Effects of Intracerebroventricular and Intra-Arcuate Nucleus Injection of Ghrelin on Pain Behavioral Responses and Met-Enkephalin and β-Endorphin Concentrations in the Periaqueductal Gray Area in Rats
Abstract
1. Introduction
2. Results
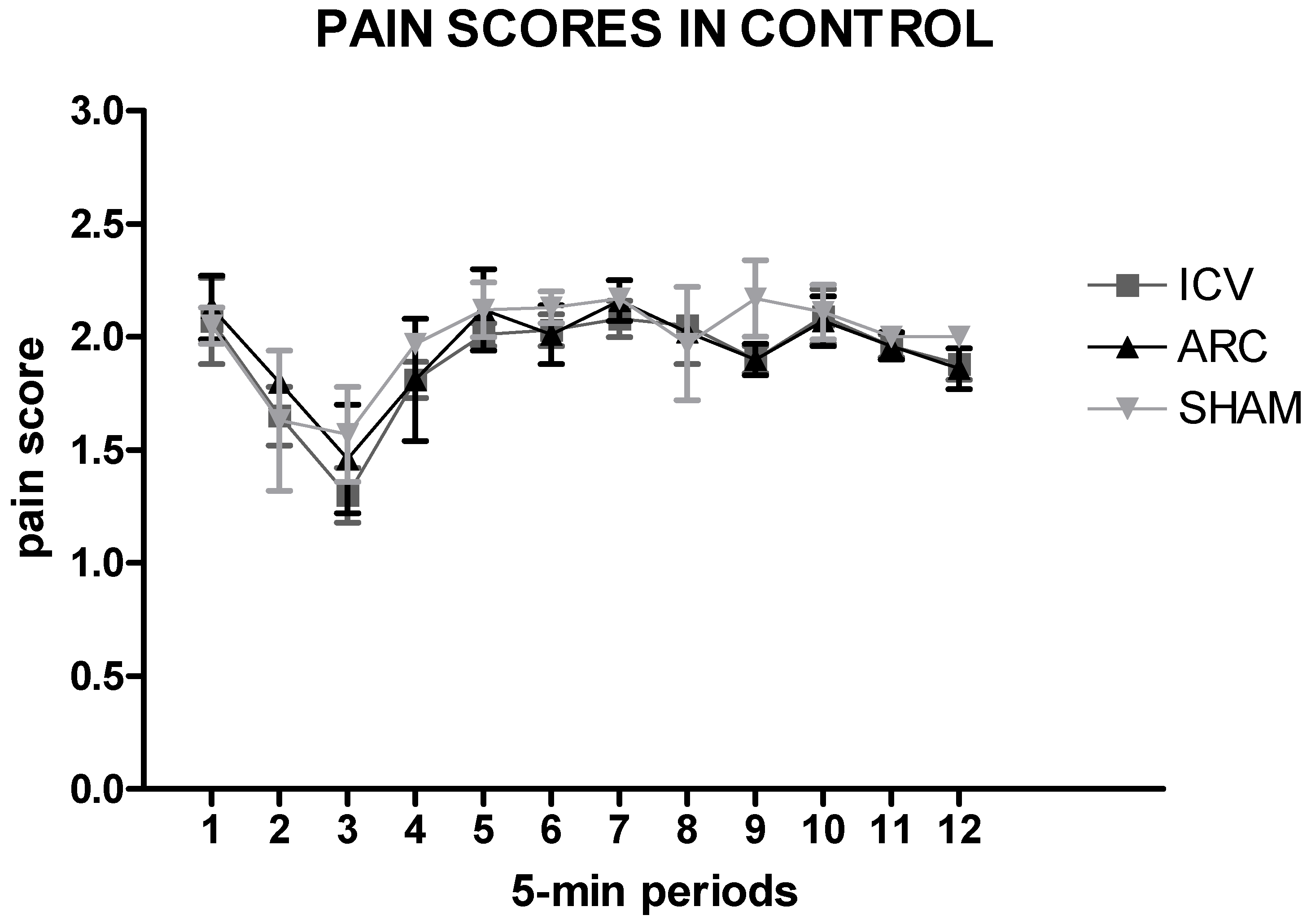
2.1. Saline Injection Versus Sham Treatment
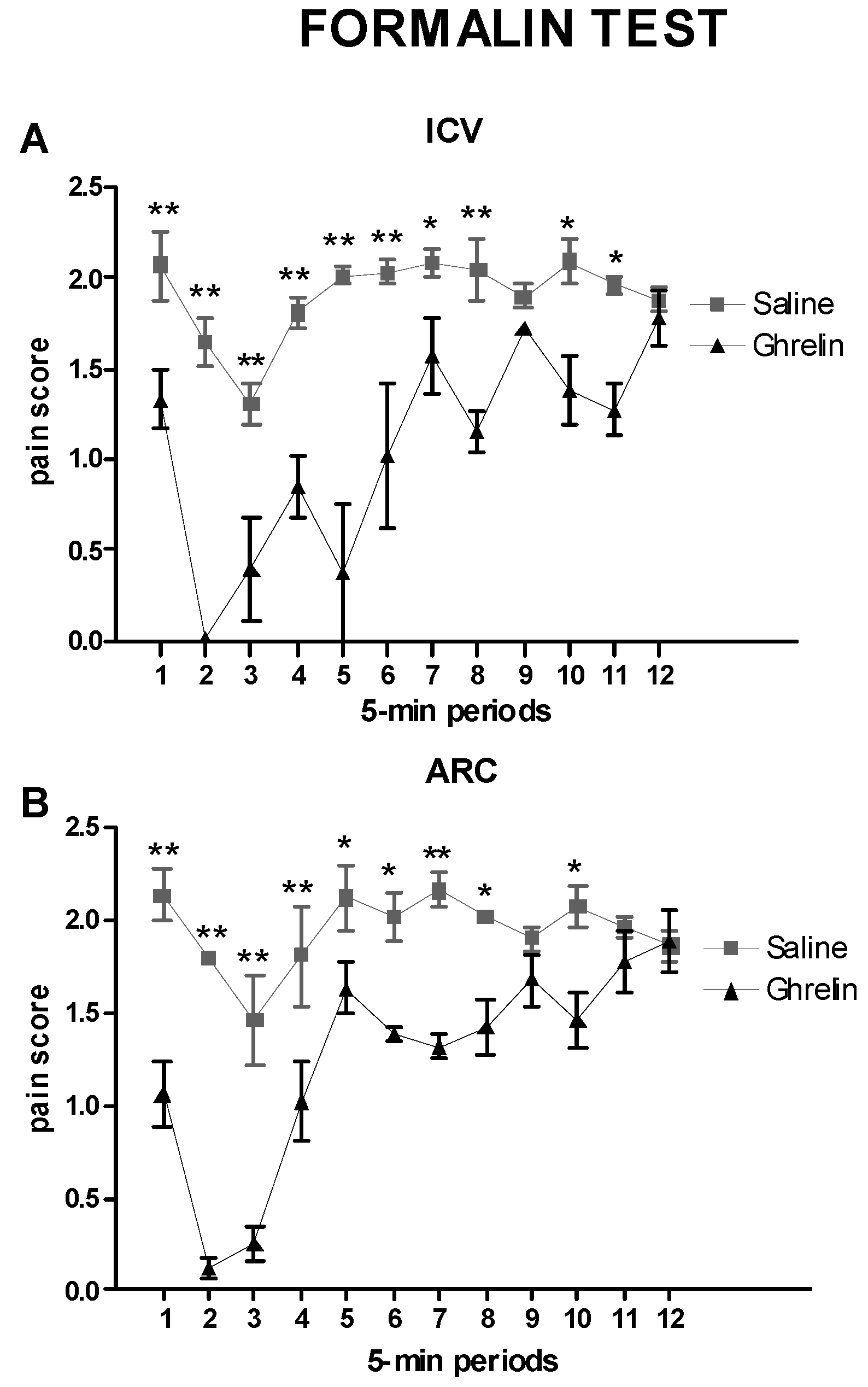
2.2. Evaluation of the Effects of Ghrelin on the Pain Score after Intracerebroventricular (ICV) or Intra-Arcuate Nucleus (ARC) Injection
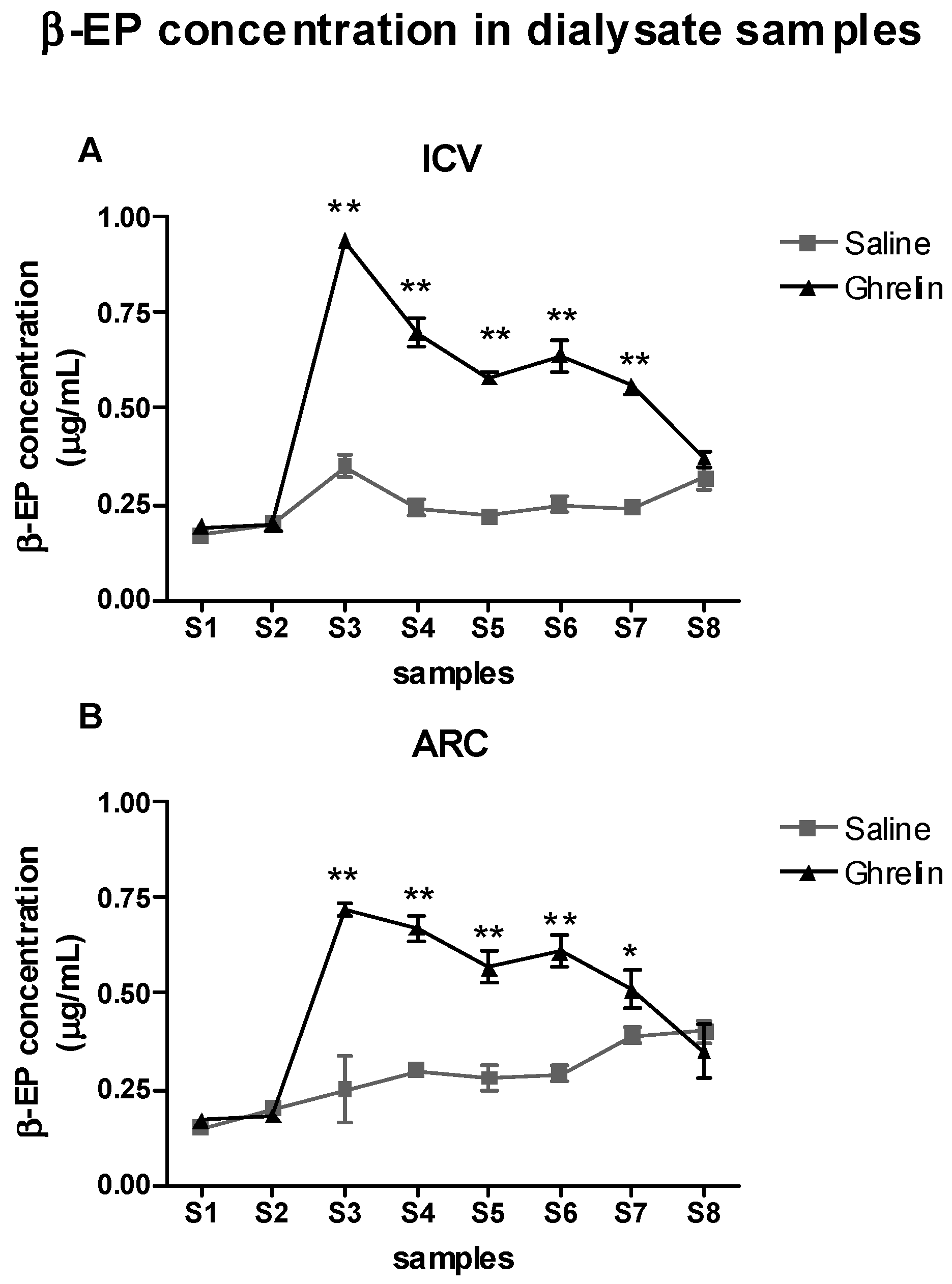
2.3. Evaluation of the Effects of Ghrelin on β-Endorphin (β-EP) Levels in the Periaqueductal Grey Area (PAG)
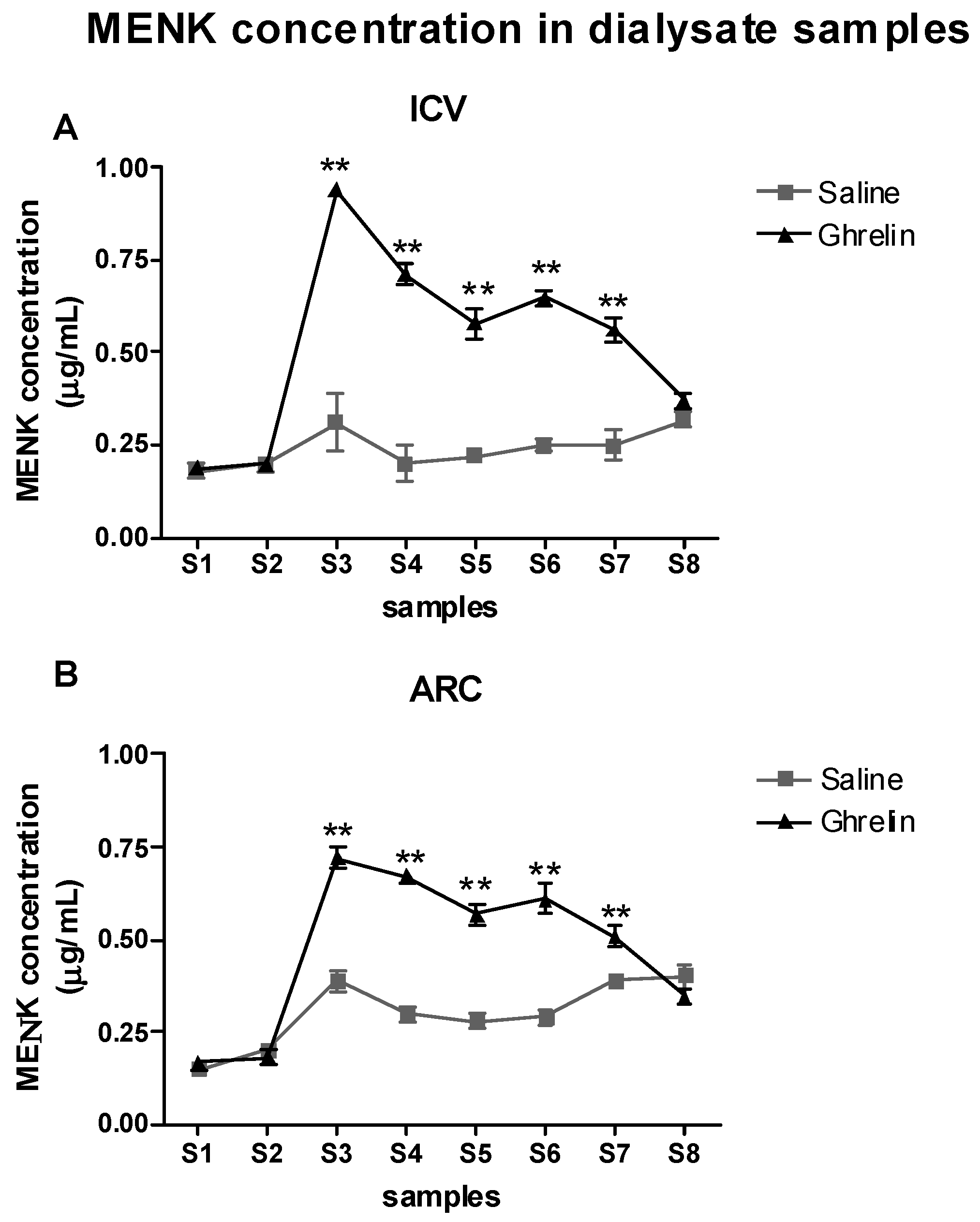
2.4. Evaluation of the Effects of Ghrelin on Met-Enkephalin (MENK) Levels in the PAG
3. Discussion
4. Materials and Methods
4.1. Ethics and Animals
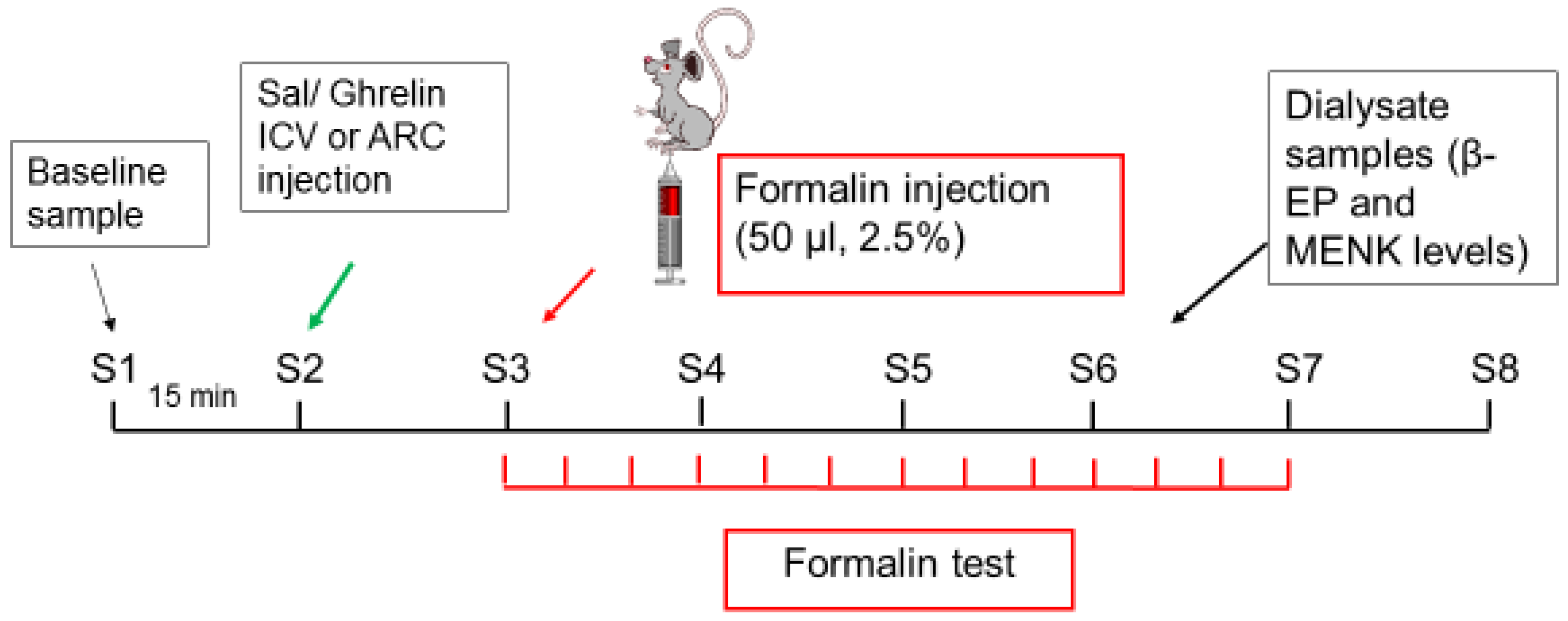
4.2. Study Design
- (1)
- Saline-ARC: receiving normal saline (1 μL) in the arcuate nucleus;
- (2)
- Saline-ICV: receiving normal saline (5 μL) ICV;
- (3)
- SHAM: receiving no injection;
- (4)
- Ghrelin-ICV: receiving 1 nmol ghrelin (5 μL) ICV;
- (5)
- Ghrelin-ARC: receiving 0.2 nmol ghrelin in the arcuate nucleus (1 μL).
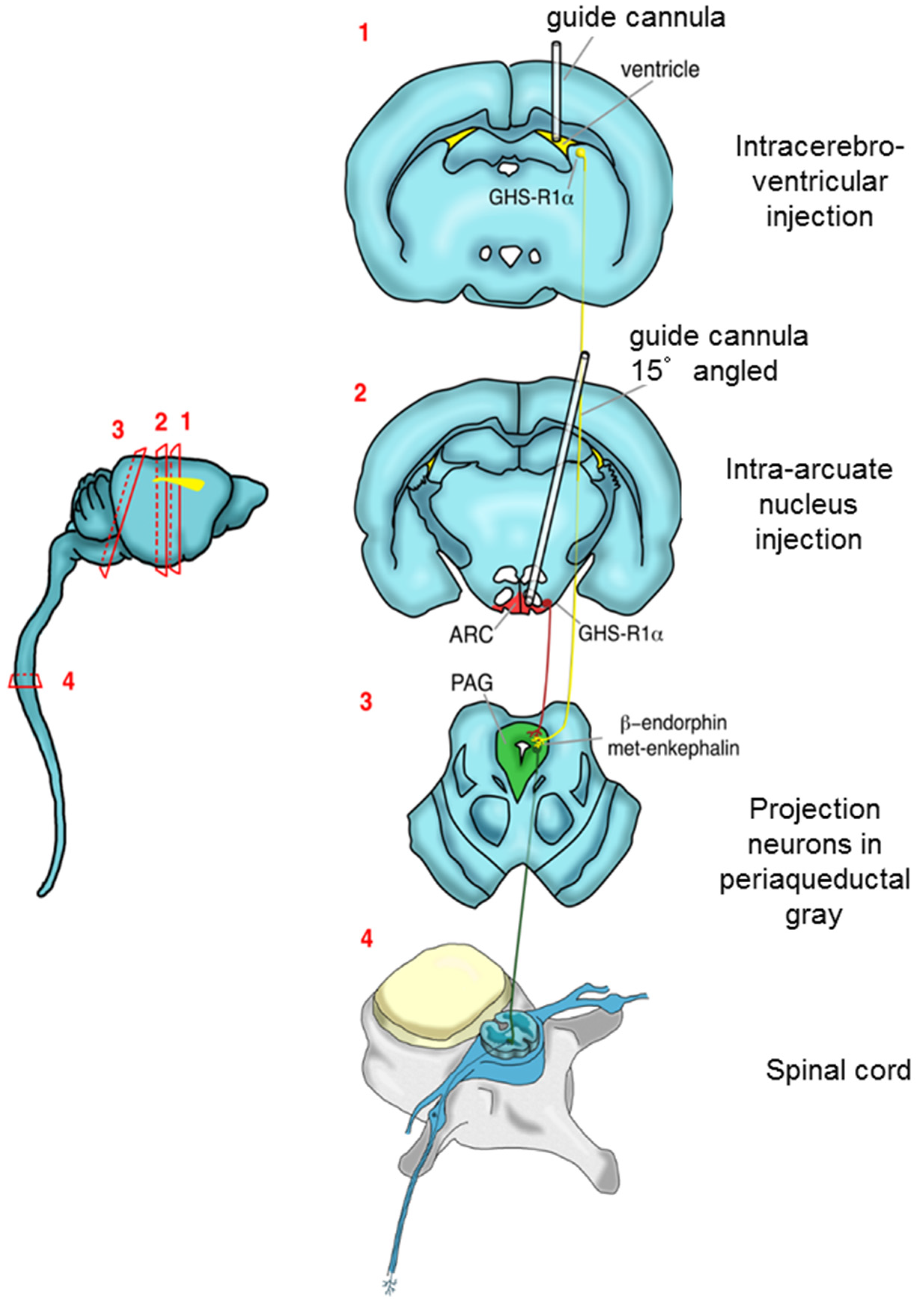
4.3. Implantation of the Probe and Guide Cannula and Injection of Drugs
4.4. Microdialysis and Sample Collection
4.5. Formalin Test
4.6. High-Performance Liquid Chromatography (HPLC) Analysis
4.7. Histological Verification
