Vitamin D and Endometrium: A Systematic Review of a Neglected Area of Research
Abstract
1. Introduction: Vitamin D, Metabolism, and Reproduction
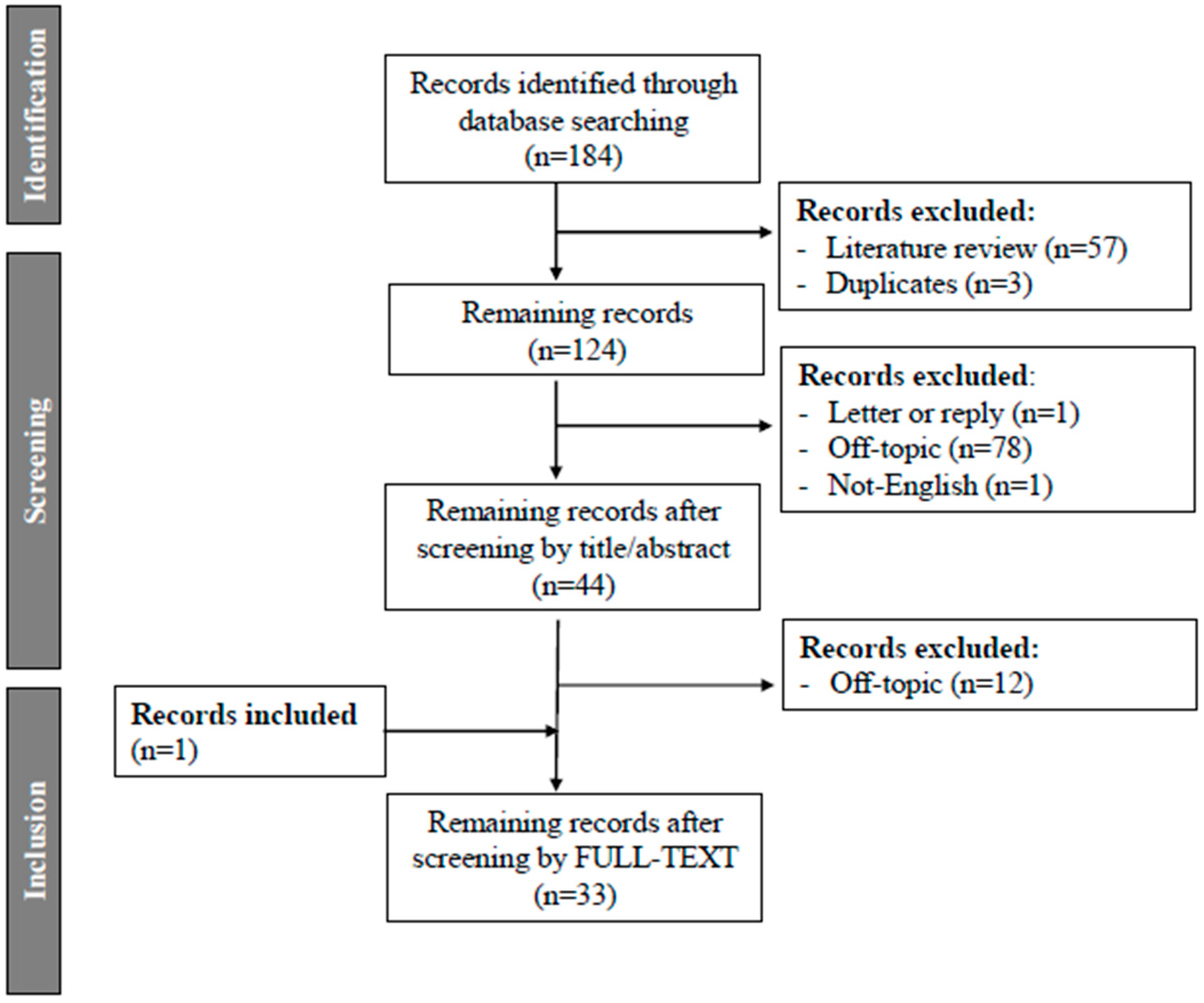
2. Methods
3. Vitamin D and Normal Endometrium
4. Vitamin D and Endometriosis
5. Vitamin D and Endometrial Cancer
5.1. VD/VDR Pathway and VD Metabolism in Endometrial Cancer
5.2. Vitamin D Action in Endometrial Cancer
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bikle, D.D. Vitamin D metabolism, mechanism of action, and clinical applications. Chem. Biol. 2014, 21, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Prosser, D.E.; Kaufmann, M. Cytochrome P450-mediated metabolism of vitamin D. J. Lipid Res. 2014, 55, 13–31. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Hewison, M. Extrarenal expression of the 25-hydroxyvitamin D-1-hydroxylase. Arch. Biochem. Biophys. 2012, 523, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Kliewer, S.A.; Umesono, K.; Mangelsdorf, D.J.; Evans, R.M. Retinoid X receptor interacts with nuclear receptors in retinoic acid, thyroid hormone and vitamin D3 signalling. Nature 1992, 355, 446–449. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.M.; Binderup, L.; Hamberg, K.J.; Carlberg, C. Vitamin D and cancer: Effects of 1,25(OH)2D3 and its analogs on growth control and tumorigenesis. Front. Biosci. 2001, 6, D820–D848. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; Mahon, B.D. D-hormone and the immune system. J. Rheumatol. Suppl. 2005, 76, 11–20. [Google Scholar] [PubMed]
- Hii, C.S.; Ferrante, A. The Non-Genomic Actions of Vitamin D. Nutrients 2016, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Grzechocinska, B.; Dabrowski, F.A.; Cyganek, A.; Wielgos, M. The role of vitamin D in impaired fertility treatment. Neuro Endocrinol. Lett. 2013, 34, 756–762. [Google Scholar] [PubMed]
- Dokoh, S.; Donaldson, C.A.; Marion, S.L.; Pike, J.W.; Haussler, M.R. The ovary: A target organ for 1,25-dihydroxyvitamin D3. Endocrinology 1983, 112, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Halloran, B.P.; DeLuca, H.F. Effect of vitamin D deficiency on fertility and reproductive capacity in the female rat. J. Nutr. 1980, 110, 1573–1580. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, T.; Handa, Y.; Uematsu, Y.; Takeda, S.; Sekine, K.; Yoshihara, Y.; Kawakami, T.; Arioka, K.; Sato, H.; Uchiyama, Y.; et al. Mice lacking the vitamin D receptor exhibit impaired bone formation, uterine hypoplasia and growth retardation after weaning. Nat. Genet. 1997, 16, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Panda, D.K.; Miao, D.; Tremblay, M.L.; Sirois, J.; Farookhi, R.; Hendy, G.N.; Goltzman, D. Targeted ablation of the 25-hydroxyvitamin D 1α-hydroxylase enzyme: Evidence for skeletal, reproductive, and immune dysfunction. Proc. Natl. Acad. Sci. USA 2001, 98, 7498–7503. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Daftary, G.S.; Lalwani, S.I.; Taylor, H.S. Direct regulation of HOXA10 by 1,25(OH)2D3 in human myelomonocytic cells and human endometrial stromal cells. Mol. Endocrinol. 2005, 19, 2222–2233. [Google Scholar] [CrossRef] [PubMed]
