Ethyl Acetate Extract of Scindapsus cf. hederaceus Exerts the Inhibitory Bioactivity on Human Non-Small Cell Lung Cancer Cells through Modulating ER Stress
Abstract
1. Introduction
2. Results
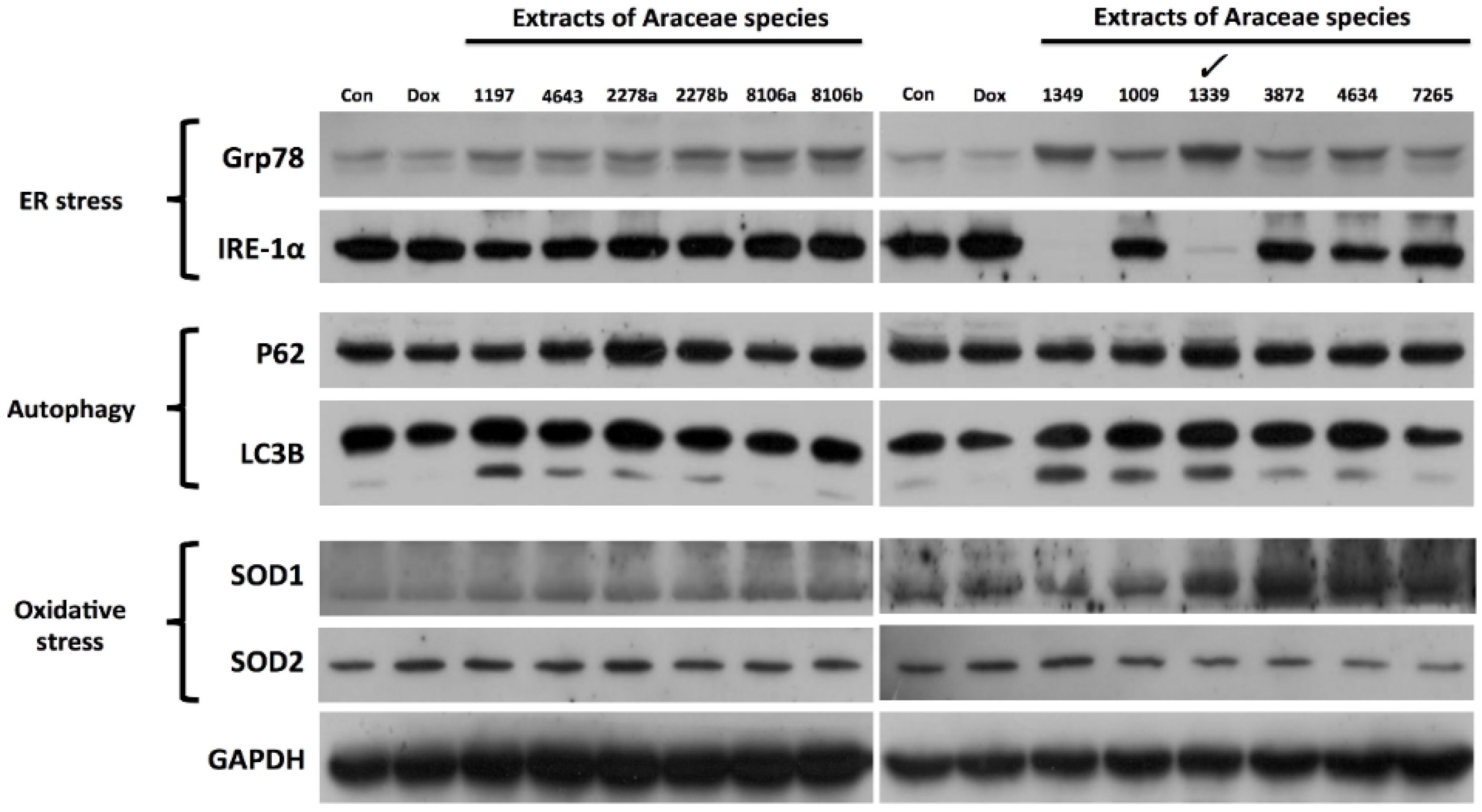
2.1. Functional Screening to Identify Crude Extract as a UPR Targeting from Plants in the Family Araceae
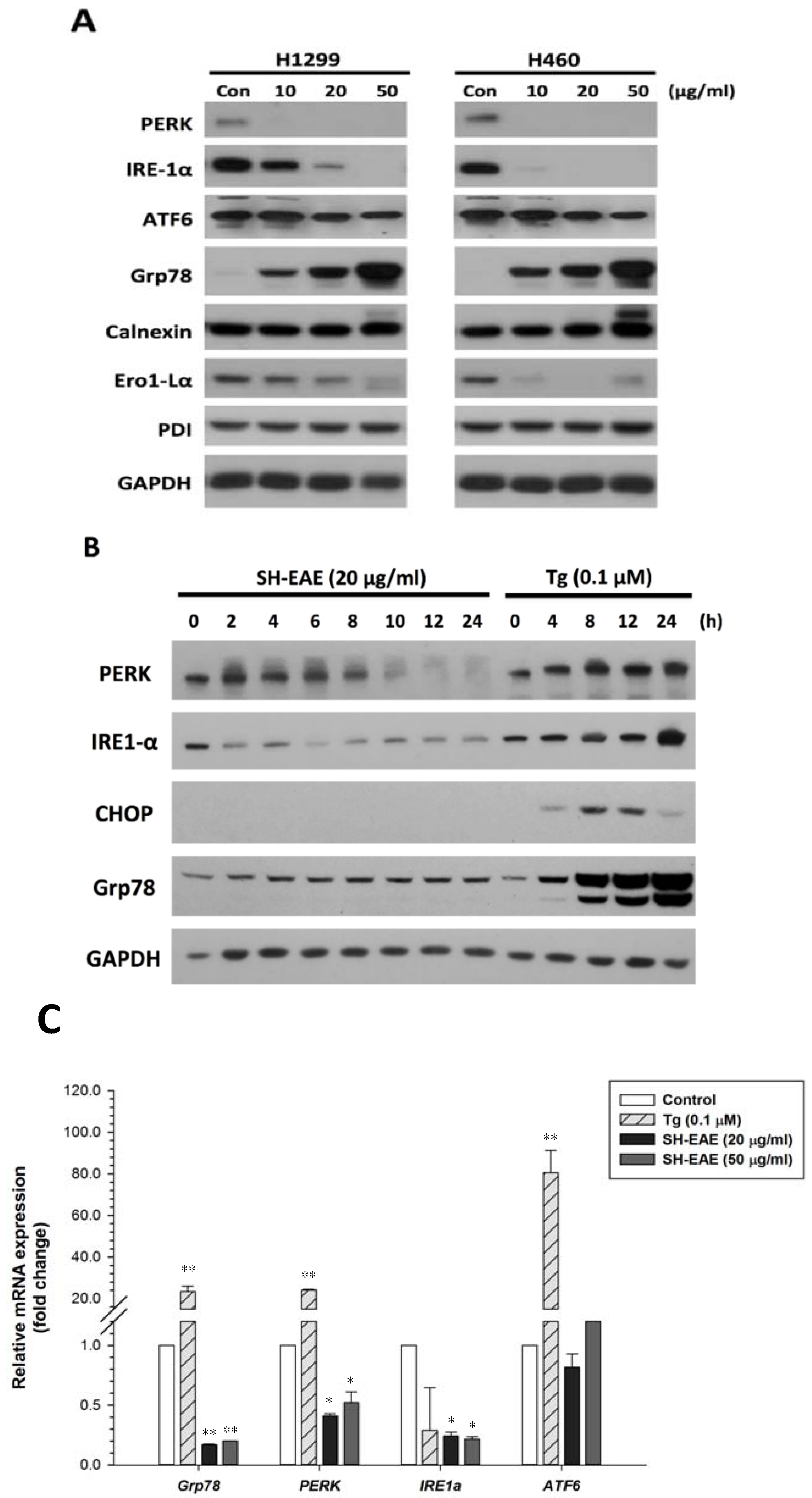
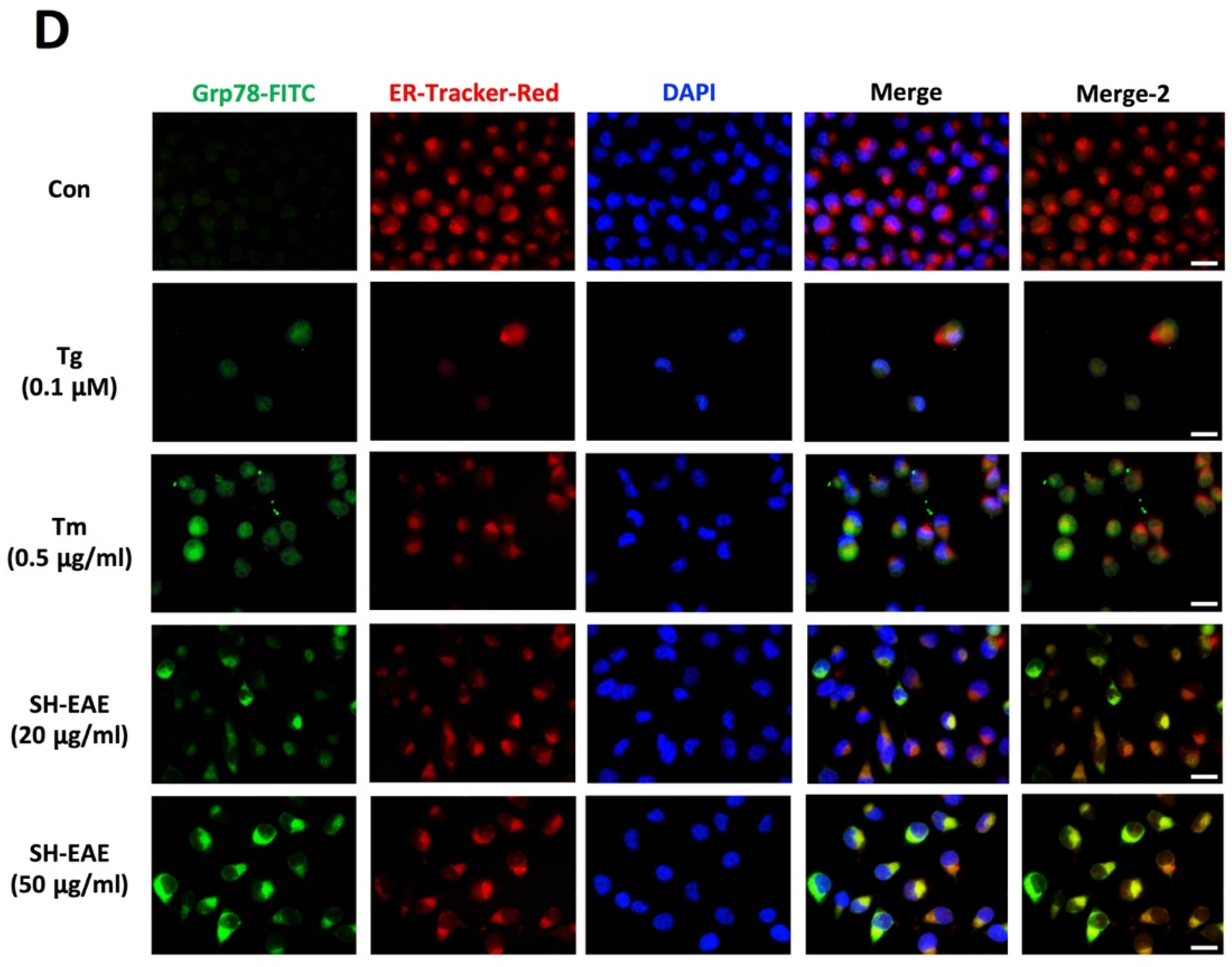
2.2. SH-EAE Altered the Key Regulators of Unfolded Protein Response (UPR)
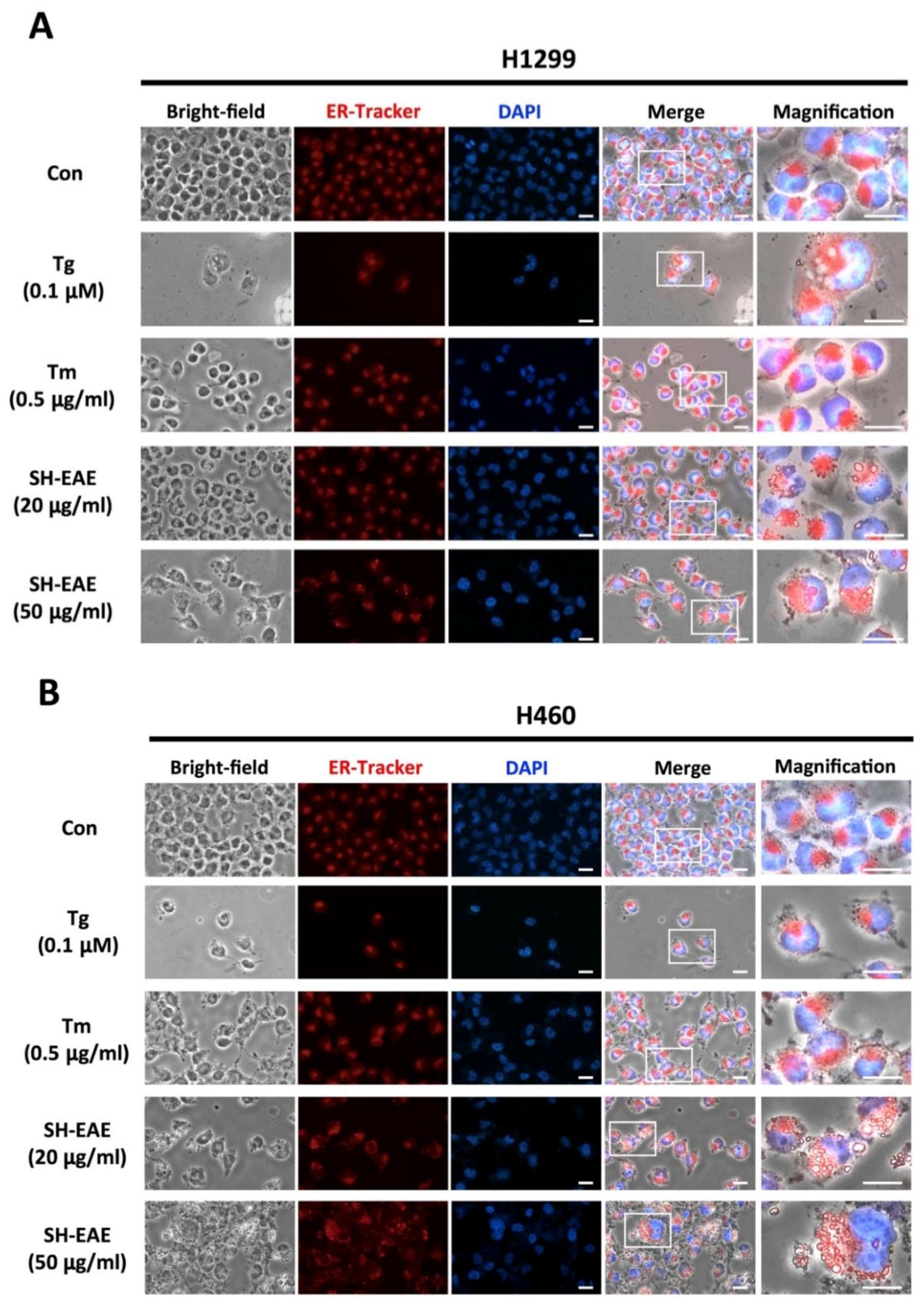
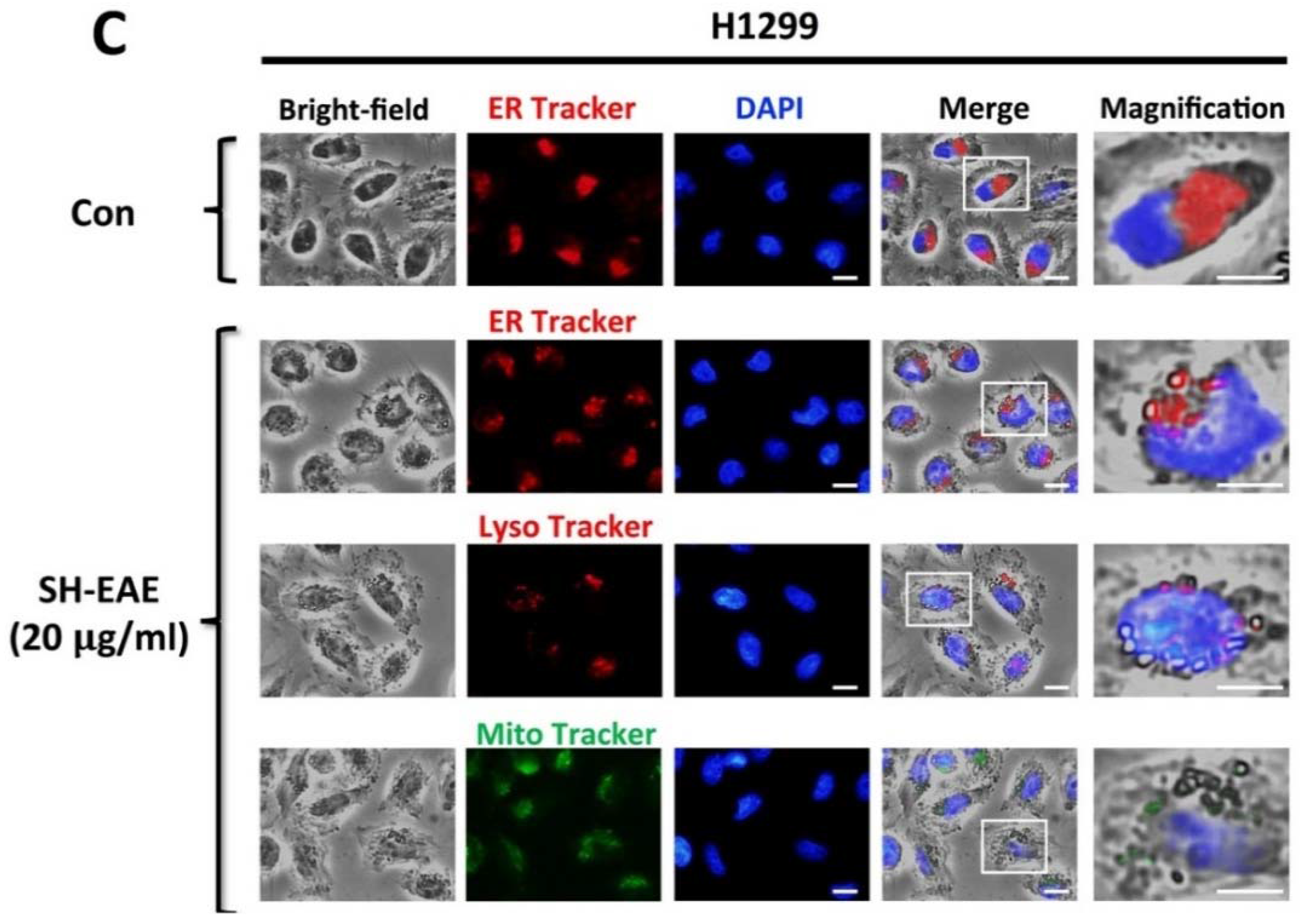
2.3. Induction of Cytoplasmic Vacuoles in SH-EAE-Treated Cells
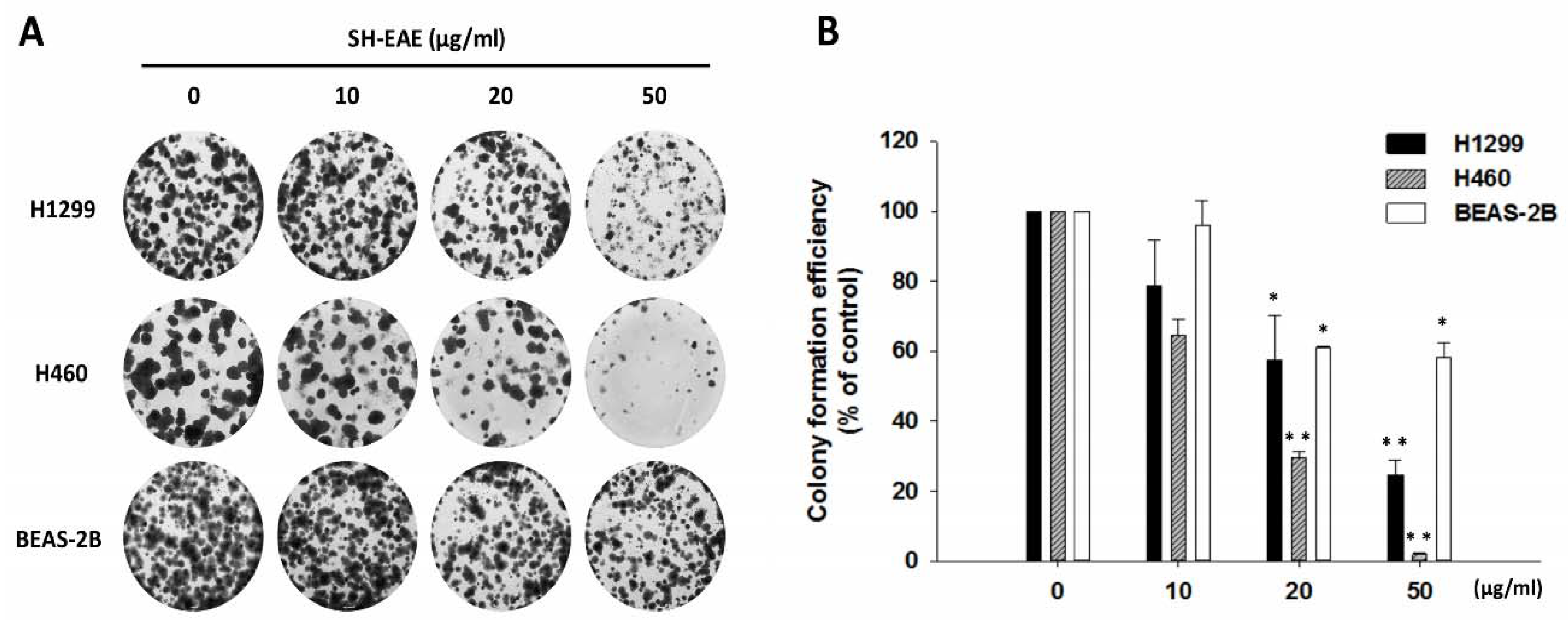
2.4. SH-EAE Selectively Decreased Colony-Forming Ability in NSCLC Cells
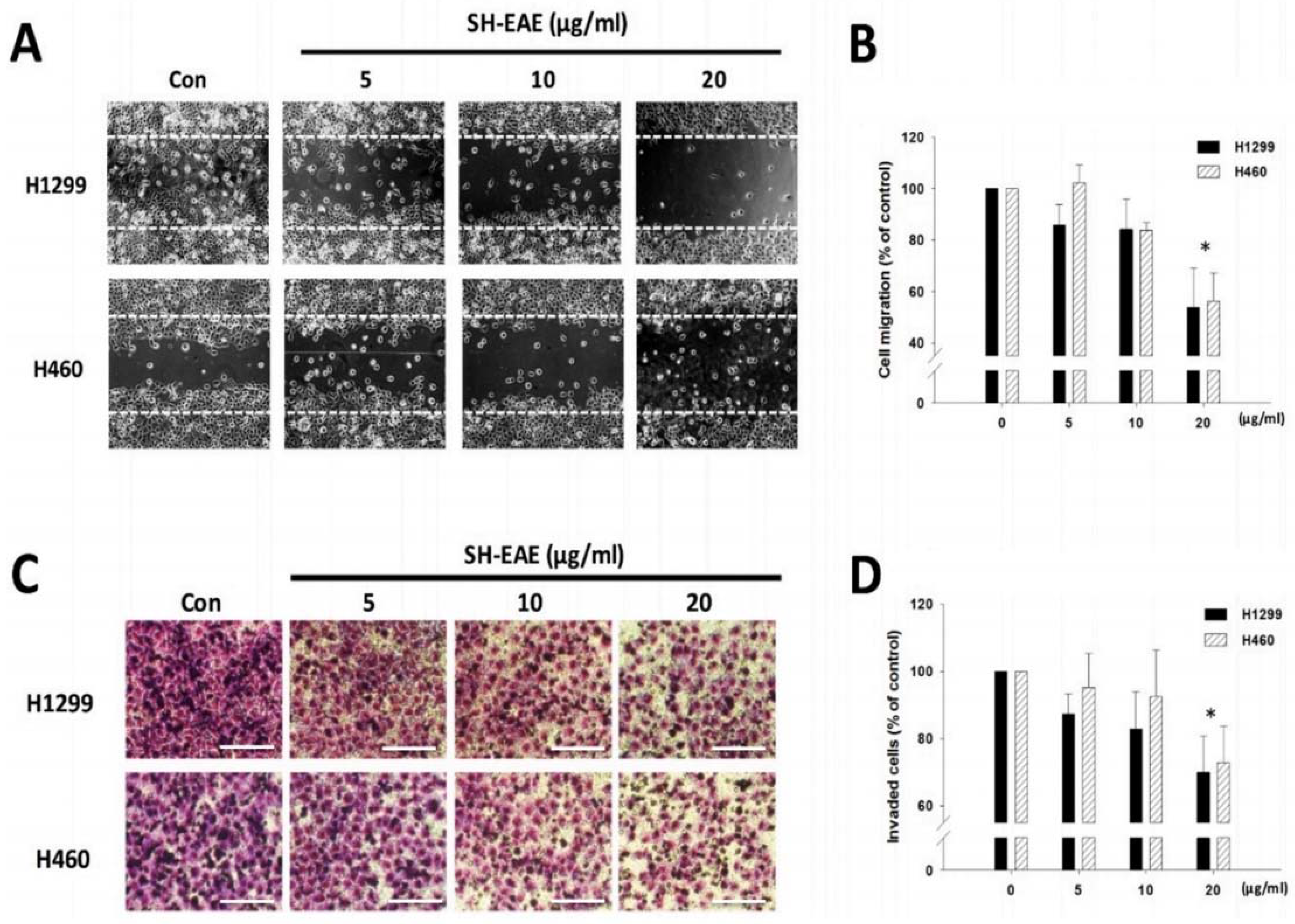
2.5. SH-EAE Reduced the Migration and Invasive Ability of NSCLC Cells
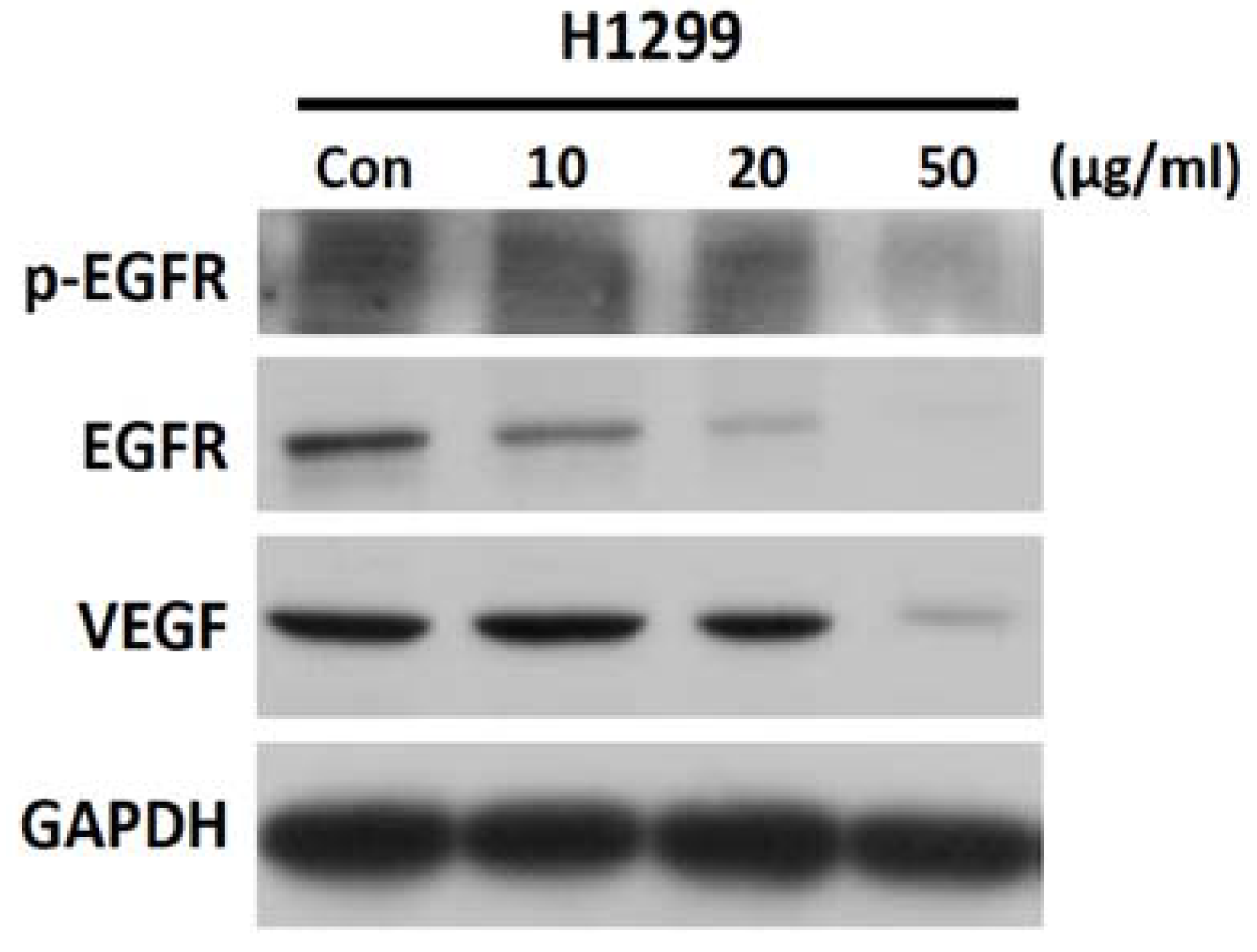
2.6. SH-EAE Reduced Phosphorylation and Expression of EGFR and VEGF Signaling in H1299 Cells
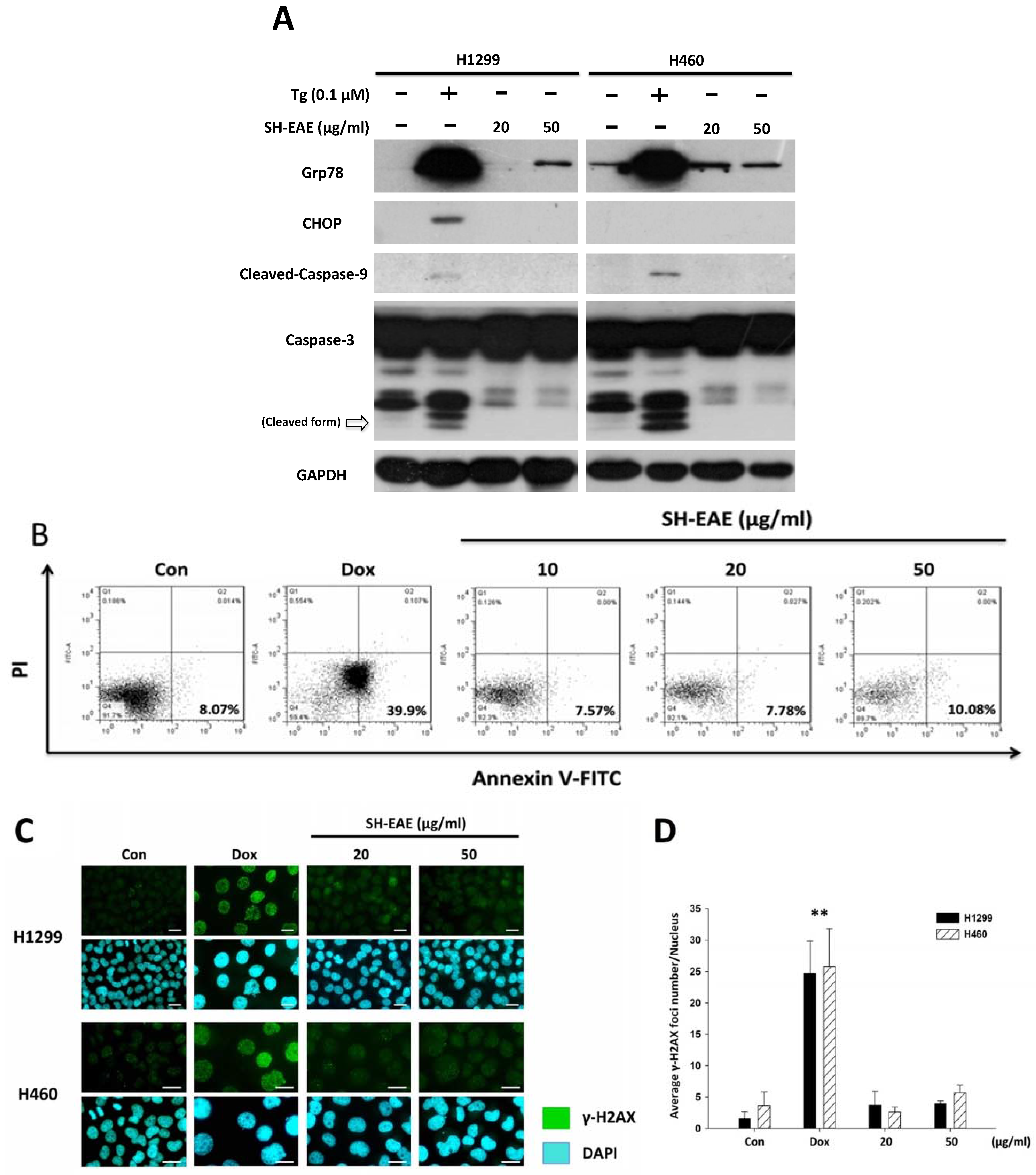
2.7. SH-EAE Was Not Poisonous Enough to Induce DNA Damage and Apoptosis in NSCLC Cells
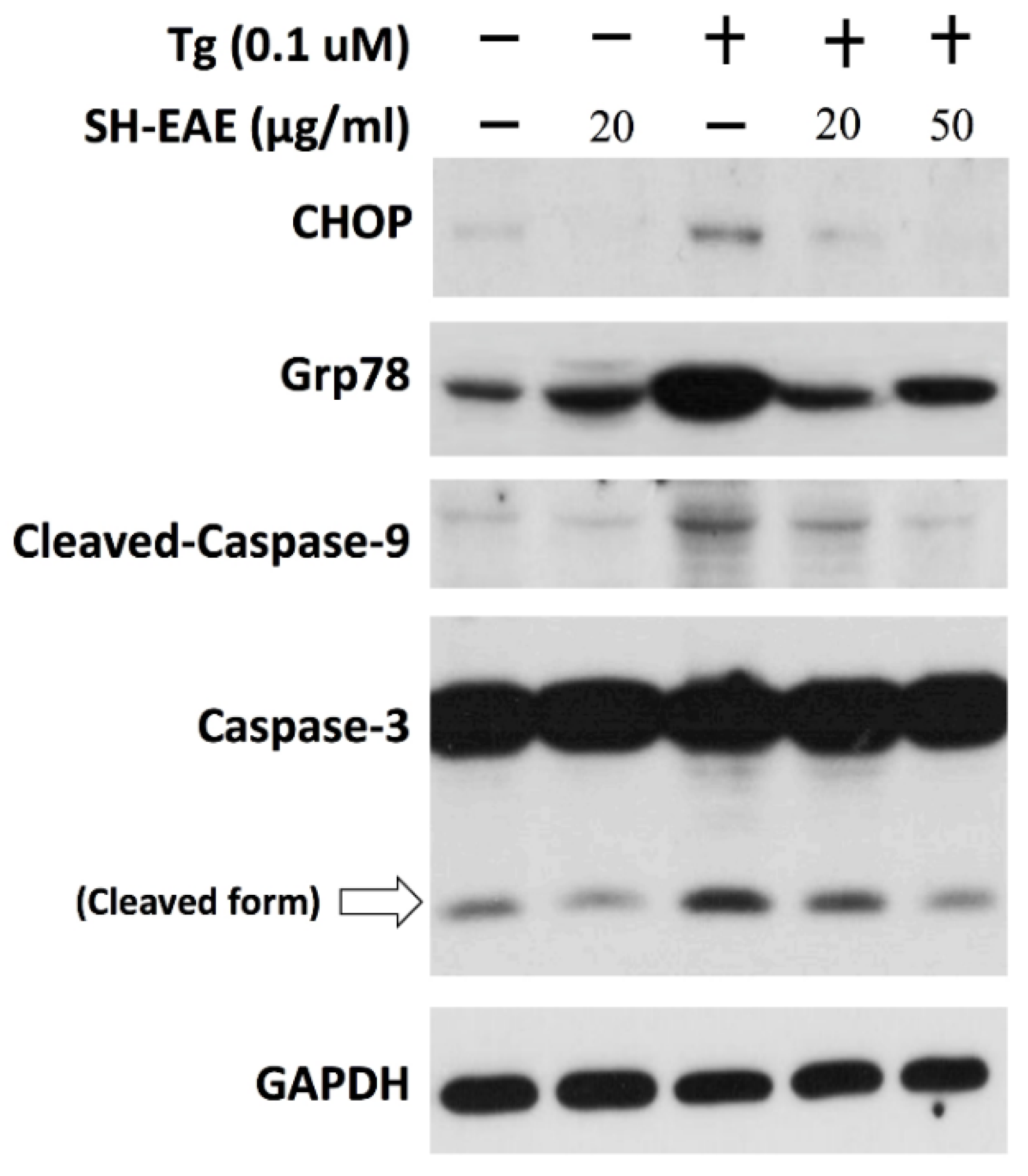
2.8. SH-EAE Antagonizes the Induction of CHOP Expression by Tg
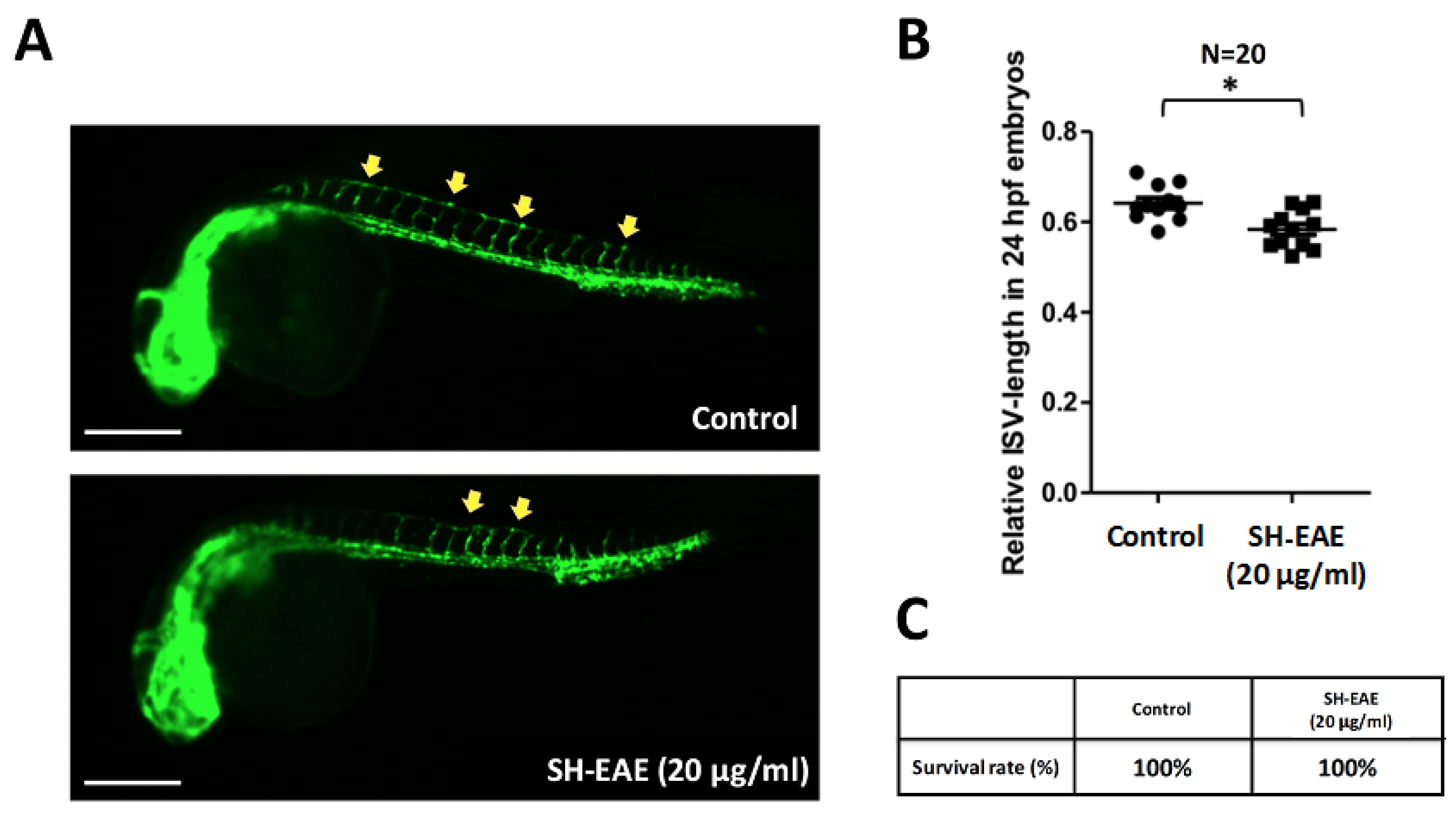
2.9. SH-EAE Attenuated the Angiogenesis in Zebrafish Model
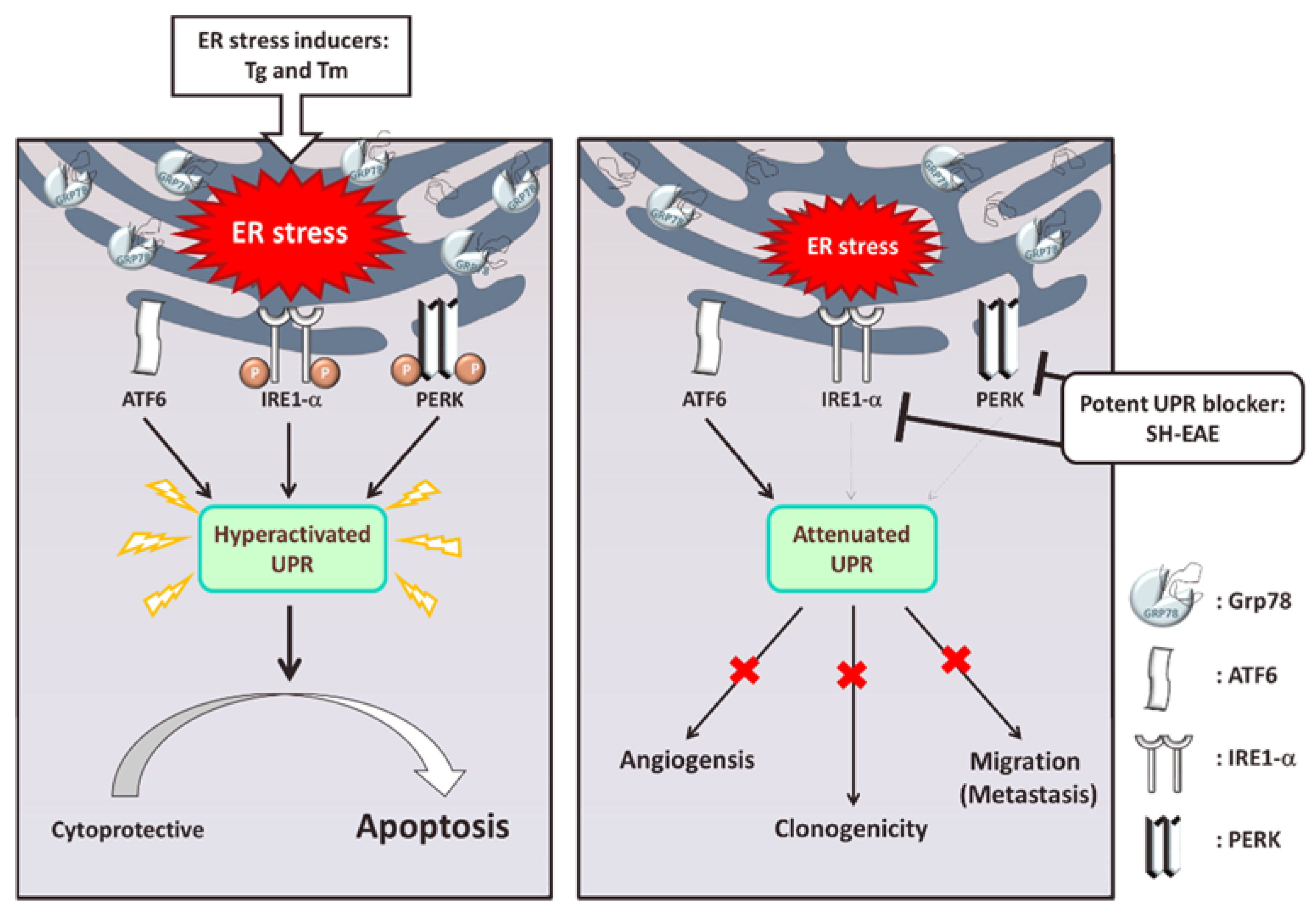
3. Discussion
4. Experimental Section
4.1. Reagents and Antibodies
4.2. Preparation of the Extract of Scindapsus cf. hederaceus
4.3. Cell Culture
4.4. Colony Formation Assay
4.5. Wound Healing Assay
4.6. Transwell Invasion Assay
4.7. Western Blotting Assay
4.8. Quantitative Polymerase Chain Reaction Analysis
4.9. Determination of ER Morphology
4.10. Apoptosis Assessment
4.11. Immunofluorescence Staining
4.12. In Vivo Assessment of Anti-Angiogenic Efficacy in Zebrafish
