M. Improving growth, flower yield, and water relations of snapdragon (Antirhinum majus L.) plants grown under well-watered and water-stress conditions using arbuscular mycorrhizal fungi. Photosynthetica 2012, 50, 305–316. [Google Scholar] [CrossRef]
- Zou, Y.N.; Wu, Q.S.; Huang, Y.M.; Ni, Q.D.; He, X.H. Mycorrhizal-mediated lower proline accumulation in Poncirus trifoliata under water deficit derives from the integration of inhibition of proline synthesis with increase of proline degradation. PLoS ONE 2013, 8, e80568. [Google Scholar] [CrossRef]
- Rapparini, F.; Peñuelas, J. Mycorrhizal fungi to alleviate drought stress on plant growth. In Use of Microbes for the Alleviation of Soil Stresses; Miransari, M., Ed.; Springer: New York, NY, USA, 2014; pp. 21–42. [Google Scholar]
- Nasim, G. The role of arbuscualr mycorrhizae in inducing resistance to drought and salinity stress in crops. In Plant Adaptation and Phytoremediation; Ashraf, M., Ozturk, M., Ahmad, M.S.A., Eds.; Springer: Dordrecht, The Netherlands, 2010; pp. 119–141. [Google Scholar]
- Sylvia, D.M.; Schenck, N.C. Application of superphosphate to mycorrhizal plants stimulates sporulation of phosphorus-tolerant vesicular-arbuscular mycorrhizal fungi. New Phytol. 1983, 95, 655–661. [Google Scholar] [CrossRef]
- Douds, D.D.; Schenck, N.S. Germination and hyphal growth of Vam fungi during and after storage in soil at five matric potentials. Soil Biol. Biochem. 1991, 33, 177–183. [Google Scholar] [CrossRef]
- Liu, T.; Sheng, M.; Wang, C.Y.; Chen, H.; Li, Z.; Tang, M. Impact of arbuscular mycorrhizal fungi on the growth, water status, and photosynthesis of hybrid poplar under drought stress and recovery. Photosynthetica 2015, 53, 250–258. [Google Scholar] [CrossRef]
- Zhu, X.C.; Song, F.B.; Liu, S.Q.; Liu, T.D.; Zhou, X. Arbuscular mycorrhizae improves photosynthesis and water status of Zea mays L. under drought stress. Plant Soil Environ. 2012, 58, 186–191. [Google Scholar] [CrossRef]
- Avio, L.; Pellegrino, E.; Bonari, E.; Giovannetti, M. Functional diversity of arbuscular mycorrhizal fungal isolates in relation to extraradical mycelial networks. New Phytol. 2006, 172, 347–357. [Google Scholar] [CrossRef]
- de la Providencia, I.E.; de Souza, F.A.; Fernandez, F.; Delmas, N.S.; Declerck, S. Arbuscular mycorrhizal fungi reveal distinct patterns of anastomosis formation and hyphal healing mechanisms between different phylogenic groups. New Phytol. 2005, 165, 261–271. [Google Scholar] [CrossRef]
- Enkhtuya, B.; Rydlová, J.; Vosátka, M. Effectiveness of indigenous and nonindigenous isolates of arbuscular mycorrhizal fungi in soils from degraded ecosystems and man-made habitats. Appl. Soil Ecol. 2002, 14, 201–211. [Google Scholar] [CrossRef]
- Lenoir, I.; Fontaine, J.; Lounes-Hadj Sahraoui, A. Arbuscular mycorrhizal fungal responses to abiotic stresses: A review. Phytochemistry 2016, 123, 4–15. [Google Scholar] [CrossRef]
- Huang, Z.; Zou, Z.; He, C.; He, Z.; Zhang, Z.; Li, J. Physiological and photosynthetic responses of melon (Cucumis melo L.) seedlings to three Glomus species under water deficit. Plant Soil 2010, 339, 391–399. [Google Scholar] [CrossRef]
- Estaun, M.V. Effect of sodium chloride and mannitol on germination and hyphal growth of the vesicular-arbuscular mycorrhizal fungus Glomus mosseae. Agric. Ecosyst. Environ. 1989, 29, 123–129. [Google Scholar] [CrossRef]