- Zondervan, K.T.; Cardon, L.R.; Kennedy, S.H. What makes a good case–control study? Hum. Reprod. 2002, 17, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Zondervan, K.T.; Becker, C.M.; Koga, K.; Missmer, S.A.; Taylor, R.N.; Viganò, P. Endometriosis. Nat. Rev. Dis. Primers. 2018, 19, 9. [Google Scholar] [CrossRef] [PubMed]
- Sasson, I.E.; Taylor, H.S. Stem Cells and the Pathogenesis of Endometriosis. Ann. N. Y. Acad. Sci. 2008, 1127, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Laganà, A.S.; Vitale, S.G.; Salmeri, F.M.; Triolo, O.; ban Frangež, H.; Vrtačnik-Bokal, E.; Stojanovska, L.; Apostolopoulos, V.; Granese, R.; Sofo, V. Unus pro omnibus, omnes pro uno: A novel, evidence-based, unifying theory for the pathogenesis of endometriosis. Med. Hypotheses 2017, 103, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Gordts, S.; Koninckx, P.; Brosens, I. Pathogenesis of deep endometriosis. Fertil. Steril. 2017, 6, 872–885. [Google Scholar] [CrossRef] [PubMed]
- Vinatier, D.; Cosson, M.; Dufour, P. Is endometriosis an endometrial disease? Eur. J. Obstet. Gynecol. Reprod. Biol. 2000, 91, 113–125. [Google Scholar] [CrossRef]
- Sharpe-Timms, K.L. Endometrial anomalies in women with endometriosis. Ann. N. Y. Acad. Sci. 2001, 943, 131–147. [Google Scholar] [CrossRef] [PubMed]
- Colonese, F.; Laganà, A.S.; Colonese, E.; Sofo, V.; Salmeri, F.M.; Granese, R.; Triolo, O. The pleiotropic effects of vitamin D in gynaecological and obstetric diseases: An overview on a hot topic. BioMed Res. Int. 2015, 2015, 986281. [Google Scholar] [CrossRef] [PubMed]
- Morice, P.; Leary, A.; Creutzberg, C.; Abu-Rustum, N.; Darai, E. Endometrial cancer. Lancet 2016, 387, 1094–1108. [Google Scholar] [CrossRef]
- Deuster, E.; Jeschke, U.; Ye, Y.; Mahner, S.; Czogalla, B. Vitamin D and VDR in Gynecological Cancers—A Systematic Review. Int. J. Mol. Sci. 2017, 18, 2328. [Google Scholar] [CrossRef] [PubMed]
- Salazar-Martinez, E.; Lazcano-Ponce, E.; Sanchez-Zamorano, L.M.; Gonzalez-Lira, G.; Escudero-DE Los Rios, P.; Hernandez-Avila, M. Dietary factors and endometrial cancer risk. Results of a case-control study in Mexico. Int. J. Gynecol. Cancer 2005, 15, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Bandera, E.V.; Gifkins, D.M.; Moore, D.F.; McCullough, M.L.; Kushi, L.H. Antioxidant vitamins and the risk of endometrial cancer: A dose-response meta-analysis. Cancer Causes Control 2009, 20, 699–711. [Google Scholar] [CrossRef] [PubMed]
- Zeleniuch-Jacquotte, A.; Gallicchio, L.; Hartmuller, V.; Helzlsouer, K.J.; McCullough, M.L.; Setiawan, V.W.; Shu, X.O.; Weinstein, S.J.; Weiss, J.M.; Arslan, A.A.; et al. Circulating 25-hydroxyvitamin D and risk of endometrial cancer: Cohort Consortium Vitamin D Pooling Project of Rarer Cancers. Am. J. Epidemiol. 2010, 172, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Vienonen, A.; Miettinen, S.; Bläuer, M.; Martikainen, P.M.; Tomás, E.; Heinonen, P.K.; Ylikomi, T. Expression of nuclear receptors and cofactors in human endometrium and myometrium. J. Soc. Gynecol. Investig. 