Non-Coding RNAs in Endometrial Physiopathology
Abstract
1. Introduction
2. ncRNAs and Embryo Implantation
2.1. Embryo Implantation
2.2. miRNAs in Endometrial Receptivity
2.3. Extracellular miRNAs in Endometrial Fluid
3. ncRNAs and Endometrial Cancer
3.1. Endometrial Cancer
3.2. miRNAs in Endometrial Cancer
3.3. Circulating miRNA and Endometrial Cancer
3.4. lncRNAs in Endometrial Cancer
4. ncRNAs and Endometriosis
4.1. Endometriosis
4.2. miRNAs and Endometriosis
4.3. Circulating miRNAs as Biomarkers of Endometriosis
4.4. lncRNAs and Endometriosis
5. ncRNAs and Chronic Endometritis
5.1. Chronic Endometritis
5.2. miRNAs and Chronic Endometritis
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Nowak, R. Mining treasures from ‘junk DNA’. Science 1994, 263, 608–610. [Google Scholar] [CrossRef] [PubMed]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar] [CrossRef] [PubMed]
- International Human Genome Sequencing Consortium. Finishing the euchromatic sequence of the human genome. Nature 2004, 431, 931–945. [Google Scholar]
- Friedman, R.C.; Farh, K.K.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Wilczynska, A.; Bushell, M. The complexity of miRNA-mediated repression. Cell Death Differ. 2015, 22, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Ragusa, M.; Barbagallo, C.; Statello, L.; Condorelli, A.G.; Battaglia, R.; Tamburello, L.; Barbagallo, D.; Di Pietro, C.; Purrello, M. Non-coding landscapes of colorectal cancer. World J. Gastroenterol. 2015, 21, 11709–11739. [Google Scholar] [CrossRef] [PubMed]
- Rosa, A.; Ballarino, M. Long Noncoding RNA Regulation of Pluripotency. Stem Cells Int. 2016, 2016, 1797692. [Google Scholar] [CrossRef] [PubMed]
- Fatica, A.; Bozzoni, I. Long non-coding RNAs: New players in cell differentiation and development. Nat. Rev. Genet. 2014, 15, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, M.; Kitagawa, K.; Kotake, Y.; Niida, H.; Ohhata, T. Cell cycle regulation by long non-coding RNAs. Cell Mol. Life Sci. 2013, 70, 4785–4794. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Wu, H.; Pavlosky, A.; Zou, L.L.; Deng, X.; Zhang, Z.X.; Jevnikar, A.M. Regulatory non-coding RNA: New instruments in the orchestration of cell death. Cell Death Dis. 2016, 7, e2333. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, T.; Wang, X.; He, A. Circles reshaping the RNA world: From waste to treasure. Mol. Cancer 2017, 16, 58. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Jensen, T.I.; Clausen, B.H.; Bramsen, J.B.; Finsen, B.; Damgaard, C.K.; Kjems, J. Natural RNA circles function as efficient microRNA sponges. Nature 2013, 495, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Ragusa, M.; Barbagallo, C.; Brex, D.; Caponnetto, A.; Cirnigliaro, M.; Battaglia, R.; Barbagallo, D.; Di Pietro, C.; Purrello, M. Molecular Crosstalking among Noncoding RNAs: A New Network Layer of Genome Regulation in Cancer. Int. J. Genom. 2017, 2017, 4723193. [Google Scholar] [CrossRef] [PubMed]
- Koot, Y.E.; Macklon, N.S. Embryo implantation: Biology, evaluation and enhancement. Curr. Opin. Obstet. Gynecol. 2013, 25, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Von Grothusen, C.; Lalitkumar, S.; Boggavarapu, N.R.; Gemzell-Danielsson, K.; Lalitkumar, P.G. Recent advances in understanding endometrial receptivity: Molecular basis and clinical applications. Am. J. Reprod. Immunol. 2014, 72, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Galliano, D.; Pellicer, A. MicroRNA and implantation. Fertil. Steril. 2014, 101, 1531–1544. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Gimeno, P.; Horcajadas, J.A.; Martínez-Conejero, J.A. A genomic diagnostic tool for human endometrial receptivity based on the transcriptomic signature. Fertil. Steril. 2011, 95, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Gimeno, P.; Ruiz-Alonso, M.; Blesa, D.; Bosch, N.; Martínez-Conejero, J.A.; Alamá, P.; Garrido, N.; Pellicer, A.; Simón, C. The accuracy and reproducibility of the endometrial receptivity array is superior to histology as a diagnostic method for endometrial receptivity. Fertil. Steril. 2013, 99, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Garrido-Gòmez, T.; Ruiz-Alonso, M.; Blesa, D.; Diaz-Gimeno, P.; Vilella, F.; Simòn, C. Profiling the gene signature of endometrial receptivity: Clinical results. Fertil. Steril. 2013, 99, 1078–1085. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Alonso, M.; Blesa, D.; Díaz-Gimeno, P.; Gómez, E.; Fernández-Sánchez, M.; Carranza, F.; Carrera, J.; Vilella, F.; Pellicer, A.; Simón, C. The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertil. Steril. 2013, 100, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Inyawilert, W.; TY, F.; Lin, C.T.; Tang, P.C. Let-7-mediated suppression of mucin 1 expression in the mouse uterus during embryo implantation. J. Reprod. Dev. 2015, 61, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Altmäe, S.; Martinez-Conejero, J.A.; Esteban, F.J.; Ruiz-Alonso, M.; Stavreus-Evers, A.; Horcajadas, J.A.; Salumets, A. MicroRNAs miR-30b, miR-30d and miR-494 regulate human endometrial receptivity. Reprod. Sci. 2013, 20, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Inyawilert, W.; TY, F.; Lin, C.T.; Tang, P.C. MicroRNA-199a mediates mucin 1 expression in mouse uterus during implantation. Reprod. Fertil. Dev. 2014, 26, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.J.; Lees, M.; Matthews, L.C.; Kimber, S.J.; Forbes, K.; Aplin, J.D. MiR-145 suppresses embryo-epithelial juxtacrine communication at implantation by modulating maternal IGF1R. J. Cell Sci. 2015, 128, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Wang, S.; Wang, Z. Role of microRNAs in embryo implantation. Reprod. Biol. Endocrinol. 2017, 15, 90. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Gou, J.; Jia, J.; Zhao, X. MicroRNA-429 functions as a regulator of epithelial-mesenchymal transition by targeting Pcdh8 during murine embryo implantation. Hum. Reprod. 2015, 30, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Jia, J.; Gou, J.; Tong, A.; Liu, X.; Zhao, X.; Yi, T. Mmu-miR-126a-3p plays a role in murine embryo implantation by regulating Itga11. Reprod. Biomed. Online 2015, 31, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Zhao, Y.; Yu, Y.; Li, R.; Qiao, J. MiR-125b regulates endometrial receptivity by targeting MMP26 in women undergoing IVF-ET with elevated progesterone on HCG priming day. Sci. Rep. 2016, 6, 25302. [Google Scholar] [CrossRef] [PubMed]
- Cha, J.; Sun, X.; Dey, S.K. Mechanisms of implantation: Strategies for successful pregnancy. Nat. Med. 2012, 18, 1754–1767. [Google Scholar] [CrossRef] [PubMed]
- Vilella, F.; Moreno-Moya, J.M.; Balaguer, N.; Grasso, A.; Herrero, M.; Martínez, S.; Marcilla, A.; Simón, C. Hsa-miR-30d, secreted by the human endometrium, is taken up by the pre-implantation embryo and might modify its transcriptome. Development 2015, 142, 3210–3221. [Google Scholar] [CrossRef] [PubMed]
