Downregulation of BTLA on NKT Cells Promotes Tumor Immune Control in a Mouse Model of Mammary Carcinoma
Abstract
:1. Introduction
2. Results
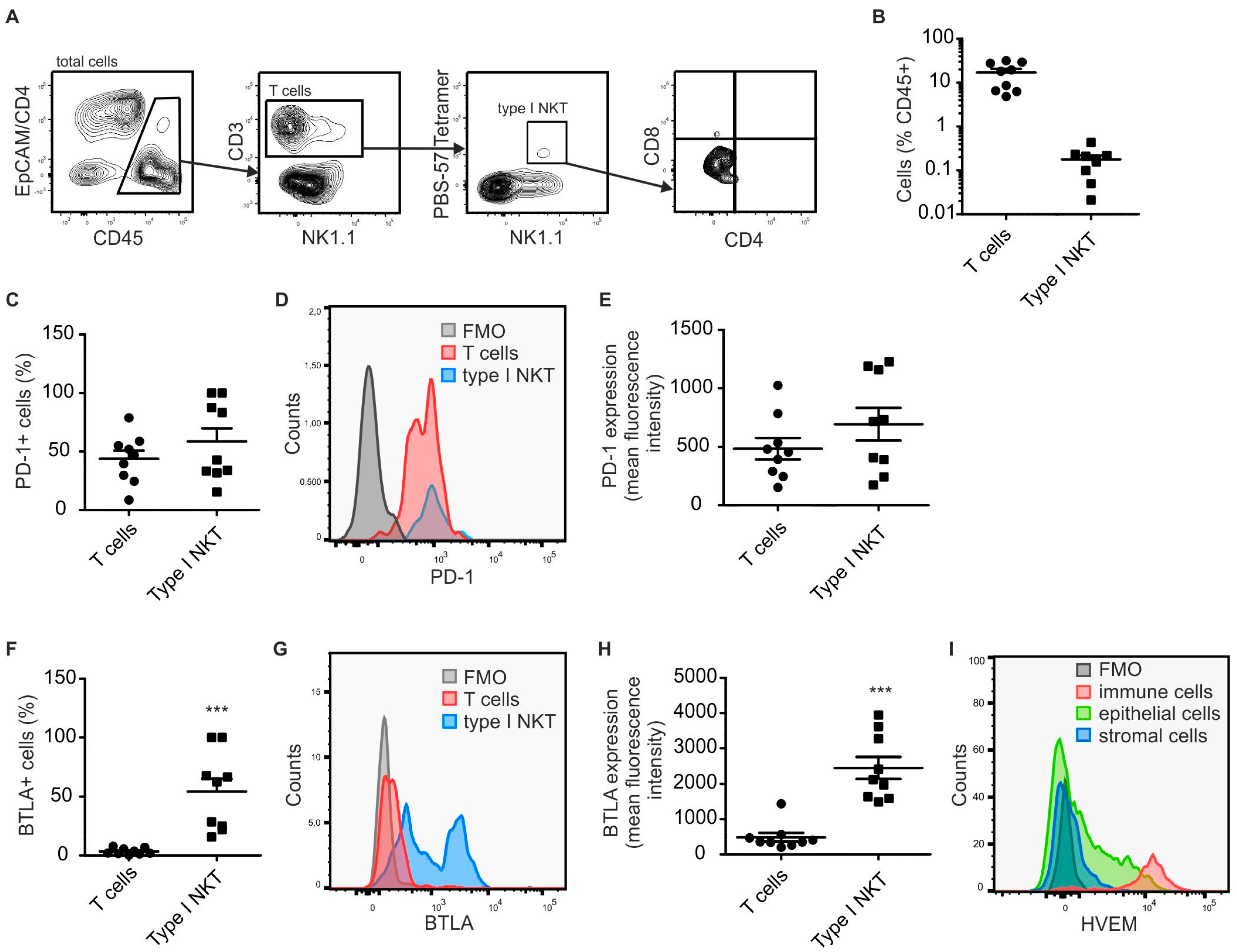
2.1. Type I NKT Express BTLA in PyMT Mammary Tumors
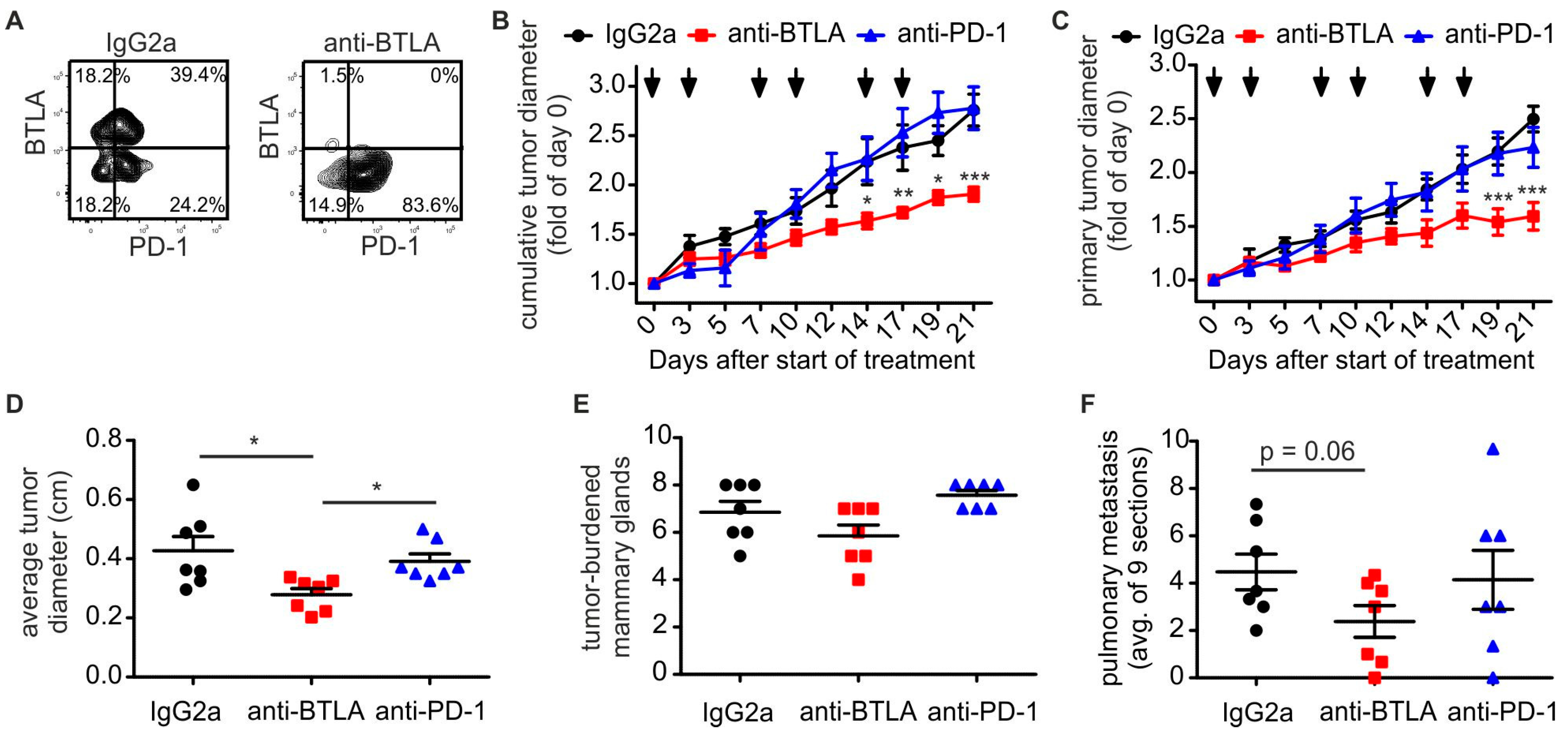
2.2. Downregulation of BTLA But Not PD-1 Blockade Reduces PyMT Mammary Tumor Progression
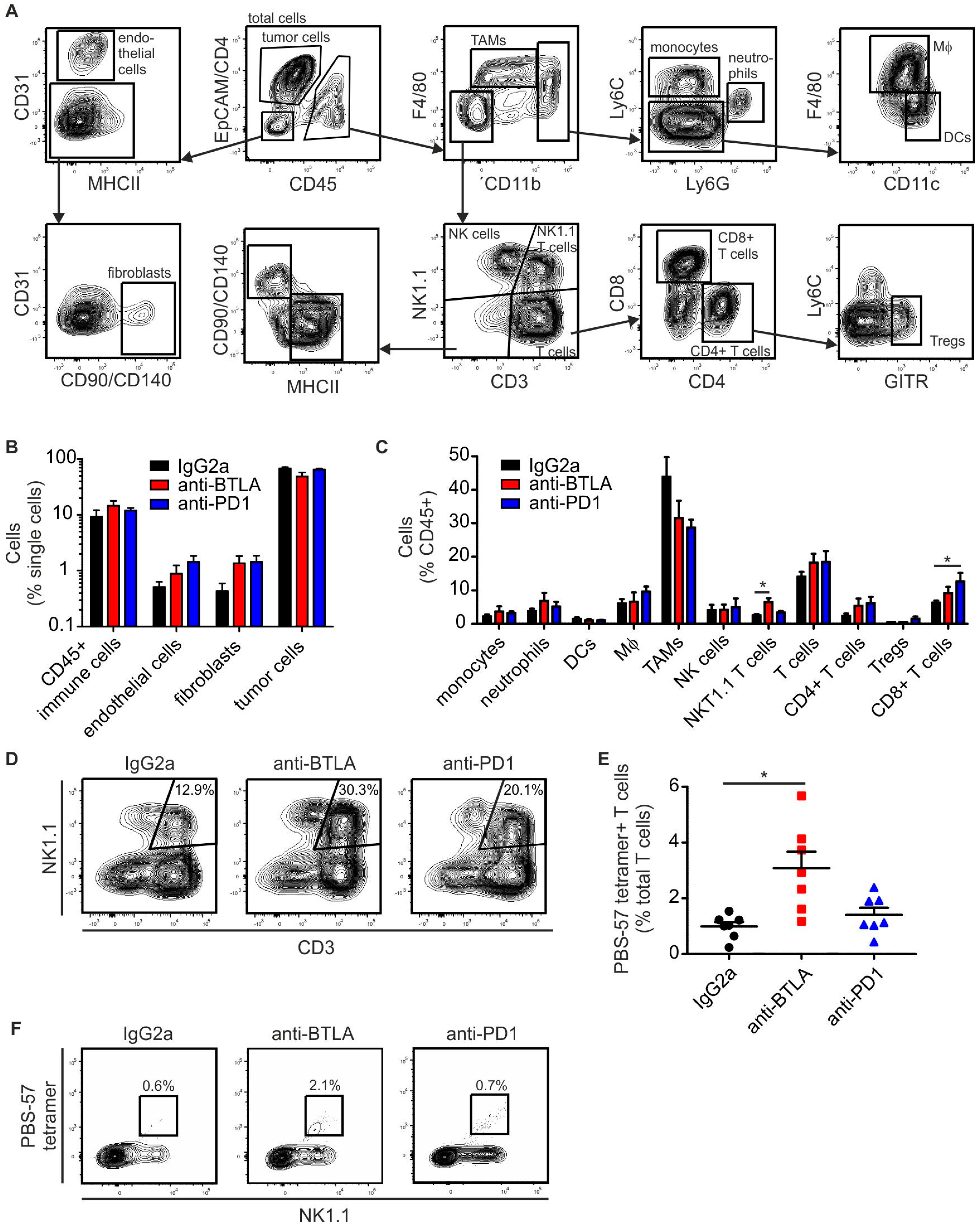
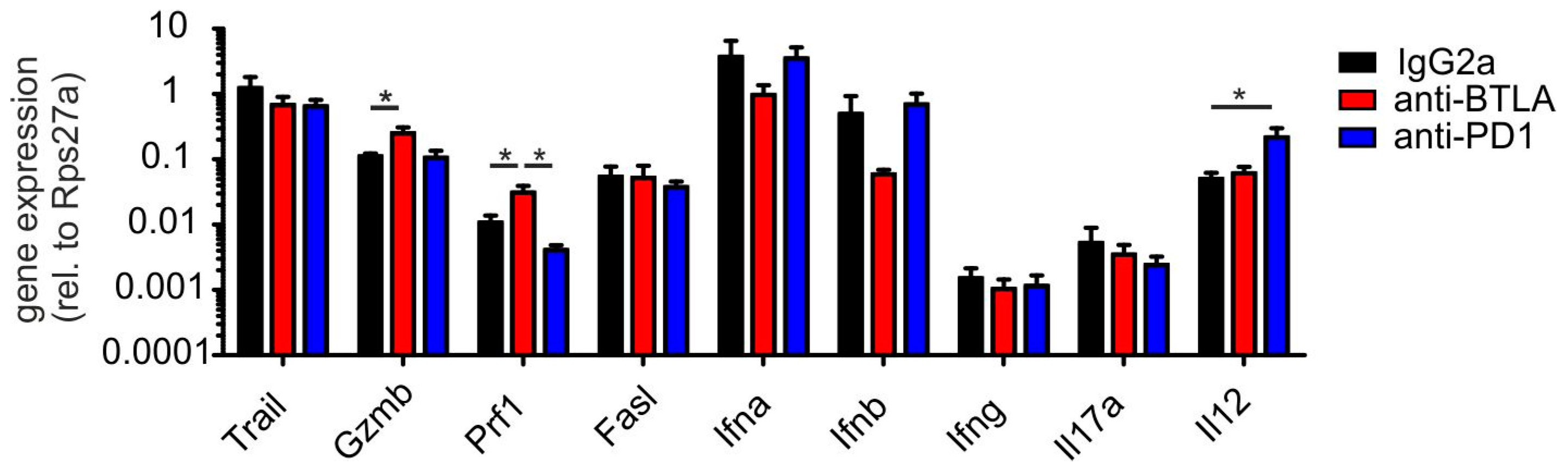
2.3. BTLA Downregulation Increases NKT Cell Numbers
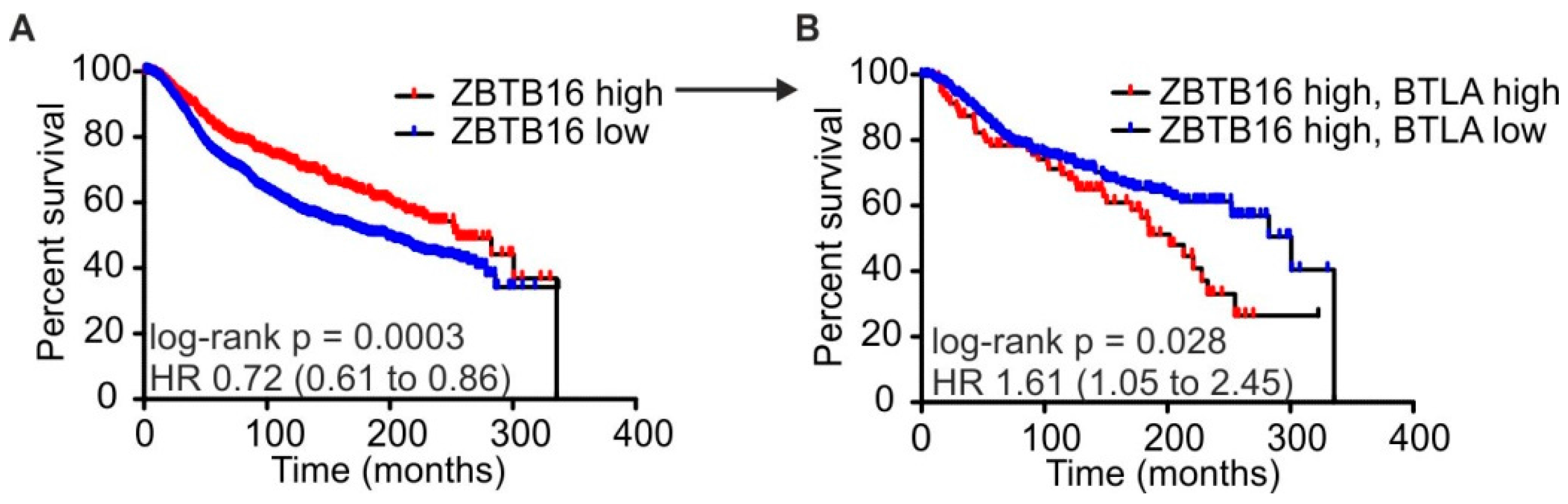
2.4. High BTLA and ZBTB16 Expression Defines a Subset of Breast Cancer Patients with Favorable Prognosis
3. Discussion
4. Materials and Methods
4.1. Animal Experiments
4.2. Immunohistochemistry
4.3. Flow Cytometry
4.4. Generation of CD1d Tetramers
4.5. RNA Isolation and RT-qPCR
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gentles, A.J.; Newman, A.M.; Liu, C.L.; Bratman, S.V.; Feng, W.; Kim, D.; Nair, V.S.; Xu, Y.; Khuong, A.; Hoang, C.D.; et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 2015, 21, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Hu-Lieskovan, S.; Wargo, J.A.; Ribas, A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 2017, 168, 707–723. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Gavrieli, M.; Sedy, J.R.; Yang, J.; Fallarino, F.; Loftin, S.K.; Hurchla, M.A.; Zimmerman, N.; Sim, J.; Zang, X.; et al. BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat. Immunol. 