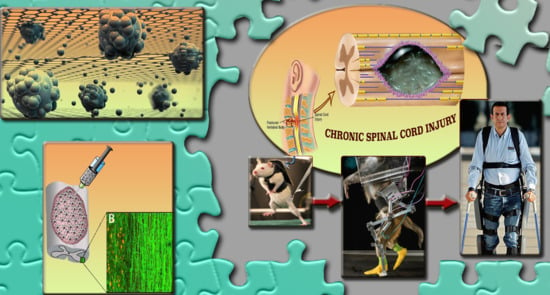
Translational Regenerative Therapies for Chronic Spinal Cord Injury
Abstract
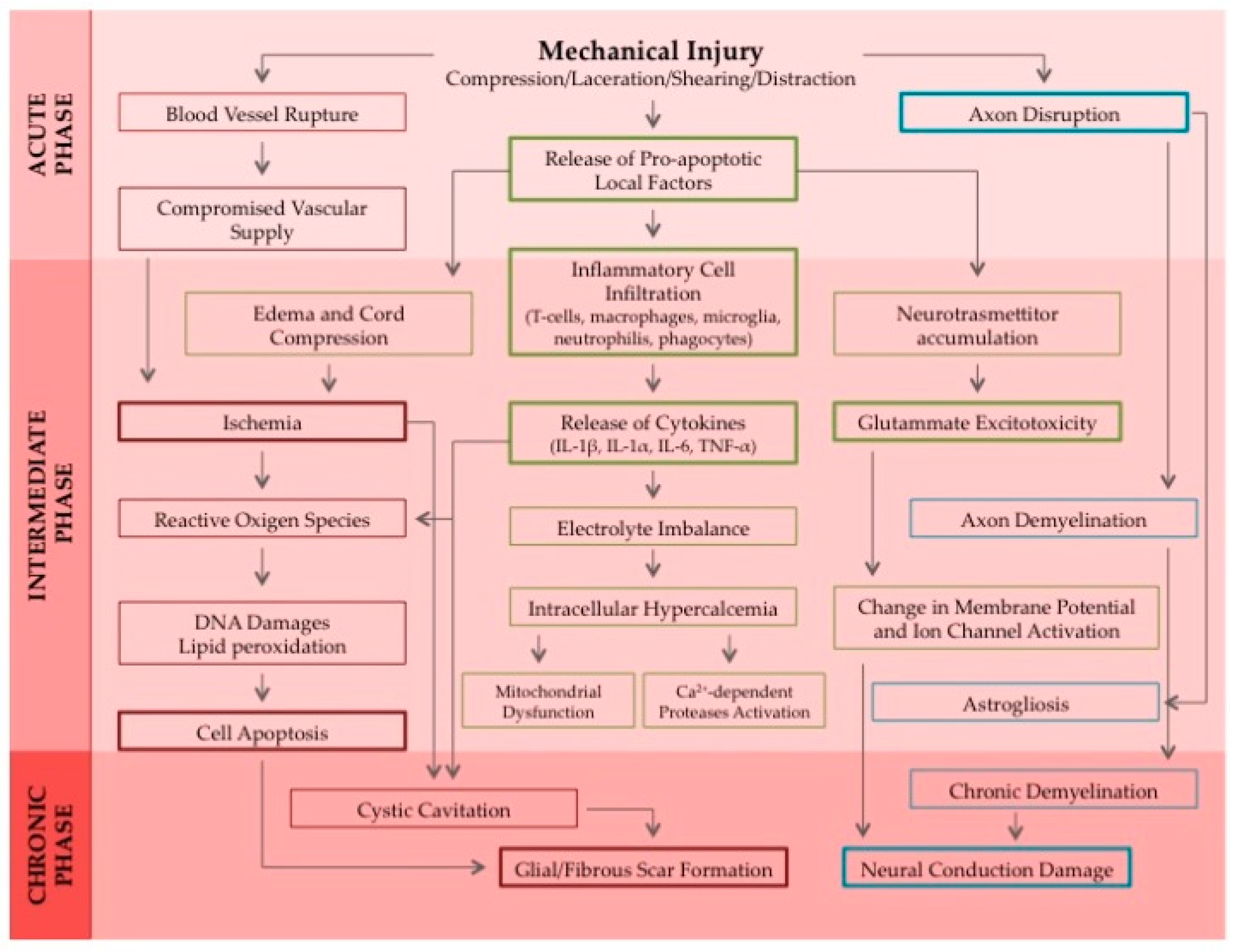
1. Introduction
2. Cell Therapies
2.1. The “Pathway Study” of Stemcell Inc. (Phase I/II Clinical Trials)
2.2. Phase I Clinical Trial of NeuralStem Inc.
2.3. The Chronic SCI Stem Cell Study of InVivo Therapeutics
2.4. “Walk Again Project”
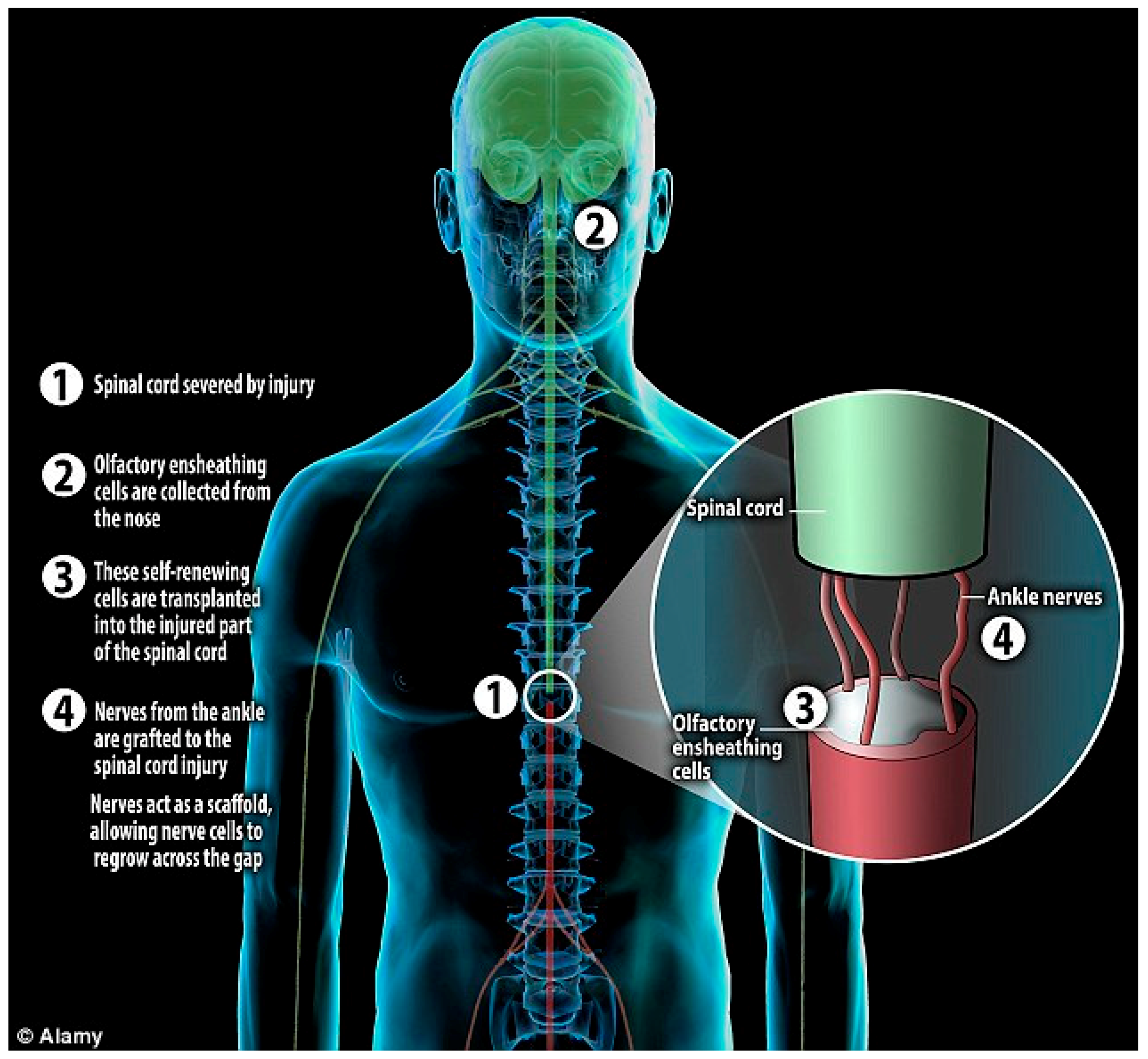
2.5. “Miami Project” Phase I Clinical Trial
2.6. Umbilical Cord Blood & Lithium ChinaSCINet Phase II Clinical Trial
2.7. The Puerta de Hierro Phase I/II Clinical Trial
2.8. “Neurocell” Pre-Clinical Study of Neuroplast (Phase I Clinical Trial in Preparation)
2.9. Less Strictly Regulated Clinical Trials
3. Molecular Therapies
3.1. Nogo Trap of ReNetX Bio (Formerly Known as Axerion Therapeutics)
3.2. CHASE-IT Preclinical Initiative of the International Spinal Research Trust (ISRT)
4. Selected Biomaterials That Hold Promise for Future Clinical Trials on Chronic Sci
4.1. Fibrin-Based Scaffolds and Hydrogels Have Shown Impressive Results in Terms of Supporting CNS Regeneration in SCI Lesions in the Right Settings
4.2. Collagen/Heparin Sulfate Scaffolds Fabricated by a 3D Bioprinter
4.3. Peripheral Nerve Grafts Combined with Chitosan-Laminin Scaffold
5. Conclusions and Future Perspectives
Author Contributions
Acknowledgments
Conflicts of Interest
References
- National Spinal Cord Injury Statistical Center. Spinal Cord Injury (SCI) Facts and Figures at a Glance; National Spinal Cord Injury Statistical Center: Birmingham, AL, USA, 2017. [Google Scholar]
- Kadoya, K.; Tsukada, S.; Lu, P.; Coppola, G.; Geschwind, D.; Filbin, M.; Blesch, A.; Tuszynski, M.H. Combined Intrinsic and Extrinsic Neuronal Mechanisms Facilitate Bridging Axonal Regeneration One Year After Spinal Cord Injury. Neuron 2009, 64, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Gelain, F.; Panseri, S.; Antonini, S.; Cunha, C.; Donega, M.; Lowery, J.; Taraballi, F.; Cerri, G.; Montagna, M.; Baldissera, F.; et al. Transplantation of Nanostructured Composite Scaffolds Results in the Regeneration of Chronically Injured Spinal Cords. ACS Nano 2011, 5, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Faccendini, A.; Vigani, B.; Rossi, S.; Sandri, G.; Bonferoni, M.C.; Caramella, C.M.; Ferrari, F. Nanofiber Scaffolds as Drug Delivery Systems to Bridge Spinal Cord Injury. Pharmaceuticals 2017, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- StemCells Inc. Pathway Study; StemCells Inc.: Newark, CA, USA, 2015. [Google Scholar]
- StemCells Inc. Phase II Trial in Cervical Spinal Cord Injury (SCI); StemCells Inc.: Newark, CA, USA, 2015. [Google Scholar]
