Anti-Apoptotic and Anti-Oxidant Proteins in Glioblastomas: Immunohistochemical Expression of Beclin and DJ-1 and Its Correlation with Prognosis
Abstract
1. Introduction
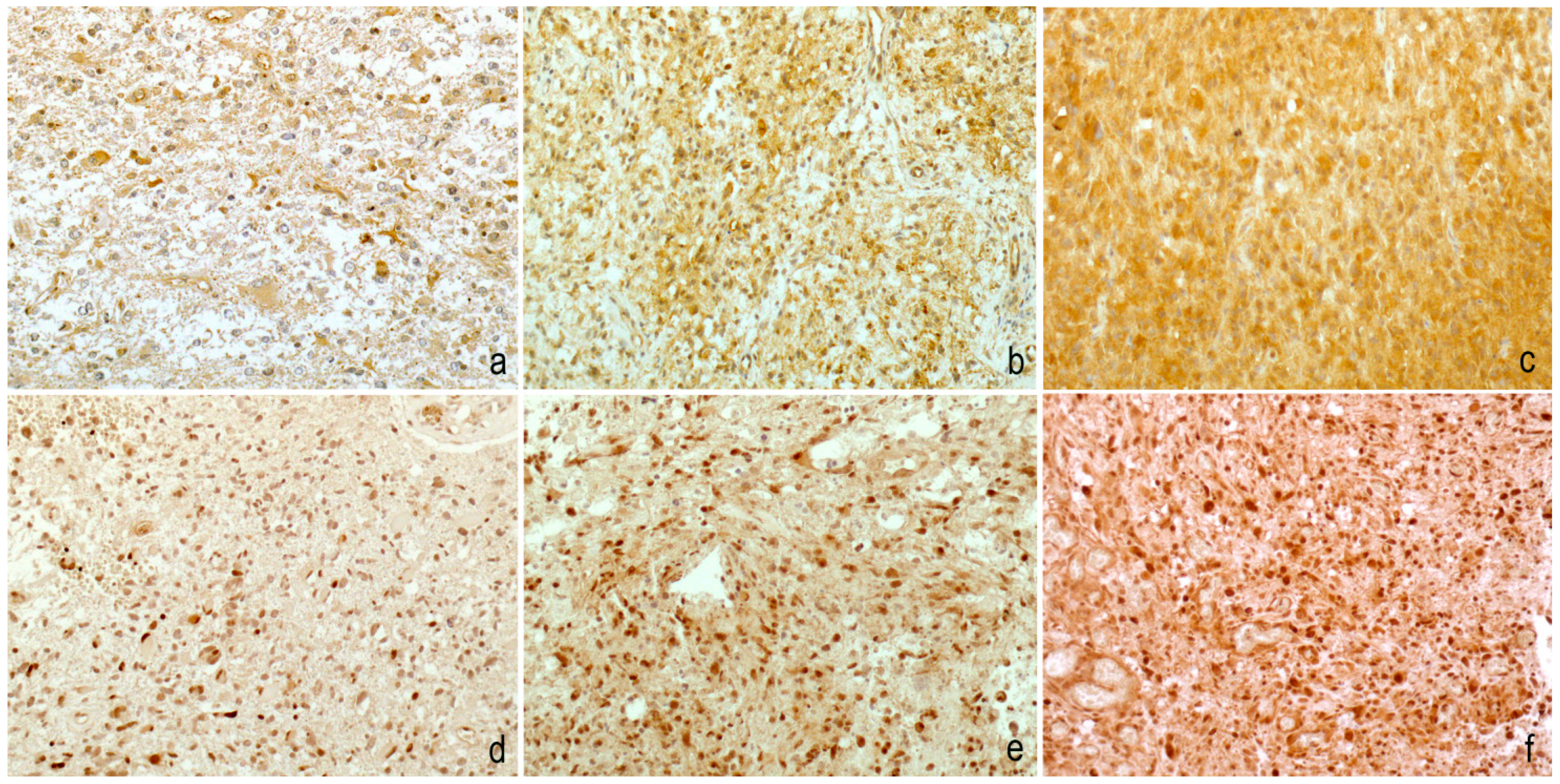
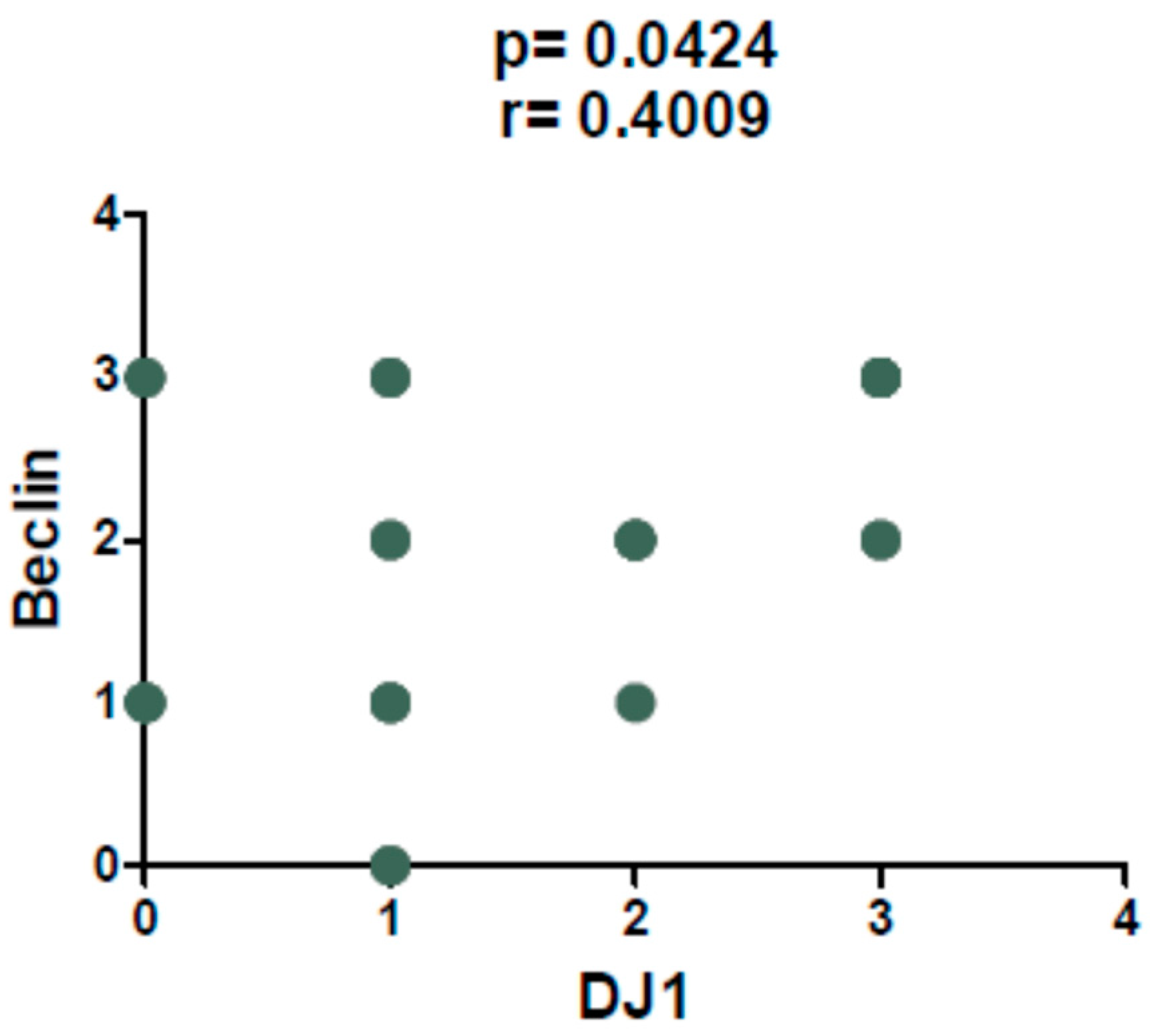
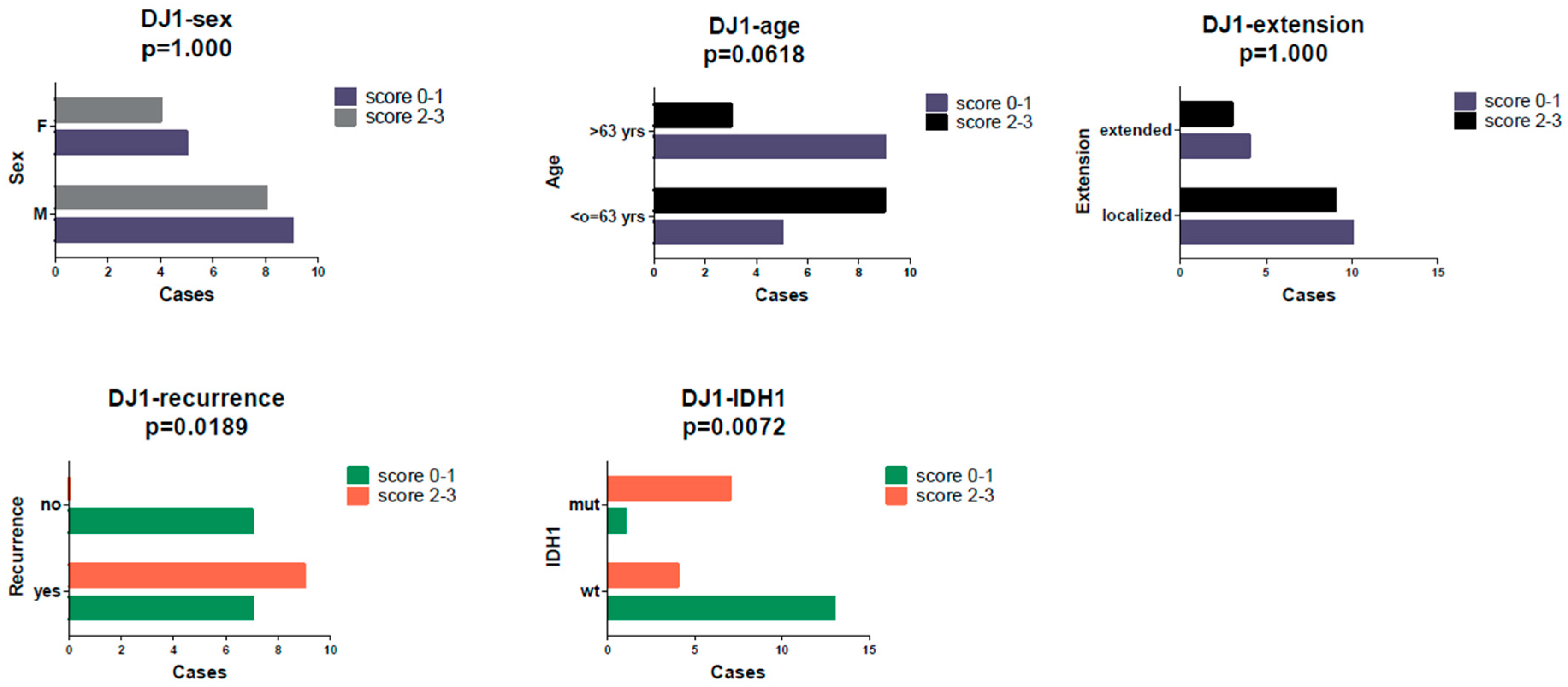
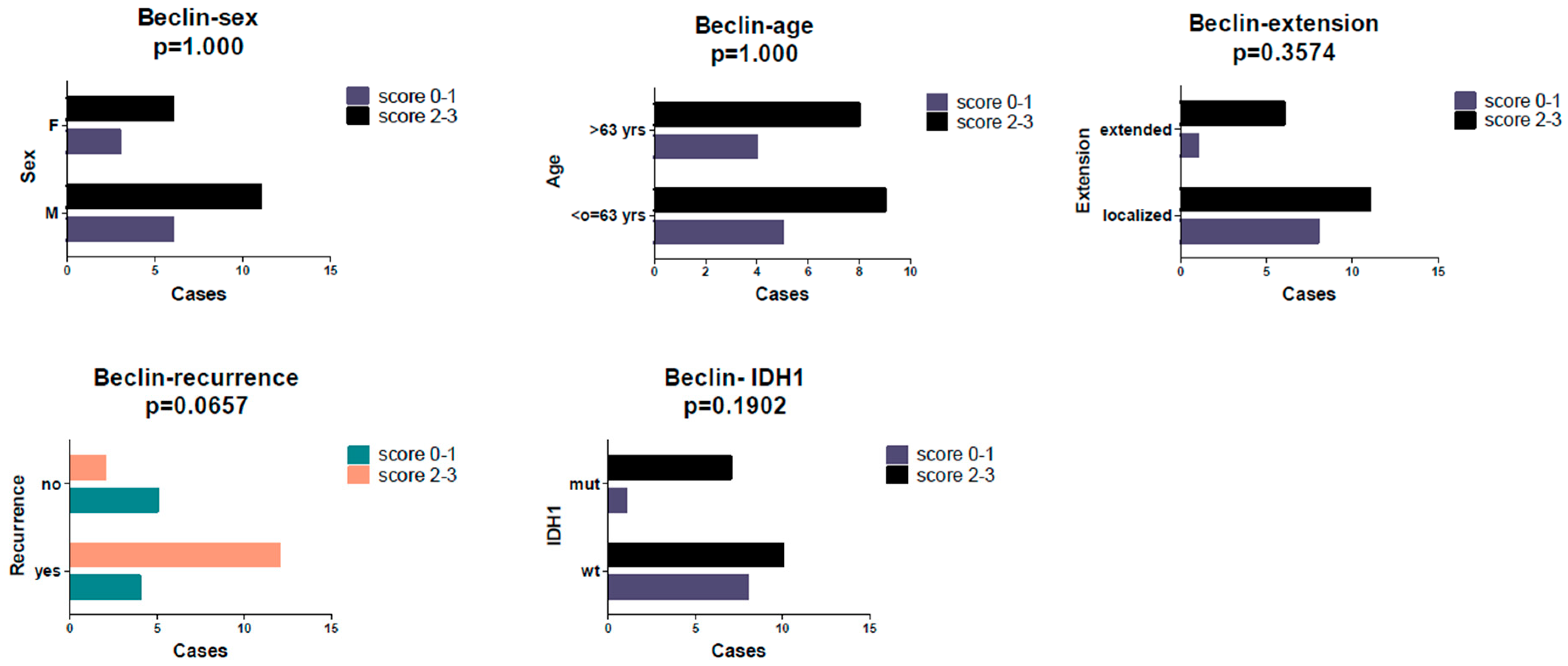
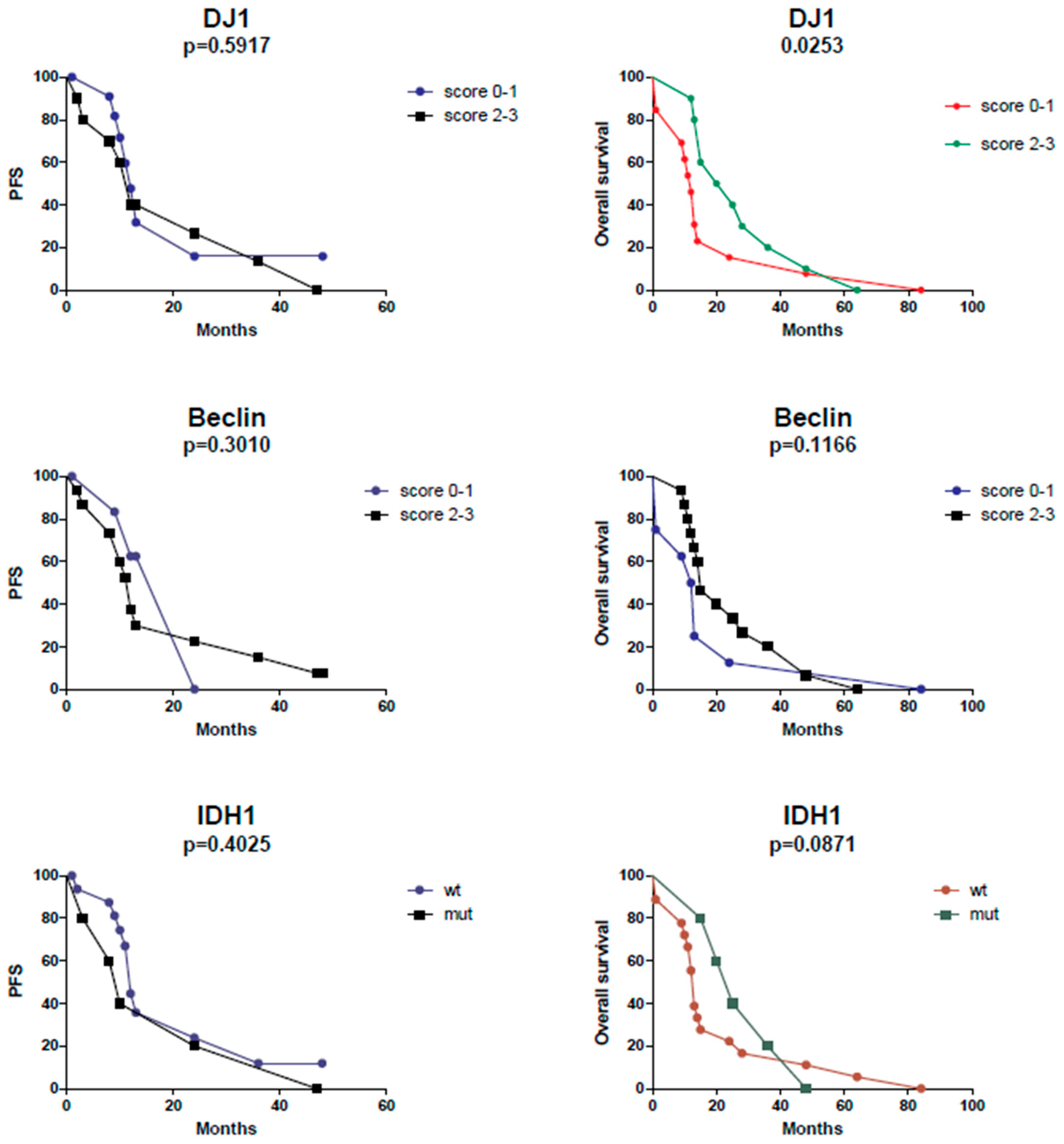
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ostrom, Q.T.; Gittleman, H.; Liao, P.; Rouse, C.; Chen, Y.; Dowling, J.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2007–2011. Neuro-Oncology. 2014, 16, iv1–iv63. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef] [PubMed]
- Shendelman, S.; Jonason, A.; Martinat, C.; Leete, T.; Abeliovich, A. DJ-1 is a redox-dependent molecular chaperone that inhibits α-synuclein aggregate formation. PLoS. Biol. 2004, 2, e362. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Zhu, M.; Wilson, M.A.; Petsko, G.A.; Fink, A.L. The oxidation state of DJ-1 regulates its chaperone activity toward α-synuclein. J. Mol. Biol. 2006, 356, 1036–1048. [Google Scholar] [CrossRef] [PubMed]
- MacKeigan, J.P.; Clements, C.M.; Lich, J.D.; Pope, R.M.; Hod, Y.; Ting, J.P. Proteomic profiling drug-induced apoptosis in non-small cell lung carcinoma: Identification of RS/DJ-1 and RhoGDIα. Cancer Res. 2003, 63, 6928–6934. [Google Scholar] [PubMed]
