Transcriptomic Studies Reveal that the Rhizobium leguminosarum Serine/Threonine Protein Phosphatase PssZ has a Role in the Synthesis of Cell-Surface Components, Nutrient Utilization, and Other Cellular Processes
Abstract
1. Introduction
2. Results
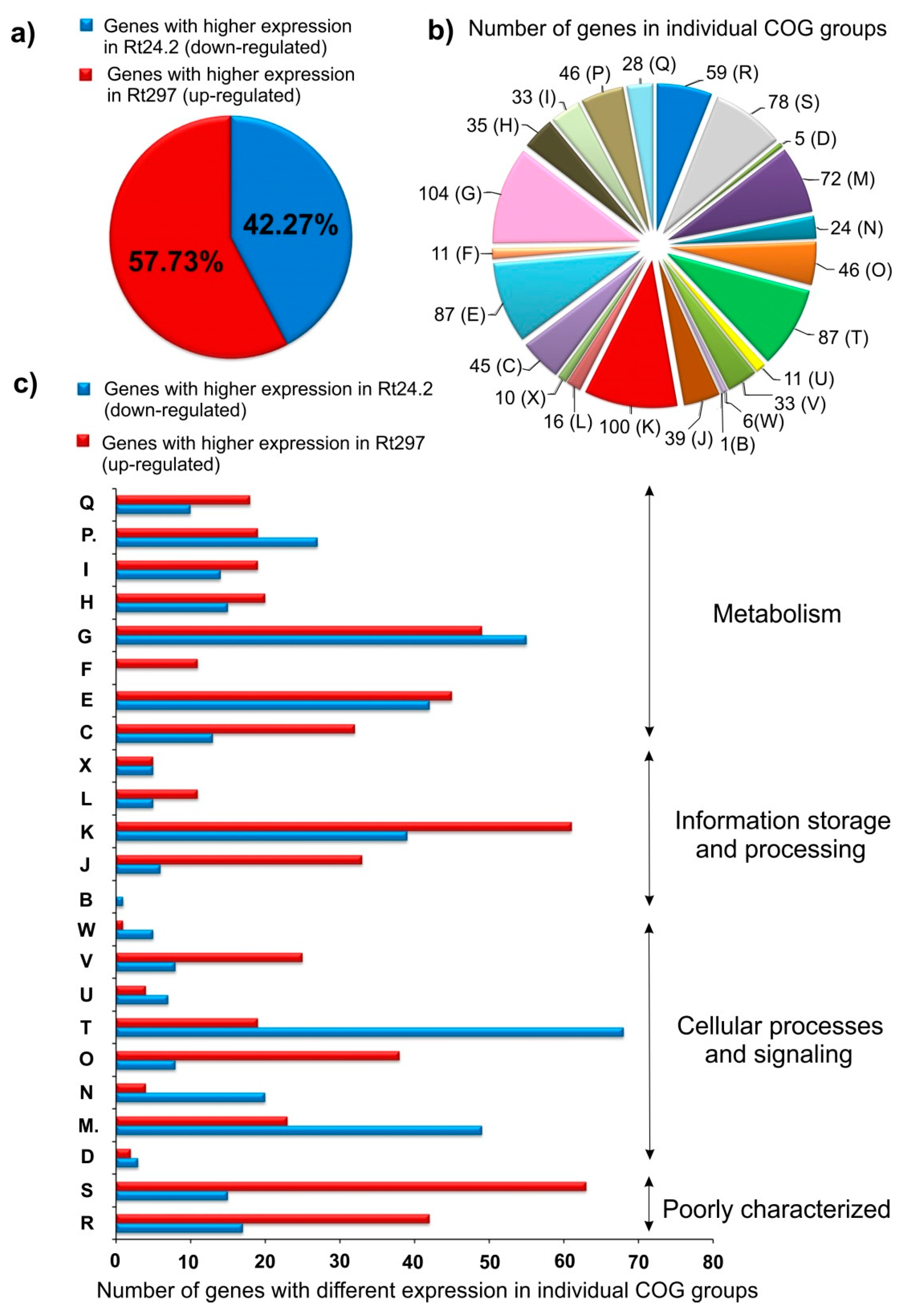
2.1. RNA-Seq Analysis of the Wild-Type Rt24.2 and pssZ Mutant Rt297 Strains
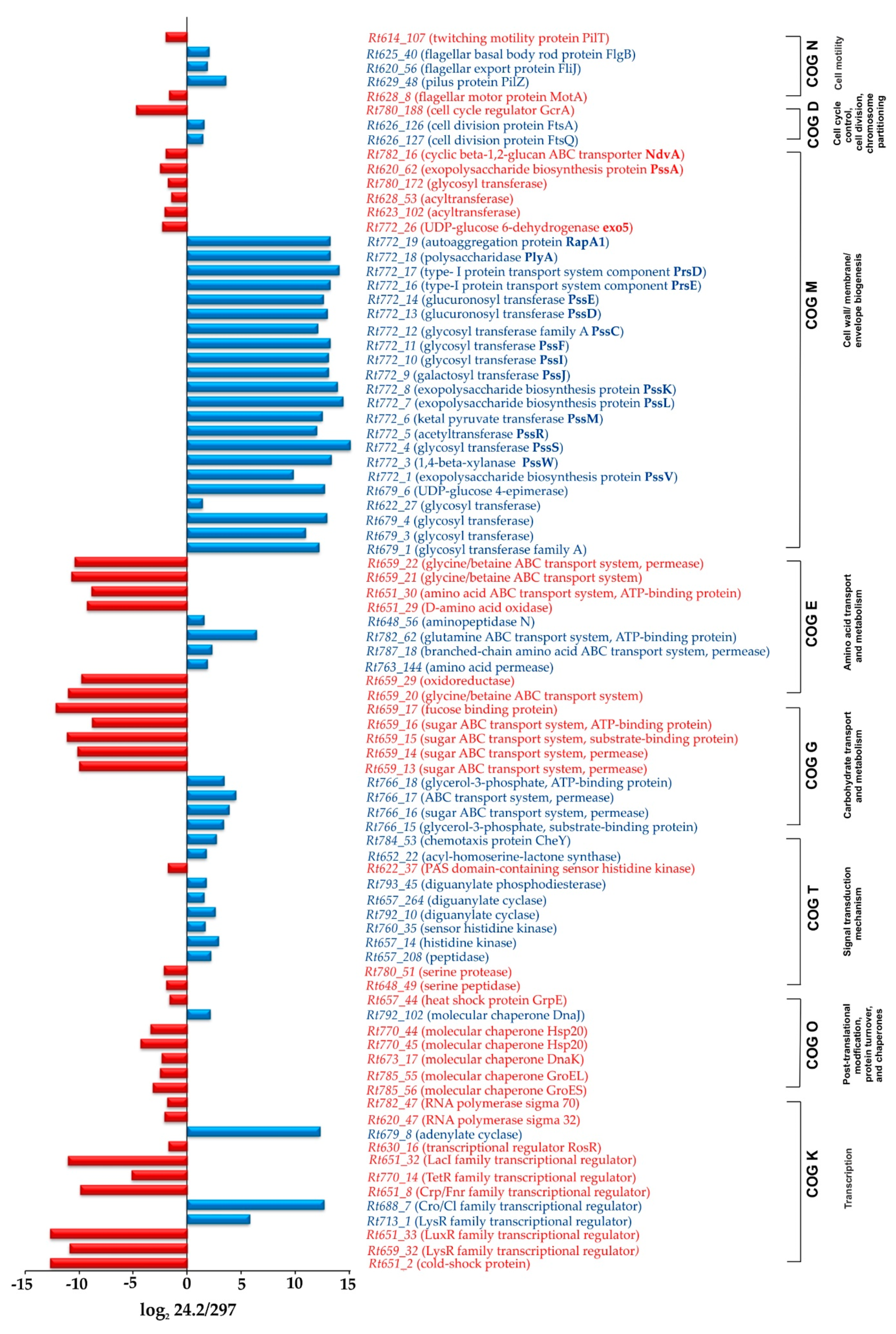
2.1.1. Transcription, Translation, and Signal Transduction Mechanisms
2.1.2. Carbon and Amino Acid Transport and Metabolism
2.1.3. Synthesis of Cell-Surface Components
2.1.4. Genes Involved in Cell Cycle and Motility
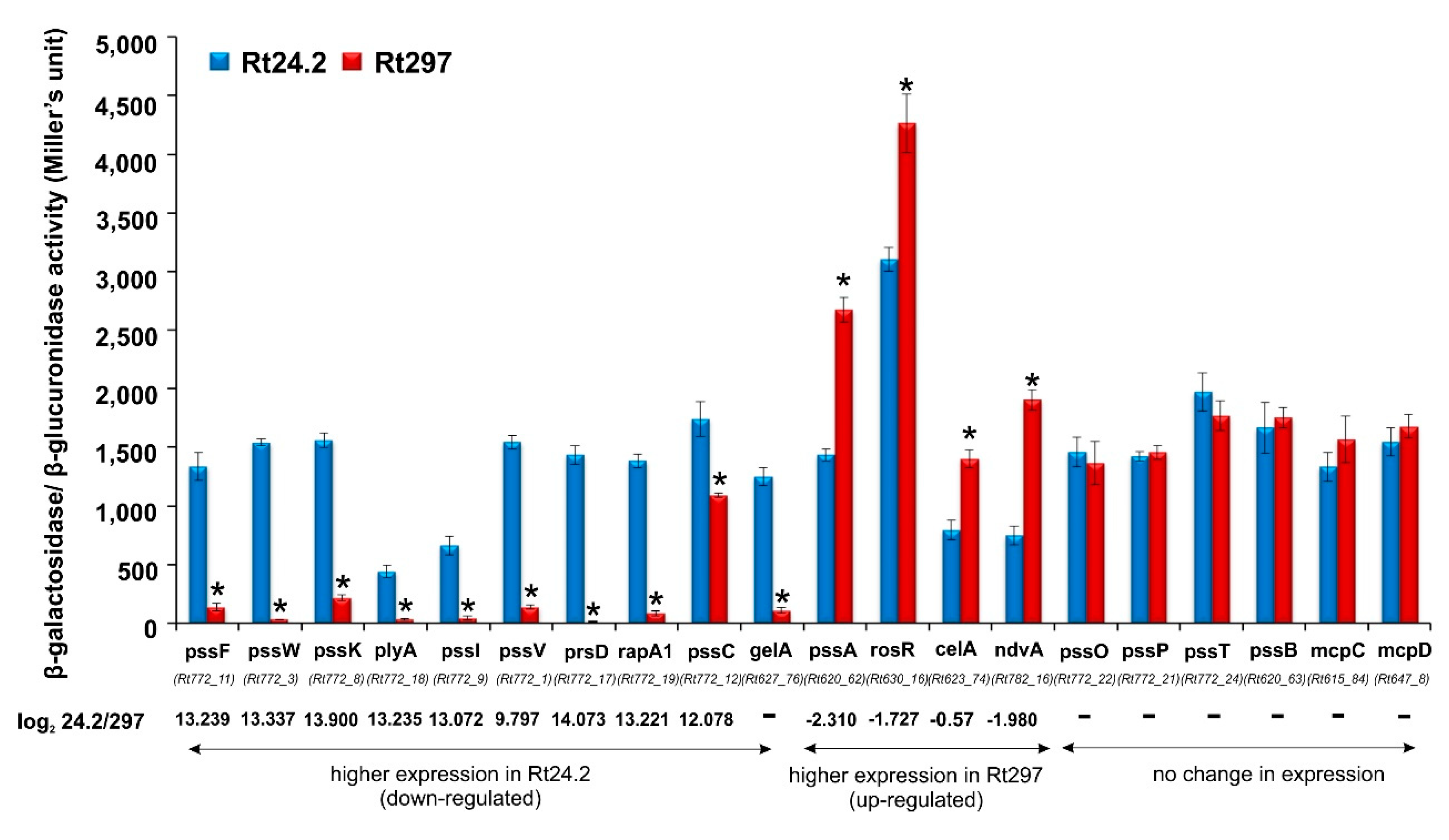
2.1.5. Analysis of Transcriptional Fusions in Rt297 and Rt24.2 Strains
2.2. Phenotypic Characteristics of the Wild-Type Strain Rt24.2 and Its Derivatives
2.2.1. Growth at a Wide Range of Temperatures
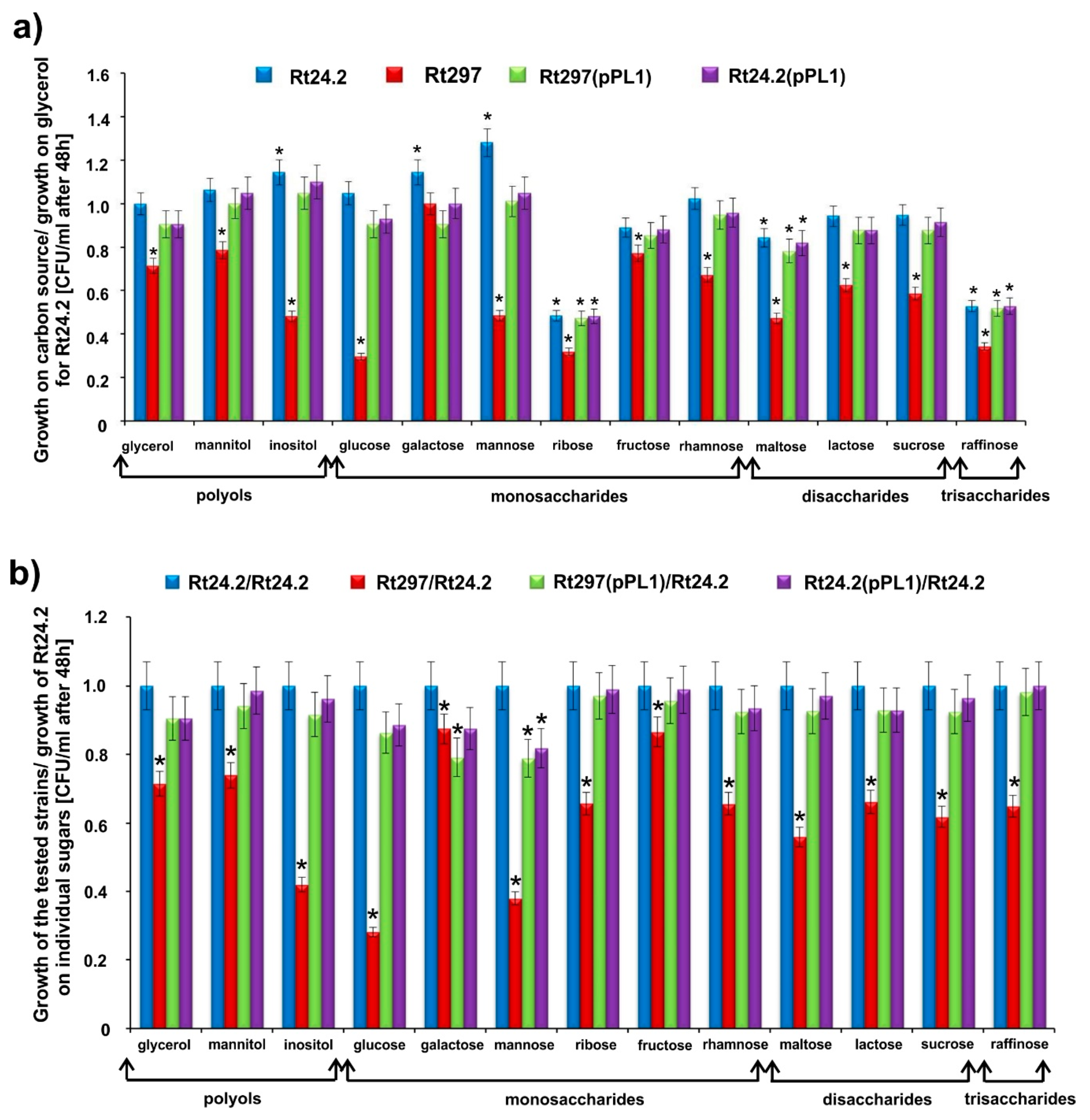
2.2.2. Utilization of Different Sugars
2.2.3. Synthesis of Different Polysaccharides
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains, Plasmids, and Culture Conditions
4.2. Isolation of Total RNA and Synthesis of cDNA Libraries
4.3. RNA-Seq Data Analysis
4.4. Analysis of Transcriptional Fusions
4.5. Isolation of Surface Polysaccharides
4.5.1. EPS
4.5.2. Gel-Forming Polysaccharide
4.5.3. Capsular Polysaccharide
4.5.4. Cyclic β-Glucans
4.5.5. Glucomannan
4.5.6. Determination of PS Amounts Synthesized by Rhizobial Strains
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EPS | exopolysaccharide |
| LPS | lipopolysaccharide |
| PS | polysaccharide |
| LMW | low-molecular-weight |
| HMW | High-molecular-weight |
| CPS | capsular polysaccharide |
| NP | neutral polysaccharide |
| GPS | gel-forming polysaccharide |
| CG | cyclic β-glucan |
| IT | infection thread |
| STP | serine/threonine protein phosphatases |
| STK | Hanks-type serine/threonine kinase |
| DEG | differentially expressed gene |
| COG | cluster of orthologous group |
References
- Raynaud, X.; Nunan, N. Spatial ecology of bacteria at the microscale in soil. PLoS ONE 2014, 9, e8721. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Guerrero, I.; Acosta-Jurado, S.; Del Cerro, P.; Navarro-Gómez, P.; López-Baena, F.J.; Ollero, F.J.; Vinardell, J.M.; Pérez-Montaño, F. Transcriptomic Studies of the Effect of nod Gene-Inducing Molecules in Rhizobia: Different Weapons, One Purpose. Genes 2018, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Janczarek, M.; Rachwał, K.; Marzec, A.; Grządziel, J.; Palusińska-Szysz, M. Signal molecules and cell-surface components involved in early stages of the legume-rhizobium interactions. Appl. Soil Ecol. 2015, 85, 94–113. [Google Scholar] [CrossRef]
- Sánchez-Cañizares, C.; Jorrín, B.; Durán, D.; Nadendla, S.; Albareda, M.; Rubio-Sanz, L.; Lanza, M.; González-Guerrero, M.; Prieto, R.I.; Brito, B.; et al. Genomic Diversity in the Endosymbiotic Bacterium Rhizobium leguminosarum. Genes 2018, 9, 60. [Google Scholar] [CrossRef] [PubMed]
- Young, J.P.; Crossman, L.C.; Johnston, A.W.; Thomson, N.R.; Ghazoui, Z.F.; Hull, K.H.; Wexler, M.; Curson, A.R.; Todd, J.D.; Poole, P.S.; et al. The genome of Rhizobium leguminosarum has recognizable core and accessory components. Genome Biol. 2006, 7, R34. [Google Scholar] [CrossRef] [PubMed]
- Zahran, H.H. Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol. Mol. Biol. Rev. 1999, 63, 968–989. [Google Scholar] [PubMed]
- Graham, P.H.; Vance, C.P. Nitrogen fixation in perspective: An overview of research an extension needs. Field Crops Res. 2000, 65, 93–106. [Google Scholar] [CrossRef]
- Santi, C.; Bogusz, D.; Franche, C. Biological nitrogen fixation in non-legume plants. Ann. Bot. 2013, 111, 743–767. [Google Scholar] [CrossRef]
- Downie, J.A. The roles of extracellular proteins, polysaccharides and signals in the interactions of rhizobia with legume roots. FEMS Microbiol. Rev. 2010, 34, 150–170. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, J.; Zhu, H. Genetic and Molecular Mechanisms Underlying Symbiotic Specificity in Legume-Rhizobium Interactions. Front. Plant Sci. 2018, 9, 313. [Google Scholar] [CrossRef]
- López-Baena, F.J.; Ruiz-Sainz, J.E.; Rodríguez-Carvajal, M.A.; Vinardell, J.M. Bacterial molecular signals in the Sinorhizobium fredii-soybean symbiosis. Int. J. Mol. Sci. 2016, 17, 755. [Google Scholar] [CrossRef] [PubMed]