- NeuralStem Inc. Neuralstem Reports Third Quarter 2015 Financial Results—Nov 9, 2015; NeuralStem Inc.: Rockville, MD, USA, 2015. [Google Scholar]
- Yan, J.; Xu, L.; Welsh, A.M.; Hatfield, G.; Hazel, T.; Johe, K.; Koliatsos, V.E. Extensive neuronal differentiation of human neural stem cell grafts in adult rat spinal cord. PLoS Med. 2007, 4, e39. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yan, J.; Chen, D.; Welsh, A.M.; Hazel, T.; Johe, K.; Hatfield, G.; Koliatsos, V.E. Human neural stem cell grafts ameliorate motor neuron disease in SOD-1 transgenic rats. Transplantation 2006, 82, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, E.S.; Brock, J.H.; Lu, P.; Kumamaru, H.; Salegio, E.A.; Kadoya, K.; Weber, J.L.; Liang, J.J.; Moseanko, R.; Hawbecker, S.; et al. Restorative effects of human neural stem cell grafts on the primate spinal cord. Nat. Med. 2018, 24, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Theodore, N.; Hlubek, R.; Danielson, J.; Neff, K.; Vaickus, L.; Ulich, T.R.; Ropper, A.E. First Human Implantation of a Bioresorbable Polymer Scaffold for Acute Traumatic Spinal Cord Injury: A Clinical Pilot Study for Safety and Feasibility. Neurosurgery 2016, 79, E305–E312. [Google Scholar] [CrossRef] [PubMed]
- Tabakow, P.; Raisman, G.; Fortuna, W.; Czyz, M.; Huber, J.; Li, D.; Szewczyk, P.; Okurowski, S.; Miedzybrodzki, R.; Czapiga, B.; et al. Functional regeneration of supraspinal connections in a patient with transected spinal cord following transplantation of bulbar olfactory ensheathing cells with peripheral nerve bridging. Cell Transpl. 2014, 23, 1631–1655. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chen, L.; Wang, H.; Xiu, B.; Li, B.; Wang, R.; Zhang, J.; Zhang, F.; Gu, Z.; Li, Y.; et al. Influence of patients’ age on functional recovery after transplantation of olfactory ensheathing cells into injured spinal cord injury. Chin. Med. J. 2003, 116, 1488–1491. [Google Scholar] [PubMed]
- Féron, F.; Perry, C.; Cochrane, J.; Licina, P.; Nowitzke, A.; Urquhart, S.; Geraghty, T.; Mackay-Sim, A. Autologous olfactory ensheathing cell transplantation in human spinal cord injury. Brain 2005, 128, 2951–2960. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.; Pratas-Vital, J.; Escada, P.; Hasse-Ferreira, A.; Capucho, C.; Peduzzi, J.D. Olfactory Mucosa Autografts in Human Spinal Cord Injury: A Pilot Clinical Study. J. Spinal Cord Med. 2006, 29, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Adnan, H.; Xu, B.; Wang, J.; Wang, C.; Li, F.; Tang, K. Effects of transplantation of olfactory ensheathing cells in chronic spinal cord injury: A systematic review and meta-analysis. Eur. Spine J. 2015, 24, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.; Gladwin, K. Olfactory Ensheathing Cells: Part II—Source of Cells and Application to Patients. World Neurosurg. 2015, 83, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.-J.; Thompson, W.J. Schwann cell processes guide regeneration of peripheral axons. Neuron 1995, 14, 125–132. [Google Scholar] [CrossRef]