4.8. Data Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ICV | Intracerebroventricular |
| ARC | Arcuate nucleus |
| β-EP | β-endorphin |
| MENK | Met-enkephalin |
| PAG | Periaqueductal gray area |
| POMC | Pro-opiomelanocortin |
| NO | Nitric oxide |
| ACSF | Artificial cerebrospinal fluid |
| HPLC | High-performance liquid chromatograph |
| ANOVA | Analysis of variance |
References
- Ossipov, M.H.; Dussor, G.O.; Porreca, F. Central modulation of pain. J. Clin. Investig. 2010, 120, 377937–377987. [Google Scholar] [CrossRef]
- Coderre, T.J.; Fundytus, M.E.; McKenna, J.E.; Dalal, S.; Melzack, R. The formalin test: A validation of the weighted-scores method of behavioural pain rating. Pain 1993, 54, 43–50. [Google Scholar] [PubMed]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef]
- Muccioli, G.; Baragli, A.; Granata, R.; Papotti, M.; Ghigo, E. Heterogeneity of ghrelin/growth hormone secretagogue receptors. Toward the understanding of the molecular identity of novel ghrelin/GHS receptors. Neuroendocrinology 2007, 86, 147–164. [Google Scholar] [CrossRef]
- Kojima, M.; Kangawa, K. Ghrelin: Structure and function. Physiol. Rev. 2005, 84, 495–522. [Google Scholar] [CrossRef] [PubMed]
- Zigman, J.M.; Jones, J.E.; Saper, C.B.; Elmquist, J.K. Expression of ghrelin receptor mRNA in the rat and mouse brain. J. Comp. Neurol. 2006, 548, 528–548. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.M.; Yu, H.; Palyha, O.C.; McKee, K.K.; Feighner, S.D.; Sirinathsinghji, D.J.; Smith, R.G.; Van der Ploeg, L.H.; Howard, A.D. Distribution of mRNA encoding the growth hormone secretagogue receptor in brain and peripheral tissues. Brain Res. Mol. Brain Res. 1997, 48, 23–29. [Google Scholar] [CrossRef]
- Ferrini, F.; Salio, C.; Lossi, L.; Merighi, A. Ghrelin in central neurons. Curr. Neuropharmacol. 2009, 7, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Riediger, T.; Traebert, M.; Schmid, H.A.; Scheel, C.; Lutz, T.A.; Scharrer, E. Site-specific effects of ghrelin on the neuronal activity in the hypothalamic arcuate nucleus. Neurosci. Lett. 2003, 341, 151–155. [Google Scholar] [CrossRef]
- Guneli, E.; Gumastekin, M.; Ates, M. Possible involvement of ghrelin on pain threshold in obesity. Med. Hypotheses 2010, 74, 452–454. [Google Scholar] [CrossRef]
- Vergnano, A.M.; Ferrini, F.; Salio, C.; Lossi, L.; Barrata, M.; Merighi, A. The gastrointestinal hormone ghrelin modulates inhibitory neurotransmission in deep laminae of mouse spinal cord dorsal horn. Endocrinology 2008, 149, 2306–2312. [Google Scholar] [CrossRef]
- Bloom, F.; Battenberg, E.; Rossier, J.; Ling, N.; Guillemin, R. Neurons containing beta-endorphin in rat brain exist separately from those containing encephalin: Immunocytochemical studies. Proc. Natl. Acad. Sci. USA 1978, 75, 1591–1595. [Google Scholar] [CrossRef]
