Classical Risk Factors and Inflammatory Biomarkers: One of the Missing Biological Links between Cardiovascular Disease and Major Depressive Disorder
Abstract
:1. Introduction
2. Results
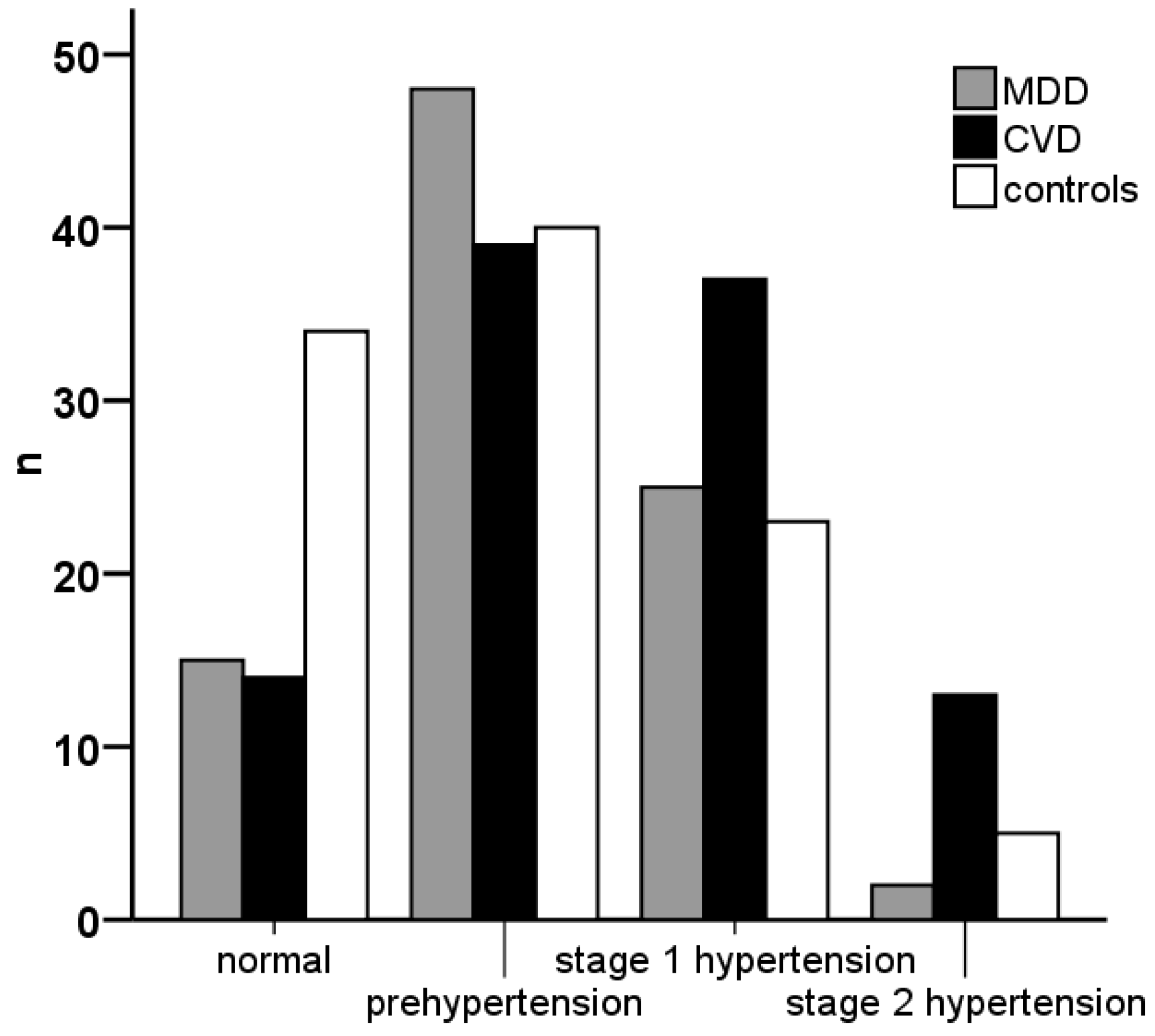
2.1. Classical Cardiovascular Risk Markers
2.2. Inflammation Biomarkers in MDD in Comparison to Healthy Controls
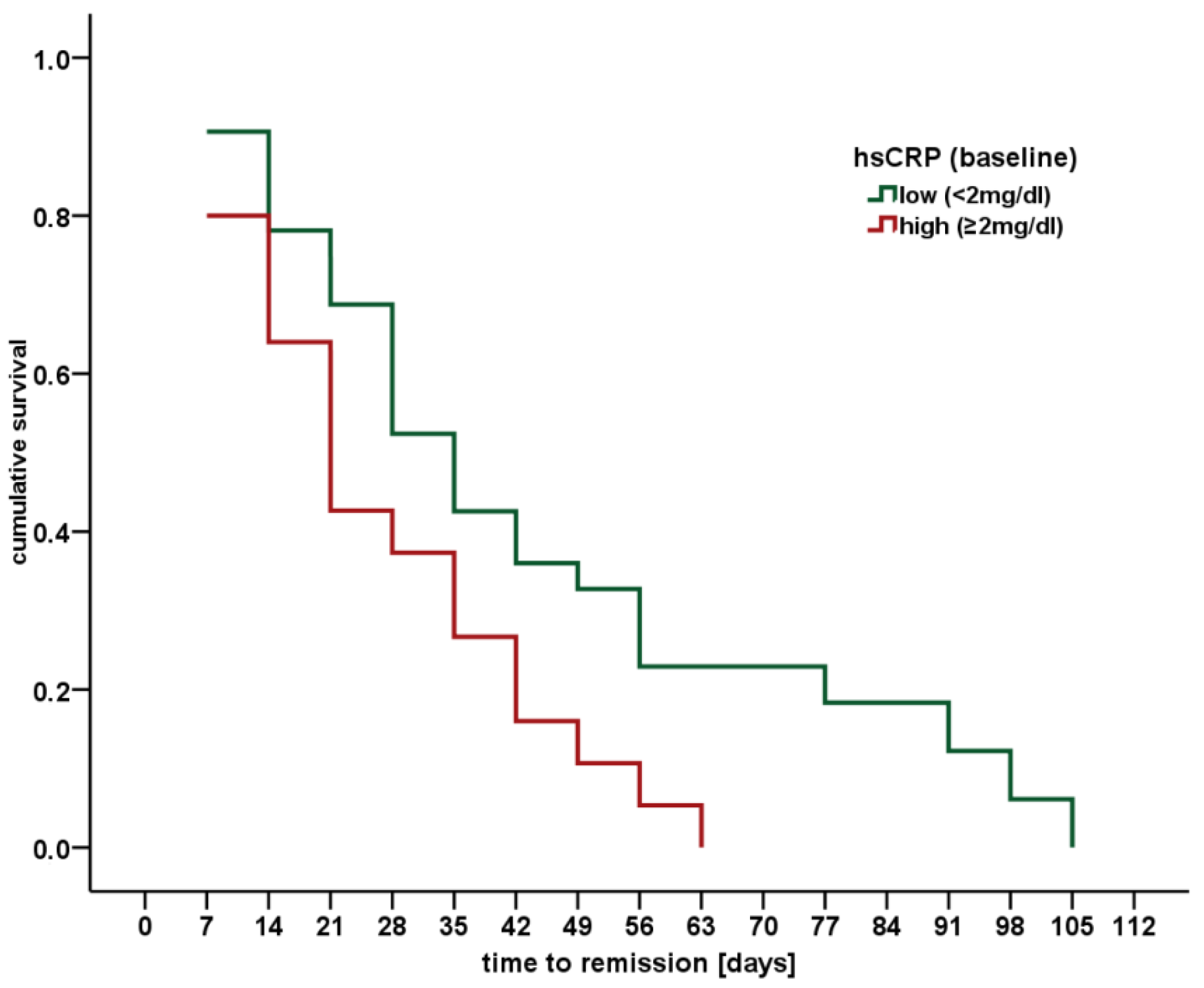
2.3. Cardiovascular Risk, Severity of Depression, and Time to Remission in MDD
3. Discussion
4. Material and Methods
4.1. Study Samples
4.2. Assessment of Vital Signs and Calculation of the Framingham-Index
4.3. Biochemical Analyses
Inflammatory Risk Factors
4.4. Statistical Analysis
4.5. Ethical Approval
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Mathers, C.D.; Stein, C.; Ma Fat, D.; Rao, C.; Inoue, M.; Tomijima, N.; Bernard, C.; De Lopez, A.; Murray, C.J.L. Global Burden of Disease 2000: Version 2 Methods and Results; Global Programme on Evidence for Health Policy Discussion Paper; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Ivanovs, R.; Kivite, A.; Ziedonis, D.; Mintale, I.; Vrublevska, J.; Rancans, E. Association of depression and anxiety with cardiovascular co-morbidity in a primary care population in Latvia: A cross-sectional study. BMC Public Health 2018, 18, 328. [Google Scholar] [CrossRef] [PubMed]
- Frasure-Smith, N.; Lesperance, F.; Talajic, M. Depression following myocardial infarction. Impact on 6-month survival. JAMA 1993, 270, 1819–1825. [Google Scholar] [CrossRef] [PubMed]
- Barefoot, J.C.; Schroll, M. Symptoms of depression, acute myocardial infarction, and total mortality in a community sample. Circulation 1996, 93, 1976–1980. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, Y.; Ma, L. Depression and cardiovascular disease in elderly: Current understanding. J. Clin. Neurosci. 2018, 47, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Penninx, B.W.; Beekman, A.T.; Honig, A.; Deeg, D.J.; Schoevers, R.A.; van Eijk, J.T.; van Tilburg, W. Depression and cardiac mortality: Results from a community-based longitudinal study. Arch. Gen. Psychiatry 2001, 58, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Lederbogen, F.; Deuschle, M.; Heuser, I. Depression—A cardiovascular risk factor. Internist 1999, 40, 1119–1121. [Google Scholar] [CrossRef] [PubMed]
