Searching for Potential Lipid Biomarkers of Parkinson’s Disease in Parkin-Mutant Human Skin Fibroblasts by HILIC-ESI-MS/MS: Preliminary Findings
Abstract
1. Introduction
2. Results
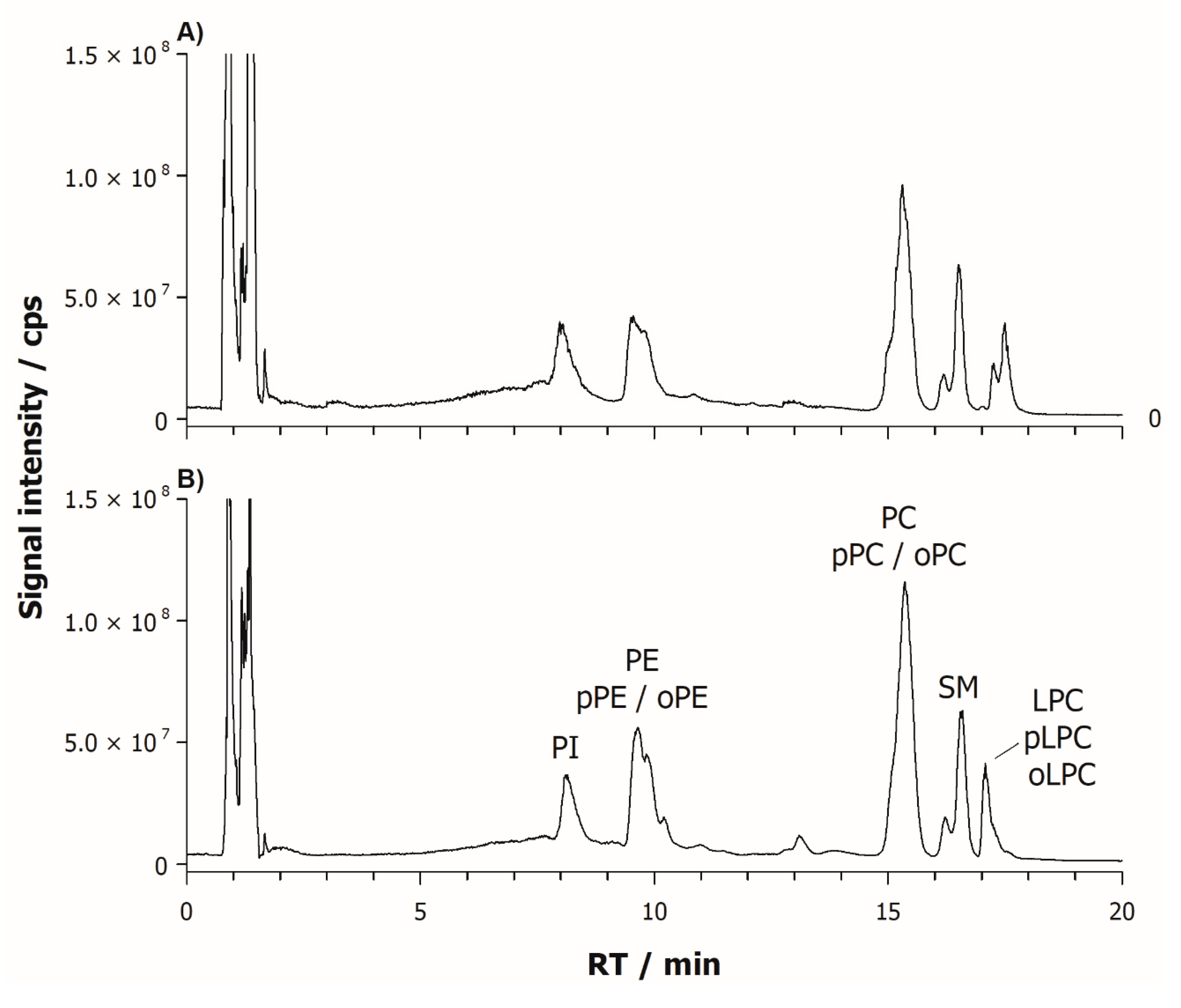
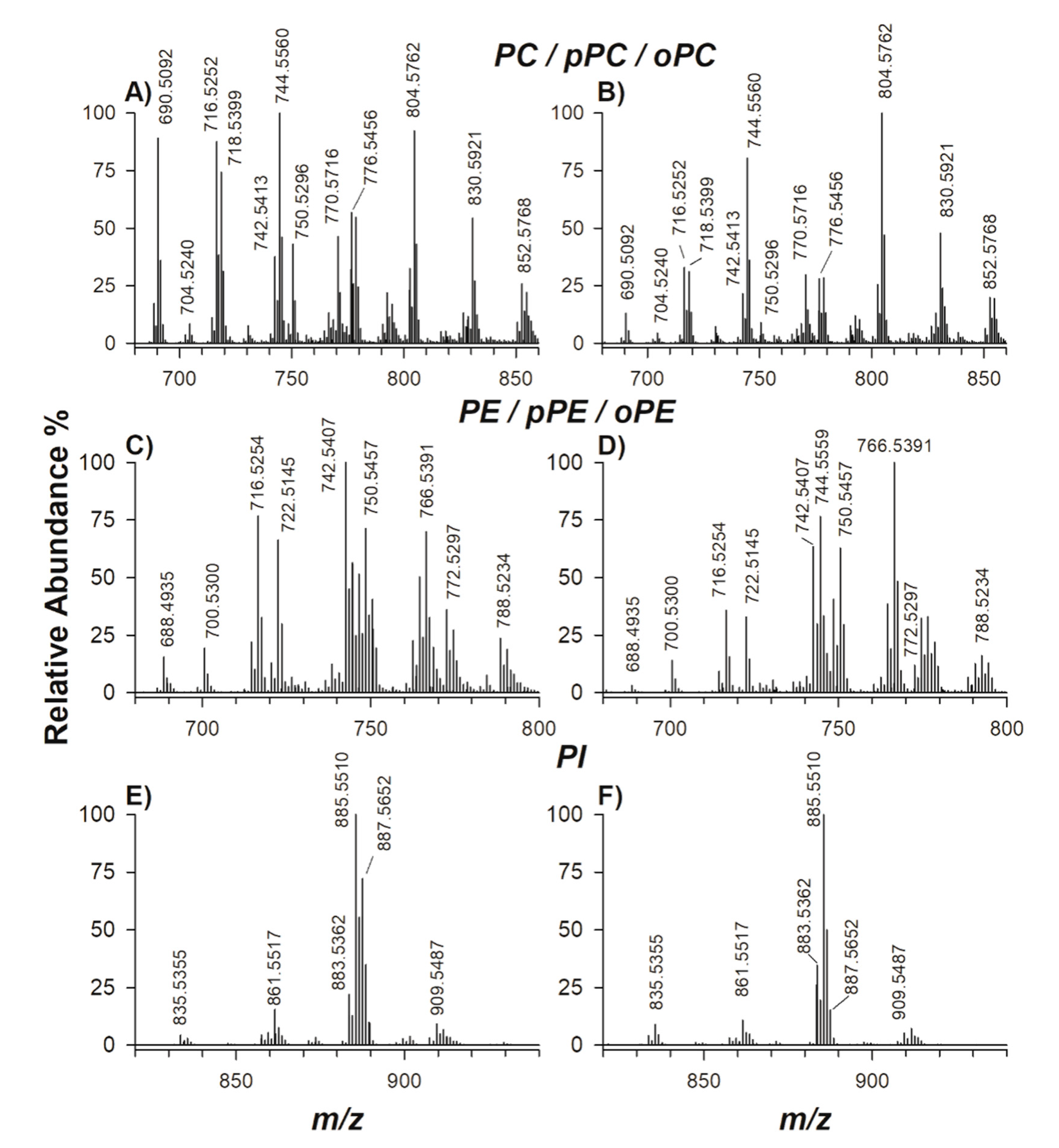
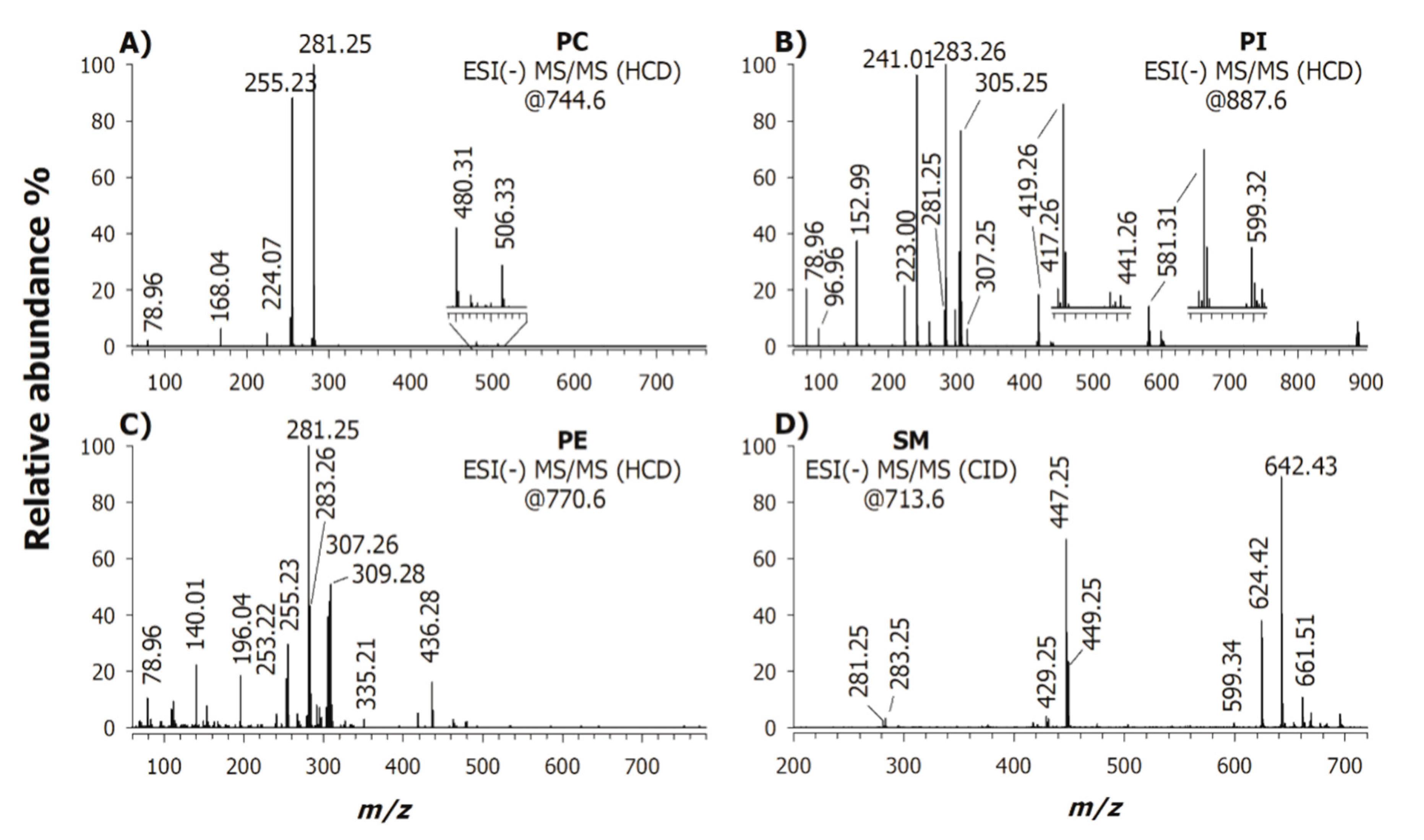
2.1. Characterization of Phospholipids
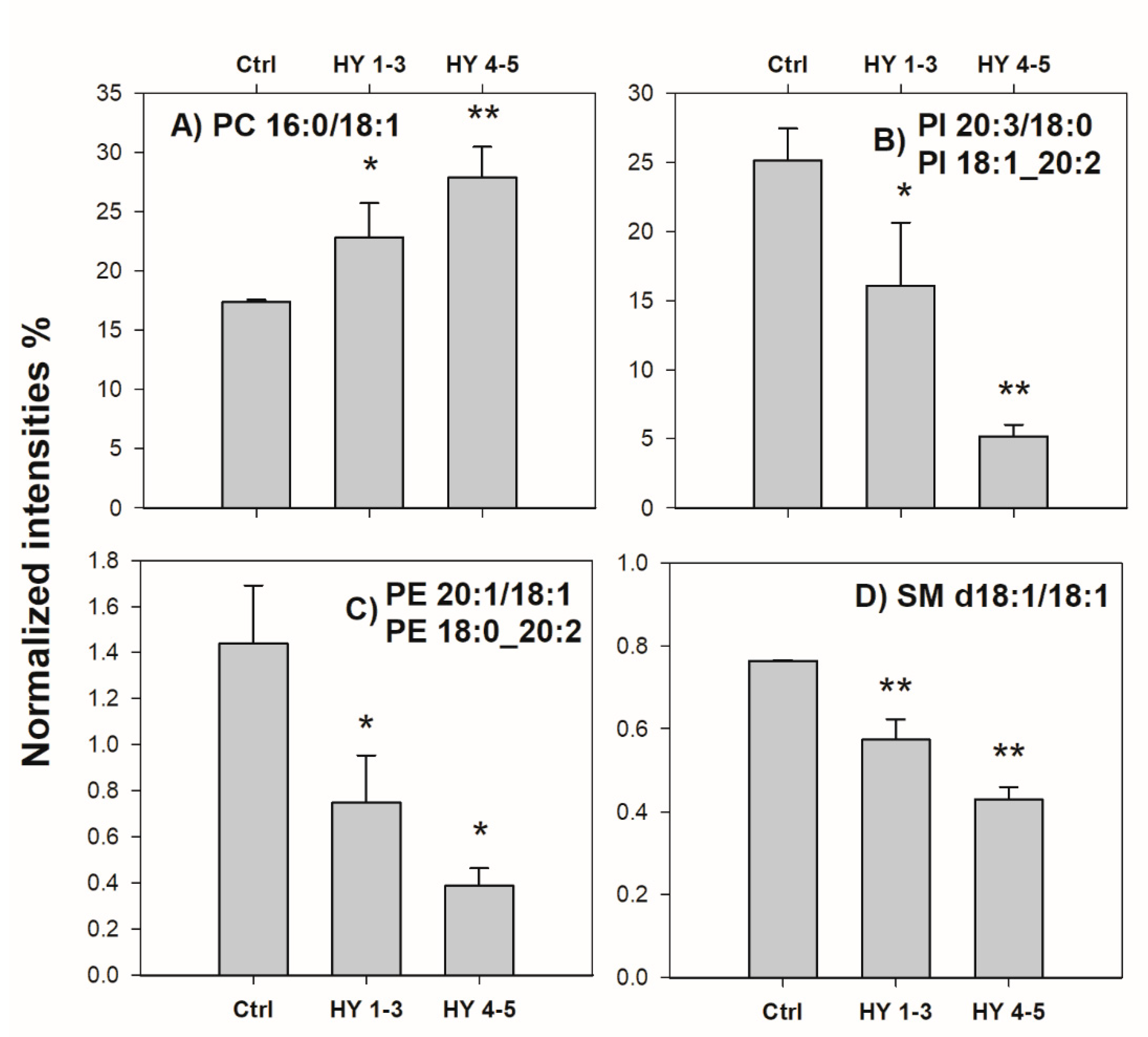
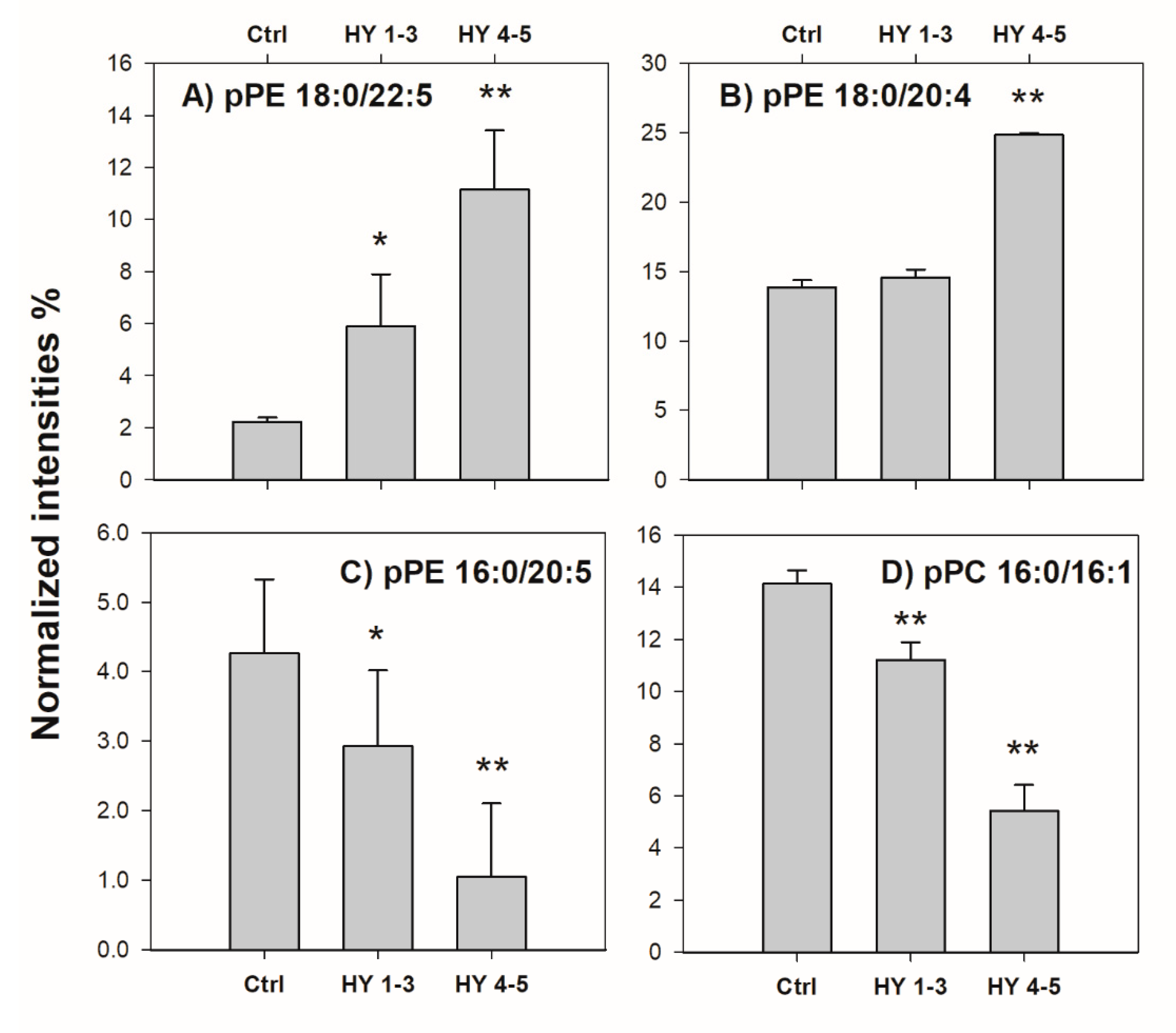
2.2. Control vs. PD Patients
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Sample Preparation
4.2.1. Samples and Cell Growth
4.2.2. Lipid Extraction
4.3. HILIC-ESI-MS Instrumentation and Operating Conditions
4.4. Data Treatment
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kitada, T.; Tomlinson, J.J.; Ao, H.S.; Grimes, D.A.; Schlossmacher, M.G. Considerations Regarding the Etiology and Future Treatment of Autosomal Recessive Versus Idiopathic Parkinson Disease. Curr. Treat. Options Neurol. 2012, 14, 230–240. [Google Scholar] [CrossRef]
- Klockgether, T. Parkinson’s disease: Clinical aspects. Cell Tissue Res. 2004, 318, 115–120. [Google Scholar] [CrossRef]
- Menza, M.; Dobkin, R.D.; Marin, H. Treatment of depression in Parkinson’s disease. Curr. Psychiatry Rep. 2006, 8, 234–240. [Google Scholar] [CrossRef]
- Feigin, V.L.; Abajobir, A.A.; Abate, K.H.; Abd-Allah, F.; Abdulle, A.M.; Abera, S.F.; Abyu, G.Y.; Ahmed, M.B.; Aichour, A.N.; Aichour, I.; et al. Global, regional, and national burden of neurological disorders during 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017, 16, 877–897. [Google Scholar] [CrossRef]
- Pringsheim, T.; Jette, N.; Frolkis, A.; Steeves, T.D.L. The prevalence of Parkinson’s disease: A systematic review and meta-analysis. Mov. Disord. 2014, 29, 1583–1590. [Google Scholar] [CrossRef]
- Dickson, D.W.; Braak, H.; Duda, J.E.; Duyckaerts, C.; Gasser, T.; Halliday, G.M.; Hardy, J.; Leverenz, J.B.; Del Tredici, K.; Wszolek, Z.K.; et al. Neuropathological assessment of Parkinson’s disease: Refining the diagnostic criteria. Lancet Neurol. 2009, 8, 1150–1157. [Google Scholar] [CrossRef]
- Halliday, G.M.; Ophof, A.; Broe, M.; Jensen, P.H.; Kettle, E.; Fedorow, H.; Cartwright, M.I.; Griffiths, F.M.; Shepherd, C.E.; Double, K.L. α-Synuclein redistributes to neuromelanin lipid in the substantia nigra early in Parkinson’s disease. Brain 2005, 128, 2654–2664. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression, and mortality. Neurology 1967, 17, 427–442. [Google Scholar] [CrossRef]
- Salat, D.; Tolosa, E. Levodopa in the treatment of Parkinson’s disease: Current status and new developments. J. Parkinson’s. Dis. 2013, 3, 255–269. [Google Scholar]