- Bruce, J.H.; Norenberg, M.D.; Kraydieh, S.; Puckett, W.; Marcillo, A.; Dietrich, D. Schwannosis: Role of gliosis and proteoglycan in human spinal cord injury. J. Neurotrauma 2000, 17, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Guest, J.D.; Hiester, E.D.; Bunge, R.P. Demyelination and Schwann cell responses adjacent to injury epicenter cavities following chronic human spinal cord injury. Exp. Neurol. 2005, 192, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Bunge, M.B.; Wood, P.M. Realizing the maximum potential of Schwann cells to promote recovery from spinal cord injury. Handb. Clin. Neurol. 2012, 109, 523–540. [Google Scholar] [CrossRef] [PubMed]
- Wiliams, R.R.; Bunge, M.B. Schwann cell transplantation: A repair strategy for spinal cord injury? Prog. Brain Res. 2012, 201, 295–312. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Poon, W.; Liu, Y.; Leung, G.K.-K.; Wong, Y.; Feng, Y.; Ng, S.C.P.; Tsang, K.S.; Sun, D.T.F.; Yeung, D.K.; et al. Phase I–II Clinical Trial Assessing Safety and Efficacy of Umbilical Cord Blood Mononuclear Cell Transplant Therapy of Chronic Complete Spinal Cord Injury. Cell Transpl. 2016, 25, 1925–1943. [Google Scholar] [CrossRef] [PubMed]
- Dasari, V.R.; Veeravalli, K.K.; Tsung, A.J.; Gondi, C.S.; Gujrati, M.; Dinh, D.H.; Rao, J.S. Neuronal Apoptosis Is Inhibited by Cord Blood Stem Cells after Spinal Cord Injury. J. Neurotrauma 2009, 26, 2057–2069. [Google Scholar] [CrossRef] [PubMed]
- Veeravalli, K.K.; Dasari, V.R.; Tsung, A.J.; Dinh, D.H.; Gujrati, M.; Fassett, D.; Rao, J.S. Human umbilical cord blood stem cells upregulate matrix metalloproteinase-2 in rats after spinal cord injury. Neurobiol. Dis. 2009, 36, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Veeravalli, K.K.; Dasari, V.R.; Tsung, A.J.; Dinh, D.H.; Gujrati, M.; Fassett, D.; Rao, J.S. Stem Cells Downregulate the Elevated Levels of Tissue Plasminogen Activator in Rats After Spinal Cord Injury. Neurochem. Res. 2009, 34, 1183–1194. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.-H.; Chen, S.-H.; Chio, C.-C.; Lin, M.-T. Human Umbilical Cord Blood-derived CD34+ cells may attenuate spinal cord injury by stimulating vascular endothelial and neurotrophic factors. Shock 2008, 29, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Chua, S.J.; Bielecki, R.; Yamanaka, N.; Fehlings, M.G.; Rogers, I.M.; Casper, R.F. The Effect of Umbilical Cord Blood Cells on Outcomes After Experimental Traumatic Spinal Cord Injury. Spine 2010, 35, 1520–1526. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Chung, W.; Lee, J.-H.; Chung, D.-J.; Yang, W.-J.; Lee, A.-J.; Choi, C.-B.; Chang, H.-S.; Kim, D.-H.; Suh, H.J.; et al. Expression of neurotrophic factors in injured spinal cord after transplantation of human-umbilical cord blood stem cells in rats. J. Vet. Sci. 2016, 17, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Dasari, V.R.; Spomar, D.G.; Gondi, C.S.; Sloffer, C.A.; Saving, K.L.; Gujrati, M.; Rao, J.S.; Dinh, D.H. Axonal Remyelination by Cord Blood Stem Cells after Spinal Cord Injury. J. Neurotrauma 2007, 24, 391–410. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.-R.; Yang, M.S.; Yim, S.H.; Park, J.H.; Lee, J.E.; Eom, Y.; Jang, I.K.; Kim, H.E.; Park, J.S.; Kim, H.O.; et al. Neurally induced umbilical cord blood cells modestly repair injured spinal cords. NeuroReport 2008, 19, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.-H.; Byeon, Y.-E.; Park, S.-S.; Kang, B.-J.; Seo, M.-S.; Park, S.-B.; Kim, W.H.; Kang, K.-S.; Kweon, O.-K. Immunohistomorphometric Analysis of Transplanted Umbilical Cord Blood-Derived Mesenchymal Stem Cells and The Resulting Anti-Inflammatory Effects on Nerve Regeneration of Injured Canine Spinal Cord. Tissue Eng. Regen. Med. 2011, 8, 173–182. [Google Scholar]
- Ning, G.; Tang, L.; Wu, Q.; Li, Y.; Li, Y.; Zhang, C.; Feng, S. Human umbilical cord blood stem cells for spinal cord injury: Early transplantation results in better local angiogenesis. Regen. Med. 2013, 8, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Vaquero, J.; Zurita, M.; Rico, M.A.; Bonilla, C.; Aguayo, C.; Montilla, J.; Bustamante, S.; Carballido, J.; Marin, E.; Martinez, F.; et al. An approach to personalized cell therapy in chronic complete paraplegia: The Puerta de Hierro phase I/II clinical trial. Cytotherapy 2016, 18, 1025–1036. [Google Scholar] [CrossRef] [PubMed]
- Deans, R.J.; Moseley, A.B. Mesenchymal stem cells. Exp. Hematol. 2000, 28, 875–884. [Google Scholar] [CrossRef]
- Kopen, G.C.; Prockop, D.J.; Phinney, D.G. Marrow stromal cells migrate throughout forebrain and cerebellum, and they differentiate into astrocytes after injection into neonatal mouse brains. Proc. Natl. Acad. Sci. USA 1999, 96, 10711–10716. [Google Scholar] [CrossRef] [PubMed]
- Hofer, H.R.; Tuan, R.S. Secreted trophic factors of mesenchymal stem cells support neurovascular and musculoskeletal therapies. Stem Cell Res. Ther. 2016, 7, 131. [Google Scholar] [CrossRef] [PubMed]
- Cizkova, D.; Cubinkova, V.; Smolek, T.; Murgoci, A.-N.; Danko, J.; Vdoviakova, K.; Humenik, F.; Cizek, M.; Quanico, J.; Fournier, I.; et al. Localized Intrathecal Delivery of Mesenchymal Stromal Cells Conditioned Medium Improves Functional Recovery in a Rat Model of Spinal Cord Injury. Int. J. Mol. Sci. 2018, 19, 870. [Google Scholar] [CrossRef] [PubMed]