- Wulsin, L.R.; Singal, B.M. Do depressive symptoms increase the risk for the onset of coronary disease? A systematic quantitative review. Psychosom. Med. 2003, 65, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Grippo, A.J.; Johnson, A.K. Biological mechanisms in the relationship between depression and heart disease. Neurosci. Biobehav. Rev. 2002, 26, 941–962. [Google Scholar] [CrossRef]
- Libby, P. Inflammation in atherosclerosis. Nature 2002, 420, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Blake, G.J.; Ridker, P.M. Novel clinical markers of vascular wall inflammation. Circ. Res. 2001, 89, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Blann, A.D.; Lip, G.Y.; McCollum, C.N. Changes in von Willebrand factor and soluble ICAM, but not soluble VCAM, soluble E selectin or soluble thrombomodulin, reflect the natural history of the progression of atherosclerosis. Atherosclerosis 2002, 165, 389–391. [Google Scholar] [CrossRef]
- Ridker, P.M. Inflammatory biomarkers, statins, and the risk of stroke: Cracking a clinical conundrum. Circulation 2002, 105, 2583–2585. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. Clinical application of C-reactive protein for cardiovascular disease detection and prevention. Circulation 2003, 107, 363–369. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Conti, C.M.; Campanella, D.; Carano, A.; Scali, M.; Valchera, A.; Serroni, N.; Pizzorno, A.M.; D’Albenzio, A.; Fulcheri, M.; et al. Evaluation of C-reactive protein and total serum cholesterol in adult patients with bipolar disorder. Int. J. Immunopathol. Pharmacol. 2008, 21, 319–324. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Conti, C.M.; Serroni, N.; Moschetta, F.S.; Carano, A.; Salerno, R.M.; Cavuto, M.; Farina, B.; Alessandrini, M.; Janiri, L.; et al. The role of cholesterol levels in mood disorders and suicide. J. Biol. Regul. Homeost. Agents 2009, 23, 133–140. [Google Scholar] [PubMed]
- De Berardis, D.; Marini, S.; Piersanti, M.; Cavuto, M.; Perna, G.; Valchera, A.; Mazza, M.; Fornaro, M.; Iasevoli, F.; Martinotti, G.; et al. The Relationships between Cholesterol and Suicide: An Update. ISRN Psychiatry 2012. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.S. The macrophage theory of depression. Med. Hypotheses 1991, 35, 298–306. [Google Scholar] [CrossRef]
- Holsboer, F. The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology 2000, 23, 477–501. [Google Scholar] [CrossRef]