- Giacomini, E.; Vago, R.; Sanchez, A.M.; Podini, P.; Zarovni, N.; Murdica, V.; Rizzo, R.; Bortolotti, D.; Candiani, M.; Viganò, P. Secretome of in vitro cultured human embryos contains extracellular vesicles that are uptaken by the maternal side. Sci. Rep. 2017, 7, 5210. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.H.; Rome, S.; Jalabert, A.; Forterre, A.; Singh, H.; Hincks, C.L.; Salamonsen, L.A. Endometrial exosomes/microvesicles in the uterine microenvironment: A new paradigm for embryo-endometrial cross talk at implantation. PLoS ONE. 2013, 8, e58502. [Google Scholar] [CrossRef] [PubMed]
- Bendifallah, S.; Ouldamer, L.; Lavoue, V.; Canlorbe, G.; Raimond, E.; Coutant, C.; Graesslin, O.; Touboul, C.; Collinet, P.; Daraï, E.; et al. Patterns of recurrence and outcomes in surgically treated women with endometrial cancer according to ESMO-ESGO-ESTRO Consensus Conference risk groups: Results from the FRANCOGYN study Group. Gynecol. Oncol. 2017, 144, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Bokhman, J.V. Two pathogenetic types of endometrial carcinoma. Gynecol. Oncol. 1983, 15, 10–17. [Google Scholar] [CrossRef]
- Rose, P.G. Endometrial carcinoma. N. Engl. J. Med. 1997, 336, 1335. [Google Scholar] [CrossRef] [PubMed]
- Sherman, M.E.; Sturgeon, S.; Brinton, L.; Kurman, R.J. Endometrial cancer chemoprevention: Implications of diverse pathways of carcinogenesis. J. Cell Biochem. Suppl. 1995, 23, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Mutter, G.L.; Lin, M.C.; Fitzgerald, J.T.; Kum, J.B.; Baak, J.P.; Lees, J.A.; Weng, L.P.; Eng, C. Altered PTEN expression as a diagnostic marker for the earliest endometrial precancers. J. Natl. Cancer Inst. 2000, 92, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Furness, S.; Roberts, H.; Marjoribanks, J.; Lethaby, A.; Hickey, M.; Farquhar, C. Hormone therapy in postmenopausal women and risk of endometrial hyperplasia. Cochrane Database Syst. Rev. 2009, 2, CD000402. [Google Scholar] [CrossRef]
- Hecht, J.L.; Mutter, G.L. Molecular and pathologic aspects of endometrial carcinogenesis. J. Clin. Oncol. 2006, 24, 4783–4791. [Google Scholar] [CrossRef] [PubMed]
- Lax, S.F.; Kendall, B.; Tashiro, H.; Slebos, R.J.; Hedrick, L. The frequency of p53, K-ras mutations and microsatellite instability differs in uterine endometrioid and serous carcinoma: Evidence of distinct molecular genetic pathways. Cancer 2000, 88, 814–824. [Google Scholar] [CrossRef]
- Zagouri, F.; Bozas, G.; Kafantari, E.; Tsiatas, M.; Nikitas, N.; Dimopoulos, M.A.; Papadimitriou, C.A. Endometrial cancer: What is new in adjuvant and molecularly targeted therapy? Obstet. Gynecol. Int. 2010, 2010, 749579. [Google Scholar] [CrossRef] [PubMed]
- Vallone, C.; Rigon, G.; Gulia, C.; Baffa, A.; Votino, R.; Morosetti, G.; Zaami, S.; Briganti, V.; Catania, F.; Gaffi, M.; et al. Non-Coding RNAs and Endometrial Cancer. Genes 2018, 9, 187. [Google Scholar] [CrossRef] [PubMed]
- Klinge, C.M. Non-coding RNAs: Long non-coding RNAs and microRNAs in endocrine-related cancers. Endocr. Relat. Cancer 2018, 25, R259–R282. [Google Scholar] [CrossRef] [PubMed]
- Smolle, M.A.; Bullock, M.D.; Ling, H.; Pichler, M.; Haybaeck, J. Long Non-Coding RNAs in Endometrial Carcinoma. Int. J. Mol. Sci. 2015, 16, 26463–26472. [Google Scholar] [CrossRef] [PubMed]
- Vasilatou, D.; Sioulas, V.D.; Pappa, V.; Papageorgiou, S.G.; Vlahos, N.F. The role of miRNAs in endometrial cancer. Epigenomics 2015, 7, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.M.; Evangelisti, C.; Chiarini, F.; Grimaldi, C.; Cappellini, A.; Ognibene, A.; McCubrey, J.A. The emerging role of the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin signaling network in normal myelopoiesis and leukemogenesis. Biochim. Biophys. Acta 2010, 1803, 991–1002. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, B.; Hennessy, B.T.; Li, J.; Barkoh, B.A.; Luthra, R.; Mills, G.B.; Broaddus, R.R. Clinical assessment of PTEN loss in endometrial carcinoma: Immunohistochemistry out-performs gene sequencing. Mod. Pathol. 2012, 25, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Hou, X.; Li, Y.; Zhao, M. MiR-205 inhibits cell apoptosis by targeting phosphatase and tensin homolog deleted on chromosome ten in endometrial cancer Ishikawa cells. BMC Cancer 2014, 14, 440. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Liang, R. miR-205 promotes epithelial-mesenchymal transition by targeting AKT signaling in endometrial cancer cells. J. Obstet. Gynaecol. Res. 2015, 41, 1653–1660. [Google Scholar] [CrossRef] [PubMed]
- Torres, A.; Kozak, J.; Korolczuk, A.; Rycak, D.; Wdowiak, P.; Maciejewski, R.; Torres, K. Locked nucleic acid-inhibitor of miR-205 decreases endometrial cancer cells proliferation in vitro and in vivo. Oncotarget 2016, 7, 73651–73663. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Yan, L.; Zhao, X.; Li, C.; Fu, Y. microrna-21 overexpression contributes to cell proliferation by targeting PTEN in endometrioid endometrial cancer. Oncol. Lett. 2012, 4, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Takano, M.; Albergaria, A.; Briese, J.; Pomeranz, K.M.; Cloke, B.; Fusi, L.; Feroze-Zaidi, F.; Maywald, N.; Sajin, M.; et al. Mechanism and functional consequences of loss of FOXO1 expression in endometrioid endometrial cancer cells. Oncogene 2008, 27, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Kajihara, T.; Brosens, J.J.; Ishihara, O. The role of FOXO1 in the decidual transformation of the endometrium and early pregnancy. Med. Mol. Morphol. 2013, 46, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Myatt, S.S.; Wang, J.; Monteiro, L.J.; Christian, M.; Ho, K.K.; Fusi, L.; Dina, R.E.; Brosens, J.J.; Ghaem-Maghami, S.; Lam, E.W. Definition of microRNAs that repress expression of the tumor suppressor gene FOXO1 in endometrial cancer. Cancer Res. 2010, 70, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.K.; Lau, T.S.; Cheung, T.H.; Yim, S.F.; Lo, K.W.; Siu, N.S.; Chan, L.K.; Yu, M.Y.; Kwong, J.; Doran, G.; et al. Dysregulation of microRNA-204 mediates migration and invasion of endometrial cancer by regulating FOXC1. Int. J. Cancer 2012, 130, 1036–1045. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Zhu, Y.; Ma, T.; Pan, C.; Wang, J.; He, Z.; Li, Z.; Qi, X.; Chen, Y. miR-204 functions as a tumor suppressor by regulating SIX1 in NSCLC. FEBS Lett. 2014, 588, 3703–3712. [Google Scholar] [CrossRef] [PubMed]
- Lam, E.K.; Wang, X.; Shin, V.Y.; Zhang, S.; Morrison, H.; Sun, J.; Ng, E.K.; Yu, J.; Jin, H. A microRNA contribution to aberrant Ras activation in gastric cancer. Am. J. Transl. Res. 2011, 3, 209–218. [Google Scholar] [PubMed]