2003, 4, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Sedy, J.R.; Gavrieli, M.; Potter, K.G.; Hurchla, M.A.; Lindsley, R.C.; Hildner, K.; Scheu, S.; Pfeffer, K.; Ware, C.F.; Murphy, T.L.; et al. B and T lymphocyte attenuator regulates T cell activation through interaction with herpesvirus entry mediator. Nat. Immunol. 2005, 6, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, M.L.; Kaye, J.; Rodriguez-Barbosa, J.I. Detection of protein on BTLAlow cells and in vivo antibody-mediated down-modulation of BTLA on lymphoid and myeloid cells of C57BL/6 and BALB/c BTLA allelic variants. Immunobiology 2010, 215, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Han, P.; Goularte, O.D.; Rufner, K.; Wilkinson, B.; Kaye, J. An inhibitory Ig superfamily protein expressed by lymphocytes and APCs is also an early marker of thymocyte positive selection. J. Immunol. 2004, 172, 5931–5939. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, M.L.; Jones, N.D.; Buhler, L.; Norris, P.; Shintani, Y.; Ware, C.F.; Rodriguez-Barbosa, J.I. Selective blockade of herpesvirus entry mediator-B and T lymphocyte attenuator pathway ameliorates acute graft-versus-host reaction. J. Immunol. 2012, 188, 4885–4896. [Google Scholar] [CrossRef] [PubMed]
- Le Mercier, I.; Lines, J.L.; Noelle, R.J. Beyond CTLA-4 and PD-1, the generation Z of negative checkpoint regulators. Front. Immunol. 2015, 6, 418. [Google Scholar] [CrossRef] [PubMed]
- Bekiaris, V.; Sedy, J.R.; Ware, C.F. Mixing signals: Molecular turn ons and turn offs for innate γδ T-cells. Front. Immunol. 2014, 5, 654. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.L.; Sun, Y.; Fu, Y.X. Cutting edge: B and T lymphocyte attenuator signaling on NKT cells inhibits cytokine release and tissue injury in early immune responses. J. Immunol. 