- Wakabayashi, K.; Takahashi, H. Pathology of familial Parkinson’s disease. Brain Nerve 2007, 59, 851–864. [Google Scholar]
- Klein, C.; Westenberger, A. Genetics of Parkinson’s disease. Cold Spring Harb. Perspect. Med. 2012, 2, a008888. [Google Scholar] [CrossRef]
- Dawson, T.M.; Dawson, V.L. The role of parkin in familial and sporadic Parkinson’s disease. Mov. Disord. 2010, 25, S32–S39. [Google Scholar] [CrossRef] [PubMed]
- Papa, S.; Sardanelli, A.M.; Capitanio, N.; Piccoli, C. Mitochondrial respiratory dysfunction and mutations in mitochondrial DNA in PINK1 familial Parkinsonism. J. Bioenerg. Biomembr. 2009, 41, 509–516. [Google Scholar] [CrossRef]
- Piccoli, C.; Sardanelli, A.; Scrima, R.; Ripoli, M.; Quarato, G.; D’Aprile, A.; Bellomo, F.; Scacco, S.; De Michele, G.; Filla, A.; et al. Mitochondrial Respiratory Dysfunction in Familiar Parkinsonism Associated with PINK1 Mutation. Neurochem. Res. 2008, 33, 2565–2574. [Google Scholar] [CrossRef]
- Calvano, C.D.; Palmisano, F.; Cataldi, T.R. Understanding neurodegenerative disorders by MS-based lipidomics. Bioanalysis 2018, 10, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Shamim, A.; Mahmood, T.; Ahsan, F.; Kumar, A.; Bagga, P. Lipids: An insight into the neurodegenerative disorders. Clin. Nutr. Exp. 2018, 20, 1–19. [Google Scholar] [CrossRef]
- Song, S.; Cheong, L.Z.; Man, Q.Q.; Pang, S.J.; Li, Y.Q.; Ren, B.; Zhang, J. Characterization of potential plasma biomarkers related to cognitive impairment by untargeted profiling of phospholipids using the HILIC-ESI-IT-TOF-MS system. Anal. Bioanal. Chem. 2018, 410, 2937–2948. [Google Scholar] [CrossRef] [PubMed]
- Farmer, K.; Smith, C.A.; Hayley, S.; Smith, J. Major alterations of phosphatidylcholine and lysophosphotidylcholine lipids in the substantia nigra using an early stage model of parkinson’s disease. Int. J. Mol. Sci. 2015, 16, 18865–18877. [Google Scholar] [CrossRef]
- Wood, P.L.; Tippireddy, S.; Feriante, J.; Woltjer, R.L. Augmented frontal cortex diacylglycerol levels in Parkinson’s disease and Lewy Body Disease. PLoS ONE 2018, 13, e0191815. [Google Scholar] [CrossRef]
- Cheng, D.; Jenner, A.M.; Shui, G.; Cheong, W.F.; Mitchell, T.W.; Nealon, J.R.; Kim, W.S.; McCann, H.; Wenk, M.R.; Halliday, G.M.; et al. Lipid pathway alterations in parkinson’s disease primary visual cortex. PLoS ONE 2011, 6, e17299. [Google Scholar] [CrossRef]
- Burbulla, L.F.; Krüger, R. The Use of Primary Human Fibroblasts for Monitoring Mitochondrial Phenotypes in the Field of Parkinson’s Disease. J. Vis. Exp. 2012, 68, e4228. [Google Scholar] [CrossRef] [PubMed]