- Leonard, B.E. The HPA and immune axes in stress: The involvement of the serotonergic system. Eur. Psychiatry 2005, 20 (Suppl. 3), S302–S306. [Google Scholar] [CrossRef]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Leonard, B.E.; Myint, A. The psychoneuroimmunology of depression. Hum. Psychopharmacol. 2009, 24, 165–175. [Google Scholar] [PubMed]
- Bremmer, M.A.; Beekman, A.T.; Deeg, D.J.; Penninx, B.W.; Dik, M.G.; Hack, C.E.; Hoogendijk, W.J. Inflammatory markers in late-life depression: Results from a population-based study. J. Affect. Disord. 2008, 106, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Leonard, B.E. Psychopathology of depression. Drugs Today 2007, 43, 705–716. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Na, K.S.; Shin, K.H.; Jung, H.Y.; Choi, S.H.; Kim, J.B. Cytokine imbalance in the pathophysiology of major depressive disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 2007, 31, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Ovaskainen, Y.; Koponen, H.; Jokelainen, J; Keinanen-Kiukaanniemi, S.; Kumpusalo, E.; Vanhala, M. Depressive symptomatology is associated with decreased interleukin-1 beta and increased interleukin-1 receptor antagonist levels in males. Psychiatry Res. 2009, 167, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L., Jr.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T., Jr.; et al. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure: The JNC 7 report. JAMA 2003, 289, 2560–2572. [Google Scholar] [CrossRef] [PubMed]
- Danner, M.; Kasl, S.V.; Abramson, J.L.; Vaccarino, V. Association between depression and elevated C.-reactive protein. Psychosom. Med. 2003, 65, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. High-sensitivity C-reactive protein, inflammation, and cardiovascular risk: From concept to clinical practice to clinical benefit. Am. Heart J. 2004, 148, S19–S26. [Google Scholar] [CrossRef] [PubMed]
- The ENRICHD investigators. Enhancing recovery in coronary heart disease (ENRICHD): Baseline characteristics. Am. J. Cardiol. 2001, 88, 316–322. [Google Scholar]