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Jorgensen, E.; Stinson, A.; Shan, L.; Yang, J.; Gietl, D.; Albino, A.P. Cigarette smoke induces endoplasmic reticulum stress and the unfolded protein response in normal and malignant human lung cells. BMC Cancer 2008, 8, 229. [Google Scholar] [CrossRef] [PubMed]
- Hazari, Y.M.; Bashir, A.; Haq, E.U.; Fazili, K.M. Emerging tale of UPR and cancer: An essentiality for malignancy. Tumour Biol. 2016, 37, 14381–14390. [Google Scholar] [CrossRef] [PubMed]
- Karagoz, G.E.; Acosta-Alvear, D.; Nguyen, H.T.; Lee, C.P.; Chu, F.; Walter, P. An unfolded protein-induced conformational switch activates mammalian ire1. eLife 2017, 6, e30700. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Jin, Z.; Chen, N.Z.; Lu, M.; Liu, C.B.; Hu, W.L.; Zheng, C.G. Activation of IRE1α-XBP1 pathway induces cell proliferation and invasion in colorectal carcinoma. Biochem. Biophys. Res. Commun. 2016, 470, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Woyach, J.A.; Johnson, A.J.; Byrd, J.C. The B-cell receptor signaling pathway as a therapeutic target in CLL. Blood 2012, 120, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Vandewynckel, Y.P.; Laukens, D.; Geerts, A.; Bogaerts, E.; Paridaens, A.; Verhelst, X.; Janssens, S.; Heindryckx, F.; van Vlierberghe, H. The paradox of the unfolded protein response in cancer. Anticancer Res. 2013, 33, 4683–4694. [Google Scholar] [PubMed]
- Ri, M.; Tashiro, E.; Oikawa, D.; Shinjo, S.; Tokuda, M.; Yokouchi, Y.; Narita, T.; Masaki, A.; Ito, A.; Ding, J.; et al. Identification of toyocamycin, an agent cytotoxic for multiple myeloma cells, as a potent inhibitor of ER stress-induced XBP1 mRNA splicing. Blood Cancer J. 2012, 2, e79. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhang, K.; Li, Z. Unfolded protein response in cancer: The physician’s perspective. J. Hematol. Oncol. 2011, 4, 8. [Google Scholar] [CrossRef] [PubMed]
- Bi, M.; Naczki, C.; Koritzinsky, M.; Fels, D.; Blais, J.; Hu, N.; Harding, H.; Novoa, I.; Varia, M.; Raleigh, J.; et al. ER stress-regulated translation increases tolerance to extreme hypoxia and promotes tumor growth. EMBO J. 2005, 24, 3470–3481. [Google Scholar] [CrossRef] [PubMed]