- Bansal, H.; Verma, P.; Agrawal, A.; Leon, J.; Sundell, I.B.; Koka, P.S. Autologous Bone Marrow-Derived Stem Cells in Spinal Cord Injury. J. Stem Cells 2016, 11, 51–61. [Google Scholar] [PubMed]
- Amr, S.M.; Gouda, A.; Koptan, W.T.; Galal, A.A.; Abdel-Fattah, D.S.; Rashed, L.A.; Atta, H.M.; Abdel-Aziz, M.T. Bridging defects in chronic spinal cord injury using peripheral nerve grafts combined with a chitosan-laminin scaffold and enhancing regeneration through them by co-transplantation with bone-marrow-derived mesenchymal stem cells: Case series of 14 patients. J. Spinal Cord Med. 2014, 37, 54–71. [Google Scholar] [CrossRef] [PubMed]
- Frolov, A.A.; Bryukhovetskiy, A.S. Effects of hematopoietic autologous stem cell transplantation to the chronically injured human spinal cord evaluated by motor and somatosensory evoked potentials methods. Cell Transpl. 2012, 21 (Suppl. 1), S49–S55. [Google Scholar] [CrossRef] [PubMed]
- El-Kheir, W.A.; Gabr, H.; Awad, M.R.; Ghannam, O.; Barakat, Y.; Farghali, H.A.M.A.; El Maadawi, Z.M.; Ewes, I.; Sabaawy, H.E. Autologous bone marrow-derived cell therapy combined with physical therapy induces functional improvement in chronic spinal cord injury patients. Cell Transpl. 2014, 23, 729–745. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.W.; Tam, S.; So, K.F.; Chen, J.Y.H.; Cheng, W.S.; Luk, K.D.K.; Tang, S.W.; Young, W. A three-month, open-label, single-arm trial evaluating the safety and pharmacokinetics of oral lithium in patients with chronic spinal cord injury. Spinal Cord 2011, 49, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Cristante, A.F.; Barros-Filho, T.E.P.; Tatsui, N.; Mendrone, A.; Caldas, J.G.; Camargo, A.; Alexandre, A.; Teixeira, W.G.J.; Oliveira, R.P.; Marcon, R.M. Stem cells in the treatment of chronic spinal cord injury: Evaluation of somatosensitive evoked potentials in 39 patients. Spinal Cord 2009, 47, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Moviglia, G.A.; Fernandez Viña, R.; Brizuela, J.A.; Saslavsky, J.; Vrsalovic, F.; Varela, G.; Bastos, F.; Farina, P.; Etchegaray, G.; Barbieri, M.; et al. Combined protocol of cell therapy for chronic spinal cord injury. Report on the electrical and functional recovery of two patients. Cytotherapy 2006, 8, 202–209. [Google Scholar] [CrossRef] [PubMed]
- ReNetX. ReNetX Bio Launched to Advance Innovative Neuro-Regenerative Technology Developed at Yale University. Available online: http://globenewswire.com/news-release/2017/07/24/1056062/0/en/ReNetX-Bio-Launched-to-Advance-Innovative-Neuro-Regenerative-Technology-Developed-at-Yale-University.html (accessed on 20 April 2018).