- Le Naour, F.; Misek, D.E.; Krause, M.C.; Deneux, L.; Giordano, T.J.; Scholl, S.; Hanash, S.M. Proteomics-based identification of RS/DJ-1 as a novel circulating tumor antigen in breast cancer. Clin. Cancer Res. 2001, 7, 3328–3335. [Google Scholar] [PubMed]
- Merikallio, H.; Pääkkö, P.; Kinnula, V.L.; Harju, T.; Soini, Y. Nuclearfactorerythroid-derived 2-like 2 (Nrf2) and DJ-1 are prognostic factors in lung cancer. Hum. Pathol. 2012, 43, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Morelli, M.; Scumaci, D.; Di Cello, A.; Venturella, R.; Donato, G.; Faniello, M.C.; Lent, B.; Cuda, G.; Zullo, F.; Costanzo, F. DJ-1 in endometrial cancer: A possible biomarker to improve differential diagnosis between subtypes. Int. J. Gynecol. Cancer 2014, 24, 649–658. [Google Scholar] [CrossRef]
- Soini, Y.; Eskelinen, M.; Juvonen, P.; Kärjä, V.; Haapasaari, K.M.; Saarela, A.; Karihtala, P. Nuclear Nrf2 expression is related to a poor survival in pancreatic adenocarcinoma. Pathol. Res. Pr. 2014, 210, 35–39. [Google Scholar] [CrossRef]