- Blais, J.D.; Addison, C.L.; Edge, R.; Falls, T.; Zhao, H.; Wary, K.; Koumenis, C.; Harding, H.P.; Ron, D.; Holcik, M.; et al. Perk-dependent translational regulation promotes tumor cell adaptation and angiogenesis in response to hypoxic stress. Mol. Cell. Biol. 2006, 26, 9517–9532. [Google Scholar] [CrossRef] [PubMed]
- Ron, D.; Walter, P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat. Rev. Mol. Cell Biol. 2007, 8, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Haze, K.; Yoshida, H.; Yanagi, H.; Yura, T.; Mori, K. Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol. Biol. Cell 1999, 10, 3787–3799. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Rawson, R.B.; Komuro, R.; Chen, X.; Dave, U.P.; Prywes, R.; Brown, M.S.; Goldstein, J.L. ER stress induces cleavage of membrane-bound ATF6 by the same proteases that process SREBPs. Mol. Cell 2000, 6, 1355–1364. [Google Scholar] [CrossRef]
- Harding, H.P.; Zhang, Y.; Ron, D. Protein translation and folding are coupled by an endoplasmic-reticulum-resident kinase. Nature 1999, 397, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Calfon, M.; Zeng, H.; Urano, F.; Till, J.H.; Hubbard, S.R.; Harding, H.P.; Clark, S.G.; Ron, D. IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 2002, 415, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Chevet, E.; Hetz, C.; Samali, A. Endoplasmic reticulum stress-activated cell reprogramming in oncogenesis. Cancer Discov. 2015, 5, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Oyadomari, S.; Mori, M. Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ. 2004, 11, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Bailly-Maitre, B.; Reed, J.C. Endoplasmic reticulum stress: Cell life and death decisions. J. Clin. Investig. 2005, 115, 2656–2664. [Google Scholar] [CrossRef] [PubMed]