- Treutlein, B.; Lee, Q.Y.; Camp, J.G.; Mall, M.; Koh, W.; Shariati, S.A.M.; Sim, S.; Neff, N.F.; Skotheim, J.M.; Wernig, M.; et al. Dissecting direct reprogramming from fibroblast to neuron using single-cell RNA-seq. Nature 2016, 534, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Hou, P.-S.; Chuang, C.-Y.; Yeh, C.-H.; Chiang, W.; Liu, H.-J.; Lin, T.-N.; Kuo, H.-C. Direct Conversion of Human Fibroblasts into Neural Progenitors Using Transcription Factors Enriched in Human ESC-Derived Neural Progenitors. Stem Cell Rep. 2017, 8, 54–68. [Google Scholar] [CrossRef] [PubMed]
- Mortiboys, H.; Thomas, K.J.; Koopman, W.J.; Klaffke, S.; Abou-Sleiman, P.; Olpin, S.; Wood, N.W.; Willems, P.H.; Smeitink, J.A.; Cookson, M.R.; et al. Mitochondrial function and morphology are impaired in parkin-mutant fibroblasts. Ann. Neurol. 2008, 64, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Hoepken, H.-H.; Gispert, S.; Morales, B.; Wingerter, O.; Del Turco, D.; Mülsch, A.; Nussbaum, R.L.; Müller, K.; Dröse, S.; Brandt, U.; et al. Mitochondrial dysfunction, peroxidation damage and changes in glutathione metabolism in PARK6. Neurobiol. Dis. 2007, 25, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Vergara, D.; Ferraro, M.M.; Cascione, M.; Loretta, L.; Leporatti, S.; Ferretta, A.; Tanzarella, P.; Pacelli, C.; Santino, A.; Maffia, M.; et al. Cytoskeletal Alterations and Biomechanical Properties of parkin-Mutant Human Primary Fibroblasts. Cell Biochem. Biophys. 2015, 71, 1395–1404. [Google Scholar] [CrossRef] [PubMed]
- Pacelli, C.; De Rasmo, D.; Signorile, A.; Grattagliano, I.; di Tullio, G.; D’Orazio, A.; Nico, B.; Comi, G.P.; Ronchi, D.; Ferranini, E.; et al. Mitochondrial defect and PGC-1α dysfunction in parkin-associated familial Parkinson’s disease. Biochim. Biophys. Acta Mol. Basis Dis. 2011, 1812, 1041–1053. [Google Scholar] [CrossRef] [PubMed]
- Lobasso, S.; Tanzarella, P.; Vergara, D.; Maffia, M.; Cocco, T.; Corcelli, A. Lipid profiling of parkin-mutant human skin fibroblasts. J. Cell. Physiol. 2017, 232, 3540–3551. [Google Scholar] [CrossRef]
- Losito, I.; Patruno, R.; Conte, E.; Cataldi, T.R.I.; Megli, F.M.; Palmisano, F. Phospholipidomics of Human Blood Microparticles. Anal. Chem. 2013, 85, 6405–6413. [Google Scholar] [CrossRef]
- Bang, D.Y.; Byeon, S.K.; Moon, M.H. Rapid and simple extraction of lipids from blood plasma and urine for liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2014, 1331, 19–26. [Google Scholar] [CrossRef]
- Calvano, C.D.; Glaciale, M.; Palmisano, F.; Cataldi, T.R.I. Glycosphingolipidomics of donkey milk by hydrophilic interaction liquid chromatography coupled to ESI and multistage MS. Electrophoresis 2018, 39, 1634–1644. [Google Scholar] [CrossRef] [PubMed]