- Ahtikoski, A.M.; Kangas, J.; Salonen, R.; Park, U.; Karihtala, P. Cytoplasmic Keap1 Expression Is Associated With Poor Prognosis in Endometrial Cancer. Anticancer Res. 2019, 39, 585–590. [Google Scholar] [CrossRef]
- Zhong, Y.; Wang, Q.J.; Li, X.; Yan, Y.; Backer, J.M.; Chait, B.T.; Heintz, N.; Yue, Z. Distinct regulation of autophagic activity by Atg14L and Rubicon associated with Beclin 1-phosphatidylinositol-3-kinase complex. Nat. Cell Biol. 2009, 11, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Zeh, H.J.; Lotze, M.T.; Tang, D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ. 2011, 18, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Yu, J.; Bhagat, G.; Furuya, N.; Hibshoosh, H.; Troxel, A.; Rosen, J.; Eskelinen, E.L.; Mizushima, N.; Ohsumi, Y.; et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J. Clin. Invest. 2003, 112, 1809–1820. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.Y.; Wang, L.Y.; Zhao, S.; Guo, X.C.; Xu, Y.Q.; Zheng, Z.H.; Lu, H.; Zheng, H.C. Effects of Beclin 1 overexpression on aggressive phenotypes of colon cancer cells. Oncol. Lett. 2019, 17, 2441–2450. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Song, J.; Yang, X.; Guo, J.; Wang, T.; Zhuo, W. ProNGF siRN Ainhibits cell proliferation and invasion of pancreatic cancer cells and promotes anoikis. Biomed. Pharm. 