2004, 11, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Viganò, P.; Lattuada, D.; Mangioni, S.; Ermellino, L.; Vignali, M.; Caporizzo, E.; Panina-Bordignon, P.; Besozzi, M.; di Blasio, A.M. Cycling and early pregnant endometrium as a site of regulated expression of the vitamin D system. J. Mol. Endocrinol. 2006, 36, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Zelenko, Z.; Aghajanova, L.; Irwin, J.C.; Giudice, L.C. Nuclear receptor, coregulator signaling, and chromatin remodeling pathways suggest involvement of the epigenome in the steroid hormone response of endometrium and abnormalities in endometriosis. Reprod. Sci. 2012, 19, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Bergadà, L.; Pallares, J.; Maria Vittoria, A.; Cardus, A.; Santacana, M.; Valls, J.; Cao, G.; Fernàndez, E.; Dolcet, X.; Dusso, A.S.; et al. Role of local bioactivation of vitamin D by CYP27A1 and CYP2R1 in the control of cell growth in normal endometrium and endometrial carcinoma. Lab. Investig. 2014, 94, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Cordes, T.; Diesing, D.; Diedrich, K.; Friedrich, M. Expression of 25 hydroxyvitamin D3-1α-hydroxylase in human endometrial tissue. J. Steroid Biochem. Mol. Biol. 2007, 103, 771–775. [Google Scholar] [CrossRef] [PubMed]
- Gellersen, B.; Brosens, J.J. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr. Rev. 2014, 35, 851–905. [Google Scholar] [CrossRef] [PubMed]
- Szczepańska, M.; Wirstlein, P.; Luczak, M.; Jagodziński, P.P.; Skrzypczak, J. Expression of HOXA-10 and HOXA-11 in the endometria of women with idiopathic infertility. Folia Histochem. Cytobiol. 2011, 49, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.A.; Burghardt, R.C.; Bazer, F.W.; Spencer, T.E. Osteopontin: Roles in implantation and placentation. Biol. Reprod. 2003, 69, 1458–1471. [Google Scholar] [CrossRef] [PubMed]
- Asadi, M.; Matin, N.; Frootan, M.; Mohamadpour, J.; Qorbani, M.; Tanha, F.D. Vitamin D improves endometrial thickness in PCOS women who need intrauterine insemination: A randomized double-blind placebo-controlled trial. Arch. Gynecol. Obstet. 2014, 289, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Detti, L.; Uhlmann, R.A.; Fletcher, N.M.; Diamond, M.P.; Saed, G.M. Endometrial signaling pathways during ovarian stimulation for assisted reproduction technology. Fertil. Steril. 2013, 100, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Rudick, B.J.; Ingles, S.A.; Chung, K.; Stanczyk, F.Z.; Paulson, R.J.; Bendikson, K.A. Influence of vitamin D levels on in vitro fertilization outcomes in donor-recipient cycles. Fertil. Steril. 2014, 101, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Fabris, A.; Pacheco, A.; Cruz, M.; Puente, J.M.; Fatemi, H.; Garcia-Velasco, J.A. Impact of circulating levels of total and bioavailable serum vitamin D on pregnancy rate in egg donation recipients. Fertil. Steril. 2014, 102, 1608–1612. [Google Scholar] [CrossRef] [PubMed]