- Debiane, D.; Garçon, G.; Verdin, A.; Fontaine, J.; Durand, R.; Shirali, P.; Grandmougin-Ferjani, A.; Lounès-Hadj Sahraoui, A. Mycorrhization alleviates benzo[a]pyrene-induced oxidative stress in an in vitro chicory root model. Phytochemistry 2009, 70, 1421–1427. [Google Scholar] [CrossRef]
- Nehls, U.; Dietz, S. Fungal aquaporins: Cellular functions and ecophysiological perspectives. Appl. Microbiol. Biotechnol. 2014, 98, 8835–8851. [Google Scholar] [CrossRef]
- Maurel, C.; Verdoucq, L.; Luu, D.T.; Santoni, V. Plant aquaporins: Membrane channels with multiple integrated functions. Annu. Rev. Plant Biol. 2008, 59, 595–624. [Google Scholar] [CrossRef]
- Reuscher, S.; Akiyama, M.; Mori, C.; Aoki, K.; Shibata, D.; Shiratake, K. Genome-wide identification and expression analysis of aquaporins in tomato. PLoS ONE 2013, 8, e79052. [Google Scholar] [CrossRef]
- Marjanovic, Z.; Uehlein, N.; Kaldenhoff, R.; Zwiazek, J.J.; Weiss, M.; Hampp, R.; Nehls, U. Aquaporins in poplar: What a difference a symbiont makes! Planta 2005, 222, 258–268. [Google Scholar] [CrossRef]
- Aroca, R.; Porcel, R.; Ruiz-Lozano, J.M. How does arbuscular mycorrhizal symbiosis regulate root hydraulic properties and plasma membrane aquaporins in Phaseolus vulgaris under drought, cold or salinity stresses? New Phytol. 2007, 173, 808–816. [Google Scholar] [CrossRef]
- Porcel, R.; Aroca, R.; Azcon, R.; Ruiz-Lozano, J.M. PIP aquaporin gene expression in arbuscular mycorrhizal Glycine max and Lactuca sativa plants in relation to drought stress tolerance. Plant Mol. Biol. 2006, 60, 389–404. [Google Scholar] [CrossRef]
- Li, T.; Hu, Y.J.; Hao, Z.P.; Li, H.; Wang, Y.S.; Chen, B.D. First cloning and characterization of two functional aquaporin genes from an arbuscular mycorrhizal fungus Glomus intraradices. New Phytol. 2013, 197, 617–630. [Google Scholar] [CrossRef]
- Li, Y.; Zou, Y.N.; Wu, Q.S. Effects of Diversispora spurca inoculation on growth, root system architecture and chlorophyll contents of four citrus genotype. Int. J. Agric. Biol. 2013, 15, 1814–9596. [Google Scholar]
- Neumann, E.; Schmid, B.; Romheld, V.; George, E. Extraradical development and contribution to plant performance of an arbuscular mycorrhizal symbiosis exposed to complete or partial rootzone drying. Mycorrhiza 2009, 20, 3–23. [Google Scholar] [CrossRef]
- Zou, Y.N.; Srivastava, A.K.; Ni, Q.D.; Wu, Q.S. Disruption of mycorrhizal extraradical mycelium and changes in leaf water status and soil aggregate stability in rootbox-grown trifoliate orange. Front. Microbiol. 2015, 6, 203. [Google Scholar] [CrossRef]
- Aroca, R.; Porcel, R.; Ruiz-Lozano, J.M. Regulation of root water uptake under abiotic stress conditions. J. Exp. Bot. 2012, 63, 43–57. [Google Scholar] [CrossRef]
- Barzana, G.; Aroca, R.; Bienert, G.P.; Chaumont, F.; Ruiz-Lozano, J.M. New insights into the regulation of aquaporins by the arbuscular mycorrhizal symbiosis in maize plants under drought stress and possible implications for plant performance. Mol. Plant Microbe Interact. 2014, 27, 349–363. [Google Scholar] [CrossRef]
- Giovannetti, M.; Balestrini, R.; Volpe, V.; Guether, M.; Straub, D.; Costa, A.; Ludewig, U.; Bonfante, P. Two putative-aquaporin genes are differentially expressed during arbuscular mycorrhizal symbiosis in Lotus japonicus. BMC Plant Biol. 2012, 12, 1–14. [Google Scholar] [CrossRef]