- Foufelle, F.; Fromenty, B. Role of endoplasmic reticulum stress in drug-induced toxicity. Pharmacol. Res. Perspect. 2016, 4, e00211. [Google Scholar] [CrossRef] [PubMed]
- Bieberich, E. Synthesis, processing, and function of N-glycans in N-glycoproteins. Adv. Neurobiol. 2014, 9, 47–70. [Google Scholar] [PubMed]
- Mekahli, D.; Bultynck, G.; Parys, J.B.; De Smedt, H.; Missiaen, L. Endoplasmic-reticulum calcium depletion and disease. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Gaut, J.R.; Hendershot, L.M. The modification and assembly of proteins in the endoplasmic reticulum. Curr. Opin. Cell Biol. 1993, 5, 589–595. [Google Scholar] [CrossRef]
- Shivhare, S.C.; Patidar, A.O.; Malviya, K.G.; Shivhare-Malviya, K.K. Antioxidant and anticancer evaluation of Scindapsus officinalis (Roxb.) schott fruits. Ayu 2011, 32, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Song, X.; Wang, D.; Wang, X.; Wang, X. Five new chromone glycosides from Scindapsus officinalis (Roxb.) schott. Fitoterapia 2017, 122, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wey, S.; Zhang, Y.; Ye, R.; Lee, A.S. Role of the unfolded protein response regulator GRP78/BiP in development, cancer, and neurological disorders. Antioxid. Redox Signal. 2009, 11, 2307–2316. [Google Scholar] [CrossRef] [PubMed]
- Ram, B.M.; Ramakrishna, G. Endoplasmic reticulum vacuolation and unfolded protein response leading to paraptosis like cell death in cyclosporine a treated cancer cervix cells is mediated by cyclophilin b inhibition. Biochim. Biophys. Acta 2014, 1843, 2497–2512. [Google Scholar] [CrossRef] [PubMed]
- Turinetto, V.; Giachino, C. Multiple facets of histone variant H2AX: A DNA double-strand-break marker with several biological functions. Nucleic Acids Res. 2015, 43, 2489–2498. [Google Scholar] [CrossRef] [PubMed]
- Atkins, C.; Liu, Q.; Minthorn, E.; Zhang, S.Y.; Figueroa, D.J.; Moss, K.; Stanley, T.B.; Sanders, B.; Goetz, A.; Gaul, N.; et al. Characterization of a novel perk kinase inhibitor with antitumor and antiangiogenic activity. Cancer Res. 2013, 73, 1993–2002. [Google Scholar] [CrossRef] [PubMed]