- Franasiak, J.M.; Molinaro, T.A.; Dubell, E.K.; Scott, K.L.; Ruiz, A.R.; Forman, E.J.; Werner, M.D.; Hong, K.H.; Scott, R.T. Vitamin D levels do not affect IVF outcomes following the transfer of euploid blastocysts. Am. J. Obstet. Gynecol. 2015, 212, 315. [Google Scholar] [CrossRef] [PubMed]
- Agic, A.; Xu, H.; Altgassen, C.; Noack, F.; Wolfler, M.M.; Diedrich, K.; Friedrich, M.; Taylor, R.N.; Hornung, D. Relative expression of 1,25-dihydroxyvitamin D3 receptor, vitamin D 1α-hydroxylase, vitamin D 24-hydroxylase, and vitamin D 25-hydroxylase in endometriosis and gynecologic cancers. Reprod. Sci. 2007, 14, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Valdivielso, J.M.; Fernandez, E. Vitamin D receptor polymorphisms and diseases. Clin. Chim. Acta 2006, 371, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Vilarino, F.L.; Bianco, B.; Lerner, T.G.; Teles, J.S.; Mafra, F.A.; Christofolini, D.M.; Barbosa, C.P. Analysis of vitamin D receptor gene polymorphisms in women with and without endometriosis. Hum. Immunol. 2011, 72, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Szczepańska, M.; Mostowska, A.; Wirstlein, P.; Skrzypczak, J.; Misztal, M.; Jagodziński, P.P. Polymorphic variants in vitamin D signaling pathway genes and the risk of endometriosis-associated infertility. Mol. Med. Rep. 2015, 12, 7109–7115. [Google Scholar] [CrossRef] [PubMed]
- Guyton, K.Z.; Kensler, T.W.; Posner, G.H. Vitamin D and vitamin D analogs as cancer chemopreventive agents. Nutr. Rev. 2003, 61, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, M.; Koga, K.; Izumi, G.; Sue, F.; Makabe, T.; Taguchi, A.; Nagai, M.; Urata, Y.; Takamura, M.; Harada, M.; et al. Effects of 1,25-Dihydroxy Vitamin D3 on Endometriosis. J. Clin. Endocrinol. Metab. 2016, 101, 2371–2379. [Google Scholar] [CrossRef] [PubMed]
- Delbandi, A.A.; Mahmoudi, M.; Shervin, A.; Zarnani, A.H. 1,25-Dihydroxy Vitamin D3 Modulates Endometriosis-Related Features of Human Endometriotic Stromal Cells. Am. J. Reprod. Immunol. 2016, 75, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Ingles, S.A.; Wu, L.; Liu, B.T.; Chen, Y.; Wang, C.Y.; Templeman, C.; Brueggmann, D. Differential gene expression by 1,25(OH)2D3 in an endometriosis stromal cell line. J. Steroid Biochem. Mol. Biol. 2017, 173, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Mariani, M.; Viganò, P.; Gentilini, D.; Camisa, B.; Caporizzo, E.; di Lucia, P.; Monno, A.; Candiani, M.; Somigliana, E.; Panina-Bordignon, P. The selective vitamin D receptor agonist, elocalcitol, reduces endometriosis development in a mouse model by inhibiting peritoneal inflammation. Hum. Reprod. 2012, 27, 2010–2019. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.A.; Taha, M.O.; Disi, A.M.; Shomaf, M. Regression of endometrial implants treated with vitamin D3 in a rat model of endometriosis. Eur. J. Pharmacol. 