2019, 111, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Valente, G.; Morani, F.; Nicotra, G.; Fusco, N.; Peracchio, C.; Titone, R.; Alabiso, O.; Arisio, R.; Katsaros, D.; Benedetto, C.; et al. Expression and clinical significance of the autophagy proteins BECLIN 1 and LC3 in ovarian cancer. Biomed Res. Int. 2014, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.E.; Mouradian, M.M. Regulation of Signal Transduction by DJ-1. Adv. Exp. Med. Biol. 2017, 1037, 97–131. [Google Scholar]
- Ren, H.; Fu, K.; Mu, C.; Li, B.; Wang, D.; Wang, G. DJ-1, a cancer and Parkinson’s disease associated protein, regulates autophagy through JNK pathway in cancer cells. Cancer Lett. 2010, 297, 101–108. [Google Scholar] [CrossRef]
- Hinkle, D.A.; Mullett, S.J.; Gabris, B.E. Hamilton, R.L. DJ-1 expression in glioblastomas shows positive correlation with p53 expression and negative correlation with epidermal growth factor receptor amplification. Neuropathology 2011, 31, 29–37. [Google Scholar] [CrossRef]
- Jin, S.; Dai, Y.; Li, C.; Fang, X.; Han, H.; Wang, D. MicroRNA-544 inhibits glioma proliferation, invasion and migration but induces cell apoptosis by targeting PARK7. Am. J. Transl. Res. 2016, 8, 1826–1837. [Google Scholar]