- Calvano, C.D.; Sardanelli, A.M.; Ventura, G.; Glaciale, M.; Savino, L.; Losito, I.; Palmisano, F.; Cataldi, T. Development and use of advanced mass spectrometry techniques for the characterization of cellular and mitochondrial lipidomic profiling in control fibroblasts and Parkinson’s disease patients. Trends Anal. Res. 2018, 1, 1–10. [Google Scholar]
- Losito, I.; Facchini, L.; Diomede, S.; Conte, E.; Megli, F.M.; Cataldi, T.R.I.; Palmisano, F. Hydrophilic interaction liquid chromatography–electrospray ionization-tandem mass spectrometry of a complex mixture of native and oxidized phospholipids. J. Chromatogr. A 2015, 1422, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Ellis, S.R.; Paine, M.R.L.; Eijkel, G.B.; Pauling, J.K.; Husen, P.; Jervelund, M.W.; Hermansson, M.; Ejsing, C.S.; Heeren, R.M. Automated, parallel mass spectrometry imaging and structural identification of lipids. Nat. Methods 2018, 15, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Dane, A.; Spijksma, G.; Wang, M.; van der Greef, J.; Luo, G.; Hankemeier, T.; Vreeken, R.J. An efficient hydrophilic interaction liquid chromatography separation of 7 phospholipid classes based on a diol column. J. Chromatogr. A 2012, 1220, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, B.; Muller, K.; Paasch, U.; Schiller, J. Lysophospholipids: Potential markers of diseases and infertility? Mini Rev. Med. Chem. 2012, 12, 74–86. [Google Scholar] [CrossRef] [PubMed]
- Granafei, S.; Losito, I.; Palmisano, F.; Cataldi, T.R. Identification of isobaric lyso-phosphatidylcholines in lipid extracts of gilthead sea bream (Sparus aurata) fillets by hydrophilic interaction liquid chromatography coupled to high-resolution Fourier-transform mass spectrometry. Anal. Bioanal. Chem. 2015, 407, 6391–6404. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.-F.; Turk, J. Electrospray ionization with low-energy collisionally activated dissociation tandem mass spectrometry of glycerophospholipids: Mechanisms of fragmentation and structural characterization. J. Chromatogr. B 2009, 877, 2673–2695. [Google Scholar] [CrossRef]
- Hsu, F.-F.; Turk, J. Characterization of phosphatidylinositol, phosphatidylinositol-4-phosphate, and phosphatidylinositol-4,5-bisphosphate by electrospray ionization tandem mass spectrometry: A mechanistic study. J. Am. Soc. Mass Spectrom. 2000, 11, 986–999. [Google Scholar] [CrossRef]
- Houjou, T.; Yamatani, K.; Nakanishi, H.; Imagawa, M.; Shimizu, T.; Taguchi, R. Rapid and selective identification of molecular species in phosphatidylcholine and sphingomyelin by conditional neutral loss scanning and MS3. Rapid Commun. Mass Spectrom. 2004, 18, 3123–3130. [Google Scholar] [CrossRef]