- Drogat, B.; Auguste, P.; Nguyen, D.T.; Bouchecareilh, M.; Pineau, R.; Nalbantoglu, J.; Kaufman, R.J.; Chevet, E.; Bikfalvi, A.; Moenner, M. IRE1 signaling is essential for ischemia-induced vascular endothelial growth factor-A expression and contributes to angiogenesis and tumor growth in vivo. Cancer Res. 2007, 67, 6700–6707. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, R.; Lipson, K.L.; Sargent, K.E.; Mercurio, A.M.; Hunt, J.S.; Ron, D.; Urano, F. Transcriptional regulation of VEGF-A by the unfolded protein response pathway. PLoS ONE 2010, 5, e9575. [Google Scholar] [CrossRef] [PubMed]
- Gifford, J.B.; Hill, R. GRP78 influences chemoresistance and prognosis in cancer. Curr. Drug Targets 2017, 19, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Rozpedek, W.; Pytel, D.; Mucha, B.; Leszczynska, H.; Diehl, J.A.; Majsterek, I. The role of the PERK/EIF2α/ATF4/CHOP signaling pathway in tumor progression during endoplasmic reticulum stress. Curr. Mol. Med. 2016, 16, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Kaul, A.; Maltese, W.A. Killing of cancer cells by the photoactivatable protein kinase c inhibitor, calphostin c, involves induction of endoplasmic reticulum stress. Neoplasia 2009, 11, 823–834. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.B.; Feng, L.X.; Yue, Q.X.; Wu, W.Y.; Guan, S.H.; Jiang, B.H.; Yang, M.; Liu, X.; Guo, D.A. Paraptosis accompanied by autophagy and apoptosis was induced by celastrol, a natural compound with influence on proteasome, ER stress and Hsp90. J. Cell. Physiol. 2012, 227, 2196–2206. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.H.; Shu, K.H.; Chang, H.R.; Chou, M.C. Cyclosporine-induced tubular vacuolization: The role of Bip/Grp78. Nephron Exp. Nephrol. 