AOX1-Subfamily Gene Members in Olea europaea cv. “Galega Vulgar”—Gene Characterization and Expression of Transcripts during IBA-Induced in Vitro Adventitious Rooting
Abstract
1. Introduction
2. Results
2.1. Characterization of AOX1-Subfamily Members
2.2. Analysis of Transcript Expression
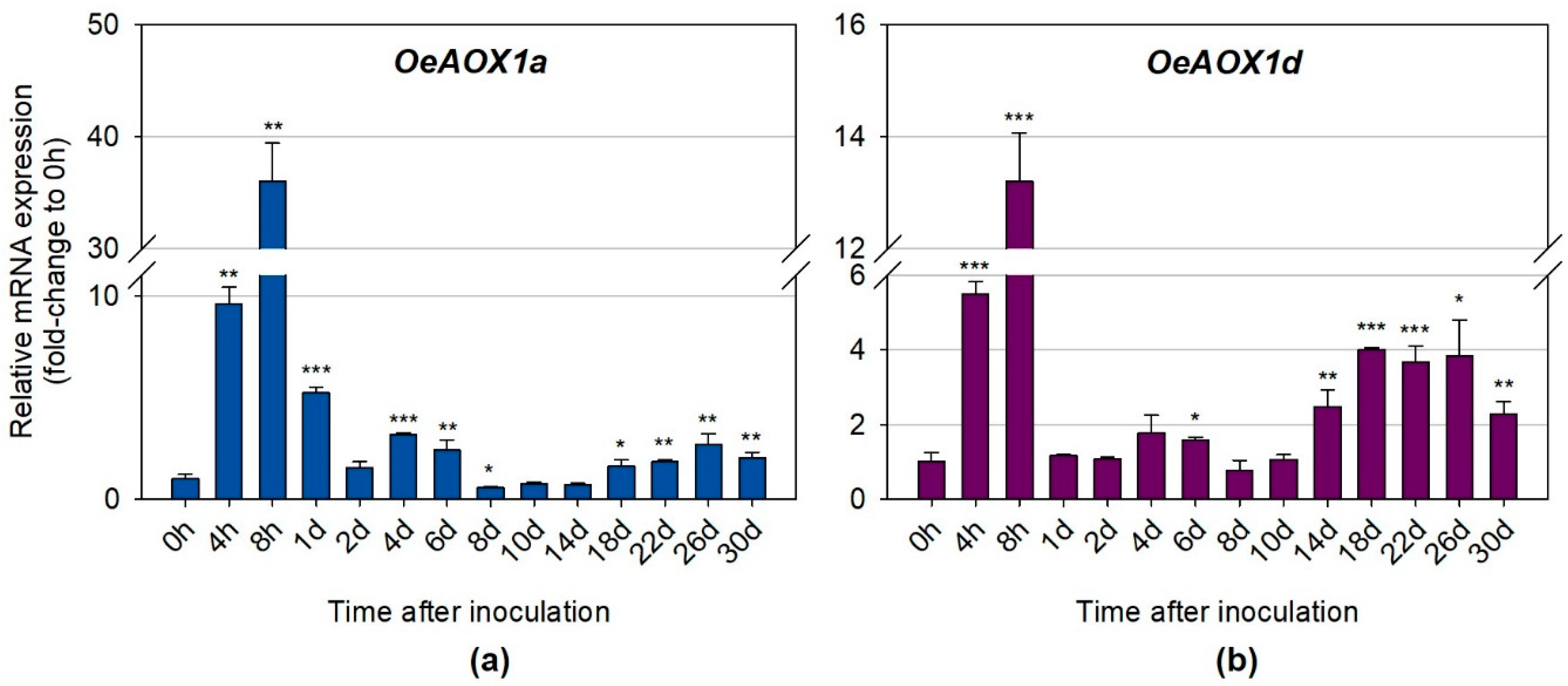
2.2.1. OeAOX1 Genes are Differentially Expressed during IBA-induced AR
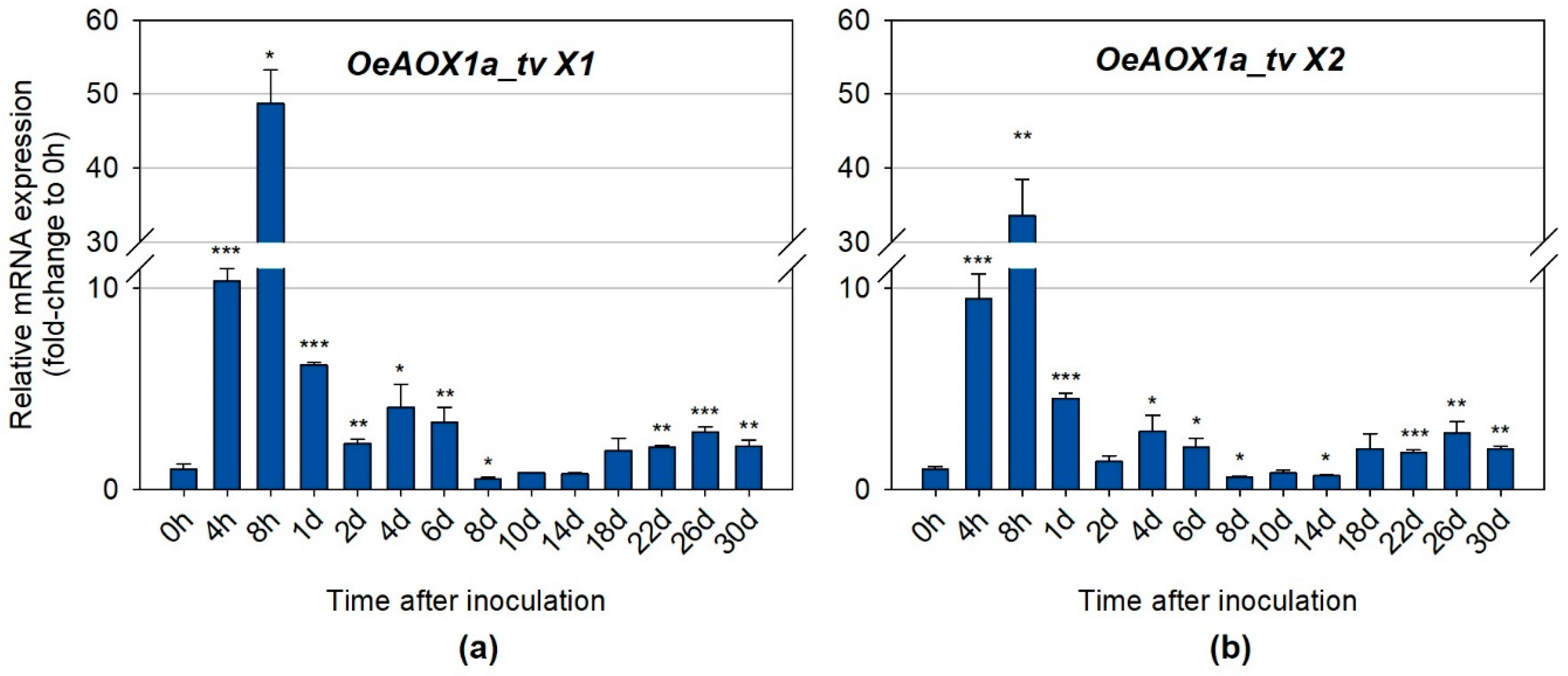
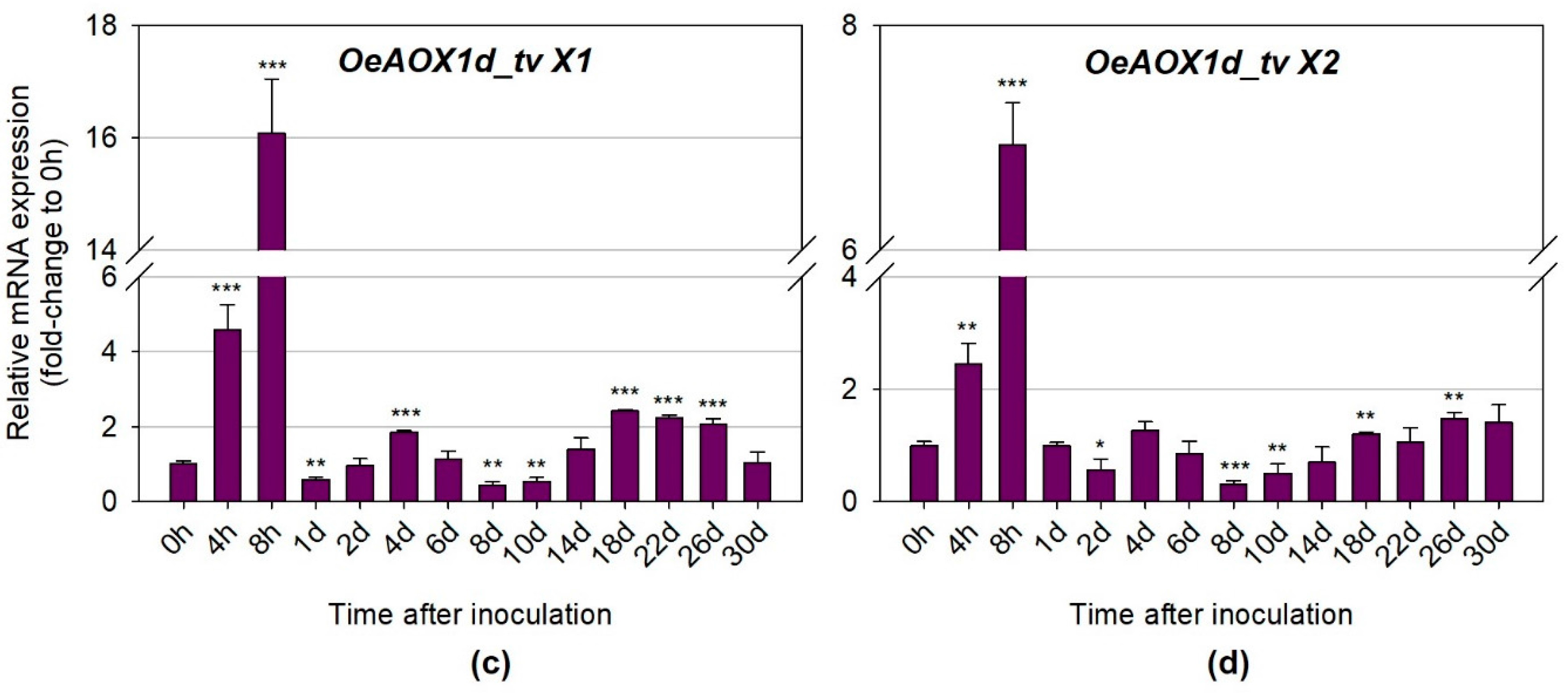
2.2.2. Distinct Transcripts Variants Show Different Expression Levels
3. Discussion
4. Materials and Methods
4.1. Characterization of the AOX Genes at the cDNA and Genomic Levels
4.1.1. Plant Material
4.1.2. Isolation of Complete OeAOX1-Subfamily Gene Members
4.1.3. Cloning and in Silico Sequence Analysis
4.1.4. Homology-Based Model
4.1.5. In Silico Identification of Regulatory Elements Located at the AOX Gene Boundaries
4.2. Transcript Expression of the AOX1-subfamily Members during AR on in Vitro Cultured Stem Segments
4.2.1. Plant Material and in Vitro Rooting Experiments
4.2.2. RNA Isolation and First-Strand cDNA Synthesis
4.2.3. Quantitative Real-Time PCR
4.2.4. Expression Analysis of Transcripts
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AOX | Alternative oxidase |
| AR | Adventitious rooting |
| IBA | indole-3-butyric acid |
| AR | adventitious rooting |
| SNP | single nucleotide polymorphism |
| QTL | Quantitative Trait Locus |
| UTR | Untranslated region |
| APA | alternative polyadenylation |
| mRNA | Messenger Ribonucleic acid |
| PAS | poly(a) site |
| gDNA | Genomic Deoxyribonucleic acid |
| NCBI | National Center for Biotechnology Information |
| TSA | transcriptome shotgun assembly |
| NJ | Neighbor-Joining |
| RACE | Rapid amplification of cDNA ends |
| RT-qPCR | Reverse transcription-quantitative real time polymerase chain reaction |
| ORF | Open reading frame |
| cDNA | complementary DNA |
| LTR | long terminal repeats |
| TSD | Target site duplications |
| PCR | polymerase chain reaction |
| mTP | mitochondrial transit peptides |
| snoRNAs | Small nucleolar RNAs |
| TEs | transposable elements |
| NMD | nonsense-mediated decay |
| RBPs | RNA-binding proteins |
| SHAM | salicylhydroxamic acid |
| ROS | Reactive oxygen species |
| ACT | actin |
| EF1a | elongation factor 1a |
| Cq | Quantification cycle |
| RQ | relative quantities |
References
- Baldoni, L.; Belaj, A. Olive. In Oil Crops; Handbook of Plant Breeding, Vollmann, J., Rajean, I., Eds.; Springer Science Business Media: New York, NY, USA, 2009; pp. 397–421. [Google Scholar]
- Peixe, A.; Santos Macedo, E.; Vieira, C.M.; Arnholdt-Schmitt, B. A histological evaluation of adventitious root formation in olive (Olea europaea L. cv. Galega vulgar) microshoots cultured in vitro. In Proceedings of the 28th International Horticultural Congress, Lisbon, Portugal, 22–27 August 2010; pp. S08–S214. [Google Scholar]
- Porfírio, S.; Gomes da Silva, M.D.R.; Cabrita, M.J.; Azadi, P.; Peixe, A. Reviewing current knowledge on olive (Olea europaea L.) adventitious root formation. Sci. Hortic. (Amsterdam) 2016, 198, 207–226. [Google Scholar] [CrossRef]
- Santos MacEdo, E.; Cardoso, H.G.; Hernández, A.; Peixe, A.A.; Polidoros, A.; Ferreira, A.; Cordeiro, A.; Arnholdt-Schmitt, B. Physiologic responses and gene diversity indicate olive alternative oxidase as a potential source for markers involved in efficient adventitious root induction. Physiol. Plant. 2009, 137, 532–552. [Google Scholar] [CrossRef] [PubMed]