- Sorensen, C.; Brandes, A.; Hendricks, O.; Thrane, J.; Friis-Hasche, E.; Haghfelt, T.; Bech, P. Psychosocial predictors of depression in patients with acute coronary syndrome. Acta Psychiatr. Scand. 2005, 111, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Duivis, H.E.; de Jonge, P.; Penninx, B.W.; Na, B.Y.; Cohen, B.E.; Whooley, M.A. Depressive symptoms, health behaviors, and subsequent inflammation in patients with coronary heart disease: Prospective findings from the heart and soul study. Am. J. Psychiatry 2011, 168, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Kannel, W.B.; McGee, D.; Gordon, T. A general cardiovascular risk profile: The Framingham Study. Am. J. Cardiol. 1976, 38, 46–51. [Google Scholar] [CrossRef]
- Yan, L.L.; Liu, K.; Matthews, K.A.; Daviglus, M.L.; Ferguson, T.F.; Kiefe, C.I. Psychosocial factors and risk of hypertension: The Coronary Artery Risk Development in Young Adults (CARDIA) study. JAMA 2003, 290, 2138–2148. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, F.; Irving, K.; Terra, J.L.; Nony, P.; Berthezene, F.; Moulin, P. Depressive symptoms are associated with unhealthy lifestyles in hypertensive patients with the metabolic syndrome. J. Hypertens. 2005, 23, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Richter, N.; Juckel, G.; Assion, H.J. Metabolic syndrome: A follow-up study of acute depressive inpatients. Eur. Arch. Psychiatry Clin. Neurosci. 2009, 260, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Willerson, J.T.; Ridker, P.M. C-reactive protein and other emerging blood biomarkers to optimize risk stratification of vulnerable patients. J. Am. Coll. Cardiol. 2006, 47, C19–C31. [Google Scholar] [CrossRef] [PubMed]
- Papanicolaou, D.A.; Wilder, R.L.; Manolagas, S.C.; Chrousos, G.P. The pathophysiologic roles of interleukin-6 in human disease. Ann. Intern. Med. 1998, 128, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Lesperance, F.; Frasure-Smith, N.; Theroux, P.; Irwin, M. The association between major depression and levels of soluble intercellular adhesion molecule 1, interleukin-6, and C-reactive protein in patients with recent acute coronary syndromes. Am. J. Psychiatry 2004, 161, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Chrysohoou, C.; Kollia, N.; Tousoulis, D. The link between depression and atherosclerosis through the pathways of inflammation and endothelium dysfunction. Maturitas 2018, 109, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Ho, C.S.; Liu, X.; Chua, A.N.; Wang, W.; McIntyre, R.S.; Ho, R.C. Chronic administration of fluoxetine and pro-inflammatory cytokine change in a rat model of depression. PLoS ONE 2017, 12, e0186700. [Google Scholar] [CrossRef] [PubMed]