- Manning, B.D.; Cantley, L.C. AKT/PKB signaling: Navigating downstream. Cell 2007, 129, 1261–1274. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; He, Y.; Zhang, X.; Xing, B.; Sheng, Y.; Lu, H.; Wei, Z. Estrogen receptor-regulated microRNAs contribute to the BCL2/BAX imbalance in endometrial adenocarcinoma and precancerous lesions. Cancer Lett. 2012, 314, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Backes, F.J.; Walker, C.J.; Goodfellow, P.J.; Hade, E.M.; Agarwal, G.; Mutch, D.; Cohn, D.E.; Suarez, A.A. Estrogen receptor-alpha as a predictive biomarker in endometrioid endometrial cancer. Gynecol. Oncol. 2016, 141, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Lebeau, A.; Grob, T.; Holst, F.; Seyedi-Fazlollahi, N.; Moch, H.; Terracciano, L.; Turzynski, A.; Choschzick, M.; Sauter, G.; Simon, R. Oestrogen receptor gene (ESR1) amplification is frequent in endometrial carcinoma and its precursor lesions. J. Pathol. 2008, 216, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Jongen, V.; Briët, J.; de Jong, R.; ten Hoor, K.; Boezen, M.; van der Zee, A.; Nijman, H.; Hollema, H. Expression of estrogen receptor-alpha and -beta and progesterone receptor-A and -B in a large cohort of patients with endometrioid endometrial cancer. Gynecol. Oncol. 2009, 112, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.S.; Lambros, M.B.; Marchiò, C.; Reis-Filho, J.S. ESR1 amplification in endometrial carcinomas: Hope or hyperbole? J. Pathol. 2008, 216, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yan, Q.; Li, S.; Zhou, L.; Yang, H.; Yang, Y.; Liu, X.; Wan, X. Expression of the tumor suppressor miR-206 is associated with cellular proliferative inhibition and impairs invasion in ERα-positive endometrioid adenocarcinoma. Cancer Lett. 2012, 314, 41–53. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, N.A.; Wright, M.W.; Brister, J.R.; Ciufo, S.; Haddad, D.; McVeigh, R.; Rajput, B.; Robbertse, B.; Smith-White, B.; Ako-Adjei, D.; et al. Reference sequence (RefSeq) database at NCBI: Current status, taxonomic expansion and functional annotation. Nucleic Acids Res. 2016, 44, D733–D745. [Google Scholar] [CrossRef] [PubMed]
- Pengchong, H.; Tao, H. Expression of IGF-1R, VEGF-C and D2-40 and their correlation with lymph node metastasis in endometrial adenocarcinoma. Eur. J. Gynaecol. Oncol. 2011, 32, 660–664. [Google Scholar] [PubMed]
- Tu, C.; Wang, F.; Wan, J. MicroRNA-381 inhibits cell proliferation and invasion in endometrial carcinoma by targeting the IGF-1R. Mol. Med. Rep. 2018, 17, 4090–4098. [Google Scholar] [CrossRef] [PubMed]
- Banno, K.; Kisu, I.; Yanokura, M.; Masuda, K.; Ueki, A.; Kobayashi, Y.; Susumu, N.; Aoki, D. Epigenetics and genetics in endometrial cancer: New carcinogenic mechanisms and relationship with clinical practice. Epigenomics 2012, 4, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Tao, M.H.; Freudenheim, J.L. DNA methylation in endometrial cancer. Epigenetics 2010, 5, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.W.; Liu, J.C.; Deatherage, D.E.; Luo, J.; Mutch, D.G.; Goodfellow, P.J.; Miller, D.S.; Huang, T.H. Epigenetic repression of microRNA-129-2 leads to overexpression of SOX4 oncogene in endometrial cancer. Cancer Res. 2009, 69, 9038–9046. [Google Scholar] [CrossRef] [PubMed]
- Zighelboim, I.; Goodfellow, P.J.; Gao, F.; Gibb, R.K.; Powell, M.A.; Rader, J.S.; Mutch, D.G. Microsatellite instability and epigenetic inactivation of MLH1 and outcome of patients with endometrial carcinomas of the endometrioid type. J. Clin. Oncol. 2007, 25, 2042–2048. [Google Scholar] [CrossRef] [PubMed]
- Bueno, M.J.; Pérez de Castro, I.; Gómez de Cedrón, M.; Santos, J.; Calin, G.A.; Cigudosa, J.C.; Croce, C.M.; Fernández-Piqueras, J.; Malumbres, M. Genetic and epigenetic silencing of microRNA-203 enhances ABL1 and BCR-ABL1 oncogene expression. Cancer Cell 2008, 13, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.W.; Kuo, C.T.; Chen, J.H.; Goodfellow, P.J.; Huang, T.H.; Rader, J.S.; Uyar, D.S. Hypermethylation of miR-203 in endometrial carcinomas. Gynecol. Oncol. 2014, 133, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Liang, Y.; Su, Y.; An, Y.; Qu, P. miR-152 inhibits proliferation of human endometrial cancer cells via inducing G2/M phase arrest by suppressing CDC25B expression. Biomed. Pharmacother. 2018, 99, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Tsuruta, T.; Kozaki, K.; Uesugi, A.; Furuta, M.; Hirasawa, A.; Imoto, I.; Susumu, N.; Aoki, D.; Inazawa, J. miR-152 is a tumor suppressor microRNA that is silenced by DNA hypermethylation in endometrial cancer. Cancer Res. 2011, 71, 6450–6462. [Google Scholar] [CrossRef] [PubMed]
- Kraljevic Pavelic, S.; Sedic, M.; Bosnjak, H.; Spaventi, S.; Pavelic, K. Metastasis: New perspectives on an old problem. Mol. Cancer 2011, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, X.; Sun, K.X.; Xiu, Y.L.; Liu, B.L.; Feng, M.X.; Sang, X.B.; Zhao, Y. MicroRNA-93 Promotes Epithelial-Mesenchymal Transition of Endometrial Carcinoma Cells. PLoS ONE 2016, 11, e0165776. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Bao, W.; Qiu, M.; Liao, Y.; Che, Q.; Yang, T.; He, X.; Qiu, H.; Wan, X. Forkhead-box A1 suppresses the progression of endometrial cancer via crosstalk with estrogen receptor α. Oncol. Rep. 2014, 31, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Xu, X.; Xun, Q.; Yu, D.; Ling, J.; Guo, F.; Yan, Y.; Shi, J.; Hu, Y. microRNA-30c negatively regulates endometrial cancer cells by targeting metastasis-associated gene-1. Oncol. Rep. 2012, 27, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Xu, X.; Yan, Y.; Guo, F.; Li, J.; Hu, Y.; Zhou, H.; Xun, Q. Estrogen regulates the tumour suppressor MiRNA-30c and its target gene, MTA-1, in endometrial cancer. PLoS ONE 2014, 9, e90810. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Kaneuchi, M.; Watari, H.; Sudo, S.; Sakuragi, N. MicroRNA-106b modulates epithelial-mesenchymal transition by targeting TWIST1 in invasive endometrial cancer cell lines. Mol. Carcinog. 2014, 53, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Xu, Y.; He, T.; Qin, C.; Xu, J. Normal and disease-related biological functions of Twist1 and underlying molecular mechanisms. Cell Res. 2012, 22, 90–106. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.Q.; Liu, F.X.; Tang, H.L.; Su, Q. Expression and its clinical significance of hsa-miR-155 in serum of endometrial cancer. Zhonghua Fu Chan Ke Za Zhi 2010, 45, 772–774. [Google Scholar] [PubMed]
- Torres, A.; Torres, K.; Pesci, A.; Ceccaroni, M.; Paszkowski, T.; Cassandrini, P.; Zamboni, G.; Maciejewski, R. Deregulation of miR-100, miR-99a and miR-199b in tissues and plasma coexists with increased expression of mTOR kinase in endometrioid endometrial carcinoma. BMC Cancer 2012, 12, 369. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wang, T.T.; Lv, X.P. Expression and prognostic value of miRNA-29b in peripheral blood for endometrial cancer. Future Oncol. 2018, 14. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chen, Y.J.; Xu, K.; Xu, H.; Shen, X.Z.; Tu, R.Q. Circulating microRNAs as a fingerprint for endometrial endometrioid adenocarcinoma. PLoS ONE 2014, 9, e110767. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Qi, M.; Ma, C.; Lao, G.; Liu, Y.; Liu, Y.; Liu, Y. Let7a inhibits the growth of endometrial carcinoma cells by targeting Aurora-B. FEBS Lett. 2013, 587, 2523–2529. [Google Scholar] [CrossRef] [PubMed]