2013, 715, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, B.; Guler, T.; Akbulut, M.; Oztekin, O.; Sariiz, G. 1-α,25-dihydroxyvitamin D3 regresses endometriotic implants in rats by inhibiting neovascularization and altering regulation of matrix metalloproteinase. Postgrad. Med. 2014, 126, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.M.; Viganò, P.; Somigliana, E.; Panina-Bordignon, P.; Vercellini, P.; Candiani, M. The distinguishing cellular and molecular features of the endometriotic ovarian cyst: From pathophysiology to the potential endometrioma-mediated damage to the ovary. Hum. Reprod. Update 2014, 20, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Yabushita, H.; Hirata, M.; Noguchi, M.; Nakanishi, M. Vitamin D receptor in endometrial carcinoma and the differentiation-inducing effect of 1,25-dihydroxyvitamin D3 on endometrial carcinoma cell lines. J. Obstet. Gynaecol. Res. 1996, 22, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Bokhari, A.A.; Lee, L.R.; Raboteau, D.; Turbov, J.; Rodriguez, I.V.; Pike, J.W.; Hamilton, C.A.; Maxwell, G.L.; Rodriguez, G.C.; Syed, V. Progesterone potentiates the growth inhibitory effects of calcitriol in endometrial cancer via suppression of CYP24A1. Oncotarget. 2016, 7, 77576–77590. [Google Scholar] [CrossRef] [PubMed]
- Cross, H.S.; Bises, G.; Lechner, D.; Manhardt, T.; Kállay, E. The Vitamin D endocrine system of the gut—Its possible role in colorectal cancer prevention. J. Steroid Biochem. Mol. Biol. 2005, 97, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.G.; Nakane, M.; Ruan, X.; Kroeger, P.E.; Wu-Wong, J.R. Expression of VDR and CYP24A1 mRNA in human tumors. Cancer Chemother. Pharmacol. 2006, 57, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Christopherson, W.A.; Porter, J.C.; MacDonald, P.C.; Casey, M.L. Responsiveness of human carcinoma cells of gynecologic origin to 1,25-dihydroxycholecalciferol. Am. J. Obstet. Gynecol. 1986, 155, 1293–1296. [Google Scholar] [CrossRef]
- Saunders, D.E.; Christensen, C.; Wappler, N.L.; Cho, Y.L.; Lawrence, W.D.; Malone, J.M.; Malviya, V.K.; Deppe, G. Additive inhibition of RL95-2 endometrial carcinoma cell growth by carboplatin and 1,25 dihydroxyvitamin D3. Gynecol. Oncol. 1993, 51, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.; Ivanova, V.S.; Kavandi, L.; Rodriguez, G.C.; Maxwell, G.L.; Syed, V. Progesterone and 1,25-dihydroxyvitamin D3 inhibit endometrial cancer cell growth by upregulating semaphorin 3B and semaphorin 3F. Mol. Cancer Res. 2011, 9, 1479–1492. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.R.; Teng, P.N.; Nguyen, H.; Hood, B.L.; Kavandi, L.; Wang, G.; Turbov, J.M.; Thaete, L.G.; Hamilton, C.A.; Maxwell, G.L.; et al. Progesterone enhances calcitriol antitumor activity by upregulating vitamin D receptor expression and promoting apoptosis in endometrial cancer cells. Cancer Prev. Res. 2013, 6, 731–743. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Park, J.; Lee, J.S.; Lee, H.H. Effects of 1α,25-dihydroxyvitamin D3 on programmed cell death of Ishikawa endometrial cancer cells through ezrin phosphorylation. J. Obstet. Gynaecol. 2017, 37, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Kasiappan, R.; Sun, Y.; Lungchukiet, P.; Quarni, W.; Zhang, X.; Bai, W. Vitamin D suppresses leptin stimulation of cancer growth through microRNA. Cancer Res. 2014, 74, 6194–6204. [Google Scholar] [CrossRef] [PubMed]