- Cowley, M.A.; Smith, R.G.; Diano, S.; Tschöp, M.; Pronchuk, N.; Grove, K.L.; Strasburger, C.J.; Bidlingmaier, M.; Esterman, M.; Heiman, M.L.; et al. The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron 2003, 37, 649–661. [Google Scholar] [CrossRef]
- Sun, Y.G.; Lundeberg, T.; Yu, L.C. Involvement of endogenous beta-endorphin in antinociception in the arcuate nucleus of hypothalamus in rats with inflammation. Pain 2003, 104, 55–63. [Google Scholar] [CrossRef]
- Joseph, S.A.; Michael, G.J. Efferent ACTH-IR opiocortin projection from nucleus tractus solitaries: A hypothalamic deafferentation study. Peptides 1988, 9, 193–201. [Google Scholar] [CrossRef]
- Sim, L.J.; Joseph, S.A. Arcuate nucleus projections to brainstem regions which modulate nociception. J. Chem. Neuroanat. 1991, 4, 97–109. [Google Scholar] [CrossRef]
- Romagnano, M.A.; Joseph, S.A. Immunocytochemical localization of ACTH in the brainstem of the rat. Brain Res. 1983, 276, 1–16. [Google Scholar] [CrossRef]
- Sibilia, V.; Lattuada, N.; Rapetti, O.; Francesca, P.; Vincenza, D.; Bulgarelli, I.; Locatelli, V.; Guidobono, F.; Netti, C. Ghrelin inhibits inflammatory pain in rats. Involvement of the opioid system. Neuropharmacology 2006, 51, 497–505. [Google Scholar] [CrossRef]
- Kyoraku, I.; Shimi, K.; Kangawa, K.; Nakazato, M. Ghrelin reverses experimental diabetic neuropathy in mice. Biochem. Biophys. Res. Commun. 2009, 389, 405–408. [Google Scholar] [CrossRef]
- Muccioli, G.; Papotti, M.; Locatelli, V.; Ghigo, E.; Deghenghi, R. Binding of 125I-labeled ghrelin to membranes from human hypothalamus and pituitary gland. J. Endocrinol. Investig. 2001, 24, RC7–RC9. [Google Scholar] [CrossRef]