- Haapasalo, T.; Nordfors, K.; Järvelä, S.; Kok, E.; Sallinen, P.; Kinnula, V.L.; Haapasalo, H.K.; Soini, Y. Peroxiredoxins and their expression in ependymomas. J. Clin. Pathol. 2013, 66, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Fang, M.; Zhang, M.; Li, W.; Guan, H.; Sun, Y.; Xie, S.; Zhong, X. The positive correlation between DJ-1 and β-cateninexpression shows prognostic value for patients with glioma. Neuropathology 2013, 33, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Jennewein, L.; Ronellenfitsch, M.W.; Antonietti, P.; Ilina, E.I.; Jung, J.; Stadel, D.; Flohr, L.M.; Zinke, J.; von Renesse, J.; Drott, U.; et al. Diagnostic and clinicalrelevance of the autophago-lysosomal network in human gliomas. Oncotarget 2016, 7, 20016–20032. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Cui, Y.Z.; Song, G.H.; Zong, M.J.; Zhou, Y.; Chen, Y.; Han, J.X. Proteomic analysis identifies MMP-9, DJ-1 and A1BG as overexpressed proteins in pancreatic juice from pancreatic ductal adenocarcinoma patients. BMC Cancer 2008, 8, 241. [Google Scholar] [CrossRef] [PubMed]
- Conte, A.; Paladino, S.; Bianco, G.; Fasano, D.; Gerlini, R.; Tornincasa, M.; Renna, M.; Fusco, A.; Tramontano, D.; Pierantoni, G.M. High mobility group A1 protein modulates autophagy in cancer cells. Cell Death Differ. 2017, 24, 1948–1962. [Google Scholar] [CrossRef] [PubMed]