- Potters, G.; Pasternak, T.P.; Guisez, Y.; Palme, K.J.; Jansen, M.A.K. Stress-induced morphogenic responses: Growing out of trouble? Trends Plant Sci. 2007, 12, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Vanlerberghe, G.C.; Cvetkovska, M.; Wang, J. Is the maintenance of homeostatic mitochondrial signaling during stress a physiological role for alternative oxidase? Physiol. Plant. 2009, 137, 392–406. [Google Scholar] [CrossRef] [PubMed]
- Vanlerberghe, G.C. Alternative oxidase: A mitochondrial respiratory pathway to maintain metabolic and signaling homeostasis during abiotic and biotic stress in plants. Int. J. Mol. Sci. 2013, 14, 6805–6847. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.; Borovskii, G.; Panda, S.K. Alternative oxidase and plant stress tolerance. Plant Signal. Behav. 2016, 11, e1256530. [Google Scholar] [CrossRef] [PubMed]
- Simons, B.H.; Lambers, H. The alternative oxidase: Is it a respiratory pathway allowing a plant to cope with stress? In Plant Responses to Environmental Stress: From Phytohormones to Gene Reorganization; Lerner, H.R., Ed.; Marcel Dekker Inc.: New York, NY, USA, 1999; pp. 265–286. [Google Scholar]
- Finnegan, P.M.; Soole, P.M.; Umbach, A.L. Alternative electron transport proteins. In Plant Mitochondria: From Gene to Function. Advances in Photosynthesis and Respiration; Day, D., Millar, H., Whelan, J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; p. 182. [Google Scholar]
- Finnegan, P.M.; Soole, K.L.; Umbach, A.L. Alternative mitochondrial electron transport proteins in higher plants. In Plant Mitochondria Advances in Photosynthesis and Respiration; Day, D.A., Millar, A.H., Whelan, J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; pp. 163–230. [Google Scholar]
- Clifton, R.; Lister, R.; Parker, K.L.; Sappl, P.G.; Elhafez, D.; Millar, A.H.; Day, D.A.; Whelan, J. Stress-induced co-expression of alternative respiratory chain components in Arabidopsis thaliana. Plant Mol. Biol. 2005, 58, 193–212. [Google Scholar] [CrossRef] [PubMed]
- Clifton, R.; Millar, A.H.; Whelan, J. Alternative oxidases in Arabidopsis: A comparative analysis of differential expression in the gene family provides new insights into function of non-phosphorylating bypasses. Biochim. Biophys. Acta-Bioenerg. 2006, 1757, 730–741. [Google Scholar] [CrossRef] [PubMed]
- Santos Macedo, E.; Sircar, D.; Cardoso, H.G.; Peixe, A.; Arnholdt-Schmitt, B. Involvement of alternative oxidase (AOX) in adventitious rooting of Olea europaea L. microshoots is linked to adaptive phenylpropanoid and lignin metabolism. Plant Cell Rep. 2012, 31, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- Hedayati, V.; Mousavi, A.; Razavi, K.; Cultrera, N.; Alagna, F.; Mariotti, R.; Hosseini-Mazinani, M.; Baldoni, L. Polymorphisms in the AOX2 gene are associated with the rooting ability of olive cuttings. Plant Cell Rep. 2015, 34, 1151–1164. [Google Scholar] [CrossRef] [PubMed]
- McDonald, A.E.; Costa, J.H.; Nobre, T.; De Melo, D.F.; Arnholdt-Schmitt, B. Evolution of AOX genes across kingdoms and the challenge of classification. In Alternative Respiratory Pathways in Higher Plants; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 267–272. ISBN 9781118789971. [Google Scholar]