- Kawaharada, Y.; Kelly, S.; Nielsen, M.W.; Hjuler, C.T.; Gysel, K.; Muszyński, A.; Carlson, R.W.; Thygesen, M.B.; Sandal, N.; Asmussen, M.H.; et al. Receptor-mediated exopolysaccharide perception controls bacterial infection. Nature 2015, 523, 308–312. [Google Scholar] [CrossRef] [PubMed]
- Muszyński, A.; Heiss, C.; Hjuler, C.T.; Sullivan, J.T.; Kelly, S.J.; Thygesen, M.B.; Stougaard, J.; Azadi, P.; Carlson, R.W.; Ronson, C.W. Structures of Exopolysaccharides Involved in Receptor-mediated Perception of Mesorhizobium loti by Lotus japonicus. J. Biol. Chem. 2016, 291, 20946–20961. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Barthelemy-Delaux, C.; Marburger, D.; Delaux, P.; Conley, S.; Ané, J. Effect of drought on Bradyrhizobium japonicum populations in Midwest soils. Plant Soil 2014, 382, 165–173. [Google Scholar] [CrossRef]
- Jaszek, M.; Janczarek, M.; Kuczyński, K.; Piersiak, T.; Grzywnowicz, K. The response of the Rhizobium leguminosarum bv. trifolii wild-type and exopolysaccharide-deficient mutants to oxidative stress. Plant Soil 2014, 376, 75–94. [Google Scholar] [CrossRef]
- López-Leal, G.; Tabche, M.L.; Castillo-Ramírez, S.; Mendoza-Vargas, A.; Ramírez-Romero, M.A.; Dávila, G. RNA-Seq analysis of the multipartite genome of Rhizobium etli CE3 shows different replicon contributions under heat and saline shock. BMC Genom. 2014, 15, 1–15. [Google Scholar] [CrossRef]
- Janczarek, M.; Rachwał, K.; Cieśla, J.; Ginalska, G.; Bieganowski, A. Production of exopolysaccharide by Rhizobium leguminosarum bv. trifolii and its role in bacterial attachment and surface properties. Plant Soil 2015, 388, 211–227. [Google Scholar] [CrossRef]
- Del Cerro, P.; Pérez-Montaño, F.; Gil-Serrano, A.; López-Baena, F.J.; Megías, M.; Hungria, M.; Ollero, F.J. The Rhizobium tropici CIAT 899 NodD2 protein regulates the production of Nod factors under salt stress in a flavonoid-independent manner. Sci. Rep. 2017, 7, 46712. [Google Scholar] [CrossRef]
- Kopycińska, M.; Lipa, P.; Cieśla, J.; Kozieł, M.; Janczarek, M. Extracellular polysaccharide protects Rhizobium leguminosarum cells against zinc stress in vitro and during symbiosis with clover. Environ. Microbiol. Rep. 2018, 10, 355–368. [Google Scholar] [CrossRef]
- Laus, M.C.; Logman, T.J.; Lamers, G.E.; Van Brussel, A.A.; Carlson, R.W.; Kijne, J.W. A novel polar surface polysaccharide from Rhizobium leguminosarum binds host plant lectin. Mol. Microbiol. 2006, 59, 1704–1713. [Google Scholar] [CrossRef] [PubMed]
- Bogino, P.C.; Oliva, M.D.; Sorroche, F.G.; Giordano, W. The role of bacterial biofilms and surface components in plant-bacterial associations. Int. J. Mol. Sci. 2013, 14, 15838–15859. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.; Wilkinson, A.; Krehenbrink, M.; Russo, D.M.; Zorreguieta, A.; Downie, J.A. Glucomannan-mediated attachment of Rhizobium leguminosarum to pea root hairs is required for competitive nodule infection. J. Bacteriol. 2008, 190, 4706–4715. [Google Scholar] [CrossRef] [PubMed]
- Russo, D.M.; Abdian, P.L.; Posadas, D.M.; Williams, A.; Vozza, N.; Giordano, W.; Kannenberg, E.; Downie, J.A.; Zorreguieta, A. Lipopolysaccharide O-chain core region required for cellular cohesion and compaction of in vitro and root biofilms developed by Rhizobium leguminosarum. Appl. Environ. Microbiol. 2015, 81, 1013–1023. [Google Scholar] [CrossRef] [PubMed]
- Sorroche, F.G.; Spesia, M.B.; Zorreguieta, A.; Giordano, W. A positive correlation between bacterial autoaggregation and biofilm formation in native Sinorhizobium meliloti isolates from Argentina. Appl. Environ. Microbiol. 2012, 78, 4092–4101. [Google Scholar] [CrossRef] [PubMed]
- Rinaudi, L.V.; Giordano, W. An integrated view of biofilm formation in rhizobia. FEMS Microbiol. Lett. 2010, 304, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.P.; Walker, G.C. Succinoglycan is required for initiation and elongation of infection threads during nodulation of alfalfa by Rhizobium meliloti. J. Bacteriol. 1998, 180, 5183–5191. [Google Scholar]