- Ihira, K.; Dong, P.; Xiong, Y.; Watari, H.; Konno, Y.; Hanley, S.J.; Noguchi, M.; Hirata, N.; Suizu, F.; Yamada, T.; et al. EZH2 inhibition suppresses endometrial cancer progression via miR-361/Twist axis. Oncotarget 2017, 8, 13509–13520. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Fan, Y.; Xu, W.; Chen, J.; Xu, C.; Wei, X.; Fang, D.; Feng, Y. miR-10b Inhibits Apoptosis and Promotes Proliferation and Invasion of Endometrial Cancer Cells via Targeting HOXB3. Cancer Biother. Radiopharm. 2016, 31, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.M.; Wan, X.H.; Sang, G.Y.; Zhao, J.D.; Zhu, Q.Y.; Wang, D.M. miR-15a-5p suppresses endometrial cancer cell growth via Wnt/β-catenin signaling pathway by inhibiting WNT3A. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4810–4818. [Google Scholar] [PubMed]
- Liu, P.; Wang, C.; Ma, C.; Wu, Q.; Zhang, W.; Lao, G. MicroRNA-23a regulates epithelial-to-mesenchymal transition in endometrial endometrioid adenocarcinoma by targeting SMAD3. Cancer Cell Int. 2016, 16, 67. [Google Scholar] [CrossRef] [PubMed]
- Lang, B.; Shang, C.; Meng, L. Targeted Silencing of S100A8 Gene by miR-24 to Increase Chemotherapy Sensitivity of Endometrial Carcinoma Cells to Paclitaxel. Med. Sci. Monit. 2016, 22, 1953–1958. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.X.; Xu, X.X.; Tan, B.Z.; Zhang, Z.; Zhou, X.D. MicroRNA-29b Inhibits Angiogenesis by Targeting VEGFA through the MAPK/ERK and PI3K/Akt Signaling Pathways in Endometrial Carcinoma. Cell Physiol. Biochem. 2017, 41, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Mitamura, T.; Watari, H.; Wang, L.; Kanno, H.; Kitagawa, M.; Hassan, M.K.; Kimura, T.; Tanino, M.; Nishihara, H.; Tanaka, S.; et al. microRNA 31 functions as an endometrial cancer oncogene by suppressing Hippo tumor suppressor pathway. Mol. Cancer. 2014, 13, 97. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Meng, W.; Zeng, J.; Hu, H.; Lu, L. MiR-34c oligonucleotide enhances chemosensitivity of Ishikawa cell to cisplatin by inducing apoptosis. Cell Biol. Int. 2013, 37, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Shang, C.; Lu, Y.M.; Meng, L.R. MicroRNA-125b down-regulation mediates endometrial cancer invasion by targeting ERBB2. Med. Sci. Monit. 2012, 18, BR149–BR155. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Qian, J.; Li, C.; Kwok, L.; Cheng, F.; Liu, P.; Perdomo, C.; Kotton, D.; Vaziri, C.; Anderlind, C.; et al. miR-129 regulates cell proliferation by downregulating Cdk6 expression. Cell Cycle 2010, 9, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Gao, F.; Liu, N. L1CAM promotes epithelial to mesenchymal transition and formation of cancer initiating cells in human endometrial cancer. Exp. Ther. Med. 2018, 15, 2792–2797. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, C.; Jiang, Y.; Wan, Y.; Zhou, S.; Cheng, W. Tumor-suppressor role of miR-139-5p in endometrial cancer. Cancer Cell Int. 2018, 18, 51. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.S.; Jeon, H.W.; Kim, Y.B.; Kim, Y.A.; Kim, M.A.; Kang, S.B. Aberrant microRNA expression in endometrial carcinoma using formalin-fixed paraffin-embedded (FFPE) tissues. PLoS ONE 2013, 8, e81421. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Zhang, D.; Shi, H.; Bian, Y.; Guo, R. miR-143 inhibits endometrial cancer cell proliferation and metastasis by targeting MAPK1. Oncotarget 2017, 8, 84384–84395. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Adila, S.; Zhang, X.; Dong, Y.; Li, W.; Zhou, M.; Li, T. MicroRNA expression signature profile and its clinical significance in endometrioid carcinoma. Zhonghua Bing Li Xue Za Zhi 2014, 43, 88–94. [Google Scholar] [PubMed]
- Qu, J.; Zhang, L.; Li, L.; Su, Y. miR-148b functions as a tumor suppressor by targeting endoplasmic reticulum metallo protease 1 in human endometrial cancer cells. Oncol. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Devor, E.J.; Miecznikowski, J.; Schickling, B.M.; Gonzalez-Bosquet, J.; Lankes, H.A.; Thaker, P.; Argenta, P.A.; Pearl, M.L.; Zweizig, S.L.; Mannel, R.S.; et al. Dysregulation of miR-181c expression influences recurrence of endometrial endometrioid adenocarcinoma by modulating NOTCH2 expression: An NRG Oncology/Gynecologic Oncology Group study. Gynecol. Oncol. 2017, 147, 648–653. [Google Scholar] [CrossRef] [PubMed]
- Ruan, H.; Liang, X.; Zhao, W.; Ma, L.; Zhao, Y. The effects of microRNA-183 promots cell proliferation and invasion by targeting MMP-9 in endometrial cancer. Biomed. Pharmacother. 2017, 89, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Dong, P.; Kaneuchi, M.; Watari, H.; Hamada, J.; Sudo, S.; Ju, J.; Sakuragi, N. MicroRNA-194 inhibits epithelial to mesenchymal transition of endometrial cancer cells by targeting oncogene BMI-1. Mol. Cancer 2011, 10, 99. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Wang, X.; Ruan, L.; Fu, J.; Liu, F.; Qu, J. MiR-200a promotes epithelial-mesenchymal transition of endometrial cancer cells by negatively regulating FOXA2 expression. Pharmazie 2017, 72, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Snowdon, J.; Zhang, X.; Childs, T.; Tron, V.A.; Feilotter, H. The microRNA-200 family is upregulated in endometrial carcinoma. PLoS ONE 2011, 6, e22828. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Nian, Z.; Jingjing, Z.; Tao, L.; Quan, L. MicroRNA-424/E2F6 feedback loop modulates cell invasion, migration and EMT in endometrial carcinoma. Oncotarget 2017, 8, 114281–114291. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Y.; Wu, H.J.; Ma, H.D.; Xu, L.P.; Huo, Y.; Yin, L.R. MicroRNA-503 suppresses proliferation and cell-cycle progression of endometrioid endometrial cancer by negatively regulating cyclin D1. FEBS J. 2013, 280, 3768–3779. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Xu, H.; Meng, Y.; Kuang, Y. miR-944 acts as a prognostic marker and promotes the tumor progression in endometrial cancer. Biomed. Pharmacother. 2017, 88, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Qu, Y.W.; Li, Y.P. Over-expression of miR-1271 inhibits endometrial cancer cells proliferation and induces cell apoptosis by targeting CDK1. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2816–2822. [Google Scholar] [PubMed]
- Hosseini, E.S.; Meryet-Figuiere, M.; Sabzalipoor, H.; Kashani, H.H.; Nikzad, H.; Asemi, Z. Dysregulated expression of long noncoding RNAs in gynecologic cancers. Mol. Cancer 2017, 16, 107. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.T.; Sun, Y.M.; Huang, W.; He, B.; Zhao, Y.N.; Chen, Y.Q. Genome-wide Long Non-coding RNA Analysis Identified Circulating LncRNAs as Novel Non-invasive Diagnostic Biomarkers for Gynecological Disease. Sci. Rep. 2016, 6, 23343. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Y.; Yang, Z.; Liu, K.; Wang, D. Genome-Wide Microarray Analysis of Long Non-Coding RNAs in Eutopic Secretory Endometrium with Endometriosis. Cell Physiol. Biochem. 2015, 37, 2231–2245. [Google Scholar] [CrossRef] [PubMed]