- Donato, G.; Martinez Hoyos, J.; Amorosi, A.; Maltese, L.; Lavano, A.; Volpentesta, G.; Signorelli, F.; Pentimalli, F.; Pallante, P.; Ferraro, G.; et al. High mobility group A1 expression correlates with the histological grade of human glial tumors. Oncol. Rep. 2004, 11, 1209–1213. [Google Scholar] [CrossRef] [PubMed]
| Case | Sex | Age | Site | IDH1 | DJ-1 | Beclin | Recurrence | O.S.* |
|---|---|---|---|---|---|---|---|---|
| 1 | F | 62 | Right temporal | wt | 1 | 0 | 24 | 84 |
| 2 | F | 70 | Right temporal | wt | 1 | 3 | no | 10 |
| 3 | M | 35 | Left temporal | mut | 2 | 2 | 47 | 48 |
| 4 | F | 41 | Right frontal | mut | 2 | 2 | 24 | 36 |
| 5 | F | 58 | Left posterior frontal | wt | 2 | 2 | 2 | 12 |
| 6 | M | 74 | Left temporal | wt | 3 | 3 | 12 | 15 |
| 7 | M | 60 | Left temporale | wt | 0 | 1 | no | 12 |
| 8 | M | 57 | Right frontal-temporal | wt | 0 | 3 | no | 48 |
| 9 | F | 36 | Left posterior temporal | wt | 1 | 1 | 12 | 24 |
| 10 | M | 69 | Right temporal | wt | 1 | 0 | 9 | 13 |
| 11 | F | 72 | Left temporal | wt | 3 | 2 | 36 | 64 |
| 12 | M | 73 | Left posterior parietal | wt | 1 | 2 | 10 | 11 |