- Margaret-Oliver, I.; Lei, W.; Parada, M.; Rodríguez-Carvajal, M.A.; Crespo-Rivas, J.C.; Hidalgo, Á.; Gil-Serrano, A.; Moreno, J.; Rodríguez-Navarro, D.N.; Buendía-Clavería, A.; et al. Sinorhizobium fredii HH103 does not strictly require KPS and/or EPS to nodulate Glycyrrhiza uralensis, an indeterminate nodule-forming legume. Arch. Microbiol. 2012, 194, 87–102. [Google Scholar] [CrossRef]
- D’Haeze, W.; Glushka, J.; De Rycke, R.; Holsters, M.; Carlson, R.W. Structural characterization of extracellular polysaccharides of Azorhizobium caulinodans and importance for nodule initiation on Sesbania rostrata. Mol. Microbiol. 2004, 52, 485–500. [Google Scholar] [CrossRef]
- Rodríguez-Navarro, D.N.; Rodríguez-Carvajal, M.A.; Acosta-Jurado, S.; Soto, M.J.; Margaret, I.; Crespo-Rivas, J.C.; Sanjuan, J.; Temprano, F.; Gil-Serrano, A.; Ruiz-Sainz, J.E.; et al. Structure and biological roles of Sinorhizobium fredii HH103 exopolysaccharide. PLoS ONE 2014, 18, e115391. [Google Scholar] [CrossRef]
- Ivashina, T.V.; Khmelnitsky, M.I.; Shlyapnikov, M.G.; Kanapin, A.A.; Ksenzenko, V.N. The pss4 gene from Rhizobium leguminosarum by viciae VF39: Cloning, sequence and the possible role in polysaccharide production and nodule formation. Gene 1994, 150, 111–116. [Google Scholar] [CrossRef]
- Janczarek, M.; Urbanik-Sypniewska, T. Expression of the Rhizobium leguminosarum bv. trifolii pssA gene, involved in exopolysaccharide synthesis, is regulated by RosR, phosphate, and the carbon source. J. Bacteriol. 2013, 195, 3412–3423. [Google Scholar] [CrossRef] [PubMed]
- Van Workum, W.A.T.; van Slageren, S.; van Brussel, A.A.N.; Kijne, J.W. Role of exopolysaccharides of Rhizobium leguminosarum bv. viciae as host plant-specific molecules required for infection thread formation during nodulation of Vicia sativa. Mol. Plant Microbe Interact. 1998, 11, 1233–1241. [Google Scholar] [CrossRef]
- Janczarek, M. Environmental signals and regulatory pathways that influence exopolysaccharide production in rhizobia. Int. J. Mol. Sci. 2011, 12, 7898–7933. [Google Scholar] [CrossRef] [PubMed]
- Marczak, M.; Mazur, A.; Koper, P.; Żebracki, K.; Skorupska, A. Synthesis of rhizobial exopolysaccharides and their importance for symbiosis with legume plants. Genes 2017, 8, 360. [Google Scholar] [CrossRef] [PubMed]
- Robertsen, B.K.; Aman, P.; Darvill, A.G.; McNeil, M.; Albersheim, P. Host-Symbiont Interactions: V. The structure of acidic extracellular polysaccharides secreted by Rhizobium leguminosarum and Rhizobium trifolii. Plant Physiol. 1981, 67, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Canter Cremers, H.C.J.; Stevens, K.; Lugtenberg, B.J.J.; Wijffelman, C.A.; Batley, M.; Redmond, J.W.; Breedveld, M.; Zevenhuizen, L.P.T.M. Unusual structure of the exopolysaccharide of Rhizobium leguminosarum biovar viciae strain 248. Carbohydr. Res. 1991, 218, 185–200. [Google Scholar] [CrossRef]
- O’Neill, M.A.; Darvill, A.G.; Albersheim, P. The degree of esterification and points of substitution by O-acetyl and O-(3-hydroxybutanoyl) groups in the acidic extracellular polysaccharides secreted by Rhizobium leguminosarum biovars viciae, trifolii, and phaseoli are not related to host range. J. Biol. Chem. 1991, 266, 9549–9555. [Google Scholar]
- Philip-Hollingsworth, S.; Hollingsworth, R.I.; Dazzo, F.B.; Djordjevic, M.A.; Rolfe, B.G. The effect of interspecies transfer of Rhizobium host-specific nodulation genes on acidic polysaccharide structure and in situ binding by host lectin. J. Biol. Chem. 1989, 264, 5710–5714. [Google Scholar]
- Breedveld, M.W.; Cremers, H.C.; Batley, M.; Posthumus, M.A.; Zevenhuizen, L.P.; Wijffelman, C.A.; Zehnder, A.J. Polysaccharide synthesis in relation to nodulation behavior of Rhizobium leguminosarum. J. Bacteriol. 1993, 175, 750–757. [Google Scholar] [CrossRef]
- Van Workum, W.A.; Canter Cremers, H.C.; Wijfjes, A.H.; van der Kolk, C.; Wijffelman, C.A.; Kijne, J.W. Cloning and characterization of four genes of Rhizobium leguminosarum bv. trifolii involved in exopolysaccharide production and nodulation. Mol. Plant Microbe Interact. 1997, 10, 290–301. [Google Scholar] [CrossRef] [PubMed]
- Pollock, T.J.; van Workum, W.A.; Thorne, L.; Mikolajczak, M.J.; Yamazaki, M.; Kijne, J.W.; Armentrout, R.W. Assignment of biochemical functions to glycosyl transferase genes which are essential for biosynthesis of exopolysaccharides in Sphingomonas strain S88 and Rhizobium leguminosarum. J. Bacteriol. 1998, 180, 586–593. [Google Scholar] [PubMed]