2009, 183, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Godfrey, D.I.; MacDonald, H.R.; Kronenberg, M.; Smyth, M.J.; Van Kaer, L. NKT cells: What’s in a name? Nat. Rev. Immunol. 2004, 4, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Salio, M.; Silk, J.D.; Jones, E.Y.; Cerundolo, V. Biology of CD1- and MR1-restricted T cells. Annu. Rev. Immunol. 2014, 32, 323–366. [Google Scholar] [CrossRef] [PubMed]
- Kawano, T.; Cui, J.; Koezuka, Y.; Toura, I.; Kaneko, Y.; Motoki, K.; Ueno, H.; Nakagawa, R.; Sato, H.; Kondo, E.; et al. CD1d-restricted and TCR-mediated activation of valpha14 NKT cells by glycosylceramides. Science 1997, 278, 1626–1629. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.; Dhodapkar, M.V. Natural killer T cells in cancer immunotherapy. Front. Immunol. 2017, 8, 1178. [Google Scholar] [CrossRef] [PubMed]
- McEwen-Smith, R.M.; Salio, M.; Cerundolo, V. The regulatory role of invariant NKT cells in tumor immunity. Cancer Immunol. Res. 2015, 3, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Berzofsky, J.A.; Terabe, M. NKT cells in tumor immunity: Opposing subsets define a new immunoregulatory axis. J. Immunol. 2008, 180, 3627–3635. [Google Scholar] [CrossRef] [PubMed]
- Exley, M.A.; Friedlander, P.; Alatrakchi, N.; Vriend, L.; Yue, S.; Sasada, T.; Zeng, W.; Mizukami, Y.; Clark, J.; Nemer, D.; et al. Adoptive transfer of invariant NKT cells as immunotherapy for advanced melanoma: A phase I clinical trial. Clin. Cancer Res. 2017, 23, 3510–3519. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.Y.; Jones, J.G.; Li, P.; Zhu, L.; Whitney, K.D.; Muller, W.J.; Pollard, J.W. Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am. J. Pathol. 2003, 163, 2113–2126. [Google Scholar] [CrossRef]