- Jia-Dong, H.; Tao, D.; Hui-Hui, W.; Ying-Ning, Z.; Qiang-Sheng, W.; Kamil, K. Mycorrhizas induce diverse responses of root TIP aquaporin gene expression to drought stress in trifoliate orange. Sci. Hortic. 2019, 243, 64–69. [Google Scholar] [CrossRef]
- Tyerman, S.D.; Bohnert, H.J.; Maurel, C.; Steudle, E.; Smith, J.A.C. Plant aquaporins: Their molecular biology, biophysics and significance for plant water relations. J. Exp. Bot. 1999, 50, 1055–1071. [Google Scholar] [CrossRef]
- Javot, H.; Maurel, C. The role of aquaporins in root water uptake. Ann. Bot. 2002, 90, 301–313. [Google Scholar] [CrossRef]
- Chaumont, F.; Tyerman, S.D. Aquaporins: Highly regulated channels controlling plant water relations. Plant Physiol. 2014, 164, 1600–1618. [Google Scholar] [CrossRef]
- Sanchez-Romera, B.; Ruiz-Lozano, J.M.; Zamarreno, A.M.; Garcia-Mina, J.M.; Aroca, R. Arbuscular mycorrhizal symbiosis and methyl jasmonate avoid the inhibition of root hydraulic conductivity caused by drought. Mycorrhiza 2016, 26, 111–122. [Google Scholar] [CrossRef]
- Grondin, A.; Mauleon, R.; Vadez, V.; Henry, A. Root aquaporins contribute to whole plant water fluxes under drought stress in rice (Oryza sativa L.). Plant Cell Environ. 2016, 39, 347–365. [Google Scholar] [CrossRef]
- He, D.; Xiang, X.; He, J.S.; Wang, C.; Cao, G.; Adams, J.; Chu, H. Composition of the soil fungal community is more sensitive to phosphorus than nitrogen addition in the alpine meadow on the Qinghai-Tibetan Plateau. Biol. Fertil. Soils 2016, 52, 1059–1072. [Google Scholar] [CrossRef]
- Aroca, R.; Bago, A.; Sutka, M.; Paz, J.A.; Cano, C.; Amodeo, G.; Ruiz-Lozano, J.M. Expression analysis of the first arbuscular mycorrhizal fungi aquaporin described reveals concerted gene expression between salt-stressed and nonstressed mycelium. Mol. Plant Microbe Interact. 2009, 22, 1169–1178. [Google Scholar] [CrossRef]
- Yu, Q.; Hu, Y.; Li, J.; Wu, Q.; Lin, Z. Sense and antisense expression of plasma membrane aquaporin BnPIP1 from Brassica napus in tobacco and its effects on plant drought resistance. Plant Sci. 2005, 169, 647–656. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Porcel, R.; Aroca, R. Regulation by arbuscular mycorrhizae of the integrated physiological response to salinity in plants: New challenges in physiological and molecular studies. J. Exp. Bot. 2012, 63, 4033–4044. [Google Scholar] [CrossRef]
| Plant Species | AMF Species | AMF Variables | Plant Variables | Reference | ||
|---|---|---|---|---|---|---|
| Morphological | Physiological | Biochemical | ||||
| Calotropis procera Ait | Glomus intraradices | Col%↑ | N↑, K↑ | CAT↑, POD↑, APX↑, SOD↑ | [52] | |
| Cynophala flexuosa L. | AMF | Col% ns | LRWC↑, PEUE↑ (Cycle 2), leaf construction cost↑ (Cycle 2), SLA↑ (Cycle 1) | [41] | ||
| Glycine max L. | AMF | Col%↑ | Soil moisture (%)↑, LAI↑, growth performance↑ | Pn↑, Leaf proline concentration↓ | [44] | |
| Glycyrrhiza uralensis Fisch. ex DC. | Rhizophagus irregularis | Col%↑, A%↓ | Leaf proline concentrations↑, P↑, C:N↑, Pn↑, WUE↑, C:P↓, N:P↓ | Root ABA↓ | [7] | |