- Janczarek, M.; Rachwał, K. Mutation in the pssA gene involved in exopolysaccharide synthesis leads to several physiological and symbiotic defects in Rhizobium leguminosarum bv. trifolii. Int. J. Mol. Sci. 2013, 14, 23711–23735. [Google Scholar] [CrossRef] [PubMed]
- Ivashina, T.V.; Ksenzenko, V.N. Exopolysaccharide biosynthesis in Rhizobium leguminosarum from genes to functions. In The Complex World of Polysaccharides; Karunaratne, D.N., Ed.; InTech: Rijeka, Croatia, 2012; pp. 99–127. ISBN 978-953-51-0819-1. [Google Scholar]
- Janczarek, M.; Rachwał, K.; Kopcińska, J. Genetic characterization of the Pss region and the role of PssS in exopolysaccharide production and symbiosis of Rhizobium leguminosarum bv. trifolii with clover. Plant Soil 2015, 396, 257–275. [Google Scholar] [CrossRef]
- Ivashina, T.V.; Fedorova, E.E.; Ashina, N.P.; Kalinchuk, N.A.; Druzhinina, T.N.; Shashkov, A.S.; Shibaev, V.N.; Ksenzenko, V.N. Mutation in the pssM gene encoding ketal pyruvate transferase leads to disruption of Rhizobium leguminosarum bv. viciae-Pisum sativum symbiosis. J. Appl. Microbiol. 2010, 109, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Mazur, A.; Marczak, M.; Król, J.E.; Skorupska, A. Topological and transcriptional analysis of pssL gene product: A putative Wzx-like exopolysaccharide translocase in Rhizobium leguminosarum bv. trifolii TA1. Arch. Microbiol. 2005, 184, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mazur, A.; Król, J.E.; Marczak, M.; Skorupska, A. Membrane topology of PssT, the transmembrane protein component of the type I exopolysaccharide transport system in Rhizobium leguminosarum bv. trifolii strain TA1. J. Bacteriol. 2003, 185, 2503–2511. [Google Scholar] [CrossRef]
- Marczak, M.; Dźwierzyńska, M.; Skorupska, A. Homo- and heterotypic interactions between Pss proteins involved in the exopolysaccharide transport system in Rhizobium leguminosarum bv. trifolii. Biol. Chem. 2013, 394, 541–559. [Google Scholar] [CrossRef]
- Marczak, M.; Matysiak, P.; Kutkowska, J.; Skorupska, A. PssP2 is a polysaccharide co-polymerase involved in exopolysaccharide chain-length determination in Rhizobium leguminosarum. PLoS ONE 2014, 9, e109106. [Google Scholar] [CrossRef]
- Sadykov, M.R.; Ivashina, T.V.; Kanapin, A.A.; Shliapnikov, M.G.; Ksenzenko, V.N. Structure-functional organization of exopolysaccharide biosynthetic genes in Rhizobium leguminosarum bv. viciae VF39. Mol. Biol. 1998, 32, 797–804. [Google Scholar]
- Król, J.E.; Mazur, A.; Marczak, M.; Skorupska, A. Syntenic arrangements of the surface polysaccharide biosynthesis genes in Rhizobium leguminosarum. Genomics 2007, 89, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Janczarek, M.; Rachwał, K.; Turska-Szewczuk, A. A mutation in pssE affects exopolysaccharide synthesis by Rhizobium leguminosarum bv. trifolii, its surface properties, and symbiosis with clover. Plant Soil 2017, 417, 331–347. [Google Scholar] [CrossRef]
- Borthakur, D.; Johnston, A.W. Sequence of psi, a gene on the symbiotic plasmid of Rhizobium phaseoli which inhibits exopolysaccharide synthesis and nodulation and demonstration that its transcription is inhibited by psr, another gene on the symbiotic plasmid. Mol. Gen. Genet. 1987, 207, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Borthakur, D.; Barker, R.F.; Latchford, J.W.; Rossen, L.; Johnston, A.W. Analysis of pss genes of Rhizobium leguminosarum required for exopolysaccharide synthesis and nodulation of peas: Their primary structure and their interaction with psi and other nodulation genes. Mol. Gen. Genet. 1988, 213, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Reeve, W.G.; Dilworth, M.J.; Tiwari, R.P.; Glenn, A.R. Regulation of exopolysaccharide production in Rhizobium leguminosarum biovar viciae WSM710 involves exoR. Microbiology 1997, 143, 1951–1958. [Google Scholar] [CrossRef] [PubMed]
- Janczarek, M.; Skorupska, A. Modulation of rosR expression and exopolysaccharide production in Rhizobium leguminosarum bv. trifolii by phosphate and clover root exudates. Int. J. Mol. Sci. 2011, 12, 4132–4155. [Google Scholar] [CrossRef] [PubMed]