- Garcia, J.M.; Cata, J.P.; Dougherty, P.M.; Smith, R.G. Ghrelin prevents cisplatin-induced mechanical hyperalgesia and cachexia. Endocrinology 2008, 149, 455–460. [Google Scholar] [CrossRef]
- Capone, F.; Aloisi, A.M. Refinement of pain evaluation techniques. The formalin test. Ann. Ist. Super. Sanita 2004, 40, 223–229. [Google Scholar] [PubMed]
- Azizzadeh, F.; Mahmoodi, J.; Sadigh-Eteghad, S.; Farajdokht, F.; Mohaddes, G. Ghrelin exerts analgesic effects through modulation of IL-10 and TGF-β levels in a rat model of inflammatory pain. Iran. Biomed. J. 2017, 21, 114–119. [Google Scholar] [CrossRef]
- Guneli, E.; Onal, A.; Ates, M.; Bagriyanik, H.A.; Resmi, H.; Orhan, C.E.; Kolatan, H.E.; Gumustekin, M. Effects of repeated administered ghrelin on chronic constriction injury of the sciatic nerve in rats. Neurosci. Lett. 2010, 479, 226–230. [Google Scholar] [CrossRef] [PubMed]
- Gaskin, F.S.; Farr, S.A.; Banks, W.A.; Kumar, V.B.; Morley, J.E. Ghrelin-induced feeding is dependent on nitric oxide. Peptides 2003, 24, 913–918. [Google Scholar] [CrossRef]
- Heinzen, E.L.; Booth, R.G.; Pollack, G.M. Neuronal nitric oxide modulates morphine antinociceptive tolerance by enhancing constitutive activity of the mu-opioid receptor. Biochem. Pharmacol. 2005, 69, 679–688. [Google Scholar] [CrossRef]