- Haselberger, M.; Springwald, A.; Konwisorz, A.; Lattrich, C.; Goerse, R.; Ortmann, O.; Treeck, O. Silencing of the icb-1 gene inhibits the induction of differentiation-associated genes by vitamin D3 and all-trans retinoic acid in gynecological cancer cells. Int. J. Mol. Med. 2011, 28, 121–127. [Google Scholar] [PubMed]
- Treeck, O.; Strunck, E.; Vollmer, G. A novel basement membrane-induced gene identified in the human endometrial adenocarcinoma cell line HEC1B. FEBS Lett. 1998, 425, 426–430. [Google Scholar] [CrossRef]
- Bansal, N.; Yendluri, V.; Wenham, R.M. The molecular biology of endometrial cancers and the implications for pathogenesis, classification, and targeted therapies. Cancer Control 2009, 16, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Zeng, N.; Salker, M.S.; Zhang, S.; Singh, Y.; Shi, B.; Stournaras, C.; Lang, F. 1α,25(OH)2D3 Induces Actin Depolymerization in Endometrial Carcinoma Cells by Targeting RAC1 and PAK1. Cell. Physiol. Biochem. 2016, 40, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.Y.; Ly, L.H.; Peehl, D.M.; Feldman, D. Induction of androgen receptor by 1α,25-dihydroxyvitamin D3 and 9-cis retinoic acid in LNCaP human prostate cancer cells. Endocrinology 1999, 140, 1205–1212. [Google Scholar] [CrossRef] [PubMed]
- Beumer, J.H.; Parise, R.A.; Kanterewicz, B.; Petkovich, M.; D’Argenio, D.Z.; Hershberger, P.A. A local effect of CYP24 inhibition on lung tumor xenograft exposure to 1,25-dihydroxyvitamin D3 is revealed using a novel LC-MS/MS assay. Steroids 2012, 77, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Modugno, F.; Ness, R.B.; Chen, C.; Weiss, N.S. Inflammation and endometrial cancer: A hypothesis. Cancer Epidemiol. Biomark. Prev. 2005, 14, 2840–2847. [Google Scholar] [CrossRef] [PubMed]
- Neill, A.S.; Nagle, C.M.; Protani, M.M.; Obermair, A.; Spurdle, A.B.; Webb, P.M.; Australian National Endometrial Cancer Study Group. Aspirin, nonsteroidal anti-inflammatory drugs, paracetamol and risk of endometrial cancer: A case-control study, systematic review and meta-analysis. Int. J. Cancer 2013, 132, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Verdoodt, F.; Friis, S.; Dehlendorff, C.; Albieri, V.; Kjaer, S.K. Non-steroidal anti-inflammatory drug use and risk of endometrial cancer: A systematic review and meta-analysis of observational studies. Gynecol. Oncol. 2016, 140, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Kavandi, L.; Collier, M.A.; Nguyen, H.; Syed, V. Progesterone and calcitriol attenuate inflammatory cytokines CXCL1 and CXCL2 in ovarian and endometrial cancer cells. J. Cell. Biochem. 2012, 113, 3143–3152. [Google Scholar] [CrossRef] [PubMed]
| Type of Samples | Target | Result | Technique | Reference |
|---|---|---|---|---|
| Endometrial tissue from control patients (premenopausal) | VDR | Presence | PCR-array | Vienonen et al., 2004 [27] |