- Rachwał, K.; Matczyńska, E.; Janczarek, M. Transcriptome profiling of a Rhizobium leguminosarum bv. trifolii rosR mutant reveals the role of the transcriptional regulator RosR in motility, synthesis of cell-surface components, and other cellular processes. BMC Genomics 2015, 16, 1111. [Google Scholar] [CrossRef]
- Lipa, P.; Vinardell, J.M.; Kopcińska, J.; Zdybicka-Barabas, A.; Janczarek, M. Mutation in the pssZ Gene Negatively Impacts Exopolysaccharide Synthesis, Surface Properties, and Symbiosis of Rhizobium leguminosarum bv. trifolii with Clover. Genes 2018, 9, 369. [Google Scholar] [CrossRef]
- Janczarek, M.; Vinardell, J.M.; Lipa, P.; Karaś, M. Hanks-Type Serine/Threonine Protein Kinases and Phosphatases in Bacteria: Roles in Signaling and Adaptation to Various Environments. Int. J. Mol. Sci. 2018, 19, 2872. [Google Scholar] [CrossRef]
- De Vinney, R.; Steele-Mortimer, O.; Finlay, B.B. Phosphatases and kinases delivered to the host cell by bacterial pathogens. Trends Microbiol. 2000, 8, 29–33. [Google Scholar] [CrossRef]
- Dworkin, J. Ser/Thr phosphorylation as a regulatory mechanism in bacteria. Curr. Opin. Microbiol. 2015, 24, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Mijakovic, I.; Grangeasse, C.; Turgay, K. Exploring the diversity of protein modifications: Special bacterial phosphorylation systems. FEMS Microbiol. Rev. 2016, 40, 398–417. [Google Scholar] [CrossRef] [PubMed]
- Rachwał, K.; Boguszewska, A.; Kopcińska, J.; Karas, M.; Tchórzewski, M.; Janczarek, M. The regulatory protein RosR affects Rhizobium leguminosarum bv. trifolii protein profiles, cell surface properties, and symbiosis with clover. Front. Microbiol. 2016, 7, 1302. [Google Scholar] [CrossRef]
- Tatusov, R.L.; Natale, D.A.; Garkavtsev, I.V.; Tatusova, T.A.; Shankavaram, U.T.; Rao, B.S.; Kiryutin, B.; Galperin, M.Y.; Fedorova, N.D.; Koonin, E.V. The COG database: New developments in phylogenetic classification of proteins from complete genomes. Nucl. Acids Res. 2001, 29, 22–28. [Google Scholar] [CrossRef]
- Schäper, S.; Krol, E.; Skotnicka, D.; Kaever, V.; Hilker, R.; Søgaard-Andersen, L.; Becker, A. Cyclic Di-GMP Regulates Multiple Cellular Functions in the Symbiotic Alphaproteobacterium Sinorhizobium meliloti. J. Bacteriol. 2015, 198, 521–535. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.T.; Matewish, J.M.; Kessler, J.L.; Hyodo, M.; Hayakawa, Y.; Lory, S. A cyclic-di-GMP receptor required for bacterial exopolysaccharide production. Mol. Microbiol. 2007, 65, 1474–1484. [Google Scholar] [CrossRef]
- Baraquet, C.; Murakami, K.; Parsek, M.R.; Harwood, C.S. The FleQ protein from Pseudomonas aeruginosa functions as both a repressor and an activator to control gene expression from the pel operon promoter in response to c-di-GMP. Nucl. Acids Res. 2012, 40, 7207–7218. [Google Scholar] [CrossRef]
- Paul, K.; Nieto, V.; Carlquist, W.C.; Blair, D.F.; Harshey, R.M. The c-di-GMP binding protein YcgR controls flagellar motor direction and speed to affect chemotaxis by a “backstop brake” mechanism. Mol. Cell 2010, 38, 128–139. [Google Scholar] [CrossRef]
- Ross, P.; Weinhouse, H.; Aloni, Y.; Michaeli, D.; Weinberger-Ohana, P.; Mayer, R.; Braun, S.; de Vroom, E.; van der Marel, G.A.; van Boom, J.H.; et al. Regulation of cellulose synthesis in Acetobacter xylinum by cyclic diguanylic acid. Nature 1987, 325, 279–281. [Google Scholar] [CrossRef]
- Ryjenkov, D.A.; Simm, R.; Römling, U.; Gomelsky, M. The PilZ domain is a receptor for the second messenger c-di-GMP: The PilZ domain protein YcgR controls motility in enterobacteria. J. Biol. Chem. 2006, 281, 30310–30314. [Google Scholar] [CrossRef]
- Libby, E.A.; Goss, L.A.; Dworkin, J. The eukaryotic-like Ser/Thr kinase PrkC regulates the essential WalRK Two-Component System in Bacillus subtilis. PLoS Genet. 2015, 11, e1005275. [Google Scholar] [CrossRef] [PubMed]
- Brautigan, D.L. Protein Ser/Thr phosphatases--the ugly ducklings of cell signalling. FEBS J. 2013, 280, 324–345. [Google Scholar] [CrossRef] [PubMed]
- Esser, D.; Hoffmann, L.; Pham, T.K.; Bräsen, C.; Qiu, W.; Wright, P.C.; Albers, S.V.; Siebers, B. Protein phosphorylation and its role in archaeal signal transduction. FEMS Microbiol. Rev. 2016, 40, 625–647. [Google Scholar] [CrossRef] [PubMed]