2012, 122, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mimnaugh, E.G.; Xu, W.; Vos, M.; Yuan, X.; Neckers, L. Endoplasmic reticulum vacuolization and valosin-containing protein relocalization result from simultaneous hsp90 inhibition by geldanamycin and proteasome inhibition by velcade. Mol. Cancer Res. 2006, 4, 667–681. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Chien, M.H.; Chow, J.M.; Chang, J.L.; Wen, Y.C.; Lin, Y.W.; Cheng, C.W.; Lai, G.M.; Hsiao, M.; Lee, L.M. Nonautophagic cytoplasmic vacuolation death induction in human PC-3M prostate cancer by curcumin through reactive oxygen species -mediated endoplasmic reticulum stress. Sci. Rep. 2015, 5, 10420. [Google Scholar] [CrossRef] [PubMed]
- Kadioglu, O.; Cao, J.; Saeed, M.E.; Greten, H.J.; Efferth, T. Targeting epidermal growth factor receptors and downstream signaling pathways in cancer by phytochemicals. Target Oncol. 2015, 10, 337–353. [Google Scholar] [CrossRef] [PubMed]
- Zahorowska, B.; Crowe, P.J.; Yang, J.L. Combined therapies for cancer: A review of EGFR-targeted monotherapy and combination treatment with other drugs. J. Cancer Res. Clin. Oncol. 2009, 135, 1137–1148. [Google Scholar] [CrossRef] [PubMed]
- Cai, B.; Tomida, A.; Mikami, K.; Nagata, K.; Tsuruo, T. Downregulation of epidermal growth factor receptor-signaling pathway by binding of GRP78/BiP to the receptor under glucose-starved stress conditions. J. Cell. Physiol. 1998, 177, 282–288. [Google Scholar] [CrossRef]
- Yu, L.; Andruska, N.; Zheng, X.; Shapiro, D.J. Anticipatory activation of the unfolded protein response by epidermal growth factor is required for immediate early gene expression and cell proliferation. Mol. Cell. Endocrinol. 2016, 422, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Paridaens, A.; Laukens, D.; Vandewynckel, Y.P.; Coulon, S.; Van Vlierberghe, H.; Geerts, A.; Colle, I. Endoplasmic reticulum stress and angiogenesis: Is there an interaction between them? Liver Int. 2014, 34, e10–e18. [Google Scholar] [CrossRef] [PubMed]