| Presence | Vigano et al., 2006 [28] | |||
| Down-regulated in mid-secretory vs. early secretory | PCR-array | Zelenko et al., 2012 [29] | ||
| Down-regulated in proliferative vs. secretory phase | Tissue array | Bergada et al., 2014 [30] | ||
| 1α-hydroxylase | Presence | RT-PCR | Vigano et al., 2006 [28] | |
| Presence | Becker et al., 2007 [31] | |||
| Down-regulated in proliferative vs. secretory phase | Tissue array | Bergada et al., 2014 [30] | ||
| 25-hydroxylase | Down-regulated in proliferative vs. secretory phase | Tissue array | Bergada et al., 2014 [30] | |
| Eutopic endometrium from endometriosis patients | 24-hydroxylase | Up-regulated endometriosis vs. control tissue | RT-PCR | Agic et al., 2007 [40] |
| 25-hydroxylase | No differences between endometriosis vs. control tissue | |||
| 1α-hydroxylase | No differences between endometriosis vs. control tissue | |||
| VDR | No differences between endometriosis vs. control tissue | |||
| No differences between endometriosis vs. control tissue | RT-PCR | Zelenko et al., 2012 [29] | ||
| Endometrial tissue from endometrial cancer patients | VDR | Up-regulated endometrial cancer vs. control tissue | RT-PCR | Agic et al., 2007 [40] |
| Nuclear VDR | Down-regulated endometrial cancer vs. control tissue | Tissue array | Bergada et al., 2014 [30] | |
| 24-hydroxylase | Up-regulated endometrial cancer vs. control tissue Correlation with tumor progression. | Tissue array | Bokhari et al., 2016 [53] | |
| Down-regulated endometrial cancer vs. control tissue No differences in tumor progression. | Bergada et al., 2014 [30] | |||
| Up-regulated endometrial cancer vs. control tissue | RT-PCR | Agic et al., 2007 [40] | ||
| 1α-hydroxylase | No differences between endometrial cancer vs. control tissue | Immunostaining | Becker et al., 2007 [31] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cermisoni, G.C.; Alteri, A.; Corti, L.; Rabellotti, E.; Papaleo, E.; Viganò, P.; Sanchez, A.M. Vitamin D and Endometrium: A Systematic Review of a Neglected Area of Research. Int. J. Mol. Sci. 2018, 19, 2320. https://doi.org/10.3390/ijms19082320
Cermisoni GC, Alteri A, Corti L, Rabellotti E, Papaleo E, Viganò P, Sanchez AM. Vitamin D and Endometrium: A Systematic Review of a Neglected Area of Research. International Journal of Molecular Sciences. 2018; 19(8):2320. https://doi.org/10.3390/ijms19082320
Chicago/Turabian StyleCermisoni, Greta Chiara, Alessandra Alteri, Laura Corti, Elisa Rabellotti, Enrico Papaleo, Paola Viganò, and Ana Maria Sanchez. 2018. "Vitamin D and Endometrium: A Systematic Review of a Neglected Area of Research" International Journal of Molecular Sciences 19, no. 8: 2320. https://doi.org/10.3390/ijms19082320
APA StyleCermisoni, G. C., Alteri, A., Corti, L., Rabellotti, E., Papaleo, E., Viganò, P., & Sanchez, A. M. (2018). Vitamin D and Endometrium: A Systematic Review of a Neglected Area of Research. International Journal of Molecular Sciences, 19(8), 2320. https://doi.org/10.3390/ijms19082320