- Muszyński, A.; Laus, M.; Kijne, J.W.; Carlson, R.W. Structures of the lipopolysaccharides from Rhizobium leguminosarum RBL5523 and its UDP-glucose dehydrogenase mutant (exo5). Glycobiology 2011, 21, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Jurado, S.; Navarro-Gómez, P.; Crespo-Rivas, J.C.; Medina, C.; Murdoch, P.S.; Cuesta-Berrio, L.; Rodríguez-Carvajal, M.A.; Ruiz-Sainz, J.E.; Vinardell, J.M. The Sinorhizobium (Ensifer) fredii HH103 rkp-2 region is involved in the biosynthesis of lipopolysaccharide and exopolysaccharide but not in K-antigen polysaccharide production. Plant Soil 2017, 417, 415–431. [Google Scholar] [CrossRef]
- Kawaharada, Y.; Kiyota, H.; Eda, S.; Minamisawa, K.; Mitsui, H. Structural characterization of neutral and anionic glucans from Mesorhizobium loti. Carbohydr. Res. 2008, 343, 2422–2427. [Google Scholar] [CrossRef]
- Acosta-Jurado, S.; Alias-Villegas, C.; Navarro-Gómez, P.; Zehner, S.; Murdoch, P.D.; Rodríguez-Carvajal, M.A.; Soto, M.J.; Ollero, F.J.; Ruiz-Sainz, J.E.; Göttfert, M.; et al. The Sinorhizobium fredii HH103 MucR1 Global Regulator Is Connected with the nod Regulon and Is Required for Efficient Symbiosis with Lotus burttii and Glycine max cv. Williams. Mol. Plant Microbe Interact. 2016, 29, 700–712. [Google Scholar] [CrossRef]
- Crespo-Rivas, J.C.; Margaret, I.; Hidalgo, A.; Buendía-Clavería, A.M.; Ollero, F.J.; López-Baena, F.J.; del Socorro Murdoch, P.; Rodríguez-Carvajal, M.A.; Soria-Díaz, M.E.; Reguera, M.; et al. Sinorhizobium fredii HH103 cgs mutants are unable to nodulate determinate- and indeterminate nodule-forming legumes and overproduce an altered EPS. Mol. Plant Microbe Interact. 2009, 22, 575–588. [Google Scholar] [CrossRef]
- Vanderlinde, E.M.; Yost, C.K. Mutation of the sensor kinase chvG in Rhizobium leguminosarum negatively impacts cellular metabolism, outer membrane stability, and symbiosis. J. Bacteriol. 2012, 194, 768–777. [Google Scholar] [CrossRef]
- Pérez-Mendoza, D.; Sanjuán, J. Exploiting the commons: Cyclic diguanylate regulation of bacterial exopolysaccharide production. Curr. Opin. Microbiol. 2016, 30, 36–43. [Google Scholar] [CrossRef]
- Tamayo, R.; Tischler, A.D.; Camilli, A. The EAL domain protein VieA is a cyclic diguanylate phosphodiesterase. J. Biol. Chem. 2005, 280, 33324–33330. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Waters, C.M. A tangled web: Regulatory connections between quorum sensing and cyclic di-GMP. J. Bacteriol. 2012, 194, 4485–4493. [Google Scholar] [CrossRef] [PubMed]
- Prada-Ramírez, H.A.; Pérez-Mendoza, D.; Felipe, A.; Martínez-Granero, F.; Rivilla, R.; Sanjuán, J.; Gallegos, M.T. AmrZ regulates cellulose production in Pseudomonas syringae pv. tomato DC3000. Mol. Microbiol. 2016, 99, 960–977. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Zhang, G.; Wood, T.K. Escherichia coli BdcA controls biofilm dispersal in Pseudomonas aeruginosa and Rhizobium meliloti. BMC Res. Notes 2011, 4, 447. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Romdhane, S.B.; Beullens, S.; Kaever, V.; Lambrichts, I.; Fauvart, M.; Michiels, J. Genomic analysis of cyclic-di-GMP-related genes in rhizobial type strains and functional analysis in Rhizobium etli. Appl. Microbiol. Biotechnol. 2014, 98, 4589–4602. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Mendoza, D.; Rodríguez-Carvajal, M.Á.; Romero-Jiménez, L.; Farias Gde, A.; Lloret, J.; Gallegos, M.T.; Sanjuán, J. Novel mixed-linkage β-glucan activated by c-di-GMP in Sinorhizobium meliloti. Proc. Natl. Acad. Sci. USA 2015, 112, E757–E765. [Google Scholar] [CrossRef]
- Pérez-Mendoza, D.; Bertinetti, D.; Lorenz, R.; Gallegos, M.T.; Herberg, F.W.; Sanjuán, J. A novel c-di-GMP binding domain in glycosyltransferase BgsA is responsible for the synthesis of a mixed-linkage β-glucan. Sci. Rep. 2017, 7, 8997. [Google Scholar] [CrossRef]
- Alexandre, A.; Laranjo, M.; Oliveira, S. Global transcriptional response to heat shock of the legume symbiont Mesorhizobium loti MAFF303099 comprises extensive gene downregulation. DNA Res. 2014, 21, 195–206. [Google Scholar] [CrossRef]
- Gomes, D.F.; Batista, J.S.; Schiavon, A.L.; Andrade, D.S.; Hungria, M. Proteomic profiling of Rhizobium tropici PRF 81: Identification of conserved and specific responses to heat stress. BMC Microbiol. 2012, 12, 84. [Google Scholar] [CrossRef]