- Ngiow, S.F.; Young, A.; Jacquelot, N.; Yamazaki, T.; Enot, D.; Zitvogel, L.; Smyth, M.J. A threshold level of intratumor CD8+ T-cell PD1 expression dictates therapeutic response to anti-PD1. Cancer Res. 2015, 75, 3800–3811. [Google Scholar] [CrossRef] [PubMed]
- Curtis, C.; Shah, S.P.; Chin, S.F.; Turashvili, G.; Rueda, O.M.; Dunning, M.J.; Speed, D.; Lynch, A.G.; Samarajiwa, S.; Yuan, Y.; et al. The genomic and transcriptomic architecture of 2000 breast tumours reveals novel subgroups. Nature 2012, 486, 346–352. [Google Scholar] [PubMed]
- Assarsson, E.; Kambayashi, T.; Sandberg, J.K.; Hong, S.; Taniguchi, M.; Van Kaer, L.; Ljunggren, H.G.; Chambers, B.J. CD8+ T cells rapidly acquire NK1.1 and NK cell-associated molecules upon stimulation in vitro and in vivo. J. Immunol. 2000, 165, 3673–3679. [Google Scholar] [CrossRef] [PubMed]
- Haas, J.D.; Gonzalez, F.H.; Schmitz, S.; Chennupati, V.; Fohse, L.; Kremmer, E.; Forster, R.; Prinz, I. CCR6 and NK1.1 distinguish between Il-17a and IFN-gamma-producing gammadelta effector T cells. Eur. J. Immunol. 2009, 39, 3488–3497. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Laouar, A.; Denzin, L.K.; Sant’Angelo, D.B. Zbtb16 (PLZF) is stably suppressed and not inducible in non-innate T cells via T cell receptor-mediated signaling. Sci. Rep. 2015, 5, 12113. [Google Scholar] [CrossRef] [PubMed]