- Angevine, J.B.; Cotman, C.W. Principles of Neuroanatomy; Oxford University Press: New York, NY, USA, 1981; pp. 253–293. [Google Scholar]
- Reynolds, D.V. Surgery in the rat during electrical analgesia induced by focal brain stimulation. Science 1969, 164, 444–445. [Google Scholar] [CrossRef] [PubMed]
- Tsou, K.; Jang, C.S. Studies on the site of analgesic action of morphine by intracerebral micro-injection. Sci. Sin. 1964, 13, 1099–1109. [Google Scholar] [CrossRef]
- Hosobuchi, Y.; Adams, J.E.; Linchitz, R. Pain relief by electrical stimulation of the central gray matter in humans and its reversal by naloxone. Science 1977, 197, 183–186. [Google Scholar] [CrossRef]
- Richardson, D.E.; Akil, H. Long term results of periventricular gray self-stimulation. Neurosurgery 1977, 1, 199–202. [Google Scholar] [CrossRef]
- Fields, H.L.; Basbaum, A.I. Endogenous pain control mechanisms. In Textbook of Pain; Wall, P.D., Melzack, R., Eds.; Churchill Livingston: New York, NY, USA, 1989; pp. 206–217. [Google Scholar]
- Yu, L.C.; Han, S. Involvement of arcuate nucleus of hypothalamus in the descending pathway from nucleus accumbens to periaqueductal grey subserving an antinociceptive effect. J. Neurosci. 1989, 48, 71–78. [Google Scholar] [CrossRef]
- Atweh, S.F.; Kuhar, M.J. Autoradiographic localization of opiate receptors in rat brain. 1. Spinal cord and lower medulla. Brain Res. 1977, 124, 53–67. [Google Scholar] [CrossRef]
- Kutlu, S.; Ozcan, M.; Canpolat, S.; Sandal, S.; Aydin, M.; Kelestimur, H. Effect of ghrelin on pain threshold in mice. Firat Tip Dergisi 2005, 10, 89–91. [Google Scholar]