- Girvan, M.S.; Bullimore, J.; Pretty, J.N.; Osborn, A.M.; Ball, A.S. Soil type is the primary determinant of the composition of the total and active bacterial communities in arable soils. Appl. Environ. Microbiol. 2003, 69, 1800–1809. [Google Scholar] [CrossRef]
- González, V.; Santamaría, R.I.; Bustos, P.; Hernández-González, I.; Medrano-Soto, A.; Moreno-Hagelsieb, G.; Janga, S.C.; Ramírez, M.A.; Jiménez-Jacinto, V.; Collado-Vides, J.; et al. The partitioned Rhizobium etli genome: Genetic and metabolic redundancy in seven interacting replicons. Proc. Natl. Acad. Sci. USA 2006, 103, 3834–3839. [Google Scholar] [CrossRef] [PubMed]
- Prell, J.; Poole, P. Metabolic changes of rhizobia in legume nodules. Trends Microbiol. 2006, 14, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Galibert, F.; Finan, T.M.; Long, S.R.; Puhler, A.; Abola, P.; Ampe, F.; Barloy-Hubler, F.; Barnett, M.J.; Becker, A.; Boistard, P.; et al. The composite genome of the legume symbiont Sinorhizobium meliloti. Science 2001, 293, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Mauchline, T.H.; Fowler, J.E.; East, A.K.; Sartor, A.L.; Zaheer, R.; Hosie, A.H.; Poole, P.S.; Finan, T.M. Mapping the Sinorhizobium meliloti 1021 solute-binding protein-dependent transportome. Proc. Natl. Acad. Sci. USA 2006, 103, 17933–17938. [Google Scholar] [CrossRef] [PubMed]
- Vanderlinde, E.M.; Hynes, M.F.; Yost, C.K. Homoserine catabolism by Rhizobium leguminosarum bv. viciae 3841 requires a plasmid-borne gene cluster that also affects competitiveness for nodulation. Environ. Microbiol. 2014, 16, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Wielbo, J.; Marek-Kozaczuk, M.; Mazur, A.; Kubik-Komar, A.; Skorupska, A. Genetic and metabolic divergence within a Rhizobium leguminosarum bv. trifolii population recovered from clover nodules. Appl. Environ. Microbiol. 2010, 76, 4593–4600. [Google Scholar] [CrossRef]
- Wielbo, J.; Marek-Kozaczuk, M.; Kubik-Komar, A.; Skorupska, A. Increased metabolic potential of Rhizobium spp. is associated with bacterial competitiveness. Can. J. Microbiol. 2007, 53, 957–967. [Google Scholar] [CrossRef]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu. Rev. Plant Biol. 2006, 57, 233–266. [Google Scholar] [CrossRef]
- Bertin, C.; Yang, X.; Weston, L.A. The role of root exudates and allelochemicals in the rhizosphere. Plant Soil 2003, 256, 67–83. [Google Scholar] [CrossRef]
- Janczarek, M.; Skorupska, A. The Rhizobium leguminosarum bv. trifolii RosR: Transcriptional regulator involved in exopolysaccharide production. Mol. Plant Microbe Interact. 2007, 20, 867–881. [Google Scholar] [CrossRef]
- Spaink, H.P.; Wijffelman, C.A.; Pees, E.; Okker, R.J.; Lugtenberg, B.J. Rhizobium nodulation gene nodD as a determinant of host specificity. Nature 1987, 328, 337–340. [Google Scholar] [CrossRef]
- Yost, C.K.; Del Bel, K.L.; Quandt, J.; Hynes, M.F. Rhizobium leguminosarum methyl-accepting chemotaxis protein genes are down-regulated in the pea nodule. Arch. Microbiol. 2004, 182, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Janczarek, M.; Kalita, M.; Skorupska, A. New taxonomic markers for identification of Rhizobium leguminosarum and discrimination between closely related species. Arch. Microbiol. 2009, 191, 207–219. [Google Scholar] [CrossRef] [PubMed]
- Beringer, J.E. R factor transfer in Rhizobium leguminosarum. J. Gen. Microbiol. 1974, 84, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Bertani, G. Studies on lysogenesis. I. The mode of phage liberation by lysogenic Escherichia coli. J. Bacteriol. 1951, 62, 293–300. [Google Scholar] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Trapnell, C.; Roberts, A.; Goff, L.; Pertea, G.; Kim, D.; Kelley, D.R.; Pimentel, H.; Salzberg, S.L.; Rinn, J.L.; Pachter, L. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 2012, 7, 562–578. [Google Scholar] [CrossRef]
- Anders, S.; Pyl, P.T.; Huber, W. HTSeq—A Python framework to work with high-throughput sequencing data. Bioinformatics 2015, 31, 166–169. [Google Scholar] [CrossRef]
- R Development Core Team R: A language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008; ISBN 3-900051-07-0.