- Empana, J.P.; Jouven, X.; Canoui-Poitrine, F.; Luc, G.; Tafflet, M.; Haas, B.; Arveiler, D.; Ferrieres, J.; Ruidavets, J.B.; Montaye, M.; et al. C-reactive protein, interleukin 6, fibrinogen and risk of sudden death in European middle-aged men: The PRIME study. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2047–2052. [Google Scholar] [CrossRef] [PubMed]
- Kiecolt-Glaser, J.K.; Glaser, R. Depression and immune function: Central pathways to morbidity and mortality. J. Psychosom. Res. 2002, 53, 873–876. [Google Scholar] [CrossRef]
- Lanquillon, S.; Krieg, J.C.; Bening-Abu-Shach, U.; Vedder, H. Cytokine production and treatment response in major depressive disorder. Neuropsychopharmacology 2000, 22, 370–379. [Google Scholar] [CrossRef]
- Aronson, D.; Avizohar, O.; Levy, Y.; Bartha, P.; Jacob, G.; Markiewicz, W. Factor analysis of risk variables associated with low-grade inflammation. Atherosclerosis 2008, 200, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Choo, C.C.; Chew, P.K.H.; Lai, S.M.; Soo, S.C.; Ho, C.S.; Ho, R.C.; Wong, R.C. Effect of Cardiac Rehabilitation on Quality of Life, Depression and Anxiety in Asian Patients. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4rd ed.; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- Hamilton, M. Development of a rating scale for primary depressive illness. Br. J. Soc. Clin. Psychol. 1967, 6, 278–296. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.W.; D’Agostino, R.B.; Levy, D.; Belanger, A.M.; Silbershatz, H.; Kannel, W.B. Prediction of coronary heart disease using risk factor categories. Circulation 1998, 97, 1837–1847. [Google Scholar] [CrossRef] [PubMed]
| Samples | Sample 1 * | Sample 2 | Sample 3 † | Kruskal-Wallis- or χ2-Test ‡ | |
|---|---|---|---|---|---|
| Variable | MDD | CVD | Controls | χ2, d.f., p | |
| n | 100 | 106 | 104 | ||
| age (mean ± SD) | 46.6 ± 14.8 | 66.9 ± 7.3 | 54.7 ± 14.4 | 100, 2, p < 0.001 | |
| sex | |||||
| (male/female) | 37.0%/63.0% | 81.1%/18.9% | 45.2%/54.8% | 46.3, 2, p < 0.001 ‡ | |
| clinical ratings (baseline) | |||||
| CGI-1 | 5.3 ± 0.5 | 1.3 ± 0.8 | 1.0 ± 0.0 | 205, 2, p < 0.001 | |
| HAM-D17 (mean ± SD) | 22.0 ± 5.3 | 2.4 ± 4.4 | 0.7 ± 1.2 | 160, 2, p < 0.001 | |