- Costa, J.H.; McDonald, A.E.; Arnholdt-Schmitt, B.; Fernandes de Melo, D. A classification scheme for alternative oxidases reveals the taxonomic distribution and evolutionary history of the enzyme in angiosperms. Mitochondrion 2014, 19, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.H.; dos Santos, C.P.; de Sousa e Lima, B.; Moreira Netto, A.N.; Saraiva, K.D.d.C.; Arnholdt-Schmitt, B. In silico identification of alternative oxidase 2 (AOX2) in monocots: A new evolutionary scenario. J. Plant Physiol. 2017, 210, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, H.G.; Nogales, A.; Frederico, A.M.; Svensson, J.T.; Macedo, E.S.; Valadas, V.; Arnholdt-Schmitt, B. Exploring AOX gene diversity. In Alternative Respiratory Pathways in Higher Plants; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 239–254. ISBN 9781118789971. [Google Scholar]
- Whelan, J.; Millar, A.H.; Day, D.A. The alternative oxidase is encoded in a multigene family in soybean. Planta 1996, 198, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Saisho, D.; Nambara, E.; Naito, S.; Tsutsumi, N.; Hirai, A.; Nakazono, M. Characterization of the gene family for alternativa oxidase from Arabidopsis thaliana. Plant Mol. Biol. 1997, 35, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Considine, M.J.; Holtzapffel, R.C.; Day, D.A.; Whelan, J.; Millar, A.H. Molecular distinction between alternative oxidase from monocots and dicots. Plant Physiol. 2002, 129, 949–953. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.H.; dos Santos, C.P.; da Cruz Saraiva, K.D.; Arnholdt-Schmitt, B. A step-by-step protocol for classifying AOX proteins in flowering plants. Methods Mol. Biol. 2017, 1670, 225–234. [Google Scholar] [PubMed]
- Feng, H.; Guan, D.; Sun, K.; Wang, Y.; Zhang, T.; Wang, R. Expression and signal regulation of the alternative oxidase genes under abiotic stresses. Acta Biochim. Biophys. Sin. (Shanghai) 2013, 45, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.H.; Mota, E.F.; Cambursano, M.V.; Lauxmann, M.A.; De Oliveira, L.M.N.; Silva Lima, M.D.G.; Orellano, E.G.; Fernandes De Melo, D. Stress-induced co-expression of two alternative oxidase (VuAox1 and 2b) genes in Vigna unguiculata. J. Plant Physiol. 2010, 167, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Cavalcanti, J.H.F.; Oliveira, G.M.; Saraiva, K.D.d.C.; Torquato, J.P.P.; Maia, I.G.; Fernandes de Melo, D.; Costa, J.H. Identification of duplicated and stress-inducible Aox2b gene co-expressed with Aox1 in species of the Medicago genus reveals a regulation linked to gene rearrangement in leguminous genomes. J. Plant Physiol. 2013, 170, 1609–1619. [Google Scholar] [CrossRef] [PubMed]
- Velada, I.; Ragonezi, C.; Arnholdt-Schmitt, B.; Cardoso, H. Reference Genes Selection and Normalization of Oxidative Stress Responsive Genes upon Different Temperature Stress Conditions in Hypericum perforatum L. PLoS ONE 2014, 9, e115206. [Google Scholar] [CrossRef] [PubMed]
- Guerra Cardoso, H.; Doroteia Campos, M.; Rita Costa, A.; Catarina Campos, M.; Nothnagel, T.; Arnholdt-Schmitt, B. Carrot alternative oxidase gene AOX2a demonstrates allelic and genotypic polymorphisms in intron 3. Physiol. Plant. 2009, 137, 592–608. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, H.; Campos, M.D.; Nothnagel, T.; Arnholdt-Schmitt, B. Polymorphisms in intron 1 of carrot AOX2b–a useful tool to develop a functional marker? Plant Genet. Resour. 2011, 9, 177–180. [Google Scholar] [CrossRef]
- Ferreira, A.O.; Cardoso, H.G.; Macedo, E.S.; Breviario, D.; Arnholdt-Schmitt, B. Intron polymorphism pattern in AOX1b of wild St John’s wort (Hypericum perforatum) allows discrimination between individual plants. Physiol. Plant. 2009, 137, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Nogales, A.; Nobre, T.; Cardoso, H.G.; Muñoz-Sanhueza, L.; Valadas, V.; Campos, M.D.; Arnholdt-Schmitt, B. Allelic variation on DcAOX1 gene in carrot (Daucus carota L.): An interesting simple sequence repeat in a highly variable intron. Plant Gene 2016, 5, 49–55. [Google Scholar] [CrossRef]