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. B 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Phylogenetic Classification of Proteins Encoded in Complete Genomes. Available online: http://www.ncbi.nlm.nih.gov/COG/ (accessed on 1 May 2019).
- Miller, J. Experiments in Molecular Genetics; Cold Spring Harbor Laboratory: New York, NY, USA, 1972; pp. 352–355. [Google Scholar]
- Loewus, F.A. Improvement in Anthrone Method for Determination of Carbohydrates. Anal. Chem. 1952, 24, 219. [Google Scholar] [CrossRef]
- Westphal, O.; Jann, K. Bacterial Lipopolysaccharides Extraction with Phenol-Water and Further Applications of the Procedure. Methods Carbohydr. Chem. 1965, 5, 83–91. [Google Scholar]


| Strains, Plasmids, and Primers | Characteristics | Source or Reference |
|---|---|---|
| Strains | ||
| Rt24.2 | wild-type strain Rhizobium leguminosarum bv. trifolii, clover microsymbiont, Rifr, Nxr | [101] |
| Rt297 | Rt24.2 pssZ::mTn5SSgusA40, Spr | [59] |
| Rt297(pPL1) | Rt297 carrying pssZ on pBBR1MCS-2 vector, Kmr | [59] |
| Rt24.2(pPL1) | Rt24.2 carrying pssZ on pBBR1MCS-2 vector, Kmr | [59] |
| Rt24.2(pMP220) | Rt24.2 carrying pMP220 vector, Rifr, Nxr,Tcr | This work |
| Rt297(pMP220) | Rt297 carrying pMP220 vector, Rifr, Nxr,Tcr | This work |
| Plasmids | ||
| pMP220 | IncP, mob, promoterless lacZ, Tcr | [102] |
| pFUS1P | pFUS1 with par cassette, promoterless gusA, Tcr | [103] |
| pPL1 | pBBR1MCS-2 carrying 1.8-kb SalI-XbaI fragment with the pssZ gene, Kmr | [59] |
| pPSS4 | pMP220 carrying 0.6-kb EcoRI-PstI fragment of the pssB promoter region | [58] |
| pNDV5 | pMP220 carrying 0.3-kb EcoRI-PstI fragment of the ndvA promoter region | [58] |
| pCEL9 | pMP220 carrying 0.72-kb EcoRI-PstI fragment of the celA promoter region | [58] |
| pGEL10 | pMP220 carrying 0.8-kbBglII-XbaI fragment of the gelA promoter region | [58] |
| pRAP11 | pMP220 carrying 0.9-kb BglII-XbaI fragment of the rapA1 promoter region | [58] |
| pPRS12 | pMP220 carrying 0.85-kb EcoRI-XbaI fragment of the prsD promoter region | [58] |
| pF65 | pMP220 carrying 0.65-kb BglII-PstI fragment of the pssF promoter region | [45] |
| pW74 | pMP220 carrying 0.74-kb EcoRI-PstI fragment of the pssW promoter region | [45] |
| pK48 | pMP220 carrying 0.48-kb EcoRI-PstI fragment of the pssK promoter region | [45] |
| pV90 | pMP220 carrying 0.9-kb KpnI-XbaI fragment of the pssV promoter region | [45] |
| pC55 | pMP220 carrying 0.55-kb EcoRI-SphI fragment of the pssC promoter region | [45] |
| pO66 | pMP220 carrying 0.65-kb BglII-PstI fragment of the pssO promoter region | [45] |
| pN76 | pMP220 carrying 0.75-kb BglII-PstI fragment of the pssN promoter region | [45] |
| pT80 | pMP220 carrying 0.8-kb BglII-PstI fragment of the pssT promoter region | [45] |
| pP85 | pMP220 carrying 0.85-kb EcoRI-XbaI fragment of the pssP promoter region | [45] |
| pI90 | pMP220 carrying 0.9-kb EcoRI-SphI fragment of the pssI promoter region | [45] |
| pPA2 | pMP220 carrying 0.9-kb EcoRI-XbaI fragment of the pssA promoter region | [32] |
| pEP1 | pMP220 carrying 0.65-bp EcoRI-PstI fragment of the rosR promoter region | [101] |
| pDGRP | pFUS1P carrying mcpD-gusA fusion | [103] |
| pCGR | pFUS1P carrying mcpC-gusA fusion | [103] |
| Primers | Sequence (5′→3′) | |
| pssAG1f | CGCACATGCGAAAGATTTGCTGCG | [104] |
| pssA2r | CCAGATCGAGGAATTCCCGACGTA | [104] |
| pssY5f | GTCGTCGATGACGATGCGGCTGTT | [104] |
| pssY5r | GAAACTATGTGCTTCCCATGTCATCG | [104] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lipa, P.; Vinardell, J.-M.; Janczarek, M. Transcriptomic Studies Reveal that the Rhizobium leguminosarum Serine/Threonine Protein Phosphatase PssZ has a Role in the Synthesis of Cell-Surface Components, Nutrient Utilization, and Other Cellular Processes. Int. J. Mol. Sci. 2019, 20, 2905. https://doi.org/10.3390/ijms20122905
Lipa P, Vinardell J-M, Janczarek M. Transcriptomic Studies Reveal that the Rhizobium leguminosarum Serine/Threonine Protein Phosphatase PssZ has a Role in the Synthesis of Cell-Surface Components, Nutrient Utilization, and Other Cellular Processes. International Journal of Molecular Sciences. 2019; 20(12):2905. https://doi.org/10.3390/ijms20122905
Chicago/Turabian StyleLipa, Paulina, José-María Vinardell, and Monika Janczarek. 2019. "Transcriptomic Studies Reveal that the Rhizobium leguminosarum Serine/Threonine Protein Phosphatase PssZ has a Role in the Synthesis of Cell-Surface Components, Nutrient Utilization, and Other Cellular Processes" International Journal of Molecular Sciences 20, no. 12: 2905. https://doi.org/10.3390/ijms20122905
APA StyleLipa, P., Vinardell, J.-M., & Janczarek, M. (2019). Transcriptomic Studies Reveal that the Rhizobium leguminosarum Serine/Threonine Protein Phosphatase PssZ has a Role in the Synthesis of Cell-Surface Components, Nutrient Utilization, and Other Cellular Processes. International Journal of Molecular Sciences, 20(12), 2905. https://doi.org/10.3390/ijms20122905