- Hsu, F.F. Complete structural characterization of ceramides as [M − H]− ions by multiple-stage linear ion trap mass spectrometry. Biochimie 2016, 130, 63–75. [Google Scholar] [CrossRef]
- Berry, K.A.Z.; Murphy, R.C. Electrospray ionization tandem mass spectrometry of glycerophosphoethanolamine plasmalogen phospholipids. J. Am. Soc. Mass Spectrom. 2004, 15, 1499–1508. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.-F.; Lodhi, I.J.; Turk, J.; Semenkovich, C.F. Structural Distinction of Diacyl-, Alkylacyl, and Alk-1-Enylacyl Glycerophosphocholines as [M-15]-Ions by Multiple-Stage Linear Ion-Trap Mass Spectrometry with Electrospray Ionization. J. Am. Soc. Mass Spectrom. 2014, 25, 1412–1420. [Google Scholar] [CrossRef] [PubMed]
- Xicoy, H.; Wieringa, B.; Martens, G.J.M.; Xicoy, H.; Wieringa, B.; Martens, G.J.M. The Role of Lipids in Parkinson’s Disease. Cells 2019, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- Valsecchi, M.; Mauri, L.; Casellato, R.; Prioni, S.; Loberto, N.; Prinetti, A.; Chigorno, V.; Sonnino, S. Ceramide and sphingomyelin species of fibroblasts and neurons in culture. J. Lipid Res. 2007, 48, 417–424. [Google Scholar] [CrossRef]
- Fuller, M.; Duplock, S.; Hein, L.K.; Rigat, B.A.; Mahuran, D.J. Liquid chromatography/electrospray ionisation-tandem mass spectrometry quantification of GM2 gangliosides in human peripheral cells and plasma. Anal. Biochem. 2014, 458, 20–26. [Google Scholar] [CrossRef]
- Olsen, A.S.B.; Færgeman, N.J. Sphingolipids: Membrane microdomains in brain development, function and neurological diseases. Open Biol. 2017, 7, 170069. [Google Scholar] [CrossRef] [PubMed]
- Guedes, L.C.; Chan, R.B.; Gomes, M.A.; Conceição, V.A.; Machado, R.B.; Soares, T.; Xu, Y.; Gaspar, P.; Carriço, J.A.; Alcalay, R.N.; et al. Serum lipid alterations in GBA-associated Parkinson’s disease. Parkinsonism Relat. Disord. 2017, 44, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Abbott, S.K.; Li, H.; Muñoz, S.S.; Knoch, B.; Batterham, M.; Murphy, K.E.; Halliday, G.M.; Garner, B. Altered ceramide acyl chain length and ceramide synthase gene expression in Parkinson’s disease. Mov. Disord. 2014, 29, 518–526. [Google Scholar] [CrossRef]
- Braverman, N.E.; Moser, A.B. Functions of plasmalogen lipids in health and disease. Biochim. Biophys. Acta Mol. Basis Dis. 2012, 1822, 1442–1452. [Google Scholar] [CrossRef]