- Nobre, T.; Campos, M.D.; Lucic-Mercy, E.; Arnholdt-Schmitt, B. Misannotation Awareness: A Tale of Two Gene-Groups. Front. Plant Sci. 2016, 7, 868. [Google Scholar] [CrossRef] [PubMed]
- Albury, M.S.; Elliott, C.; Moore, A.L. Towards a structural elucidation of the alternative oxidase in plants. Physiol. Plant. 2009, 137, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Abe, F.; Saito, K.; Miura, K.; Toriyama, K. A single nucleotide polymorphism in the alternative oxidase gene among rice varieties differing in low temperature tolerance. FEBS Lett. 2002, 527, 181–185. [Google Scholar] [CrossRef]
- Di Giammartino, D.C.; Nishida, K.; Manley, J.L. Mechanisms and Consequences of Alternative Polyadenylation. Mol. Cell 2011, 43, 853–866. [Google Scholar] [CrossRef] [PubMed]
- Polidoros, A.N.; Mylona, P.V.; Arnholdt-Schmitt, B. Aox gene structure, transcript variation and expression in plants. Physiol. Plant. 2009, 137, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Umbach, A.L.; Siedow, J.N. Covalent and Noncovalent Dimers of the Cyanide-Resistant Alternative Oxidase Protein in Higher Plant Mitochondria and Their Relationship to Enzyme Activity. Plant Physiol. 1993, 103, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.L.; Shiba, T.; Young, L.; Harada, S.; Kita, K.; Ito, K. Unraveling the Heater: New Insights into the Structure of the Alternative Oxidase. Annu. Rev. Plant Biol. 2013, 64, 637–663. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Manley, J.L. Alternative polyadenylation of mRNA precursors. Nat. Rev. Mol. Cell Biol. 2017, 18, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Macedo, E.; Vieira, C.; Carrizo, D.; Porfírio, S.; Hegewald, H.; Arnholdt-Schmitt, B.; Calado, M.L.; Peixe, A. Adventitious root formation in olive (Olea europaea L.) microshoots: Anatomical evaluation and associated biochemical changes in peroxidase and polyphenoloxidase activities. J. Hortic. Sci. Biotechnol. 2013, 88, 53–59. [Google Scholar] [CrossRef]
- Campos, M.D.; Cardoso, H.G.; Linke, B.; Costa, J.H.; De Melo, D.F.; Justo, L.; Frederico, A.M.; Arnholdt-Schmitt, B. Differential expression and co-regulation of carrot AOX genes (Daucus carota). Physiol. Plant. 2009, 137, 578–591. [Google Scholar] [CrossRef] [PubMed]
- Velada, I.; Cardoso, H.G.; Ragonezi, C.; Nogales, A.; Ferreira, A.; Valadas, V.; Arnholdt-Schmitt, B. Alternative Oxidase Gene Family in Hypericum perforatum L.: Characterization and Expression at the Post-germinative Phase. Front. Plant Sci. 2016, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.-S. Respiratory pathways in bulky tissues and storage organs. In Alternative Respiratory Pathways in Higher Plants; Gupta, K.J., Mur, L.A.J., Neelwarne, B., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2015; pp. 221–232. ISBN 9781118789971. [Google Scholar]
- Vishwakarma, A.; Dalal, A.; Tetali, S.D.; Kirti, P.B.; Padmasree, K. Genetic engineering of AtAOX1a in Saccharomyces cerevisiae prevents oxidative damage and maintains redox homeostasis. FEBS Open Bio 2016, 6, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Taylor, N.L.; Whelan, J.; Millar, A.H. Refining the Definition of Plant Mitochondrial Presequences through Analysis of Sorting Signals, N-Terminal Modifications, and Cleavage Motifs. Plant Physiol. 2009, 150, 1272–1285. [Google Scholar] [CrossRef] [PubMed]