- Urra, H.; Hetz, C. A novel ER stress-independent function of the UPR in angiogenesis. Mol. Cell 2014, 54, 542–544. [Google Scholar] [CrossRef] [PubMed]
- Philippe, C.; Dubrac, A.; Quelen, C.; Desquesnes, A.; van den Berghe, L.; Segura, C.; Filleron, T.; Pyronnet, S.; Prats, H.; Brousset, P.; et al. PERK mediates the IRES-dependent translational activation of mRNAs encoding angiogenic growth factors after ischemic stress. Sci. Signal. 2016, 9, ra44. [Google Scholar] [CrossRef] [PubMed]
- Karali, E.; Bellou, S.; Stellas, D.; Klinakis, A.; Murphy, C.; Fotsis, T. VEGF signals through ATF6 and PERK to promote endothelial cell survival and angiogenesis in the absence of ER stress. Mol. Cell 2014, 54, 559–572. [Google Scholar] [CrossRef] [PubMed]
- Duan, Q.; Ni, L.; Wang, P.; Chen, C.; Yang, L.; Ma, B.; Gong, W.; Cai, Z.; Zou, M.H.; Wang, D.W. Deregulation of XBP1 expression contributes to myocardial vascular endothelial growth factor-A expression and angiogenesis during cardiac hypertrophy in vivo. Aging Cell 2016, 15, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Harding, H.P.; Zhang, Y.; Bertolotti, A.; Zeng, H.; Ron, D. PERK is essential for translational regulation and cell survival during the unfolded protein response. Mol. Cell 2000, 5, 897–904. [Google Scholar] [CrossRef]
- Fan, C.F.; Miao, Y.; Lin, X.Y.; Zhang, D.; Wang, E.H. Expression of a phosphorylated form of ATF4 in lung and non-small cell lung cancer tissues. Tumour Biol. 2014, 35, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.K.; Chae, S.W.; Kim, H.R.; Chae, H.J. Endoplasmic reticulum stress and cancer. J. Cancer Prev. 2014, 19, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Cubillos-Ruiz, J.R.; Bettigole, S.E.; Glimcher, L.H. Tumorigenic and immunosuppressive effects of endoplasmic reticulum stress in cancer. Cell 2017, 168, 692–706. [Google Scholar] [CrossRef] [PubMed]

| Genes | Primers |
|---|---|
| GRP78 | F′: 5′-CAACCCCGAGAACACGGTC-3′ |