- Han, X. Lipid alterations in the earliest clinically recognizable stage of Alzheimer’s disease: Implication of the role of lipids in the pathogenesis of Alzheimer’s disease. Curr. Alzheimer Res. 2005, 2, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Fabelo, N.; Martin, V.; Santpere, G.; Marín, R.; Torrent, L.; Ferrer, I.; Díaz, M. Severe alterations in lipid composition of frontal cortex lipid rafts from Parkinson’s disease and incidental Parkinson’s disease. Mol. Med. 2011, 17, 1107–1118. [Google Scholar] [CrossRef]
- Broniec, A.; Klosinski, R.; Pawlak, A.; Wrona-Krol, M.; Thompson, D.; Sarna, T. Interactions of plasmalogens and their diacyl analogs with singlet oxygen in selected model systems. Free Radic. Biol. Med. 2011, 50, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Liebisch, G.; Ekroos, K.; Hermansson, M.; Ejsing, C.S. Reporting of lipidomics data should be standardized. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2017, 1862, 747–751. [Google Scholar] [CrossRef] [PubMed]
- Liebisch, G.; Vizcaíno, J.A.; Köfeler, H.; Trötzmüller, M.; Griffi, W.J.; Schmitz, G.; Spener, F.; Wakelam, M.J.O. Shorthand notation for lipid structures derived from mass spectrometry. J. Lipid Res. 2013, 54, 1523–1530. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dier, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Calvano, C.D.; Ventura, G.; Sardanelli, A.M.; Losito, I.; Palmisano, F.; Cataldi, T. Identification of neutral and acidic glycosphingolipids in the human dermal fibroblasts. Anal. Biochem. 2019, 581, 113348. [Google Scholar] [CrossRef]
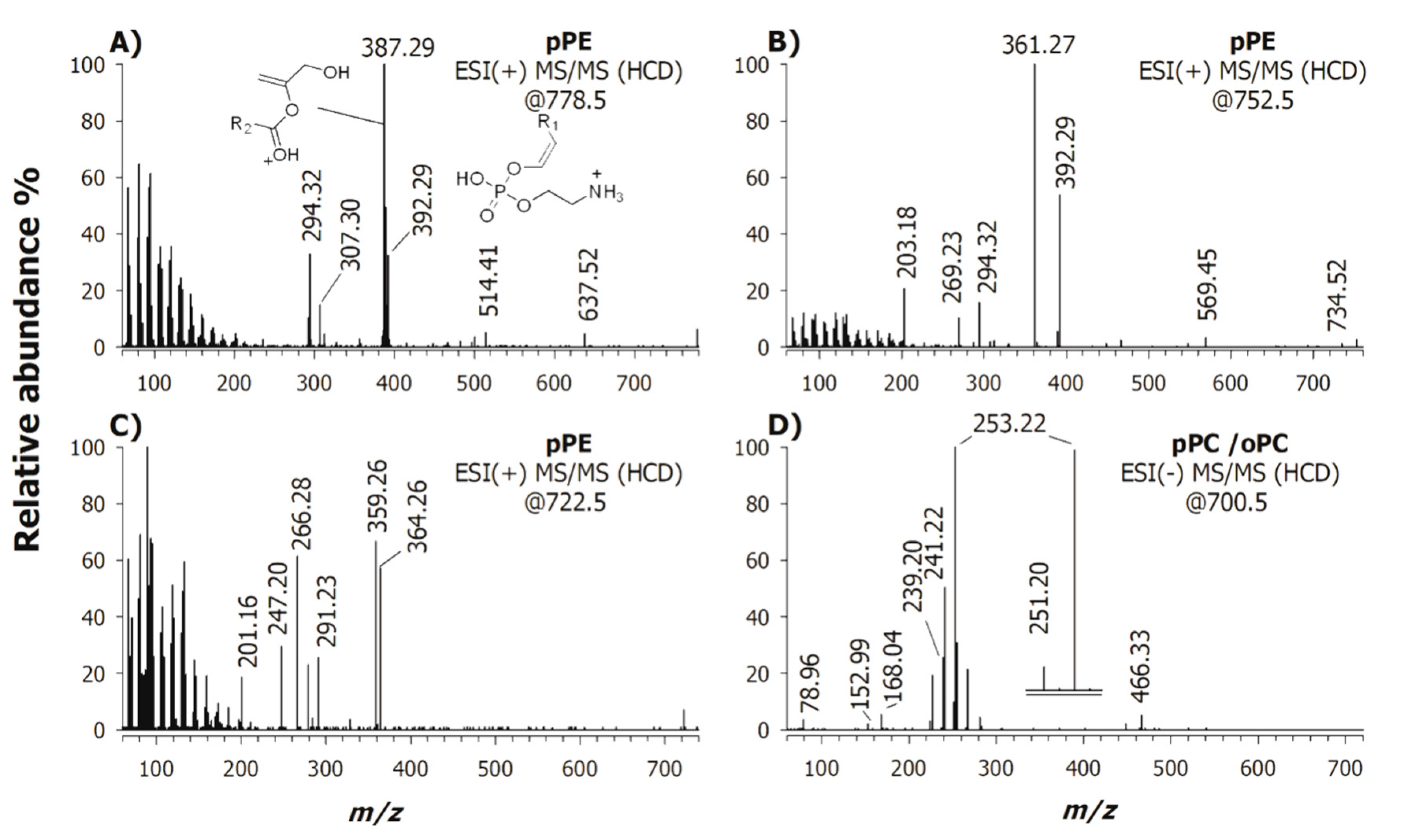
| Experimental Value (m/z) | Adduct | Regiochemical Assignment | Molecular Formula (M) |
|---|---|---|---|
| Plasmenyl-phosphatidylcholine(p-PC) | |||
| 700.5286 | [M − CH3 − H]− | (16:0/16:1) | C39H75NO7P |
| 702.5448 | [M − CH3 − H]− | (16:0/16:0) | C39H77NO7P |
| Plasmanyl-phosphatidylcholine(o-PC) | |||
| 700.5286 | [M − CH3 − H]− | (16:0/16:2) | C39H75NO7P |
| 702.5448 | [M − CH3 − H]− | (16:0/16:1); (15:0_17:1) | C39H77NO7P |
| 714.5443 | [M − CH3 − H]− | (16:1_19:1) | C40H77NO7P |
| 716.599 | [M − CH3 − H]− | (18:1/15:0); (16:1/17:0) | C40H79NO7P |
| 718.5756 | [M − CH3 − H]− | (17:0_16:0) | C40H81NO7P |
| 732.5912 | [M − CH3 − H]− | (16:0/18:0); (18:0/16:0); (17:0/17:0) | C41H83NO7P |
| 752.5599 | [M − CH3 − H]− | (16:0/20:4) | C43H79NO7P |
| 756.5912 | [M − CH3 − H]− | (16:0/20:2) | C43H83NO7P |
| 762.5654 | [M + HCOO]− | (16:0/16:1) | C41H81NO9P |
| 780.5912 | [M − CH3 − H]− | (18:0/20:4) | C45H83NO7P |
| 802.5967 | [M + HCOO]− | (17:1/18:1) | C44H85NO9P |
| 812.5811 | [M + HCOO]− | (16:0/20:4) | C45H83NO9P |
| Plasmenyl-phosphatidylethanolamine(p-PE) | |||
| 672.4973 | [M − H]− | (16:0/16:1) | C37H71NO7P |