- CHASE IT. Available online: https://www.spinal-research.org/chase-it (accessed on 20 April 2018).
- Tran, A.P.; Sundar, S.; Yu, M.; Lang, B.T.; Silver, J. Modulation of receptor protein tyrosine phosphatase sigma increases chondroitin sulfate proteoglycan degradation through Cathepsin B secretion to enhance axon outgrowth. J. Neurosci. 2018, 3214–3217. [Google Scholar] [CrossRef] [PubMed]
- Bartus, K.; James, N.D.; Didangelos, A.; Bosch, K.D.; Verhaagen, J.; Yáñez-Muñoz, R.J.; Rogers, J.H.; Schneider, B.L.; Muir, E.M.; Bradbury, E.J. Large-scale chondroitin sulfate proteoglycan digestion with chondroitinase gene therapy leads to reduced pathology and modulates macrophage phenotype following spinal cord contusion injury. J. Neurosci. 2014, 34, 4822–4836. [Google Scholar] [CrossRef] [PubMed]
- Muir, E.; Raza, M.; Ellis, C.; Burnside, E.; Love, F.; Heller, S.; Elliot, M.; Daniell, E.; Dasgupta, D.; Alves, N.; et al. Trafficking and processing of bacterial proteins by mammalian cells: Insights from chondroitinase ABC. PLoS ONE 2017, 12, e0186759. [Google Scholar] [CrossRef] [PubMed]
- James, N.D.; Shea, J.; Muir, E.M.; Verhaagen, J.; Schneider, B.L.; Bradbury, E.J. Chondroitinase gene therapy improves upper limb function following cervical contusion injury. Exp. Neurol. 2015, 271, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.M.; Carvalho, L.; Silva-Correia, J.; Vieira, S.; Majchrzak, M.; Lukomska, B.; Stanaszek, L.; Strymecka, P.; Malysz-Cymborska, I.; Golubczyk, D.; et al. Hydrogel-based scaffolds to support intrathecal stem cell transplantation as a gateway to the spinal cord: Clinical needs, biomaterials, and imaging technologies. NPJ Regen. Med. 2018, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Führmann, T.; Tam, R.Y.; Ballarin, B.; Coles, B.; Elliott Donaghue, I.; van der Kooy, D.; Nagy, A.; Tator, C.H.; Morshead, C.M.; Shoichet, M.S. Injectable hydrogel promotes early survival of induced pluripotent stem cell-derived oligodendrocytes and attenuates longterm teratoma formation in a spinal cord injury model. Biomaterials 2016, 83, 23–36. [Google Scholar] [CrossRef] [PubMed]
- López-Dolado, E.; González-Mayorga, A.; Gutiérrez, M.C.; Serrano, M.C. Immunomodulatory and angiogenic responses induced by graphene oxide scaffolds in chronic spinal hemisected rats. Biomaterials 2016, 99, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Mattei, T.A. How graphene is expected to impact neurotherapeutics in the near future. Expert Rev. Neurother. 2014, 14, 845–847. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Bajo, A.; González-Mayorga, A.; López-Dolado, E.; Serrano, M.C. Graphene-Derived Materials Interfacing the Spinal Cord: Outstanding in Vitro and in Vivo Findings. Front. Syst. Neurosci. 2017, 11, 71. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Motamed, S.; Thouas, G.A.; Bernard, C.C.; Li, D.; Parkington, H.C.; Coleman, H.A.; Finkelstein, D.I.; Forsythe, J.S. Graphene Functionalized Scaffolds Reduce the Inflammatory Response and Supports Endogenous Neuroblast Migration when Implanted in the Adult Brain. PLoS ONE 2016, 11, e0151589. [Google Scholar] [CrossRef] [PubMed]
- González-Mayorga, A.; López-Dolado, E.; Gutiérrez, M.C.; Collazos-Castro, J.E.; Ferrer, M.L.; del Monte, F.; Serrano, M.C. Favorable Biological Responses of Neural Cells and Tissue Interacting with Graphene Oxide Microfibers. ACS Omega 2017, 2, 8253–8263. [Google Scholar] [CrossRef]
- Singh, Z. Applications and Toxicity of Graphene Family Nanomaterials and Their Composites. Available online: https://www.dovepress.com/applications-and-toxicity-of-graphene-family-nanomaterials-and-their-c-peer-reviewed-fulltext-article-NSA (accessed on 19 April 2018).