- Akrami, R.; Jacobsen, A.; Hoell, J.; Schultz, N.; Sander, C.; Larsson, E. Comprehensive analysis of long non-coding RNAs in ovarian cancer reveals global patterns and targeted DNA amplification. PLoS ONE 2013, 8, e80306. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yang, Y.; Trovik, J.; Sun, K.; Zhou, L.; Jiang, P.; Lau, T.S.; Hoivik, E.A.; Salvesen, H.B.; Sun, H.; et al. A novel wnt regulatory axis in endometrioid endometrial cancer. Cancer Res. 2014, 74, 5103–5117. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Qu, X.; Li, W.; Zhong, X.; Li, P.; Yang, S.; Chen, X.; Shao, M.; Zhang, L. The long non-coding RNA, GAS5, enhances gefitinib-induced cell death in innate EGFR tyrosine kinase inhibitor-resistant lung adenocarcinoma cells with wide-type EGFR via downregulation of the IGF-1R expression. J. Hematol. Oncol. 2015, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, W.; Jiang, J.; Bao, E.; Xu, D.; Zeng, Y.; Tao, L.; Qiu, J. Downregulation of GAS5 promotes bladder cancer cell proliferation, partly by regulating CDK6. PLoS ONE 2013, 8, e73991. [Google Scholar] [CrossRef] [PubMed]
- Mourtada-Maarabouni, M.; Pickard, M.R.; Hedge, V.L.; Farzaneh, F.; Williams, G.T. GAS5, a non-protein-coding RNA, controls apoptosis and is downregulated in breast cancer. Oncogene 2009, 28, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Jin, F.Y.; Xia, R.; Kong, R.; Li, J.H.; Xu, T.P.; Liu, Y.W.; Zhang, E.B.; Liu, X.H.; De, W. Decreased expression of long noncoding RNA GAS5 indicates a poor prognosis and promotes cell proliferation in gastric cancer. BMC Cancer 2014, 14, 319. [Google Scholar] [CrossRef] [PubMed]
- Tu, Z.Q.; Li, R.J.; Mei, J.Z.; Li, X.H. Down-regulation of long non-coding RNA GAS5 is associated with the prognosis of hepatocellular carcinoma. Int. J. Clin. Exp. Pathol. 2014, 7, 4303–4309. [Google Scholar] [PubMed]
- Guo, C.; Song, W.Q.; Sun, P.; Jin, L.; Dai, H.Y. LncRNA-GAS5 induces PTEN expression through inhibiting miR-103 in endometrial cancer cells. J. Biomed. Sci. 2015, 22, 100. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.A.; Shah, N.; Wang, K.C.; Kim, J.; Horlings, H.M.; Wong, D.J.; Tsai, M.C.; Hung, T.; Argani, P.; Rinn, J.L.; et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature 2010, 464, 1071–1076. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Bao, W.; Li, X.; Chen, Z.; Che, Q.; Wang, H.; Wan, X.P. The long non-coding RNA HOTAIR is upregulated in endometrial carcinoma and correlates with poor prognosis. Int. J. Mol. Med. 2014, 33, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Ke, P.; Guo, L.; Wang, W.; Tan, H.; Liang, Y.; Yao, S. Lentivirus-mediated RNA interference targeting the long noncoding RNA HOTAIR inhibits proliferation and invasion of endometrial carcinoma cells in vitro and in vivo. Int. J. Gynecol. Cancer 2014, 24, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Zemel, S.; Bartolomei, M.S.; Tilghman, S.M. Physical linkage of two mammalian imprinted genes, H19 and insulin-like growth factor 2. Nat. Genet. 1992, 2, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Onyango, P.; Brandenburg, S.; Wu, Y.; Hsieh, C.L.; Feinberg, A.P. Loss of imprinting in colorectal cancer linked to hypomethylation of H19 and IGF2. Cancer Res. 2002, 62, 6442–6446. [Google Scholar] [PubMed]
- Ariel, I.; Miao, H.Q.; Ji, X.R.; Schneider, T.; Roll, D.; de Groot, N.; Hochberg, A.; Ayesh, S. Imprinted H19 oncofetal RNA is a candidate tumour marker for hepatocellular carcinoma. Mol. Pathol. 1998, 51, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.M.; Wong, H.L.; Birnstein, E.A.; Wolff, E.M.; Liang, G.; Yang, A.S. Examination of IGF2 and H19 loss of imprinting in bladder cancer. Cancer Res. 2007, 67, 10753–10758. [Google Scholar] [CrossRef] [PubMed]
- Tanos, V.; Prus, D.; Ayesh, S.; Weinstein, D.; Tykocinski, M.L.; De-Groot, N.; Hochberg, A.; Ariel, I. Expression of the imprinted H19 oncofetal RNA in epithelial ovarian cancer. Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 85, 7–11. [Google Scholar] [CrossRef]
- Fu, X.; Liu, Y.; Zhuang, C.; Liu, L.; Cai, Z.; Huang, W. Synthetic artificial microRNAs targeting UCA1-MALAT1 or c-Myc inhibit malignant phenotypes of bladder cancer cells T24 and 5637. Mol. Biosyst. 2015, 11, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, D.; Vittone, G.; Romani, M.; Purrello, M. Noncoding RNAs in Health and Disease. Int. J. Genom. 2018, 2018, 9135073. [Google Scholar] [CrossRef]
- Tian, Y.; Zhang, X.; Hao, Y.; Fang, Z.; He, Y. Potential roles of abnormally expressed long noncoding RNA UCA1 and Malat-1 in metastasis of melanoma. Melanoma Res. 2014, 24, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Li, Y.; Lv, W.; Zhang, G.; Tian, X.; Li, X.; Cheng, H.; Zhu, C. UCA1 promotes cell proliferation and invasion and inhibits apoptosis through regulation of the miR129-SOX4 pathway in renal cell carcinoma. Oncol. Targets Ther. 2018, 11, 2475–2487. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ye, C.; Liu, J.; Hu, Y. UCA1 confers paclitaxel resistance to ovarian cancer through miR-129/ABCB1 axis. Biochem. Biophys. Res. Commun. 2018, 501, 1034–1040. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Shen, Y.; Tseng, K.F.; Liu, W.; Duan, H.; Meng, W. Silencing of UCA1, a poor prognostic factor, inhibited the migration of endometrial cancer cell. Cancer Biomark. 2016, 17, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, Q.; Bao, C.; Li, S.; Guo, W.; Zhao, J.; Chen, D.; Gu, J.; He, X.; Huang, S. Circular RNA is enriched and stable in exosomes: A promising biomarker for cancer diagnosis. Cell Res. 2015, 25, 981–984. [Google Scholar] [CrossRef] [PubMed]
- Kulcheski, F.R.; Christoff, A.P.; Margis, R. Circular RNAs are miRNA sponges and can be used as a new class of biomarker. J. Biotechnol. 2016, 238, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Gong, Z.; Shen, Y.; Fang, Y.; Zhong, S. Circular RNA expression in extracellular vesicles isolated from serum of patients with endometrial cancer. Epigenomics 2018, 10, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.J.; Byrne, F.L.; Takenaka, K.; Modesitt, S.C.; Olzomer, E.M.; Mills, J.D.; Farrell, R.; Hoehn, K.L.; Janitz, M. Analysis of the circular RNA transcriptome in endometrial cancer. Oncotarget 2017, 9, 5786–5796. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.; Li, X.; Wu, S.; Zhang, Y.; Pang, H.; Chen, W. Microarray expression profile of lncRNAs and the upregulated ASLNC04080 lncRNA in human endometrial carcinoma. Int. J. Oncol. 2015, 46, 2125–2137. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wang, D.; Wang, N.; Long, Z.; Ren, X. Long Non-Coding RNA BANCR Promotes Endometrial Cancer Cell Proliferation and Invasion by Regulating MMP2 and MMP1 via ERK/MAPK Signaling Pathway. Cell Physiol. Biochem. 2016, 40, 644–656. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wei, X.; Zhao, L.; Shi, L.; Cheng, J.; Kang, S.; Zhang, H.; Zhang, J.; Li, L.; Zhang, H.; et al. The rs6983267 SNP and long non-coding RNA CARLo-5 are associated with endometrial carcinoma. Environ. Mol. Mutagen. 2016, 57, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.; Cao, H.; Li, Y.; Wang, J.; Cui, Z. Knockdown of lncRNA CCAT2 inhibits endometrial cancer cells growth and metastasis via sponging miR-216b. Cancer Biomark. 2017, 21, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Q.; Li, H. LncRNA FER1L4 suppresses cancer cell proliferation and cycle by regulating PTEN expression in endometrial carcinoma. Biochem. Biophys. Res. Commun. 2016, 478, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, C.C.; Lee, W.Y.W.; Trovik, J.; Chung, T.K.H.; Kwong, J. Long non-coding RNA HAND2-AS1 inhibits invasion and metastasis in endometrioid endometrial carcinoma through inactivating neuromedin U. Cancer Lett. 2018, 413, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Xin, W.; Liu, X.; Ding, J.; Zhao, J.; Zhou, Y.; Wu, Q.; Hua, K. Long non-coding RNA derived miR-205-5p modulates human endometrial cancer by targeting PTEN. Am. J. Transl. Res. 2015, 7, 2433–2441. [Google Scholar] [PubMed]