| 698.5130 | [M − H]− | (16:0/18:2) | C39H73NO7P |
| 700.5286 | [M − H]− | (16:0/18:1) | C39H75NO7P |
| 720.4973 | [M − H]− | (16:0/20:5) | C41H71NO7P |
| 722.5130 | [M − H]− | (16:1/20:4) | C41H73NO7P |
| 728.5599 | [M − H]− | (18:0/18:1) | C41H79NO7P |
| 748.5286 | [M − H]− | (18:1/20:4) | C43H75NO7P |
| 750.5443 | [M − H]− | (18:0/20:4) | C43H77NO7P |
| 772.5286 | [M − H]− | (18:1/22:6) | C45H75NO7P |
| 774.5442 | [M − H]− | (18:1/22:5) | C45H77NO7P |
| 776.5599 | [M − H]− | (18:0/22:5) | C45H79NO7P |
| Plasmanyl-phosphatidylethanolamine(o-PE) | |||
| 702.5443 | [M − H]− | (16:0/18:1) | C39H77NO7P |
| 716.5588 | [M − H]− | (17:0/18:2) | C40H77NO7P |
| 728.5599 | [M − H]− | (16:0/20:2) | C41H79NO7P |
| 762.5443 | [M − H]− | (17:0/22:5) | C44H79NO7P |
| 764.5599 | [M − H]− | (17:0/22:5) (19:0/20:5) | C44H79NO7P |
| 776.5599 | [M − H]− | (18:0/22:6) | C45H79NO7P |
| Phosphatidylserine(PS) | |||
| 700.5304 | [M − H]− | (18:1/12:2); (18:2/12:1) | C36H63NO10P |
| 702.5433 | [M − H]− | (18:0/12:2); (14:1/16:1) | C36H65NO10P |
| 732.482 | [M − H]− | (16:0/16:1); (18:1_14:0) | C38H71NO10P |
| 734.498 | [M − H]− | (16:1/16:1) | C38H73NO10P |
| 756.4820 | [M − H]− | (18:2_16:1); (18:1_16:2) | C40H71NO10P |
| 758.4980 | [M − H]− | (16:0/18:2); (18:1/16:1) | C40H73NO10P |
| 760.5149 | [M − H]− | 16:0/18:1; 16:1_18:0 | C40H75NO10P |
| 774.5303 | [M − H]− | (18:0/17:1); (18:1_17:0); (16:0_19:1); (16:1_19:0) | C41H77NO10P |
| 782.4977 | [M − H]− | (16:0/20:4); (18:2_18:2) | C42H73NO10P |
| 784.5134 | [M − H]− | (16:0/20:4); (18:1_18:3) | C42H75NO10P |
| 786.5309 | [M − H]− | (18:0/18:2); (18:1/18:1); (16:0/20:2); (16:1_20:1) | C42H77NO10P |
| 788.4977 | [M − H]− | (18:0/18:1); (16:0_20:1) | C42H79NO10P |
| 790.5532 | [M − H]− | (18:0/18:0) | C42H81NO10P |
| 796.5134 | [M − H]− | (17:0/20:4) | C43H75NO10P |
| 800.5447 | [M − H]− | (19:1_18:1) | C43H75NO10P |
| 806.4977 | [M − H]− | (16:0/22:6); (18:1_20:5) | C44H73NO10P |
| 808.5134 | [M − H]− | (16:0/22:5); (18:1_20:4); (18:0_20:5) | C44H75NO10P |
| 810.5297 | [M − H]− | (18:1/20:3); (18:0_20:4) | C44H77NO10P |
| 812.5450 | [M − H]− | (18:0/20:3); (18:1_20:2) | C44H79NO10P |
| 816.5760 | [M − H]− | (18:0/20:1); (18:1_20:0) | C44H81NO10P |
| 826.5603 | [M − H]− | (18:0/21:3) | C45H81NO10P |
| 832.5130 | [M − H]− | (18:0/22:7); (18:1_22:6) | C46H75NO10P |
| 834.5290 | [M − H]− | (18:0/22:6); (20:4_20:2) | C46H77NO10P |
| 836.5450 | [M − H]− | (18:0/22:5); (18:1_22:4); (20:1/20:4) | C46H79NO10P |
| 838.5603 | [M − H]− | (18:0_22:4) | C46H81NO10P |
| 840.5760 | [M − H]− | (18:0/22:3) | C46H83NO10P |