- Kim, C.-Y.; Sikkema, W.K.A.; Hwang, I.-K.; Oh, H.; Kim, U.J.; Lee, B.H.; Tour, J.M. Spinal cord fusion with PEG-GNRs (TexasPEG): Neurophysiological recovery in 24 hours in rats. Surg. Neurol. Int. 2016, 7, S632–S636. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, M.C.P.; Soares, E.S.; de Jesus, M.B.; Ceragioli, H.J.; Batista, Â.G.; Nyúl-Tóth, Á.; Molnár, J.; Wilhelm, I.; Maróstica, M.R.; Krizbai, I.; et al. PEGylation of Reduced Graphene Oxide Induces Toxicity in Cells of the Blood-Brain Barrier: An in Vitro and in Vivo Study. Mol. Pharm. 2016, 13, 3913–3924. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yin, J.; Peng, C.; Hu, W.; Zhu, Z.; Li, W.; Fan, C.; Huang, Q. Distribution and biocompatibility studies of graphene oxide in mice after intravenous administration. Carbon 2011, 49, 986–995. [Google Scholar] [CrossRef]
- Yang, K.; Gong, H.; Shi, X.; Wan, J.; Zhang, Y.; Liu, Z. In vivo biodistribution and toxicology of functionalized nano-graphene oxide in mice after oral and intraperitoneal administration. Biomaterials 2013, 34, 2787–2795. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Wang, Y.; Xu, X.; Zeng, C.; Hou, J.; Lin, M.; Xu, J.; Sun, F.; Huang, X.; Dai, L.; et al. Can Graphene Oxide Cause Damage to Eyesight? Chem. Res. Toxicol. 2012, 25, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Sahu, A.; Il Choi, W.; Tae, G. A stimuli-sensitive injectable graphene oxide composite hydrogel. Chem. Commun. 2012, 48, 5820–5822. [Google Scholar] [CrossRef] [PubMed]
- Palejwala, A.H.; Fridley, J.S.; Mata, J.A.; Samuel, E.L.G.; Luerssen, T.G.; Perlaky, L.; Kent, T.A.; Tour, J.M.; Jea, A. Biocompatibility of reduced graphene oxide nanoscaffolds following acute spinal cord injury in rats. Surg. Neurol. Int. 2016, 7, 75. [Google Scholar] [CrossRef] [PubMed]
- Wieken, K.; Angioi-Duprez, K.; Lim, A.; Marchal, L.; Merle, M. Nerve anastomosis with glue: Comparative histologic study of fibrin and cyanoacrylate glue. J. Reconstr. Microsurg. 2003, 19, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Brodbaker, E.; Bahar, I.; Slomovic, A.R. Novel use of fibrin glue in the treatment of conjunctivochalasis. Cornea 2008, 27, 950–952. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-C.; Huang, W.-C.; Chen, Y.-C.; Tu, T.-H.; Tsai, Y.-A.; Huang, S.-F.; Huang, H.-C.; Cheng, H. Acidic fibroblast growth factor for repair of human spinal cord injury: A clinical trial. J. Neurosurg. Spine 2011, 15, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Iwakawa, M.; Mizoi, K.; Tessler, A.; Itoh, Y. Intraspinal implants of fibrin glue containing glial cell line-derived neurotrophic factor promote dorsal root regeneration into spinal cord. Neurorehabil. Neural Repair 2001, 15, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Huang, S.S.; Lin, S.M.; Lin, M.J.; Chu, Y.C.; Chih, C.L.; Tsai, M.J.; Lin, H.C.; Huang, W.C.; Tsai, S.K. The neuroprotective effect of glial cell line-derived neurotrophic factor in fibrin glue against chronic focal cerebral ischemia in conscious rats. Brain Res. 2005, 1033, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Petter-Puchner, A.H.; Froetscher, W.; Krametter-Froetscher, R.; Lorinson, D.; Redl, H.; van Griensven, M. The long-term neurocompatibility of human fibrin sealant and equine collagen as biomatrices in experimental spinal cord injury. Exp. Toxicol. Pathol. 2007, 58, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Graham, L.; Wang, Y.; Wu, D.; Tuszynski, M. Promotion of Survival and Differentiation of Neural Stem Cells with Fibrin and Growth Factor Cocktails after Severe Spinal Cord Injury. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
- Willerth, S.M.; Faxel, T.E.; Gottlieb, D.I.; Sakiyama-Elbert, S.E. The Effects of Soluble Growth Factors on Embryonic Stem Cell Differentiation Inside of Fibrin Scaffolds. Stem Cells 2007, 25, 2235–2244. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Wang, Y.; Graham, L.; McHale, K.; Gao, M.; Wu, D.; Brock, J.; Blesch, A.; Rosenzweig, E.S.; Havton, L.A.; et al. Long-Distance Growth and Connectivity of Neural Stem Cells after Severe Spinal Cord Injury. Cell 2012, 150, 1264–1273. [Google Scholar] [CrossRef] [PubMed]