| Ipomoea batatas (L.) Lam. | Commercial inoculum containing Glomus sp. and Acaulospora sp. | Plant growth↑, tubers per plant↑, tuber fresh weight↑ | P content↑, soluble sugars↑, leaf osmotic potential↑, chlorophyll degradation↓, photosynthetic pigments↑, maximum quantum yield of PSII (Fv/Fm)↑, photon yield of PSII (ΦPSII) ↑, net photosynthetic rate↑ | Proline↑ | [53] | |
| Pelargonium graveolens (L.) Herit. | Funneliformis mosseae, Rhizophagus irregularis | Col%↓ | Essential oil content↑, oil yield↑ | MDA↓, H2O2↓, CAT↑, APX↑, SOD↑, GPX↑ | [54] | |
| Phoenix dactylifera L. | Funneliformis monosporum, Rhizophagus clarus, Glomus deserticola ∆ | Plant growth performance↑ | Nutrient absorption↑, RWC↑, water potential↑, stomatal resistance↓ | [25] | ||
| Poncirus trifoliata (L.) | Diversispora versiformis | Col%↓, hyphal length↓ | Plant growth performance↑, root morphology↑ | LWP↓ | IAA↑, MeJA↑, NO↑ | [37] |
| Poncirus trifoliata (L.) | Funneliformis mosseae | Col%↓ | Shoot↑, root↑, total biomass↑, surface area of lateral roots↑ | O2·−↓, H2O2↓, MDA↓ | [55] | |
| Poncirus Trifoliata (L.) | Funneliformis mosseae, Paraglomus occultum | Col%↓ | Plant height↑, stem diameter↑, leaf number↑, leaf, stem, and root dry weight↑ | LRWC↑ | Leaf sucrose↑, glucose↑, fructose↑, leaf proline concentration↓ | [40] |
| Poncirus trifoliate (L.) | Funneliformis mosseae | Col%↓ | Plant height↑, shoot and root biomass↑, root hairs density↑, length↑, diameter↑ | Root IAA↑ | [56] | |
| Poncirus trifoliata (L.) | Funneliformis mosseae, Paraglomus occultum | Col%↓ | Root biomass↑, taproot length↑, number of lateral roots↑ | Root sucrose↓, glucose↑, fructose↑ root sucrose relevant enzymes↑, root proline↓ | [1] | |
| Solanum lycopersicum L. mutant notabilis and its wild-type | Rhizophagus intraradices | Col%↑, Col%↓ | Shoot biomass↑ | Shoot and root P concentrations↑, WUE↑, Tr↑ | ABA↓ | [20] |
| Solanum lycopersicum L. | Funneliformis mosseae, Rhizophagus intraradices | Plant height↑, root fresh weight↓ | Stomatal density↑, WUE↑, Tr↑ | ABA↓, H2O2↓, proline↑ | [18] | |
| Zea mays L. | Rhizophagus irregularis | Col%↓ | Plant growth↑ | P↑, WUE↑, Rehydration rate↑, leaf moisture percentage↑ | Proline↑, C:P↓, N:P↓, MDA↓ | [57] |
| Zea mays L. | Rhizophagus irregularis | Col% ns | Shoot dry weight↑, root dry weight↑ | gs↑, Lpr↑, Lo↑ | Root ABA↑ | [58] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahadur, A.; Batool, A.; Nasir, F.; Jiang, S.; Mingsen, Q.; Zhang, Q.; Pan, J.; Liu, Y.; Feng, H. Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants. Int. J. Mol. Sci. 2019, 20, 4199. https://doi.org/10.3390/ijms20174199
Bahadur A, Batool A, Nasir F, Jiang S, Mingsen Q, Zhang Q, Pan J, Liu Y, Feng H. Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants. International Journal of Molecular Sciences. 2019; 20(17):4199. https://doi.org/10.3390/ijms20174199
Chicago/Turabian StyleBahadur, Ali, Asfa Batool, Fahad Nasir, Shengjin Jiang, Qin Mingsen, Qi Zhang, Jianbin Pan, Yongjun Liu, and Huyuan Feng. 2019. "Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants" International Journal of Molecular Sciences 20, no. 17: 4199. https://doi.org/10.3390/ijms20174199
APA StyleBahadur, A., Batool, A., Nasir, F., Jiang, S., Mingsen, Q., Zhang, Q., Pan, J., Liu, Y., & Feng, H. (2019). Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants. International Journal of Molecular Sciences, 20(17), 4199. https://doi.org/10.3390/ijms20174199