- Li, W.; Li, H.; Zhang, L.; Hu, M.; Li, F.; Deng, J.; An, M.; Wu, S.; Ma, R.; Lu, J.; et al. Long non-coding RNA LINC00672 contributes to p53 protein-mediated gene suppression and promotes endometrial cancer chemosensitivity. J. Biol. Chem. 2017, 292, 5801–5813. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Li, D.; Huo, J.; Kong, F.; Yang, H.; Ma, X. LINC01016 promotes the malignant phenotype of endometrial cancer cells by regulating the miR-302a-3p/miR-3130-3p/NFYA/SATB1 axis. Cell Death Dis. 2018, 9, 303. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Qian, Z.; Yan, D.; Li, L.; Huang, L. LncRNA-MEG3 inhibits cell proliferation of endometrial carcinoma by repressing Notch signaling. Biomed. Pharmacother. 2016, 82, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wei, D.; Yang, C.; Sun, H.; Lu, T.; Kuang, D. Overexpression of long noncoding RNA, NEAT1 promotes cell proliferation, invasion and migration in endometrial endometrioid adenocarcinoma. Biomed. Pharmacother. 2016, 84, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.H.; Zhang, X.Y.; Zhou, L.N.; Wan, Y.; Song, L.L.; Gu, W.L.; Liu, R.; Ma, Y.N.; Meng, H.R.; Tian, Y.L.; et al. LncRNA SNHG8 participates in the development of endometrial carcinoma through regulating c-MET expression by miR-152. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 1629–1637. [Google Scholar] [CrossRef] [PubMed]
- Shang, C.; Lang, B.; Ao, C.N.; Meng, L. Long non-coding RNA tumor suppressor candidate 7 advances chemotherapy sensitivity of endometrial carcinoma through targeted silencing of miR-23b. Tumour Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed]
- Fuldeore, M.J.; Soliman, A.M. Prevalence and Symptomatic Burden of Diagnosed Endometriosis in the United States: National Estimates from a Cross-Sectional Survey of 59,411 Women. Gynecol. Obstet. Investig. 2017, 82, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.W. Endometriosis and ovarian cancer: Potential benefits and harms of screening and risk-reducing surgery. Fertil. Steril. 2015, 104, 813–830. [Google Scholar] [CrossRef] [PubMed]
- Garai, J.; Molnar, V.; Varga, T.; Koppan, M.; Torok, A.; Bodis, J. Endometriosis: Harmful survival of an ectopic tissue. Front. Biosci. 2006, 11, 595–619. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Mutlu, L.; Grechukhina, O.; Taylor, H.S. Circulating microRNAs as potential biomarkers for endometriosis. Fertil. Steril. 2015, 103, 1252.e1–1260.e1. [Google Scholar] [CrossRef] [PubMed]
- Saare, M.; Rekker, K.; Laisk-Podar, T.; Rahmioglu, N.; Zondervan, K.; Salumets, A.; Götte, M.; Peters, M. Challenges in endometriosis miRNA studies—From tissue heterogeneity to disease specific miRNAs. Biochim. Biophys. Acta 2017, 1863, 2282–2292. [Google Scholar] [CrossRef] [PubMed]
- Braza-Boïls, A.; Marí-Alexandre, J.; Gilabert, J.; Sánchez-Izquierdo, D.; España, F.; Estellés, A.; Gilabert-Estellés, J. MicroRNA expression profile in endometriosis: Its relation to angiogenesis and fibrinolytic factors. Hum. Reprod. 2014, 29, 978–988. [Google Scholar] [CrossRef] [PubMed]
- Filigheddu, N.; Gregnanin, I.; Porporato, P.; Surico, D.; Perego, B.; Galli, L.; Patrignani, C.; Graziani, A.; Surico, N. Differential expression of micrornas between eutopic and ectopic endometrium in ovarian endometriosis. J. Biomed. Biotechnol. 2010, 2010, 369549. [Google Scholar] [CrossRef] [PubMed]
- Burney, R.O.; Hamilton, A.E.; Aghajanova, L.; Vo, K.C.; Nezhat, C.N.; Lessey, B.A.; Giudice, L.C. MicroRNA expression profiling of eutopic secretory endometrium in women with versus without endometriosis. Mol. Hum. Reprod. 2009, 15, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Laudanski, P.; Charkiewicz, R.; Kuzmicki, M.; Szamatowicz, J.; Charkiewicz, A.; Niklinski, J. MicroRNAs expression profiling of eutopic proliferative endometrium in women with ovarian endometriosis. Reprod. Biol. Endocrinol. 2013, 11, 78. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Lee, S.K.; Park, J.H.; Lee, J.H.; Yun, B.H.; Park, J.H.; Seo, S.K.; Cho, S.; Choi, Y.S. Ginsenoside Rg3 Decreases Fibrotic and Invasive Nature of Endometriosis by Modulating miRNA-27b: In Vitro and In Vivo Studies. Sci. Rep. 2017, 7, 17670. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gu, C.; Ye, M.; Zhang, Z.; Li, L.; Fan, W.; Meng, Y. Integration analysis of microRNA and mRNA paired expression profiling identifies deregulated microRNA-transcription factor-gene regulatory networks in ovarian endometriosis. Reprod. Biol. Endocrinol. 2018, 16, 4. [Google Scholar] [CrossRef] [PubMed]
- Laschke, M.W.; Menger, M.D. In vitro and in vivo approaches to study angiogenesis in the pathophysiology and therapy of endometriosis. Hum. Reprod. Update 2007, 13, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Ohlsson—Teague, E.M.; Print, C.G.; Hull, M.L. The role of microRNAs in endometriosis and associatedreproductive conditions. Hum. Reprod. Update 2010, 16, 142–165. [Google Scholar] [CrossRef] [PubMed]
- Braza-Boïls, A.; Gilabert-Estellés, J.; Ramón, L.A.; Gilabert, J.; Marí-Alexandre, J.; Chirivella, M.; España, F.; Estellés, A. Peritoneal fluid reduces angiogenesis-related microRNA expression in cell cultures of endometrial and endometriotic tissues from women with endometriosis. PLoS ONE 2013, 8, e62370. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.J.; Bai, X.G.; Teng, Y.L.; Song, L.; Lu, N.; Yang, R.Q. miRNA-15a-5p regulates VEGFA in endometrial mesenchymal stem cells and contributes to the pathogenesis of endometriosis. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 3319–3326. [Google Scholar] [PubMed]