- Heyn, P.; Kalinka, A.T.; Tomancak, P.; Neugebauer, K.M. Introns and gene expression: Cellular constraints, transcriptional regulation, and evolutionary consequences. BioEssays 2015, 37, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Oswald, A.; Oates, A.C. Control of endogenous gene expression timing by introns. Genome Biol. 2011, 12, 107. [Google Scholar] [CrossRef] [PubMed]
- Brinster, R.L.; Allen, J.M.; Behringer, R.R.; Gelinas, R.E.; Palmiter, R.D. Introns increase transcriptional efficiency in transgenic mice. Proc. Natl. Acad. Sci. USA 1988, 85, 836–840. [Google Scholar] [CrossRef] [PubMed]
- Pawlicki, J.M.; Steitz, J.A. Primary microRNA transcript retention at sites of transcription leads to enhanced microRNA production. J. Cell Biol. 2008, 182, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, C.D.; Springer, N.M. Transposable element influences on gene expression in plants. Biochim. Biophys. Acta-Gene Regul. Mech. 2017, 1860, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Schnable, P.S.; Ware, D.; Fulton, R.S.; Stein, J.C.; Wei, F.; Pasternak, S.; Liang, C.; Zhang, J.; Fulton, L.; Graves, T.A.; et al. The B73 Maize Genome: Complexity, Diversity, and Dynamics. Science 2009, 326, 1112–1115. [Google Scholar] [CrossRef] [PubMed]
- Ohtsu, K.; Hirano, H.Y.; Tsutsumi, N.; Hirai, A.; Nakazono, M. Anaconda, a new class of transposon belonging to the Mu superfamily, has diversified by acquiring host genes during rice evolution. Mol. Genet. Genom. 2005, 274, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.H.; De Melo, D.F.; Gouveia, Z.; Cardoso, H.G.; Peixe, A.; Arnholdt-Schmitt, B. The alternative oxidase family of Vitis vinifera reveals an attractive model to study the importance of genomic design. Physiol. Plant. 2009, 137, 553–565. [Google Scholar] [CrossRef] [PubMed]
- Macko-Podgorni, A.; Nowicka, A.; Grzebelus, E.; Simon, P.W.; Grzebelus, D. DcSto: Carrot Stowaway-like elements are abundant, diverse, and polymorphic. Genetica 2013, 141, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.Q.; Liu, Z.; Lu, W.; Liu, M. Interplay between Alternative Splicing and Alternative Polyadenylation Defines the Expression Outcome of the Plant Unique OXIDATIVE TOLERANT-6 Gene. Sci. Rep. 2017, 7, 2052. [Google Scholar] [CrossRef] [PubMed]
- Tantikanjana, T. An Alternative Transcript of the S Locus Glycoprotein Gene in a Class II Pollen-Recessive Self-Incompatibility Haplotype of Brassica oleracea Encodes a Membrane-Anchored Protein. Plant Cell Online 1993, 5, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Simpson, G.G.; Dijkwel, P.P.; Quesada, V.; Henderson, I.; Dean, C. FY is an RNA 3′ end-processing factor that interacts with FCA to control the Arabidopsis floral transition. Cell 2003, 113, 777–787. [Google Scholar] [CrossRef]
- Lykke-Andersen, S.; Jensen, T.H. Nonsense-mediated mRNA decay: An intricate machinery that shapes transcriptomes. Nat. Rev. Mol. Cell Biol. 2015, 16, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Fabian, M.R.; Sonenberg, N.; Filipowicz, W. Regulation of mRNA Translation and Stability by microRNAs. Annu. Rev. Biochem. 2010, 79, 351–379. [Google Scholar] [CrossRef] [PubMed]
- Lianoglou, S.; Garg, V.; Yang, J.L.; Leslie, C.S.; Mayr, C. Ubiquitously transcribed genes use alternative polyadenylation to achieve tissue-specific expression. Genes Dev. 2013, 27, 2380–2396. [Google Scholar] [CrossRef] [PubMed]