- Bekiaris, V.; Sedy, J.R.; Macauley, M.G.; Rhode-Kurnow, A.; Ware, C.F. The inhibitory receptor BTLA controls gammadelta T cell homeostasis and inflammatory responses. Immunity 2013, 39, 1082–1094. [Google Scholar] [CrossRef] [PubMed]
- Bos, P.D.; Plitas, G.; Rudra, D.; Lee, S.Y.; Rudensky, A.Y. Transient regulatory T cell ablation deters oncogene-driven breast cancer and enhances radiotherapy. J. Exp. Med. 2013, 210, 2435–2466. [Google Scholar] [CrossRef] [PubMed]
- Iwata, A.; Watanabe, N.; Oya, Y.; Owada, T.; Ikeda, K.; Suto, A.; Kagami, S.; Hirose, K.; Kanari, H.; Kawashima, S.; et al. Protective roles of B and T lymphocyte attenuator in Nkt cell-mediated experimental hepatitis. J. Immunol. 2010, 184, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Kain, L.; Webb, B.; Anderson, B.L.; Deng, S.; Holt, M.; Costanzo, A.; Zhao, M.; Self, K.; Teyton, A.; Everett, C.; et al. The identification of the endogenous ligands of natural killer T cells reveals the presence of mammalian alpha-linked glycosylceramides. Immunity 2014, 41, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Fox, L.M.; Cox, D.G.; Lockridge, J.L.; Wang, X.; Chen, X.; Scharf, L.; Trott, D.L.; Ndonye, R.M.; Veerapen, N.; Besra, G.S.; et al. Recognition of lyso-phospholipids by human natural killer T lymphocytes. PLoS Biol. 2009, 7, e1000228. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.H.; Deng, H.; Matthews, P.; Krasovsky, J.; Ragupathi, G.; Spisek, R.; Mazumder, A.; Vesole, D.H.; Jagannath, S.; Dhodapkar, M.V. Inflammation-associated lysophospholipids as ligands for CD1d-restricted T