- Guest, J.D.; Hesse, D.; Schnell, L.; Schwab, M.E.; Bunge, M.B.; Bunge, R.P. Influence of IN-1 antibody and acidic FGF-fibrin glue on the response of injured corticospinal tract axons to human Schwann cell grafts. J. Neurosci. Res. 1997, 50, 888–905. [Google Scholar] [CrossRef]
- Kuo, H.-S.; Tsai, M.-J.; Huang, M.-C.; Chiu, C.-W.; Tsai, C.-Y.; Lee, M.-J.; Huang, W.-C.; Lin, Y.-L.; Kuo, W.-C.; Cheng, H. Acid fibroblast growth factor and peripheral nerve grafts regulate Th2 cytokine expression, macrophage activation, polyamine synthesis, and neurotrophin expression in transected rat spinal cords. J. Neurosci. 2011, 31, 4137–4147. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Tu, Y.; Zhao, M.; Chen, C.; Liang, H.; Wang, J.; Zhang, S.; Li, X. Preparation of Bionic Collagen-Heparin Sulfate Spinal Cord Scaffold with Three-dimentional print technology. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2015, 29, 1022–1027. [Google Scholar] [PubMed]
- Chen, B.; Bohnert, D.; Borgens, R.B.; Cho, Y. Pushing the science forward: Chitosan nanoparticles and functional repair of CNS tissue after spinal cord injury. J. Biol. Eng. 2013, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Nomura, H.; Baladie, B.; Katayama, Y.; Morshead, C.M.; Shoichet, M.S.; Tator, C.H. Delayed implantation of intramedullary chitosan channels containing nerve grafts promotes extensive axonal regeneration after spinal cord injury. Neurosurgery 2008, 63, 127–141, discussion 141–143. [Google Scholar] [CrossRef] [PubMed]
- Seth, N.; Simmons, H.A.; Masood, F.; Graham, W.A.; Rosene, D.L.; Westmoreland, S.V.; Cummings, S.M.; Gwardjan, B.; Sejdic, E.; Hoggatt, A.F.; et al. Model of Traumatic Spinal Cord Injury for Evaluating Pharmacologic Treatments in Cynomolgus Macaques (Macaca fasicularis). Comp. Med. 2018, 68, 63–73. [Google Scholar] [PubMed]
| Project Name | Mechanism | Current Progress | Future Outlook | Ref. |
|---|---|---|---|---|
| Nogo Trap of ReNetX Bio | A decoy receptor that binds growth inhibitors, allowing for the nerve fibers to grow naturally and directly. | Nogo Trap has demonstrated improved neurologic function following CNS damage in several animal models. | Planning phase Ib–IIa clinical trials to test safety and efficacy for patients with a chronic cervical incomplete SCI. | [46] |
| CHASE-IT Preclinical Initiative of the International Spinal Research Trust (ISRT) | The application of the biological enzyme Ch’ase in animal models is reported to have degraded scar tissue, promoted growth and improved activity. | Ch’ase has proven to be effective in rats, delivered to both thoracic and cervical contusion injury sites. Latest animal studies took place in 2016 and proved that longer-term application of the enzyme led to more significant motor control improvement. | Promising outcome, but one should bear in mind that data is based on rodent in vivo models; will this translate to humans? | [47] |
| Intracellular sigma peptide (ISP), Ch’ase and combinations preclinical projects | Using the biological enzyme Ch’ase in combinations with intracellular sigma peptide in order to restore breathing after long chronic C2 hemisection injury. | These projects are at a single center led by Jerry Silver. Currently these applications are at a pre-clinical stage. | Development of the product and preparation for clinical trials. | [48] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalamagkas, K.; Tsintou, M.; Seifalian, A.; Seifalian, A.M. Translational Regenerative Therapies for Chronic Spinal Cord Injury. Int. J. Mol. Sci. 2018, 19, 1776. https://doi.org/10.3390/ijms19061776
Dalamagkas K, Tsintou M, Seifalian A, Seifalian AM. Translational Regenerative Therapies for Chronic Spinal Cord Injury. International Journal of Molecular Sciences. 2018; 19(6):1776. https://doi.org/10.3390/ijms19061776
Chicago/Turabian StyleDalamagkas, Kyriakos, Magdalini Tsintou, Amelia Seifalian, and Alexander M. Seifalian. 2018. "Translational Regenerative Therapies for Chronic Spinal Cord Injury" International Journal of Molecular Sciences 19, no. 6: 1776. https://doi.org/10.3390/ijms19061776
APA StyleDalamagkas, K., Tsintou, M., Seifalian, A., & Seifalian, A. M. (2018). Translational Regenerative Therapies for Chronic Spinal Cord Injury. International Journal of Molecular Sciences, 19(6), 1776. https://doi.org/10.3390/ijms19061776