- Smibert, P.; Miura, P.; Westholm, J.O.; Shenker, S.; May, G.; Duff, M.O.; Zhang, D.; Eads, B.D.; Carlson, J.; Brown, J.B.; et al. Global Patterns of Tissue-Specific Alternative Polyadenylation in Drosophila. Cell Rep. 2012, 1, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Sandberg, R.; Neilson, J.R.; Sarma, A.; Sharp, P.A.; Burge, C.B. Proliferating cells express mRNAs with shortened 3′ UTRs and fewer microRNA target sites. Science 2008, 320, 1643–1647. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C.; Bartel, D.P. Widespread Shortening of 3’UTRs by Alternative Cleavage and Polyadenylation Activates Oncogenes in Cancer Cells. Cell 2009, 138, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Gupta, I.; Clauder-Münster, S.; Klaus, B.; Järvelin, A.I.; Aiyar, R.S.; Benes, V.; Wilkening, S.; Huber, W.; Pelechano, V.; Steinmetz, L.M. Alternative polyadenylation diversifies post-transcriptional regulation by selective RNA-protein interactions. Mol. Syst. Biol. 2014, 10. [Google Scholar] [CrossRef] [PubMed]
- Gruber, A.R.; Martin, G.; Müller, P.; Schmidt, A.; Gruber, A.J.; Gumienny, R.; Mittal, N.; Jayachandran, R.; Pieters, J.; Keller, W.; et al. Global 3′UTR shortening has a limited effect on protein abundance in proliferating T cells. Nat. Commun. 2014, 5, 5465. [Google Scholar] [CrossRef] [PubMed]
- Arnholdt-Schmitt, B.; Santos Macedo, E.; Peixe, A.; Cardoso, H.; Cordeiro, A. AOX—A potential functional marker for efficient rooting in olive shoot cuttings. In Proceedings of the Second International Seminar Olivebioteq, Mazara del Vallo, Italy, 5–10 November 2006; pp. 249–254. [Google Scholar]
- Arnholdt-Schmitt, B.; Costa, J.H.; de Melo, D.F. AOX-a functional marker for efficient cell reprogramming under stress? Trends Plant Sci. 2006, 11, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Porfirio, S.; Calado, M.L.; Noceda, C.; Cabrita, M.J.; da Silva, M.G.; Azadi, P.; Peixe, A. Tracking biochemical changes during adventitious root formation in olive (Olea europaea L.). Sci. Hortic. (Amsterdam) 2016, 204, 41–53. [Google Scholar] [CrossRef]
- De Klerk, G. Markers of adventitious root formation. Agronomie 1996, 16, 609–616. [Google Scholar] [CrossRef]
- Gaspar, T.; Kevers, C.; Hausman, J.; Berthon, J.; Ripetti, V. Practical uses of peroxidase activity as a predictive marker of rooting performance of micropropagated shoots. Agronomie 1992, 12, 757–765. [Google Scholar] [CrossRef]
- Gaspar, T.; Kevers, C.; Hausman, J.-F. Indissociable Chief Factors in the Inductive Phase of Adventitious Rooting BT-Biology of Root Formation and Development; Altman, A., Waisel, Y., Eds.; Springer: Boston, MA, USA, 1997; pp. 55–63. ISBN 978-1-4615-5403-5. [Google Scholar]
- Jarvis, B.C. Endogenous Control of Adventitious Rooting in Non-Woody Cuttings. In BT-New Root Formation in Plants and Cuttings; Jackson, M.B., Ed.; Springer: Dordrecht, The Netherlands, 1986; pp. 191–222. ISBN 978-94-009-4358-2. [Google Scholar]
- Kevers, C.; Hausman, J.F.; Faivre-Rampant, O.; Evers, D.; Gaspar, T. Hormonal control of adventitious rooting: Progress and questions. J. Appl. Bot. 1997, 71, 71–79. [Google Scholar]
- Rout, G.R.; Samantaray, S.; Das, P. In vitro rooting of Psoralea corylifolia Linn: Peroxidase activity as a marker. Plant Growth Regul. 2000, 30, 215–219. [Google Scholar] [CrossRef]
- Berthon, J.Y.; Ben Tahar, S.; Gaspar, T.; Boyer, N. Rooting phases of shoots of Sequoiadendron giganteum in vitro and their requirements. Plant Physiol. Biochem. 1990, 28, 631–638. [Google Scholar]