- Wei, J.; Zhi, X.; Wang, X.L.; Zeng, P.; Zou, T.; Yang, B.; Wang, J.L. In vivo characterization of the effects of ghrelin on the modulation of acute pain at the supraspinal levels in mice. Peptides 2013, 43, 76–82. [Google Scholar] [CrossRef]
- Zeng, P.; Chen, J.X.; Yang, B.; Zhi, X.; Guo, F.X.; Sun, M.L.; Wang, J.L.; Wei, J. Attenuation of systemic morphine-induced analgesia by central administration of ghrelin and related peptides in mice. Peptides 2013, 50, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Zeng, P.; Li, S.; Zheng, Y.H.; Liu, F.Y.; Wang, J.L.; Zhang, D.L.; Wei, J. Ghrelin receptor agonist, GHRP-2, produces antinociceptive effects at the supraspinal level via the opioid receptor in mice. Peptides 2014, 55, 103–109. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates, 5th ed.; Academic Press: San Diego, CA, USA, 1977; p. 80. [Google Scholar]
- Dubuisson, D.; Dennis, S.G. The formalin test: A quantitative study of the analgesic effects of morphine, meperidine, and brain stem stimulation in rats and cats. Pain 1977, 4, 161–174. [Google Scholar] [CrossRef]
- Sauernheimer, C.; Williams, K.M.; Brune, K.; Geisslinger, G. Application of microdialysis to the pharmacokinetics of analgesics: Problems with reduction of dialysis efficiency in vivo. J. Pharmacol. Toxicol. Methods 1994, 32, 149–154. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pirzadeh, S.; Sajedianfard, J.; Aloisi, A.M.; Ashrafi, M. Effects of Intracerebroventricular and Intra-Arcuate Nucleus Injection of Ghrelin on Pain Behavioral Responses and Met-Enkephalin and β-Endorphin Concentrations in the Periaqueductal Gray Area in Rats. Int. J. Mol. Sci. 2019, 20, 2475. https://doi.org/10.3390/ijms20102475
Pirzadeh S, Sajedianfard J, Aloisi AM, Ashrafi M. Effects of Intracerebroventricular and Intra-Arcuate Nucleus Injection of Ghrelin on Pain Behavioral Responses and Met-Enkephalin and β-Endorphin Concentrations in the Periaqueductal Gray Area in Rats. International Journal of Molecular Sciences. 2019; 20(10):2475. https://doi.org/10.3390/ijms20102475
Chicago/Turabian StylePirzadeh, Samaneh, Javad Sajedianfard, Anna Maria Aloisi, and Mahboobeh Ashrafi. 2019. "Effects of Intracerebroventricular and Intra-Arcuate Nucleus Injection of Ghrelin on Pain Behavioral Responses and Met-Enkephalin and β-Endorphin Concentrations in the Periaqueductal Gray Area in Rats" International Journal of Molecular Sciences 20, no. 10: 2475. https://doi.org/10.3390/ijms20102475
APA StylePirzadeh, S., Sajedianfard, J., Aloisi, A. M., & Ashrafi, M. (2019). Effects of Intracerebroventricular and Intra-Arcuate Nucleus Injection of Ghrelin on Pain Behavioral Responses and Met-Enkephalin and β-Endorphin Concentrations in the Periaqueductal Gray Area in Rats. International Journal of Molecular Sciences, 20(10), 2475. https://doi.org/10.3390/ijms20102475