- Harp, D.; Driss, A.; Mehrabi, S.; Chowdhury, I.; Xu, W.; Liu, D.; Garcia-Barrio, M.; Taylor, R.N.; Gold, B.; Jefferson, S.; et al. Exosomes derived from endometriotic stromal cells have enhanced angiogenic effects in vitro. Cell Tissue Res. 2016, 365, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Hull, M.L.; Nisenblat, V. Tissue and circulating microRNA influence reproductive function in endometrial disease. Reprod. Biomed. Online 2013, 27, 515–529. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Liu, H.Y. Small RNA molecules in endometriosis: Pathogenesis and therapeutic aspects. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 183, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Nothnick, W.B.; Al-Hendy, A.; Lue, J.R. Circulating Micro-RNAs as Diagnostic Biomarkers for Endometriosis: Privation and Promise. J. Minim. Invasive Gynecol. 2015, 22, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.T.; Zhao, Y.N.; Han, B.W.; Hong, S.J.; Chen, Y.Q. Circulating microRNAs identified in a genome-wide serum microRNA expression analysis as noninvasive biomarkers for endometriosis. J. Clin. Endocrinol. Metab. 2013, 98, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Jia, S.Z.; Yang, Y.; Lang, J.; Sun, P.; Leng, J. Plasma miR-17–5p, miR-20a and miR-22 are down-regulated in women with endometriosis. Hum. Reprod. 2013, 28, 322–330. [Google Scholar] [CrossRef] [PubMed]
- Rekker, K.; Saare, M.; Roost, A.M.; Kaart, T.; Sõritsa, D.; Karro, H.; Sõritsa, A.; Simón, C.; Salumets, A.; Peters, M. Circulating miR-200-family micro-RNAs have altered plasma levels in patients with endometriosis and vary with blood collection time. Fertil. Steril. 2015, 104, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Ghazal, S.; McKinnon, B.; Zhou, J.; Mueller, M.; Men, Y.; Yang, L.; Mueller, M.; Flannery, C.; Huang, Y.; Taylor, H.S. H19 lncRNA alters stromal cell growth via IGF signaling in the endometrium of women with endometriosis. EMBO Mol. Med. 2015, 7, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Liu, Y.; Xu, W.; Sun, Y.; Lu, J.; Wang, F.; Wei, M.; Shen, J.; Hou, J.; Gao, X.; et al. Long noncoding RNA MALAT-1 is a new potential therapeutic target for castration resistant prostate cancer. J. Urol. 2013, 190, 2278–2287. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Shen, B.; Tan, M.; Mu, X.; Qin, Y.; Zhang, F.; Liu, Y. TGF-beta-induced upregulation of malat1 promotes bladder cancer metastasis by associating with suz12. Clin. Cancer Res. 2014, 20, 1531–1541. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Chen, C.; Liu, Y.; Wu, C. 17 beta-Estradiol treatment inhibits breast cell proliferation, migration and invasion by decreasing MALAT-1 RNA level. Biochem. Biophys. Res. Commun. 2014, 445, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhang, C.; Chen, R.; Xiong, H.; Qiu, F.; Liu, S.; Zhang, M.; Wang, F.; Wang, Y.; Zhou, X.; et al. Disrupting MALAT1/miR-200c sponge decreases invasion and migration in endometrioid endometrial carcinoma. Cancer Lett. 2016, 383, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Chen, Y.; Zhao, Y.; Xu, C.; Zhang, A.; Zhang, Q.; Wang, D.; He, J.; Hua, W.; Duan, P. miR-200c suppresses endometriosis by targeting MALAT1 in vitro and in vivo. Stem Cell Res. Ther. 2017, 8, 251. [Google Scholar] [CrossRef] [PubMed]
- Cravello, L.; Porcu, G.; D’Ercole, C.; Roger, V.; Blanc, B. Identification and treatment of endometritis. Contracept. Fertil. Sex. 1997, 25, 585–586. [Google Scholar] [PubMed]
- Gilmore, H.; Fleischhacker, D.; Hecht, J.L. Diagnosis of chronic endometritis in biopsies with stromal breakdown. Hum. Pathol. 2007, 38, 581–584. [Google Scholar] [CrossRef] [PubMed]
- Johnston-MacAnanny, E.B.; Hartnett, J.; Engmann, L.L.; Nulsen, J.C.; Sanders, M.M.; Benadiva, C.A. Chronic endometritis is a frequent finding in women with recurrent implantation failure after in vitro fertilization. Fertil. Steril. 2010, 93, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Cicinelli, E.; Matteo, M.; Tinelli, R.; Pinto, V.; Marinaccio, M.; Indraccolo, U.; De Ziegler, D.; Resta, L. Chronic endometritis due to common bacteria is prevalent in women with recurrent miscarriage as confirmed by improved pregnancy outcome after antibiotic treatment. Reprod. Sci. 2014, 21, 640–647. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.; Frydman, R. Hysteroscopy and sperm infection. Contracept. Fertil. Sex. 1996, 24, 549–551. [Google Scholar] [PubMed]
- Féghali, J.; Bakar, J.; Mayenga, J.M.; Ségard, L.; Hamou, J.; Driguez, P.; Belaisch-Allart, J. Systematic hysteroscopy prior to in vitro fertilization. Gynecol. Obstet. Fertil. 2003, 31, 127–131. [Google Scholar] [CrossRef]
- Espinoza, J.; Erez, O.; Romero, R. Preconceptional antibiotic treatment to prevent preterm birth in women with a previous preterm delivery. Am. J. Obstet. Gynecol. 2006, 194, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, C.; Cicinelli, E.; Guglielmino, M.R.; Ragusa, M.; Farina, M.; Palumbo, M.A.; Cianci, A. Altered transcriptional regulation of cytokines, growth factors and apoptotic proteins in the endometrium of infertile women with chronic endometritis. Am. J. Reprod. Immunol. 2013, 69, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Matteo, M.; Cicinelli, E.; Greco, P.; Massenzio, F.; Baldini, D.; Falagario, T.; Rosenberg, P.; Castellana, L.; Specchia, G.; Liso, A. Abnormal pattern of lymphocyte subpopulations in the endometrium of infertile women with chronic endometritis. Am. J. Reprod. Immunol. 2009, 61, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Kitaya, K.; Yasuo, T. Immunohistochemistrical and clinicopathological characterization of chronic endometritis. Am. J. Reprod. Immunol. 2011, 66, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Kim, Y.S.; Yoon, T.K.; Lee, W.S. Chronic endometritis and infertility. Clin. Exp. Reprod. Med. 2016, 43, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Guzeloglu-Kayisli, O.; Kayisli, U.A.; Taylor, H.S. The role of growth factors and cytokines during implantation: Endocrine and paracrine interactions. Semin. Reprod. Med. 2009, 27, 62–79. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, C.; Caruso, S.; Battaglia, R.; Iraci Sareri, M.; La Ferlita, A.; Strino, F.; Bonaventura, G.; Di Mauro, M.; Barcellona, M.L.; Perciavalle, V.; et al. MiR-27a-3p and miR-124-3p, upregulated in endometrium and serum from women affected by Chronic Endometritis, are new potential molecular markers of endometrial receptivity. Am. J. Reprod. Immunol. 2018, 16, e12858. [Google Scholar] [CrossRef] [PubMed]
- Hailemariam, D.; Ibrahim, S.; Hoelker, M.; Drillich, M.; Heuwieser, W.; Looft, C.; Cinar, M.U.; Tholen, E.; Schellander, K.; Tesfaye, D. MicroRNA-regulated molecular mechanism underlying bovine subclinical endometritis. Reprod. Fertil. Dev. 2014, 26, 898–913. [Google Scholar] [CrossRef] [PubMed]
- Dimitriadis, E.; White, C.A.; Jones, R.L.; Salamonsen, L.A. Cytokines, chemokines and growth factors in endometrium related to implantation. Hum. Reprod. Update 2005, 11, 613–630. [Google Scholar] [CrossRef] [PubMed]
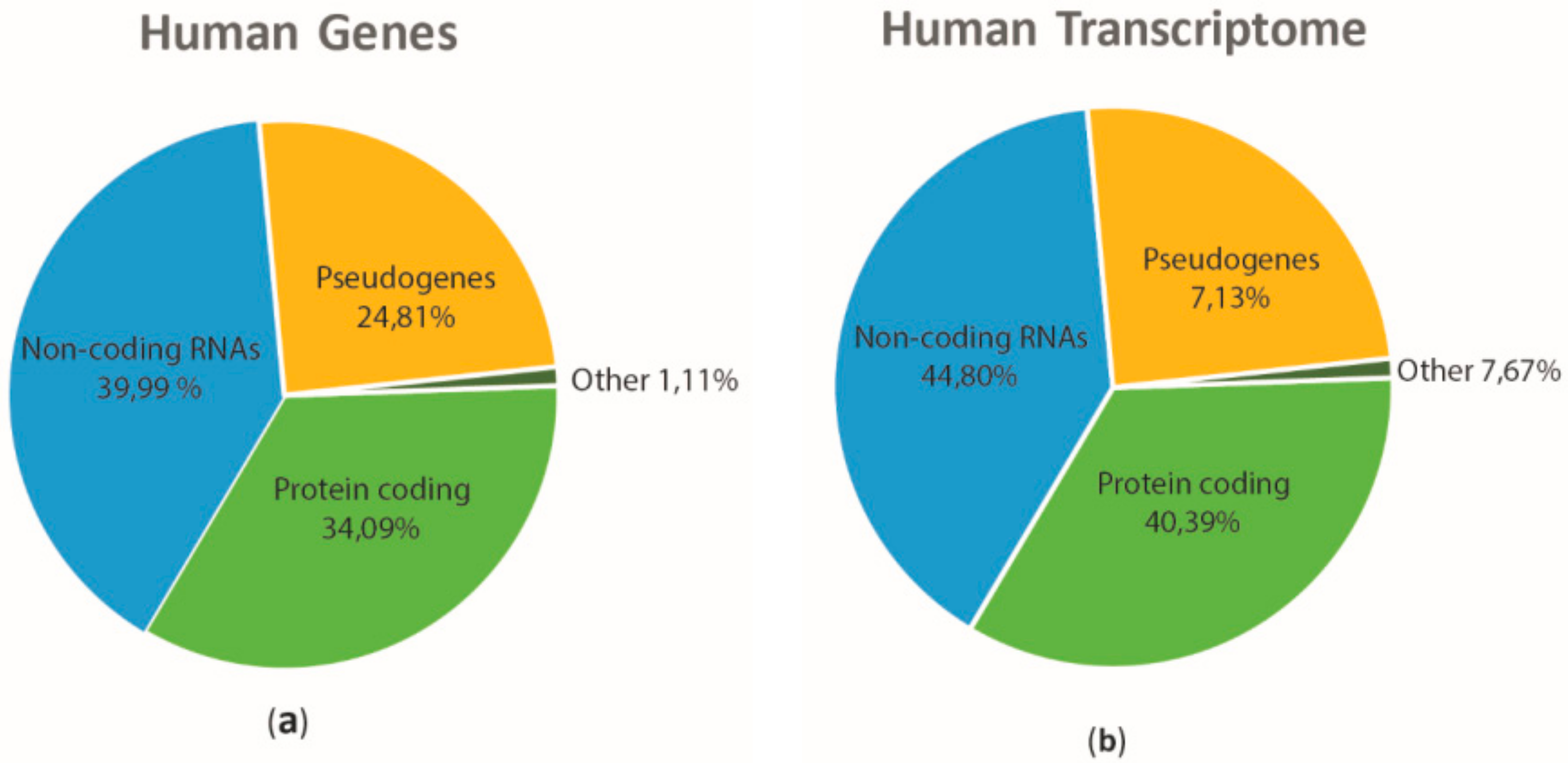

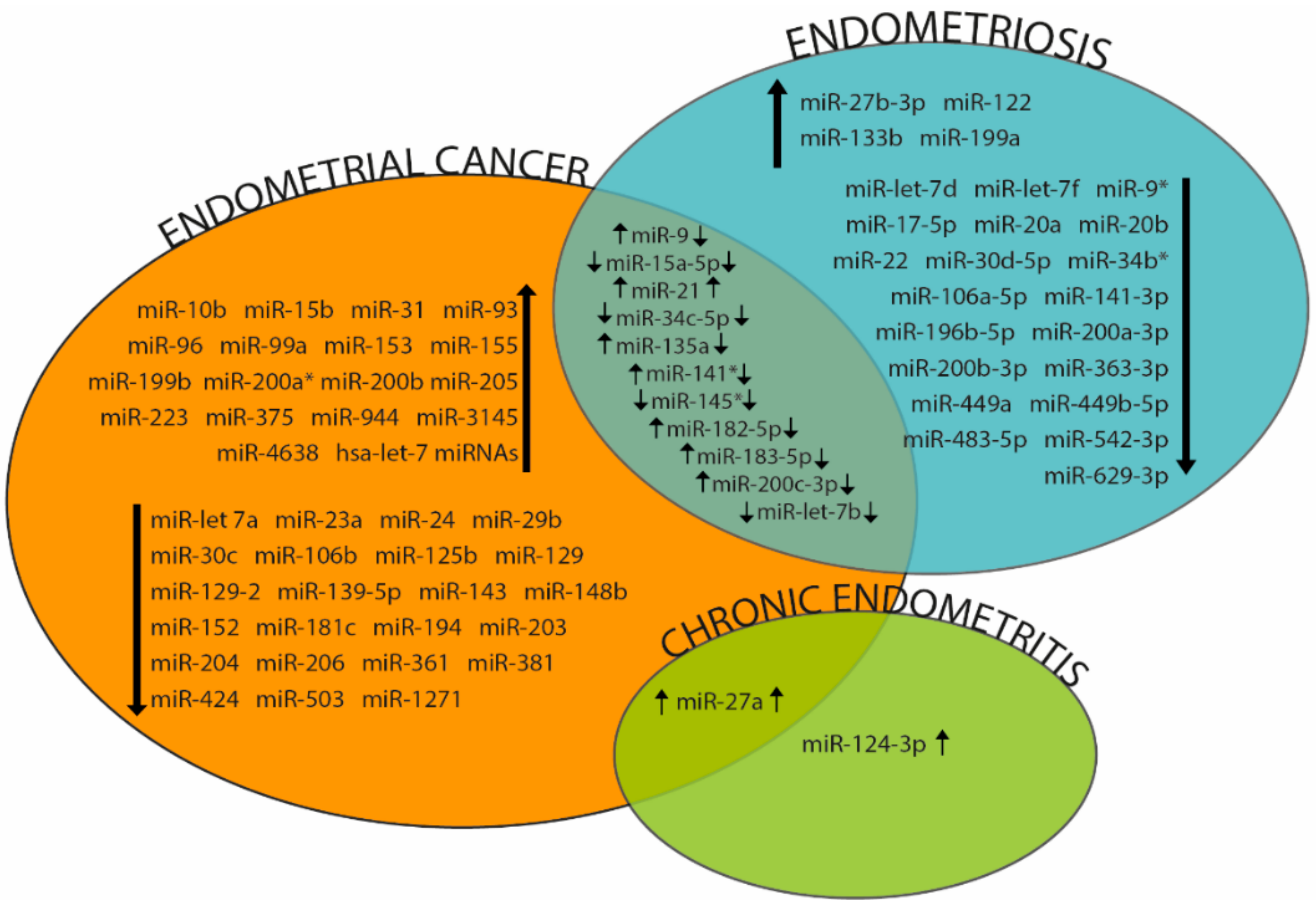
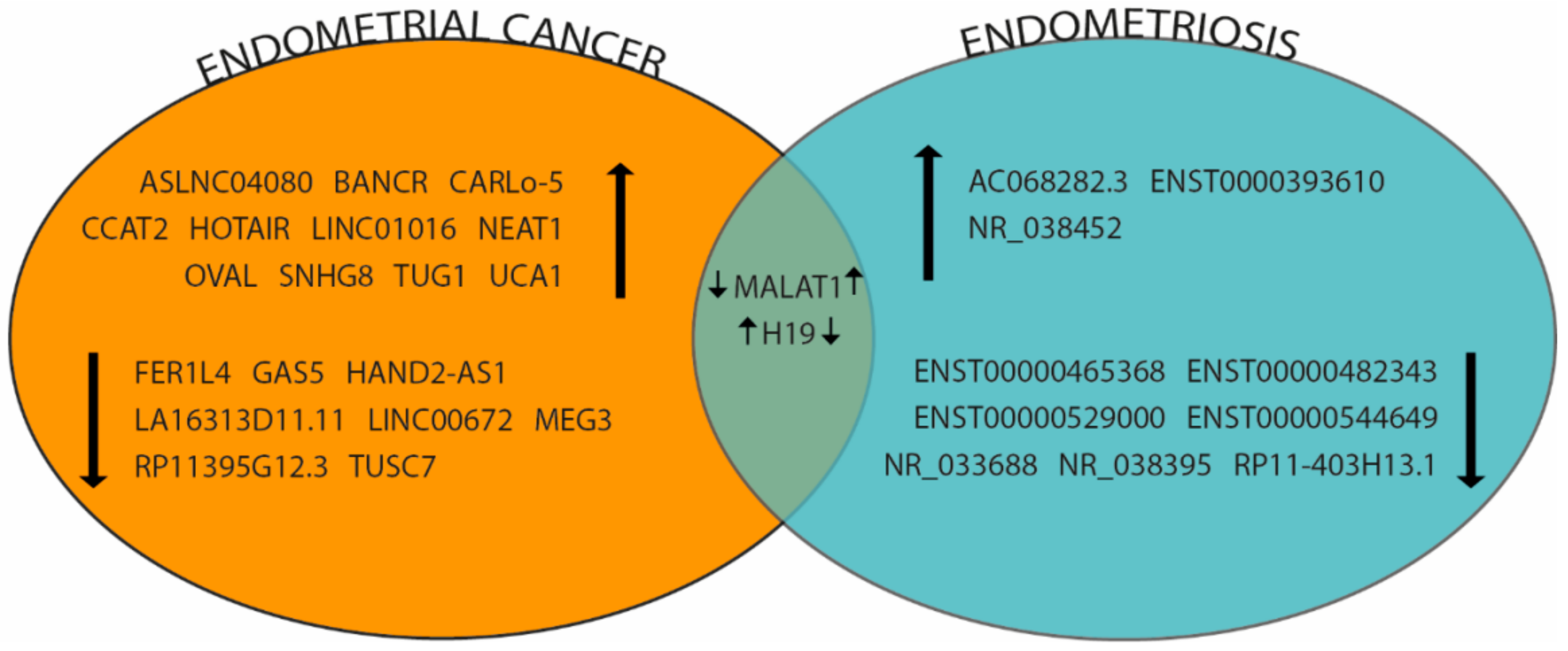
| miRNAs | Species | Sample | Reference |
|---|---|---|---|
| mmu-let-7a | mouse | endometrium | [23] |
| mmu-let-7b | mouse | endometrium | [23] |