cells in human cancer. Blood 2008, 112, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Facciotti, F.; Ramanjaneyulu, G.S.; Lepore, M.; Sansano, S.; Cavallari, M.; Kistowska, M.; Forss-Petter, S.; Ni, G.; Colone, A.; Singhal, A.; et al. Peroxisome-derived lipids are self antigens that stimulate invariant natural killer T cells in the thymus. Nat. Immunol. 2012, 13, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, D.I.; Cozzo, A.; Ji, X.; Roberts, L.S.; Louie, S.M.; Mulvihill, M.M.; Luo, K.; Nomura, D.K. Ether lipid generating enzyme AGPS alters the balance of structural and signaling lipids to fuel cancer pathogenicity. Proc. Natl. Acad. Sci. USA 2013, 110, 14912–14917. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.M.; Nelson, C.A.; Sedy, J.R. Balancing co-stimulation and inhibition with BTLA and HVEM. Nat. Rev. Immunol. 2006, 6, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Oya, Y.; Watanabe, N.; Owada, T.; Oki, M.; Hirose, K.; Suto, A.; Kagami, S.; Nakajima, H.; Kishimoto, T.; Iwamoto, I.; et al. Development of autoimmune hepatitis-like disease and production of autoantibodies to nuclear antigens in mice lacking B and T lymphocyte attenuator. Arthritis Rheum. 2008, 58, 2498–2510. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Barbosa, J.I.; Fernandez-Renedo, C.; Moral, A.M.B.; Buhler, L.; Del Rio, M.L. T follicular helper expansion and humoral-mediated rejection are independent of the HVEM/BTLA pathway. Cell. Mol. Immunol. 