| R′: 5′-CTGCACAGACGGGTCATTC-3′ | |
| ATF6 | F′: 5′-ATGTCTCCCCTTTCCTTATATGGT-3′ |
| R′: 5′-AAGGCTTGGGCTGAATTGAA-3′ | |
| PERK | F′: 5′-TGTCGCCAATGGGATAGTGACGAA-3′ |
| R′: 5′-AATCCGGCTCTCGTTTCCATGTCT-3′ | |
| IRE1α | F′: 5′-GGGAAATACTCTACCAGCCT-3′ |
| R′: 5′-GAAATCTCTCCAGCATCTTG-3′ | |
| GAPDH | F′: 5′-GTCTTCACCACCATGGAGAA-3′ |
| R′: 5′-ATGGCATGGACTGTGGTCAT-3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chou, C.-K.; Liu, W.; Hong, Y.-J.; Dahms, H.-U.; Chiu, C.-H.; Chang, W.-T.; Chien, C.-M.; Yen, C.-H.; Cheng, Y.-B.; Chiu, C.-C. Ethyl Acetate Extract of Scindapsus cf. hederaceus Exerts the Inhibitory Bioactivity on Human Non-Small Cell Lung Cancer Cells through Modulating ER Stress. Int. J. Mol. Sci. 2018, 19, 1832. https://doi.org/10.3390/ijms19071832
Chou C-K, Liu W, Hong Y-J, Dahms H-U, Chiu C-H, Chang W-T, Chien C-M, Yen C-H, Cheng Y-B, Chiu C-C. Ethyl Acetate Extract of Scindapsus cf. hederaceus Exerts the Inhibitory Bioactivity on Human Non-Small Cell Lung Cancer Cells through Modulating ER Stress. International Journal of Molecular Sciences. 2018; 19(7):1832. https://doi.org/10.3390/ijms19071832
Chicago/Turabian StyleChou, Chon-Kit, Wangta Liu, Yu-Jie Hong, Hans-Uwe Dahms, Chen-Hao Chiu, Wen-Tsan Chang, Ching-Ming Chien, Chia-Hung Yen, Yuan-Bin Cheng, and Chien-Chih Chiu. 2018. "Ethyl Acetate Extract of Scindapsus cf. hederaceus Exerts the Inhibitory Bioactivity on Human Non-Small Cell Lung Cancer Cells through Modulating ER Stress" International Journal of Molecular Sciences 19, no. 7: 1832. https://doi.org/10.3390/ijms19071832
APA StyleChou, C.-K., Liu, W., Hong, Y.-J., Dahms, H.-U., Chiu, C.-H., Chang, W.-T., Chien, C.-M., Yen, C.-H., Cheng, Y.-B., & Chiu, C.-C. (2018). Ethyl Acetate Extract of Scindapsus cf. hederaceus Exerts the Inhibitory Bioactivity on Human Non-Small Cell Lung Cancer Cells through Modulating ER Stress. International Journal of Molecular Sciences, 19(7), 1832. https://doi.org/10.3390/ijms19071832