- Heloir, M.C.; Kevers, C.; Hausman, J.F.; Gaspar, T. Changes in the concentrations of auxins and polyamines during rooting of in-vitro-propagated walnut shoots. Tree Physiol. 1996, 16, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Li, S.W.; Xue, L.; Xu, S.; Feng, H.; An, L. Mediators, genes and signaling in adventitious rooting. Bot. Rev. 2009, 75, 230–247. [Google Scholar] [CrossRef]
- Pacurar, D.I.; Perrone, I.; Bellini, C. Auxin is a central player in the hormone cross-talks that control adventitious rooting. Physiol. Plant. 2014, 151, 83–96. [Google Scholar] [CrossRef] [PubMed]
- Campos, D.M.; Nogales, A.; Cardoso, H.G.; Kumar, S.R.; Nobre, T.; Sathishkumar, R.; Arnholdt-Schmitt, B. Stress-induced accumulation of DcAoX1 and DcAoX2a transcripts coincides with critical time point for structural biomass prediction in carrot primary cultures (Daucus carota L.). Front. Genet. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Frederico, A.M.; Campos, M.D.; Cardoso, H.G.; Imani, J.; Arnholdt-Schmitt, B. Alternative oxidase involvement in Daucus carota somatic embryogenesis. Physiol. Plant. 2009, 137, 498–508. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, A.; Law, S.R.; Narsai, R.; Duncan, O.; Lee, J.-H.; Zhang, B.; Van Aken, O.; Radomiljac, J.D.; van der Merwe, M.; Yi, K.; et al. A Functional Antagonistic Relationship between Auxin and Mitochondrial Retrograde Signaling Regulates Alternative Oxidase1a Expression in Arabidopsis. Plant Physiol. 2014, 165, 1233–1254. [Google Scholar] [CrossRef] [PubMed]
- Kerchev, P.I.; De Clercq, I.; Denecker, J.; Mühlenbock, P.; Kumpf, R.; Nguyen, L.; Audenaert, D.; Dejonghe, W.; Van Breusegem, F. Mitochondrial perturbation negatively affects auxin signaling. Mol. Plant 2014, 7, 1138–1150. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, D.M.; Umbach, A.L.; Subbaiah, C.C.; Siedow, J.N. Mitochondrial reactive oxygen species. Contribution to oxidative stress and interorganellar signaling. Plant Physiol. 2006, 141, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Tognetti, V.B.; Mühlenbock, P.; van Breusegem, F. Stress homeostasis-the redox and auxin perspective. Plant Cell Environ. 2012, 35, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, T.; Potters, G.; Caubergs, R.; Jansen, M.A.K. Complementary interactions between oxidative stress and auxins control plant growth responses at plant, organ, and cellular level. J. Exp. Bot. 2005, 56, 1991–2001. [Google Scholar] [CrossRef] [PubMed]
- Guilfoyle, T.J.; Hagen, G. Auxin response factors. Curr. Opin. Plant Biol. 2007, 10, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Peixe, A.; Raposo, A.; Lourenço, R.; Cardoso, H.; Macedo, E. Coconut water and BAP successfully replaced zeatin in olive (Olea europaea L.) micropropagation. Sci. Hortic. (Amsterdam) 2007, 113, 1–7. [Google Scholar] [CrossRef]
- Karlin, S.; Altschul, S.F. Applications and statistics for multiple high-scoring segments in molecular sequences. Proc. Natl. Acad. Sci. USA 1993, 90, 5873–5877. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Emanuelsson, O.; Nielsen, H.; Brunak, S.; von Heijne, G. Predicting Subcellular Localization of Proteins Based on their N-terminal Amino Acid Sequence. J. Mol. Biol. 2000, 300, 1005–1016. [Google Scholar] [CrossRef] [PubMed]