| 13 | M | 71 | Left parietal | wt | 1 | 2 | 8 | 9 |
| 14 | M | 41 | Right temporal | mut | 3 | 3 | 8 | 15 |
| 15 | M | 52 | Left temporal | mut | 3 | 3 | 3 | 20 |
| 16 | M | 63 | Left frontal | mut | 3 | 3 | 10 | 25 |
| 17 | M | 61 | Left temporal-parietal | wt | 2 | 2 | 12 | 28 |
| 18 | M | 69 | Right frontal-temporal | wt | 1 | 3 | 13 | 13 |
| 19 | F | 81 | Right parietal-occipital | wt | 0 | 3 | 11 | 14 |
| 20 | F | 72 | temporal-parietal-occipital | wt | 1 | 1 | no | 1 |
| 21 | M | 62 | Right temporal | wt | 1 | 1 | no | 9 |
| 22 | M | 75 | Right temporal | wt | 0 | 1 | no | 1 |
| 23 | M | 63 | Right frontal | wt | 2 | 1 | no | 13 |
| 24 | M | 70 | Right temporal-parietal | mut | 3 | 3 | n.a. | n.a. |
| 25 | M | 72 | Left temporale | mut | 0 | 1 | n.a. | n.a. |
| 26 | F | 39 | Left frontal-parietal | mut | 3 | 2 | n.e. | n.e. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guadagno, E.; Borrelli, G.; Pignatiello, S.; Donato, A.; Presta, I.; Arcidiacono, B.; Malara, N.; Solari, D.; Somma, T.; Cappabianca, P.; et al. Anti-Apoptotic and Anti-Oxidant Proteins in Glioblastomas: Immunohistochemical Expression of Beclin and DJ-1 and Its Correlation with Prognosis. Int. J. Mol. Sci. 2019, 20, 4066. https://doi.org/10.3390/ijms20164066