2017, 14, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Weichand, B.; Popp, R.; Dziumbla, S.; Mora, J.; Strack, E.; Elwakeel, E.; Frank, A.C.; Scholich, K.; Pierre, S.; Syed, S.N.; et al. S1PR1 on tumor-associated macrophages promotes lymphangiogenesis and metastasis via NLRP3/Il-1beta. J. Exp. Med. 2017, 214, 2695–2713. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sekar, D.; Govene, L.; Del Río, M.-L.; Sirait-Fischer, E.; Fink, A.F.; Brüne, B.; Rodriguez-Barbosa, J.I.; Weigert, A. Downregulation of BTLA on NKT Cells Promotes Tumor Immune Control in a Mouse Model of Mammary Carcinoma. Int. J. Mol. Sci. 2018, 19, 752. https://doi.org/10.3390/ijms19030752
Sekar D, Govene L, Del Río M-L, Sirait-Fischer E, Fink AF, Brüne B, Rodriguez-Barbosa JI, Weigert A. Downregulation of BTLA on NKT Cells Promotes Tumor Immune Control in a Mouse Model of Mammary Carcinoma. International Journal of Molecular Sciences. 2018; 19(3):752. https://doi.org/10.3390/ijms19030752
Chicago/Turabian StyleSekar, Divya, Luisa Govene, María-Luisa Del Río, Evelyn Sirait-Fischer, Annika F. Fink, Bernhard Brüne, José I. Rodriguez-Barbosa, and Andreas Weigert. 2018. "Downregulation of BTLA on NKT Cells Promotes Tumor Immune Control in a Mouse Model of Mammary Carcinoma" International Journal of Molecular Sciences 19, no. 3: 752. https://doi.org/10.3390/ijms19030752
APA StyleSekar, D., Govene, L., Del Río, M.-L., Sirait-Fischer, E., Fink, A. F., Brüne, B., Rodriguez-Barbosa, J. I., & Weigert, A. (2018). Downregulation of BTLA on NKT Cells Promotes Tumor Immune Control in a Mouse Model of Mammary Carcinoma. International Journal of Molecular Sciences, 19(3), 752. https://doi.org/10.3390/ijms19030752