| MADRS (mean ± SD) | 31.8 ± 7.4 | 3.2 ± 6.0 | 0.7 ± 1.3 | 162, 2, p < 0.001 | |
| BDI (mean ± SD) | 26.4 ± 9.1 | 7.0 ± 5.1 | 2.8 ± 3.2 | 130, 2, p < 0.001 | |
| Vital signs and classical cardiovascular risk factors | |||||
| blood pressure (systolic) | 129.2 ± 13.5 | 138.6 ± 19.1 | 128.6 ± 19.1 | 18.7, 2, p < 0.001 | |
| blood pressure (diastolic) | 79.7 ± 8.2 | 78.3 ± 10.9 | 76.8 ± 11.9 | 4.63, 2, n.s. § | |
| heart rate (beats/minute) | 85.7 ± 14.7 | 63.8 ± 10.7 | 68.9 ± 10.1 | 104, 2, p < 0.001 | |
| total cholesterol (mg/dL) | 206.2 ± 58.1 | n.d. | 223.8 ± 48.1 | 4.15, 1, p = 0.042 | |
| LDL (mg/dL) | 125.2 ± 42.8 | n.d. | 136.9 ± 45.6 | 2.30, 1, n.s. | |
| triglycerides (mg/dL) | 148.8 ± 100.3 | n.d. | 100.6 ± 51.6 | 10.5, 1, p = 0.001 | |
| HDL (mg/dL) | 58.4 ± 17.1 | n.d. | 66.1 ± 19.8 | 5.67, 1, p = 0.017 | |
| fasting glucose (mg/dL) | 95.1 ± 18.2 | n.d. | 86.9 ± 11.6 | 5.41, 1, p = 0.02 | |
| body weight (kg) | 72.9 ± 13.6 | 82.8 ± 14.7 | 72.3 ± 11.8 | 32.9, 2, p < 0.001 | |
| body mass index (kg/m2) | 25.3 ± 4.0 | 28.0 ± 4.4 | 24.4 ± 2.9 | 35.8, 2, p < 0.001 | |
| waist circumference (cm) | 96.9 ± 12.3 | 101.9 ± 12.9 | 90.1 ± 12.1 | 34.2, 2, p < 0.001 | |
| hip circumference (cm) | 105.3 ± 10.7 | 105.8 ± 9.5 | 100.6 ± 8.4 | 12.5, 2, p = 0.002 | |
| waist-hip-ratio | 0.92 ± 0.12 | 0.96 ± 0.07 | 0.90 ± 0.09 | 35.9, 2, p < 0.001 | |
| smoker/non-smoker (%) | 42.9%/57.1% | 9.0%/91.0% | 14.6%/85.4% | 34.1, 2, p < 0.001 ‡ | |
| pack years (20 cigarettes/day * years) | 18.8 ± 13.4 | 35.7 ± 30.7 | 18.8 ± 20.3 | 9.96, 2, p = 0.007 | |
| Framingham-index (total) | 2.77 ± 5.74 | 9.69 ± 2.69 | 4.09 ± 5.86 | 99.3, 2, p < 0.001 | |
| Framingham-index, 10 years-risk (%) | 4.94 ± 5.62 | 11.47 ± 7.11 | 5.85 ± 5.78 | 96.4, 2, p < 0.001 | |
| Samples | Sample 1 | t-Test * | Sample 3 | t-Test † | ||
|---|---|---|---|---|---|---|
| MDD | Controls | |||||
| Variable | Baseline | Discharge | T, p | Baseline | T, p | |
| inflammation marker | ||||||
| hsCRP (mg/L) | 3.07 ± 3.7 | 3.97 ± 4.4 | 0.23, n.s. | 1.37 ± 1.2 | 4.25, p < 0.001 | |
| pro-inflammatory cytokines | ||||||
| interleukin 1β (IL-1 β) (pg/mL) | 1.08 ± 1.1 | 1.34 ± 1.0 | 0.88, n.s. | 0.54 ± 0.6 | 4.10, p < 0.001 | |
| interleukin 6 (IL-6) (pg/mL) | 1.58 ± 1.5 | 1.35 ± 1.7 | 2.26, p = 0.027 | 1.32 ± 1.3 | 2.64, p = 0.009 | |