- Šali, A.; Blundell, T.L. Comparative Protein Modelling by Satisfaction of Spatial Restraints. J. Mol. Biol. 1993, 234, 779–815. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Barghini, E.; Natali, L.; Giordani, T.; Cossu, R.M.; Scalabrin, S.; Cattonaro, F.; Šimková, H.; Vrána, J.; Doležel, J.; Morgante, M.; et al. LTR retrotransposon dynamics in the evolution of the olive (Olea europaea) genome. DNA Res. 2015, 22, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Higo, K.; Ugawa, Y.; Iwamoto, M.; Korenaga, T. Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res. 1999, 27, 297–300. [Google Scholar] [CrossRef] [PubMed]
- Lescot, M.; Déhais, P.; Thijs, G.; Marchal, K.; Moreau, Y.; Van de Peer, Y.; Rouzé, P.; Rombauts, S. PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res. 2002, 30, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Rugini, E. In vitro propagation of some olive (Olea europaea sativa L.) cultivars with different root-ability, and medium development using analytical data from developing shoots and embryos. Sci. Hortic. (Amsterdam) 1984, 24, 123–134. [Google Scholar] [CrossRef]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002, 3. [Google Scholar] [CrossRef]
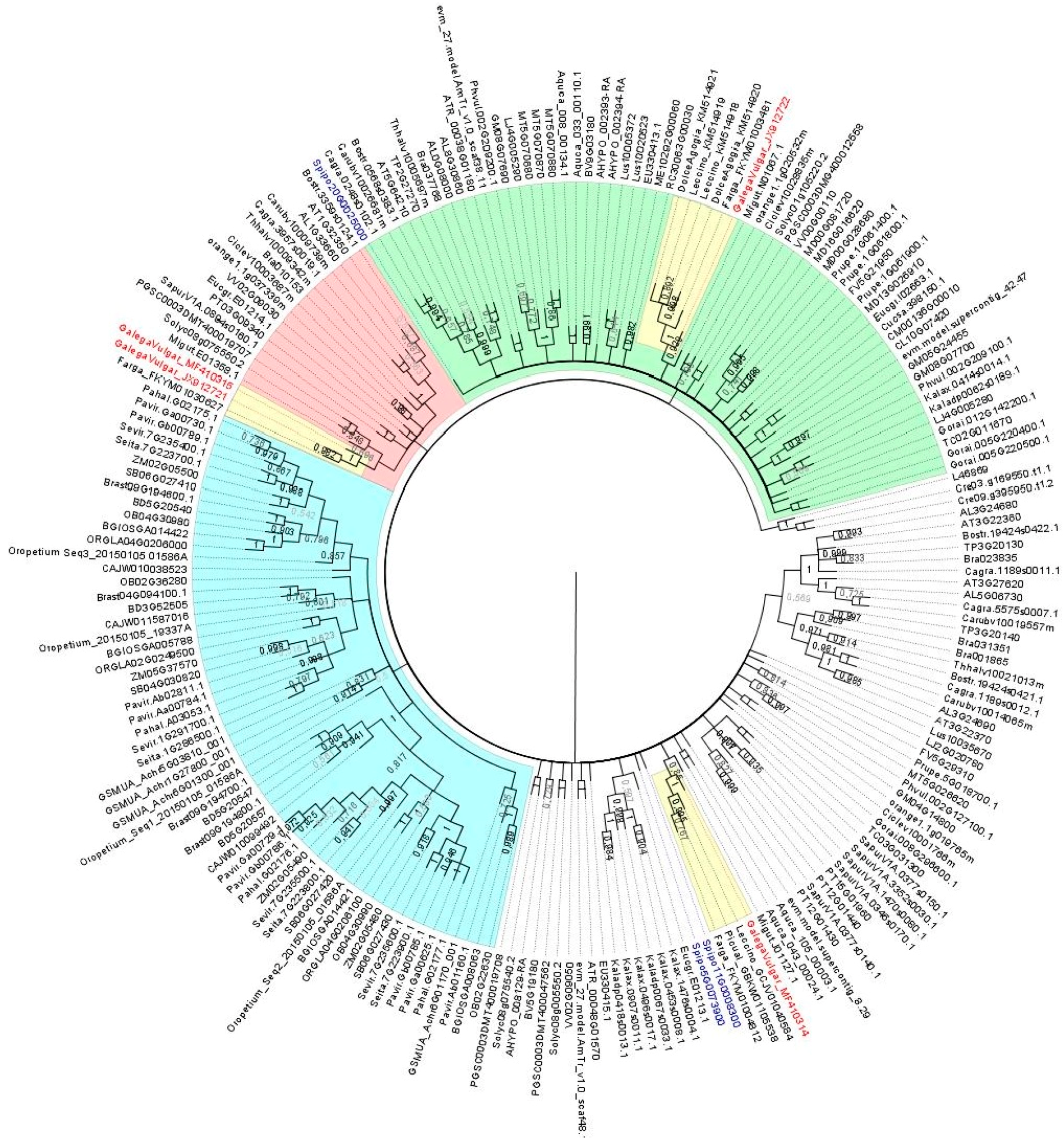


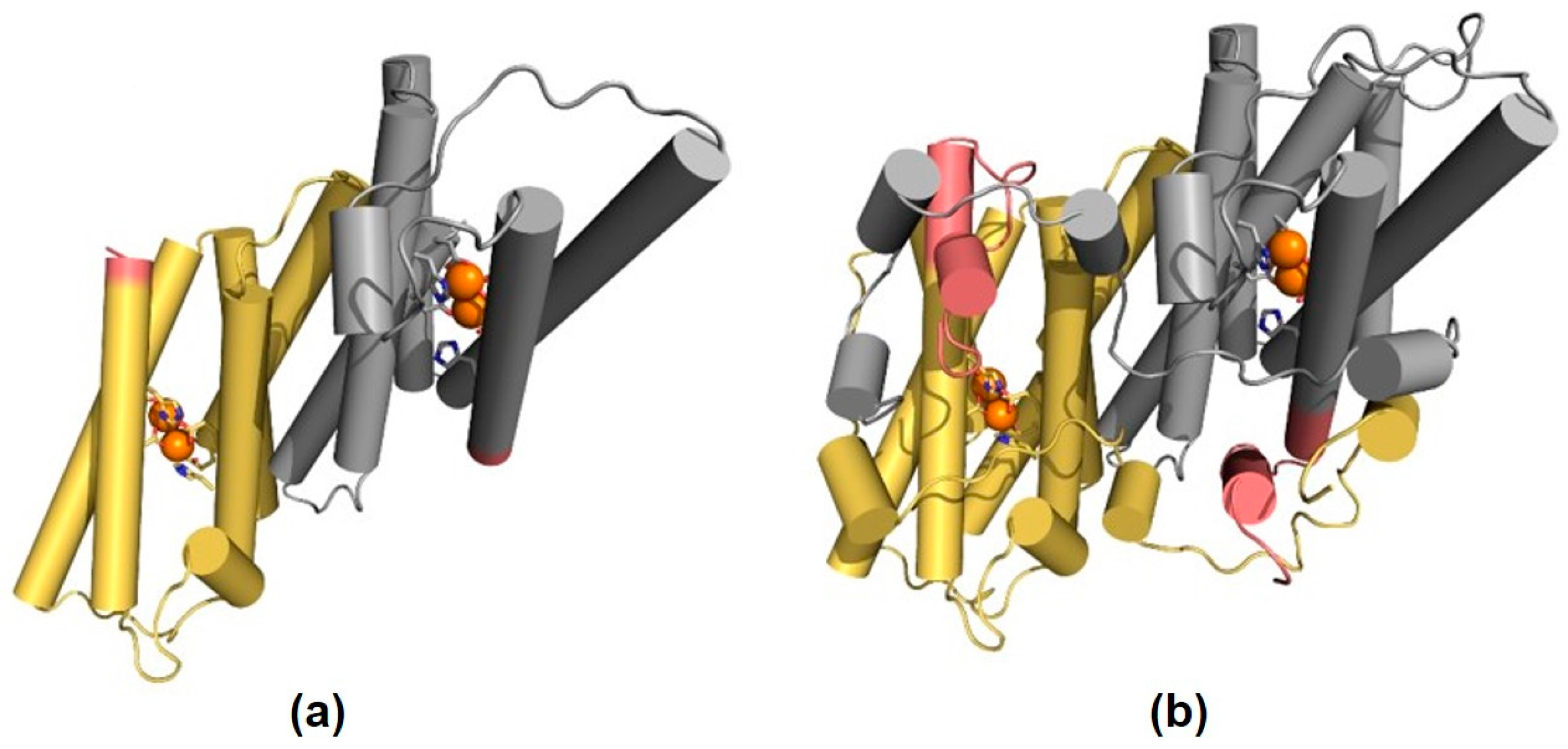
| Element | Length (bp) | LTR Length | TSD | Position Relative to Start Codon |
|---|---|---|---|---|
| OeAOX1a | ||||
| isolate 84856_A retrotransposon gypsy-type [KM577525] | 13,288 | 1791 (left) 1752 (right) | GAAAG | −18,801/−5513 |
| isolate 95401_B retrotransposon gypsy-type [KM577546] | 12,998 | 782 (left) 773 (right) | GTCAT | −27,411/−1125 |
| OeAOX1d | ||||
| isolate 95401_B retrotransposon gypsy-type [KM577546] | 12,948 | 767 (left) 773 (right) | CAATT | −16,375/−3427 |
| isolate 70744_E retrotransposon copia-type [KM577454] | 6023 | 750 (left) 750 (right) | TTATC | undetermined (downstream) |
| unknown, copia-type, similar to Copia-63_VV-I | 4959 | 275 (left) 283 (right) | [A/G]TAGC | undetermined (downstream) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Velada, I.; Grzebelus, D.; Lousa, D.; M. Soares, C.; Santos Macedo, E.; Peixe, A.; Arnholdt-Schmitt, B.; G. Cardoso, H. AOX1-Subfamily Gene Members in Olea europaea cv. “Galega Vulgar”—Gene Characterization and Expression of Transcripts during IBA-Induced in Vitro Adventitious Rooting. Int. J. Mol. Sci. 2018, 19, 597. https://doi.org/10.3390/ijms19020597
Velada I, Grzebelus D, Lousa D, M. Soares C, Santos Macedo E, Peixe A, Arnholdt-Schmitt B, G. Cardoso H. AOX1-Subfamily Gene Members in Olea europaea cv. “Galega Vulgar”—Gene Characterization and Expression of Transcripts during IBA-Induced in Vitro Adventitious Rooting. International Journal of Molecular Sciences. 2018; 19(2):597. https://doi.org/10.3390/ijms19020597
Chicago/Turabian StyleVelada, Isabel, Dariusz Grzebelus, Diana Lousa, Cláudio M. Soares, Elisete Santos Macedo, Augusto Peixe, Birgit Arnholdt-Schmitt, and Hélia G. Cardoso. 2018. "AOX1-Subfamily Gene Members in Olea europaea cv. “Galega Vulgar”—Gene Characterization and Expression of Transcripts during IBA-Induced in Vitro Adventitious Rooting" International Journal of Molecular Sciences 19, no. 2: 597. https://doi.org/10.3390/ijms19020597
APA StyleVelada, I., Grzebelus, D., Lousa, D., M. Soares, C., Santos Macedo, E., Peixe, A., Arnholdt-Schmitt, B., & G. Cardoso, H. (2018). AOX1-Subfamily Gene Members in Olea europaea cv. “Galega Vulgar”—Gene Characterization and Expression of Transcripts during IBA-Induced in Vitro Adventitious Rooting. International Journal of Molecular Sciences, 19(2), 597. https://doi.org/10.3390/ijms19020597