Guadagno E, Borrelli G, Pignatiello S, Donato A, Presta I, Arcidiacono B, Malara N, Solari D, Somma T, Cappabianca P, et al. Anti-Apoptotic and Anti-Oxidant Proteins in Glioblastomas: Immunohistochemical Expression of Beclin and DJ-1 and Its Correlation with Prognosis. International Journal of Molecular Sciences. 2019; 20(16):4066. https://doi.org/10.3390/ijms20164066
Chicago/Turabian StyleGuadagno, Elia, Giorgio Borrelli, Sara Pignatiello, Annalidia Donato, Ivan Presta, Biagio Arcidiacono, Natalia Malara, Domenico Solari, Teresa Somma, Paolo Cappabianca, and et al. 2019. "Anti-Apoptotic and Anti-Oxidant Proteins in Glioblastomas: Immunohistochemical Expression of Beclin and DJ-1 and Its Correlation with Prognosis" International Journal of Molecular Sciences 20, no. 16: 4066. https://doi.org/10.3390/ijms20164066
APA StyleGuadagno, E., Borrelli, G., Pignatiello, S., Donato, A., Presta, I., Arcidiacono, B., Malara, N., Solari, D., Somma, T., Cappabianca, P., Donato, G., & Del Basso De Caro, M. (2019). Anti-Apoptotic and Anti-Oxidant Proteins in Glioblastomas: Immunohistochemical Expression of Beclin and DJ-1 and Its Correlation with Prognosis. International Journal of Molecular Sciences, 20(16), 4066. https://doi.org/10.3390/ijms20164066