| hsa-let-7e* | human | exosomes from endometrial cell lines | [34] |
| hsa-let-7f-2* | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-30b | human | endometrium | [24] |
| hsa-miR-30d | human | endometrium/endometrial fluid | [24,32] |
| hsa-miR-122 | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-124 | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-125b | human | endometrial cell lines | [30] |
| mmu-miR-126-3p | mouse | endometrium | [29] |
| hsa-miR-129* | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-142-3p | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-145 | human | endometrium | [26] |
| mmu-miR-199a | mouse | endometrium | [25] |
| hsa-miR-222* | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-376c | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-409-3p | human | exosomes from endometrial cell lines | [34] |
| mmu-miR-429 | mouse | endometrium | [28] |
| hsa-miR-432 | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-451 | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-494 | human | endometrium | [24] |
| hsa-miR-520h | human | exosomes from endometrial cell lines | [34] |
| hsa-miR-1248 | human | exosomes from endometrial cell lines | [34] |
| miRNAs | Expression | Sample | Reference |
|---|---|---|---|
| Has-let-7 miRNAs | up | EEC | [61] |
| miR-let-7a | down | EC | [89] |
| miR-let-7b | down | EC | [90] |
| miR-9 | up | EEC | [56] |
| miR-10b | up | EC | [91] |
| miR-15a-5p | down | EC | [92] |
| miR-15b | up | Plasma in EEC patients | [88] |
| miR-21 | up | EEC | [53] |
| miR-23a | down | EEC | [93] |
| miR-24 | down | EC | [94] |
| miR-27a | up | EEC, Plasma in EEC patients | [56,88] |
| miR-29b | down | EC, Plasma | [87,95] |
| miR-30c | down | EC | [81,82] |
| miR-31 | up | EC | [96] |
| miR-34c | down | EC | [97] |
| miR-96 | up | EEC | [56] |
| miR-99a | up | Plasma in EEC | [86] |
| miR-106b | down | EEC | [83] |
| miR-125b | down | EEC | [98] |
| miR-129 | down | EC | [99] |
| miR-129-2 | down | EC | [72] |
| miR-135a | up | EC | [100] |
| miR-139-5p | down | EC | [101] |
| miR-141 | up | EEC | [102] |
| miR-143 | down | EC | [103] |
| miR-145 | down | EEC | [104] |
| miR-148b | down | EC | [105] |
| miR-152 | down | EC | [76] |
| miR-153 | up | EC | [56] |
| miR-155 | Up | Serum | [85] |
| miR-181c | down | EEC | [106] |
| miR-182-5p | up | EEC | [56,102] |
| miR-183-5p | up | EC | [108] |
| miR-194 | down | EC | [109] |
| miR-199b | up | Plasma in EEC patients | [86] |
| miR-200a* | up | EC | [102,108] |
| miR-200b | up | EEC | [102] |
| miR-200c-3p | up | EC | [110] |
| miR-203 | down | EC | [74] |
| miR-204 | down | EC | [57] |
| miR-205 | up | EEC | [50,51,102] |
| miR-206 | down | EC | [66] |
| miR-223 | up | Plasma in EEC | [88] |
| miR-361 | down | EC | [90] |
| miR-375 | up | EC | [100] |
| miR-381 | down | EC | [69] |
| miR-424 | down | EC | [111] |
| miR-503 | down | EEC | [112] |
| miR-944 | up | EC | [113] |
| miR-1271 | down | EC | [114] |
| miR-3145 | up | Plasma in EEC patients | [88] |
| miR-4638 | up | Plasma in EEC patients | [88] |
| lncRNAs | Expression | EC Type | Reference |
|---|---|---|---|
| ASLNC04080 | up | EC | [144] |
| BANCR | up | EEC | [145] |
| CARLo-5 | up | EC | [146] |
| CCAT2 | up | EC | [147] |
| FER1L4 | down | EC | [148] |
| GAS5 | down | EC | [125] |
| H19 | up | EC | [115] |
| HAND2-AS1 | down | EEC | [149] |
| HOTAIR | up | EC | [127] |
| LA16313D11.11 | down | EC | [150] |
| LINC00672 | down | EC | [151] |
| LINC01016 | up | EC | [152] |
| MALAT1 | down | EEC | [119] |
| MEG3 | down | EC | [153] |
| NEAT1 | up | EEC | [154] |
| OVAL | up | EEC | [46,118] |
| RP11395G12.3 | down | EC | [150] |
| SNHG8 | up | EC | [155] |
| TUSC7 | down | EC | [156] |
| UCA1 | up | EC | [139] |
| miRNAs | Expression | Sample | Reference |
|---|---|---|---|
| let-7b | down | serum | [160] |
| let-7d | down | serum | [160] |
| let-7f | down | serum | [160] |
| miR-9 | down | endometrium | [164] |
| miR-9* | down | endometrium\serum | [164,176] |
| miR-15a-5p | down | endometrium | [171] |
| miR-17-5p | down | ovarian endometrium\plasma | [165,169,177] |
| miR-20a | down | ovarian endometrium\plasma | [165,169,177] |
| miR-20b-5p | down | endometrium | [167] |
| miR-21 | up | endometrial exosomes | [172] |
| miR-22 | down | plasma | [175] |
| miR-27b-3p | up | endometrium | [166] |
| miR-30d-5p | down | endometrium | [167] |
| miR-34b* | down | endometrium | [164] |
| miR-34c-5p | down | endometrium | [164] |
| miR-106a-5p | down | endometrium | [167] |
| miR-122 | up | serum | [176] |
| miR-133b | up | endometrium | [167] |
| miR-135a | down | serum | [160] |
| miR-141* | down | serum | [176] |
| miR-141-3p | down | endometrium\plasma | [167,176] |
| miR-145* | down | serum | [176] |
| miR-182-5p | down | endometrium | [167] |
| miR-183-5p | down | endometrium | [167] |
| miR-196b-5p | down | endometrium | [167] |
| miR-199a | up | serum | [176] |
| miR-200a-3p | down | endometrium\plasma | [161,167,178] |
| miR-200b-3p | down | endometrium\plasma | [161,167,178] |
| miR-200c-3p | down | endometrium | [161,167] |
| miR-363-3p | down | endometrium | [167] |
| miR-449a | down | endometrium | [167] |
| miR-449b-5p | down | endometrium | [167] |
| miR-483-5p | down | endometrium | [165] |
| miR-542-3p | down | serum | [176] |
| miR-629-3p | down | endometrium | [165] |
| lncRNAs | Expression | Sample | Reference |
|---|---|---|---|
| AC068282.3 | up | endometrium | [117] |
| ENST00000393610 | up | serum | [116] |
| ENST00000465368 | down | serum | [116] |
| ENST00000482343 | down | serum | [116] |
| ENST00000529000 | down | serum | [116] |
| ENST00000544649 | down | serum | [116] |
| H19 | down | endometrium | [179] |
| MALAT1 | up | endometrium | [184] |
| NR_033688 | down | serum | [116] |
| NR_038395 | down | serum | [116] |
| NR_038452 | up | serum | [116] |
| RP11-403H13.1 | down | endometrium | [117] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
La Ferlita, A.; Battaglia, R.; Andronico, F.; Caruso, S.; Cianci, A.; Purrello, M.; Di Pietro, C. Non-Coding RNAs in Endometrial Physiopathology. Int. J. Mol. Sci. 2018, 19, 2120. https://doi.org/10.3390/ijms19072120
La Ferlita A, Battaglia R, Andronico F, Caruso S, Cianci A, Purrello M, Di Pietro C. Non-Coding RNAs in Endometrial Physiopathology. International Journal of Molecular Sciences. 2018; 19(7):2120. https://doi.org/10.3390/ijms19072120
Chicago/Turabian StyleLa Ferlita, Alessandro, Rosalia Battaglia, Francesca Andronico, Salvatore Caruso, Antonio Cianci, Michele Purrello, and Cinzia Di Pietro. 2018. "Non-Coding RNAs in Endometrial Physiopathology" International Journal of Molecular Sciences 19, no. 7: 2120. https://doi.org/10.3390/ijms19072120
APA StyleLa Ferlita, A., Battaglia, R., Andronico, F., Caruso, S., Cianci, A., Purrello, M., & Di Pietro, C. (2018). Non-Coding RNAs in Endometrial Physiopathology. International Journal of Molecular Sciences, 19(7), 2120. https://doi.org/10.3390/ijms19072120