| 844.6070 | [M − H]− | (18:0/22:1); (16:0_24:1); (18:1_22:0) | C46H87NO10P |
| 856.5134 | [M − H]− | (20:3_22:6) | C48H75NO10P |
| 858.5290 | [M − H]− | (20:3_22:5) | C48H77NO10P |
| 860.5450 | [M − H]− | (20:0_22:7); (20:1/22:6) | C48H79NO10P |
| Patient | Sex | Age at Skin Biopsy (yr) | Age at PD Onset (yr) | PARK2 Mutation | HY Stage |
|---|---|---|---|---|---|
| #1 | F | 36 | 28 | p.Cys253Tyr */ex5del | 1 |
| #2 | M | 64 | 47 | ex3-4del/ex3-4del | 3 |
| #3 | M | 38 | 18 | ex2-3del/ex2del | 4 |
| #4 | M | 65 | 33 | ex2del/ex2-4del | 4 |
| #5 | M | 62 | 20 | ex2del/ex2-4del | 5 |
| PC | oPC/pPC | PE | oPE/pPE | PI | SM |
|---|---|---|---|---|---|
| 31:2 ↑ 33:0 ↑ 33:1 ↑ 33:2 ↑ 34:0 ↑ 34:1 ↑ 34:4 ↑ 35:1 ↑ 35:2 ↑ 36:1 ↑ 36:2 ↑ 37:1 ↑ 38:4 ↑ 40:5 ↑ 40:6 ↑ 28:0 ↓ 28:1 ↓ 29:1 ↓ 30:0 ↓ 30:1 ↓ 32:1 ↓ 32:2 ↓ 36:4 ↓ 38:2 ↓ 39:3 ↓ | 34:0 ↑ 36:4 ↑ 38:4 ↑ 32:1 ↓ 32:2 ↓ | 36:1 ↑ 37:4 ↑ 38:4 ↑ 40:5 ↑ 32:1 ↓ 36:2 ↓ 36:4 ↓ 36:5 ↓ 38:2 ↓ 38:3 ↓ 38:6 ↓ | 38:5 ↑ 39:5 ↑ 39:6 ↑ 40:6 ↑ 32:2 ↓ 34:3 ↓ 36:6 ↓ 38:6 ↓ | 35:1 ↑ 35:2 ↑ 38:4 ↑ 38:5 ↑ 40:4 ↑ 40:5 ↑ 37:3 ↓ 38:3 ↓ 39:4 ↓ | 34:0;2 ↑ 35:1;2 ↑ 36:2:2 ↓ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calvano, C.D.; Ventura, G.; Sardanelli, A.M.M.; Savino, L.; Losito, I.; De Michele, G.; Palmisano, F.; Cataldi, T.R.I. Searching for Potential Lipid Biomarkers of Parkinson’s Disease in Parkin-Mutant Human Skin Fibroblasts by HILIC-ESI-MS/MS: Preliminary Findings. Int. J. Mol. Sci. 2019, 20, 3341. https://doi.org/10.3390/ijms20133341
Calvano CD, Ventura G, Sardanelli AMM, Savino L, Losito I, De Michele G, Palmisano F, Cataldi TRI. Searching for Potential Lipid Biomarkers of Parkinson’s Disease in Parkin-Mutant Human Skin Fibroblasts by HILIC-ESI-MS/MS: Preliminary Findings. International Journal of Molecular Sciences. 2019; 20(13):3341. https://doi.org/10.3390/ijms20133341
Chicago/Turabian StyleCalvano, Cosima D., Giovanni Ventura, Anna Maria M. Sardanelli, Laura Savino, Ilario Losito, Giuseppe De Michele, Francesco Palmisano, and Tommaso R. I. Cataldi. 2019. "Searching for Potential Lipid Biomarkers of Parkinson’s Disease in Parkin-Mutant Human Skin Fibroblasts by HILIC-ESI-MS/MS: Preliminary Findings" International Journal of Molecular Sciences 20, no. 13: 3341. https://doi.org/10.3390/ijms20133341
APA StyleCalvano, C. D., Ventura, G., Sardanelli, A. M. M., Savino, L., Losito, I., De Michele, G., Palmisano, F., & Cataldi, T. R. I. (2019). Searching for Potential Lipid Biomarkers of Parkinson’s Disease in Parkin-Mutant Human Skin Fibroblasts by HILIC-ESI-MS/MS: Preliminary Findings. International Journal of Molecular Sciences, 20(13), 3341. https://doi.org/10.3390/ijms20133341