| adhesion molecules | ||||||
| P selectin (ng/mL) | 150.4 ± 102.4 | 114.4 ± 78.9 | 2.56, p = 0.013 | 184.2 ± 146.0 | −1.47, n.s. | |
| E selectin (ng/mL) | 54.6 ± 29.0 | 58.0 ± 32.0 | −0.69, n.s. | 46.5 ± 27.2 | 1.76, n.s. | |
| MCP-1 (pg/mL) | 221.3 ± 149.7 | 301.8 ± 179.2 | −2.92, p = 0.005 | 256.5 ± 140.8 | −1.54, n.s. | |
| sICAM-1 (ng/mL) | 535.4 ± 210.0 | 555.0 ± 209.5 | −0.92, n.s. | 360.6 ± 107.7 | 5.90, p < 0.001 | |
| sVCAM-1 (ng/mL) | 486.1 ± 182.0 | 533.2 ± 221.4 | −1.62, n.s. | 552.3 ± 142.2 | −4.41, p = 0.017 | |
| costimulatory glycoprotein | ||||||
| sCD40 (ng/mL) | 10.4 ± 3.7 | 11.3 ± 3.7 | −2.42, p = 0.019 | 10.1 ± 4.2 | 0.59, n.s. | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baghai, T.C.; Varallo-Bedarida, G.; Born, C.; Häfner, S.; Schüle, C.; Eser, D.; Zill, P.; Manook, A.; Weigl, J.; Jooyandeh, S.; et al. Classical Risk Factors and Inflammatory Biomarkers: One of the Missing Biological Links between Cardiovascular Disease and Major Depressive Disorder. Int. J. Mol. Sci. 2018, 19, 1740. https://doi.org/10.3390/ijms19061740
Baghai TC, Varallo-Bedarida G, Born C, Häfner S, Schüle C, Eser D, Zill P, Manook A, Weigl J, Jooyandeh S, et al. Classical Risk Factors and Inflammatory Biomarkers: One of the Missing Biological Links between Cardiovascular Disease and Major Depressive Disorder. International Journal of Molecular Sciences. 2018; 19(6):1740. https://doi.org/10.3390/ijms19061740
Chicago/Turabian StyleBaghai, Thomas C., Gabriella Varallo-Bedarida, Christoph Born, Sibylle Häfner, Cornelius Schüle, Daniela Eser, Peter Zill, André Manook, Johannes Weigl, Somayeh Jooyandeh, and et al. 2018. "Classical Risk Factors and Inflammatory Biomarkers: One of the Missing Biological Links between Cardiovascular Disease and Major Depressive Disorder" International Journal of Molecular Sciences 19, no. 6: 1740. https://doi.org/10.3390/ijms19061740
APA StyleBaghai, T. C., Varallo-Bedarida, G., Born, C., Häfner, S., Schüle, C., Eser, D., Zill, P., Manook, A., Weigl, J., Jooyandeh, S., Nothdurfter, C., Von Schacky, C., Bondy, B., & Rupprecht, R. (2018). Classical Risk Factors and Inflammatory Biomarkers: One of the Missing Biological Links between Cardiovascular Disease and Major Depressive Disorder. International Journal of Molecular Sciences, 19(6), 1740. https://doi.org/10.3390/ijms19061740





